Musculoskeletal Manifestations in Sjogren’s Syndrome: An Orthopedic Point of View
Abstract
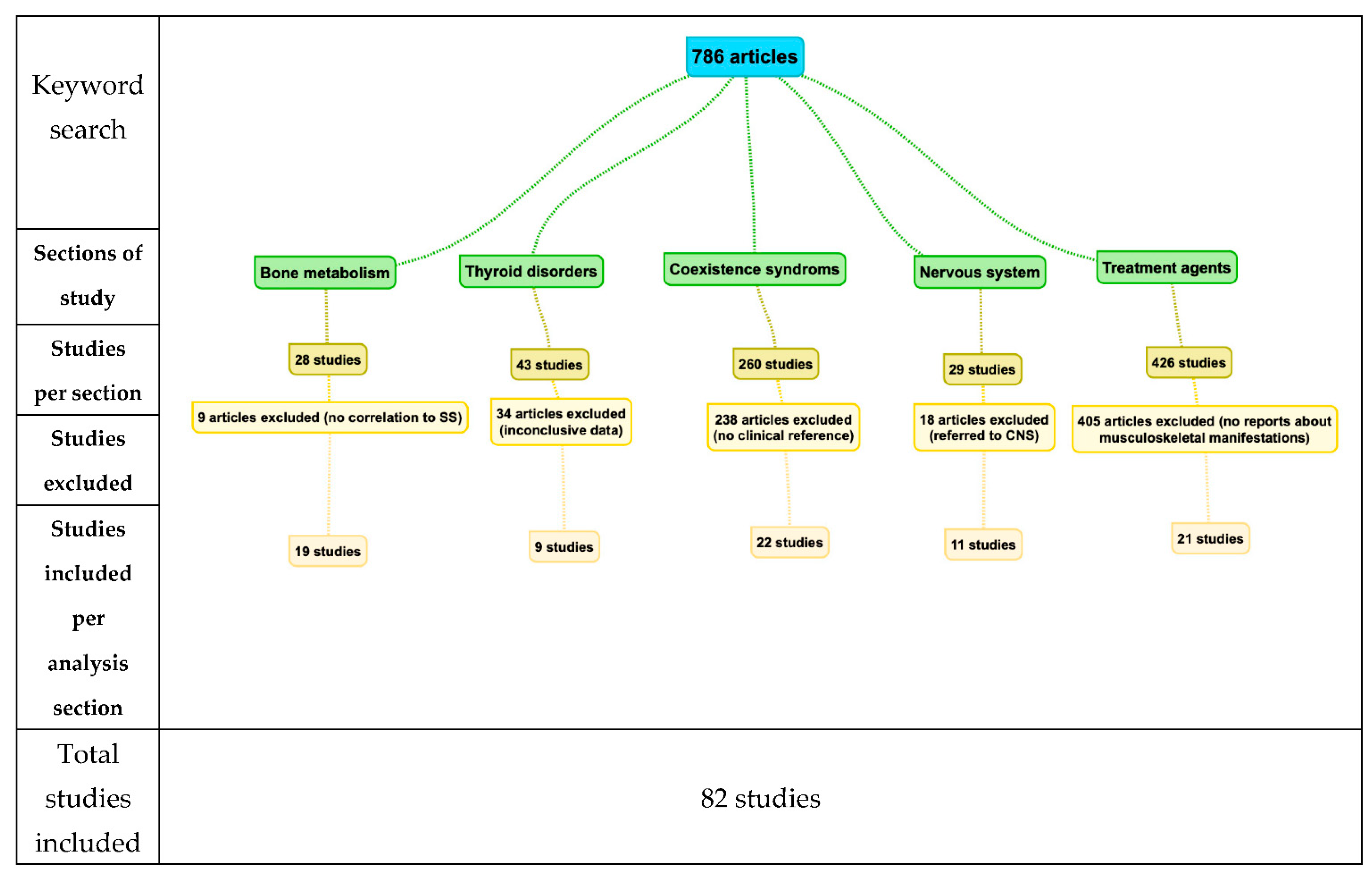
1. Introduction
2. Discussion
2.1. Primary Sjogren’s Syndrome (pSS) and Its Relationship to Bone Metabolic Diseases
2.1.1. pSS and Osteoporosis
2.1.2. pSS and Osteomalacia
2.2. Primary Sjogren’s Syndrome (pSS) and Peripheral Joints
2.3. Sjogren’s Syndrome Associated with Other Diseases
2.4. Treatment-Related Manifestations
2.4.1. Typical Treatment Agents
2.4.2. Second-Line Treatments
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Nair, J.J.; Singh, T.P. Sjogren’s syndrome: Review of the aetiology, Pathophysiology & Potential therapeutic interventions. J. Clin. Exp. Dent. 2017, 9, 584–589. [Google Scholar]
- Mavragani, C.P.; Moutsopoulos, H.M. Sjögren syndrome. CMAJ 2014, 186, 579–586. [Google Scholar] [CrossRef] [PubMed]
- Malladi, A.S.; Sack, K.E.; Shiboski, S.C.; Shiboski, C.H.; Baer, A.N.; Banushree, R.; Dong, Y.; Helin, P.; Kirkham, B.W.; Li, M.; et al. Primary Sjogren’s syndrome as a systemic disease: A study of participants enrolled in an international Sjogren’s syndrome registry. Arthritis Care Res. 2012, 64, 911–918. [Google Scholar] [CrossRef] [PubMed]
- Helmick, C.G.; Felson, D.T.; Lawrence, R.C.; Gabriel, S.; Hirsch, R.; Kwoh, C.K.; Liang, M.H.; Kremers, H.M.; Mayes, M.D.; Merkel, P.A.; et al. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part, I. Arthritis Rheum. 2008, 58, 15–25. [Google Scholar] [CrossRef]
- Soyfoo, M.S.; Chivasso, C.; Perret, J.; Delporte, C. Involvement of Aquaporins in the Pathogenesis, Diagnosis and Treatment of Sjogren’s Syndrome. Int. J. Mol. Sci. 2018, 19, 3392. [Google Scholar] [CrossRef]
- Both, T.; Dalm, V.A.; van Hagen, P.M.; van Daele, P.L. Reviewing primary Sjogren’s syndrome: Beyond the dryness—From pathophysiology to diagnosis and treatment. Int. J. Med. Sci. 2017, 14, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Moutsopoulos, H.M. Sjögren’s syndrome: A forty-year scientific journey. J.Autoimmun. 2014, 51, 1–9. [Google Scholar] [CrossRef]
- Rivièr, E.; Pascaud, J.; Tchitchek, N.; Boudaoud, S.; Paoletti, A.; Ly, B.; Dupré, A.; Chen, H.; Thai, A.; Allaire, N.; et al. Salivary gland epithelial cells from patients with Sjögren’s syndrome induce B-lymphocyte survival and activation. Ann. Rheum. Dis. 2020, 79, 1468–1477. [Google Scholar] [CrossRef]
- Ibanez-Cabellos, J.S.; Seco-Cervera, M.; Osca-Verdegal, R.; Pallardo, F.V.; Garcia-Gimenez, J.L. Epigenetic Regulation in the Pathogenesis of Sjogren Syndrome and Rheumatoid Arthritis. Front Genet. 2019, 10, 1104. [Google Scholar] [CrossRef] [PubMed]
- Marketos, N.; Cinoku, I.; Rapti, A.; Mavragani, C.P. Type I interferon signature in Sjögren’s syndrome: Pathophysiological and clinical implications. Clin. Exp. Rheumatol. 2019, 37, 185–191. [Google Scholar]
- Bodewes, I.L.A.; Björk, A.; Versnel, M.A.; Wahren-Herlenius, M. Innate immunity and interferons in the pathogenesis of Sjögren’s syndrome. Rheumatology 2019, 360. [Google Scholar] [CrossRef]
- Nordmark, G.; Alm, G.V.; Ronnblom, L. Mechanisms of Disease: Primary Sjogren’s syndrome and the type I interferon system. Nat. Clin.Pract. Rheumatol. 2006, 2, 262–269. [Google Scholar] [CrossRef]
- Jonsson, R.; Brokstad, K.A.; Jonsson, M.V.; Delaleu, N.; Skarstein, K. Current concepts on Sjogren’s syndrome—Classification criteria and biomarkers. Eur. J. Oral Sci. 2018, 126, 37–48. [Google Scholar] [CrossRef] [PubMed]
- Fox, R.I. Sjogren’s syndrome. Lancet 2005, 366, 321–331. [Google Scholar] [CrossRef]
