Lipodystrophy as a Late Effect after Stem Cell Transplantation
Abstract
1. Introduction
2. Clinical Cases
3. Common Disease Pattern in HSCT-Associated LD
3.1. Body Fat Distribution
3.2. Metabolic Alterations
3.3. Endocrinopathies
4. Treatment Strategies for Metabolic Derangements in HSCT-Associated LD
5. Potential Risk Factors for the Development of LD after HSCT
5.1. Total Body Irradiation
5.2. Graft-versus-Host Disease
5.3. Glucocorticoid Treatment
5.4. Transplantation
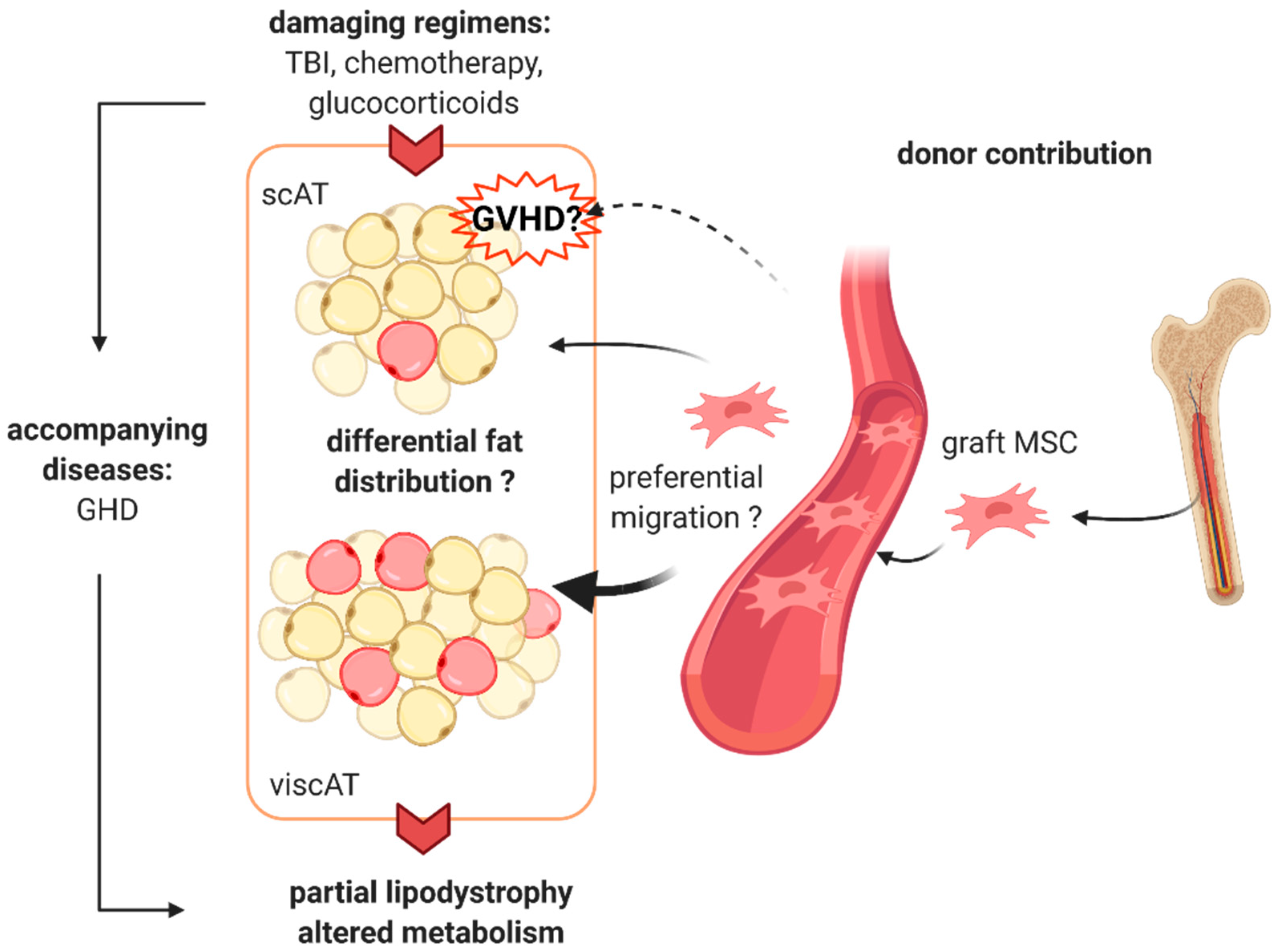
6. Potential Mechanism
Author Contributions
Funding
Conflicts of Interest
References
- Rose, S.R.; Horne, V.E.; Howell, J.; Lawson, S.A.; Rutter, M.M.; Trotman, G.E.; Corathers, S.D. Late endocrine effects of childhood cancer. Nat. Rev. Endocrinol. 2016, 12, 319–336. [Google Scholar] [CrossRef]
- Nandagopal, R.; Laverdière, C.; Mulrooney, D.; Hudson, M.M.; Meacham, L. Endocrine late effects of childhood cancer therapy: A report from the children’s oncology group. Horm. Res. 2008, 69, 65–74. [Google Scholar] [CrossRef]
- Gunasekaran, U.; Agarwal, N.; Jagasia, M.H.; Jagasia, S.M. Endocrine complications in long-term survivors after allogeneic stem cell transplant. Semin. Hematol. 2012, 49, 66–72. [Google Scholar] [CrossRef]
- Orio, F.; Muscogiuri, G.; Palomba, S.; Serio, B.; Sessa, M.; Giudice, V.; Ferrara, I.; Tauchmanovà, L.; Colao, A.; Selleri, C. Endocrinopathies after allogeneic and autologous transplantation of hematopoietic stem cells. Sci. World J. 2014, 2014. [Google Scholar] [CrossRef]
- Taskinen, M.; Saarinen-Pihkala, U.M.; Hovi, L.; Lipsanen-Nyman, M. Impaired glucose tolerance and dyslipidaemia as late effects after bone-marrow transplantation in childhood. Lancet 2000, 356, 993–997. [Google Scholar] [CrossRef]
- Tomita, Y.; Ishiguro, H.; Yasuda, Y.; Hyodo, H.; Koike, T.; Shimizu, T.; Morimoto, T.; Hattori, K.; Matsumoto, M.; Inoue, H.; et al. High incidence of fatty liver and insulin resistance in long-term adult survivors of childhood SCT. Bone Marrow Transplant. 2011, 46, 416–425. [Google Scholar] [CrossRef][Green Version]
- Mayson, S.; Parker, V.; Schutta, M.; Semple, R.; Rickels, M. Severe Insulin Resistance and Hypertriglyceridemia after Childhood Total Body Irradiation. Endocr. Pract. 2013, 19, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Baker, K.S.; Ness, K.K.; Steinberger, J.; Carter, A.; Francisco, L.; Burns, L.J.; Sklar, C.; Forman, S.; Weisdorf, D.; Gurney, J.G.; et al. Diabetes, hypertension, and cardiovascular events in survivors of hematopoietic cell transplantation: A report from the bone marrow transplantation survivor study. Blood 2007, 109, 1765–1772. [Google Scholar] [CrossRef] [PubMed]
- Rajendran, R.; Abu, E.; Fadl, A.; Byrne, C.D. Late effects of childhood cancer treatment: Severe hypertriglyceridaemia, central obesity, non alcoholic fatty liver disease and diabetes as complications of childhood total body irradiation. Diabet. Med. 2013, 30, 239–242. [Google Scholar] [CrossRef] [PubMed]
