The Impact of Diagnostic Criteria for Gestational Diabetes Mellitus on Adverse Maternal Outcomes: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
- To study the pooled risk of adverse maternal outcomes among pregnant women with GDM compared to non-GDM counterparts, regardless of diagnostic criteria;
- To study the pooled risk of adverse maternal outcomes among pregnant women with GDM compared to non-GDM women, according to the various diagnostic criteria;
- To study the association between adverse maternal outcomes and GDM criteria.
2.1. Eligibility Criteria
2.2. Search Strategy
2.3. Study Selection and Data Extraction
2.4. Study Subgroups and Outcomes of Study
- (i)
- IADPSG criteria, one step screening with oral glucose tolerance test (2 h, 75 g GTT); GDM diagnosis: any of the given values are met or exceeded (fasting: 92 mg/dL, BS-1 h: 180 mg/dL, BS-2 h: 153 mg/dL);
- (ii)
- One step screening with 2 h, 75 g OGTT. GDM diagnosis: any of the given valued are met or exceeded (fasting 100 mg/dL, 2 h: 144 mg/dL;
- (iii)
- One step screening with 2 h, 75 g OGTT. GDM diagnosis: any of the given valued are met or exceeded (fasting: 110 mg/dL, 2 h: 140 mg/dL);
- (iv)
- Group 4, one step screening with 2 h, 75 g OGTT. GDM diagnosis: any of the given values are met or exceeded (fasting 100 mg/dL, BS 2 h: 162 mg/dL);
- (v)
- Two step screening with 1 h-50 g Glucose challenge test (1 h-50 g-GCT), values > 140 mg/dL following 100 g OGTT. GDM diagnosis: two values are met or exceeded (fasting: 95 mg/dL, BS-1 h: 180 mg/dL, BS-2 h: 155 mg/dL, BS-3 h: 140 mg/dL or two step screening with 1 h-50 g-GCT, values > 140 mg/dL following 75 g OGTT. GDM diagnosis: two values are met or exceeded (fasting: 95 mg/dL, BS-1 h: 180 mg/dL, BS-2 h: 155 mg/dL, BS-3 h: 140 mg/dL);
- (vi)
- Two step screening with 1 h-50 g-GCT, values > 140 mg/dL following 100 g OGTT. GDM diagnosis: two values are met or exceeded (fasting: 105 mg/dL, BS-1 h: 155 mg/dL, BS-2 h: 165 mg/dL, BS-1 h: 145 mg/dL);
- (vii)
- One step screening with 75 g OGTT. GDM diagnosis: any of the given valued are met or exceeded (fasting: 128 mg/dl, BS2 h: 140 mg/dl).
2.5. Statistical Analysis
3. Results
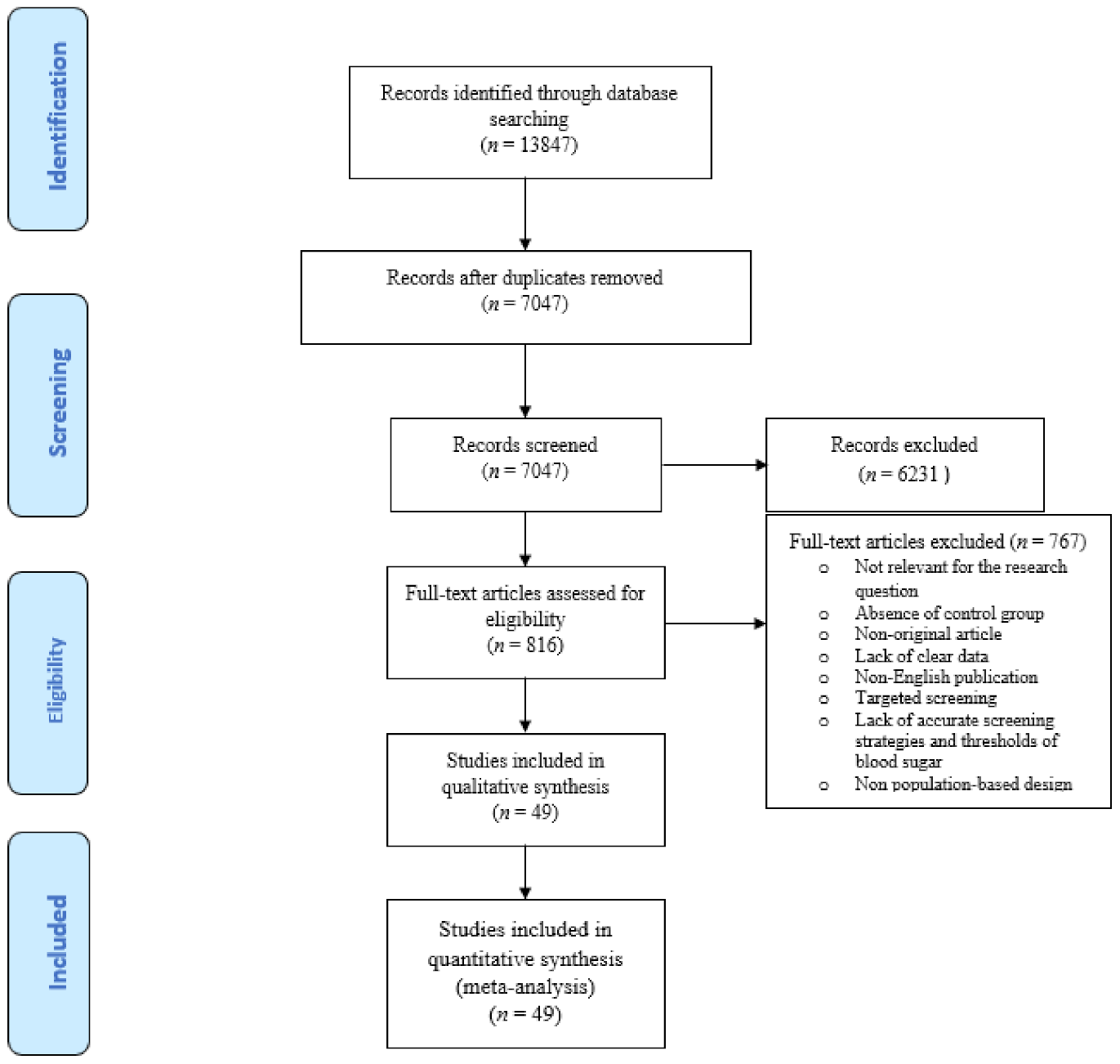
3.1. Literature Search Results and Quality assessment
3.2. Meta-Analysis and Meta-Regression Results
3.3. Results of Publication Bias and Risk of Bias evaluation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- American Diabetes Association. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes—2020. Diabetes Care 2020, 43, S14–S31. [Google Scholar] [CrossRef]
- Behboudi-Gandevani, S.; Amiri, M.; Yarandi, R.B.; Tehrani, F.R. The impact of diagnostic criteria for gestational diabetes on its prevalence: A systematic review and meta-analysis. Diabetol. Metab. Syndr. 2019, 11, 1–18. [Google Scholar] [CrossRef]
- Gabbay-Benziv, R.; Doyle, L.E.; Blitzer, M.; Baschat, A.A. First trimester prediction of maternal glycemic status. J. Périnat. Med. 2015, 43, 283–289. [Google Scholar] [CrossRef]
- Giannakou, K.; Evangelou, E.; Yiallouros, P.; Christophi, C.A.; Middleton, N.; Papatheodorou, E.; Papatheodorou, S.I. Risk factors for gestational diabetes: An umbrella review of meta-analyses of observational studies. PLoS ONE 2019, 14, e0215372. [Google Scholar] [CrossRef]
- Plows, J.F.; Stanley, J.L.; Baker, P.; Reynolds, C.M.; Vickers, M.H. The Pathophysiology of Gestational Diabetes Mellitus. Int. J. Mol. Sci. 2018, 19, 3342. [Google Scholar] [CrossRef]
- Morikawa, M.; Sugiyama, T.; Sagawa, N.; Hiramatsu, Y.; Ishikawa, H.; Hamada, H.; Kameda, T.; Hara, E.; Toda, S.; Minakami, H. Perinatal mortality in Japanese women diagnosed with gestational diabetes mellitus and diabetes mellitus. J. Obstet. Gynaecol. Res. 2017, 43, 1700–1707. [Google Scholar] [CrossRef] [PubMed]
- Gorgal, R.; Gonçalves, E.; Barros, M.; Namora, G.; Magalhães, Â.; Rodrigues, T.; Montenegro, N. Gestational diabetes mellitus: A risk factor for non-elective cesarean section. J. Obstet. Gynaecol. Res. 2011, 38, 154–159. [Google Scholar] [CrossRef]
- Corrado, F.; D’Anna, R.; Laganà, A.S.; Di Benedetto, A. Abnormal glucose tolerance later in life in women affected by glucose intolerance during pregnancy. J. Obstet. Gynaecol. 2014, 34, 123–126. [Google Scholar] [CrossRef] [PubMed]
- Vitagliano, A.; Saccone, G.; Cosmi, E.; Visentin, S.; Dessole, F.; Ambrosini, G.; Berghella, V. Inositol for the prevention of gestational diabetes: A systematic review and meta-analysis of randomized controlled trials. Arch. Gynecol. Obstet. 2019, 299, 55–68. [Google Scholar] [CrossRef] [PubMed]
- Facchinetti, F.; Appetecchia, M.; Aragona, C.; Bevilacqua, A.; Espinola, M.S.B.; Bizzarri, M.; D’Anna, R.; Dewailly, D.; Diamanti-Kandarakis, E.; Marín, I.H.; et al. Experts’ opinion on inositols in treating polycystic ovary syndrome and non-insulin dependent diabetes mellitus: A further help for human reproduction and beyond. Expert Opin. Drug Metab. Toxicol. 2020, 16, 255–274. [Google Scholar] [CrossRef]
- Jacklin, P.B.; Maresh, M.J.; Patterson, C.C.; Stanley, K.P.; Dornhorst, A.; Burman-Roy, S.; Bilous, R.W. A cost-effectiveness comparison of the NICE 2015 and WHO 2013 diagnostic criteria for women with gestational diabetes with and without risk factors. BMJ Open 2017, 7, e016621. [Google Scholar] [CrossRef] [PubMed]
