Complex Liver Resections for Intrahepatic Cholangiocarcinoma
Abstract
1. Introduction
2. Methods
2.1. Patient Selection and Definitions
- (a)
- Conventional liver resections: major anatomical or non-anatomical hepatectomy without vascular reconstruction and any minor anatomical or non-anatomical hepatectomy.
- (b)
- Complex liver resections: extended liver resection or major anatomical hepatectomy with vascular reconstruction, ALPPS and ante situm resection.
- (c)
- Exploration: Patients were preoperative, considered as resectable but were found to be unresectable during exploration.
2.2. Statistical Analysis
3. Results
3.1. Study Population
3.2. Preoperative Characteristics
3.3. Operative and Resection Details
3.4. Outcome and Complications
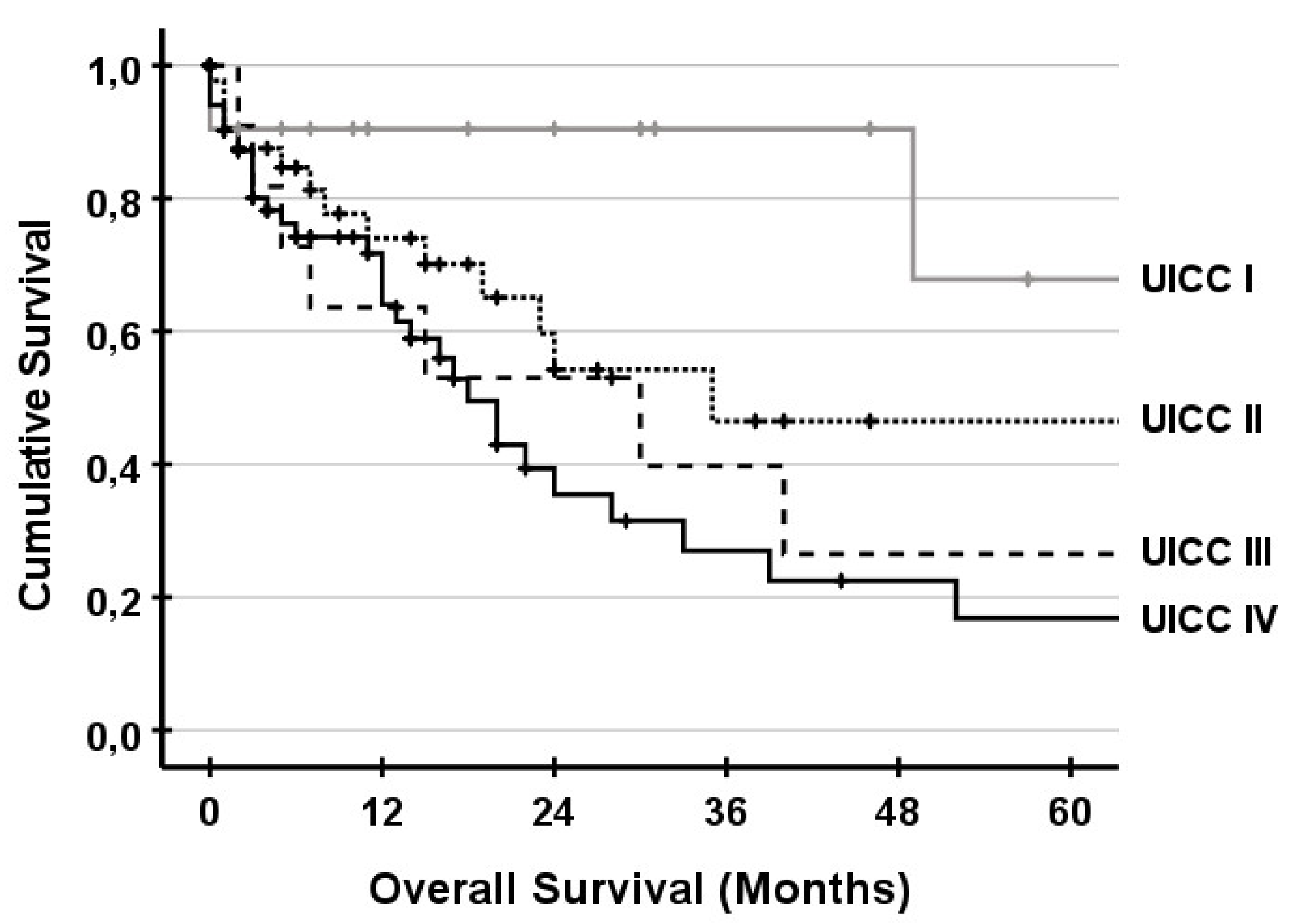
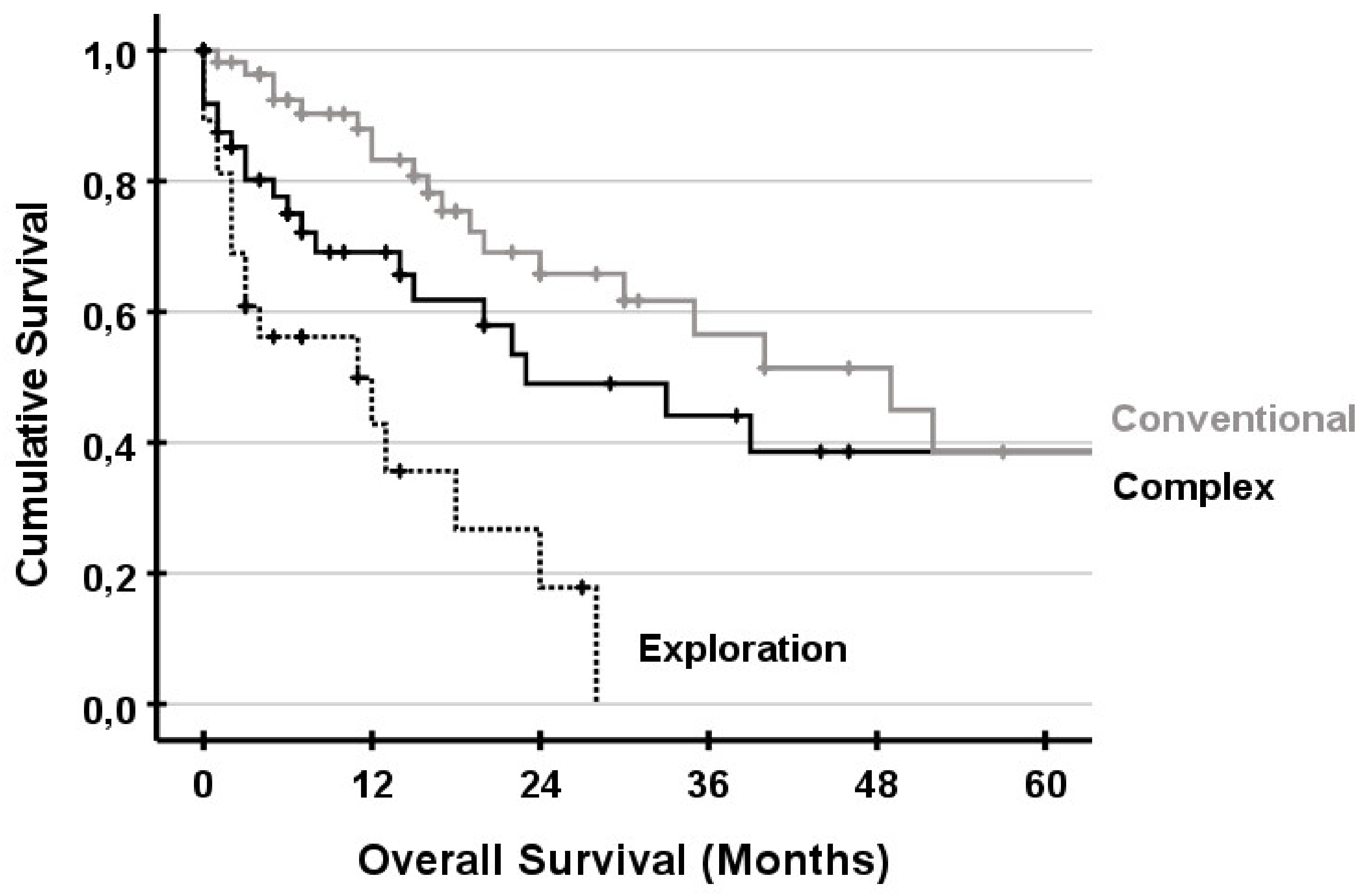
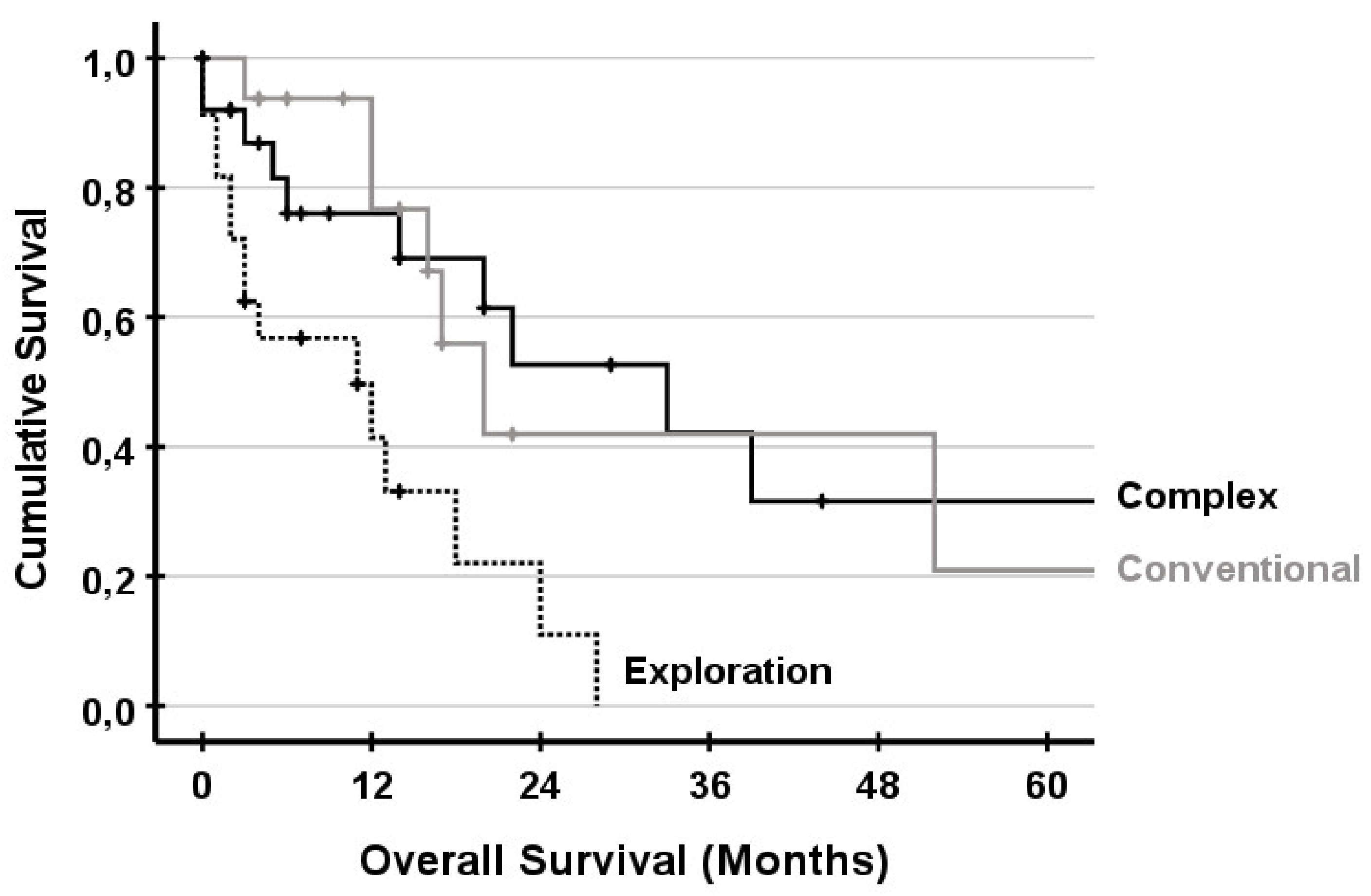
3.5. Survival
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Von Hahn, T.; Ciesek, S.; Wegener, G.; Plentz, R.R.; Weismüller, T.J.; Wedemeyer, H.; Manns, M.P.; Greten, T.F.; Malek, N.P. Epidemiological Trends in Incidence and Mortality of Hepatobiliary Cancers in Germany. Scand. J. Gastroenterol. 2011, 46, 1092–1098. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.A.; Emadossadaty, S.; Ladep, N.G.; Thomas, H.C.; Elliott, P.; Taylor-Robinson, S.D.; Toledano, M.B. Rising Trends in Cholangiocarcinoma: Is the ICD Classification System Misleading Us? J. Hepatol. 2012, 56, 848–854. [Google Scholar] [CrossRef] [PubMed]
- Altekruse, S.F.; Petrick, J.L.; Rolin, A.I.; Cuccinelli, J.E.; Zou, Z.; Tatalovich, Z.; McGlynn, K.A. Geographic Variation of Intrahepatic Cholangiocarcinoma, Extrahepatic Cholangiocarcinoma, and Hepatocellular Carcinoma in the United States. PLoS ONE 2015, 10, e0120574. [Google Scholar] [CrossRef] [PubMed]
- Bertuccio, P.; Malvezzi, M.; Carioli, G.; Hashim, D.; Boffetta, P.; El-Serag, H.B.; La Vecchia, C.; Negri, E. Global Trends in Mortality from Intrahepatic and Extrahepatic Cholangiocarcinoma. J. Hepatol. 2019, 71, 104–114. [Google Scholar] [CrossRef] [PubMed]
- Fostea, R.M.; Fontana, E.; Torga, G.; Arkenau, H.-T. Recent Progress in the Systemic Treatment of Advanced/Metastatic Cholangiocarcinoma. Cancers 2020, 12, 2599. [Google Scholar] [CrossRef] [PubMed]
