Visual Evoked Potential Responses after Photostress in Migraine Patients and Their Correlations with Clinical Features
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Visual Evoked Potentials
2.3. Procedure
2.4. Statistical Analysis
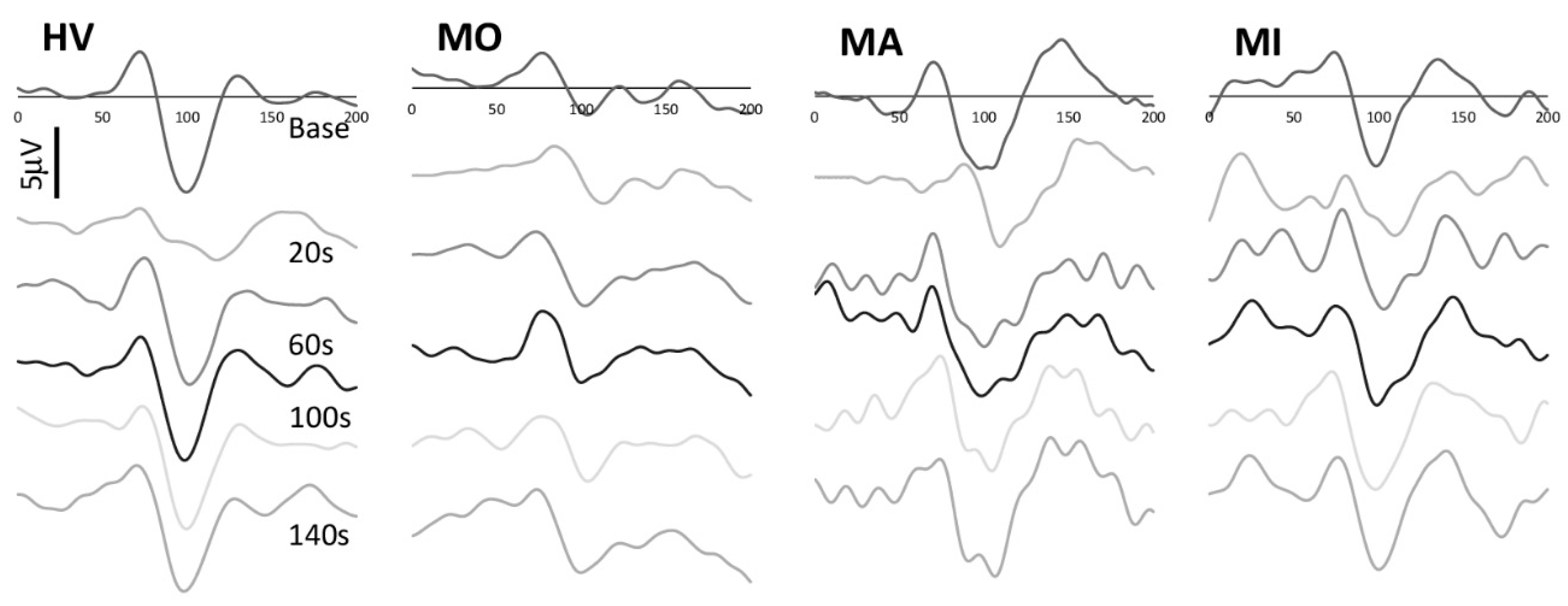
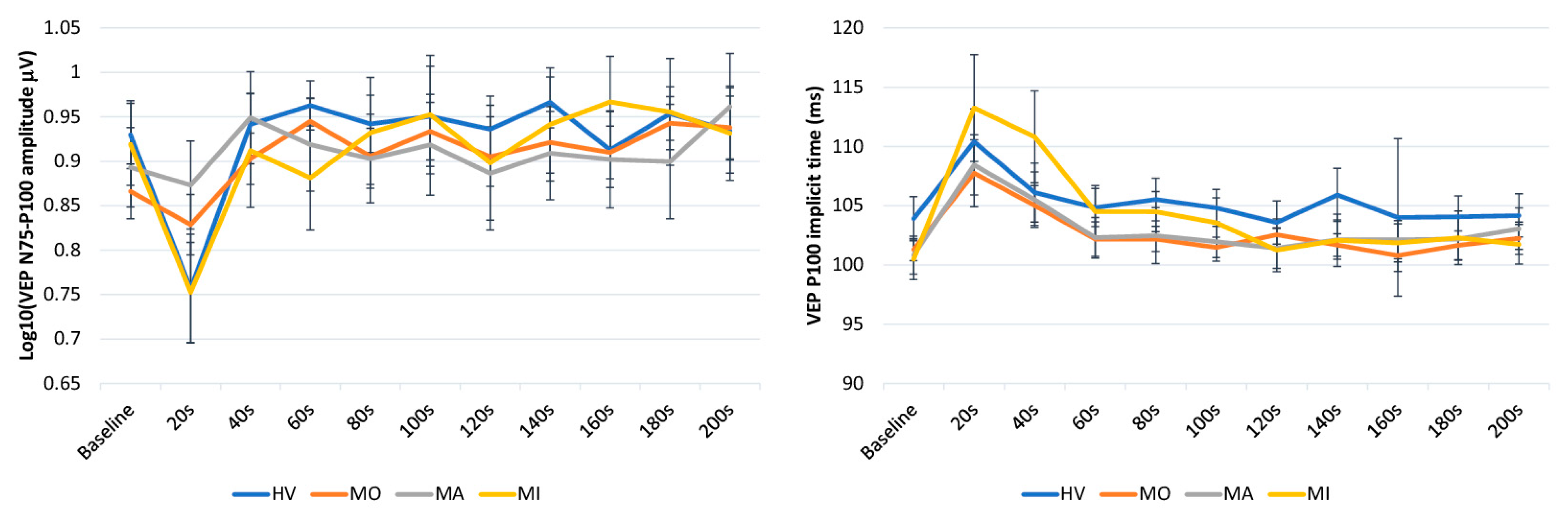
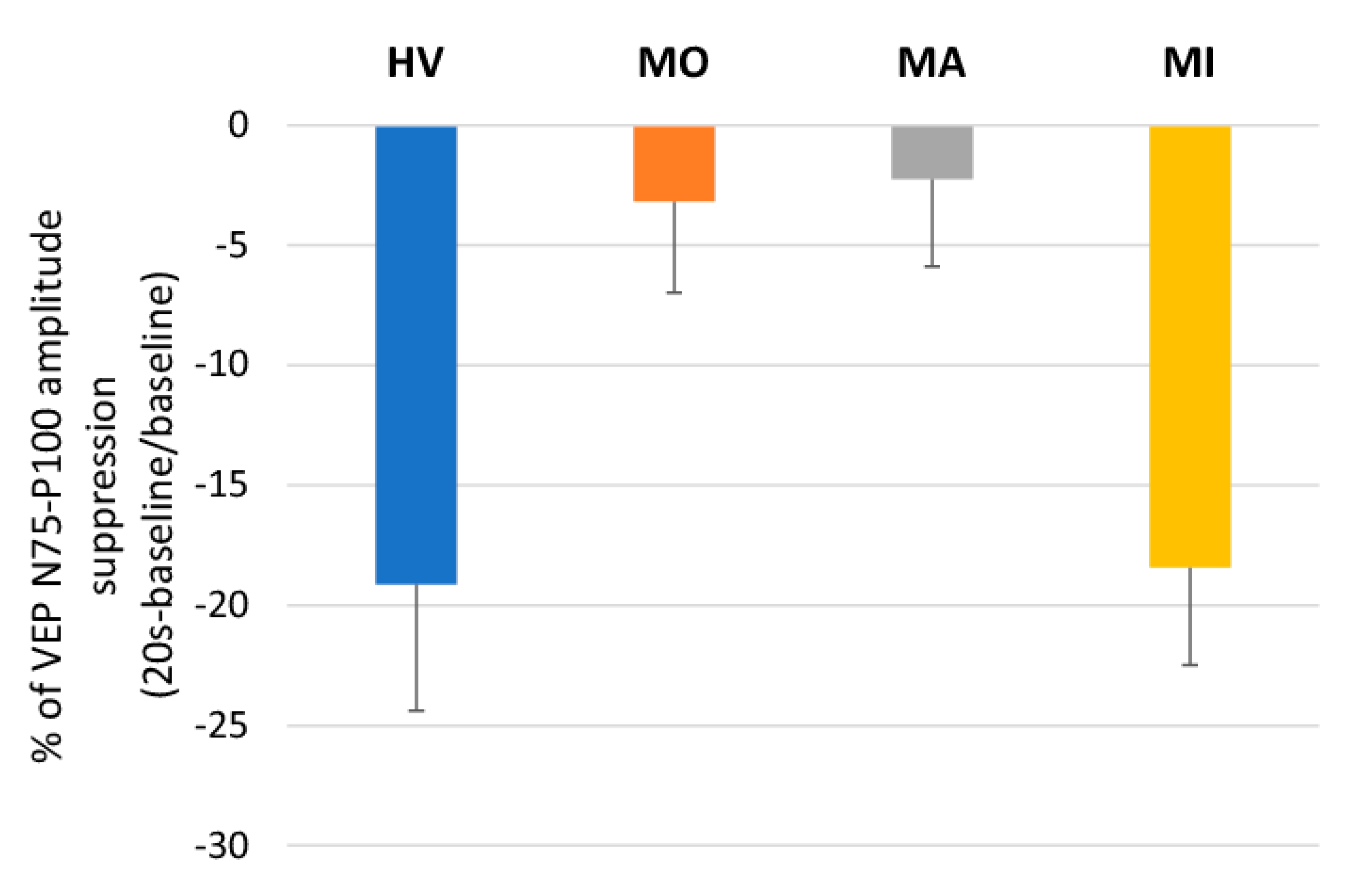
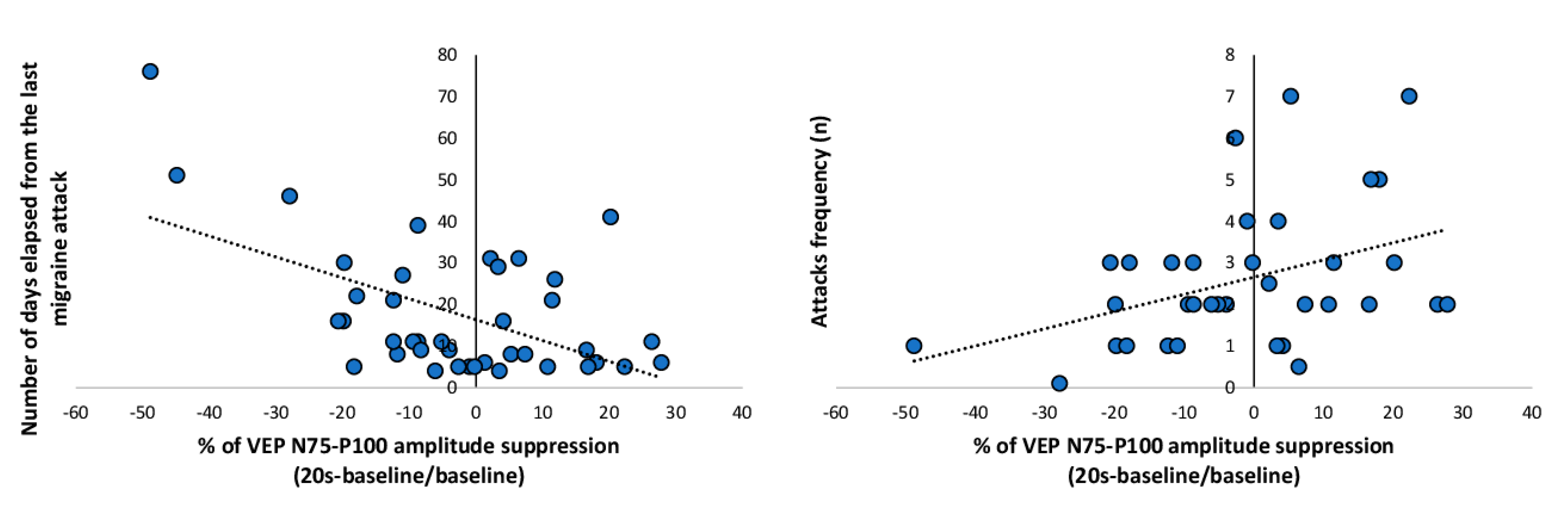
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| HV | healthy volunteers |
| ICHD | international classification of headache disorders |
| MA | patients with migraine with aura between attacks |
| MI | patients with migraine recorded during an attack |
| MO | patients with migraine without aura between attacks |
| PS | photostress |
| VEP | visual evoked potential |
References
- Noseda, R.; Bernstein, C.A.; Nir, R.-R.; Lee, A.J.; Fulton, A.B.; Bertisch, S.M.; Hovaguimian, A.; Cestari, D.M.; Saavedra-Walker, R.; Borsook, D.; et al. Migraine photophobia originating in cone-driven retinal pathways. Brain 2016, 139, 1971–1986. [Google Scholar] [CrossRef] [PubMed]
- Coppola, G.; Parisi, V.; Di Lorenzo, C.; Serrao, M.; Magis, D.; Schoenen, J.; Pierelli, F. Lateral inhibition in visual cortex of migraine patients between attacks. J. Headache Pain 2013, 14, 20. [Google Scholar] [CrossRef]
- Datta, R.; Aguirre, G.K.; Hu, S.; Detre, J.A.; Cucchiara, B. Interictal cortical hyperresponsiveness in migraine is directly related to the presence of aura. Cephalalgia 2013, 33, 365–374. [Google Scholar] [CrossRef] [PubMed]
- Lisicki, M.; D’Ostilio, K.; Coppola, G.; De Noordhout, A.M.; Parisi, V.; Schoenen, J.; Magis, D. Brain correlates of single trial visual evoked potentials in migraine: More than meets the eye. Front. Neurol. 2018, 9, 393. [Google Scholar] [CrossRef]
- Granziera, C.; DaSilva, A.F.M.; Snyder, J.; Tuch, D.S.; Hadjikhani, N. Anatomical alterations of the visual motion processing network in migraine with and without aura. PLoS Med. 2006, 3, e402. [Google Scholar] [CrossRef]
- Rocca, M.A.; Pagani, E.; Colombo, B.; Tortorella, P.; Falini, A.; Comi, G.; Filippi, M. Selective diffusion changes of the visual pathways in patients with migraine: A 3-T tractography study. Cephalalgia 2008, 28, 1061–1068. [Google Scholar] [CrossRef]
