Bendamustine-Based Regimens as Salvage Therapy in Refractory/Relapsed Multiple Myeloma Patients: A Retrospective Real-Life Analysis by the Polish Myeloma Group
Abstract
:1. Introduction
2. Material and Methods
2.1. Patients Characteristics
2.2. Treatment Regimens
2.3. Treatment Response and Toxicity Assessment
2.4. Statistical Analysis
3. Results
3.1. Response to Treatment
3.2. Progression-Free Survival
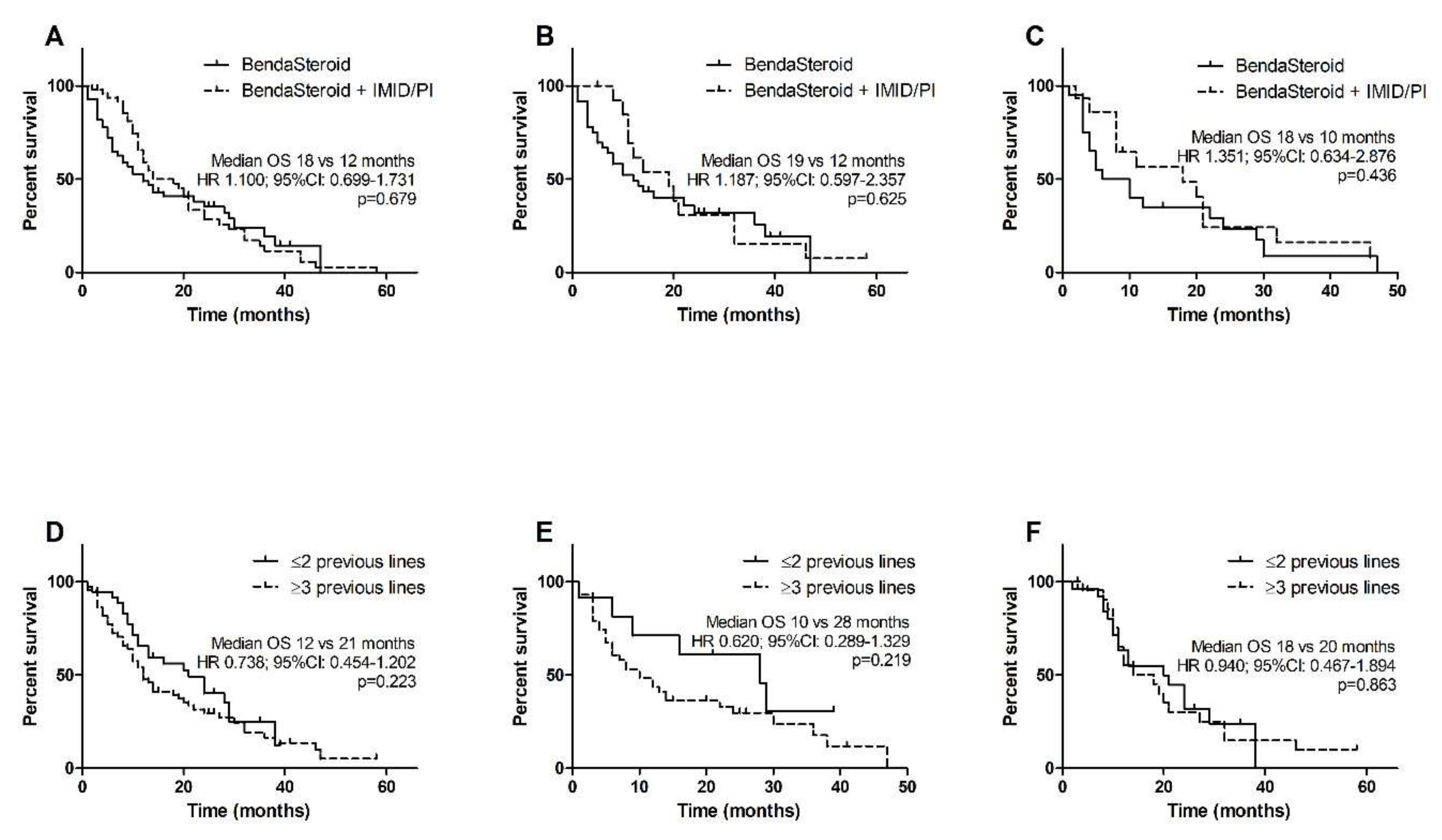
3.3. Overall Survival
3.4. Patients with High-Risk Cytogenetics
3.5. Toxicity and Tolerability of Bendamustine-Based Therapies
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kyle, R.A.; Rajkumar, S.V. Multiple myeloma. Blood 2008, 111, 2962–2972. [Google Scholar] [CrossRef] [Green Version]
- Larocca, A.; Mina, R.; Gay, F.; Bringhen, S.; Boccadoro, M. Emerging drugs and combinations to treat multiple myeloma. Oncotarget 2017, 36, 60656–60672. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, S.K.; Rajkumar, S.V.; Dispenzieri, A.; Lacy, M.Q.; Hayman, S.R.; Buadi, F.K.; Zeldenrust, S.R.; Dingli, D.; Russell, S.J.; Lust, J.A.; et al. Improved survival in multiple myeloma and the impact of novel therapies. Blood 2008, 111, 2516–2520. [Google Scholar] [CrossRef] [Green Version]
- Balfour, J.A.; Goa, K.L. Bendamustine. Drugs 2001, 61, 631–638. [Google Scholar] [CrossRef] [PubMed]
- Cheson, B.D.; Rummel, M.J. Bendamustine: Rebirth of an old drug. J. Clin. Oncol. 2009, 27, 1492–1501. [Google Scholar] [CrossRef] [Green Version]
- Stoehr, E.; Schmeel, F.C.; Schmeel, L.C.; Hänel, M.; Schmidt-Wolf, I.G.H. Bendamustine in heavily pre-treated patients with relapsed or refractory multiple myeloma. J. Cancer Res. Clin. Oncol. 2015, 141, 2205–2212. [Google Scholar] [CrossRef] [PubMed]
- Damaj, G.; Malard, F.; Hulin, C.; Caillot, D.; Garidi, R.; Royer, B.; Marit, G.; Stoppa, A.; Baños, A.; Morineau, N.; et al. Efficacy of bendamustine in relapsed/refractory myeloma patients: Results from the French compassionate use program. Leuk. Lymphoma 2012, 53, 632–634. [Google Scholar] [CrossRef]