- Amini, N.; Ejaz, A.; Spolverato, G.; Kim, Y.; Herman, J.M.; Pawlik, T.M. Temporal Trends in Liver-Directed Therapy of Patients with Intrahepatic Cholangiocarcinoma in the United States: A Population-Based Analysis. J. Surg. Oncol. 2014, 110, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Edeline, J.; Benabdelghani, M.; Bertaut, A.; Watelet, J.; Hammel, P.; Joly, J.-P.; Boudjema, K.; Fartoux, L.; Bouhier-Leporrier, K.; Jouve, J.-L.; et al. Gemcitabine and Oxaliplatin Chemotherapy or Surveillance in Resected Biliary Tract Cancer (PRODIGE 12-ACCORD 18-UNICANCER GI): A Randomized Phase III Study. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2019, 37, 658–667. [Google Scholar] [CrossRef]
- Rizzo, A.; Brandi, G. Pitfalls, Challenges, and Updates in Adjuvant Systemic Treatment for Resected Biliary Tract Cancer. Expert Rev. Gastroenterol. Hepatol. 2021, 1–8. [Google Scholar] [CrossRef]
- Beal, E.W.; Cloyd, J.M.; Pawlik, T.M. Surgical Treatment of Intrahepatic Cholangiocarcinoma: Current and Emerging Principles. J. Clin. Med. 2020, 10, 104. [Google Scholar] [CrossRef]
- Mazzaferro, V.; Gorgen, A.; Roayaie, S.; Droz dit Busset, M.; Sapisochin, G. Liver Resection and Transplantation for Intrahepatic Cholangiocarcinoma. J. Hepatol. 2020, 72, 364–377. [Google Scholar] [CrossRef]
- Endo, I.; Gonen, M.; Yopp, A.C.; Dalal, K.M.; Zhou, Q.; Klimstra, D.; D’Angelica, M.; DeMatteo, R.P.; Fong, Y.; Schwartz, L.; et al. Intrahepatic Cholangiocarcinoma: Rising Frequency, Improved Survival, and Determinants of Outcome After Resection. Ann. Surg. 2008, 248, 84–96. [Google Scholar] [CrossRef]
- Machairas, N.; Lang, H.; Jayant, K.; Raptis, D.A.; Sotiropoulos, G.C. Intrahepatic Cholangiocarcinoma: Limitations for Resectability, Current Surgical Concepts and Future Perspectives. Eur. J. Surg. Oncol. 2020, 46, 740–746. [Google Scholar] [CrossRef]
- Tan, J.C.C.; Coburn, N.G.; Baxter, N.N.; Kiss, A.; Law, C.H.L. Surgical Management of Intrahepatic Cholangiocarcinoma-A Population-Based Study. Ann. Surg. Oncol. 2008, 15, 600–608. [Google Scholar] [CrossRef]
- Lurje, G.; Bednarsch, J.; Roderburg, C.; Trautwein, C.; Neumann, U.P. Intrahepatic cholangiocarcinoma-current perspectives and treatment algorithm. Chir. Z. Gebiete Oper. Medizen 2018, 89, 858–864. [Google Scholar] [CrossRef]
- De Jong, M.C.; Nathan, H.; Sotiropoulos, G.C.; Paul, A.; Alexandrescu, S.; Marques, H.; Pulitano, C.; Barroso, E.; Clary, B.M.; Aldrighetti, L.; et al. Intrahepatic Cholangiocarcinoma: An International Multi-Institutional Analysis of Prognostic Factors and Lymph Node Assessment. J. Clin. Oncol. 2011, 29, 3140–3145. [Google Scholar] [CrossRef]
