Prevalence of Sarcopenia Employing Population-Specific Cut-Points: Cross-Sectional Data from the Geelong Osteoporosis Study, Australia
Abstract
1. Introduction
2. Methods
2.1. Study Design
2.2. Participants
2.3. Measures
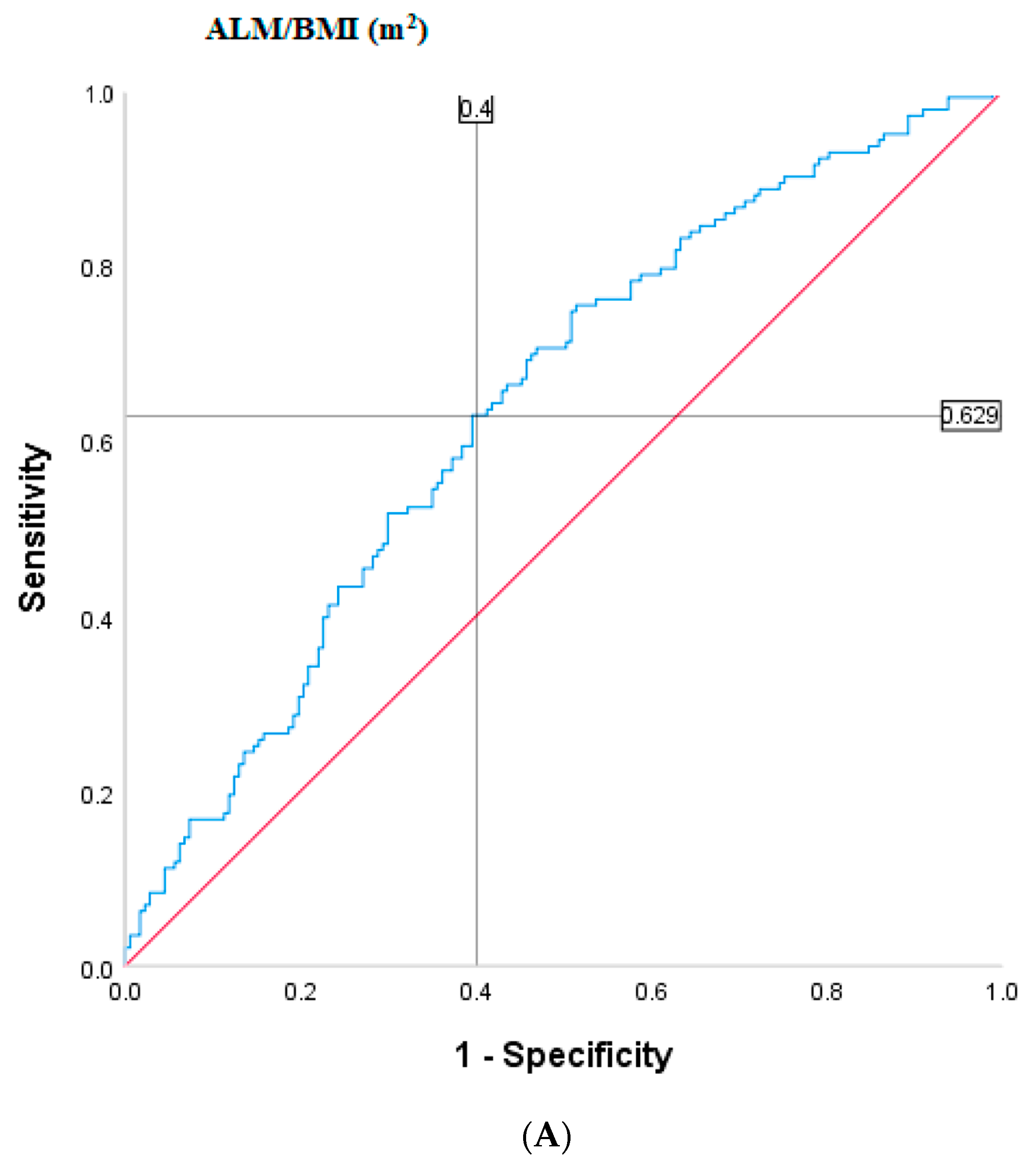
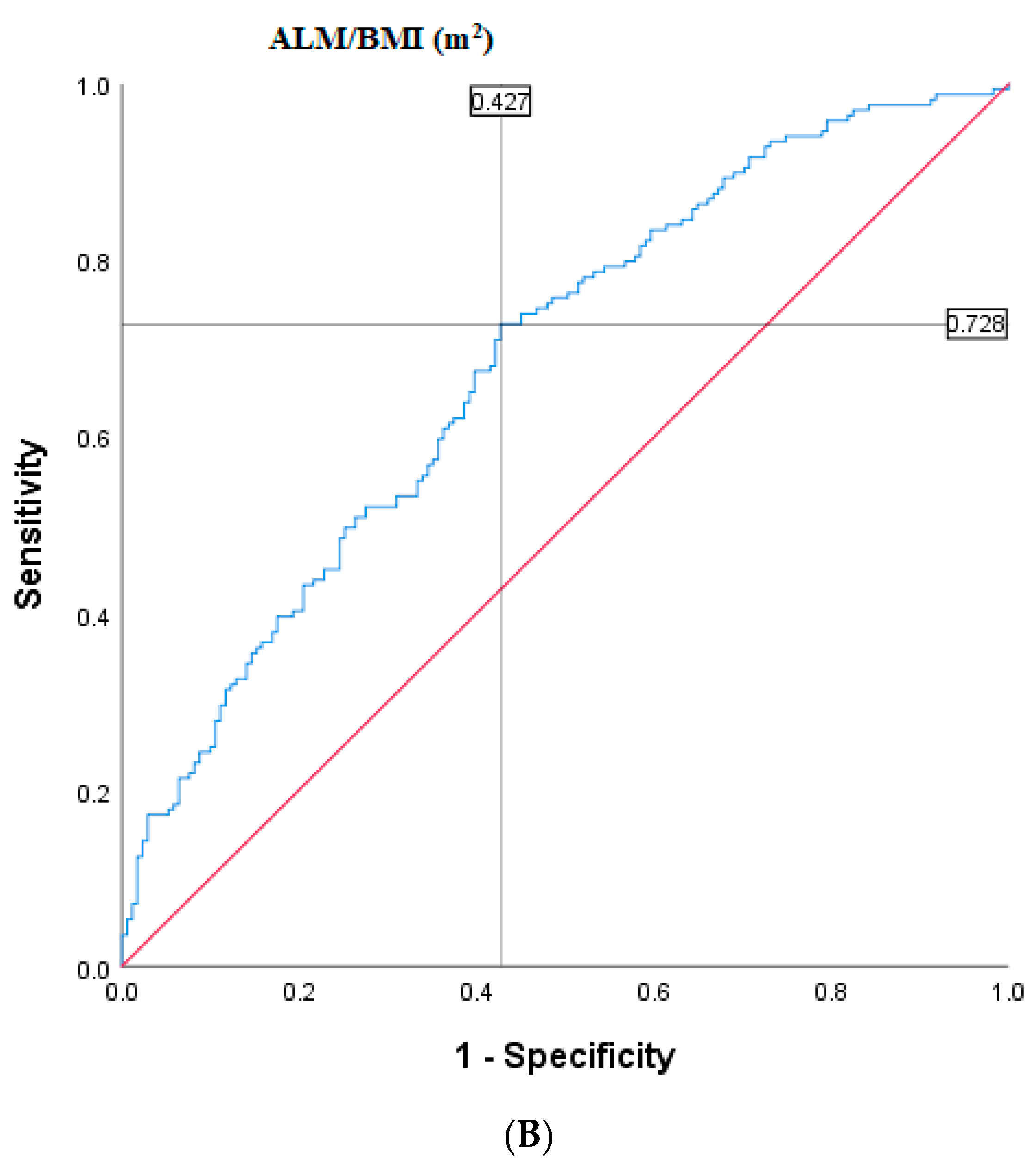
2.4. Population-Specific Cut-Points
2.5. Sarcopenia Ascertainment
2.6. Statistical Analysis
3. Results
3.1. Participant Characteristics
3.2. Sarcopenia Prevalence in Men Compared with Women
3.3. Age-Standardized Estimates of Sarcopenia
3.4. Agreement
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Ethical Approval
Abbreviations
| ALM | Appendicular lean mass |
| ANZSSFR | Australian and New Zealand Society for Sarcopenia and Frailty Research |
| BMI | Body mass index |
| CI | Confidence interval |
| DXA | Dual-energy X-ray absorptiometry |
| EWGSOP | European Working Group on Sarcopenia in Older People |
| FNIH | Foundation for the National Institutes of Health |
| GOS | Geelong Osteoporosis Study |
| GS | Gait speed |
| HGS | Handgrip strength |
| TUG | Timed up-and-go |
| ROC | Receiver operating characteristic |
Appendix A
References
- Fielding, R.A.; Vellas, B.; Evans, W.J.; Bhasin, S.; Morley, J.E.; Newman, A.B.; Van Kan, G.A.; Andrieu, S.; Bauer, J.; Breuille, D.; et al. Sarcopenia: An undiagnosed condition in older adults. Current consensus definition: Prevalence, etiology, and consequences. International Working Group on Sarcopenia. J. Am. Med. Dir. Assoc. 2011, 12, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Sim, M.; Prince, R.L.; Scott, D.; Daly, R.M.; Duque, G.; Inderjeeth, C.A.; Zhu, K.; Woodman, R.J.; Hodgson, J.M.; Lewis, J.R. Sarcopenia definitions and their associations with mortality in older Australian women. J. Am. Med. Dir. Assoc. 2019, 20, 76–82.e2. