Effects of Hyperbaric Oxygen Therapy in Children with Severe Atopic Dermatitis
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Severity of Skin Lesions
2.3. Immunological Parameters
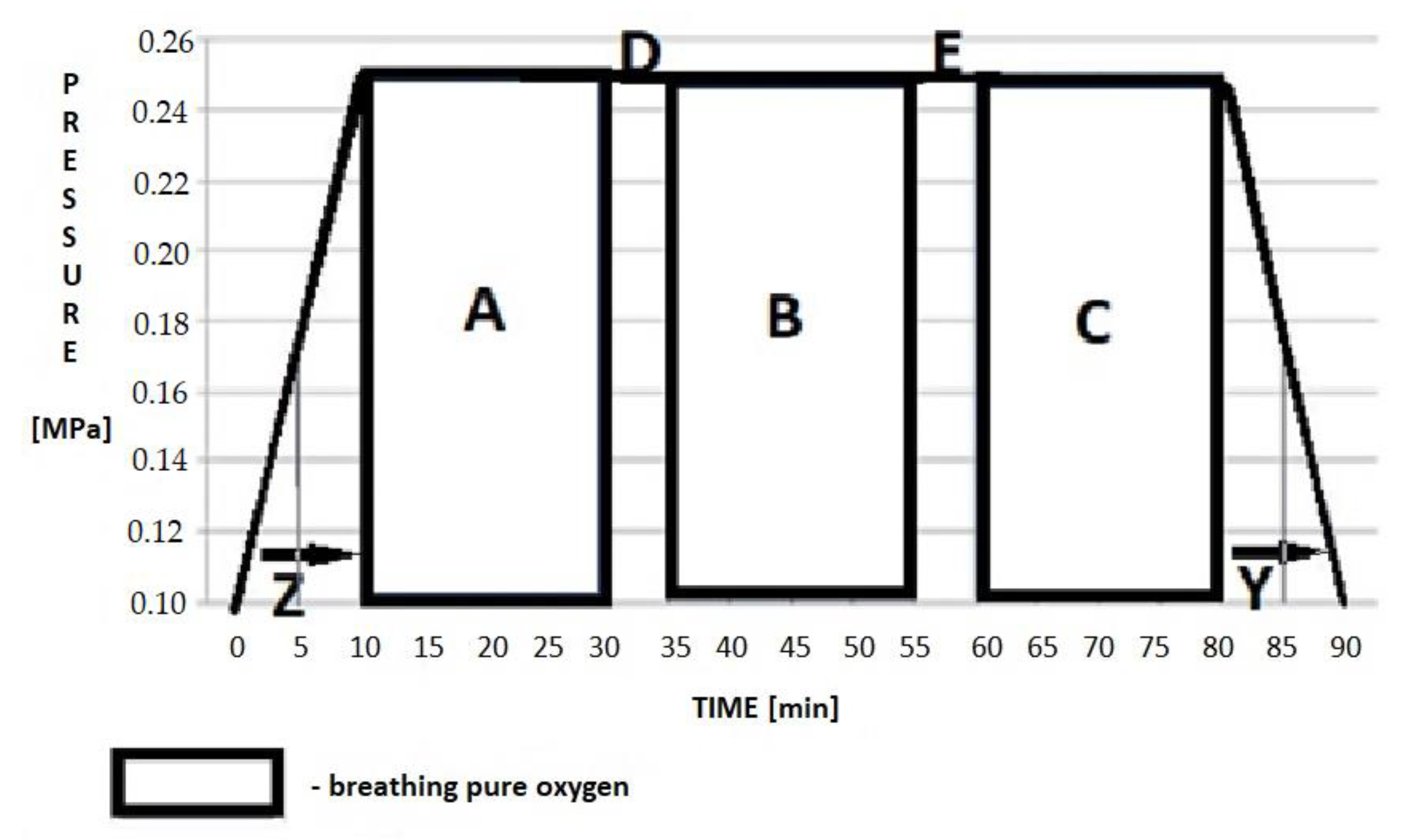
2.4. Hyperbaric Oxygen Therapy
2.5. Statistical Analysis
3. Results
3.1. Characteristics of the Study Group
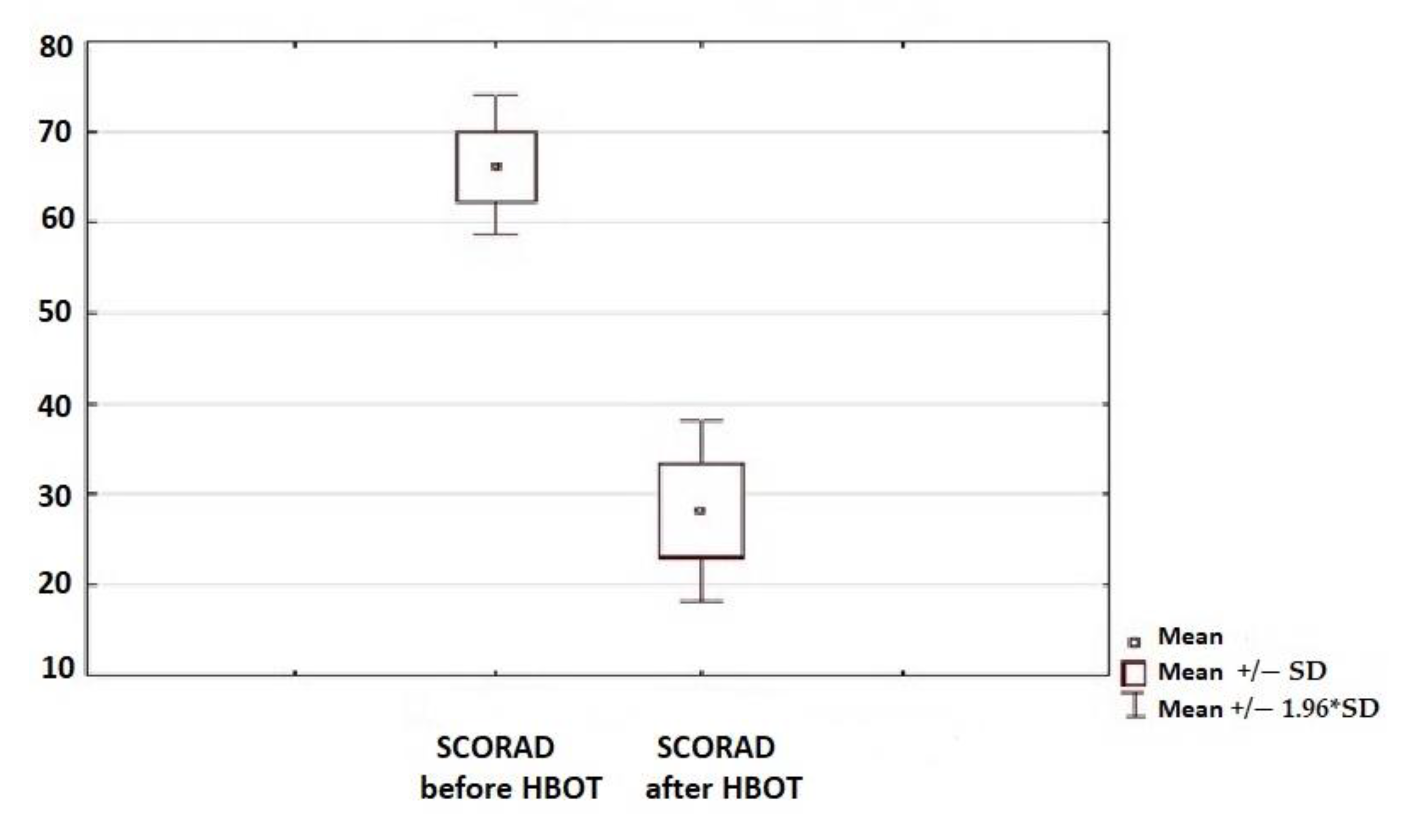
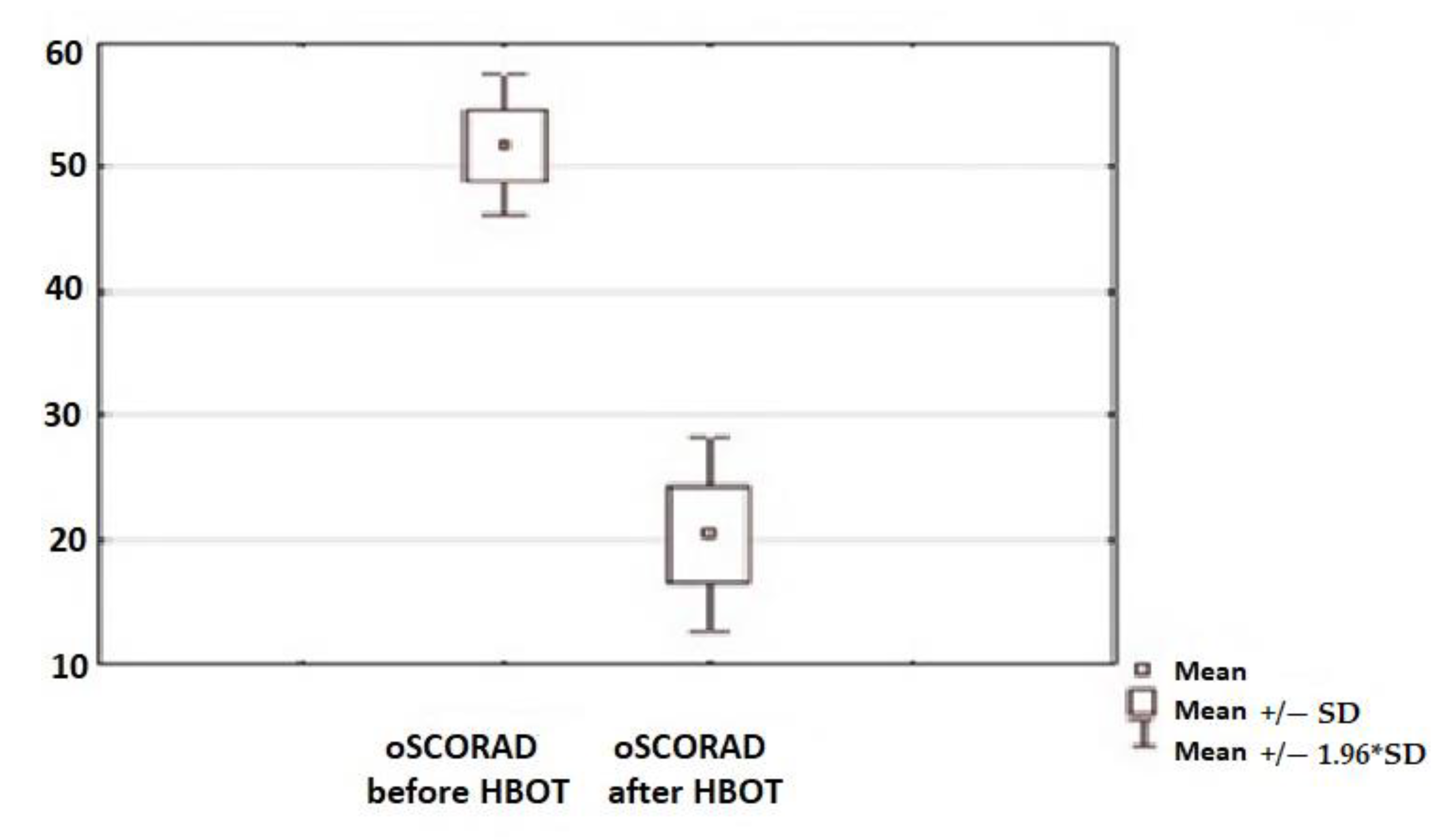
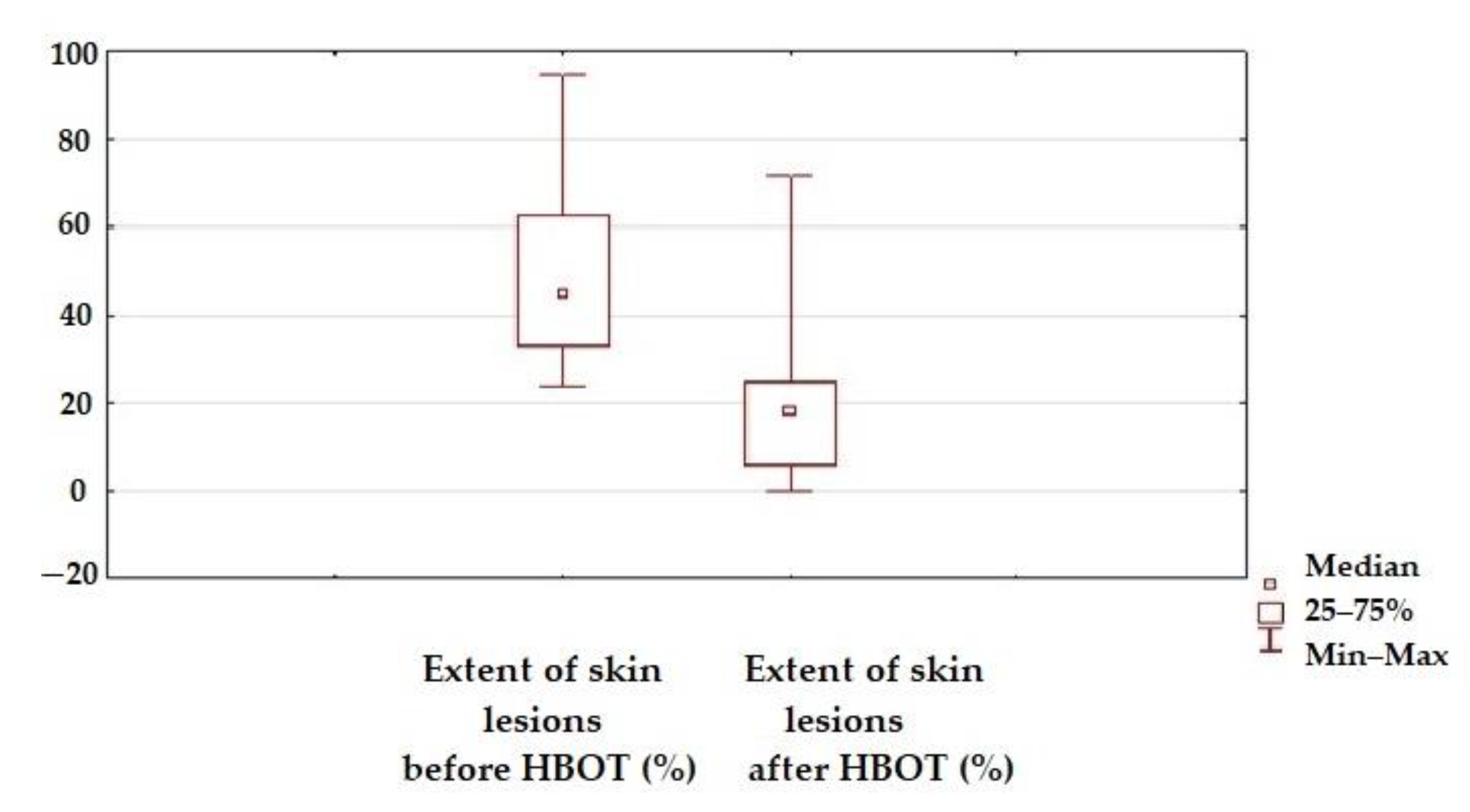
3.2. Skin Condition
3.3. Immunological Parameters
4. Discussion
5. Conclusions
- The use of hyperbaric therapy in children with a severe course of atopic dermatitis has a positive impact on the treatment results of this disease. It reduces the severity of skin lesions, as well as its dryness, the presence of redness, swelling, oozing/crusting, scratch marks and lichenification. HBOT has a positive effect on the reduction of pruritus intensity and improves sleep quality in patients with severe AD. This has a significant impact on improving the quality of life of these patients.
- Hyperbaric oxygen therapy may be a therapeutic option for some patients with atopic dermatitis, especially in severe cases that are resistant to the standard methods of treatment.
- The study did not show a clear effect of hyperbaric oxygen therapy on the functioning of the immune system. Immunological parameters presented in this study are not sufficient to discuss all of the molecular mechanisms underlying the immunomodulatory effects of HBOT.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lyons, J.; Milner, J.; Stone, K. Atopic dermatitis in children: Clinical features, pathophysiology and treatment. Immunol. Allergy Clin. 2015, 35, 161–183. [Google Scholar] [CrossRef]
- Hanifin, J.M.; Rajka, G. Diagnostic features of atopic dermatitis. Acta Derm. Venereol. 1980, 92, 44–47. [Google Scholar]
- Bieber, T. Atopic dermatitis. Ann. Dermatol. 2010, 22, 125–137. [Google Scholar] [CrossRef] [PubMed]
- Samochocki, Z.; Alifier, M.; Bodera, P.; Jeziorkowska, R.; Rosiak, E.; Jurkiewicz, B.; Glińska, O.; Gliński, W.; Stankiewicz, W. T-regulatory cells in severe atopic dermatitis: Alterations related to cytokines and other lymphocyte subpopulations. Arch. Dermatol. Res. 2012, 304, 795–801. [Google Scholar] [CrossRef]