- Doussot, A.; Gonen, M.; Wiggers, J.K.; Groot-Koerkamp, B.; DeMatteo, R.P.; Fuks, D.; Allen, P.J.; Farges, O.; Kingham, T.P.; Regimbeau, J.M.; et al. Recurrence Patterns and Disease-Free Survival after Resection of Intrahepatic Cholangiocarcinoma: Preoperative and Postoperative Prognostic Models. J. Am. Coll. Surg. 2016, 223, 493–505.e2. [Google Scholar] [CrossRef]
- Oldhafer, F.; Ringe, K.I.; Timrott, K.; Kleine, M.; Beetz, O.; Ramackers, W.; Cammann, S.; Klempnauer, J.; Vondran, F.W.R.; Bektas, H. Modified Ante Situm Liver Resection without Use of Cold Perfusion nor Veno-Venous Bypass for Treatment of Hepatic Lesions Infiltrating the Hepatocaval Confluence. Langenbecks Arch. Surg. 2018, 403, 379–386. [Google Scholar] [CrossRef]
- Oldhafer, K.J.; Lang, H.; Malagó, M.; Testa, G.; Broelsch, C.E. Ex situ Resektion und Resektion an der in situ perfundierten Leber–Gibt es noch Indikationen? Der Chirurg 2001, 72, 131–137. [Google Scholar] [CrossRef]
- Lang, H.; Sotiropoulos, G.C.; Sgourakis, G.; Schmitz, K.J.; Paul, A.; Hilgard, P.; Zöpf, T.; Trarbach, T.; Malagó, M.; Baba, H.A.; et al. Operations for Intrahepatic Cholangiocarcinoma: Single-Institution Experience of 158 Patients. J. Am. Coll. Surg. 2009, 208, 218–228. [Google Scholar] [CrossRef]
- Fard-Aghaie, M.H.; Kantas, A.; Makridis, G.; Reese, T.; Wagner, K.C.; Oldhafer, K.J. Critical Appraisal of the Modified Ante Situm Liver Resection: Is the Original Method the Better Choice? Langenbecks Arch. Surg. 2019, 404, 647. [Google Scholar] [CrossRef]
- Machado, M.A.; Bacchella, T.; Makdissi, F.F.; Surjan, R.T.; Machado, M.C. Extended Left Trisectionectomy Severing All Hepatic Veins Preserving Segment 6 and Inferior Right Hepatic Vein. Eur. J. Surg. Oncol. EJSO 2008, 34, 247–251. [Google Scholar] [CrossRef]
- Reames, B.N.; Ejaz, A.; Koerkamp, B.G.; Alexandrescu, S.; Marques, H.P.; Aldrighetti, L.; Maithel, S.K.; Pulitano, C.; Bauer, T.W.; Shen, F.; et al. Impact of Major Vascular Resection on Outcomes and Survival in Patients with Intrahepatic Cholangiocarcinoma: A Multi-Institutional Analysis. J. Surg. Oncol. 2017, 116, 133–139. [Google Scholar] [CrossRef]
- Conci, S.; Viganò, L.; Ercolani, G.; Gonzalez, E.; Ruzzenente, A.; Isa, G.; Salaris, C.; Fontana, A.; Bagante, F.; Pedrazzani, C.; et al. Outcomes of Vascular Resection Associated with Curative Intent Hepatectomy for Intrahepatic Cholangiocarcinoma. Eur. J. Surg. Oncol. J. Eur. Soc. Surg. Oncol. Br. Assoc. Surg. Oncol. 2020, 46, 1727–1733. [Google Scholar] [CrossRef]