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Baeyens, J.P.; Bauer, J.M.; Boirie, Y.; Cederholm, T.; Landi, F.; Martin, F.C.; Michel, J.-P.; Rolland, Y.; Schneider, S.M.; et al. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing 2010, 39, 412–423. [Google Scholar] [CrossRef] [PubMed]
- Studenski, S.A.; Peters, K.W.; Alley, D.E.; Cawthon, P.M.; McLean, R.R.; Harris, T.B.; Ferrucci, L.; Guralnik, J.M.; Fragala, M.S.; Kenny, A.M.; et al. The FNIH Sarcopenia Project: Rationale, study description, conference recommendations, and final estimates. J. Gerontol. Ser. A Boil. Sci. Med. Sci. 2014, 69, 547–558. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyère, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef] [PubMed]
- Pagotto, V.; Silveira, E.A. Applicability and agreement of different diagnostic criteria for sarcopenia estimation in the elderly. Arch. Gerontol. Geriatr. 2014, 59, 288–294. [Google Scholar] [CrossRef]
- Dam, T.-T.; Peters, K.W.; Fragala, M.; Cawthon, P.M.; Harris, T.B.; McLean, R.; Shardell, M.; Alley, D.E.; Kenny, A.; Ferrucci, L.; et al. An evidence-based comparison of operational criteria for the presence of sarcopenia. J. Gerontol. Ser. A: Boil. Sci. Med. Sci. 2014, 69, 584–590. [Google Scholar] [CrossRef]
- Sui, S.X.; Holloway-Kew, K.L.; Hyde, N.K.; Williams, L.J.; Tembo, M.C.; Leach, S.; Pasco, J.A. Definition-specific prevalence estimates for sarcopenia in Australian population: The Geelong Osteoporosis Study. JCSM Clin. Rep. 2020, 5, 89–98. [Google Scholar]
- Bahat, G.; Tufan, A.; Tufan, F.; Kilic, C.; Akpinar, T.S.; Kose, M.; Erten, N.; Karan, M.A.; Cruz-Jentoft, A.J. Cut-off points to identify sarcopenia according to European Working Group on Sarcopenia in Older People (EWGSOP) definition. Clin. Nutr. 2016, 35, 1557–1563. [Google Scholar] [CrossRef]