- Lipińska-Opałka, A.; Wawrzyniak, A.; Lewicki, S.; Zdanowski, R.; Kalicki, B. Evaluation of Immune Indices and Serum Vitamin D Content in Chil-dren with Atopic Dermatitis. Adv. Exp. Med. Biol. 2017, 42, 101–106. [Google Scholar]
- Chiarelli, F.; Canfora, G.; Verrotti, A.; Amerio, P.; Morgese, G. Humoral and cellular immunity in children with active and quiescent atopic dermati-tis. Br. J. Dermatol. 1980, 116, 651–660. [Google Scholar] [CrossRef] [PubMed]
- Bacchetta, R.; Gambineri, E.; Roncarolo, M.-G. Role of regulatory T cells and FOXP3 in human diseases. J. Allergy Clin. Immunol. 2007, 120, 227–235. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhou, B. Functions of Thymic Stromal Lymphopoietin in Immunity and Disease. Immunol. Res. 2012, 52, 211–223. [Google Scholar] [CrossRef]
- Wong, L.-S.; Wu, T.; Lee, C.-H. Inflammatory and Noninflammatory Itch: Implications in Pathophysiology-Directed Treatments. Int. J. Mol. Sci. 2017, 18, 1485. [Google Scholar] [CrossRef]
- Paprocki, J.; Gackowska, M.; Pawłowska, M.; Woźniak, A. The current use of hyperbaric oxygen treatment. Med. Rodz. 2016, 19, 217–222. [Google Scholar]
- Roekevisch, E.; Spuls, P.I.; Kuester, D.; Limpens, J.; Schmitt, J. Efficacy and safety of systemic treatments for moderate to severe atopic dermatitis: A systematic review. J. Allergy Clin. Immunol. 2014, 133, 429–437. [Google Scholar] [CrossRef]
- Nowicki, R.; Trzeciak, M.; Wilkowska, A.; Sokołowska-Wojdyło, M.; Ługowska-Umer, H.; Barańska-Rybak, W.; Kaczmarski, M.; Kowalewski, C.; Kruszewski, J.; Maj, J.; et al. Special paper Atopic dermatitis: Current treatment guidelines. Statement of the experts of the Dermatological Section, Polish Society of Allergology, and the Allergology Section, Polish Society of Dermatology. Adv. Dermatol. Allergol. 2015, 4, 239–249. [Google Scholar] [CrossRef]
- Wawrzyniak, A.; Lipińska-Opałka, A.; Zdanowski, R.; Lewicki, S.; Murawski, P.; Kalicki, B. Evaluation of selected immunological parameters and the concentra-tion of vitamin D in children with asthma. Case-control study. Cent Eur. J. Immunol. 2017, 42, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Bartlet, L.B.; Westbroek, R.; White, E.J. Sleep patterns in children with atopic eczema. Acta Derm. Venereol. 1997, 77, 446–448. [Google Scholar] [PubMed]