- Bartsch, F.; Tripke, V.; Baumgart, J.; Hoppe-Lotichius, M.; Heinrich, S.; Lang, H. Extended Resection of Intrahepatic Cholangiocarcinoma: A Retrospective Single-Center Cohort Study. Int. J. Surg. 2019, 67, 62–69. [Google Scholar] [CrossRef]
- Jonas, S.; Thelen, A.; Benckert, C.; Biskup, W.; Neumann, U.; Rudolph, B.; Lopez-Häänninen, E.; Neuhaus, P. Extended Liver Resection for Intrahepatic Cholangiocarcinoma: A Comparison of the Prognostic Accuracy of the Fifth and Sixth Editions of the TNM Classification. Ann. Surg. 2009, 249, 303–309. [Google Scholar] [CrossRef]
- Zhang, X.-F.; Bagante, F.; Chakedis, J.; Moris, D.; Beal, E.W.; Weiss, M.; Popescu, I.; Marques, H.P.; Aldrighetti, L.; Maithel, S.K.; et al. Perioperative and Long-Term Outcome for Intrahepatic Cholangiocarcinoma: Impact of Major Versus Minor Hepatectomy. J. Gastrointest. Surg. Off. J. Soc. Surg. Aliment. Tract 2017, 21, 1841–1850. [Google Scholar] [CrossRef]
- Nagino, M.; Kamiya, J.; Nishio, H.; Ebata, T.; Arai, T.; Nimura, Y. Two Hundred Forty Consecutive Portal Vein Embolizations Before Extended Hepatectomy for Biliary Cancer: Surgical Outcome and Long-Term Follow-Up. Ann. Surg. 2006, 243, 364–372. [Google Scholar] [CrossRef]
- Li, J.; Moustafa, M.; Linecker, M.; Lurje, G.; Capobianco, I.; Baumgart, J.; Ratti, F.; Rauchfuss, F.; Balci, D.; Fernandes, E.; et al. ALPPS for Locally Advanced Intrahepatic Cholangiocarcinoma: Did Aggressive Surgery Lead to the Oncological Benefit? An International Multi-Center Study. Ann. Surg. Oncol. 2020, 27, 1372–1384. [Google Scholar] [CrossRef]
- Lang, H.; de Santibañes, E.; Schlitt, H.J.; Malagó, M.; van Gulik, T.; Machado, M.A.; Jovine, E.; Heinrich, S.; Ettorre, G.M.; Chan, A.; et al. 10th Anniversary of ALPPS—Lessons Learned and Quo Vadis. Ann. Surg. 2019, 269, 114–119. [Google Scholar] [CrossRef]
- Linecker, M.; Björnsson, B.; Stavrou, G.A.; Oldhafer, K.J.; Lurje, G.; Neumann, U.; Adam, R.; Pruvot, F.-R.; Topp, S.A.; Li, J.; et al. Risk Adjustment in ALPPS Is Associated with a Dramatic Decrease in Early Mortality and Morbidity. Ann. Surg. 2017, 266, 779–786. [Google Scholar] [CrossRef]
- Clavien, P.A.; Barkun, J.; de Oliveira, M.L.; Vauthey, J.N.; Dindo, D.; Schulick, R.D.; de Santibañes, E.; Pekolj, J.; Slankamenac, K.; Bassi, C.; et al. The Clavien-Dindo Classification of Surgical Complications: Five-Year Experience. Ann. Surg. 2009, 250, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Slankamenac, K.; Graf, R.; Barkun, J.; Puhan, M.A.; Clavien, P.