- Metzger, B.E.; Lowe, L.; Dyer, A.; Trimble, E.; Chaovarindr, U.; Coustan, D.; Hadden, D.; McCance, D.; Hod, M.; McIntyre, H.; et al. Hyperglycemia and Adverse Pregnancy Outcomes. Obstet. Anesth. Dig. 2009, 29, 39–40. [Google Scholar] [CrossRef]
- International Association of Diabetes and Pregnancy Study Groups Consensus Panel. International association of diabetes and pregnancy study groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy. Diabetes Care 2010, 33, 676–682. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Diagnostic Criteria and Classification of Hyperglycaemia First Detected in Pregnancy; World Health Organization: Geneva, Switzerland, 2013. [Google Scholar]
- Hosseini, E.; Janghorbani, M. Systematic review and meta-analysis of diagnosing gestational diabetes mellitus with one-step or two-step approaches and associations with adverse pregnancy outcomes. Int. J. Gynaecol. Obstet. 2018, 143, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Wong, C.S. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Int. J. Sur. 2010, 8, 336–341. [Google Scholar] [CrossRef]
- Stang, A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur. J. Epidemiol. 2010, 25, 603–605. [Google Scholar] [CrossRef] [PubMed]
- Sterne, J.A.; Hernán, M.A.; Reeves, B.C.; Savović, J.; Berkman, N.D.; Viswanathan, M.; Henry, D.; Altman, D.G.; Ansari, M.T.; Boutron, I.; et al. ROBINS-I: A tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016, 355, i4919. [Google Scholar] [CrossRef]
- Higgins, J. Analysing data and undertaking meta-analyses In Cochrane Handbook for Systematic Reviews of Interventions (Version 5.1. 0); Higgins, J., Green, S., Eds.; Wiley: New York, NY, USA, 2011. [Google Scholar]
- Shand, A.W.; Bell, J.C.; McElduff, A.; Morris, J.; Roberts, C.L. Outcomes of pregnancies in women with pre-gestational diabetes mellitus and gestational diabetes mellitus; a population-based study in New South Wales, Australia, 19982002. Diabet. Med. 2008, 25, 708–715. [Google Scholar] [CrossRef] [PubMed]
- Erjavec, K.; Poljičanin, T.; Matijević, R. Impact of the Implementation of New WHO Diagnostic Criteria for Gestational Diabetes Mellitus on Prevalence and Perinatal Outcomes: A Population-Based Study. J. Pregnancy 2016, 2016, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Black, M.H.; Sacks, D.A.; Xiang, A.H.; Lawrence, J.M. Clinical Outcomes of Pregnancies Complicated by Mild Gestational Diabetes Mellitus Differ by Combinations of Abnormal Oral Glucose Tolerance Test Values. Diabetes Care 2010, 33, 2524–2530. [Google Scholar] [CrossRef]
- Lamminpää, R.; Vehviläinen-Julkunen, K.; Gissler, M.; Gissler, M.; Selander, T.; Heinonen, S. Pregnancy outcomes in women aged 35 years or older with gestational diabetes—A registry-based study in Finland. J. Matern. Fetal Neonatal Med. 2016, 29, 55–59. [Google Scholar] [CrossRef] [PubMed]
- Carr, D.B.; Newton, K.M.; Utzschneider, K.M.; Faulenbach, M.V.; Kahn, S.E.; Easterling, T.R.; Heckbert, S.R. Gestational Diabetes or Lesser Degrees of Glucose Intolerance and Risk of Preeclampsia. Hypertens. Pregnancy 2010, 30, 153–163. [Google Scholar] [CrossRef]
- Metcalfe, A.; Sabr, Y.; Hutcheon, J.A.; Donovan, L.; Lyons, J.; Burrows, J.; Joseph, K.S. Trends in Obstetric Intervention and Pregnancy Outcomes of Canadian Women with Diabetes in Pregnancy From 2004 to 2015. J. Endocr. Soc. 2017, 1, 1540–1549. [Google Scholar] [CrossRef] [PubMed]
- Su, W.-J.; Chen, Y.-L.; Huang, P.-Y.; Shi, X.-L.; Yan, F.-F.; Chen, Z.; Yan, B.; Song, H.-Q.; Lin, M.-Z.; Li, X. Effects of Prepregnancy Body Mass Index, Weight Gain, and Gestational Diabetes Mellitus on Pregnancy Outcomes: A Population-Based Study in Xiamen, China, 2011–2018. Ann. Nutr. Metab. 2019, 75, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Van Hoorn, J.; Dekker, G.; Jeffries, B. Gestational diabetes versus obesity as risk factors for pregnancy-induced hypertensive disorders and fetal macrosomia. Aust. N. Z. J. Obstet. Gynaecol. 2002, 42, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Zeki, R.; Oats, J.J.; Wang, A.Y.; Li, Z.; Homer, C.S.E.; Sullivan, E.A. Cesarean section and diabetes during pregnancy: An NSW population study using the Robson classification. J. Obstet. Gynaecol. Res. 2018, 44, 890–898. [Google Scholar] [CrossRef]
- Sugaya, A.; Sugiyama, T.; Nagata, M.; Toyoda, N. Comparison of the validity of the criteria for gestational diabetes mellitus by WHO and by the Japan Society of Obstetrics and Gynecology by the outcomes of pregnancy. Diabetes Res. Clin. Pr. 2000, 50, 57–63. [Google Scholar] [CrossRef]
- Sletner, L.; Jenum, A.K.; Yajnik, C.S.; Mørkrid, K.; Nakstad, B.; Rognerud-Jensen, O.H.; Birkeland, K.I.; Vangen, S. Fetal growth trajectories in pregnancies of European and South Asian mothers with and without gestational diabetes, a population-based cohort study. PLoS ONE 2017, 12, e0172946. [Google Scholar] [CrossRef]
- Ellerbe, C.N.; Gebregziabher, M.; Korte, J.E.; Mauldin, J.; Hunt, K.J. Quantifying the Impact of Gestational Diabetes Mellitus, Maternal Weight and Race on Birthweight via Quantile Regression. PLoS ONE 2013, 8, e65017. [Google Scholar] [CrossRef]
- Mahanta, T.G.; Deuri, A.; Mahanta, B.N.; Bordoloi, P.; Rasaily, R.; Mahanta, J.; Baruah, S.; Gogoi, P. Maternal and foetal outcome of gestational diabetes mellitus in a rural block of Assam, India. Clin. Epidemiol. Glob. Health 2014, 2, 9–15. [Google Scholar] [CrossRef]