- Ben-Gashir, M.A.; Seed, P.T.; Hay, R.J. Are quality of family life and disease severity related in childhood atopic dermatitis? J. Eur. Acad. Dermatol. Venereol. 2002, 16, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Siewiera, J.; Mews, J.; Królikowska, K.; Kalicki, B.; Jobs, K. Hyperbaric oxygenation in pediatrics: Indications in the light of evidence—Based medicine. Dev. Period Med. 2019, 23, 142–148. [Google Scholar]
- Mews, J.; Królikowska, K.; Siewiera, J.; Wawrzyniak, A.; Kalicki, B. Assessment of hyperbaric oxygen efficacy in children with carbon monoxide poison-ing. Pediatr. Med. Rodz. 2019, 15, 157–163. [Google Scholar] [CrossRef]
- Mathieu, D.; Marroni, A.; Kot, J. Tenth European Consensus Conference on Hyperbaric Medicine: Recommendations for accept-ed and nonaccepted clinical indications and practice of hyperbaric oxygen treatment. Diving Hyperb. Med. 2017, 47, 24–32. [Google Scholar] [CrossRef]
- Edwards, M.L. Hyperbaric oxygen therapy. Part 1: History and principles. J. Veter. Emerg. Crit. Care 2010, 20, 284–288. [Google Scholar] [CrossRef]
- Edwards, M.L. Hyperbaric oxygen therapy. Part 2: Application in disease. J. Veter. Emerg. Crit. Care 2010, 20, 289–297. [Google Scholar] [CrossRef]
- Dulai, P.S.; Buckey, J.C., Jr.; Raffals, L.E. Hyperbaric oxygen therapy is well tolerated and effective for ulcerative colitis patients hospitalized for moderate–severe flares: A phase 2A pilot multi-center, randomized, double-blind, sham-controlled trial. Am. J. Gastroenterol. 2018, 10, 1516–1523. [Google Scholar] [CrossRef]
- Jeter, J.P.; Wong, E.B. Hyperbaric oxygen therapy in dermatology. Cutis 2020, 105, 24–27. [Google Scholar]
- Bennardop, L.; Del Duca, E.; Dastoli, S.; Schipani, G.; Scali, E.; Silvestri, M.; Nisticò, S.P.; Bennardo, L. Potential applications of topical oxygen therapy in dermatology. Dermatol. Pract. Concept. 2018, 8, 272–276. [Google Scholar] [CrossRef]
- Hao, Y.; Zhang, L.; Chen, Y. Hyperbaric oxygen for the treatment of skin diseases. Chin. J. Dermatol. 2016, 49, 672–675. [Google Scholar]
- Olszański, R.; Konarski, M.; Siermontowski, P. Leczenie tlenoterapią hiperbaryczną (HBOT) jako opcja terapeutyczna dla cho-rych na atopowe zapalenie skóry (AZS)—Doświadczenia własne i przegląd piśmiennictwa. Pol. Hyperb. Res. 2017, 60, 3. [Google Scholar]