- Granziera, C.; Daducci, A.; Romascano, D.; Roche, A.; Helms, G.; Krueger, G.; Hadjikhani, N. Structural abnormalities in the thalamus of migraineurs with aura: A multiparametric study at 3 T. Hum. Brain Mapp. 2013, 35, 1461–1468. [Google Scholar] [CrossRef]
- Shepherd, A.J. Colour vision in migraine: Selective deficits for S-Cone discriminations. Cephalalgia 2005, 25, 412–423. [Google Scholar] [CrossRef]
- Shepherd, A.J. Color vision but not visual attention is altered in migraine. Headache J. Head Face Pain 2006, 46, 611–621. [Google Scholar] [CrossRef] [PubMed]
- Main, A.; Vlachonikolis, I.; Dowson, A. The wavelength of light causing photophobia in migraine and tension-type headache between attacks. Headache J. Head Face Pain 2000, 40, 194–199. [Google Scholar] [CrossRef]
- Good, P.; Taylor, R.; Mortimer, M. The use of tinted glasses in childhood migraine. Headache J. Head Face Pain 1991, 31, 533–536. [Google Scholar] [CrossRef] [PubMed]
- Wilkins, A.J.; Patel, R.; Adjamian, P.; Evans, B.J.W. Tinted spectacles and visually sensitive migraine. Cephalalgia 2002, 22, 711–719. [Google Scholar] [CrossRef]
- Afra, J.; Mascia, A.; Genicot, R.; Albert, A.; Schoenen, J. Influence of colors on habituation of visual evoked potentials in patients with migraine with aura and in healthy volunteers. Headache J. Head Face Pain 2000, 40, 36–40. [Google Scholar] [CrossRef]
- Noseda, R.; Kainz, V.; Jakubowski, M.; Gooley, J.J.; Saper, C.B.; Digre, K.B.; Burstein, R. A neural mechanism for exacerbation of headache by light. Nat. Neurosci. 2010, 13, 239–245. [Google Scholar] [CrossRef]
- McAdams, H.; Kaiser, E.A.; Igdalova, A.; Haggerty, E.B.; Cucchiara, B.; Brainard, D.H.; Aguirre, G.K. Selective amplification of ipRGC signals accounts for interictal photophobia in migraine. Proc. Natl. Acad. Sci. USA 2020, 117, 17320–17329. [Google Scholar] [CrossRef] [PubMed]
- Severin, S.L.; Tour, R.L.; Kershaw, R.H. Macular function and the photostress test 1. Arch. Ophthalmol. 1967, 77, 2–7. [Google Scholar] [CrossRef] [PubMed]
- Severin, S.L.; Tour, R.L.; Kershaw, R.H. Macular function and the photostress Test 2. Arch. Ophthalmol. 1967, 77, 163–167. [Google Scholar] [CrossRef]