- Kieffer, E.C.; Tabaei, B.P.; Carman, W.J.; Nolan, G.H.; Guzman, J.R.; Herman, W.H. The Influence of Maternal Weight and Glucose Tolerance on Infant Birthweight in Latino Mother–Infant Pairs. Am. J. Public Health 2006, 96, 2201–2208. [Google Scholar] [CrossRef] [PubMed]
- Nerenberg, K.A.; Johnson, J.A.; Leung, B.; Savu, A.; Ryan, E.A.; Chik, C.L.; Kaul, P. Risks of Gestational Diabetes and Preeclampsia Over the Last Decade in a Cohort of Alberta Women. J. Obstet. Gynaecol. Can. 2013, 35, 986–994. [Google Scholar] [CrossRef]
- Soliman, A.; Salama, H.; Al Rifai, H.; De Sanctis, V.; Al-Obaidly, S.; Al Qubasi, M.; Olukade, T. The effect of different forms of dysglycemia during pregnancy on maternal and fetal outcomes in treated women and comparison with large cohort studies. Acta Biomed. Atenei Parm. 2018, 89, 11–21. [Google Scholar]
- Oster, R.T.; King, M.; Morrish, D.W.; Mayan, M.; Toth, E.L. Diabetes in pregnancy among First Nations women in Alberta, Canada: A retrospective analysis. BMC Pregnancy Childbirth 2014, 14, 136. [Google Scholar] [CrossRef]
- Xiong, X.; Saunders, L.; Wang, F.; Demianczuk, N. Gestational diabetes mellitus: Prevalence, risk factors, maternal and infant outcomes. Int. J. Gynecol. Obstet. 2001, 75, 221–228. [Google Scholar] [CrossRef]
- Sacks, D.A.; Black, M.H.; Li, X.; Montoro, M.N.; Lawrence, J.M. Adverse pregnancy outcomes using the International Association of the Diabetes and Pregnancy Study Groups criteria: Glycemic thresholds and associated risks. Obstet. Gynecol. 2015, 126, 67–73. [Google Scholar] [CrossRef]
- Son, K.H.; Lim, N.K.; Lee, J.; Cho, M.; Park, H. Comparison of maternal morbidity and medical costs during pregnancy and delivery between patients with gestational diabetes and patients with pre-existing diabetes. Diabet. Med. 2015, 32, 477–486. [Google Scholar] [CrossRef]
- Pan, L.; Leng, J.; Liu, G.; Zhang, C.; Liu, H.; Li, M.; Tan, L.; Tian, H.; Chan, J.C.; Hu, G.; et al. Pregnancy outcomes of Chinese women with gestational diabetes mellitus defined by the IADPSG’s but not by the 1999 WHO’s criteria. Clin. Endocrinol. 2015, 83, 684–693. [Google Scholar] [CrossRef]
- Jacobson, J.D.; Cousins, L. A population-based study of maternal and perinatal outcome in patients with gestational diabetes. Am. J. Obstet. Gynecol. 1989, 161, 981–986. [Google Scholar] [CrossRef]
- Leybovitz-Haleluya, N.; Wainstock, T.; Landau, D.; Sheiner, E. Maternal gestational diabetes mellitus and the risk of subsequent pediatric cardiovascular diseases of the offspring: A population-based cohort study with up to 18 years of follow up. Acta Diabetol. 2018, 55, 1037–1042. [Google Scholar] [CrossRef] [PubMed]
- Gortazar, L.; Roux, J.A.F.-L.; Benaiges, D.; Sarsanedas, E.; Payà, A.; Mañé, L.; Pedro-Botet, J.; Goday, A. Trends in prevalence of gestational diabetes and perinatal outcomes in Catalonia, Spain, 2006 to 2015: The Diagestcat Study. Diabetes Metabol. Res. Rev. 2019, 35, e3151. [Google Scholar] [CrossRef] [PubMed]
- Hedderson, M.M.; Ferrara, A.; Sacks, D.A. Gestational diabetes mellitus and lesser degrees of pregnancy hyperglycemia: Association with increased risk of spontaneous preterm birth. Obstet. Gynecol. 2003, 102, 850–856. [Google Scholar] [CrossRef]
- Kun, A. Insulin Resistance Is Associated with Gestational Hypertension and Not with Preeclampsia: A Population-Based Screening Study. Gynecol. Obstet. Investig. 2011, 71, 256–261. [Google Scholar] [CrossRef]
- Davey, S.; Jain, R.; Davey, A.; Raghav, S.K.; Singh, J.V. Can the management of blood sugar levels in gestational diabetes mellitus cases be an indicator of maternal and fetal outcomes? The results of a prospective cohort study from India. J. Fam. Community Med. 2016, 23, 94–99. [Google Scholar] [CrossRef]
- Hosseini, E.; Janghorbani, M.; Aminorroaya, A. Incidence, risk factors, and pregnancy outcomes of gestational diabetes mellitus using one-step versus two-step diagnostic approaches: A population-based cohort study in Isfahan, Iran. Diabetes Res. Clin. Pr. 2018, 140, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, E.; Janghorbani, M.; Shahshahan, Z. Comparison of risk factors and pregnancy outcomes of gestational diabetes mellitus diagnosed during early and late pregnancy. Midwifery 2018, 66, 64–69. [Google Scholar] [CrossRef]
- Zamstein, O.; Sheiner, E.; Wainstock, T.; Landau, D.; Walfisch, A. Maternal gestational diabetes and long-term respiratory related hospitalizations of the offspring. Diabetes Res. Clin. Pr. 2018, 140, 200–207. [Google Scholar] [CrossRef]
- Aung, Y.Y.M.; Sowter, M.; Kenealy, T.; Herman, J.; Ekeroma, A. Gestational diabetes mellitus screening, management and outcomes in the Cook Islands. N. Z. Med. J. 2015, 128. [Google Scholar]
- Ekeroma, A.J.; Chandran, G.S.; McCowan, L.; Ansell, D.; Eagleton, C.; Kenealy, T. Impact of using the international association of diabetes and pregnancy study groups criteria in South Auckland: Prevalence, interventions and outcomes. Aust. N. Z. J. Obstet. Gynaecol. 2014, 55, 34–41. [Google Scholar] [CrossRef]
- Kieffer, E.C.; Nolan, G.H.; Carman, W.J.; Sanborn, C.Z.; Guzman, R.; Ventura, A. Glucose Tolerance During Pregnancy and Birth Weight in a Hispanic Population. Obstet. Gynecol. 1999, 94, 741–746. [Google Scholar] [CrossRef] [PubMed]
