Emerging Comorbidities in Inflammatory Bowel Disease: Eating Disorders, Alcohol and Narcotics Misuse
Abstract
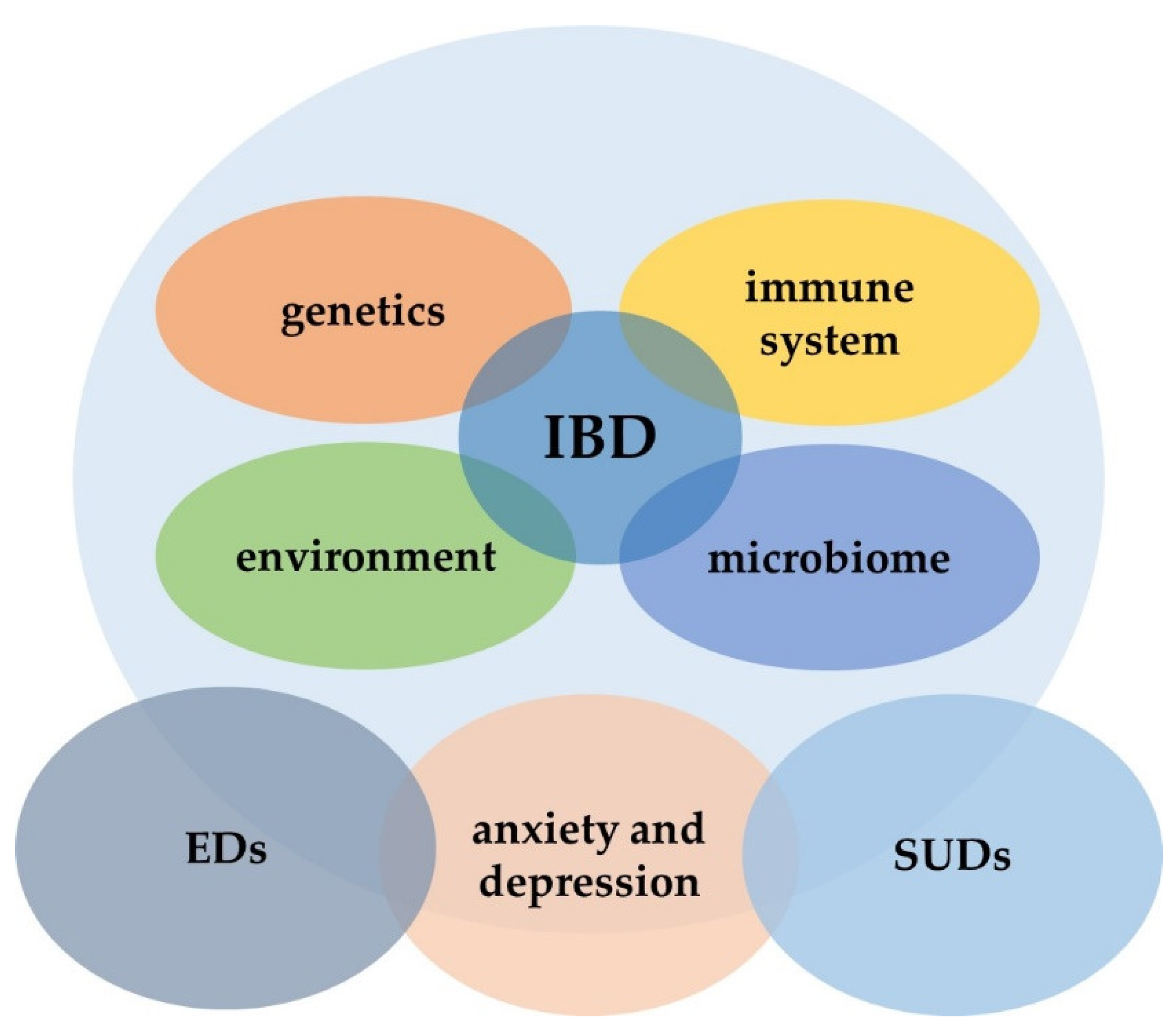
:1. Introduction
2. Materials and Methods