- Parisi, V. Electrophysiological evaluation of the macular cone adaptation: VEP after photostress. A review. Doc. Ophthalmol. 2001, 102, 251–262. [Google Scholar] [CrossRef] [PubMed]
- Lovasik, J.V. An electrophysiological investigation of the macular photostress test. Investig. Ophthalmol. Vis. Sci. 1983, 24, 437–441. [Google Scholar]
- Parisi, V.; Uccioli, L. Visual electrophysiological responses in persons with type 1 diabetes. Diabetes/Metab. Res. Rev. 2001, 17, 12–18. [Google Scholar] [CrossRef]
- Parisi, V.; Bucci, M.G. Visual evoked potentials after photostress in patients with primary open-angle glaucoma and ocular hypertension. Investig. Ophthalmol. Vis. Sci. 1992, 33, 436–442. [Google Scholar]
- Parisi, V.; Pierelli, F.; Restuccia, R.; Spadaro, M.; Parisi, L.; Colacino, G.; Bucci, M.G. Impaired VEP after photostress response in multiple sclerosis patients previously affected by optic neuritis. Electroencephalogr. Clin. Neurophysiol. 1998, 108, 73–79. [Google Scholar] [CrossRef]
- Afra, J.; Proietti Cecchini, A.; Sándor, P.S.; Schoenen, J. Comparison of visual and auditory evoked cortical potentials in migraine patients between attacks. Clin. Neurophysiol. 2000, 111, 1124–1129. [Google Scholar] [CrossRef]
- Parisi, V.; Canu, D.; Iarossi, G.; Olzi, D.; Falsini, B. Altered recovery of macular function after bleaching in Stargardt’s disease-fundus flavimaculatus: Pattern VEP evidence. Investig. Ophthalmol. Vis. Sci. 2002, 43, 2741–2748. [Google Scholar]
- Odom, J.V.; International Society for Clinical Electrophysiology of Vision; Bach, M.; Brigell, M.; Holder, G.E.; McCulloch, D.L.; Mizota, A.; Tormene, A.P. ISCEV standard for clinical visual evoked potentials: (2016 update). Doc. Ophthalmol. 2016, 133, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, P.; Boynton, R. Bleaching and regeneration kinetics. In Human Color Vision; Kaiser, P., Boynton, R., Eds.; Optical Society of America: Washington, DC, USA, 1996; pp. 208–214. [Google Scholar]
- Futterman, S. Metabolism of the retina. III. The role of reduced triphoshopyridine nucleotide in the visual cycle. J. Biol. Chem. 1963, 238, 1145–1150. [Google Scholar] [CrossRef]