- Breiman, L.; Friedman, J.H.; Olshen, R.A.; Stone, C.J. Classification and Regression Trees; Wadsworth International Group: Belmont, CA, USA, 1984. [Google Scholar]
- Gould, H.; Brennan, S.L.; Kotowicz, M.A.; Nicholson, G.C.; Pasco, J.A. Total and appendicular lean mass reference ranges for Australian men and women: The Geelong Osteoporosis Study. Calcif. Tissue Int. 2014, 94, 363–372. [Google Scholar] [CrossRef]
- Zanker, J.; Scott, D.; Reijnierse, E.M.; Brennan-Olsen, S.L.; Daly, R.M.; Girgis, C.M.; Grossmann, M.; Hayes, A.; Henwood, T.; Hirani, V.; et al. Establishing an operational definition of sarcopenia in Australia and New Zealand: Delphi method based consensus statement. J. Nutr. Health Aging 2019, 23, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Pasco, J.A.; Nicholson, G.C.; Kotowicz, M.A. Cohort profile: Geelong Osteoporosis Study. Int. J. Epidemiol. 2011, 41, 1565–1575. [Google Scholar] [CrossRef] [PubMed]
- Bischoff, H.A.; Stähelin, H.B.; Monsch, A.U.; Iversen, M.D.; Weyh, A.; Von Dechend, M.; Akos, R.; Conzelmann, M.; Dick, W.; Theiler, R. Identifying a cut-off point for normal mobility: A comparison of the timed ‘up and go’ test in community-dwelling and institutionalised elderly women. Age Ageing 2003, 32, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Pasco, J.A.; Holloway-Kew, K.L.; Tembo, M.C.; Sui, S.X.; Anderson, K.B.; Rufus-Membere, P.; Hyde, N.K.; Williams, L.J.; Kotowicz, M.A. Normative data for lean mass using FNIH criteria in an Australian setting. Calcif. Tissue Int. 2018, 104, 475–479. [Google Scholar] [CrossRef] [PubMed]
- Sui, S.X.; Holloway-Kew, K.L.; Hyde, N.K.; Williams, L.J.; Tembo, M.C.; Mohebbi, M.; Gojanovic, M.; Leach, S.; Pasco, J.A. Handgrip strength and muscle quality in Australian women: Cross-sectional data from the Geelong Osteoporosis Study. J. Cachex Sarcopenia Muscle 2020, 11, 690–697. [Google Scholar] [CrossRef] [PubMed]