- Noval Rivas, M.; Chatila, T. Regulatory T cells in Allergic Diseases. J. Allergy Clin. Immunol. 2016, 138, 639–652. [Google Scholar] [CrossRef] [PubMed]
- Ulewicz, K.; Zannini, D. On the possibility of hyperbaric oxygen therapy in some pathological reactions of immunological hy-persensitivity. Bull. Inst. Mar. Trop. Med. Gdynia 1986, 37, 71–79. [Google Scholar]
- Matsuda, J.; Mallevaey, T.; Scott-Browne, J.; Gapin, L. CD1d-restricted iNKT cells, the “Swiss-Army knife” of the immune system. Curr. Opin. Immunol. 2008, 20, 358–368. [Google Scholar] [CrossRef]
- Scordamaglia, F.; Balsamo, M.; Scordamaglia, A.; Moretta, A.; Mingari, M.C.; Canonica, G.W.; Moretta, L.; Vitale, M. Perturbations of natural killer cell regulatory functions in respiratory aller-gic diseases. J. Allergy Clin. Immunol. 2008, 121, 479–485. [Google Scholar] [CrossRef]
- Stelmaszczyk-Emmel, A.; Zawadzka-Krajewska, A.; Szypowska, A.; Kulus, M.; Demkow, U. Frequency and activation of CD4+CD25 FoxP3+ regu-latory T cells in peripheral blood from children with atopic allergy. Int. Arch. Allergy Immunol. 2013, 162, 16–24. [Google Scholar] [CrossRef]
- Lee, J.-H.; Yu, H.-H.; Wang, L.-C.; Yang, Y.-H.; Lin, Y.-T.; Chiang, B.-L. The levels of CD4+CD25+ regulatory T cells in paediatric patients with allergic rhinitis and bronchial asthma. Clin. Exp. Immunol. 2007, 148, 53–63. [Google Scholar] [CrossRef] [PubMed]
- Xu, G.; Mou, Z.; Jiang, H.; Cheng, L.; Shi, J.; Xu, R.; Oh, Y.; Li, H. A Possible Role of CD4+CD25+ T Cells as Well as Transcription Factor Foxp3 in the Dysregulation of Allergic Rhinitis. Laryngoscope 2007, 117, 876–880. [Google Scholar] [CrossRef]
- De Vries, J.E. Immunosuppressive and anti-inflammatory properties of IL-10. Ann. Med. 1995, 27, 537–541. [Google Scholar] [CrossRef] [PubMed]
- Nedoszytko, B.; Lange, M.; Sokołowska-Wojdyło, M.; Renke, J.; Trzonkowski, P.; Sobjanek, M.; Szczerkowska-Dobosz, A.; Niedoszytko, M.; Górska, A.; Romantowski, J.; et al. The role of regulatory T cells and genes involved in their differentia-tion in pathogenesis of selected inflammatory and neoplastic skin diseases. Part II: The Treg role in skin diseases pathogenesis. Adv. Dermatol. Allergol. 2017, 34, 405–417. [Google Scholar] [CrossRef] [PubMed]