- Saraux, A.; Pers, J.O.; Devauchelle-Pensec, V. Treatment of primary Sjogren syndrome. Nat. Rev. Rheumatol. 2016, 12, 456–471. [Google Scholar] [CrossRef]
- Skarlis, C.; Palli, E.; Nezos, A.; Koutsilieris, M.; Mavragani, C.P. Study of the incidence of osteoporosis in patients with Sjogren’s syndrome (pSS) and investigation of activation of the RANKL /RANK and osteoprotegerin (OPG) system. Mediterr. J. Rheumatol. 2018, 29, 224–227. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.M.; Sheen, D.H.; Lee, Y.J.; Lee, E.B.; Song, Y.W. Sjogren’s syndrome presenting as remitting seronegative symmetric synovitis with pitting edema (RS3PE). J. Korean Med. Sci. 2003, 18, 606–608. [Google Scholar] [CrossRef]
- Fernandez-Torres, J.; Perez-Hernandez, N.; Hernandez-Molina, G.; Martinez-Nava, G.A.; Garrido-Rodriguez, D.; Lopez-Reyes, A.; Rodríguez-Pérez, J.M. Risk of Wnt/beta-catenin signalling pathway gene polymorphisms in primary Sjogren’s syndrome. Rheumatology 2020, 59, 418–425. [Google Scholar] [CrossRef]
- Gravani, F.; Papadaki, I.; Antypa, E.; Nezos, A.; Masselou, K.; Ioakeimidis, D.; Koutsilieris, M.; Moutsopoulos, H.M.; Mavragani, C.P. Subclinical atherosclerosis and impaired bone health in patients with primary Sjogren’s syndrome: Prevalence, clinical and laboratory associations. Arthritis Res. Ther. 2015, 17, 99. [Google Scholar] [CrossRef]
- Pasoto, S.G.; Augusto, K.L.; Alvarenga, J.C.; Takayama, L.; Oliveira, R.M.; Bonfa, E.; Pereira, R.M. Cortical bone density and thickness alterations by high-resolution peripheral quantitative computed tomography: Association with vertebral fractures in primary Sjogren’s syndrome. Rheumatology 2016, 55, 2200–2211. [Google Scholar] [CrossRef]
- Salman-Monte, T.C.; Sanchez-Piedra, C.; Fernandez Castro, M.; Andreu, J.L.; Martinez Taboada, V.; Olive, A.; Rosas, J.; Menor, R.; Rodríguez, B.; Aparicio, A.G.; et al. Prevalence and factors associated with osteoporosis and fragility fractures in patients with primary Sjogren syndrome. Rheumatol. Int. 2020, 40, 1259–1265. [Google Scholar] [CrossRef] [PubMed]
- Tiefenbach, M.; Scheel, M.; Maier, A.; Gehlen, M.; Schwarz-Eywill, M.; Werner, M.; Siebers-Renelt, U. Hammer, M.Osteomalacia-Clinical aspects, diagnostics and treatment. Z. Rheumatol. 2018, 77, 703–718. [Google Scholar] [CrossRef] [PubMed]
- Geng, Y.; Zhao, Y.; Zhang, Z. Tubulointerstitial nephritis-induced hypophosphatemicosteomalacia in Sjogren’s syndrome: A case report and review of the literature. Clin.Rheumatol. 2018, 37, 257–263. [Google Scholar] [CrossRef]
- Fulop, M.; Mackay, M. Renal tubular acidosis, Sjogren syndrome, and bone disease. Arch. Intern. Med. 2004, 164, 905–909. [Google Scholar] [CrossRef]
- Gutierrez, J.O.; Zurita, M.F.; Zurita, L.A. Sjogren’s Syndrome Associated with Fanconi’s Syndrome and Osteomalacia. Am. J. Case Rep. 2018, 19, 392–396. [Google Scholar] [CrossRef] [PubMed]
- El Ati, Z.; Fatma, L.B.; Boulahya, G.; Rais, L.; Krid, M.; Smaoui, W.; Maiz, H.B.; Beji, S.; Zouaghi, K.; Moussa, F.B. Osteomalacia complicating renal tubular acidosis in association with Sjogren’s syndrome. Saudi. J. Kidney Dis. Transpl. 2014, 25, 1072–1077. [Google Scholar] [PubMed]
- Shen, G.; Zhang, Y.; Hu, S.; Liu, B.; Kuang, A. Adult-onset hypophosphatemicosteomalacia associated with Sjogren syndrome: Clinical case report. Medicine 2017, 96, e6493. [Google Scholar] [CrossRef] [PubMed]
- Nagae, H.; Noguchi, Y.; Ogata, S.; Ogata, C.; Matsui, R.; Shimomura, Y.; Katafuchi, R. Osteomalacia secondary to renal tubular acidosis due to Sjogren’s syndrome: A case report and review of the literature. CEN Case Rep. 2012, 1, 123–127. [Google Scholar] [CrossRef][Green Version]