- Donovan, L.; Edwards, A.; Savu, A.; Butalia, S.; Ryan, E.A.; Johnson, J.A.; Kaul, P. Population-level outcomes with a 2-step approach for gestational diabetes screening and diagnosis. Can. J. Diabetes 2017, 41, 596–602. [Google Scholar] [CrossRef]
- Kgosidialwa, O.; Egan, A.M.; Carmody, L.; Kirwan, B.; Gunning, P.; Dunne, F.P. Treatment with Diet and Exercise for Women With Gestational Diabetes Mellitus Diagnosed Using IADPSG Criteria. J. Clin. Endocrinol. Metab. 2015, 100, 4629–4636. [Google Scholar] [CrossRef] [PubMed]
- Kaul, P.; Savu, A.; Nerenberg, K.A.; Donovan, L.; Donovan, E.; Chik, C.L.; Ryan, E.A.; Johnson, J.A. Impact of gestational diabetes mellitus and high maternal weight on the development of diabetes, hypertension and cardiovascular disease: A population-level analysis. Diabet. Med. 2015, 32, 164–173. [Google Scholar] [CrossRef]
- Brand, J.S.; West, J.; Tuffnell, D.; Bird, P.K.; Wright, J.; Tilling, K.; Lawlor, D.A. Gestational diabetes and ultrasound-assessed fetal growth in South Asian and White European women: Findings from a prospective pregnancy cohort. BMC Med. 2018, 16, 203. [Google Scholar] [CrossRef]
- Kawakita, T.; Bowers, K.; Hazrati, S.; Zhang, C.; Grewal, J.; Chen, Z.; Sun, L.; Grantz, K.L.; Gtantz, K. Increased Neonatal Respiratory Morbidity Associated with Gestational and Pregestational Diabetes: A Retrospective Study. Am. J. Perinatol. 2017, 34, 1160–1168. [Google Scholar] [CrossRef] [PubMed]
- Boghossian, N.S.; Yeung, E.; Albert, P.S.; Mendola, P.; Laughon, S.K.; Hinkle, S.N.; Zhang, C. Changes in diabetes status between pregnancies and impact on subsequent newborn outcomes. Am. J. Obstet. Gynecol. 2014, 210, 431.e1. [Google Scholar] [CrossRef] [PubMed]
- Meek, C.L.; Lewis, H.B.; Patient, C.; Davies, G.A.; Poitras, V.; Gray, C.; Jaramillo Garcia, A.; Barrowman, N.; Adamo, K.B.; Duggan, M.; et al. Diagnosis of gestational diabetes mellitus: Falling through the net. Diabetologia 2015, 58, 2003–2012. [Google Scholar] [CrossRef] [PubMed]
- Wahabi, H.; Fayed, A.; Esmaeil, S.; Mamdouh, H.; Kotb, R. Prevalence and complications of pregestational and gestational diabetes in Saudi women: Analysis from Riyadh Mother and Baby cohort study (RAHMA). Biomed. Res. Int. 2017, 2017, 6878263. [Google Scholar] [CrossRef]
- Avalos, G.E.; Owens, L.A.; Dunne, F.; ATLANTIC DIP Collaborators. Applying current screening tools for gestational diabetes mellitus to a European population: Is it time for change? Diabetes Care 2013, 36, 3040–3044. [Google Scholar] [CrossRef]
- Anderberg, E.; Källén, K.; Berntorp, K. The impact of gestational diabetes mellitus on pregnancy outcome comparing different cut-off criteria for abnormal glucose tolerance. Acta Obstet. Gynecol. Scand. 2010, 89, 1532–1537. [Google Scholar] [CrossRef]
- Gu, Y.; Lu, J.; Li, W.; Liu, H.; Wang, L.; Leng, J.; Li, W.; Zhang, S.; Wang, S.; Tuomilehto, J.; et al. Joint Associations of Maternal Gestational Diabetes and Hypertensive Disorders of Pregnancy with Overweight in Offspring. Front. Endocrinol. 2019, 10. [Google Scholar] [CrossRef]
- Waters, T.P.; Dyer, A.R.; Scholtens, D.M.; Dooley, S.L.; Herer, E.; Lowe, L.P.; Oats, J.J.; Persson, B.; Sacks, D.A.; HAPO Cooperative Study Research Group; et al. Maternal and Neonatal Morbidity for Women Who Would Be Added to the Diagnosis of GDM Using IADPSG Criteria: A Secondary Analysis of the Hyperglycemia and Adverse Pregnancy Outcome Study. Diabetes Care 2016, 39, 2204–2210. [Google Scholar] [CrossRef] [PubMed]
- Moses, R.; Griffiths, R. Can a diagnosis of gestational diabetes be an advantage to the outcome of pregnancy? J. Soc. Gynecol. Investig. 1995, 2, 523–525. [Google Scholar] [CrossRef]
- Karmon, A.; Levy, A.; Holcberg, G.; Wiznitzer, A.; Mazor, M.; Sheiner, E. Decreased perinatal mortality among women with diet-controlled gestational diabetes mellitus. Int. J. Gynaecol. Obstet. 2009, 104, 199–202. [Google Scholar] [CrossRef]
- Capula, C.; Chiefari, E.; Vero, A.; Arcidiacono, B.; Iiritano, S.; Puccio, L.; Pullano, V.; Foti, D.P.; Brunetti, A.; Vero, R. Gestational Diabetes Mellitus: Screening and Outcomes in Southern Italian Pregnant Women. ISRN Endocrinol. 2013, 2013, 1–8. [Google Scholar] [CrossRef]
- Von Katterfeld, B.; Li, J.; McNamara, B.; Langridge, A.T. Maternal and neonatal outcomes associated with gestational diabetes in women from culturally and linguistically diverse backgrounds in Western Australia. Diabet. Med. 2012, 29, 372–377. [Google Scholar] [CrossRef] [PubMed]
- López Stewart, G. Diagnostic Criteria and Classification of Hyperglycaemia First Detected in Pregnancy: A World Health Organization Guideline; World Health Organization: Geneva, Switzerland, 2014. [Google Scholar]
- Bogdanet, D.; O’Shea, P.M.; Lyons, C.; Shafat, A.; Dunne, F. The Oral Glucose Tolerance Test—Is It Time for a Change?—A Literature Review with an Emphasis on Pregnancy. J. Clin. Med. 2020, 9, 3451. [Google Scholar] [CrossRef] [PubMed]
- Ansarzadeh, S.; Salehi, L.; Mahmoodi, Z.; Mohammadbeigi, A. Factors affecting the quality of life in women with gestational diabetes mellitus: A path analysis model. Health Qual. Life Outcomes 2020, 18, 1–9. [Google Scholar] [CrossRef]