-A. The Comprehensive Complication Index: A Novel Continuous Scale to Measure Surgical Morbidity. Ann. Surg. 2013, 258, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Charlson, M.E.; Pompei, P.; Ales, K.L.; MacKenzie, C.R. A New Method of Classifying Prognostic Comorbidity in Longitudinal Studies: Development and Validation. J. Chronic Dis. 1987, 40, 373–383. [Google Scholar] [CrossRef]
- Strasberg, S.M.; Belghiti, J.; Clavien, P.-A.; Gadzijev, E.; Garden, J.O.; Lau, W.-Y.; Makuuchi, M.; Strong, R.W. The Brisbane 2000 Terminology of Liver Anatomy and Resections. HPB 2000, 2, 333–339. [Google Scholar] [CrossRef]
- Rahbari, N.N.; Garden, O.J.; Padbury, R.; Brooke-Smith, M.; Crawford, M.; Adam, R.; Koch, M.; Makuuchi, M.; Dematteo, R.P.; Christophi, C.; et al. Posthepatectomy Liver Failure: A Definition and Grading by the International Study Group of Liver Surgery (ISGLS). Surgery 2011, 149, 713–724. [Google Scholar] [CrossRef]
- Farid, S.G.; White, A.; Khan, N.; Toogood, G.J.; Prasad, K.R.; Lodge, J.P.A. Clinical Outcomes of Left Hepatic Trisectionectomy for Hepatobiliary Malignancy. Br. J. Surg. 2016, 103, 249–256. [Google Scholar] [CrossRef]
- Kron, P.; Kimura, N.; Farid, S.; Lodge, J.P.A. Current Role of Trisectionectomy for Hepatopancreatobiliary Malignancies. Ann. Gastroenterol. Surg. 2019, 3, 606–619. [Google Scholar] [CrossRef]
- Linecker, M.; Stavrou, G.A.; Oldhafer, K.J.; Jenner, R.M.; Seifert, B.; Lurje, G.; Bednarsch, J.; Neumann, U.; Capobianco, I.; Nadalin, S.; et al. The ALPPS Risk Score: Avoiding Futile Use of ALPPS. Ann. Surg. 2016, 264, 763–771. [Google Scholar] [CrossRef]
- Panaro, F.; Giannone, F.; Riviere, B.; Sgarbura, O.; Cusumano, C.; Deshayes, E.; Navarro, F.; Guiu, B.; Quenet, F. Perioperative Impact of Liver Venous Deprivation Compared with Portal Venous Embolization in Patients Undergoing Right Hepatectomy: Preliminary Results from the Pioneer Center. Hepatobiliary Surg. Nutr. 2019, 8, 329–337. [Google Scholar] [CrossRef]
- Kobayashi, K.; Yamaguchi, T.; Denys, A.; Perron, L.; Halkic, N.; Demartines, N.; Melloul, E. Liver Venous Deprivation Compared to Portal Vein Embolization to Induce Hypertrophy of the Future Liver Remnant before Major Hepatectomy: A Single Center Experience. Surgery 2020, 167, 917–923. [Google Scholar] [CrossRef]
