A COVID-19 Rehabilitation Prospective Surveillance Model for Use by Physiotherapists
Abstract
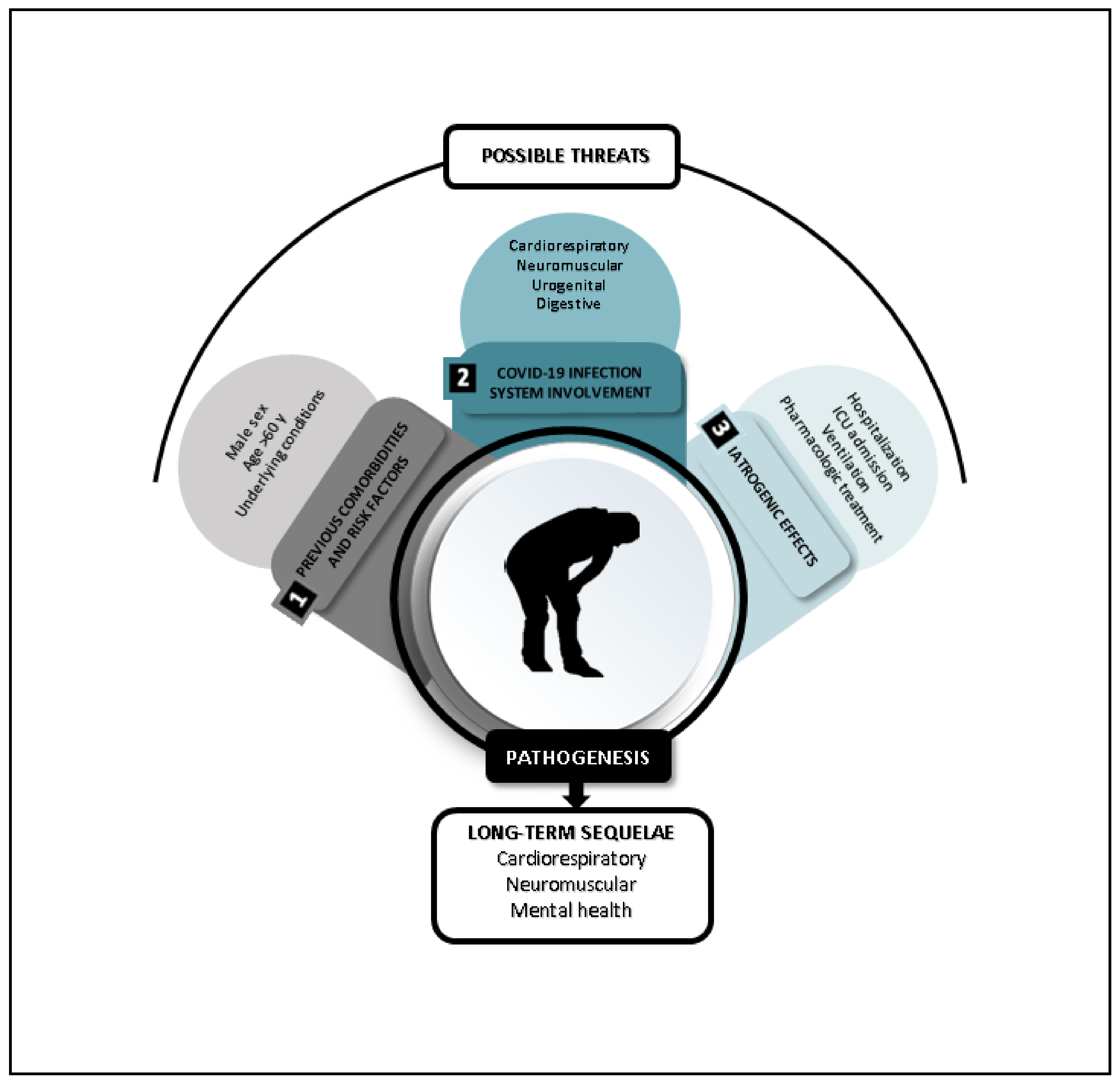
1. Introduction
2. Methodology
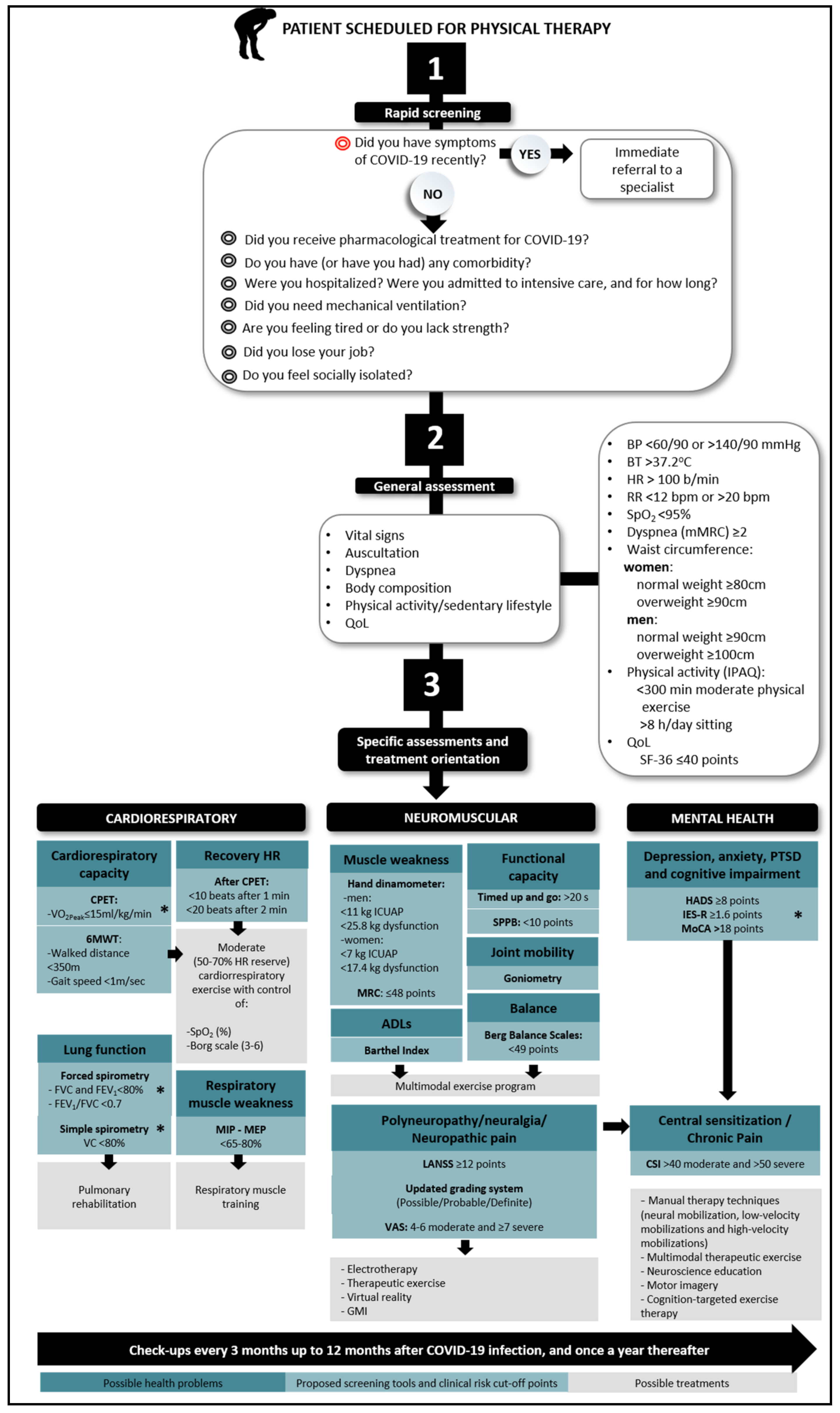
3. The Proposed Prospective Surveillance Model
3.1. Rapid Screening
3.2. General Assessment
3.3. Specific Assessments
3.3.1. Cardiorespiratory System
3.3.2. Neuromuscular System
3.3.3. Mental Health
4. Discussion
Limitations, Key Findings, and Future Directions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sheehy, L.M. Considerations for Postacute Rehabilitation for Survivors of COVID-19. JMIR Public Health Surveill. 2020, 6, e19462. [Google Scholar] [CrossRef] [PubMed]
- Stam, H.; Stucki, G.; Bickenbach, J. Covid-19 and Post Intensive Care Syndrome: A Call for Action. J. Rehabil. Med. 2020, 52, jrm00044. [Google Scholar] [CrossRef] [PubMed]
- Qureshi, H. The Digestive System and the COVID-19. J. Pak. Med. Assoc. 2020, S98–S100. [Google Scholar] [CrossRef] [PubMed]
- ECDC (European Centre for Disease Prevention and Control). COVID-19 Surveillance Report. Available online: https://covid19-surveillance-report.ecdc.europa.eu/ (accessed on 19 August 2020).
- Elliott, D.; Davidson, J.E.; Harvey, M.A.; Bemis-Dougherty, A.; Hopkins, R.O.; Iwashyna, T.J.; Wagner, J.; Weinert, C.R.; Wunsch, H.; Bienvenu, O.J.; et al. Exploring the Scope of Post-Intensive Care Syndrome Therapy and Care. Crit. Care Med. 2014, 42, 2518–2526. [Google Scholar] [CrossRef]
- Ahmed, H.; Patel, K.; Greenwood, D.C.; Halpin, S.; Lewthwaite, P.; Salawu, A.; Eyre, L.; Breen, A.; O’Connor, R.; Jones, A.; et al. Long-term clinical outcomes in survivors of severe acute respiratory syndrome and Middle East respiratory syndrome coronavirus outbreaks after hospitalisation or ICU admission: A systematic review and meta-analysis. J. Rehabil. Med. 2020. [Google Scholar] [CrossRef]
- Bettger, J.P.; Thoumi, A.; Marquevich, V.; De Groote, W.; Battistella, L.R.; Imamura, M.; Ramos, V.D.; Wang, N.; Dreinhoefer, K.E.; Mangar, A.; et al. COVID-19: Maintaining essential rehabilitation services across the care continuum. BMJ Glob. Health 2020, 5, e002670. [Google Scholar] [CrossRef]
- Dong, E.; Du, H.; Gardner, L. An interactive web-based dashboard to track COVID-19 in real time. Lancet Infect. Dis. 2020, 20, 533–534. [Google Scholar] [CrossRef]
- Wang, B.; Li, R.; Lu, Z.; Huang, Y. Does comorbidity increase the risk of patients with COVID-19: Evidence from meta-analysis. Aging 2020, 12, 6049–6057. [Google Scholar] [CrossRef]
- Rees, E.M.; Nightingale, E.S.; Jafari, Y.; Waterlow, N.R.; Clifford, S.; Pearson, C.A.B.; CMMID Working Group; Jombart, T.; Procter, S.R.; Knight, G.M. COVID-19 length of hospital stay: A systematic review and data synthesis. BMC Med. 2020, 18, 1–22. [Google Scholar] [CrossRef]