- Hoffman, L.; Nolan, C.; Wilson, J.D.; Oats, J.J.N.; Simmons, D. Gestational diabetes mellitus—Management guidelines: The Australasian Diabetes in Pregnancy Society. Med. J. Aust. 1998, 169, 93–97. [Google Scholar] [CrossRef] [PubMed]
- ACOG. Gestational Diabetes Mellitus; ACOG Practice Bulletin; ACOG: Washington, DC, USA, 2018. [Google Scholar]
- Lash, R.W. Diabetes and Pregnancy—An Endocrine Society Clinical Practice Guideline Publication Note. J. Clin. Endocrinol. Metab. 2018, 103, 4042. [Google Scholar] [CrossRef] [PubMed]
- Mission, J.F.; Ohno, M.S.; Cheng, Y.W.; Caughey, A.B. Gestational diabetes screening with the new IADPSG guidelines: A cost-effectiveness analysis. Am. J. Obstet. Gynecol. 2012, 207, 326.e1. [Google Scholar] [CrossRef]
- Weile, L.K.; Kahn, J.G.; Marseille, E.; Jensen, D.M.; Damm, P.; Lohse, N. Global cost-effectiveness of GDM screening and management: Current knowledge and future needs. Best Pract. Res. Clin. Obstet. Gynaecol. 2015, 29, 206–224. [Google Scholar] [CrossRef]
- Fitria, N.; Van Asselt, A.D.I.; Postma, M.J. Cost-effectiveness of controlling gestational diabetes mellitus: A systematic review. Eur. J. Health Econ. 2019, 20, 407–417. [Google Scholar] [CrossRef] [PubMed]
- Moss, J.R.; Crowther, C.A.; Hiller, J.E.; Willson, K.J.; Robinson, J.S.; Australian Carbohydrate Intolerance Study in Pregnant Women Group. Costs and consequences of treatment for mild gestational diabetes mellitus–evaluation from the ACHOIS randomised trial. BMC Pregnancy Childbirth 2007, 7, 27. [Google Scholar] [CrossRef] [PubMed]
- Ohno, M.S.; Sparks, T.N.; Cheng, Y.W.; Caughey, A.B. Treating mild gestational diabetes mellitus: A cost-effectiveness analysis. Am. J. Obstet. Gynecol. 2011, 205, 282.e1. [Google Scholar] [CrossRef]
- Poncet, B.; Touzet, S.; Rocher, L.; Berland, M.; Orgiazzi, J.; Colin, C. Cost-effectiveness analysis of gestational diabetes mellitus screening in France. Eur. J. Obstet. Gynecol. Reprod. Biol. 2002, 103, 122–129. [Google Scholar] [CrossRef]
- Kalra, S.; Baruah, M.P.; Gupta, Y.; Kalra, B. Gestational diabetes: An onomastic opportunity. Lancet Diabetes Endocrinol. 2013, 1, 91. [Google Scholar] [CrossRef]
- Marchetti, D.; Carrozzino, D.; Fraticelli, F.; Fulcheri, M.; Vitacolonna, E. Quality of Life in Women with Gestational Diabetes Mellitus: A Systematic Review. J. Diabetes Res. 2017, 2017, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Kalra, B.; Gupta, Y.; Baruah, M.P. Renaming gestational diabetes mellitus: A psychosocial argument. Indian J. Endocrinol. Metab. 2013, 17, 593–595. [Google Scholar] [CrossRef]
- Pantzartzis, K.A.; Manolopoulos, P.P.; Paschou, S.A.; Kazakos, K.; Kotsa, K.; Goulis, D.G. Gestational diabetes mellitus and quality of life during the third trimester of pregnancy. Qual. Life Res. 2019, 28, 1349–1354. [Google Scholar] [CrossRef]
- Weissgerber, T.L.; Mudd, L.M. Preeclampsia and diabetes. Curr. Diabetes Rep. 2015, 15, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Naylor, C.D.; Sermer, M.; Chen, E.; Sykora, K. Cesarean Delivery in Relation to Birth Weight and Gestational Glucose Tolerance. JAMA 1996, 275, 1165–1170. [Google Scholar] [CrossRef] [PubMed]

| Author, Year | Country | GDM Diagnostic Criteria | GDM Characteristics * | Non-GDM Characteristics * | Adverse Maternal Outcome in Women with vs. without GDM, % or Mean (SD) |
|---|---|---|---|---|---|
| Capula et al., 2013 | Italy | IADPSG | n = 171, Age: 30.8 (3.2), BMI: 22.8 (1.9) | n = 367, Age: 29.3 (3.5), BMI: 21.4 (2.0) | Hypertension: 4.1 vs. 1.6; Preeclampsia: 2.9 vs. 1.4; Labor induction: 1.2 vs. 0.3; gestational weight gain: 10.3 (3.4) vs. 8 (2.8); Primary cesarean section: 29.8 vs. 15.3 |
| Karmon et al., 2009 | Israel | CC | n = 10,227 | n = 174,029 | Hypertensive disorders: 11.6 vs. 5.5 Abruption: 0.8 vs. 0.7; Labor induction: 42.1 vs. 27.0. |
| Moses et al., 1995 | Australia | ADIPS | n = 138, Age: 29.5 (5.3) | n = 144, Age: 28.2 (5.4) | PIH:13.8 vs. 13.2; Labor induction: 26.8 vs. 26.4 |
| Waters et al., 2016 | North American | 1) IADPSG 2) CC | 1) n = 878, Age: 31.0 (5.6), BMI: 31.5 (6.4) 2) n = 261, Age: 32.3 (5.3), BMI: 31.6 (5.8) | n = 5020, Age: 30.1 (5.8), BMI: 28.2 (4.9) | 1) Preeclampsia: 14.9 vs. 6.4; Primary cesarean section: 23.9 vs. 17.2 2) Preeclampsia: 14 vs. 6.4; Primary cesarean section: 30.4 vs. 17.2 |
| Gu et al., 2019 | China | WHO-1999 | GDM with hypertensive disorders of pregnancy: n = 91, Age: 33.8 (3.59), Pre-pregnancy BMI: 25.1 (3.64) GDM without hypertensive disorders of pregnancy: n = 1172, Age: 33.3 (3.49), Pre-pregnancy BMI: 22.9 (3.24) | Non-GDM with hypertensive disorders of pregnancy: n = 261, Age: 32.9 (2.68), Pre-pregnancy BMI: 22.2 (3.04) Non-GDM without hypertensive disorders of pregnancy: n= 261, Age: 32.9 (2.84), Pre-pregnancy BMI: 21.4 (2.96) | Non hypertensive disorder: Gestational weight gain, kg: 16.6 (5.87) vs. 18.2 (6.67) Hypertensive disorder: Gestational weight gain, kg: 19.0 (7.01) vs. 21.3 (6.14) |
| Shand et al., 2008 | Australia | ADIPS | n = 16,727 | n = 349,933 | Pre-eclampsia: 6.7 vs. 4.4; Gestational hypertension: 6.9 vs. 4.2; Placenta Previa or abruption: 1.6 vs. 1.1; APH: 1.5 vs. 1.1; PPH: 6.3 vs. 6; Severe PPH: 0.9 vs. 0.7; Labor induction: 32.7 vs. 23.9 |
| Anderberg et al., 2010 | Sweden | WHO-1999 | n = 306, Age: 32 (18–46) | n = 329, Age: 31 (20–42) | Labor induction: 18.6 vs. 6.4 |