- Schnopp, C.; Rad, R.; Weidinger, A.; Weidinger, S.; Ring, J.; Eberlein, B.; Ollert, M.; Mempel, M. Fox-P3-positive regulatory T cells are present in the skin of generalized atopic eczema patients and are not particulary affected by medium-dose UVA1 therapy. Photodermatol. Photoimmunol. Photomed. 2007, 23, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Verhagen, J.; Akdis, M.; Traidl-Hoffmann, C.; Schmid-Grendelmeier, P.; Hijnen, D.; Knol, E.F.; Behrendt, H.; Blaser, K.; Akdis, C.A. Absence of T-regulatory cell expression and function in atopic dermatitis skin. J. Allergy Clin. Immunol. 2006, 117, 176–183. [Google Scholar] [CrossRef]
- Di Cesare, A.; Di Meglio, P.; Nestle, F.O. A role for Th17 cells in the immunopathogenesis of atopic dermatitis? J. Investig. Dermatol. 2008, 128, 2569–2571. [Google Scholar] [CrossRef]
- Butler, G.; Michaels, J.C.; Al-Waili, N.; Finkelstein, M.; Allen, M.; Petrillo, R.; Carrey, Z.; Kolanuvada, B.; Lee, B.Y.; Riera, A.G.; et al. Therapeutic effect of hyperbaric oxygen in psoriasis vulgaris: Two case reports and a review of the literature. J. Med. Case Rep. 2009, 3, 7023. [Google Scholar] [CrossRef][Green Version]
- Kindwall, E.P.; Whelan, H.T. Hiperbaric Medicine Practice; Best Publishing Company: North Palm Beach, FL, USA, 2008; pp. 3–115, 301–316, 655–676. [Google Scholar]
- Kranke, P.; Bennett, M.H.; Martyn-St James, M.; Schnabel, A.; Debus, S.E.; Weibel, S. Hyperbaric oxygen therapy for chronic wounds. Ochr. Database Syst. Rev. 2015, 6, CD004123. [Google Scholar] [CrossRef]
- Kraaij, M.D.; Savage, N.D.L.; Van Der Kooij, S.W.; Koekkoek, K.; Wang, J.; Berg, J.M.V.D.; Ottenhoff, T.H.M.; Kuijpers, T.W.; Holmdahl, R.; Van Kooten, C.; et al. Induction of regulatory T cells by macrophages is dependent on production of reactive oxygen species. Proc. Natl. Acad. Sci. USA 2010, 107, 17686–17691. [Google Scholar] [CrossRef]
- Dang, E.V.; Barbi, J.; Yang, H.Y.; Jinasena, D.; Yu, H.; Zheng, Y.; Bordman, Z.; Fu, J.; Kim, Y.; Yen, H.R.; et al. Control of T(H)17/T(reg) balance by hypoxia-inducible factor 1. Cell 2011, 146, 772–784. [Google Scholar] [CrossRef]