- Both, T.; Hoorn, E.J.; Zietse, R.; van Laar, J.A.; Dalm, V.A.; Brkic, Z.; Versnel, M.A.; van Hagen, P.M.; van Daele, P.L. Prevalence of distal renal tubular acidosis in primary Sjogren’s syndrome. Rheumatology 2015, 54, 933–939. [Google Scholar] [CrossRef]
- Pertovaara, M.; Korpela, M.; Kouri, T.; Pasternack, A. The occurrence of renal involvement in primary Sjogren’s syndrome: A study of 78 patients. Rheumatology 1999, 38, 1113–1120. [Google Scholar] [CrossRef]
- Kim, J.H.; Jin, H.Y.; Baek, H.S.; Lee, K.A. HypophosphatemicOsteomalacia with Multiple Bone Fractures: ADV-Induced Fanconi’s Syndrome. Chonnam. Med. J. 2018, 54, 78–79. [Google Scholar] [CrossRef][Green Version]
- Both, T.; Zillikens, M.C.; Hoorn, E.J.; Zietse, R.; van Laar, J.A.; Dalm, V.A.; van Duijn, C.M.; Versnel, M.A.; Maria, N.I.; van Hagen, P.M.; et al. Bone Mineral Density in Sjogren Syndrome Patients with and Without Distal Renal Tubular Acidosis. Calcif. Tissue Int. 2016, 98, 573–579. [Google Scholar] [CrossRef][Green Version]
- Aksoy, A.; Solmaz, D.; Can, G.; Cetin, P.; Balci, A.; Akar, S.; Birlik, M.; Akkoc, N.; Onen, F. Increased Frequency of Hand Osteoarthritis in Patients with Primary Sjogren Syndrome Compared with Systemic Lupus Erythematosus. J. Rheumatol. 2016, 43, 1068–1071. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Casals, M.; Solans, R.; Rosas, J.; Camps, M.T.; Gil, A.; Del Pino-Montes, J.; Calvo-Alen, J.; Jiménez-Alonso, J.; Micó, M.L.; Beltrán, J.; et al. Primary Sjogren syndrome in Spain: Clinical and immunologic expression in 1010 patients. Medicine 2008, 87, 210–219. [Google Scholar] [CrossRef]
- Guedes, L.K.N.; Leon, E.P.; Bocate, T.S.; Bonfigliolli, K.R.; Lourenco, S.V.; Bonfa, E.; Pasoto, S.G. Characterizing hand and wrist ultrasound pattern in primary Sjogren’s syndrome: A case-control study. Clin. Rheumatol. 2020, 39, 1907–1918. [Google Scholar] [CrossRef] [PubMed]
- Jacques, T.; Sudol-Szopinska, I.; Larkman, N.; O’Connor, P.; Cotten, A. Musculoskeletal Manifestations of Non-RA Connective Tissue Diseases: Scleroderma, Systemic Lupus Erythematosus, Still’s Disease, Dermatomyositis/Polymyositis, Sjogren’s Syndrome, and Mixed Connective Tissue Disease. Semin. Musculoskelet. Radiol. 2018, 22, 166–179. [Google Scholar]
- Riente, L.; Scire, C.A.; DelleSedie, A.; Baldini, C.; Filippucci, E.; Meenagh, G.; Iagnocco, A.; Montecucco, C.; Grassi, W.; Guido, V.; et al. Ultrasound imaging for the rheumatologist. XXIII. Sonographic evaluation of hand joint involvement in primary Sjogren’s syndrome. Clin. Exp. Rheumatol. 2009, 27, 747–750. [Google Scholar] [PubMed]
- Mirouse, A.; Seror, R.; Vicaut, E.; Mariette, X.; Dougados, M.; Fauchais, A.L.; Deroux, A.; Dellal, A. Arthritis in primary Sjogren’s syndrome: Characteristics, outcome and treatment from French multicenter retrospective study. Autoimmun. Rev. 2019, 18, 9–14. [Google Scholar] [CrossRef]
- Haga, H.J.; Peen, E. A study of the arthritis pattern in primary Sjogren’s syndrome. Clin. Exp. Rheumatol. 2007, 25, 88–91. [Google Scholar] [PubMed]
- Pease, C.T.; Shattles, W.; Barrett, N.K.; Maini, R.N. The arthropathy of Sjogren’s syndrome. Br. J. Rheumatol. 1993, 32, 609–613. [Google Scholar] [CrossRef] [PubMed]
- Fauchais, A.L.; Ouattara, B.; Gondran, G.; Lalloue, F.; Petit, D.; Ly, K.; Lambert, M.; Launay, D.; Loustaud-Ratti, V.; Bezanahari, H.; et al. Articular manifestations in primary Sjogren’s syndrome: Clinical significance and prognosis of 188 patients. Rheumatology 2010, 49, 1164–1172. [Google Scholar] [CrossRef]