- Kim, S.J.; Bang, S.M.; Choi, Y.S.; Jo, D.-Y.; Kim, J.S.; Lee, H.; Eom, H.S.; Yoon, D.H.; Suh, C.; Lee, J.-J.; et al. Bendamustine in heavily pre-treated multiple myeloma patients: Results of a retrospective analysis from the Korean Multiple Myeloma Working Party. Blood Res. 2016, 51, 193–199. [Google Scholar] [CrossRef] [Green Version]
- Poenisch, W.; Rozanski, M.; Goldschmidt, H.; Hoffmann, F.A.; Boldt, T.; Schwarzer, A.; Ritter, U.; Rohrberg, R.; Schwalbe, E.; Uhlig, J.; et al. Combined bendamustine, prednisolone and thalidomide for refractory or relapsed multiple myeloma after autologous stem-cell transplantation or conventional chemotherapy: Results of a Phase I clinical trial. Br. J. Haematol. 2008, 143, 191–200. [Google Scholar] [CrossRef]
- Grey-Davies, E.; Bosworth, J.L.; Boyd, K.D.; Ebdon, C.; Saso, R.; Chitnavis, D.; Mercieca, J.E.; Morgan, G.J.; Davies, F.E. Bendamustine, Thalidomide and Dexamethasone is an effective salvage regimen for advanced stage multiple myeloma. Br. J. Haematol. 2012, 156, 552–555. [Google Scholar] [CrossRef]
- Schey, S.; Brown, S.R.; Tillotson, A.L.; Yong, K.; Williams, C.; Davies, F.; Morgan, G.; Cavenagh, J.; Cook, G.; Cook, M.; et al. Bendamustine, thalidomide and dexamethasone combination therapy for relapsed/refractory myeloma patients: Results of the MUKone randomized dose selection trial. Br. J. Haematol. 2015, 170, 336–348. [Google Scholar] [CrossRef] [PubMed]
- Lau, I.J.; Smith, D.; Aitchison, R.; Blesing, N.; Roberts, P.; Peniket, A.; Yong, K.; Rabin, N.; Ramasamy, K. Bendamustine in combination with thalidomide and dexamethasone is a viable salvage option in myeloma relapsed and/or refractory to bortezomib and lenalidomide. Ann. Hematol. 2015, 94, 643–649. [Google Scholar] [CrossRef]
- Pozzi, S.; Gentile, M.; Sacchi, S.; Marcheselli, R.; Corso, A.; Cocito, F.; Musto, P.; Guarini, A.; Minoia, C.; Vincelli, I.; et al. Bendamustine, Low-dose dexamethasone, and lenalidomide (BdL) for the treatment of patients with relapsed/refractory multiple myeloma confirms very promising results in a phase I/II study. Leuk Lymphoma 2017, 58, 552–559. [Google Scholar] [CrossRef]
- Mey, U.J.M.; Brugger, W.; Schwarb, H.; Pederiva, S.; Schwarzer, A.; Dechow, T.; Jehner, P.; Rauh, J.; Taverna, C.J.; Schmid, M.; et al. Bendamustine, lenalidomide and dexamethasone (BRd) has high activity as 2 nd–line therapy for relapsed and refractory multiple myeloma—A phase II trial. Br. J. Haematol. 2017, 176, 770–782. [Google Scholar] [CrossRef] [PubMed]