- Pasco, J.A.; Stuart, A.L.; Holloway-Kew, K.L.; Tembo, M.C.; Sui, S.X.; Anderson, K.B.; Hyde, N.K.; Williams, L.J.; Kotowicz, M.A. Lower-limb muscle strength: Normative data from an observational population-based study. BMC Musculoskelet. Disord. 2020, 21, 89. [Google Scholar] [CrossRef]
- Cawthon, P.; Peters, K.W.; Shardell, M.D.; McLean, R.R.; Dam, T.-T.L.; Kenny, A.M.; Fragala, M.S.; Harris, T.B.; Kiel, D.P.; Guralnik, J.M.; et al. Cutpoints for low appendicular lean mass that identify older adults with clinically significant weakness. J. Gerontol. Ser. A Boil. Sci. Med. Sci. 2014, 69, 567–575. [Google Scholar] [CrossRef]
- Unal, I. Defining an optimal cut-point value in ROC analysis: An alternative approach. Comput. Math. Methods Med. 2017, 2017, 1–14. [Google Scholar] [CrossRef]
- Census of Population and Ageing: Age by Sex. Australian Bureau of Statistics. 2011. Available online: www.abs.gov.au (accessed on 19 November 2019).
- Auyeung, T.W.; Lee, S.W.J.; Leung, J.; Kwok, T.; Woo, J. Age-associated decline of muscle mass, grip strength and gait speed: A 4-year longitudinal study of 3018 community-dwelling older Chinese. Geriatr. Gerontol. Int. 2014, 14, 76–84. [Google Scholar] [CrossRef]
- Yoshida, D.; Suzuki, T.; Shimada, H.; Park, H.; Makizako, H.; Doi, T.; Anan, Y.; Tsutsumimoto, K.; Uemura, K.; Ito, T.; et al. Using two different algorithms to determine the prevalence of sarcopenia. Geriatr. Gerontol. Int. 2014, 14, 46–51. [Google Scholar] [CrossRef]
- Lees, M.; Wilson, O.J.; Hind, K.; Ispoglou, T. Muscle quality as a complementary prognostic tool in conjunction with sarcopenia assessment in younger and older individuals. Graefe’s Arch. Clin. Exp. Ophthalmol. 2019, 119, 1171–1181. [Google Scholar] [CrossRef]