- Payet, J.; Belkhir, R.; Gottenberg, J.; Berge, E.; Desmoulins, F.; Meyer, O.; Mariette, X.; Seror, R. ACPA-positive primary Sjogren’s syndrome: True primary or rheumatoid arthritis-associated Sjogren’s syndrome? RMD Open 2015, 1, e000066. [Google Scholar] [CrossRef]
- Ryu, Y.S.; Park, S.H.; Lee, J.; Kwok, S.K.; Ju, J.H.; Kim, H.Y.; Jeon, C.H. Follow-up of primary Sjogren’s syndrome patients presenting positive anti-cyclic citrullinated peptides antibody. Rheumatol. Int. 2013, 33, 1443–1446. [Google Scholar] [CrossRef]
- Atzeni, F.; Sarzi-Puttini, P.; Lama, N.; Bonacci, E.; Bobbio-Pallavicini, F.; Montecucco, C.; Caporali, R. Anti-cyclic citrullinated peptide antibodies in primary Sjogren syndrome may be associated with non-erosive synovitis. Arthritis Res. Ther. 2008, 10, 51. [Google Scholar] [CrossRef]
- Molano-Gonzalez, N.; Olivares-Martinez, E.; Anaya, J.M.; Hernandez-Molina, G. Anti-citrullinated protein antibodies and arthritis in Sjogren’s syndrome: A systematic review and meta-analysis. Scand. J. Rheumatol. 2019, 48, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Vitali, C.; Del Papa, N. Pain in primary Sjogren’s syndrome. Best Pract. Res. Clin. Rheumatol. 2015, 29, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.A.; Cleveland, J.D. Sjogren’s syndrome is associated with higher rate of non-home discharge after primary hip arthroplasty and higher transfusion rates after primary hip or knee arthroplasty: A U.S. cohort study. BMC Musculoskelet. Disord. 2020, 21, 492. [Google Scholar] [CrossRef] [PubMed]
- Applbaum, E.; Lichtbroun, A. Novel Sjögren’s autoantibodies found in fibromy- algia patients with sicca and/or xerostomia. Autoimmun. Rev. 2019, 18, 199–202. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.; Kim, D.H.; Park, G.; Park, S.; Kim, H.-S. Clinical significance of anti-dense fine speck- led 70 antibody in patients with fibromyalgia. Korean J. Intern. Med. 2019, 34, 426–433. [Google Scholar] [CrossRef] [PubMed]
- Torrente-Segarra, V.; Corominas, H.; Sánchez-Piedra, C.; Fernández-Castro, M.; Andreu, J.L.; Martínez-Taboada, V.M.; Olivé, A.; Rosas, J.; Sánchez-Alonso, F. SJOGRENSER Study Group of the Spanish Society of Rheumatology. Fibromyalgia prevalence and associated factors in primary Sjögren’s syndrome patients in a large cohort from the Spanish Society of Rheumatology registry (SJOGRENSER). Clin. Exp. Rheumatol. 2017, 35, 28–34. [Google Scholar]
- Zhao, G.W.; Huang, L.F.; Li, D.; Zeng, Y. Ankylosing spondylitis coexists with rheumatoid arthritis and Sjogren’s syndrome: A case report with literature review. Clin. Rheumatol. 2020, 168, 1–4. [Google Scholar] [CrossRef]
- Toro-Dominguez, D.; Carmona-Saez, P.; Alarcon-Riquelme, M.E. Shared signatures between rheumatoid arthritis, systemic lupus erythematosus and Sjogren’s syndrome uncovered through gene expression meta-analysis. Arthritis Res. Ther. 2014, 16, 489. [Google Scholar] [CrossRef]
- Ciccacci, C.; Latini, A.; Perricone, C.; Conigliaro, P.; Colafrancesco, S.; Ceccarelli, F.; Priori, R.; Conti, F.; Perricone, R.; Novelli, G.; et al. TNFAIP3 Gene Polymorphisms in Three Common Autoimmune Diseases: Systemic Lupus Erythematosus, Rheumatoid Arthritis, and Primary Sjogren Syndrome-Association with Disease Susceptibility and Clinical Phenotypes in Italian Patients. J. Immunol. Res. 2019, 2019, 6728694. [Google Scholar] [CrossRef] [PubMed]