- Poenisch, W.; Heyn, S.; Beck, J.; Wagner, I.; Mohren, M.; Hoffmann, F.A.; Lange, T.; Schmalfeld, M.; Zehrfeld, T.; Schwarzer, A.; et al. Lenalidomide, bendamustine and prednisolone exhibits a favourable safety and efficacy profile in relapsed or refractory multiple myeloma: Final results of a phase 1 clinical trial OSHO-#077. Br. J. Haematol. 2013, 162, 202–209. [Google Scholar]
- Lentzsch, S.; O’Sullivan, A.; Kennedy, R.C.; Abbas, M.; Dai, L.; Pregja, S.L.; Burt, S.; Boyiadzis, M.; Roodman, G.D.; Mapara, M.Y.; et al. Combination of bendamustine, lenalidomide, and dexamethasone (BLD) in patients with relapsed or refractory multiple myeloma is feasible and highly effective: Results of phase 1/2 open-label, dose escalation study. Blood 2012, 119, 4608–4613. [Google Scholar] [CrossRef] [PubMed]
- Poenisch, W.; Bourgeois, M.; Moll, B.; Heyn, S.; Jäkel, N.; Wagner, I.; Rohrberg, R.; Hurtz, H.-J.; Schmalfeld, M.; Aßmann, M.; et al. Combined bendamustine, prednisone and bortezomib (BPV) in patients with relapsed or refractory multiple myeloma. J. Cancer Res. Clin. Oncol. 2013, 139, 499–508. [Google Scholar] [CrossRef]
- Ludwig, H.; Kasparu, H.; Leitgeb, C.; Rauch, E.; Linkesch, W.; Zojer, N.; Greil, R.; Seebacher, A.; Pour, L.; Weißmann, A.; et al. Bendamustine-bortezomib-dexamethasone is an active and well-tolerated regimen in patients with relapsed or refractory multiple myeloma. Blood 2014, 123, 985–991. [Google Scholar] [CrossRef] [Green Version]
- Offidani, M.; Corvatta, L.; Maracci, L.; Liberati, A.M.; Ballanti, S.; Attolico, I.; Caraffa, P.; Alesiani, F.; Di Toritto, T.C.; Gentili, S.; et al. Efficacy and tolerability of bendamustine, bortezomib and dexamethasone in patients with relapsed-refractory multiple myeloma: A phase II study. Blood Cancer J. 2013, 3, e162. [Google Scholar] [CrossRef]
- National Comprehensive Cancer Network. Multiple Myeloma (Version 4.2021). Available online: http://www.nccn.org/professionals/physician_gls/pdf/myeloma.pdf (accessed on 10 December 2020).