- Parisi, V.; Falsini, B. Electrophysiological evaluation of the macular cone system: Focal electroretinography and visual evoked potentials after photostress. Semin. Ophthalmol. 1998, 13, 178–188. [Google Scholar] [CrossRef]
- Tak, A.Z.A.; Sengul, Y.; Bilak, Ş. Evaluation of white matter hyperintensities and retinal fiber layer, ganglion cell layer, inner-plexiform layer, and choroidal layer in migraine patients. Neurol. Sci. 2018, 39, 489–496. [Google Scholar] [CrossRef] [PubMed]
- Reggio, E.; Chisari, C.G.; Ferrigno, G.; Patti, F.; Donzuso, G.; Sciacca, G.; Avitabile, T.; Faro, S.; Zappia, M. Migraine causes retinal and choroidal structural changes: Evaluation with ocular coherence tomography. J. Neurol. 2016, 264, 494–502. [Google Scholar] [CrossRef]
- Can, G.D.; Can, M.E.; Ekici, A. Evaluation of retinal microvasculature and foveal avascular zone by the optical coherence tomography angiography in pediatric migraine patients. Acta Neurol. Belg. 2020, 1–7. [Google Scholar] [CrossRef]
- Rushton, W.A.H.; Baker, H.D. Effect of a very bright flash on cone vision and cone pigments in man. Nature 1963, 200, 421–423. [Google Scholar] [CrossRef]
- Main, A.; Dowson, A.; Gross, M. Photophobia and phonophobia in migraineurs between attacks. Headache J. Head Face Pain 1997, 37, 492–495. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, K.; Tashiro, A.; Chang, Z.; Bereiter, D.A. Bright light activates a trigeminal nociceptive pathway. Pain 2010, 149, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Mainster, M.A.; Turner, P.L. Glare’s causes, consequences, and clinical challenges after a century of ophthalmic study. Am. J. Ophthalmol. 2012, 153, 587–593. [Google Scholar] [CrossRef] [PubMed]
- Du Pasquier, R.A.; Genoud, D.; Safran, A.B.; Landis, T. Monocular central dazzle after thalamic infarcts. J. Neuro-Ophthalmol. 2000, 20, 97–99. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Coppola, G.; Vandenheede, M.; Di Clemente, L.; Ambrosini, A.; Fumal, A.; De Pasqua, V.; Schoenen, J. Somatosensory evoked high-frequency oscillations reflecting thalamo-cortical activity are decreased in migraine patients between attacks. Brain 2004, 128, 98–103. [Google Scholar] [CrossRef]