- Harrold, L.R.; Shan, Y.; Rebello, S.; Kramer, N.; Connolly, S.E.; Alemao, E.; Kelly, S.; Kremer, J.M.; Rosenstein, E.D. Prevalence of Sjogren’s syndrome associated with rheumatoid arthritis in the USA: An observational study from the Corrona registry. Clin. Rheumatol. 2020, 39, 1899–1905. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Bian, S.; Chen, H.; Wang, L.; Zhao, L.; Zhang, X.; Zhao, Y.; Zeng, X.; Zhang, F. Clinical characteristics and risk factors for overlapping rheumatoid arthritis and Sjogren’s syndrome. Sci. Rep. 2018, 8, 6180. [Google Scholar] [CrossRef]
- He, J.; Ding, Y.; Feng, M.; Guo, J.; Sun, X.; Zhao, J.; Yu, D.; Li, Z. Characteristics of Sjogren’s syndrome in rheumatoid arthritis. Rheumatology 2013, 52, 1084–1089. [Google Scholar] [CrossRef]
- Iagnocco, A.; Coari, G.; Palombi, G.; Valesini, G. Knee joint synovitis in Sjogren’s syndrome. Sonographic study. Scand. J. Rheumatol. 2002, 31, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Shabana, K.; Okamoto, N.; Sugita, Y.; Shindo, K.; Murata, T.; Tamai, H.; Fujiwara, K. The findings of musculoskeletal ultrasonography on primary Sjogren’s syndrome patients in childhood with articular manifestations and the impact of anti-cyclic citrullinated peptide antibody. Mod. Rheumatol. 2019, 29, 821–828. [Google Scholar] [CrossRef] [PubMed]
- Tsampoulas, C.G.; Skopouli, F.N.; Sartoris, D.J.; Kaplan, P.; Kursunoglu, S.; Pineda, C.; Resnick, D.; Moutsopoulos, H.M. Hand radiographic changes in patients with primary and secondary Sjogren’s syndrome. Scand. J. Rheumatol. 1986, 15, 333–339. [Google Scholar] [CrossRef]
- Amezcua-Guerra, L.M.; Hofmann, F.; Vargas, A.; Rodriguez-Henriquez, P.; Solano, C.; Hernandez-Diaz, C.; Castillo-Martinez, D.; Ventura-Ríos, L.; Gutiérrez, M.; Pineda, C. Joint involvement in primary Sjogren’s syndrome: An ultrasound “target area approach to arthritis”. Biomed. Res. Int. 2013, 2013, 640265. [Google Scholar] [CrossRef]
- Lopez, P.J.T.; Lopez, C.F.; de Mora, F.N.; Montes, J.A.; Albero, J.S.; Manez, A.N.; Casas, A.G. Osteoporosis in patients with subclinical hypothyroidism treated with thyroid hormone. Clin. Cases Miner. Bone Metab. 2011, 8, 44–48. [Google Scholar]
- Tan, T.L.; Rajeswaran, H.; Haddad, S.; Shahi, A.; Parvizi, J. Increased Risk of Periprosthetic Joint Infections in Patients with Hypothyroidism Undergoing Total Joint Arthroplasty. J. Arthroplasty 2016, 31, 868–871. [Google Scholar] [CrossRef]
- Buller, L.T.; Rosas, S.; Sabeh, K.G.; Roche, M.W.; McLawhorn, A.S.; Barsoum, W.K. Hypothyroidism Increases 90-Day Complications and Costs Following Primary Total Knee Arthroplasty. J. Arthroplasty 2018, 33, 1003–1007. [Google Scholar] [CrossRef]
- Loviselli, A.; Mathieu, A.; Pala, R.; Mariotti, S.; Cau, S.; Marongiu, C.; Mazzoleni, A.P.; Maggio, P.; Martino, E. Development of thyroid disease in patients with primary and secondary Sjogren’s syndrome. J. Endocrinol. Investig. 1988, 11, 653–656. [Google Scholar] [CrossRef] [PubMed]
- Tunc, R.; Gonen, M.S.; Acbay, O.; Hamuryudan, V.; Yazici, H. Autoimmune thyroiditis and anti-thyroid antibodies in primary Sjogren’s syndrome: A case-control study. Ann. Rheum. Dis. 2004, 63, 575–577. [Google Scholar] [CrossRef]
- Baldini, C.; Ferro, F.; Mosca, M.; Fallahi, P.; Antonelli, A. The Association of Sjogren Syndrome and Autoimmune Thyroid Disorders. Front. Endocrinol. 2018, 9, 121. [Google Scholar] [CrossRef] [PubMed]