| Avalos et al., 2013 | Ireland | IADPSG | n = 622, Age: 32.8 | n = 4225, Age: 31 (4.9) | GDM without risk factor vs. GDM with risk factor vs. Non-GDM Hypertension: 13 vs. 15 vs. 7 |
| Wahabi et al., 2017 | Saudi Arabia | WHO-2013 | n = 2354, Age: 31.5 (5.9) | n = 6951, Age: 29.5 (5.7) | Gestational hypertension: 1.8 vs. 1.3; Preeclampsia/superimposed: 1 vs. 1.1; Labor induction: 17.9 vs. 16 |
| Meek et al., 2015 | UK | 1) IADPSG 2) NICE | 1) n = 387, Age: 32.6, BMI: 27.4 2) n = 261, Age: 32.1, BMI: 25.5 | n = 2406, Age: 31.4, BMI: 26 | 1) Pre-eclampsia: 10.1 vs. 7.2; PPH:1 vs. 2; APH: 1.6 vs. 2.4 2) Pre-eclampsia: 9.2 vs. 7.2; PPH:0.4 vs. 2; APH: 2.7 vs. 2.4 |
| Boghossian et al., 2014 | USA | ICD-9 | n = 1279, Age: 30.3 (4.9); Prepregnancy BMI: 28.9 (7.2) | n = 58,224, Age: 28.1 (4.5), Prepregnancy BMI: 24.9 (5.6) | Gestational hypertension: 4.7 vs. 2.2; Preeclampsia: 3 vs. 1.6; Labor induction: 40.2 vs. 39.4 |
| Kawakita et al., 2017 | USA | ICD-9 | n = 11,327, Age: 30.8 (6.0), BMI: 34.1 (7.5) | n= 208,355, Age: 27.4 (6.1), BMI: 30.6 (6.1) | Pregnancy-associated hypertension: 11.7 vs. 7.2 |
| Brand et al.2018 | UK | Modified WHO-1999 | White European: n = 210, Age: 30.2 (5.4), BMI: 28.6 (6.3) South Asian: n = 622, Age: 30.7 (5.3), BMI: 28.2 (5.8) | White European: n = 4537, Age: 26.6 (6.0), BMI: 26.5 (5.9) South Asian: n = 5336, Age: 27.7 (5.0), BMI: 25.2 (5.3) | White European Hypertensive disorders of pregnancy: 6.7 vs. 6.7 South Asian Hypertensive disorders of pregnancy: 5.6 vs. 5.2 |
| Kaul et al., 2014 | Canada | CDA-2013 | GDM only: n = 7332, Age: 31.9 (5.5) GDM and overweight: n = 1399, Age: 31 (5.2) | n = 213,765, Age: 28.6 (5.6) | GDM only vs. GDM and overweight vs. No GDM, not overweight Pre-eclampsia or eclampsia: 1.9 vs. 5.5 vs. 1.2; Labor induction: 42.1 vs. 58.4 vs. 28.5 |
| Kgosidialwa et al., 2015 | Ireland | IADPSG | n = 567, Age: 33.4 (4.9), BMI: 30.5 (6.1) | n = 2499, Age: 31.5 (5.2), BMI: 26.7 (4.8) | Pre-eclampsia: 4.2 vs. 3.8; Hypertensive pregnancy disorders: 11.6 vs. 8.3 PIH: 11.6 vs. 7.7 |
| Donovan et al., 2017 | Canada | CDA IADPSG | HAPO 1.75: n = 4308, Age: 31.2 (5.1) HAPO 2–1: n = 5528, Age: 31.6 (5.2) HAPO 2–2: n = 3252, Age: 32.1 (5.2) | Normal 50 g screen: n = 144,191, Age: 28.8 (5.3) Normal 75 g OGTT: n = 21,248, Age: 30.3 (5.3) | Normal 50 g screen: Hypertensive disorders of pregnancy: 5.6; Labor induction: 27.5 Normal 75 g OGTT: Hypertensive disorders of pregnancy: 7.3; Labor induction: 27.7 HAPO 1.75: Hypertensive disorders of pregnancy: 9.1; Labor induction: 29.6 HAPO 2–1: Hypertensive disorders of pregnancy: 9.6; Labor induction: 38.2 HAPO 2–2: Hypertensive disorders of pregnancy: 11.7; Labor induction: 42.3 |
| Kieffer et al., 1999 | Michigan | NDDG | n = 19, Age: 29.4 (6.2), BMI: 28.7 (5.7) | n = 353, Age: 24.79 (4.85), BMI: 25.1 (4.21) | Hypertensive disorder: 21.1 vs. 7.16 |
| Ekeroma et al., 2014 | New Zealand | 1) NZSSD 2) IADPSG 3) ADIPS | 1) n = 381, Age: 31.7 (5.5), BMI: 31.8 (10.8) 2) n = 238, Age: 31.4 (5.8), BMI: 32.9 (11.7) 3) n = 608, Age: 31.5 (5.4), BMI: 30.5 (9.8) | n = 1672, Age: 30.0 (5.7), BMI: 30.7 (9.1) | 1) Pre-eclampsia: 8 vs. 6 2) Pre-eclampsia: 7 vs. 6 3) Pre-eclampsia: 7 vs. 6 |
| Aung et al., 2015 | Cook Islands | Modified IADPSG | n = 94, Age: 36 (28–40), BMI: 34 (30–39) | n = 28 (23–34), Age: 24.79 (4.85), BMI: 31 (26–36) | Pregnancy weight gain (kg): 6 (3–11) vs. 10 (6–14) |
| Erjavec et al., 2016 | Croatia | 1) WHO-1999 2) IADPSG | 1) n = 953, Age: 30.88 (5.23), BMI: 25.84 (5.28) 2) n = 1829, Age: 31.34 (5.19), BMI: 26.03 (5.64) | 1) n = 41,703, Age: 28.77 (5.23), BMI: 23.38 (3.99) 2) n = 37,263, Age: 29.49 (5.33), BMI: 23.38 (4.11) | 1) Weight gain: 12.57 (5.62) vs. 14.51 (5.29) 2) Weight gain: 12.50 (5.76) vs. 14.19 (5.71) |
| Gortazar et al., 2018 | Spain | NDDG | n = 35,729, Age: 33.42 | n = 704,148, Age: 31.27 | Pre-eclampsia: 2.56 vs. 1.44 |
| Zamstein et al., 2018 | Israel | ACOG | GDM A1: n = 9460, Age: 32.1 (5.8) GDM A2: n = 724, Age: 33.7 (5.6) | n = 206,013, Age: 28 (5.7) | GDM A1 vs. GDM A2 vs. Non-GDM Hypertensive disorders of pregnancy: 11.2 vs. 18.1 vs. 4.8 |
| Hedderson et al., 2003 | California | 1) NDDG 2) CC | 1) n = 1523 2) n = 840 | n = 38,515 | 1) Pregnancy-induced hypertension: 3.4 vs. 1.9; Preeclampsia or eclampsia:5.8 vs. 2.9; Placenta previa: 0.6 vs. 0.1; Abruptio placentae: 1 vs. 0.8; Labor induction: 18.4 vs. 14.5 2) Pregnancy-induced hypertension: 3.6 vs. 1.9; Preeclampsia or eclampsia: 5.6 vs. 2.9; Placenta previa: 0.8 vs. 0.1; Abruptio placentae: 0.5 vs. 0.8; Labor induction: 13.5 vs. 14.5 |
| Hosseini et al., 2018 | Iran | IADPSG | Early-onset GDM: n = 93, Age: 30.7 (4.6), Pre-pregnancy BMI: 26.5 (4.2) Late-onset GDM: n = 78, Age: 31.1 (4.9), Pre-pregnancy BMI: 26.2 (4.7) | n = 758, Age: 28.8 (4.6), Pre-pregnancy BMI: 24.2 (4.1) | Early-onset GDM vs. Late-onset GDM vs. Normal Preeclampsia: 6.5 vs. 6.4 vs. 3.6 Gestational hypertension: 8.6 vs. 12.8 vs. 6.1 |
| Hosseini et al., 2018 | Iran | 1) IADPSG 2) CC | 1) n = 78, Age: 18–45 2) n = 35, Age: 18–45 | 1) n = 35, Age: 18–45 2) n = 801, Age: 18–45 | 1) Preeclampsia (OR): 1.5; Gestational hypertension (OR): 1.9 2) Preeclampsia (OR): 2.8; Gestational hypertension (OR): 2.4 |
| Jain et al., 2016 | India | DIPSI | N = 8000 | n = 7641 | PIH: 9 vs. 6; APH/PPH: 0.84 vs. 0.32 |