- Lane, J.C.; Weaves, J.; Kostka, K.; Alser, O.; Prats-Uribe, A.; Newby, D.; Prieto-Alhambra, D. Safety of hydroxychloroquine, alone and in combination with azithromycin, in light of rapid wide-spread use for COVID-19: A multinational, network cohort and self-controlled case series study. MedRxiv 2020. [Google Scholar] [CrossRef]
- Thomas, P.; Baldwin, C.; Bissett, B.; Boden, I.; Gosselink, R.; Granger, C.L.; Hodgson, C.; Jones, A.Y.; Kho, M.E.; Moses, R.; et al. Physiotherapy management for COVID-19 in the acute hospital setting: Clinical practice recommendations. J. Physiother. 2020, 66, 73–82. [Google Scholar] [CrossRef]
- Falvey, J.R.; Krafft, C.; Kornetti, D. The Essential Role of Home- and Community-Based Physical Therapists during the COVID-19 Pandemic. Phys. Ther. 2020, 100, 1058–1061. [Google Scholar] [CrossRef]
- Taoum, A.; Mourad-Chehade, F.; Amoud, H. Evidence-based model for real-time surveillance of ARDS. Biomed. Signal Process. Control. 2019, 50, 83–91. [Google Scholar] [CrossRef]
- Ding, X.-F.; Li, J.-B.; Liang, H.-Y.; Wang, Z.-Y.; Jiao, T.-T.; Liu, Z.; Yi, L.; Bian, W.-S.; Wang, S.-P.; Zhu, X.; et al. Predictive model for acute respiratory distress syndrome events in ICU patients in China using machine learning algorithms: A secondary analysis of a cohort study. J. Transl. Med. 2019, 17, 1–10. [Google Scholar] [CrossRef]
- Stout, N.L.; Binkley, J.M.; Schmitz, K.H.; Andrews, K.; Hayes, S.C.; Campbell, K.L.; Pt, M.L.M.; Soballe, P.W.; Berger, A.M.; Cheville, A.L.; et al. A prospective surveillance model for rehabilitation for women with breast cancer. Cancer 2012, 118, 2191–2200. [Google Scholar] [CrossRef]
- Stubblefield, M.D.; McNeely, M.L.; Alfano, C.M.; Mayer, D.K. A prospective surveillance model for physical rehabilitation of women with breast cancer. Cancer 2012, 118, 2250–2260. [Google Scholar] [CrossRef]
- Koelmeyer, L.A.; Moloney, E.; Boyages, J.; Sherman, K.A.; Dean, C.M. Prospective surveillance model in the home for breast cancer-related lymphoedema: A feasibility study. Breast Cancer Res. Treat. 2021, 185, 401–412. [Google Scholar] [CrossRef]
- Cheville, A.L.; McLaughlin, S.A.; Haddad, T.C.; Lyons, K.D.; Newman, R.; Ruddy, K.J. Integrated Rehabilitation for Breast Cancer Survivors. Am. J. Phys. Med. Rehabil. 2019, 98, 154–164. [Google Scholar] [CrossRef]
- Koelmeyer, L.; Gaitatzis, K.; Ridner, S.H.; Boyages, J.; Nelms, J.; Hughes, T.M.; Elder, E.; French, J.; Ngui, N.; Hsu, J.; et al. Implementing a prospective surveillance and early intervention model of care for breast cancer-related lymphedema into clinical practice: Application of the RE-AIM framework. Support. Care Cancer 2021, 29, 1081–1089. [Google Scholar] [CrossRef]