- Mayhew, A.J.; Amog, K.; Phillips, S.; Parise, G.; McNicholas, P.D.; De Souza, R.J.; Thabane, L.; Raina, P. The prevalence of sarcopenia in community-dwelling older adults, an exploration of differences between studies and within definitions: A systematic review and meta-analyses. Age Ageing 2019, 48, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Masanés, F.; Luque, X.R.I.; Salvà, A.; Serra-Rexach, J.A.; Artaza, I.; Formiga, F.; Cuesta, F.; Soto, A.L.; Ruiz, D.; Cruz-Jentoft, A.J. Cut-off points for muscle mass—not grip strength or gait speed—determine variations in sarcopenia prevalence. J. Nutr. Heal. Aging 2017, 21, 825–829. [Google Scholar] [CrossRef]
| Population-Specific Cut-Points | ||
|---|---|---|
| Women | Men | |
| ALM/height2 + HGS | <5.30 kg/m2 + <16 kg | <6.94 kg/m2 + <31 kg |
| ALM/height2 + TUG | <5.30 kg/m2 + >9.3 s | <6.94 kg/m2 + >9.3 s |
| ALM/height2 + GS | - | <6.94 kg/m2 + <0.8 m/s |
| ALM/BMI + HGS | <0.512 m2 + <16 kg | <0.827 m2 + <31 kg |
| ALM/BMIROC + HGS | <0.579 m2 + <16 kg | 0.913 m2 + <31 kg |
| International Cut-Points | ||
| ALM/height2EWGSOP1 + HGS (3) | <5.67 kg/m2 + <20 kg | <7.23 kg/m2 + <30 kg |
| ALM/height2EWGSOP1 + GS (3) | - | <7.23 kg/m2 + <0.8 m/s |
| ALM/height2EWGSOP2 + HGS (5) | <5.5 kg/m2 + <16 kg | <7.0 kg/m2 + <27 kg |
| ALM/height2EWGSOP2 + TUG (5) | <5.5 kg/m2 + >20 s | <7.0 kg/m2 + >20 s |
| ALM/BMIFNIH + HGS (4) | <0.512 m2 + <16 kg | <0.789 m2 + <26 kg |
| Women (n = 323) | Men (n = 342) | |
|---|---|---|
| Age (yr) | 70 (64–75) | 70 (66–78) |
| Weight (kg) | 74.0 (±15.4) | 83.9 (±13.8) |
| Height (m) | 1.59 (±0.06) | 1.73 (±0.07) |
| BMI (kg/m2) | 29.0 (±5.8) | 28.0 (±4.1) |
| HGS (kg) | 21 (±6) | 36 (±6) |
| ALM/height2 (kg/m2) | 6.60 (±0.79) | 8.25 (±0.93) |