- Porcaro, C.; Di Lorenzo, G.; Seri, S.; Pierelli, F.; Tecchio, F.; Coppola, G. Impaired brainstem and thalamic high-frequency oscillatory EEG activity in migraine between attacks. Cephalalgia 2016, 37, 915–926. [Google Scholar] [CrossRef]
- Coppola, G.; Ambrosini, A.; Di Clemente, L.; Magis, D.; Fumal, A.; Gerard, P.; Pierelli, F.; Schoenen, J. Interictal abnormalities of gamma band activity in visual evoked responses in migraine: An indication of thalamocortical dysrhythmia? Cephalalgia 2007, 27, 1360–1367. [Google Scholar] [CrossRef]
- Lisicki, M.; D’Ostilio, K.; Coppola, G.; Nonis, R.; de Noordhout, A.M.; Parisi, V.; Magis, D.; Schoenen, J. Headache related alterations of visual processing in migraine patients. J. Pain 2020, 21, 593–602. [Google Scholar] [CrossRef]
- Coppola, G.; Bracaglia, M.; Di Lenola, D.; Iacovelli, E.; Di Lorenzo, C.; Serrao, M.; Evangelista, M.; Parisi, V.; Schoenen, J.; Pierelli, F. Lateral inhibition in the somatosensory cortex during and between migraine without aura attacks: Correlations with thalamocortical activity and clinical features. Cephalalgia 2015, 36, 568–578. [Google Scholar] [CrossRef] [PubMed]
- Strigaro, G.; Cerino, A.; Falletta, L.; Mittino, D.; Comi, C.; Varrasi, C.; Cantello, R. Impaired visual inhibition in migraine with aura. Clin. Neurophysiol. 2015, 126, 1988–1993. [Google Scholar] [CrossRef]
- Höffken, O.; Stude, P.; Lenz, M.; Bach, M.; Dinse, H.R.; Tegenthoff, M. Visual paired-pulse stimulation reveals enhanced visual cortex excitability in migraineurs. Eur. J. Neurosci. 2009, 30, 714–720. [Google Scholar] [CrossRef]
- Coppola, G.; Crémers, J.; Gérard, P.; Pierelli, F.; Schoenen, J. Effects of light deprivation on visual evoked potentials in migraine without aura. BMC Neurol. 2011, 11, 91. [Google Scholar] [CrossRef] [PubMed]
- Coppola, G.; Currà, A.; Serrao, M.; Di Lorenzo, C.; Gorini, M.; Porretta, E.; Alibardi, A.; Parisi, V.; Pierelli, F. Lack of cold pressor test-induced effect on visual-evoked potentials in migraine. J. Headache Pain 2009, 11, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Abagnale, C.; Ranieri, F.; Di Renzo, A.; Parisi, V.; Serrao, M.; Di Lazzaro, V.; Lisicki, M.; Coppola, G.; Pierelli, F. Impaired short-term visual paired associative plasticity in patients with migraine between attacks. Pain 2020. [Google Scholar] [CrossRef] [PubMed]