- Meacham, L.R.; Sklar, C.A.; Li, S.; Liu, Q.; Gimpel, N.; Yasui, Y.; Whitton, J.A.; Stovall, M.; Robison, L.L.; Oeffinger, K.C. Diabetes mellitus in long-term survivors of childhood cancer - Increased risk associated with radiation therapy: A report for the childhood cancer survivor study. Arch. Intern. Med. 2009, 169, 1381–1388. [Google Scholar] [CrossRef]
- Neville, K.A.; Cohn, R.J.; Steinbeck, K.S.; Johnston, K.; Walker, J.L. Hyperinsulinemia, impaired glucose tolerance, and diabetes mellitus in survivors of childhood cancer: Prevalence and risk factors. J. Clin. Endocrinol. Metab. 2006, 91, 4401–4407. [Google Scholar] [CrossRef]
- Kawarasaki, S.; Kuwata, H.; Sawazaki, H.; Sakamoto, T.; Nitta, T.; Kim, C.; Jheng, H.; Takahashi, H.; Nomura, W.; Ara, T.; et al. A new mouse model for noninvasive fluorescence-based monitoring of mitochondrial UCP 1 expression. FEBS Lett. 2019, 593, 1201–1212. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.; Thyagiarajan, M.S.; Hunt, L.P.; Shield, J.P.H.; Stevens, M.C.G.; Crowne, E.C. Reduced insulin sensitivity in childhood survivors of haematopoietic stem cell transplantation is associated with lipodystropic and sarcopenic phenotypes. Pediatr. Blood Cancer 2015, 62, 1992–1999. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.J.; Araujo-Vilar, D.; Cheung, P.T.; Dunger, D.; Garg, A.; Jack, M.; Mungai, L.; Oral, E.A.; Patni, N.; Rother, K.I.; et al. The Diagnosis and Management of Lipodystrophy Syndromes: A Multi-Society Practice Guideline. J. Clin. Endocrinol. Metab. 2016, 101, 4500–4511. [Google Scholar] [CrossRef]
- Rubio-Cabezas, O.; Puri, V.; Murano, I.; Saudek, V.; Semple, R.K.; Dash, S.; Hyden, C.S.S.; Bottomley, W.; Vigouroux, C.; Magré, J.; et al. Partial lipodystrophy and insulin resistant diabetes in a patient with a homozygous nonsense mutation in CIDEC. EMBO Mol. Med. 2009, 1, 280–287. [Google Scholar] [CrossRef]
- Von Schnurbein, J.; Adams, C.; Akinci, B.; Ceccarini, G.; D’Apice, M.R.; Gambineri, A.; Hennekam, R.C.M.; Jeru, I.; Lattanzi, G.; Miehle, K.; et al. European lipodystrophy registry: Background and structure. Orphanet J. Rare Dis. 2020, 15, 1–11. [Google Scholar] [CrossRef]
- Ajluni, N.; Meral, R.; Neidert, A.H.; Brady, G.F.; Buras, E.; McKenna, B.; DiPaola, F.; Chenevert, T.L.; Horowitz, J.F.; Buggs-Saxton, C.; et al. Spectrum of disease associated with partial lipodystrophy: Lessons from a trial cohort. Clin. Endocrinol. (Oxf). 2017, 86, 698–707. [Google Scholar] [CrossRef] [PubMed]
- Adachi, M.; Asakura, Y.; Muroya, K.; Goto, H.; Kigasawa, H. Abnormal adipose tissue distribution with unfavorable metabolic profile in five children following hematopoietic stem cell transplantation: A new etiology for acquired partial lipodystrophy. Clin. Pediatr. Endocrinol. 2013, 22, 53–64. [Google Scholar] [CrossRef] [PubMed]