- Stout, N.L.; Andrews, K.; Binkley, J.M.; Schmitz, K.H.; Smith, R.A. Stakeholder perspectives on dissemination and implementation of a prospective surveillance model of rehabilitation for breast cancer treatment. Cancer 2012, 118, 2331–2334. [Google Scholar] [CrossRef]
- Stout, N.L.; Pfalzer, L.A.; Springer, B.; Levy, E.; McGarvey, C.L.; Danoff, J.V.; Gerber, L.H.; Soballe, P.W. Breast Cancer-Related Lymphedema: Comparing Direct Costs of a Prospective Surveillance Model and a Traditional Model of Care. Phys. Ther. 2012, 92, 152–163. [Google Scholar] [CrossRef] [PubMed]
- Coatsworth, N.; Myles, P.S.; Mann, G.J.; Cockburn, I.A.; Forbes, A.B.; Gardiner, E.E.; Lum, G.; Cheng, A.C.; Gruen, R.L. Prevalence of asymptomatic SARS-CoV-2 infection in elective surgical patients in Australia: A prospective surveillance study. ANZ J. Surg. 2021, 91, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Reforma, L.G.; Duffy, C.; Collier, A.-R.Y.; Wylie, B.J.; Shainker, S.A.; Golen, T.H.; Herlihy, M.; Lydeard, A.; Zera, C.A. A multidisciplinary telemedicine model for management of coronavirus disease 2019 (COVID-19) in obstetrical patients. Am. J. Obstet. Gynecol. MFM 2020, 2, 100180. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, W.; Wan, C.; Cheng, G.; Yin, Y.; Cao, K.; Zhang, X.; Wang, Z.; Miao, S.; Yu, Y.; et al. A predictive model for respiratory distress in patients with COVID-19: A retrospective study. Ann. Transl. Med. 2020, 8, 1585. [Google Scholar] [CrossRef]
- O’Brien, H.; Tracey, M.J.; Ottewill, C.; O’Brien, M.E.; Morgan, R.K.; Costello, R.W.; Gunaratnam, C.; Ryan, D.; McElvaney, N.G.; McConkey, S.J.; et al. An integrated multidisciplinary model of COVID-19 recovery care. Ir. J. Med Sci. 2020, 1–8. [Google Scholar] [CrossRef]
- Huang, C.; Huang, L.; Wang, Y.; Li, X.; Ren, L.; Gu, X.; Kang, L.; Guo, L.; Liu, M.; Zhou, X.; et al. 6-month consequences of COVID-19 in patients discharged from hospital: A cohort study. Lancet 2021, 397, 220–232. [Google Scholar] [CrossRef]
- Sudre, C.H.; Murray, B.; Varsavsky, T.; Graham, M.S.; Penfold, R.S.; Bowyer, R.C.; Pujol, J.C.; Klaser, K.; Antonelli, M.; Canas, L.S.; et al. Attributes and predictors of long COVID. Nat. Med. 2021, 1–6. [Google Scholar] [CrossRef]
- Osikomaiya, B.; Erinoso, O.; Wright, K.O.; Odusola, A.O.; Thomas, B.; Adeyemi, O.; Bowale, A.; Adejumo, O.; Falana, A.; Abdus-Salam, I.; et al. ‘Long COVID’: Persistent COVID-19 symptoms in survivors managed in Lagos State, Nigeria. BMC Infect. Dis. 2021, 21, 1–7. [Google Scholar] [CrossRef]
- Oxford Centre for Evidence-based Medicine—Levels of Evidence March 2009—CEBM. Available online: https://www.cebm.net/2009/06/oxford-centre-evidence-based-medicine-levels-evidence-march-2009/ (accessed on 5 May 2020).