| Kun et al., 2010 | Tolna | WHO-1999 | n = 139, Age: 29.6 (5.2), Pregnancy BMI: 25.4 (5.3) | n = 2583, Age: 27.1 (4.9), Pregnancy BMI: 23.1 (4.5) | Weight gain, kg: 9.1 (4.8) vs. 12.9 (5.0) |
| Leybovitz-Haleluya et al., 2018 | Israel | ACOG | GDM A1: n = 9460, Age: 32.1 (5.8) GDM A2: n = 724, Age: 33.7 (5.6) | n = 206,013, Age: 28 (5.7) | GDM A1 vs. GDM A2 vs. No GDM Preeclampsia: 7 vs. 6.4 vs. 3.9 |
| Jacobson et al., 1989 | California | NDDG | n = 97, Age: 28.8 (0.5), BMI: 27.6 (0.8) | n = 2107, Age: 26.3 (0.1), BMI: 22.8 (0.1) | Pregnancy-induced hypertension: 3.8 vs. 3.7; Weight gain: 30.2 (1.8) (pounds) vs. 33.0 (0.3) |
| Pan et al., 2015 | China | 1) WHO-1999 2) IADPSG | 1) n = 257, Age: 29 (2.6), Prepregnancy BMI: 22.9 (3.5) 2) n = 429, Age: 28.8 (2.9), Prepregnancy BMI: 23.9 (4) | n = 16 173, Age: 28.4 (2.8), Prepregnancy BMI: 22.1 (3.3) | 1) PIH: 15.8 vs. 4.8 2) PIH: 7.5 vs. 4.8 |
| Son et al., 2014 | Korea | ICD-10 | n = 78,716, Age: 15–49 | n = 1171575, Age: 15–49 | Pregnancy-induced hypertension without significant proteinuria: 1.71 vs. 1; Pregnancy-induced hypertension with significant proteinuria: 1.66 vs. 1.13; Eclampsia: 0.08 vs. 0.05; Placenta previa: 1.41 vs. 1.16; Premature separation of placenta: 0.42 vs. 0.42; Postpartum hemorrhage: 7.03 vs. 7.30; Antepartum hemorrhage: 2.29 vs. 2.39 |
| Katterfeld et al., 2011 | Australia | ADIPS | Australian born n = 4765 CALD n = 1686 Non-CALD n = 1273 | Australian born n = 142,537 CALD n = 23,541 Non-CALD n = 31,814 | Australian born Pre-eclampsia: 8.4 vs. 5; Labor induction: 54.3 vs. 37.3 CALD Pre-eclampsia: 5.6 vs. 3.6; Labor induction: 37.6 vs. 25.7 Non-CALD Pre-eclampsia: 7.2 vs. 4.6; Labor induction: 51.9 vs. 35 |
| Sacks et al., 2015 | California | IADPSG | 1) GDM-1: n = 771, Age: 30.9 (5.6) 2) GDM-2: n = 1121, Age: 31 (5.7) | n = 7943, Age: 26.3 (0.1) | GDM-1 vs. GDM-2 vs. normal Preeclampsia–eclampsia: 4.3 vs. 7.7 vs. 4.4; Primary cesarean delivery: 20.6 vs. 22.3 vs. 16.6 |
| Soliman et al., 2018 | Qatar | IADPSG | n = 3027 | n = 8995 | Hypertensive disorders: 5.5 vs. 3.5; Labor induction: 26.5 vs. 12.4 |
| Xiong et al., 2001 | Canada | CDA | n = 2755 | n = 8995 | Gestational hypertension: 11.4 vs. 4.8; Pre-eclampsia: 1.1 vs. 1.1 |
| Oster et al., 2014 | Canada | CDA | n = 1224, Age: 28.8 (6.27) | n = 26,793, Age: 24.7 (5.8) | Pregnancy induced hypertension: 11.3 vs. 4.4; Labor induction: 43.6 vs. 23.8 |
| Sugaya et al., 2000 | Japan | 1) JSOG 2) WHO-1998 | 1) n = 55, Age: 29.7 (4.3), BMI: 26.2 (3.4) 2) n = 51, Age: 32.8 (4.3), BMI: 26.5 (4.3) | n = 281, Age: 30 (4.7), BMI: 25.5 (3.3) | 1) preeclampsia: 18 vs. 17 2) preeclampsia: 28 vs. 17 |
| Nerenberg et al., 2013 | Canada | CDA | n = 15,404, Age: 31.5 (5.4) | n = 407,268, Age: 28.4 (5.6) | Preeclampsia/eclampsia: 2.6 vs. 1.2; Labor induction: 41.9 vs. 27.1 |
| Edith Kieffer et al., 2006 | Mexico | ADA-2003 | n = 68, Age: 28.6 (0.6), BMI: 25.7 (0.2) | n = 933, Age: 24.8 (0.2), BMI: 28.4 (0.8) | Weight gain (kg): 10.0 (0.6) vs. 13 (0.2) |
| Goswami Mahanta et al., 2014 | India | DIPSI | N = 28 | n = 749 | Gestational hypertension: 53.6 vs. 28.1 |
| Ellerbe et al., 2013 | USA | ICD-9 | Non-Hispanic White: n = 8567, Age: 29.6 (5.9), BMI: 29.3(7.3) Non-Hispanic Black n = 4724, Age: 27.5 (6.2), BMI: 31.7 (7.5) | Non-Hispanic White: n = 126,524, Age: 27.0 (5.9), BMI: 25.7 (6.1) Non-Hispanic Black n = 71,939, Age: 24.3 (5.6), BMI: 28.1(7.0) | Non-Hispanic White Gestational weight gain (kg): 11.7 (7.7) vs. 13.7 (7.6). Non-Hispanic Black Gestational weight gain (kg): 11.5 (8.3) vs. 11.1 (8.0) |
| Sletner et al., 2017 | Norway | WHO-1999 | Europe Mild: n = 30, Age: 31.2 (29.5), BMI: 25.5 (23.8, 27.2) Moderate/severe: n = 9, Age: 30.6 (27.6, 33.5), BMI: 30.5 (27.4, 33.6) South Asia Mild: n = 9, Age: 30.7 (28.3, 33.0), BMI: 25.3 (23.2, 27.5) Moderate/severe: n = 4724, Age: 30.4 (28.0, 32.7), BMI: 22.7 (20.6, 24.9) | Europe n = 310, Age: 30.6 (30.1, 31.1), BMI: 24.3 (23.8, 24.8) South Asia n = 156, Age: 28.4 (27.7, 29.1), BMI: 23.7 (23.0, 24.3) | Europe Mild vs. Moderate/Severe vs. Non-GDM Mild hypertension/preeclampsia: 10 vs. 0 vs. 7; Severe hypertension/ preeclampsia: 2 vs. 0 vs. 2; inclusion to week 28 GWG: 6.2 (5.2, 7.2) vs. 5.2 (3.4, 7.1) vs. 7.1 (6.8, 7.4), week 28 to birth: 4.0 (2.6, 5.5) vs. 2.0 (-0.4, 4.4) vs. 5.9 (5.5, 6.4) South Asia Mild hypertension/preeclampsia: 7 vs. 14 vs. 3; Severe hypertension/ preeclampsia: 0 vs. 7 vs. 2; inclusion to week 28, GWG: 5.6 (3.9, 7.4) vs. 6.5 (4.7, 8.2) vs. 6.6 (6.0, 7.1), week 28 to birth, GWG: 5.1 (2.9, 7.4) vs. 4.8 (2.5, 7.0) vs. 5.2 (4.5, 5.9) |
| Zeki et al., 2018 | Australia | ADIPS | n = 51135, Age: 32.2 (5.3) | n = 950 678, Age: 29.9 (5.6) | Primary Cesarean: Relative % 13.8 vs. 13.5 |
| Hoorn et al., 2002 | Australia | ADIPS | n = 51, Age: 30.9 (5.7), BMI:31.5 (.1) | n = 258, Age: 24.9 (6.3), BMI: 25.5 (5.9) | Gestational hypertension: 45.1 vs. 29.1; Preeclampsia: 19.6 vs. 17.1 |
| Su et al., 2019 | China | China National criteria | Underweight n = 1466, BMI: 17.55 (0.79) Normal weight n = 6905, BMI: 20.80 (1.21) Overweight n = 2220, BMI: 23.86 (0.57) Obese n = 2252, BMI: 27.21 (2.15) | Underweight n = 12,336, BMI: 17.54 (0.79) Normal weight n = 36,935, BMI: 20.54 (1.2) Overweight n = 6654, BMI: 23.82 (0.56) Obese n = 4730, BMI: 26.97 (1.97) | Normal weight weight gain, kg: 11.45 (3.98) vs. 13.15 (0.25) Underweight weight gain, kg: 12.53 (3.94) vs. 13.76 (3.93) Overweight weight gain, kg: 10.92 (4.49) vs. 12.29 (4.48) Obese weight gain, kg: 8.87 (4.38) vs. 10.50 (4.35) |