- Adachi, M.; Oto, Y.; Muroya, K.; Hanakawa, J.; Asakura, Y.; Goto, H. Partial lipodystrophy in patients who have undergone hematopoietic stem cell transplantation during childhood: An institutional cross-sectional survey. Clin. Pediatr. Endocrinol. 2017, 26, 99–108. [Google Scholar] [CrossRef]
- Ceccarini, G.; Ferrari, F.; Santini, F. Acquired partial lipodystrophy after bone marrow transplant during childhood: A novel syndrome to be added to the disease classification list. J. Endocrinol. Invest. 2017, 40, 1273–1274. [Google Scholar] [CrossRef] [PubMed]
- Rooney, D.P.; Ryan, M.F. Diabetes with partial lipodystrophy following sclerodermatous chronic graft vs. host disease. Diabet. Med. 2006, 23, 436–440. [Google Scholar] [CrossRef] [PubMed]
- Kimura, L.; Alvarez, G.; Li, N.; Pawlikowska-Haddal, A.; Moore, T.B.; Casillas, J.; Lee, K.W. Temporary resolution of insulin requirement in acquired partial lipodystrophy associated with chronic graft-versus-host disease. Pediatr. Blood Cancer 2017, 64, 24–26. [Google Scholar] [CrossRef] [PubMed]
- Hosokawa, M.; Shibata, H.; Hosokawa, T.; Irie, J.; Ito, H.; Hasegawa, T. Acquired partial lipodystrophy with metabolic disease in children following hematopoietic stem cell transplantation: A report of two cases and a review of the literature. J. Pediatr. Endocrinol. Metab. 2019, 32, 537–541. [Google Scholar] [CrossRef] [PubMed]
- Shibata, Y.; Nakatsuka, A.; Eguchi, J.; Miyamoto, S.; Masuda, Y.; Awazawa, M.; Takaki, A.; Yoshida, R.; Yagi, T.; Wada, J. Acquired partial lipoatrophy as graft-versus-host disease and treatment with metreleptin: Two case reports. J. Med. Case Rep. 2018, 12, 10–14. [Google Scholar] [CrossRef]
- Adachi, M.; Muroya, K.; Hanakawa, J.; Asakura, Y. Metreleptin worked in a diabetic woman with a history of hematopoietic stem cell transplantation (HSCT) during infancy: Further support for the concept of ‘HSCT-associated lipodystrophy’. Endocr. J. 2020. [Google Scholar] [CrossRef]
- Lorenc, A.; Hamilton-Shield, J.; Perry, R.; Stevens, M.; Wootton, S.; Feelisch, M.; Dragsted, L.O.; Dirks, M.; Shoaie, S.; Mardinoglu, A.; et al. Body composition after allogeneic haematopoietic cell transplantation/total body irradiation in children and young people: A restricted systematic review. J. Cancer Surviv. 2020, 14, 624–642. [Google Scholar] [CrossRef]
- Haque, W.A.; Shimomura, I.; Matsuzawa, Y.; Garg, A. Serum Adiponectin and Leptin Levels in Patients with Lipodystrophies. J. Clin. Endocrinol. Metab. 2002, 87, 2395. [Google Scholar] [CrossRef]
- Wong, S.P.Y.; Huda, M.; English, P.; Bargiotta, A.; Wilding, J.P.H.; Johnson, A.; Corrall, R.; Pinkney, J.H. Adipokines and the insulin resistance syndrome in familial partial lipodystrophy caused by a mutation in lamin A/C. Diabetologia 2005, 48, 2641–2649. [Google Scholar] [CrossRef][Green Version]