- WHO. Global Surveillance for Human Infection with Coronavirus Disease (COVID-19); World Health Organization: Geneva, Switzerland, 2020; pp. 27–29. [Google Scholar]
- Smith, J.M.; Lee, A.C.; Zeleznik, H.; Scott, J.P.C.; Fatima, A.; Needham, D.M.; Ohtake, P.J. Home and Community-Based Physical Therapist Management of Adults with Post-Intensive Care Syndrome. Phys. Ther. 2020, 100, 1062–1073. [Google Scholar] [CrossRef]
- Ozemek, C.; Lavie, C.J.; Rognmo, Ø. Global physical activity levels—Need for intervention. Prog. Cardiovasc. Dis. 2019, 62, 102–107. [Google Scholar] [CrossRef]
- Hall, G.; Laddu, D.R.; Phillips, S.A.; Lavie, C.J.; Arena, R. A tale of two pandemics: How will COVID-19 and global trends in physical inactivity and sedentary behavior affect one another? Prog. Cardiovasc. Dis. 2021, 64, 108–110. [Google Scholar] [CrossRef]
- Bauer, U.E.; Briss, P.A.; Goodman, R.A.; Bowman, B.A. Prevention of chronic disease in the 21st century: Elimination of the leading preventable causes of premature death and disability in the USA. Lancet 2014, 384, 45–52. [Google Scholar] [CrossRef]
- Antonovsky, A. Health, Stress and Coping; Jossey Bass: San Francisco, CA, USA, 1979. [Google Scholar]
- Mo, X.; Jian, W.; Su, Z.; Chen, M.; Peng, H.; Peng, P.; Lei, C.; Chen, R.; Zhong, N.; Li, S. Abnormal pulmonary function in COVID-19 patients at time of hospital discharge. Eur. Respir. J. 2020, 55, 2001217. [Google Scholar] [CrossRef]
- Wang, L.; He, W.; Yu, X.; Hu, D.; Bao, M.; Liu, H.; Zhou, J.; Jiang, H. Coronavirus disease 2019 in elderly patients: Characteristics and prognostic factors based on 4-week follow-up. J. Infect. 2020, 80, 639–645. [Google Scholar] [CrossRef]
- Shi, S.; Qin, M.; Shen, B.; Cai, Y.; Liu, T.; Yang, F.; Gong, W.; Liu, X.; Liang, J.; Zhao, Q.; et al. Association of Cardiac Injury with Mortality in Hospitalized Patients with COVID-19 in Wuhan, China. JAMA Cardiol. 2020, 5, 802. [Google Scholar] [CrossRef]
- Trejo-Gabriel-Galán, J. Ictus como complicación y como factor pronóstico de COVID-19. Neurología 2020, 35, 318–322. [Google Scholar] [CrossRef]
- Mao, L.; Wang, M.; Chen, S.; He, Q.; Chang, J.; Hong, C.; Zhou, Y.; Wang, D.; Miao, X.; Hu, Y.; et al. Neurological Manifestations of Hospitalized Patients with COVID-19 in Wuhan, China: A Retrospective Case Series Study. SSRN Electron. J. 2020. [Google Scholar] [CrossRef]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- Report of the WHO-China Joint Mission on Coronavirus Disease 2019 (COVID-19). February 2020. Available online: https://www.who.int/docs/default-source/coronaviruse/who-china-joint-mission-on-covid-19-final-report.pdf (accessed on 25 March 2020).
- Guidon, A.C.; Amato, A.A. COVID-19 and neuromuscular disorders. Neurology 2020, 94, 959–969. [Google Scholar] [CrossRef]
- Bercker, S.; Weber-Carstens, S.; Deja, M.; Grimm, C.; Wolf, S.; Behse, F.; Busch, T.; Falke, K.J.; Kaisers, U. Critical illness polyneuropathy and myopathy in patients with acute respiratory distress syndrome. Crit. Care Med. 2005, 33, 711–715. [Google Scholar] [CrossRef]
- Kim, J.-E.; Heo, J.-H.; Kim, H.-O.; Song, S.-H.; Park, S.-S.; Park, T.-H.; Ahn, J.-Y.; Kim, M.-K.; Choi, J.-P. Neurological Complications during Treatment of Middle East Respiratory Syndrome. J. Clin. Neurol. 2017, 13, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Tsai, L.-K.; Hsieh, S.-T.; Chang, Y.-C. Neurological manifestations in severe acute respiratory syndrome. Acta Neurol. Taiwanica 2005, 14, 113–119. [Google Scholar]
- Needham, D.M.; Sepulveda, K.A.; Dinglas, V.D.; Chessare, C.M.; Friedman, L.A.; Bingham, C.O.; Turnbull, A.E. Core Outcome Measures for Clinical Research in Acute Respiratory Failure Survivors. An International Modified Delphi Consensus Study. Am. J. Respir. Crit. Care Med. 2017, 196, 1122–1130. [Google Scholar] [CrossRef] [PubMed]
- Herridge, M.S.; Moss, M.; Hough, C.L.; Hopkins, R.O.; Rice, T.W.; Bienvenu, O.J.; Azoulay, E. Recovery and outcomes after the acute respiratory distress syndrome (ARDS) in patients and their family caregivers. Intensive Care Med. 2016, 42, 725–738. [Google Scholar] [CrossRef] [PubMed]
- Conti, P.; Ronconi, G.; Caraffa, A.L.; Gallenga, C.E.; Ross, R.; Frydas, I.; Kritas, S.K. Induction of pro-inflammatory cytokines (IL-1 and IL-6) and lung inflammation by Coronavirus-19 (COVI-19 or SARS-CoV-2): Anti-inflammatory strategies. J. Biol. Regul. Homeost. Agents 2020, 34, 327–331. [Google Scholar] [CrossRef] [PubMed]
- Laisné, F.; LeComte, C.; Corbière, M. Biopsychosocial predictors of prognosis in musculoskeletal disorders: A systematic review of the literature (corrected and republished). Disabil. Rehabil. 2012, 34, 1912–1941. [Google Scholar] [CrossRef]
- Brown, S.M.; Bose, S.; Banner-Goodspeed, V.; Beesley, S.J.; Dinglas, V.D.; Hopkins, R.O.; Jackson, J.C.; Mir-Kasimov, M.; Needham, D.M.; Sevin, C.M.; et al. Approaches to Addressing Post-Intensive Care Syndrome among Intensive Care Unit Survivors. A Narrative Review. Ann. Am. Thorac. Soc. 2019, 16, 947–956. [Google Scholar] [CrossRef]
- Zhao, H.-M.; Xie, Y.-X.; Wang, C. Recommendations for respiratory rehabilitation in adults with coronavirus disease 2019. Chin. Med. J. 2020, 133, 1595–1602. [Google Scholar] [CrossRef]
- Holland, A.E.; Spruit, M.A.; Troosters, T.; Puhan, M.A.; Pepin, V.; Saey, D.; McCormack, M.C.; Carlin, B.W.; Sciurba, F.C.; Pitta, F.; et al. An official European Respiratory Society/American Thoracic Society technical standard: Field walking tests in chronic respiratory disease. Eur. Respir. J. 2014, 44, 1428–1446. [Google Scholar] [CrossRef]
- Hunter, J.; Rawlings-Anderson, K. Respiratory assessment. Nurs. Stand. 2008, 22, 41–43. [Google Scholar] [CrossRef] [PubMed]
- Ross, R.; Neeland, I.J.; Yamashita, S.; Shai, I.; Seidell, J.; Magni, P.; Santos, R.D.; Arsenault, B.; Cuevas, A.; Hu, F.B.; et al. Waist circumference as a vital sign in clinical practice: A Consensus Statement from the IAS and ICCR Working Group on Visceral Obesity. Nat. Rev. Endocrinol. 2020, 16, 177–189. [Google Scholar] [CrossRef] [PubMed]
- Snijder, M.B.; Dekker, J.M.; Visser, M.; Bouter, L.M.; Stehouwer, C.D.A.; Kostense, P.J.; Yudkin, J.S.; Heine, R.J.; Nijpels, G.; Seidell, J.C. Associations of hip and thigh circumferences independent of waist circumference with the incidence of type 2 diabetes: The Hoorn Study. Am. J. Clin. Nutr. 2003, 77, 1192–1197. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; for the DECODE Study Group; Jousilahti, P.; Stehouwer, C.D.A.; Söderberg, S.; Onat, A.; Laatikainen, T.; Yudkin, J.S.; Dankner, R.; Morris, R.; et al. Comparison of various surrogate obesity indicators as predictors of cardiovascular mortality in four European populations. Eur. J. Clin. Nutr. 2013, 67, 1298–1302. [Google Scholar] [CrossRef] [PubMed]
- Waist Circumference and Waist-Hip Ratio: Report of a WHO Expert Consultation. World Health Organization: Geneva, Switzerland. Available online: https://www.who.int/publications/i/item/9789241501491 (accessed on 20 May 2020).