- Ruggeri, R.M.; Galletti, M.; Mandolfino, M.G.; Aragona, P.; Bartolone, S.; Giorgianni, G.; Alesci, D.; Trimarchi, F.; Benvenga, S. Thyroid hormone autoantibodies in primary Sjogren syndrome and rheumatoid arthritis are more prevalent than in autoimmune thyroid disease, becoming progressively more frequent in these diseases. J. Endocrinol. Investig. 2002, 25, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Lu, L.; Li, Y.; Yang, R.; Shan, L.; Wang, Y. Increased risk of thyroid disease in patients with Sjogren’s syndrome: A systematic review and meta-analysis. PeerJ 2019, 7, e6737. [Google Scholar] [CrossRef] [PubMed]
- Foster, H.; Fay, A.; Kelly, C.; Charles, P.; Walker, D.; Griffiths, I. Thyroid disease and other autoimmune phenomena in a family study of primary Sjogren’s syndrome. Br. J. Rheumatol. 1993, 32, 36–40. [Google Scholar] [CrossRef]
- Anaya, J.M.; Restrepo-Jimenez, P.; Rodriguez, Y.; Rodriguez-Jimenez, M.; Acosta-Ampudia, Y.; Monsalve, D.M.; Pacheco, Y.; Ramírez-Santana, C.; Molano-González, N.; Mantilla, R.D. Sjogren’s Syndrome and Autoimmune Thyroid Disease: Two Sides of the Same Coin. Clin. Rev. Allergy Immunol. 2019, 56, 362–374. [Google Scholar] [CrossRef]
- Page, M.J.; Massy-Westropp, N.; O’Connor, D.; Pitt, V. Splinting for carpal tunnel syndrome. Cochrane Database Syst. Rev. 2012, 7, e010003. [Google Scholar] [CrossRef] [PubMed]
- Laymon, M.; Petrofsky, J.; McKivigan, J.; Lee, H.; Yim, J. Effect of heat, cold, and pressure on the transverse carpal ligament and median nerve: A pilot study. Med. Sci. Monit. 2015, 21, 446–451. [Google Scholar] [PubMed]
- Fauchais, A.L.; Magy, L.; Vidal, E. Central and peripheral neurological complications of primary Sjogren’s syndrome. Presse Med. 2012, 41, 485–493. [Google Scholar] [CrossRef]
- Pavlakis, P.P.; Alexopoulos, H.; Kosmidis, M.L.; Stamboulis, E.; Routsias, J.G.; Tzartos, S.J.; Tzioufas, A.G.; Moutsopoulos, H.M.; Dalakas, M.C. Peripheral neuropathies in Sjogren syndrome: A new reappraisal. J. Neurol. Neurosurg. Psychiatry 2011, 82, 798–802. [Google Scholar] [CrossRef]
- Goransson, L.G.; Herigstad, A.; Tjensvoll, A.B.; Harboe, E.; Mellgren, S.I.; Omdal, R. Peripheral neuropathy in primary sjogren syndrome: A population-based study. Arch. Neurol. 2006, 63, 1612–1615. [Google Scholar] [CrossRef]
- Gemignani, F.; Marbini, A.; Pavesi, G.; Di Vittorio, S.; Manganelli, P.; Cenacchi, G.; Mancia, D. Peripheral neuropathy associated with primary Sjogren’s syndrome. J. Neurol.Neurosurg. Psychiatry 1994, 57, 983–986. [Google Scholar] [CrossRef] [PubMed]
- Delalande, S.; de Seze, J.; Fauchais, A.L.; Hachulla, E.; Stojkovic, T.; Ferriby, D.; Dubucquoi, S.; Pruvo, J.P.; Vermersch, P.; Hatron, P.Y. Neurologic manifestations in primary Sjogren syndrome: A study of 82 patients. Medicine 2004, 83, 280–291. [Google Scholar] [CrossRef]
- Tobon, G.J.; Pers, J.O.; Devauchelle-Pensec, V.; Youinou, P. Neurological Disorders in Primary Sjogren’s Syndrome. Autoimmune Dis. 2012, 2012, 645967. [Google Scholar] [CrossRef]
- Carvajal Alegria, G.; Guellec, D.; Mariette, X.; Gottenberg, J.E.; Dernis, E.; Dubost, J.J.; Trouvin, A.P.; Hachulla, E.; Larroche, C.; Le Guern, V.; et al. Epidemiology of neurological manifestations in Sjogren’s syndrome: Data from the French ASSESS Cohort. RMD Open 2016, 2, e000179. [Google Scholar] [CrossRef]
- Mochizuki, H.; Kamakura, K.; Masaki, T.; Hirata, A.; Nakamura, R.; Motoyoshi, K. Motor dominant neuropathy in Sjogren’s syndrome: Report of two cases. Intern. Med. 2002, 41, 142–146. [Google Scholar] [CrossRef] [PubMed]