- Gebauer, J.; Higham, C.; Langer, T.; Denzer, C.; Brabant, G. Long-Term Endocrine and Metabolic Consequences of Cancer Treatment: A Systematic Review. Endocr. Rev. 2018, 40, 711–767. [Google Scholar] [CrossRef]
- Johansson, J.O.; Fowelin, J.; Landin, K.; Lager, I.; Bengtsson, B.Å. Growth hormone-deficient adults are insulin-resistant. Metabolism 1995, 44, 1126–1129. [Google Scholar] [CrossRef]
- Shankar, S.M.; Marina, N.; Hudson, M.M.; Hodgson, D.C.; Adams, M.J.; Landier, W.; Bhatia, S.; Meeske, K.; Ming, H.C.; Kinahan, K.E.; et al. Monitoring for cardiovascular disease in survivors of childhood cancer: Report from the cardiovascular disease task force of the children’s oncology group. Pediatrics 2008, 121, e387–e396. [Google Scholar] [CrossRef] [PubMed]
- Bovelli, D.; Plataniotis, G.; Roila, F. Cardiotoxicity of chemotherapeutic agents and radiotherapy-related heart disease: ESMO clinical practice guidelines. Ann. Oncol. 2010, 21, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Gan, H.-W.; Spoudeas, H.A. Long-term follow-up of survivors of childhood cancer (SIGN Clinical Guideline 132). Arch. Dis. Child. - Educ. Pract. Ed. 2014, 99, 138–143. [Google Scholar] [CrossRef]
- DeFilipp, Z.; Duarte, R.F.; Snowden, J.A.; Majhail, N.S.; Greenfield, D.M.; Miranda, J.L.; Arat, M.; Baker, K.S.; Burns, L.J.; Duncan, C.N.; et al. Metabolic syndrome and cardiovascular disease following hematopoietic cell transplantation: Screening and preventive practice recommendations from CIBMTR and EBMT. Bone Marrow Transplant. 2017, 52, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Chow, E.J.; Anderson, L.; Baker, K.S.; Bhatia, S.; Guilcher, G.M.T.; Huang, J.T.; Pelletier, W.; Perkins, J.L.; Rivard, L.S.; Schechter, T.; et al. Late Effects Surveillance Recommendations among Survivors of Childhood Hematopoietic Cell Transplantation: A Children’s Oncology Group Report. Biol. Blood Marrow Transplant. 2016, 22, 782–795. [Google Scholar] [CrossRef]
- Davis, N.L.; Tolfrey, K.; Jenney, M.; Elson, R.; Stewart, C.; Moss, A.D.; Cornish, J.M.; Stevens, M.C.G.; Crowne, E.C. Combined resistance and aerobic exercise intervention improves fitness, insulin resistance and quality of life in survivors of childhood haemopoietic stem cell transplantation with total body irradiation. Pediatr. Blood Cancer 2020, 67, 1–11. [Google Scholar] [CrossRef]
- Chou, K.; Perry, C.M. Metreleptin: First global approval. Drugs 2013, 73, 989–997. [Google Scholar] [CrossRef]
- Myalepta | European Medicines Agency. Available online: https://www.ema.europa.eu/en/medicines/human/EPAR/myalepta (accessed on 1 February 2021).
- NICE Metreleptin for treating lipodystrophy. Available online: https://www.nice.org.uk/guidance/hst14 (accessed on 1 February 2021).