| ALM/BMI (m2) | 0.593 (±0.102) | 0.888 (±0.124) |
| TUG (s) | 9.1 (7.9–10.8) | 9.2 (8.0–10.7) |
| Gait speed (m/s) | - | 0.9 (±0.2) |
| Criteria | 60–69 yr | 70–79 yr | ≥80 yr | All | Standardized Prevalence | |||
|---|---|---|---|---|---|---|---|---|
| Women | n = 151 | n = 124 | n = 48 | n = 323 | ||||
| n, %, | 95%CI | n, %, | 95%CI | n, %, | 95%CI | n, %, | Mean (%, 95%CI) | |
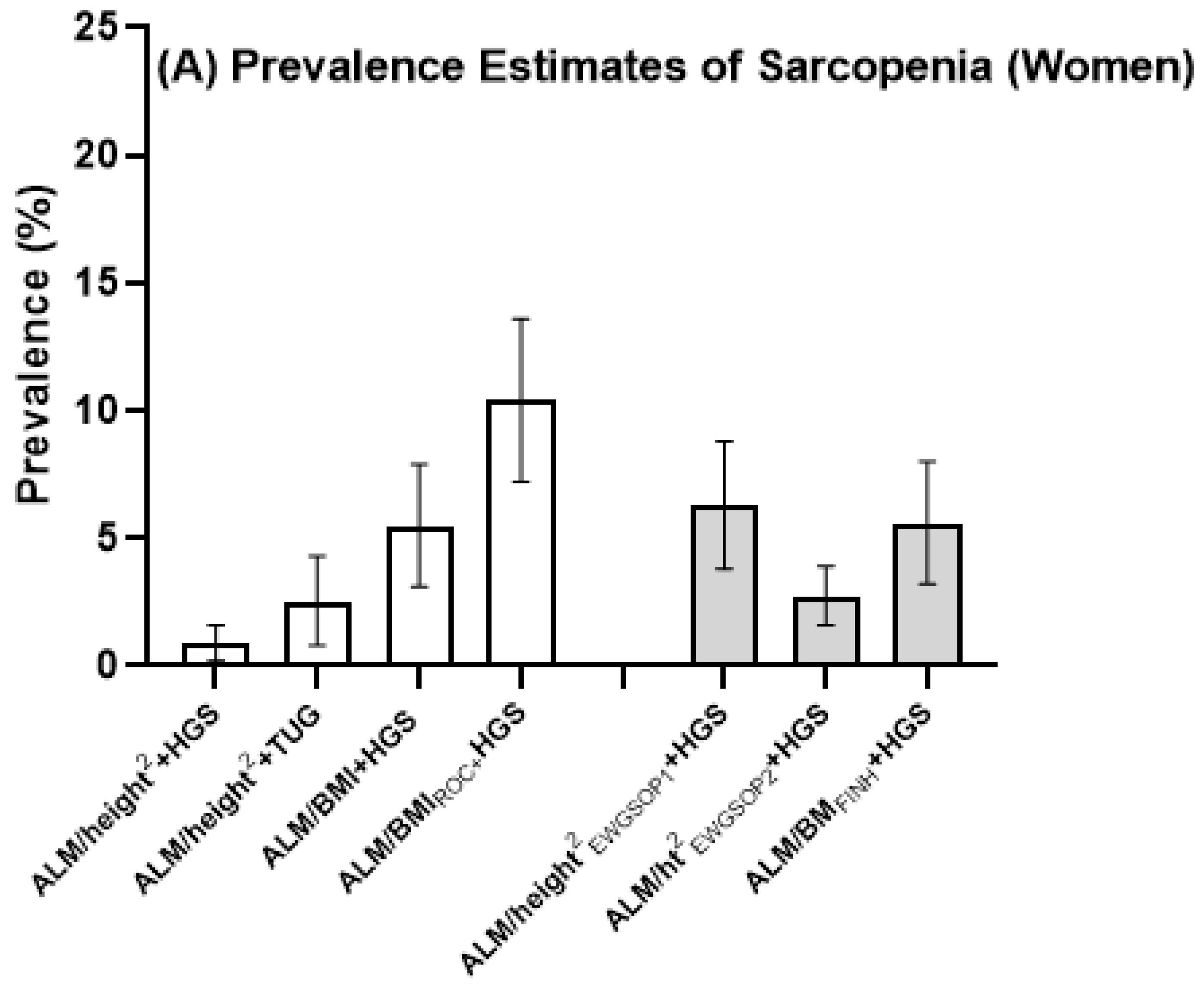
| ALM/height2 + HGS | 0 (0) | - | 2 (1.6) | - | 1 (2.1) | - | 3 (0.9) | 0.9 (0.2–1.6) |
| ALM/height2 + TUG | 3 (2.0) | 0.6–5.6 | 3(2.4) | 0.7–6.7 | 2 (4.3) | 0.9–13.9 | 8 (2.7) | 2.5 (0.8–4.3) |
| ALM/BMI + HGS | 3 (2.0) | 0.6–5.5 | 5 (4.0) | 1.6–8.9 | 8 (16.7) | 8.2–29.3 | 16 (5.0) | 5.5 (3.1–7.9) |
| ALM/BMIROC + HGS | 5 (3.3) | 1.3–7.7 | 8 (6.5) | 3.3–12.4 | 16 (33.3) | 21.5–47.7 | 29 (9.0) | 10.4 (7.2–13.6) |
| Severe sarcopenia | ||||||||
| ALM/height2 + HGS + TUG | 1 (0.8) | - | 1 (2.1) | - | 1 (2.1) | - | 2 (0.6) | 1.0 (0–2.1) |
| ALM/BMI + HGS + TUG | 2 (1.3) | 0.3–5.2 | 3 (2.4) | 0.7–7.2 | 8 (17.0) | 8.7–30.5 | 13 (4.1) | 4.8 (2.6–6.9) |
| ALM/BMIROC + HGS + TUG | 4 (2.7) | 1.0–6.9 | 5 (4.0) | 1.7–9.3 | 15 (31.9) | 20.2–46.4 | 24 (7.5) | 8.9 (6.0–11.8) |
| Men | n = 152 | n = 117 | n = 73 | n = 342 | ||||
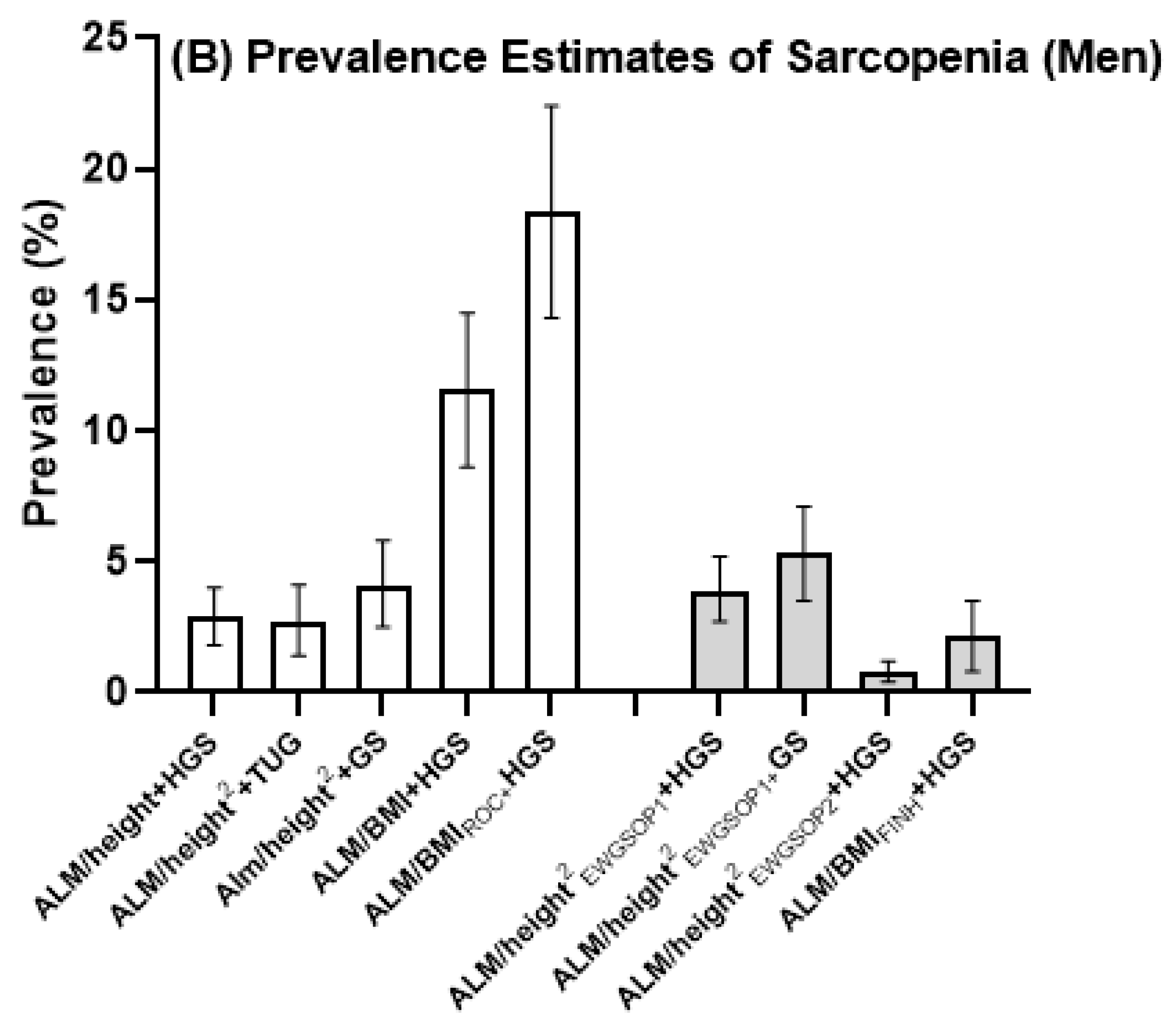
| ALM/height2 + HGS | 0 (0) | - | 3 (2.6) | - | 8 (11.1) | - | 11 (3.2) | 2.9 (1.8–4.0) |
| ALM/height2 + TUG | 2 (1.3) | 0.3–4.6 | 1 (0.9) | 0.09–4.8 | 12 (16.4) | 9.3–26.3 | 15 (4.4) | 4.1 (2.4–5.8) |
| ALM/height2 + GS | 0 | - | 0 | - | 6 (8.3) | - | 6 (1.8) | 1.6 (1.0–2.2) |
| ALM/BMI + HGS | 4 (2.6) | 0.9–6.4 | 13 (11.2) | 6.4–18.0 | 26 (36.1) | 25.7–47.6 | 43 (12.6) | 11.6 (8.6–14.5) |
| ALM/BMIROC + HGS | 13 (8.6) | 5.0–14.2 | 21 (18.1) | 12.1–26.1 | 33 (45.8) | 34.7–57.4 | 67 (19.7) | 18.4 (14.3–22.4) |