- Deshayes, E.; Schadde, E.; Piron, L.; Quenet, F.; Guiu, B. Extended Liver Venous Deprivation Leads to a Higher Increase in Liver Function That ALPPS in Early Assessment: A Comment to Sparrelid, E. et al. Dynamic Evaluation of Liver Volume and Function in Associating Liver Partition and Portal Vein Ligation for Staged Hepatectomy. J. Gastrointest. Surg. Off. J. Soc. Surg. Aliment. Tract 2017, 21, 1754–1755. [Google Scholar] [CrossRef]
- Guiu, B.; Quenet, F.; Escal, L.; Bibeau, F.; Piron, L.; Rouanet, P.; Fabre, J.-M.; Jacquet, E.; Denys, A.; Kotzki, P.-O.; et al. Extended Liver Venous Deprivation before Major Hepatectomy Induces Marked and Very Rapid Increase in Future Liver Remnant Function. Eur. Radiol. 2017, 27, 3343–3352. [Google Scholar] [CrossRef]
- Heil, J.; Korenblik, R.; Heid, F.; Bechstein, W.O.; Bemelmans, M.; Binkert, C.; Björnsson, B.; Breitenstein, S.; Detry, O.; Dili, A.; et al. Preoperative Portal Vein or Portal and Hepatic Vein Embolization: DRAGON Collaborative Group Analysis. Br. J. Surg. 2021. [Google Scholar] [CrossRef]
- Malde, D.J.; Khan, A.; Prasad, K.R.; Toogood, G.J.; Lodge, J.P.A. Inferior Vena Cava Resection with Hepatectomy: Challenging but Justified. HPB 2011, 13, 802–810. [Google Scholar] [CrossRef]
- Heinrich, S.; Baumgart, J.; Mittler, J.; Lang, H. Vascular reconstruction in hepatic surgery. Chir. Z. Geb. Oper. Medizen 2016, 87, 100–107. [Google Scholar] [CrossRef]
- Spolverato, G.; Ejaz, A.; Kim, Y.; Sotiropoulos, G.C.; Pau, A.; Alexandrescu, S.; Marques, H.; Pulitano, C.; Barroso, E.; Clary, B.M.; et al. Tumor Size Predicts Vascular Invasion and Histologic Grade among Patients Undergoing Resection of Intrahepatic Cholangiocarcinoma. J. Gastrointest. Surg. Off. J. Soc. Surg. Aliment. Tract 2014, 18, 1284–1291. [Google Scholar] [CrossRef]
- Spolverato, G.; Kim, Y.; Alexandrescu, S.; Popescu, I.; Marques, H.P.; Aldrighetti, L.; Clark Gamblin, T.; Miura, J.; Maithel, S.K.; Squires, M.H.; et al. Is Hepatic Resection for Large or Multifocal Intrahepatic Cholangiocarcinoma Justified? Results from a Multi-Institutional Collaboration. Ann. Surg. Oncol. 2015, 22, 2218–2225. [Google Scholar] [CrossRef]
| Complex (n = 49) | Conventional (n = 66) | Exploration (n = 28) | |||||
|---|---|---|---|---|---|---|---|
| Females, n (%) | 23 | (47) | 36 | (55) | 11 | (39) | 0.377 |
| Age, median (IQR) | 70 | (60–75) | 66 | (60–73) | 66 | (57–75) | 0.326 |
| BMI, median (IQR) | 25.9 | (22.9–29.5) | 24.8 | (22.5–28.7) | 25.7 | (23.9–29.1) | 0.931 |
| CKD, n (%) | 6 | (12) | 4 | (6) | 4 | (14) | 0.365 |
| CHD, n (%) | 3 | (6) | 3 | (5) | 4 | (14) | 0.228 |
| Hypertension, n (%) | 68 | (53) | 25 | (38) | 13 | (46) | 0.264 |