- Crisan, M.; Yap, S.; Casteilla, L.; Chen, C.W.; Corselli, M.; Park, T.S.; Andriolo, G.; Sun, B.; Zheng, B.; Zhang, L.; et al. A Perivascular Origin for Mesenchymal Stem Cells in Multiple Human Organs. Cell Stem Cell 2008, 3, 301–313. [Google Scholar] [CrossRef]
- Spalding, K.L.; Arner, E.; Westermark, P.O.; Bernard, S.; Buchholz, B.A.; Bergmann, O.; Blomqvist, L.; Hoffstedt, J.; Näslund, E.; Britton, T.; et al. Dynamics of fat cell turnover in humans. Nature 2008, 453, 783–787. [Google Scholar] [CrossRef]
- Shreder, K.; Rapp, F.; Tsoukala, I.; Rzeznik, V.; Wabitsch, M.; Fischer-Posovszky, P.; Fournier, C. Impact of x-ray exposure on the proliferation and differentiation of human pre-adipocytes. Int. J. Mol. Sci. 2018, 19, 2717. [Google Scholar] [CrossRef] [PubMed]
- Poglio, S.; Galvani, S.; Bour, S.; André, M.; Prunet-Marcassus, B.; Pénicaud, L.; Casteilla, L.; Cousin, B. Adipose tissue sensitivity to radiation exposure. Am. J. Pathol. 2009, 174, 44–53. [Google Scholar] [CrossRef] [PubMed]
- Ablamunits, V.; Weisberg, S.P.; Lemieux, J.E.; Combs, T.P.; Klebanov, S. Reduced adiposity in ob/ob mice following total body irradiation and bone marrow transplantation. Obesity 2007, 15, 1419–1429. [Google Scholar] [CrossRef] [PubMed]
- Zeiser, R.; Blazar, B.R. Acute Graft-versus-Host Disease—Biologic Process, Prevention, and Therapy. N. Engl. J. Med. 2017, 377, 2167–2179. [Google Scholar] [CrossRef] [PubMed]
- Schoemans, H.M.; Lee, S.J.; Ferrara, J.L.; Wolff, D.; Levine, J.E.; Schultz, K.R.; Shaw, B.E.; Flowers, M.E.; Ruutu, T.; Greinix, H.; et al. EBMT−NIH−CIBMTR Task Force position statement on standardized terminology & guidance for graft-versus-host disease assessment. Bone Marrow Transplant. 2018, 53, 1401–1415. [Google Scholar] [CrossRef]
- Stevens, A.M.; Sullivan, K.M.; Nelson, J.L. Polymyositis as a manifestation of chronic graft-versus-host disease. Rheumatology 2003, 42, 34–39. [Google Scholar] [CrossRef]
- Shimabukuro-Vornhagen, A.; Hallek, M.J.; Storb, R.F.; Von Bergwelt-Baildon, M.S. The role of B cells in the pathogenesis of graft-versus-host disease. Blood 2009, 114, 4919–4927. [Google Scholar] [CrossRef] [PubMed]
- Misra, A.; Peethambaram, A.; Garg, A. Clinical Features and Metabolic and Autoimmune Derangements in Acquired Partial Lipodystrophy: Report of 35 Cases and Review of the Literature. Medicine (Baltimore) 2004, 83, 18–34. [Google Scholar] [CrossRef]
- Levy, J.; Burnett, M.E.; Magro, C.M. Lipophagic Panniculitis of Childhood: A Case Report and Comprehensive Review of the Literature. Am. J. Dermatopathol. 2017, 39, 217–224. [Google Scholar] [CrossRef]
- Corvillo, F.; Aparicio, V.; López-Lera, A.; Garrido, S.; Araújo-Vilar, D.; De Miguel, M.P.; López-Trascasa, M. Autoantibodies against perilipin 1 as a cause of acquired generalized lipodystrophy. Front. Immunol. 2018, 9, 1–13. [Google Scholar] [CrossRef]
- Corvillo, F.; Ceccarini, G.; Nozal, P.; Magno, S.; Pelosini, C.; Garrido, S.; López-Lera, A.; Moraru, M.; Vilches, C.; Fornaciari, S.; et al. Immunological features of patients affected by Barraquer-Simons syndrome. Orphanet J. Rare Dis. 2020, 15, 9. [Google Scholar] [CrossRef]