| Severe sarcopenia | ||||||||
| ALM/height2 + HGS + TUG | 0 | - | 0 | - | 7 (9.7%) | - | 7 (2.1) | 1.9 (1.3–2.5) |
| ALM/BMI + HGS + TUG | 2 (1.3) | 0.3–5.2 | 9 (7.8) | 4.1–14.2 | 25 (34.7) | 24.7–46.4 | 36 (10.7) | 9.5 (7.0–12.0) |
| ALM/BMIROC + HGS + TUG | 4 (2.7) | 1.0–6.9 | 14 (12.1) | 7.3–19.4 | 32 (44.4) | 33.4–56.0 | 50 (14.8) | 13.4 (10.3–16.6) |
| Women | ALM/height2EWGSOP1 + HGS | ALM/height2 EWGSOP2 + HGS | ALM/BMI FNIH + HGS | ALM/height2 + HGS | ALM/height2 + TUG | ALM/BMI + HGS | ||
|---|---|---|---|---|---|---|---|---|
| ALM/height2 + HGS | 0.2 (0.0–0.5) | 0.4 (0.0–0.7) | 0.1 (−0.1–0.2) | - | ||||
| p-value | <0.001 | <0.001 | 0.02 | |||||
| ALM/height2 + TUG | 0.3 (0.0–0.5) | 0.2 (0.0–0.4) | 0.1 (0.1–0.2) | 0.4 (0–0.7) | - | |||
| p-value | <0.001 | 0.001 | 0.32 | <0.001 | ||||
| ALM/BMI + HGS | 0.2 (0.0–0.4) | 0.2 (0.0–0.4) | 1 | 0.1 (0.0–0.3) | 0.1 (−0.1–0.2) | - | ||
| p-value | 0.001 | 0.001 | <0.001 | 0.02 | 0.3 | |||
| ALM/BMIROC + HGS | 0.3 (0.1–0.5) | 0.3 (0.1–0.5) | 0.7 (0.5–0.8) | 0.1 (0.0–0.3) | 0.1 (0.0–0.2) | 0.7 (0.5–0.8) | ||
| p-value | <0.001 | <0.001 | <0.001 | <0.001 | 0.11 | <0.001 | ||
| Men | ALM/height2 EWGSOP1 + HGS | ALM/height2 EWGSOP1 + GS | ALM/height2 EWGSOP2 + HGS | ALM/BMI FNIH + HGS | ALM/height2 + HGS | ALM/height2 + TUG | ALM/heigh2 + GS | ALM/BMI + HGS |
| ALM/height2 + HGS | 0.8 (0.6–1.0) | 0.4 (0.2–0.7) | 0.4 (0.1–0.7) | 0.1 (-0.1–0.3) | - | |||
| p-value | <0.001 | <0.001 | <0.001 | 0.13 | ||||
| ALM/height2 + TUG | 0.4 (0.2–0.7) | 0.5 (0.4–0.7) | 0.3 (0.0–0.6) | 0.1 (−0.1–0.2) | 0.5 (0.3–0.8) | - | ||
| p-value | <0.001 | <0.001 | <0.001 | 0.26 | <0.001 | |||
| ALM/height2 + GS | 0.5 (0.3–0.8) | 0.7 (0.4–0.9) | 0.5 (0.1–0.8) | 0.1 (−0.1–0.3) | 0.7 (0.4–0.9) | 0.8 (0.6–1.0) | - | |