| Neoadj. Chemo, n (%) | 6 | (12) | 4 | (6) | 4 | (14) | 0.365 |
| Charlson Com. Index, median (IQR) | 5 | (4–6) | 5 | (4–6) | 7 | (4–8) | 0.582 |
| UICC, n (%) | |||||||
| Stage I | 5 | (10) | 15 | (23) | 1 | (4) | 0.031 |
| Stage II | 13 | (27) | 27 | (41) | 4 | (14) | 0.028 |
| Stage III | 6 | (12) | 5 | (8) | 0 | (0) | 0.152 |
| Stage IV | 25 | (51) | 19 | (29) | 23 | (82) | <0.001 |
| Complex (n = 49) | Conventional (n = 66) | Exploration (n = 28) | |||||
|---|---|---|---|---|---|---|---|
| Operation Time, median (IQR) | 346 | (215–429) | 210 | (161–280) | 132 | (93–165) | <0.001 |
| Hepaticojejunostomy, n (%) | 16 | (33) | 6 | (9) | 3 | (11) | 0.003 |
| Intraop. Transfusions, n (%) | 22 | (47) | 7 | (11) | 2 | (7) | <0.001 |
| Blood loss, median (IQR) | 1000 | (0–1500) | 0 | (0–500) | 0 | (0–0) | <0.001 |
| Pringle-Maneuver, n (%) | 10 | (21) | 15 | (23) | 0 | (0) | 0.022 |
| Resection Margin | |||||||
| R0 | 31 | (63) | 57 | (86) | n/a | 0.004 | |
| R1 | 18 | (37) | 9 | (14) | n/a | - | |
| Lymph Node Status | |||||||
| No lymphadenectomy | 0 | (0) | 11 | (17) | n/a | 0.011 | |
| Negative | 32 | (68) | 40 | (61) | n/a | - | |
| Positive | 15 | (32) | 15 | (23) | n/a | - | |
| No. | Age | UICC | Hepatectomy/Bypass | Type of Vascular Reconstruction | OR Time (min) | EKs (n) | Highest Complication /CCI | PHLF /LOS (days) | Resection Margin | Survival Status (Month) |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 29 | IVa | Right Trisectionectomy/Femoral-Axillary Bypass/Portal Perfusion with HTK | Resection IVC and reconstruction with interposition of graft reinsertion of left hepatic vein | 624 | 8 | Grade I /8.7 | No /16 | R0 | Dead (33 Months) |
| 2 | 48 | IVa | Left Trisectionectomy/Femoral-Axillary Bypass/Portal Perfusion with HTK | Resection of IVC with Goretex graft, reconstruction of right posterior and right anterior vein with pericardial interposition portal-vein reconstruction end-to-end | 564 | 0 | Grade II /20.9 | No /27 | R0 | Alive (95 Months) |
| 3 | 60 | I | Left Hemi-hepatectomy + Seg. 1/Femoral-Axillary Bypass/Portal Perfusion with HTK | Resection of IVC with Goretex graft, reinsertion of right hepatic vein | 495 | 5 | Grade VIa /69.8 | No /69 | R0 | Alive (10 Months) |
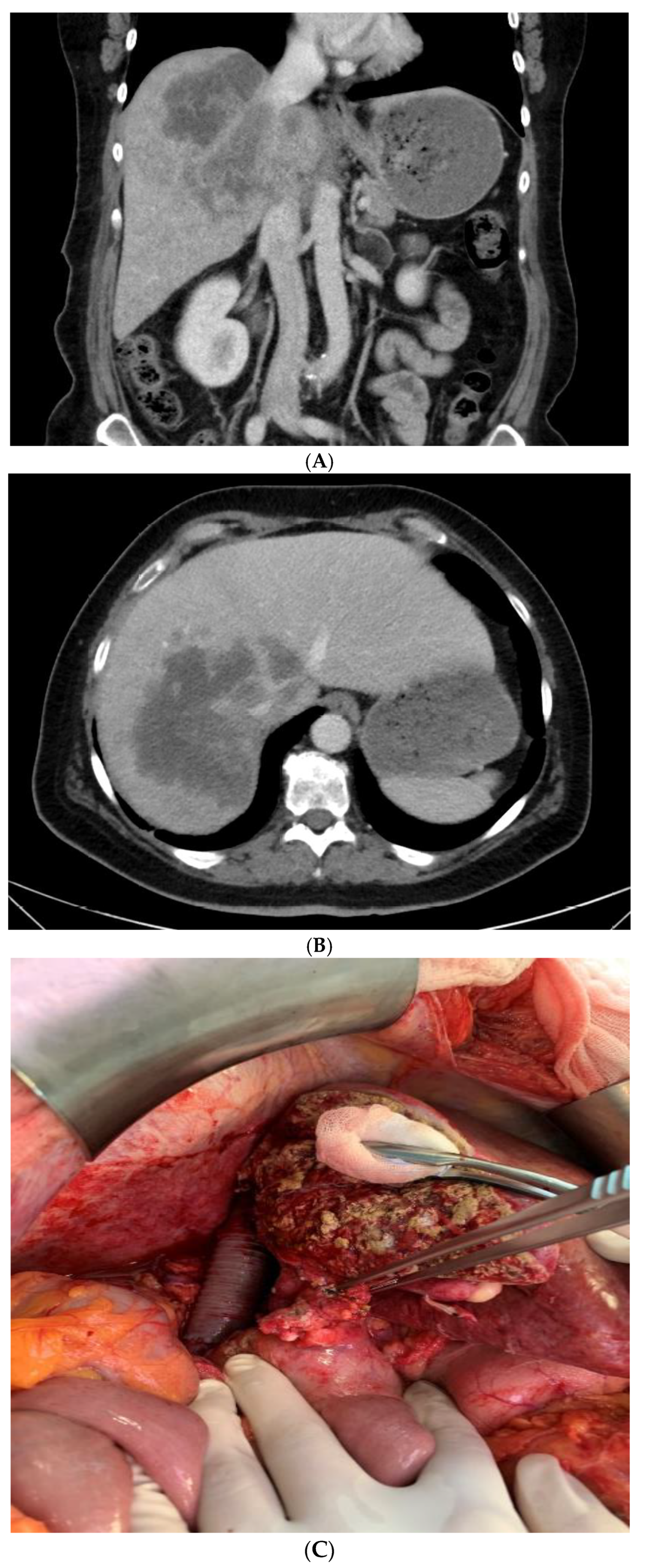
| 4 | 55 | IVa | Right Trisectionectomy/Femoral-Axillary Bypass/Portal Perfusion with HTK (Figure 1) | Resection of IVC with Goretex graft, reinsertion of left hepatic vein, portal-vein reconstruction end-to-end | 470 | 8 | Grade I /8.7 | No /17 | R0 | Alive (8 Months) |