- Bagchi, D.; Sreejayan, N. Nutritional and Therapeutic Interventions for Diabetes and Metabolic Syndrome; Elsevier Inc.: Amsterdam, The Netherlands, 2012; ISBN 9780123850836. [Google Scholar]
- Petersen, A.; Bressem, K.; Albrecht, J.; Thieß, H.-M.; Vahldiek, J.; Hamm, B.; Makowski, M.R.; Niehues, A.; Niehues, S.M.; Adams, L.C. The role of visceral adiposity in the severity of COVID-19: Highlights from a unicenter cross-sectional pilot study in Germany. Metabolism 2020, 110, 154317. [Google Scholar] [CrossRef]
- Watanabe, M.; Caruso, D.; Tuccinardi, D.; Risi, R.; Zerunian, M.; Polici, M.; Pucciarelli, F.; Tarallo, M.; Strigari, L.; Manfrini, S.; et al. Visceral fat shows the strongest association with the need of intensive care in patients with COVID-19. Metabolism 2020, 111, 154319. [Google Scholar] [CrossRef]
- Woods, J.A.; Hutchinson, N.T.; Powers, S.K.; Roberts, W.O.; Gomez-Cabrera, M.C.; Radak, Z.; Berkes, I.; Boros, A.; Boldogh, I.; Leeuwenburgh, C.; et al. The COVID-19 pandemic and physical activity. Sports Med. Health Sci. 2020, 2, 55–64. [Google Scholar] [CrossRef]
- Lim, M.A.; Pranata, R. The Danger of Sedentary Lifestyle in Diabetic and Obese People during the COVID-19 Pandemic. Clin. Med. Insights Endocrinol. Diabetes 2020, 13. [Google Scholar] [CrossRef]
- Alomari, M.A.; Khabour, O.F.; Alzoubi, K.H. Changes in Physical Activity and Sedentary Behavior Amid Confinement: The BKSQ-COVID-19 Project. Risk Manag. Healthc. Policy 2020, 13, 1757–1764. [Google Scholar] [CrossRef]
- Ali, A.M.; Kunugi, H. COVID-19: A pandemic that threatens physical and mental health by promoting physical inactivity. Sports Med. Health Sci. 2020, 2, 221–223. [Google Scholar] [CrossRef]
- Rubio Castaneda, F.J.; Tomas Aznar, C.; Muro Baquero, C. Medición de la actividad física en personas mayores de 65 años mediante el IPAQ-E: Validez de contenido, fiabilidad y factores asociados [Validity, Reliability and Associated Factors of the International Physical Activity Questionnaire Adapted to Elderly (IPAQ-E)]. Rev. Esp. Salud Publica 2017, 91, e201701004. [Google Scholar]
- Adult Physical Inactivity Prevalence Maps by Race/Ethnicity|Physical Activity|CDC. Available online: https://www.cdc.gov/physicalactivity/data/inactivity-prevalence-maps/index.html (accessed on 31 March 2021).
- Brug, J.; on behalf of the DEDIPAC Consortium; Van Der Ploeg, H.P.; Loyen, A.; Ahrens, W.; Allais, O.; Andersen, L.F.; Cardon, G.; Capranica, L.; Chastin, S.; et al. Determinants of diet and physical activity (DEDIPAC): A summary of findings. Int. J. Behav. Nutr. Phys. Act. 2017, 14, 1–24. [Google Scholar] [CrossRef]
- Filgueira, T.O.; Castoldi, A.; Santos, L.E.R.; de Amorim, G.J.; Fernandes, M.S.D.S.; de Lima do Nascimento Anastácio, W.; Campos, E.Z.; Santos, T.M.; Souto, F.O. The Relevance of a Physical Active Lifestyle and Physical Fitness on Immune Defense: Mitigating Disease Burden, with Focus on COVID-19 Consequences. Front. Immunol. 2021, 12, 587146. [Google Scholar] [CrossRef]
- Ricci, F.; Izzicupo, P.; Moscucci, F.; Sciomer, S.; Maffei, S.; Di Baldassarre, A.; Mattioli, A.V.; Gallina, S. Recommendations for Physical Inactivity and Sedentary Behavior During the Coronavirus Disease (COVID-19) Pandemic. Front. Public Health 2020, 8, 199. [Google Scholar] [CrossRef]
- Division AGD of HPH. Australia’s Physical Activity and Sedentary Behaviour Guidelines and the Australian 24-Hour Movement Guidelines 2019. Available online: https://www1.health.gov.au/internet/main/publishing.nsf/Content/pasb (accessed on 19 August 2020).