- Llinás, R.R.; Ribary, U.; Jeanmonod, D.; Kronberg, E.; Mitra, P.P. Thalamocortical dysrhythmia: A neurological and neuropsychiatric syndrome characterized by magnetoencephalography. Proc. Natl. Acad. Sci. USA 1999, 96, 15222–15227. [Google Scholar] [CrossRef] [PubMed]
- Coppola, G.; Tinelli, E.; Lepre, C.; Iacovelli, E.; Di Lorenzo, C.; Serrao, M.; Pauri, F.; Fiermonte, G.; Bianco, F.; Pierelli, F. Dynamic changes in thalamic microstructure of migraine without aura patients: A diffusion tensor magnetic resonance imaging study. Eur. J. Neurol. 2013, 21, 287-e13. [Google Scholar] [CrossRef]
- Coppola, G.; Di Renzo, A.; Tinelli, E.; Lepre, C.; Di Lorenzo, C.; Di Lorenzo, G.; Scapeccia, M.; Parisi, V.; Serrao, M.; Colonnese, C.; et al. Thalamo-cortical network activity between migraine attacks: Insights from MRI-based microstructural and functional resting-state network correlation analysis. J. Headache Pain 2016, 17, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.-L.; Zhou, X.; Chen, Y.-C.; Yu, Y.-S.; Guo, X.; Zhou, G.-P.; Zhou, Q.-Q.; Qu, L.-J.; Yin, X.; Li, J.; et al. Impaired intrinsic functional connectivity between the thalamus and visual cortex in migraine without aura. J. Headache Pain 2019, 20, 116. [Google Scholar] [CrossRef] [PubMed]
- Shin, K.J.; Lee, H.-J.; Park, K.M. Alterations of individual thalamic nuclei volumes in patients with migraine. J. Headache Pain 2019, 20, 112. [Google Scholar] [CrossRef] [PubMed]
- Tu, Y.; Fu, Z.; Zeng, F.; Maleki, N.; Lan, L.; Li, Z.; Park, J.; Wilson, G.; Gao, Y.; Liu, M.; et al. Abnormal thalamocortical network dynamics in migraine. Neurology 2019, 92, e2706–e2716. [Google Scholar] [CrossRef] [PubMed]
- Marciszewski, K.K.; Meylakh, N.; Di Pietro, F.; Macefield, V.G.; Macey, P.M.; Henderson, L.A. Fluctuating regional brainstem diffusion imaging measures of microstructure across the migraine cycle. eNeuro 2019, 6, ENEURO.0005-19.2019. [Google Scholar] [CrossRef] [PubMed]