- Fischer-Posovszky, P.; Hebestreit, H.; Hofmann, A.K.; Strauss, G.; Möller, P.; Debatin, K.-M.; Wabitsch, M. Role of {CD}95-mediated adipocyte loss in autoimmune lipodystrophy. J. Clin. Endocrinol. Metab. 2006, 91, 1129–1135. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.F.; Rodday, A.M.; Kelly, M.J.; Must, A.; Macpherson, C.; Roberts, S.B.; Saltzman, E.; Parsons, S.K. Predictors of being overweight or obese in survivors of pediatric acute lymphoblastic leukemia (ALL). Pediatr. Blood Cancer 2014, 61, 1263–1269. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, H.C.; Marsh, J.A.; Rath, S.R.; Kotecha, R.S.; Gough, H.; Taylor, M.; Walwyn, T.; Gottardo, N.G.; Cole, C.H.; Choong, C.S. Increased Body Mass Index during Therapy for Childhood Acute Lymphoblastic Leukemia: A Significant and Underestimated Complication. Int. J. Pediatr. 2015, 2015, 1–10. [Google Scholar] [CrossRef]
- Zhang, F.F.; Liu, S.; Chung, M.; Kelly, M.J. Growth patterns during and after treatment in patients with pediatric ALL: A meta-analysis. Pediatr. Blood Cancer 2015, 62, 1452–1460. [Google Scholar] [CrossRef]
- Withycombe, J.S.; Smith, L.M.; Meza, J.L.; Merkle, C.; Faulkner, M.S.; Ritter, L.; Seibel, N.L.; Moore, K. Weight change during childhood acute lymphoblastic leukemia induction therapy predicts obesity: A report from the children’s oncology group. Pediatr. Blood Cancer 2015, 62, 434–439. [Google Scholar] [CrossRef]
- Reilly, J.J.; Brougham, M.; Montgomery, C.; Richardson, F.; Kelly, A.; Gibson, B.E.S. Effect of Glucocorticoid Therapy on Energy Intake in Children Treated for Acute Lymphoblastic Leukemia. J. Clin. Endocrinol. Metab. 2001, 86, 3742–3745. [Google Scholar] [CrossRef]
- Zhang, F.F.; Kelly, M.J.; Saltzman, E.; Must, A.; Roberts, S.B.; Parsons, S.K. Obesity in pediatric ALL survivors: A meta-analysis. Pediatrics 2014, 133, e704–e715. [Google Scholar] [CrossRef]
- Wilson, C.L.; Liu, W.; Yang, J.J.; Kang, G.; Ojha, R.P.; Neale, G.A.; Srivastava, D.K.; Gurney, J.G.; Hudson, M.M.; Robison, L.L.; et al. Genetic and clinical factors associated with obesity among adult survivors of childhood cancer: A report from the St. Jude Lifetime Cohort. Cancer 2015, 121, 2262–2270. [Google Scholar] [CrossRef] [PubMed]
- Lee, R.A.; Harris, C.A.; Wang, J.-C. Glucocorticoid Receptor and Adipocyte Biology. Nucl. Recept. Res. 2018, 5, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Hauner, H.; Entenmann, G.; Wabitsch, M.; Gaillard, D.; Ailhaud, G.; Negrel, R.; Pfeiffer, E.F. Promoting effect of glucocorticoids on the differentiation of human adipocyte precursor cells cultured in a chemically defined medium. J. Clin. Invest. 1989, 84, 1663–1670. [Google Scholar] [CrossRef] [PubMed]
- Bauerle, K.T.; Hutson, I.; Scheller, E.L.; Harris, C.A. Glucocorticoid Receptor Signaling Is Not Required for in Vivo Adipogenesis. Endocrinology 2018, 159, 2050–2061. [Google Scholar] [CrossRef] [PubMed]
- Rydén, M.; Uzunel, M.; Hård, J.L.; Borgström, E.; Mold, J.E.; Arner, E.; Mejhert, N.; Andersson, D.P.; Widlund, Y.; Hassan, M.; et al. Transplanted bone marrow-derived cells contribute to human adipogenesis. Cell Metab. 2015, 22, 408–417. [Google Scholar] [CrossRef] [PubMed]
- Gavin, K.M.; Gutman, J.A.; Kohrt, W.M.; Wei, Q.; Shea, K.L.; Miller, H.L.; Sullivan, T.M.; Erickson, P.F.; Helm, K.M.; Acosta, A.S.; et al. De novo generation of adipocytes from circulating progenitor cells in mouse and human adipose tissue. FASEB J. 2016, 30, 1096–1108. [Google Scholar] [CrossRef] [PubMed]
- Tsiloulis, T.; Watt, M.J. Exercise and the Regulation of Adipose Tissue Metabolism. Prog. Mol. Biol. Transl. Sci. 2015, 135, 175–201. [Google Scholar] [CrossRef]
- Berry, R.; Jeffery, E.; Rodeheffer, M.S. Weighing in on adipocyte precursors. Cell Metab. 2014, 19, 8–20. [Google Scholar] [CrossRef]