- Pereira, P.R.; Viala, K.; Maisonobe, T.; Haroche, J.; Mathian, A.; Hie, M.; Amoura, Z.; Aubart, F.C. Sjogren Sensory Neuronopathy (Sjogren Ganglionopathy): Long-Term Outcome and Treatment Response in a Series of 13 Cases. Medicine 2016, 95, e3632. [Google Scholar] [CrossRef] [PubMed]
- Perzynska-Mazan, J.; Maslinska, M.; Gasik, R. Neurological manifestations of primary Sjogren’s syndrome. Reumatologia 2018, 56, 99–105. [Google Scholar] [CrossRef]
- Qin, B.; Wang, J.; Liang, Y.; Yang, Z.; Zhong, R. The association between TNF-alpha, IL-10 gene polymorphisms and primary Sjogren’s syndrome: A meta-analysis and systemic review. PLoS ONE 2013, 8, e63401. [Google Scholar]
- Fox, R.I.; Fox, C.M.; Gottenberg, J.E.; Dorner, T. Treatment of Sjogren’s syndrome: Current therapy and future directions. Rheumatology 2019, 142. [Google Scholar] [CrossRef]
- Vivino, F.B.; Carsons, S.E.; Foulks, G.; Daniels, T.E.; Parke, A.; Brennan, M.T.; Forstot, S.L.; Scofield, R.H.; Hammitt, K.M. New Treatment Guidelines for Sjogren’s Disease. Rheum. Dis. Clin. N. Am. 2016, 42, 531–551. [Google Scholar] [CrossRef] [PubMed]
- Stefanski, A.L.; Tomiak, C.; Pleyer, U.; Dietrich, T.; Burmester, G.R.; Dorner, T. The Diagnosis and Treatment of Sjogren’s Syndrome. Dtsch.Arztebl. Int. 2017, 114, 354–361. [Google Scholar] [CrossRef]
- Mercuro, N.J.; Yen, C.F.; Shim, D.J. Risk of QT Interval Prolongation Associated With Use of Hydroxychloroquine With or Without Concomitant Azithromycin Among Hospitalized Patients Testing Positive for Coronavirus Disease 2019 (COVID-19). JAMA Cardiol. 2020, 5, 1036–1041. [Google Scholar] [CrossRef] [PubMed]
- Hussein, H.; Brown, R. Hydroxychloroquine and the treatment of Sjogren syndrome, chronic ulcerative stomatitis, and oral lichen planus in the age of COVID-19. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2021, 131, 9–13. [Google Scholar] [CrossRef]
- Garcia-Bustinduy, M.; Gantes, M.A. Corticosteroids and osteoporosis. Actas Dermo-Sifiliogr. 2007, 98, 526–530. [Google Scholar] [CrossRef]
- Carsons, S.E.; Vivino, F.B.; Parke, A.; Carteron, N.; Sankar, V.; Brasington, R.; Brennan, M.T.; Ehlers, W.; Fox, R.; Scofield, H.; et al. Treatment Guidelines for Rheumatologic Manifestations of Sjogren’s Syndrome: Use of Biologic Agents, Management of Fatigue, and Inflammatory Musculoskeletal. Pain Arthritis Care Res. 2017, 69, 517–527. [Google Scholar] [CrossRef]
- Fox, P.C.; Datiles, M.; Atkinson, J.C.; Macynski, A.A.; Scott, J.; Fletcher, D.; Valdez, I.H.; Kurrasch, R.H.; Delapenha, R.; Jackson, W. Prednisone and piroxicam for treatment of primary Sjogren’s syndrome. Clin. Exp. Rheumatol. 1993, 11, 149–156. [Google Scholar]
- Ramos-Casals, M.; Tzioufas, A.G.; Stone, J.H.; Siso, A.; Bosch, X. Treatment of primary Sjogren syndrome: A systematic review. JAMA 2010, 304, 452–460. [Google Scholar] [CrossRef]
- Liu, Y.; Cui, Y.; Chen, Y.; Gao, X.; Su, Y.; Cui, L. Effects of dexamethasone, celecoxib, and methotrexate on the histology and metabolism of bone tissue in healthy Sprague Dawley rats. Clin. Interv. Aging 2015, 10, 1245–1253. [Google Scholar] [CrossRef] [PubMed]
- Pountos, I.; Giannoudis, P.V. Effect of methotrexate on bone and wound healing. Expert Opin Drug Saf. 2017, 16, 535–545. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.M.; Shandala, T.; Soo, P.P.; Su, Y.W.; King, T.J.; Chen, K.M.; Howe, P.R.; Xian, C.J. Effects of Resveratrol Supplementation on Methotrexate Chemotherapy-Induced Bone Loss. Nutrients 2017, 9, 255. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, Y.; Ueyama, S.; Arai, Y.; Yoshida, Y.; Kaneda, T.; Sato, T.; Shin, K.; Kumegawa, M.; Hakeda, Y. The active metabolite of leflunomide, A771726, inhibits both the generation of and the bone-resorbing activity of osteoclasts by acting directly on cells of the osteoclast lineage. J. Bone Miner. Metab. 2004, 22, 318–328. [Google Scholar] [CrossRef] [PubMed]