| p-value | <0.001 | <0.001 | <0.001 | 0.11 | <0.001 | <0.001 | ||
| ALM/BMI + HGS | 0.2 (0.0–0.3) | 0.1 (0.0–0.3) | 0.02 (0.0–0.1) | 0.3 (0.1–0.4) | 0.1 (0.0–0.3) | 0.0, (−0.8–0.2) | 0.07 (0–0.2) | - |
| p-value | <0.001 | 0.02 | 0.30 | <0.001 | 0.001 | 0.39 | 0.09 | |
| ALM/BMIROC + HGS | 0.2 (0.1–0.3) | 0.2 (0.0–0.3) | 0.0 (0.0–0.1) | 0.2 (0.1–0.3) | 0.1 (0.0–0.1) | 0.0 (0–0.1) | 0.1 (0.0–0.2) | 0.7 (0.6–0.8) |
| p-value | <0.001 | 0.001 | 0.04 | <0.001 | 0.172 | 0.182 | 0.02 | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sui, S.X.; Holloway-Kew, K.L.; Hyde, N.K.; Williams, L.J.; Tembo, M.C.; Leach, S.; Pasco, J.A. Prevalence of Sarcopenia Employing Population-Specific Cut-Points: Cross-Sectional Data from the Geelong Osteoporosis Study, Australia. J. Clin. Med. 2021, 10, 343. https://doi.org/10.3390/jcm10020343
Sui SX, Holloway-Kew KL, Hyde NK, Williams LJ, Tembo MC, Leach S, Pasco JA. Prevalence of Sarcopenia Employing Population-Specific Cut-Points: Cross-Sectional Data from the Geelong Osteoporosis Study, Australia. Journal of Clinical Medicine. 2021; 10(2):343. https://doi.org/10.3390/jcm10020343
Chicago/Turabian StyleSui, Sophia X., Kara L. Holloway-Kew, Natalie K. Hyde, Lana J. Williams, Monica C. Tembo, Sarah Leach, and Julie A. Pasco. 2021. "Prevalence of Sarcopenia Employing Population-Specific Cut-Points: Cross-Sectional Data from the Geelong Osteoporosis Study, Australia" Journal of Clinical Medicine 10, no. 2: 343. https://doi.org/10.3390/jcm10020343
APA StyleSui, S. X., Holloway-Kew, K. L., Hyde, N. K., Williams, L. J., Tembo, M. C., Leach, S., & Pasco, J. A. (2021). Prevalence of Sarcopenia Employing Population-Specific Cut-Points: Cross-Sectional Data from the Geelong Osteoporosis Study, Australia. Journal of Clinical Medicine, 10(2), 343. https://doi.org/10.3390/jcm10020343