- Raajendiran, A.; Ooi, G.; Bayliss, J.; O’Brien, P.E.; Schittenhelm, R.B.; Clark, A.K.; Taylor, R.A.; Rodeheffer, M.S.; Burton, P.R.; Watt, M.J. Identification of Metabolically Distinct Adipocyte Progenitor Cells in Human Adipose Tissues. Cell Rep. 2019, 27, 1528–1540. [Google Scholar] [CrossRef]
- Jeffery, E.; Church, C.D.; Holtrup, B.; Colman, L.; Rodeheffer, M.S. Rapid depot-specific activation of adipocyte precursor cells at the onset of obesity. Nat. Cell Biol. 2015, 17, 376–385. [Google Scholar] [CrossRef]
- Jiang, Y.; Berry, D.C.; Tang, W.; Graff, J.M. Independent stem cell lineages regulate adipose organogenesis and adipose homeostasis. Cell Rep. 2014, 9, 1007–1022. [Google Scholar] [CrossRef]
- Stoltz, J.F.; De Isla, N.; Li, Y.P.; Bensoussan, D.; Zhang, L.; Huselstein, C.; Chen, Y.; Decot, V.; Magdalou, J.; Li, N.; et al. Stem Cells and Regenerative Medicine: Myth or Reality of the 21th Century. Stem Cells Int. 2015, 2015, 1–19. [Google Scholar] [CrossRef]
- Spyridonidis, A.; Schmitt-Gräff, A.; Tomann, T.; Dwenger, A.; Follo, M.; Behringer, D.; Finke, J. Epithelial Tissue Chimerism after Human Hematopoietic Cell Transplantation Is a Real Phenomenon. Am. J. Pathol. 2004, 164, 1147–1155. [Google Scholar] [CrossRef][Green Version]
- Tran, S.D.; Pillemer, S.R.; Dutra, A.; Barrett, A.J.; Brownstein, M.J.; Key, S.; Pak, E.; Leakan, R.A.; Kingman, A.; Yamada, K.M.; et al. Differentiation of human bone marrow-derived cells into buccal epithelial cells in vivo: A molecular analytical study. Lancet 2003, 361, 1084–1088. [Google Scholar] [CrossRef]
- Themeli, M.; Waterhouse, M.; Finke, J.; Spyridonidis, A. DNA chimerism and its consequences after allogeneic hematopoietic cell transplantation. Chimerism 2011, 2, 25. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dalakas, E.; Newsome, P.N.; Harrison, D.J.; Plevris, J.N. Hematopoietic stem cell trafficking in liver injury. FASEB J. 2005, 19, 1225–1231. [Google Scholar] [CrossRef] [PubMed]
- Filip, S.; Mokrý, J.; Vávrová, J.; Šinkorová, Z.; Mičuda, S.; Šponer, P.; Filipová, A.; Hrebíková, H.; Dayanithi, G. The peripheral chimerism of bone marrow-derived stem cells after transplantation: Regeneration of gastrointestinal tissues in lethally irradiated mice. J. Cell. Mol. Med. 2014, 18, 832–843. [Google Scholar] [CrossRef]
- Crossno, J.T.; Majka, S.M.; Grazia, T.; Gill, R.G.; Klemm, D.J. Rosiglitazone promotes development of a novel adipocyte population from bone marrow–derived circulating progenitor cells. J. Clin. Invest. 2006, 116, 3220–3228. [Google Scholar] [CrossRef] [PubMed]
- Majka, S.M.; Fox, K.E.; Psilas, J.C.; Helm, K.M.; Childs, C.R.; Acosta, A.S.; Janssen, R.C.; Friedman, J.E.; Woessner, B.T.; Shade, T.R.; et al. De novo generation of white adipocytes from the myeloid lineage via mesenchymal intermediates is age, adipose depot, and gender specific. Proc. Natl. Acad. Sci. USA 2010, 107, 14781–14786. [Google Scholar] [CrossRef] [PubMed]
- Tomiyama, K.; Murase, N.; Stolz, D.B.; Toyokawa, H.; O’Donnell, D.R.; Smith, D.M.; Dudas, J.R.; Rubin, J.P.; Marra, K.G. Characterization of transplanted GFP+ bone marrow cells into adipose tissue. Stem Cells 2008, 26, 330–338. [Google Scholar] [CrossRef] [PubMed]
| Ceccarini et al. 2017 [20] | Rooney and Ryan 2006 [21] | Adachi et al. 2013 [18] | Kimura et al. 2017 [22] | Shibata et al. 2018 [24] | Hosokawa et al. 2019 [23] | Adachi et al. 2020 [25] | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sex | f | f | f | f | m | m | f | f | F | f | f | f |
| Primary diagnosis | AML | ALL | AML | AML | ALL | NB | NB | ALL | AML | AML | ALL | AML |