| Metcalfe et al., 2017 | Canada | ICD-10 | n = 149,780 | n = 2,688,231 | Gestational hypertension: 7.93 vs. 4; Mild/unspecified Preeclampsia: 0.32 vs. 0.1; Severe preeclampsia: 2.05 vs. 1.18; Placenta previa: 0.9 vs. 0.58; Labor induction: 35.33 (Rate per 100 deliveries) vs. 22.04 |
| Carr et al., 2011 | USA | ICD-9&10 | n = 1314, Age: 32.7 (5.7) | One abnormal: n= 1242, Age: 32.3 (5.3) Non abnormal: n= 3620, Age: 32 (5.7) | Preeclampsia (n): 111 vs. 102 vs. 226 |
| Lamminpää et al., 2014 | Finland | ICD-10 | <35 y: n = 19,422 >35 y: n = 7732 | <35 y: n = 210,581 >35 y: n = 45,589.00 | <35 y: Normal glucose tol. vs. Diet-treated vs. Insulin-treated Preeclampsia: 4.2 vs. 6.7 vs. 7.7; Placenta previa: 0.2 vs. 0.2 vs. 0.2 Late pregnancy bleeding: 1 vs. 1.2 vs. 1.8 >35 y: Normal glucose tol. vs. Diet-treated vs. Insulin-treated Preeclampsia: 5.1 vs. 8.2 vs. 8.6; Placenta previa: 0.4 vs. 0.5 vs. 0.1; Late pregnancy bleeding: 1.3 vs. 1.3 vs. 1.4 |
| Black et al., 2010 | California | IADPSG | single isolated impaired glucose tolerance (i-IGT1) n =391, Age: 32.1 (5.4), BMI: 28.1 (5.6) isolated impaired fasting glucose (i-IFG) n = 886, Age: 30.4 (5.6), BMI: 30.8 (7.1) double-isolated impaired glucose tolerance (i-IGT2) n = 83, Age: 32.3 (5.2), BMI: 27.5 (4.7) IFG + IGT n = 331, Age: 32 (5.1), BMI: 31.8 (7) | n = 7020, Age: 28.6 (5.9), BMI: 26.9 (5.8) | i-IGT1 vs. i-IFG vs. i-IGT2 vs. IFG + IGT vs. No GDM Gestational hypertension: 9.8 vs. 10.8 vs. 13.6 vs. 15.4 vs. 7.2; Primary cesarean section: 12.8 vs. 9.1 vs. 18.1 vs. 8.2 vs. 6.6; gestational weight gain: 119 (30.4) (Ib) vs. 427 (48.2) vs. 23 (27.7) vs. 175 (52.9) vs. 1737 (24.7) |
| Outcomes £ | GDM Classification | Sample Size | Heterogenicity | Publication Bias Begg’s Test | Effect Size * (95% CI) | p-Value from Meta- Regression | ||
|---|---|---|---|---|---|---|---|---|
| GDM Group | Non-GDM Group | I2 (%) | p-Value | |||||
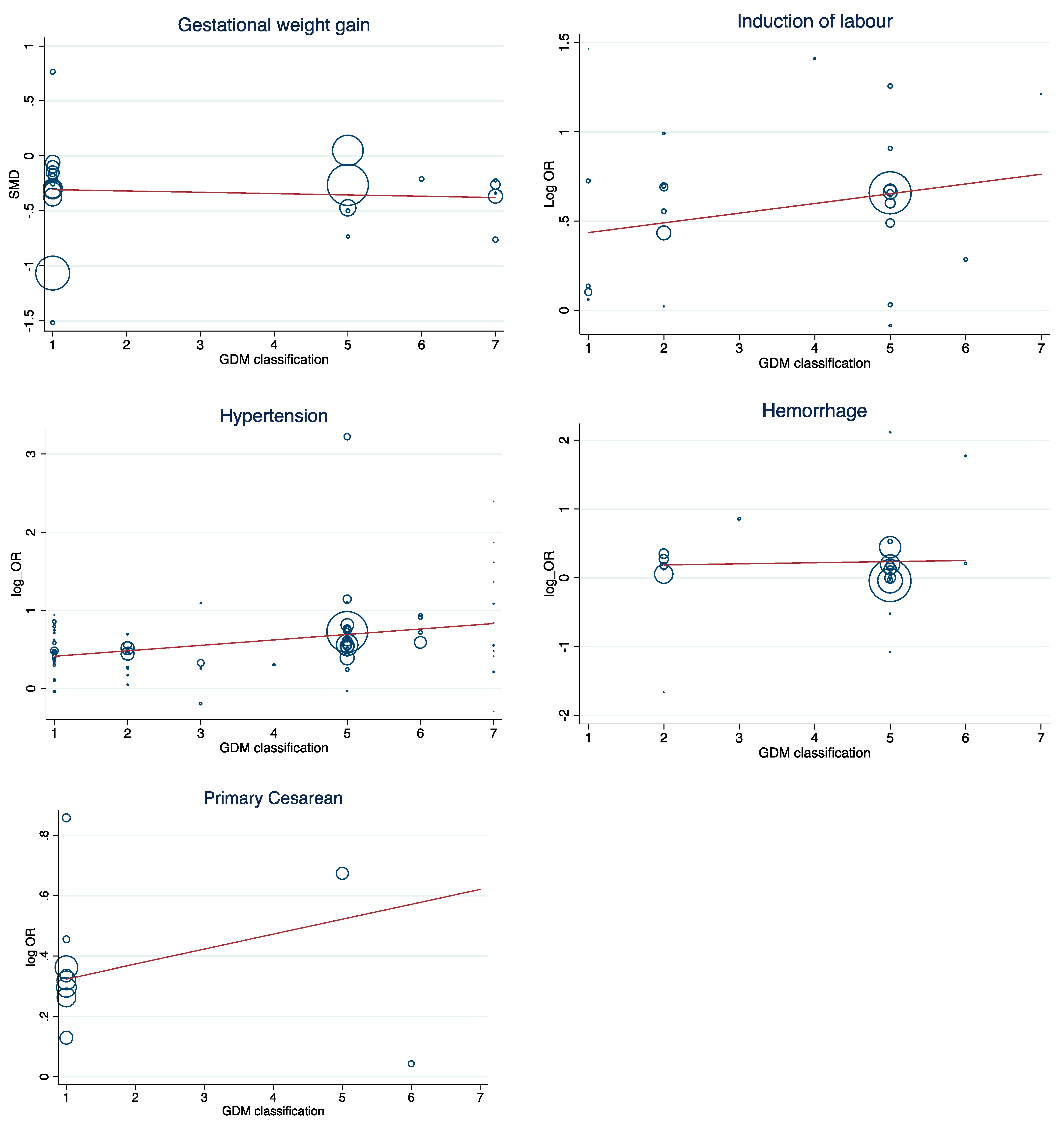
| Primary Cesarean | 1 | 4632 | 49,353 | 21.1 | 0.262 | 0.621 | 1.3 (1.2, 1.5) | Ref |
| Overall | 4990 | 56,480 | 41 | 0.084 | 0.655 | 1.4 (1.2, 1.5) | -- | |
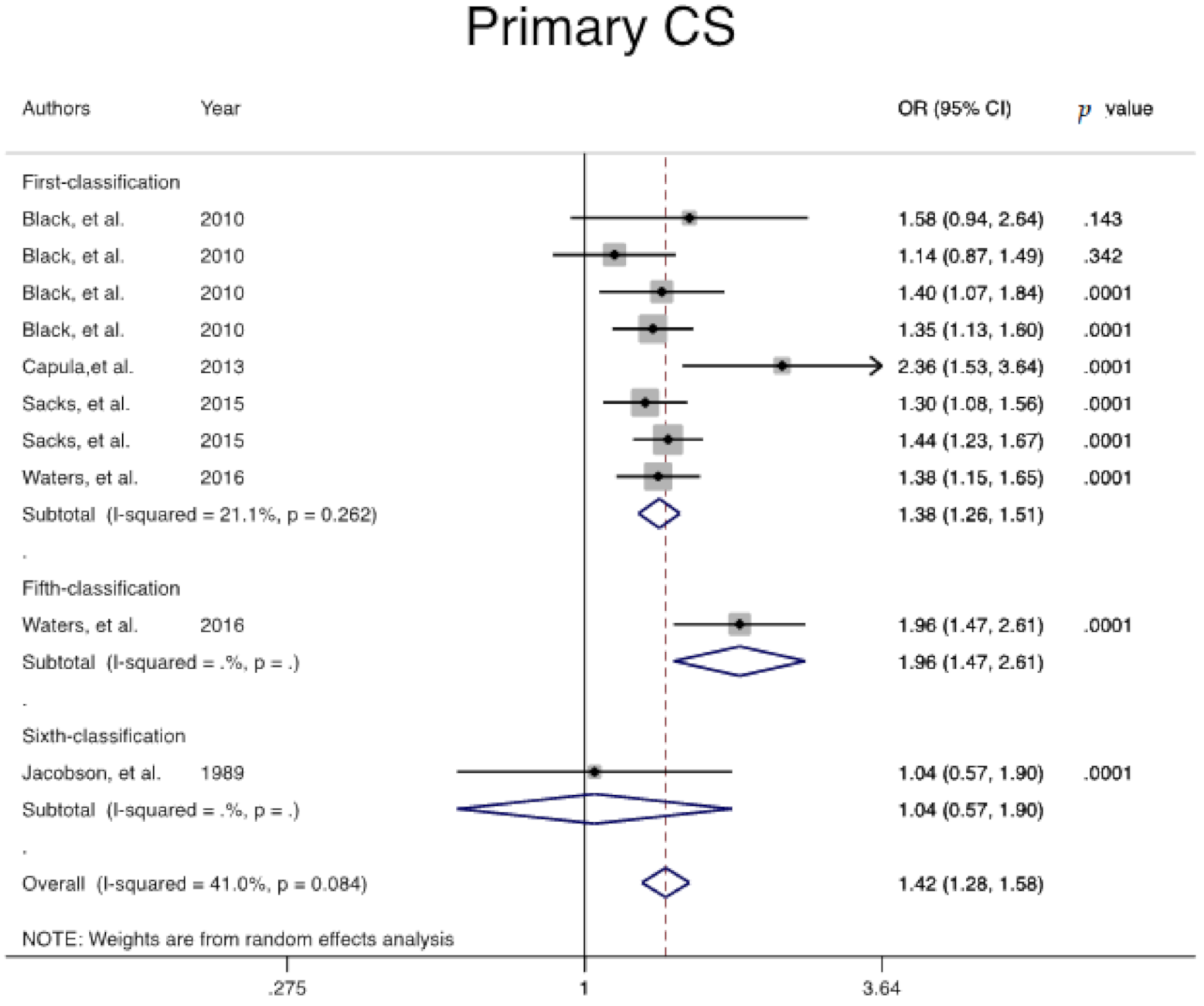
| Induction of labor | 1 | 10,098 | 183,424 | 95.2 | 0.001 | 0.327 | 1.3 (0.9, 1.8) | Ref |
| 2 | 25,197 | 549,639 | 94.7 | 0.001 | 0.851 | 1.8 (1.5, 2.1) | 0.144 | |
| 5 | 196,263 | 4,151,466 | 97.4 | 0.001 | 0.371 | 1.8 (1.6, 2.0) | 0.112 | |
| Overall | 233,767 | 4,925,044 | 97.5 | 0.001 | 0.766 | 1.7 (1.6, 1.9) | -- | |
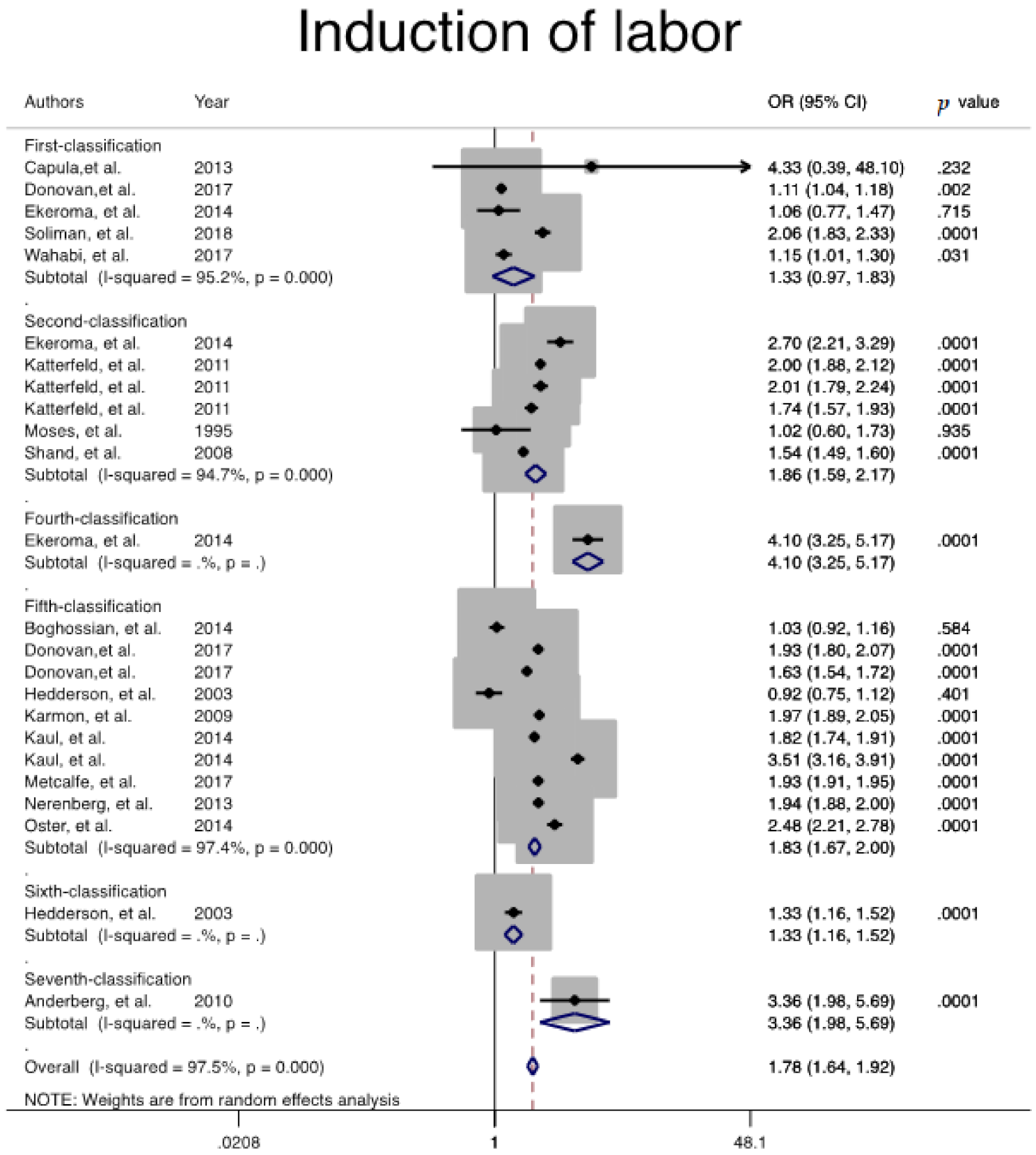
| Maternal Hemorrhage | 2 | 67,430 | 1,404,544 | 79.9 | 0.001 | 0.348 | 1.2 (1.0, 1.4) | € Ref |
| 5 | 609,575 | 9,821,846 | 95 | 0.001 | 0.680 | 1.1 (1.0, 1.3) | 0.867 | |
| 6 | 3046 | 77,031 | 91.9 | 0.001 | 0.317 | 2.6 (0.5, 12.6) | 0.126 | |
| Overall | 688,825 | 11,315,874 | 93 | 0.001 | 0.523 | 1.2 (1.0, 1.3) | -- | |
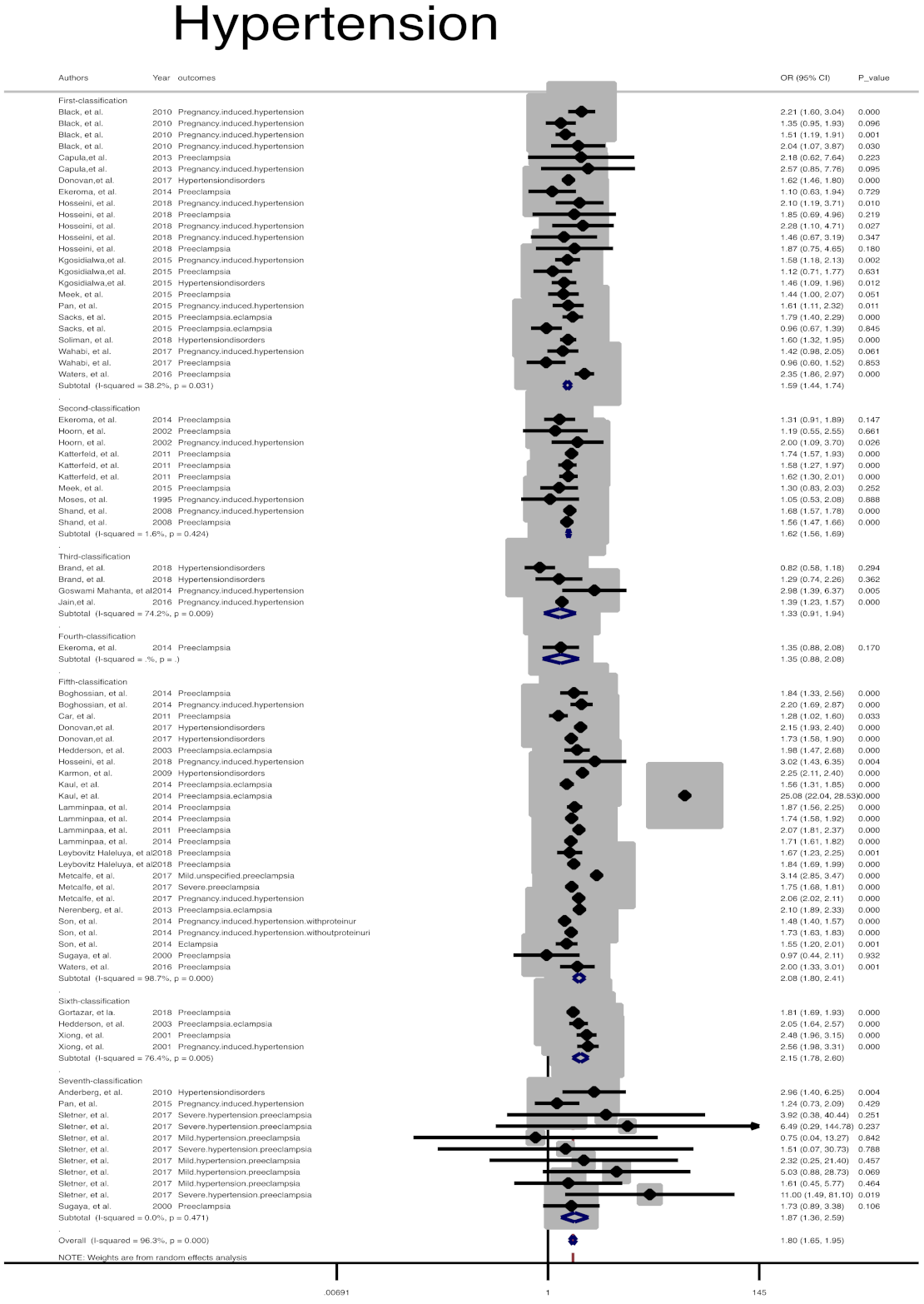
| Pregnancy related Hypertension | 1 | 20,021 | 269,637 | 38.2 | 0.031 | 0.766 | 1.5 (1.4, 1.7) | Ref |
| 2 | 42,287 | 902,497 | 1.6 | 0.424 | 0.325 | 1.6 (1.5, 1.6) | 0.784 | |
| 3 | 8860 | 18,263 | 74.2 | 0.009 | 0.497 | 1.3 (0.9, 1.9) | 0.535 | |
| 5 | 771,027 | 14,009,374 | 98.7 | 0.001 | 0.207 | 2.0 (1.8, 2.4) | 0.38 | |
| 6 | 42,762 | 959,991 | 76.4 | 0.005 | 0.051 | 2.1 (1.7, 2.6) | 0.160 | |
| 7 | 751 | 18,674 | 0 | 0.471 | 0.484 | 1.8 (1.3, 2.5) | 0.248 | |
| Overall | 886,089 | 1,618,008 | 96.3 | 0.001 | 0.541 | 1.7 (1.6, 1.9) | -- | |
| Gestational weight gain | 1 | 18,518 | 142,679 | 99.5 | 0.001 | 0.337 | −0.307 (−0.560, −0.054) | Ref |
| 5 | 14,689 | 257,901 | 90 | 0.001 | 0.624 | −0.353 (−0.569, −0.137) | 0.911 | |
| 7 | 2410 | 45,271 | 84.7 | 0.001 | 1.000 | −0.400 (−0.567, −0.233) | 0.988 | |
| Overall | 35,714 | 447,958 | 99.4 | 0.001 | 0.564 | −0.333 (−0.492, −0.174) | -- | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramezani Tehrani, F.; Naz, M.S.G.; Yarandi, R.B.; Behboudi-Gandevani, S. The Impact of Diagnostic Criteria for Gestational Diabetes Mellitus on Adverse Maternal Outcomes: A Systematic Review and Meta-Analysis. J. Clin. Med. 2021, 10, 666. https://doi.org/10.3390/jcm10040666
Ramezani Tehrani F, Naz MSG, Yarandi RB, Behboudi-Gandevani S. The Impact of Diagnostic Criteria for Gestational Diabetes Mellitus on Adverse Maternal Outcomes: A Systematic Review and Meta-Analysis. Journal of Clinical Medicine. 2021; 10(4):666. https://doi.org/10.3390/jcm10040666
Chicago/Turabian StyleRamezani Tehrani, Fahimeh, Marzieh Saei Ghare Naz, Razieh Bidhendi Yarandi, and Samira Behboudi-Gandevani. 2021. "The Impact of Diagnostic Criteria for Gestational Diabetes Mellitus on Adverse Maternal Outcomes: A Systematic Review and Meta-Analysis" Journal of Clinical Medicine 10, no. 4: 666. https://doi.org/10.3390/jcm10040666
APA StyleRamezani Tehrani, F., Naz, M. S. G., Yarandi, R. B., & Behboudi-Gandevani, S. (2021). The Impact of Diagnostic Criteria for Gestational Diabetes Mellitus on Adverse Maternal Outcomes: A Systematic Review and Meta-Analysis. Journal of Clinical Medicine, 10(4), 666. https://doi.org/10.3390/jcm10040666