- Coppola, G.; Bracaglia, M.; Di Lenola, D.; Di Lorenzo, C.; Serrao, M.; Parisi, V.; Di Renzo, A.; Martelli, F.; Fadda, A.; Schoenen, J.; et al. Visual evoked potentials in subgroups of migraine with aura patients. J. Headache Pain 2015, 16, 92. [Google Scholar] [CrossRef] [PubMed]
- Mehnert, J.; Bader, D.; Nolte, G.; May, A. Visual input drives increased occipital responsiveness and harmonized oscillations in multiple cortical areas in migraineurs. NeuroImage Clin. 2019, 23, 101815. [Google Scholar] [CrossRef] [PubMed]
- Cortese, F.; Coppola, G.; Di Lenola, D.; Serrao, M.; Di Lorenzo, C.; Parisi, V.; Pierelli, F. Excitability of the motor cortex in patients with migraine changes with the time elapsed from the last attack. J. Headache Pain 2017, 18, 2. [Google Scholar] [CrossRef]
- Stankewitz, A.; Aderjan, D.; Eippert, F.; May, A. Trigeminal nociceptive transmission in migraineurs predicts migraine attacks. J. Neurosci. 2011, 31, 1937–1943. [Google Scholar] [CrossRef] [PubMed]
- Deen, M.; Hansen, H.D.; Hougaard, A.; Da Cunha-Bang, S.; Nørgaard, M.; Svarer, C.; Keller, S.H.; Thomsen, C.; Ashina, M.; Knudsen, G.M. Low 5-HT1B receptor binding in the migraine brain: A PET study. Cephalalgia 2017, 38, 519–527. [Google Scholar] [CrossRef] [PubMed]
- Schulte, L.H.; May, A. The migraine generator revisited: Continuous scanning of the migraine cycle over 30 days and three spontaneous attacks. Brain 2016, 139, 1987–1993. [Google Scholar] [CrossRef]
- Maniyar, F.H.; Sprenger, T.; Monteith, T.; Schankin, C.; Goadsby, P.J. Brain activations in the premonitory phase of nitroglycerin-triggered migraine attacks. Brain 2014, 137, 232–241. [Google Scholar] [CrossRef]
- Schulte, L.H.; Mehnert, J.; May, A. Longitudinal neuroimaging over 30 days: Temporal characteristics of migraine. Ann. Neurol. 2020, 87, 646–651. [Google Scholar] [CrossRef]
- Matthews, G.A.; Tye, K.M. Neural mechanisms of social homeostasis. Ann. N. Y. Acad. Sci. 2019, 1457, 5–25. [Google Scholar] [CrossRef] [PubMed]
- Coppola, G.; Pierelli, F.; Schoenen, J.; Wang, S.-J.; Chen, W.-T. Neurophysiological Model of Migraine Pathophysiology: Bringing the Past into the Future; Springer: Cham, Switzerland, 2021; pp. 223–236. [Google Scholar]