- Dennog, C.; Radermacher, P.; Barnett, A.Y.; Speit, G. Antioxidant status in humans after exposure to hyperbaric oxygen. Mutat. Res. Mol. Mech. Mutagen. 1999, 428, 83–89. [Google Scholar] [CrossRef]
- Saito, K.; Tanaka, Y.; Ota, T.; Eto, S.; Yamashita, U. Suppressive effect of hyperbaric oxygenation on immune responses of normal and autoim-mune mice. Clin. Exp. Immunol. 1991, 86, 322–327. [Google Scholar] [CrossRef]
- Hultqvist, M.; Olsson, L.M.; Gelderman, K.A.; Holmdahl, R. The protective role of ROS in autoimmune disease. Trends Immunol. 2009, 30, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Thom, S.R. Hyperbaric oxygen: Its mechanisms and efficacy. Plast. Reconstr. Surg. 2011, 127, 131–141. [Google Scholar] [CrossRef] [PubMed]
- Munn, D.H.; Mellor, A.L. Indoleamine 2,3 dioxygenase and metabolic control of immune responses. Trends Immunol. 2013, 34, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Won, H.Y.; Sohn, J.H.; Min, H.J.; Lee, K.; Woo, H.A.; Ho, Y.-S.; Park, J.W.; Rhee, S.; Hwang, E.S. Glutathione Peroxidase 1 Deficiency Attenuates Allergen-Induced Airway Inflammation by Suppressing Th2 and Th17 Cell Development. Antioxid. Redox Signal. 2010, 13, 575–587. [Google Scholar] [CrossRef]
- Faleo, G.; Fotino, C.; Bocca, N.; Molano, R.D.; Zahr-Akrawi, E.; Molina, J.; Villate, S.; Umland, O.; Skyler, J.S.; Bayer, A.L.; et al. Prevention of autoimmune diabetes and induction of beta-cell proliferation in NOD mice by hyperbaric oxygen therapy. Diabetes 2012, 61, 1769–1778. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kim, H.-R.; Kim, J.-H.; Choi, E.-J.; Lee, Y.K.; Kie, J.-H.; Jang, M.H.; Seoh, J.-Y. Hyperoxygenation Attenuated a Murine Model of Atopic Dermatitis through Raising Skin Level of ROS. PLoS ONE 2014, 9, e109297. [Google Scholar] [CrossRef] [PubMed]
- Bouaziz, J.D.; Yanaba, K.; Venturi, G.M.; Wang, Y.; Tisch, R.M.; Poe, J.C.; Tedder, T.F. Therapeutic B cell depletion impairs adaptive and autoreactive CD4+ T cell activa-tion in mice. Proc. Natl. Acad. Sci. USA 2007, 104, 20882–20887. [Google Scholar] [CrossRef] [PubMed]
- Simon, D.; Hösli, S.; Kostylina, G.; Yawalkar, N.; Simon, H.-U. Anti-CD20 (rituximab) treatment improves atopic eczema. J. Allergy Clin. Immunol. 2008, 121, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Kasperkiewicz, M.; Schmidt, E.; Ludwig, R.J.; Zillikens, D. Targeting IgE Antibodies by Immuno-adsorption in Atopic Dermatitis. Front. Immunol. 2018, 9, 254. [Google Scholar] [CrossRef]