- Moreau, P.; San Miguel, J.; Sonneveld, P.; Mateos, M.V.; Zamagni, E.; Avet-Loiseau, H.; Hajek, R.; Dimopoulos, M.; Ludwig, H.; Einsele, H.; et al. Multiple Myeloma: ESMO Clinical Practice Guidelines. Ann. Oncol. 2017, 28, iv52–iv61. [Google Scholar] [CrossRef]
- Michael, M.; Bruns, I.; Boelke, E.; Zohren, F.; Czibere, A.; Safaian, N.N.; Neumann, F.; Haas, R.; Kobbe, G.; Fenk, R. Bendamustine in patients with relapsed or refractory multiple myeloma. Eur. J. Med. Res. 2010, 15, 13–19. [Google Scholar] [CrossRef] [Green Version]
- Knop, S.; Straka, C.; Haen, M.; Schwedes, R.; Hebart, H.; Einsele, H. The efficacy and toxicity of bendamustine in recurrent multiple myeloma after high-dose chemotherapy. Haematologica 2005, 90, 1287–1288. [Google Scholar] [PubMed]
- Sivaraj, D.; Green, M.M.; Kang, Y.; Long, D.G.; Rizzieri, A.D.; Li, G.Z.; Garrett, H.A.; McIntyre, L.J.; Chao, J.N.; Gasparetto, C. Bendamustine, pomalidomide, and dexamethasone for relapsed and/or refractory multiple myeloma. Blood Cancer J. 2018, 8, 71. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dhakal, B.; D’Souza, A.; Hamadani, M.; Arce-Lara, C.; Schroeder, K.; Chhabra, S.; Shah, N.N.; Gauger, K.; Keaton, T.; Pasquini, M.; et al. Phase I/II trial of bendamustine, ixazomib, and dexamethasone in relapsed/refractory multiple myeloma. Blood Cancer J. 2019, 9, 56. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, H.C.; Feng, L.; Oriabure, O.; Graham, V.; Chen, W.; Badillo, M.; Lu, R.; Lee, H.J.; Jain, P.; Manasanch, E.E.; et al. A phase one trial of carfilzomib, bendamustine, and dexamethasone in relapsed and/or refractory multiple myeloma. Am. J. Hematol. 2021, 96, 243–246. [Google Scholar] [CrossRef] [PubMed]
- Leng, S.; Bhutani, D.; Raza, S.; Assal, A.; Pan, S.; Hu, J.; Wei, A.; Mapara, M.; Lentzsch, S. Phase I/II study of carfilzomib, bendamustine, and dexamethasone (CBD) in patients with newly diagnosed multiple myeloma. Blood Cancer J. 2020, 10, 13. [Google Scholar] [CrossRef] [Green Version]

| All Patients | Bendamustine + Steroid | Bendamustine + Steroid + Thal/Len/Bort | |
|---|---|---|---|
| n = 105 | n = 52 | n = 53 | |
| Sex | |||
| male | 50 (48%) | 24 (46%) | 26 (49%) |
| female | 55 (52%) | 28 (54%) | 27 (51%) |
| Age | |||
| median (range) | 64 (45–84) | 64 (45–82) | 65 (49–84) |
| >65 years | 43 (41%) | 22 (42%) | 21 (40%) |
| Monoclonal protein type | |||
| IgG | 68 (65%) | 35 (67%) | 33 (62%) |
| IgA | 25 (24%) | 12 (23%) | 13 (25%) |
| LCD | 12 (11%) | 5 (10%) | 7 (13%) |
| Light chain | |||