- Kamali, A.; Karbasian, N.; Sherbaf, F.G.; Wilken, L.A.; Aein, A.; Sair, H.I.; Espejo, O.A.; Rabiei, P.; Choi, S.J.; Mirbagheri, S.; et al. Uncovering the dorsal thalamo-hypothalamic tract of the human limbic system. Neuroscience 2020, 432, 55–62. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | HV (n = 14) | MO (n = 22) | MA (n = 19) | MI (n = 10) |
|---|---|---|---|---|
| Women (n) | 10 | 16 | 12 | 8 |
| Age (years) | 29.7 ± 6.1 | 29.2 ± 8.2 | 30.3 ± 10.2 | 32.7 ± 11.7 |
| Duration of migraine history (years) | 17.3 ± 9.7 | 18.1 ± 11.5 | 15.6 ± 7.6 | |
| Attack frequency/month (n) | 2.4 ± 1.6 | 2.5 ± 1.5 | 2.1 ± 1.8 | |
| Attack duration (hours) | 33.2 ± 26.6 | 24.9 ± 26.5 | 40.7 ± 27.6 | |
| N° of days since the last attack | 17.4 ± 17.2 | 14.4 ± 12.5 | ||
| Scintillating scotoma/Fortification spectra | 100% | |||
| Sensory symptoms | 36.8% | |||
| Language/Speech symptoms | 15.8% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Coppola, G.; Di Lorenzo, C.; Di Lenola, D.; Serrao, M.; Pierelli, F.; Parisi, V. Visual Evoked Potential Responses after Photostress in Migraine Patients and Their Correlations with Clinical Features. J. Clin. Med. 2021, 10, 982. https://doi.org/10.3390/jcm10050982
Coppola G, Di Lorenzo C, Di Lenola D, Serrao M, Pierelli F, Parisi V. Visual Evoked Potential Responses after Photostress in Migraine Patients and Their Correlations with Clinical Features. Journal of Clinical Medicine. 2021; 10(5):982. https://doi.org/10.3390/jcm10050982
Chicago/Turabian StyleCoppola, Gianluca, Cherubino Di Lorenzo, Davide Di Lenola, Mariano Serrao, Francesco Pierelli, and Vincenzo Parisi. 2021. "Visual Evoked Potential Responses after Photostress in Migraine Patients and Their Correlations with Clinical Features" Journal of Clinical Medicine 10, no. 5: 982. https://doi.org/10.3390/jcm10050982
APA StyleCoppola, G., Di Lorenzo, C., Di Lenola, D., Serrao, M., Pierelli, F., & Parisi, V. (2021). Visual Evoked Potential Responses after Photostress in Migraine Patients and Their Correlations with Clinical Features. Journal of Clinical Medicine, 10(5), 982. https://doi.org/10.3390/jcm10050982