- Thom, S.R. Oxidative stress is fundamental to hyperbaric oxygen therapy. J. Appl. Physiol. 2009, 106, 988–995. [Google Scholar] [CrossRef] [PubMed]
- Narożny, W.; Siebert, J. Możliwości i ograniczenia stosowania tlenu hiperbarycznego w medycynie. Forum Med. Rodz. 2007, 4, 368–375. [Google Scholar]
| Inclusion Criteria | Exclusion Criteria |
|---|---|
| Children ≥3 years of age and <18 years of age | Children with symptoms of respiratory tract infection |
| Cooperative patients | Neurological contraindications (history of seizures or epilepsy) |
| Tolerance of confined spaces (assessment of the guardians and the team of the Clinical Department of Hyperbaric Medicine) | Pulmonary contraindications (uncontrolled asthma, pulmonary hypertension) |
| Severe atopic dermatitis (>50 points on SCORAD and >40 points on the oSCORAD scale) | Otolaryngological contraindications |
| Elevated serum total IgE above age norm | Cardiological contraindications (heart defects, hemodynamically significant PFO) |
| No improvement after the use of available treatment methods | Oncological contraindications (immunosuppressive treatment for neoplastic disease) |
| Written consent of the legal guardians/patient (if they are above 16 years old) |
| Age (Years) | (Mean ± SD) | 10.4 ± 4.4 |
| Sex (Girls/Boys) | 7:8 | |
| Weight (percentiles) | (Mean ± SD) | 57 ± 20 |
| Height (percentiles) | (Mean ± SD) | 52 ± 29 |
| Serum total IgE antibodies (IU/mL) | (Median, Q1, Q3) | 1803 (Q1, 1000; Q3, 9000) |
| SCORAD points | (Mean ± SD) | 66.2 ± 15.1 |
| oSCORAD points | (Mean ± SD) | 51.7 ± 11.2 |
| WBC (×109/L) | (Mean ± SD) | 7.9 ± 2.3 |
| Eosinophils (×109/L) | (Mean ± SD) | 0.6 ± 0.5 |
| Eosinophils (%) | (Mean ± SD) | 8.1 ± 5.6 |
| CRP (ng/dl) | (Mean ± SD) | 0.1 ± 0.05 |
| Duration of the disease (Years) | (Mean ± SD) | 8.8 ± 4.2 |
| Positive family history of allergies | 2 patients | |
| Coexisting atopic diseases | 1 patient: asthma | |
| 2 patients: allergic rhinitis | ||
| 12 patients: inhaled and/or food allergy | ||
| Antihistamines used | 11 patients | |