| kappa | 61 (58%) | 31 (60%) | 30 (57%) |
| lambda | 44 (42%) | 21 (40%) | 23 (43%) |
| ISS stage | |||
| I | 20 (19%) | 11 (21%) | 9 (17%) |
| II | 31 (30%) | 16 (31%) | 15 (28%) |
| III | 54 (51%) | 25 (48%) | 29 (55%) |
| Cytogenetics | |||
| unknown | 84 (80%) | 43 (83%) | 41 (77%) |
| del(17p13) | 5 (24%) 1 | 3 (33%) 1 | 2 (17%) 1 |
| del(13q14) | 3 (14%) 1 | 1 (11%) 1 | 2 (17%) 1 |
| t(4;14)(p16;q32) | 4 (19%) 1 | 1 (11%) 1 | 3 (25%) 1 |
| t(14;16)(q32;q23) | 1 (5%) 1 | 1 (11%) 1 | 0 (0%) 1 |
| Serum monoclonal protein | 23.9 (0.87–95.6) | 22.7 (0.87–95.6) | 25.2 (1.37–84.56) |
| (g/L; median, range) | |||
| Bone marrow plasma cells | 46.5 (7–90) | 48.0 (10–90) | 42.5 (7–85) |
| (%; median, range) | |||
| Albumin | 3.6 (2.01–5.33) | 3.6 (2.01–5.33) | 3.6 (2.01–5.33) |
| (g/dL; median, range) | |||
| Beta-2-microglobulin | 4.93 (1.1–40) | 4.65 (2.5–36) | 5.11 (1.1–40) |
| (mg/L; median, range) | |||
| LDH | 211 (75–2205) | 238 (75–1872) | 281 (107–2205) |
| (IU/L; median, range) | |||
| Creatinine | 0.965 (0.4–9.33) | 1.053 (0.64–7.83) | 0.924 (0.4–9.33) |
| (mg/dL; median, range) | |||
| eGFR | 69 (5.37–185.9) | 68 (5.37–152.7) | 71 (9.5–185.9) |
| (mL/min; median, range) | |||
| Calcium | 2.37 (1.79–4.94) | 2.45 (2.08–4.94) | 2.29 (1.79–4.63) |
| (mmol/L; median, range) | |||
| Neutrophils | 3.08 (0.59–8.87) | 3.42 (1.15–8.87) | 2.84 (0.59–7.59) |
| (G/L; median, range) | |||
| Hemoglobin | 10.25 (6.6–14.7) | 10.13 (6.6–14.7) | 10.37 (6.9–13.6) |
| (g/dL; median, range) | |||
| Platelets | 159 (22–409) | 149 (22–378) | 161 (29–409) |
| (G/L; median, range) | |||
| Previous treatment | |||
| Number of lines (median, range) | 3 (1–8) | 3 (1–8) | 3 (1–6) |
| IMID | 100 (95%) | 51(98%) | 49 (92%) |
| Thalidomide | 93 (89%) | 48 (92%) | 45 (85%) |
| Lenalidomide | 56 (53%) | 38 (73%) 2 | 18 (34%) 2 |
| PI | 92 (88%) | 48 (92%) | 44 (83%) |
| Bortezomib | 83 (79%) | 45 (87%) | 38 (72%) |
| Carfilzomib | 9 (9%) | 3 (6%) | 5 (9%) |
| Lenalidomide + Bortezomib | 51 (49%) | 37 (71%) 2 | 14 (26%) 2 |
| IMID + PI | 91 (87%) | 47 (90%) | 44 (83%) |
| ASCT | 33 (31%) | 16 (31%) | 17 (32%) |
| All Patients n = 105 | Bendamustine + Steroid n = 52 | Bendamustine + Steroid + Thal/Len/Bort n = 53 | |
|---|---|---|---|
| ORR | 48 (46%) | 19 (37%) | 29 (55%) |
| CR | 8 (8%) | 1 (2%) * | 7 (13%) * |
| VGPR | 9 (9%) | 3 (6%) | 6 (11%) |
| CR + VGPR | 17 (16%) | 4 (8%) * | 13 (25%) * |
| PR | 31 (30%) | 15 (29%) | 16 (30%) |
| SD | 37 (35%) | 24 (46%) | 13 (25%) |
| PD | 20 (19%) | 9 (17%) | 11 (21%) |