| Complex (n = 49) | Conventional (n = 66) | Exploration (n = 28) | |||||
|---|---|---|---|---|---|---|---|
| Complications, n (%) | |||||||
| None | 7 | (14) | 30 | (46) | 19 | (68) | <0.001 |
| Minor | 20 | (41) | 29 | (44) | 4 | (14) | - |
| Major | 17 | (35) | 7 | (11) | 3 | (11) | - |
| In-hospital death | 5 | (10) | 0 | (0) | 2 | (7) | 0.036 |
| CCI, median (IQR) | 29.6 | (20.9–42.6) | 8.7 | (0–24.2) | 0 | (0–20.9) | <0.001 |
| Bile Leakage, n (%) | 12 | (25) | 8 | (12) | 0 | (0) | 0.010 |
| Bleeding, n (%) | 14 | (29) | 7 | (11) | 3 | (11) | 0.025 |
| Infectious, n (%) | 11 | (22) | 12 | (18) | 0 | (0) | 0.029 |
| PHLF, n (%) | 5 | (10) | 4 | (6) | 2 | (7) | 0.707 |
| Hospital Stay, median (IQR) | 17 | (13–24) | 8 | (7–12) | 8 | (6–11) | <0.001 |
| 90-day Mortality, n (%) | 7 | (14) | 1 | (1) | 7 | (25) | 0.002 |
| Parameter | HR (95% CI) | p Value |
|---|---|---|
| Gender | ||
| Male | Ref | |
| Female | 1.074 (0.618–1.868) | 0.800 |
| Neoadjuvant Chemotherapy | ||
| Yes | Ref | |
| No | 1.307 (0.567–3.011) | 0.530 |
| Resection | ||
| Conventional | Ref | |
| Complex | 1.219 (0.643–2.309) | 0.544 |
| Exploration | 1.873 (0.775–4.525) | 0.163 |
| Resection Margin | ||
| R0 | Ref | |
| No R0 | 2.964 (1.638–5.363) | <0.001 |
| UICC | ||
| Stage I | Ref | |
| Stage II | 4.160 (1.171–14.780) | 0.028 |
| Stage III | 4.335 (1.107–16.986) | 0.035 |
| Stage IV | 5.329 (1.604–17.705) | 0.006 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reese, T.; Pagel, G.; Bause, B.A.; von Rittberg, Y.; Wagner, K.C.; Oldhafer, K.J. Complex Liver Resections for Intrahepatic Cholangiocarcinoma. J. Clin. Med. 2021, 10, 1672. https://doi.org/10.3390/jcm10081672
Reese T, Pagel G, Bause BA, von Rittberg Y, Wagner KC, Oldhafer KJ. Complex Liver Resections for Intrahepatic Cholangiocarcinoma. Journal of Clinical Medicine. 2021; 10(8):1672. https://doi.org/10.3390/jcm10081672
Chicago/Turabian StyleReese, Tim, Gregor Pagel, Bettina A. Bause, York von Rittberg, Kim C. Wagner, and Karl J. Oldhafer. 2021. "Complex Liver Resections for Intrahepatic Cholangiocarcinoma" Journal of Clinical Medicine 10, no. 8: 1672. https://doi.org/10.3390/jcm10081672
APA StyleReese, T., Pagel, G., Bause, B. A., von Rittberg, Y., Wagner, K. C., & Oldhafer, K. J. (2021). Complex Liver Resections for Intrahepatic Cholangiocarcinoma. Journal of Clinical Medicine, 10(8), 1672. https://doi.org/10.3390/jcm10081672