| SCORAD—scoring atopic dermatitis; oSCORAD—objective scoring atopic dermatitis; WBC—white blood cells | ||
| SCORAD Objective Parameters | Before HBOT Median, n = 15 | After HBOT Median, n = 15 | p |
|---|---|---|---|
| Skin dryness | 2 | 1 | 0.0400 |
| Redness | 2 | 1 | 0.0090 |
| Swelling | 1 | 0 | 0.0200 |
| Oozing/crusting | 2 | 1 | 0.0005 |
| Scratch marks | 2 | 1 | 0.0020 |
| Skin lichenification | 2 | 1 | 0.0002 |
| SCORAD subjective parameters | Before HBOT median, n = 15 | After HBOT median, n = 15 | p |
| Pruritus | 9 | 4 | 0.030 |
| Sleep disorders | 7 | 3 | 0.040 |
| Immunological Parameter | Before HBOT | After HBOT | p | ||||
|---|---|---|---|---|---|---|---|
| Q1 | Median | Q3 | Q1 | Median | Q3 | ||
| Total serum IgE (IU/mL) | 1000 | 1803 | 9000 | 1000 | 1661 | 6000 | 0.002 |
| IL-4 (pg/mL) | 2.5 | 3.83 | 4.2 | 1.8 | 2.98 | 4 | 1 |
| IL-6 (pg/mL) | 1.9 | 1.93 | 3.6 | 1.7 | 1.65 | 2.2 | 0.26 |
| IL-10 (pg/mL) | 1.4 | 1.99 | 3.4 | 1.6 | 2.88 | 3.6 | 0.42 |
| Treg CD4 + CD25highCD127-FOXP3+ (%) | 0.7 | 0.98 | 1.4 | 0.55 | 0.71 | 0.9 | 0.14 |
| NKT (%) | 1.8 | 2.01 | 3.5 | 1.5 | 1.71 | 3.8 | 0.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mews, J.; Tomaszewska, A.; Siewiera, J.; Lewicki, S.; Kuczborska, K.; Lipińska-Opałka, A.; Kalicki, B. Effects of Hyperbaric Oxygen Therapy in Children with Severe Atopic Dermatitis. J. Clin. Med. 2021, 10, 1157. https://doi.org/10.3390/jcm10061157
Mews J, Tomaszewska A, Siewiera J, Lewicki S, Kuczborska K, Lipińska-Opałka A, Kalicki B. Effects of Hyperbaric Oxygen Therapy in Children with Severe Atopic Dermatitis. Journal of Clinical Medicine. 2021; 10(6):1157. https://doi.org/10.3390/jcm10061157
Chicago/Turabian StyleMews, Judyta, Agata Tomaszewska, Jacek Siewiera, Sławomir Lewicki, Karolina Kuczborska, Agnieszka Lipińska-Opałka, and Bolesław Kalicki. 2021. "Effects of Hyperbaric Oxygen Therapy in Children with Severe Atopic Dermatitis" Journal of Clinical Medicine 10, no. 6: 1157. https://doi.org/10.3390/jcm10061157
APA StyleMews, J., Tomaszewska, A., Siewiera, J., Lewicki, S., Kuczborska, K., Lipińska-Opałka, A., & Kalicki, B. (2021). Effects of Hyperbaric Oxygen Therapy in Children with Severe Atopic Dermatitis. Journal of Clinical Medicine, 10(6), 1157. https://doi.org/10.3390/jcm10061157