| Bendamustine + Steroid n = 52 | Bendamustine + Steroid + Len/Thal/Bort n = 53 | |||
|---|---|---|---|---|
| All grades | Grade ≥3 | All grades | Grade ≥3 | |
| Neutropenia | 28 (54%) | 15 (29%) | 32 (60%) | 25 (47%) |
| Anemia | 18 (35%) | 11 (21%) | 21 (40%) | 6 (11%) |
| Thrombocytopenia | 17 (33%) | 10 (19%) | 15 (28%) | 10 (19%) |
| Infection | 27 (52%) | 10 (19%) | 20 (38%) | 5 (11%) |
| Gastrointestinal toxicity | 5 (10%) | 0 (0%) | 3 (7%) | 2 (4%) |
| Thromboembolism | 1 (2%) | 1 (2%) | 2 (4%) | 1 (2%) |
| Neuropathy | 0 (0%) | 0 (0%) | 1 (2%) | 0 (0%) |
| Renal function impairment | 2 (4%) | 0 (0%) | 1 (2%) | 0 (0%) |
| Hepatic toxisity | 0 (0%) | 0 (0%) | 1 (2%) | 0 (0%) |
| Hypertension | 1 (2%) | 1 (2%) | 0 (0%) | 0 (0%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grzasko, N.; Charlinski, G.; Morawska, M.; Kicinski, P.; Waszczuk-Gajda, A.; Drozd-Sokolowska, J.; Subocz, E.; Blonska, D.; Razny, M.; Druzd-Sitek, A.; et al. Bendamustine-Based Regimens as Salvage Therapy in Refractory/Relapsed Multiple Myeloma Patients: A Retrospective Real-Life Analysis by the Polish Myeloma Group. J. Clin. Med. 2021, 10, 5504. https://doi.org/10.3390/jcm10235504
Grzasko N, Charlinski G, Morawska M, Kicinski P, Waszczuk-Gajda A, Drozd-Sokolowska J, Subocz E, Blonska D, Razny M, Druzd-Sitek A, et al. Bendamustine-Based Regimens as Salvage Therapy in Refractory/Relapsed Multiple Myeloma Patients: A Retrospective Real-Life Analysis by the Polish Myeloma Group. Journal of Clinical Medicine. 2021; 10(23):5504. https://doi.org/10.3390/jcm10235504
Chicago/Turabian StyleGrzasko, Norbert, Grzegorz Charlinski, Marta Morawska, Pawel Kicinski, Anna Waszczuk-Gajda, Joanna Drozd-Sokolowska, Edyta Subocz, Danuta Blonska, Malgorzata Razny, Agnieszka Druzd-Sitek, and et al. 2021. "Bendamustine-Based Regimens as Salvage Therapy in Refractory/Relapsed Multiple Myeloma Patients: A Retrospective Real-Life Analysis by the Polish Myeloma Group" Journal of Clinical Medicine 10, no. 23: 5504. https://doi.org/10.3390/jcm10235504
APA StyleGrzasko, N., Charlinski, G., Morawska, M., Kicinski, P., Waszczuk-Gajda, A., Drozd-Sokolowska, J., Subocz, E., Blonska, D., Razny, M., Druzd-Sitek, A., Holojda, J., Swiderska, A., Usnarska-Zubkiewicz, L., Masternak, A., & Giannopoulos, K. (2021). Bendamustine-Based Regimens as Salvage Therapy in Refractory/Relapsed Multiple Myeloma Patients: A Retrospective Real-Life Analysis by the Polish Myeloma Group. Journal of Clinical Medicine, 10(23), 5504. https://doi.org/10.3390/jcm10235504