- Salman, D.; Vishnubala, D.; Le Feuvre, P.; Beaney, T.; Korgaonkar, J.; Majeed, A.; McGregor, A.H. Returning to physical activity after covid-19. BMJ 2021, 372, m4721. [Google Scholar] [CrossRef]
- Chen, Y.-C.; Chen, K.-C.; Lu, L.-H.; Wu, Y.-L.; Lai, T.-J.; Wang, C.-H. Validating the 6-minute walk test as an indicator of recovery in patients undergoing cardiac surgery. Medicine 2018, 97, e12925. [Google Scholar] [CrossRef]
- Stojanov, J.; Malobabic, M.; Stanojevic, G.; Stevic, M.; Milosevic, V.; Stojanov, A. Quality of sleep and health-related quality of life among health care professionals treating patients with coronavirus disease-19. Int. J. Soc. Psychiatry 2020. [Google Scholar] [CrossRef]
- Raman, B.; Cassar, M.P.; Tunnicliffe, E.M.; Filippini, N.; Griffanti, L.; Alfaro-Almagro, F.; Okell, T.; Sheerin, F.; Xie, C.; Mahmod, M.; et al. Medium-term effects of SARS-CoV-2 infection on multiple vital organs, exercise capacity, cognition, quality of life and mental health, post-hospital discharge. EClinicalMedicine 2021, 31, 100683. [Google Scholar] [CrossRef]
- Dweck, M.R.; Bularga, A.; Hahn, R.T.; Bing, R.; Lee, K.K.; Chapman, A.R.; White, A.; Di Salvo, G.; Sade, L.E.; Pearce, K.; et al. Global evaluation of echocardiography in patients with COVID-19. Eur. Hear. J. Cardiovasc. Imaging 2020, 21, 949–958. [Google Scholar] [CrossRef]
- Frontera, W.R.; Slovik, D.M.; Dawson, D.M.; David, M. Exercise in Rehabilitation Medicine; Human Kinetics: Champaign, IL, USA, 2006. [Google Scholar]
- Ingle, L.; Cleland, J.G.; Clark, A.L. The Long-Term Prognostic Significance of 6-Minute Walk Test Distance in Patients with Chronic Heart Failure. BioMed Res. Int. 2014, 2014, 1–7. [Google Scholar] [CrossRef]
- Hunt, S.A.; Abraham, W.T.; Chin, M.H.; Feldman, A.M.; Francis, G.S.; Ganiats, T.G.; Jessup, M.; Konstam, M.A.; Mancini, D.M.; et al. ACC/AHA 2005 Guideline Update for the Diagnosis and Management of Chronic Heart Failure in the Adult. Circulation 2005, 112, e154–e235. [Google Scholar] [CrossRef]
- American Thoracic Society. American College of Chest Physicians ATS/ACCP Statement on Cardiopulmonary Exercise Testing. Am. J. Respir. Crit. Care Med. 2003, 167, 211–277. [Google Scholar] [CrossRef]
- Mantha, S.; Tripuraneni, S.L.; Roizen, M.F.; Fleisher, L.A. Proposed Modifications in the 6-Minute Walk Test for Potential Application in Patients with Mild COVID-19: A Step to Optimize Triage Guidelines. Anesth. Analg. 2020, 131, 398–402. [Google Scholar] [CrossRef]
- Guazzi, M.; Adams, V.; Conraads, V.M.; Halle, M.; Mezzani, A.; Vanhees, L.; Arena, R.; Fletcher, G.F.; Forman, D.E.; Kitzman, D.W.; et al. Clinical Recommendations for Cardiopulmonary Exercise Testing Data Assessment in Specific Patient Populations. Circulation 2012, 126, 2261–2274. [Google Scholar] [CrossRef] [PubMed]
- Clavario, P.; de Marzo, V.; Lotti, R.; Barbara, C.; Porcile, A.; Russo, C.; Beccaria, F.; Bonavia, M.; Bottaro, L.C.; Caltabellotta, M.; et al. Assessment of functional capacity with cardiopulmonary exercise testing in non-severe COVID-19 patients at three months follow-up. MedRxiv 2020. [Google Scholar] [CrossRef]
- Dorelli, G.; Braggio, M.; Gabbiani, D.; Busti, F.; Caminati, M.; Senna, G.; Girelli, D.; Laveneziana, P.; Ferrari, M.; Sartori, G.; et al. Importance of Cardiopulmonary Exercise Testing amongst Subjects Recovering from COVID-19. Diagnostics 2021, 11, 507. [Google Scholar] [CrossRef]
- Hodgson, C.L.; Stiller, K.; Needham, D.M.; Tipping, C.J.; Harrold, M.; Baldwin, C.E.; Bradley, S.; Berney, S.; Caruana, L.R.; Elliott, D.; et al. Expert consensus and recommendations on safety criteria for active mobilization of mechanically ventilated critically ill adults. Crit. Care 2014, 18, 1–9. [Google Scholar] [CrossRef]
- Okechukwu, C.E.; Deb, A.A.; Emara, S.; Abbas, S.A. Physical activity as preventive therapy for older adults: A narrative review. Niger. J. Exp. Clin. Biosci. 2019, 7, 82. [Google Scholar] [CrossRef]
- Qiu, S.; Cai, X.; Sun, Z.; Li, L.; Zuegel, M.; Steinacker, J.M.; Schumann, U. Heart Rate Recovery and Risk of Cardiovascular Events and All-Cause Mortality: A Meta-Analysis of Prospective Cohort Studies. J. Am. Hear. Assoc. 2017, 6, e005505. [Google Scholar] [CrossRef]
- Peçanha, T.; Silva-Júnior, N.D.; Forjaz, C.L.D.M. Heart rate recovery: Autonomic determinants, methods of assessment and association with mortality and cardiovascular diseases. Clin. Physiol. Funct. Imaging 2014, 34, 327–339. [Google Scholar] [CrossRef]
- Pellegrino, R.; Viegi, G.; Brusasco, V.; Crapo, R.O.; Burgos, F.; Casaburi, R.; Coates, A.; Van Der Grinten, C.P.M.; Gustafsson, P.; Hankinson, J.; et al. Interpretative strategies for lung function tests. Eur. Respir. J. 2005, 26, 948–968. [Google Scholar] [CrossRef]
- American Thoracic Society. ATS/ERS Statement on Respiratory Muscle Testing. Am. J. Respir. Crit. Care Med. 2002, 166, 518–624. [Google Scholar] [CrossRef]
- Barreiro, E.; Bustamante, V.; Cejudo, P.; Gáldiz, J.B.; Gea, J.; de Lucas, P.; Martínez-Llorens, J.; Ortega, F.; Puente-Maestu, L.; Roca, J.; et al. Normativa SEPAR sobre disfunción muscular de los pacientes con enfermedad pulmonar obstructiva crónica. Archivos de Bronconeumología 2015, 51, 384–395. [Google Scholar] [CrossRef] [PubMed]