| Graft | allogeneic | allogeneic | allogeneic | unrelated BMT | allogeneic | Scheduled PBSCT | allogeneic | allogeneic | allogeneic | Allogeneic | allogeneic | allogeneic |
| Age at transplant | 2 | 14 | 1 | 8 | 0 | 1 | 1 | 4 | 4 | 2 | 7 | 1 |
| Irradiation | TBI | TBI | TBI | TBI | TBI | TBI | TBI | TBI | TBI | TBI | TBI | |
| Age at diagnosis | 20 | 23 | 17 | 15 | 19 | 19 | 17 | 10 | 28 | 14 | 17 | 17 |
| Treatment-related complications | GVHD, GH deficiency, hypothyroidism, and hypogonadism. | GVHD. | GVHD, GH deficiency, hypothyroidism, leukoencephalopathy, epilepsy, and hypogonadism. | GVHD, femoral neck necrosis, aplastic anemia, hepatic angioma, hypothyroidism, and primary hypogonadism. | GVHD, GH deficiency, and chronic thyroiditis. | No GVHD, hypothyroidism, empty sella, GH deficiency, and hypogonadism. | GVHD, GH deficiency, hypogonadism, high-frequency deafness, and cataracts. | GVHD. | GVHD. | GVHD. | GVHD, leukoencephalopathy, intractable epilepsy, moderate intellectual impairment, cataracts, GH deficiency, hypothyroidism, and hypogonadism. | |
| Metabolic complications | T2DM fatty liver | T2DM low serum AdipoQ | DM Dyslipidemia fatty liver | Hyperinsulinemia dyslipidemia | hyperinsulinemia dyslipidemia | fatty liver increased visceral fat hyperinsulinemia | fatty liver | Hyperglycemia | T2DM fatty liver | Hyperglycemia dyslipidemia | T2DM dyslipidemia | Hyperinsulinemia hypertriglyceridemia fatty liver |
| BMI | 14 | x | 17.7 | 12.2 | 16.5 | 18.3 | 14.1 | x | 16.9 | 13.2 | 17.0 | x |
| HbA1c (%) (4–5.9) | 7.46 | 10.5 | 6.10 | 5.40 | 5.30 | 5.70 | 5.20 | 9.2 | 8.7 | 6.3 | 7.3 | 9.50 |
| fTG (mg/dL) (<100) | 654 | 1301 | 675 | 965 | 901 | 1073 | 402 | 3090 | 986 | 332 | 927 | 490 |
| total cholesterol (mg/dL) | 277 | 228 | 322 | 375 | 284 | 314 | 203 | x | x | x | 336 | x |
| HDL cholesterol (mg/dL) | x | 39 | 39 | 50 | 44 | 44 | 35 | x | 40 | 33 | 34 | x |
| LDL cholesterol (mg/dL) | x | 168 | 203 | 179 | 176 | 124 | x | x | x | x | x | |
| fInsulin (µU/dL) | x | 115.1 | x | x | x | x | x | x | x | 54 | 53.9 | 53.9 |
| fBG (mg/dL) | 126 | x | x | x | x | x | 354 | 232 | 128 | 132 | 100 | |
| LEP (ng/mL) | 7.4 | 10.60 | 18.70 | 9.50 | 10.70 | 17.90 | 11.90 | x | 6.5 | 5.6 | 3.5 | x |
| AdipoQ (µg/mL) | x | 0.90 | 1.60 | 6.80 | 8.50 | 1.70 | 3.80 | x | x | 1.6 | <1.9 | 1.80 |
| ALT (U/L) | 62 | 90 | 56 | 102 | 85 | 137 | 250 | x | 19 | 40 | 78 | 110 |
| AST (U/L) | 141 | x | x | x | x | x | x | x | 21 | 49 | 85 | x |
| GGT (U/L) | 140 | 91 | 387 | x | x | x | x | x | 32 | x | x | 177 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tews, D.; Schulz, A.; Denzer, C.; von Schnurbein, J.; Ceccarini, G.; Debatin, K.-M.; Wabitsch, M. Lipodystrophy as a Late Effect after Stem Cell Transplantation. J. Clin. Med. 2021, 10, 1559. https://doi.org/10.3390/jcm10081559
Tews D, Schulz A, Denzer C, von Schnurbein J, Ceccarini G, Debatin K-M, Wabitsch M. Lipodystrophy as a Late Effect after Stem Cell Transplantation. Journal of Clinical Medicine. 2021; 10(8):1559. https://doi.org/10.3390/jcm10081559
Chicago/Turabian StyleTews, Daniel, Ansgar Schulz, Christian Denzer, Julia von Schnurbein, Giovanni Ceccarini, Klaus-Michael Debatin, and Martin Wabitsch. 2021. "Lipodystrophy as a Late Effect after Stem Cell Transplantation" Journal of Clinical Medicine 10, no. 8: 1559. https://doi.org/10.3390/jcm10081559
APA StyleTews, D., Schulz, A., Denzer, C., von Schnurbein, J., Ceccarini, G., Debatin, K.-M., & Wabitsch, M. (2021). Lipodystrophy as a Late Effect after Stem Cell Transplantation. Journal of Clinical Medicine, 10(8), 1559. https://doi.org/10.3390/jcm10081559