- Verstappen, G.M.; van Nimwegen, J.F.; Vissink, A.; Kroese, F.G.M.; Bootsma, H. The value of rituximab treatment in primary Sjogren’s syndrome. Clin. Immunol. 2017, 182, 62–71. [Google Scholar] [CrossRef]
- Gottenberg, J.E.; Cinquetti, G.; Larroche, C.; Combe, B.; Hachulla, E.; Meyer, O.; Meyer, O.; Pertuiset, E.; Kaplanski, G.; Chiche, L.; et al. Efficacy of rituximab in systemic manifestations of primary Sjogren’s syndrome: Results in 78 patients of the AutoImmune and Rituximab registry. Ann. Rheum. Dis. 2013, 72, 1026–1031. [Google Scholar] [CrossRef] [PubMed]
- Tony, H.P.; Burmester, G.; Schulze-Koops, H.; Grunke, M.; Henes, J.; Kotter, I.; Kötter, I.; Haas, J.; Unger, L.; Lovric, S.; et al. Safety and clinical outcomes of rituximab therapy in patients with different autoimmune diseases: Experience from a national registry (GRAID). Arthritis Res. Ther. 2011, 13, 75. [Google Scholar] [CrossRef]
- Shi, Y.; Wu, Y.; Ren, Y.; Jiang, Y.; Chen, Y. Infection risks of rituximab versus non-rituximab treatment for rheumatoid arthritis: A systematic review and meta-analysis. Int. J. Rheum. Dis. 2019, 22, 1361–1370. [Google Scholar] [CrossRef]
- Tsuboi, H.; Matsumoto, I.; Hagiwara, S.; Hirota, T.; Takahashi, H.; Ebe, H.; Yokosawa, M.; Yagishita, M.; Takahashi, H.; Kurata, I.; et al. Effectiveness of abatacept for patients with Sjogren’s syndrome associated with rheumatoid arthritis. An open label, multicenter, one-year, prospective study: ROSE (Rheumatoid Arthritis with Orencia Trial toward Sjogren’s syndrome Endocrinopathy) trial. Mod. Rheumatol. 2016, 26, 891–899. [Google Scholar] [CrossRef] [PubMed]
- Machado, A.C.; Dos Santos, L.C.; Fidelix, T.; Lekwitch, I.; Soares, S.B.; Gasparini, A.F.; Augusto, J.V.; Junior, N.C.; Trevisani, V.F.M. Effectiveness and safety of abatacept for the treatment of patients with primary Sjogren’s syndrome. Clin. Rheumatol. 2020, 39, 243–248. [Google Scholar] [CrossRef] [PubMed]
- Tada, M.; Inui, K.; Sugioka, Y.; Mamoto, K.; Okano, T.; Koike, T. Abatacept might increase bone mineral density at femoral neck for patients with rheumatoid arthritis in clinical practice: AIRTIGHT study. Rheumatol. Int. 2018, 38, 777–784. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rozis, M.; Vlamis, J.; Vasiliadis, E.; Mavragani, C.; Pneumaticos, S.; Evangelopoulos, D.S. Musculoskeletal Manifestations in Sjogren’s Syndrome: An Orthopedic Point of View. J. Clin. Med. 2021, 10, 1574. https://doi.org/10.3390/jcm10081574
Rozis M, Vlamis J, Vasiliadis E, Mavragani C, Pneumaticos S, Evangelopoulos DS. Musculoskeletal Manifestations in Sjogren’s Syndrome: An Orthopedic Point of View. Journal of Clinical Medicine. 2021; 10(8):1574. https://doi.org/10.3390/jcm10081574
Chicago/Turabian StyleRozis, Meletios, John Vlamis, Elias Vasiliadis, Clio Mavragani, Spiros Pneumaticos, and Dimitrios Stergios Evangelopoulos. 2021. "Musculoskeletal Manifestations in Sjogren’s Syndrome: An Orthopedic Point of View" Journal of Clinical Medicine 10, no. 8: 1574. https://doi.org/10.3390/jcm10081574
APA StyleRozis, M., Vlamis, J., Vasiliadis, E., Mavragani, C., Pneumaticos, S., & Evangelopoulos, D. S. (2021). Musculoskeletal Manifestations in Sjogren’s Syndrome: An Orthopedic Point of View. Journal of Clinical Medicine, 10(8), 1574. https://doi.org/10.3390/jcm10081574