- Simonelli, C.; Paneroni, M.; Fokom, A.G.; Saleri, M.; Speltoni, I.; Favero, I.; Garofali, F.; Scalvini, S.; Vitacca, M. How the COVID-19 infection tsunami revolutionized the work of respiratory physiotherapists: An experience from Northern Italy. Monaldi Arch. Chest Dis. 2020, 90. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.-L.; Yang, T. Pulmonary rehabilitation for patients with coronavirus disease 2019 (COVID-19). Chronic Dis. Transl. Med. 2020, 6, 79–86. [Google Scholar] [CrossRef]
- Liu, K.; Zhang, W.; Yang, Y.; Zhang, J.; Li, Y.; Chen, Y. Respiratory rehabilitation in elderly patients with COVID-19: A randomized controlled study. Complement. Ther. Clin. Pr. 2020, 39, 101166. [Google Scholar] [CrossRef]
- Paliwal, V.K.; Garg, R.K.; Gupta, A.; Tejan, N. Neuromuscular presentations in patients with COVID-19. Neurol. Sci. 2020, 41, 3039–3056. [Google Scholar] [CrossRef]
- Vasconcelos, K.S.D.S.; Dias, J.M.D.; Bastone, A.D.C.; Vieira, R.A.; Andrade, A.C.D.S.; Perracini, M.R.; Guerra, R.O.; Dias, R.C. Handgrip strength cutoff points to identify mobility limitation in community-dwelling older people and associated factors. J. Nutr. Health Aging 2016, 20, 306–315. [Google Scholar] [CrossRef]
- Ali, N.A.; O’Brien, J.M.; Hoffmann, S.P.; Phillips, G.; Garland, A.; Finley, J.C.W.; Almoosa, K.; Hejal, R.; Wolf, K.M.; Lemeshow, S.; et al. Acquired Weakness, Handgrip Strength, and Mortality in Critically Ill Patients. Am. J. Respir. Crit. Care Med. 2008, 178, 261–268. [Google Scholar] [CrossRef]
- Sidiras, G.; Patsaki, I.; Karatzanos, E.; Dakoutrou, M.; Kouvarakos, A.; Mitsiou, G.; Routsi, C.; Stranjalis, G.; Nanas, S.; Gerovasili, V. Long term follow-up of quality of life and functional ability in patients with ICU acquired Weakness—A post hoc analysis. J. Crit. Care 2019, 53, 223–230. [Google Scholar] [CrossRef]
- Mahoney, F.I.; Barthel, D.W. Functional Evaluation: The Barthel Index. Md. State Med. J. 1965, 14, 61–65. [Google Scholar]
- Elliott, D.; Denehy, L.; Berney, S.; Alison, J.A. Assessing physical function and activity for survivors of a critical illness: A review of instruments. Aust. Crit. Care 2011, 24, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Vélez, R.; Pérez-Sousa, M.A.; Venegas-Sanabria, L.C.; Cano-Gutierrez, C.A.; Hernández-Quiñonez, P.A.; Rincón-Pabón, D.; García-Hermoso, A.; Zambom-Ferraresi, F.; De Asteasu, M.L.S.; Izquierdo, M. Normative Values for the Short Physical Performance Battery (SPPB) and Their Association With Anthropometric Variables in Older Colombian Adults. The SABE Study, 2015. Front. Med. 2020, 7, 52. [Google Scholar] [CrossRef] [PubMed]
- Norkin, C.C.; White, D.J. Measurement of Joint Motion: A Guide to Goniometry; F. A. Davis Company: Philadelphia, PA, USA, 2016; ISBN 9780803645660. [Google Scholar]
- Santos, G.M.; Souza, A.C.S.; Virtuoso, J.F.; Tavares, G.M.S.; Mazo, G.Z. Predictive values at risk of falling in physically active and no active elderly with Berg Balance Scale. Braz. J. Phys. Ther. 2011, 15, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Bennett, M. The LANSS Pain Scale: The Leeds assessment of neuropathic symptoms and signs. Pain 2001, 92, 147–157. [Google Scholar] [CrossRef]
- Finnerup, N.B.; Haroutounian, S.; Kamerman, P.; Baron, R.; Bennett, D.L.; Bouhassira, D.; Cruccu, G.; Freeman, R.; Hansson, P.; Nurmikko, T.; et al. Neuropathic pain: An updated grading system for research and clinical practice. Pain 2016, 157, 1599–1606. [Google Scholar] [CrossRef]
- Boonstra, A.M.; Stewart, R.E.; Köke, A.J.A.; Oosterwijk, R.F.A.; Swaan, J.L.; Schreurs, K.M.G.; Preuper, H.R.S. Cut-Off Points for Mild, Moderate, and Severe Pain on the Numeric Rating Scale for Pain in Patients with Chronic Musculoskeletal Pain: Variability and Influence of Sex and Catastrophizing. Front. Psychol. 2016, 7, 1466. [Google Scholar] [CrossRef]
- Daniela, S. The Post-Intensive Care Syndrome (PICS): Impact of ICU-Stay on Functioning and Implications for Rehabilitation Care; Dettling-Ihnenfeldt: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Ekelund, U.; Steene-Johannessen, J.; Brown, W.J.; Fagerland, M.W.; Owen, N.; Powell, K.E.; Bauman, A.; Lee, I.-M. Does physical activity attenuate, or even eliminate, the detrimental association of sitting time with mortality? A harmonised meta-analysis of data from more than 1 million men and women. Lancet 2016, 388, 1302–1310. [Google Scholar] [CrossRef]
- Alawna, M.; Amro, M.; Mohamed, A.A. Aerobic exercises recommendations and specifications for patients with COVID-19: A systematic review. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 13049–13055. [Google Scholar]
- Sagarra-Romero, L.; Viñas-Barros, A. COVID-19: Short and Long-Term Effects of Hospitalization on Muscular Weakness in the Elderly. Int. J. Environ. Res. Public Health 2020, 17, 8715. [Google Scholar] [CrossRef]
- Akyuz, G.; Kenis, O. Physical Therapy Modalities and Rehabilitation Techniques in the Management of Neuropathic Pain. Am. J. Phys. Med. Rehabil. 2014, 93, 253–259. [Google Scholar] [CrossRef]
- Orsucci, D.; Ienco, E.C.; Nocita, G.; Napolitano, A.; Vista, M. Neurological features of COVID-19 and their treatment: A review. Drugs Context 2020, 9, 1–12. [Google Scholar] [CrossRef]
- Kemp, H.I.; Corner, E.; Colvin, L.A. Chronic pain after COVID-19: Implications for rehabilitation. Br. J. Anaesth. 2020, 125, 436–440. [Google Scholar] [CrossRef]
- Hans, G.H.; Wildemeersch, D. Impact of SARS-CoV-2 Infection on the Epidemiology of Chronic Pain and Long-Term Disability: Prepare for the Next Perfect Storm. Front. Pain Res. 2020, 1, 616284. [Google Scholar] [CrossRef]
- Mayer, T.G.; Neblett, R.; Cohen, H.; Howard, K.J.; Choi, Y.H.; Williams, M.J.; Perez, Y.; Gatchel, R.J. The Development and Psychometric Validation of the Central Sensitization Inventory. Pain Pract. 2011, 12, 276–285. [Google Scholar] [CrossRef]
- Arribas-Romano, A.; Fernández-Carnero, J.; Molina-Rueda, F.; Angulo-Diaz-Parreño, S.; Navarro-Santana, M.J. Efficacy of Physical Therapy on Nociceptive Pain Processing Alterations in Patients with Chronic Musculoskeletal Pain: A Systematic Review and Meta-analysis. Pain Med. 2020, 21, 2502–2517. [Google Scholar] [CrossRef]
- Malfliet, A.; Kregel, J.; Meeus, M.; Cagnie, B.; Roussel, N.; Dolphens, M.; Danneels, L.; Nijs, J. Applying contemporary neuroscience in exercise interventions for chronic spinal pain: Treatment protocol. Braz. J. Phys. Ther. 2017, 21, 378–387. [Google Scholar] [CrossRef]
- Curatolo, M.; Arendt-Nielsen, L.; Petersen-Felix, S. Central Hypersensitivity in Chronic Pain: Mechanisms and Clinical Implications. Phys. Med. Rehabil. Clin. N. Am. 2006, 17, 287–302. [Google Scholar] [CrossRef] [PubMed]
- Mazza, M.G.; De Lorenzo, R.; Conte, C.; Poletti, S.; Vai, B.; Bollettini, I.; Melloni, E.M.T.; Furlan, R.; Ciceri, F.; Rovere-Querini, P.; et al. Anxiety and depression in COVID-19 survivors: Role of inflammatory and clinical predictors. Brain Behav. Immun. 2020, 89, 594–600. [Google Scholar] [CrossRef]
- Garrigues, E.; Janvier, P.; Kherabi, Y.; Le Bot, A.; Hamon, A.; Gouze, H.; Doucet, L.; Berkani, S.; Oliosi, E.; Mallart, E.; et al. Post-discharge persistent symptoms and health-related quality of life after hospitalization for COVID-19. J. Infect. 2020, 81, e4–e6. [Google Scholar] [CrossRef]
- Rogers, J.P.; Chesney, E.; Oliver, D.; Pollak, T.A.; McGuire, P.; Fusar-Poli, P.; Zandi, M.S.; Lewis, G.; David, A.S. Psychiatric and neuropsychiatric presentations associated with severe coronavirus infections: A systematic review and meta-analysis with comparison to the COVID-19 pandemic. Lancet Psychiatry 2020, 7, 611–627. [Google Scholar] [CrossRef]
- Jutte, J.E.; Needham, D.M.; Pfoh, E.R.; Bienvenu, O.J. Psychometric evaluation of the Hospital Anxiety and Depression Scale 3 months after acute lung injury. J. Crit. Care 2015, 30, 793–798. [Google Scholar] [CrossRef] [PubMed]
- Bienvenu, O.J.; Williams, J.B.; Yang, A.; Hopkins, R.O.; Needham, D.M. Posttraumatic Stress Disorder in Survivors of Acute Lung Injury. Chest 2013, 144, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Nasreddine, Z.S.; Phillips, N.A.; Bédirian, V.; Charbonneau, S.; Whitehead, V.; Collin, I.; Cummings, J.L.; Chertkow, H. The Montreal Cognitive Assessment, MoCA: A Brief Screening Tool for Mild Cognitive Impairment. J. Am. Geriatr. Soc. 2005, 53, 695–699. [Google Scholar] [CrossRef] [PubMed]
- Palem, S.P.; Palem, H.P. The effect of COVID-19 on global population and its fatality rate: Retrospective study by online database. Indian J. Med. Sci. 2020, 72, 13–16. [Google Scholar] [CrossRef]
- Nalbandian, A.; Sehgal, K.; Gupta, A.; Madhavan, M.V.; McGroder, C.; Stevens, J.S.; Cook, J.R.; Nordvig, A.S.; Shalev, D.; Sehrawat, T.S.; et al. Post-acute COVID-19 syndrome. Nat. Med. 2021, 1–15. [Google Scholar] [CrossRef]
| Condition | |
|---|---|
| 1 | (COVID-19 [tiab]) OR (Coronavirus disease 2019[tiab]) OR (COVID *[tiab]) |
| Cardiopulmonary system | |
| 2 | (exercise[Mesh]) OR (physical activity [tiab]) OR (physical exercise[tiab]) OR (cardiovascular system[Mesh]) OR (cardiovascular systems[tiab]) OR (circulatory system[tiab]) or (circulatory systems[tiab]) OR (cardiorespiratory fitness [Mesh]) OR (cardiorespiratory exercise testing [tiab]) OR (functional capacity [tiab]) OR (respiratory system[Mesh]) OR (respiratory systems[tiab]) OR (respiratory tract[tiab]) OR (respiratory tracts[tiab]) OR (respiratory function test[Mesh]) OR (maximal respiratory pressures[tiab]) OR (breathing exercises[tiab]) |
| Neuromuscular system | |
| 3 | (musculoskeletal system[Mesh]) OR (musculoskeletal systems[tiab]) OR (nervous system[Mesh]) OR (neuralgia [Mesh]) OR (neuropathic pain[tiab]) OR (neuropathies[tiab]) OR (myalgia[tiab]) OR (paresthesia[Mesh]) OR (neurologic manifestation[Mesh]) |
| Mental health | |
| 4 | (mental health [Mesh terms]) OR (mental hygiene[tiab]). |
| Definitive search | |
| 5 | 1 AND 2 |
| 6 | 1 AND 3 |
| 7 | 1 AND 4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Postigo-Martin, P.; Cantarero-Villanueva, I.; Lista-Paz, A.; Castro-Martín, E.; Arroyo-Morales, M.; Seco-Calvo, J. A COVID-19 Rehabilitation Prospective Surveillance Model for Use by Physiotherapists. J. Clin. Med. 2021, 10, 1691. https://doi.org/10.3390/jcm10081691
Postigo-Martin P, Cantarero-Villanueva I, Lista-Paz A, Castro-Martín E, Arroyo-Morales M, Seco-Calvo J. A COVID-19 Rehabilitation Prospective Surveillance Model for Use by Physiotherapists. Journal of Clinical Medicine. 2021; 10(8):1691. https://doi.org/10.3390/jcm10081691
Chicago/Turabian StylePostigo-Martin, Paula, Irene Cantarero-Villanueva, Ana Lista-Paz, Eduardo Castro-Martín, Manuel Arroyo-Morales, and Jesús Seco-Calvo. 2021. "A COVID-19 Rehabilitation Prospective Surveillance Model for Use by Physiotherapists" Journal of Clinical Medicine 10, no. 8: 1691. https://doi.org/10.3390/jcm10081691
APA StylePostigo-Martin, P., Cantarero-Villanueva, I., Lista-Paz, A., Castro-Martín, E., Arroyo-Morales, M., & Seco-Calvo, J. (2021). A COVID-19 Rehabilitation Prospective Surveillance Model for Use by Physiotherapists. Journal of Clinical Medicine, 10(8), 1691. https://doi.org/10.3390/jcm10081691