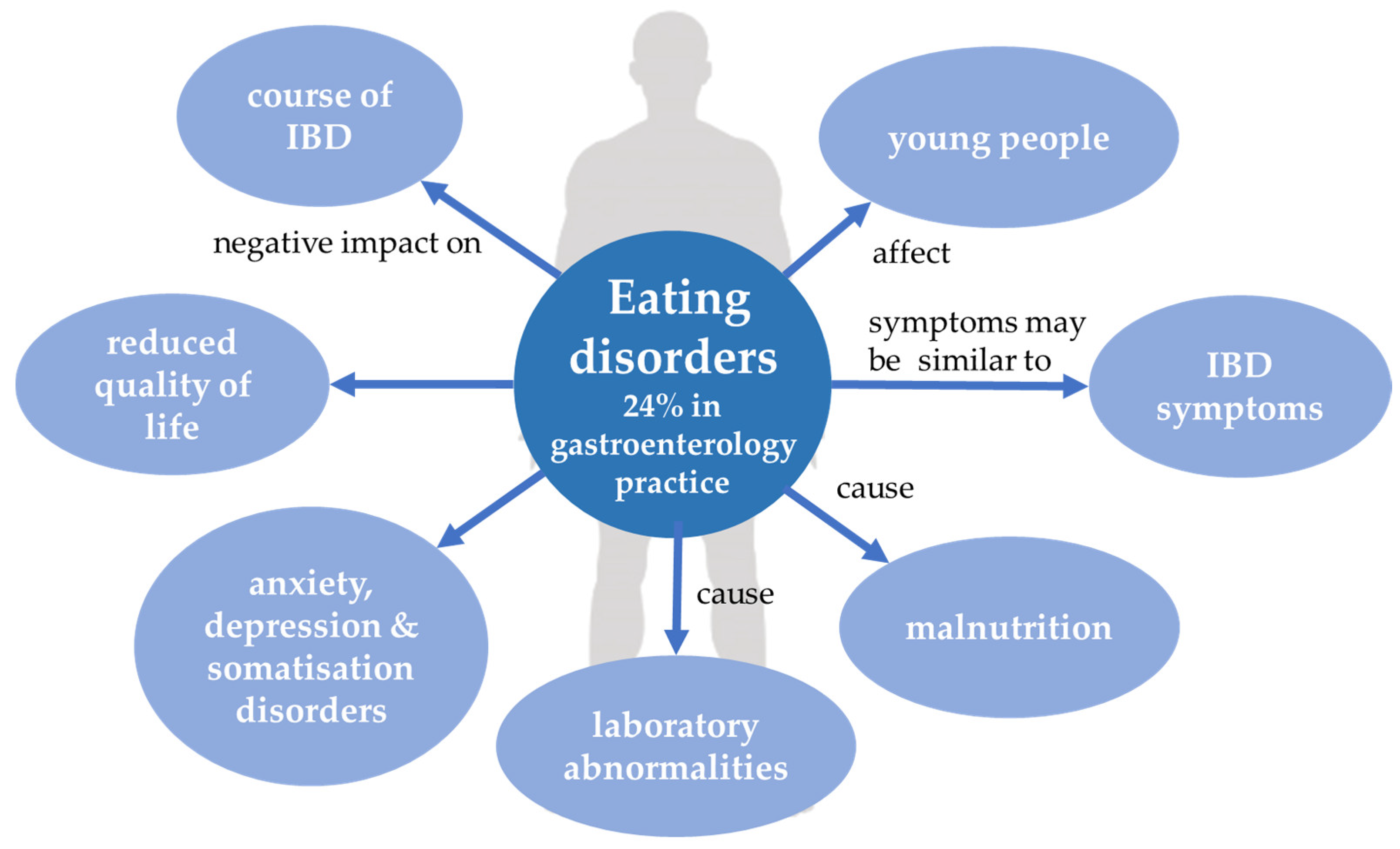
3. Eating Disorders
3.1. Introduction
3.2. EDs and IBD—What Do They Have in Common?
3.3. ED and IBD—Diagnostic Challenge
3.4. EDs in IBD Patients
3.5. IBD in EDs Patients
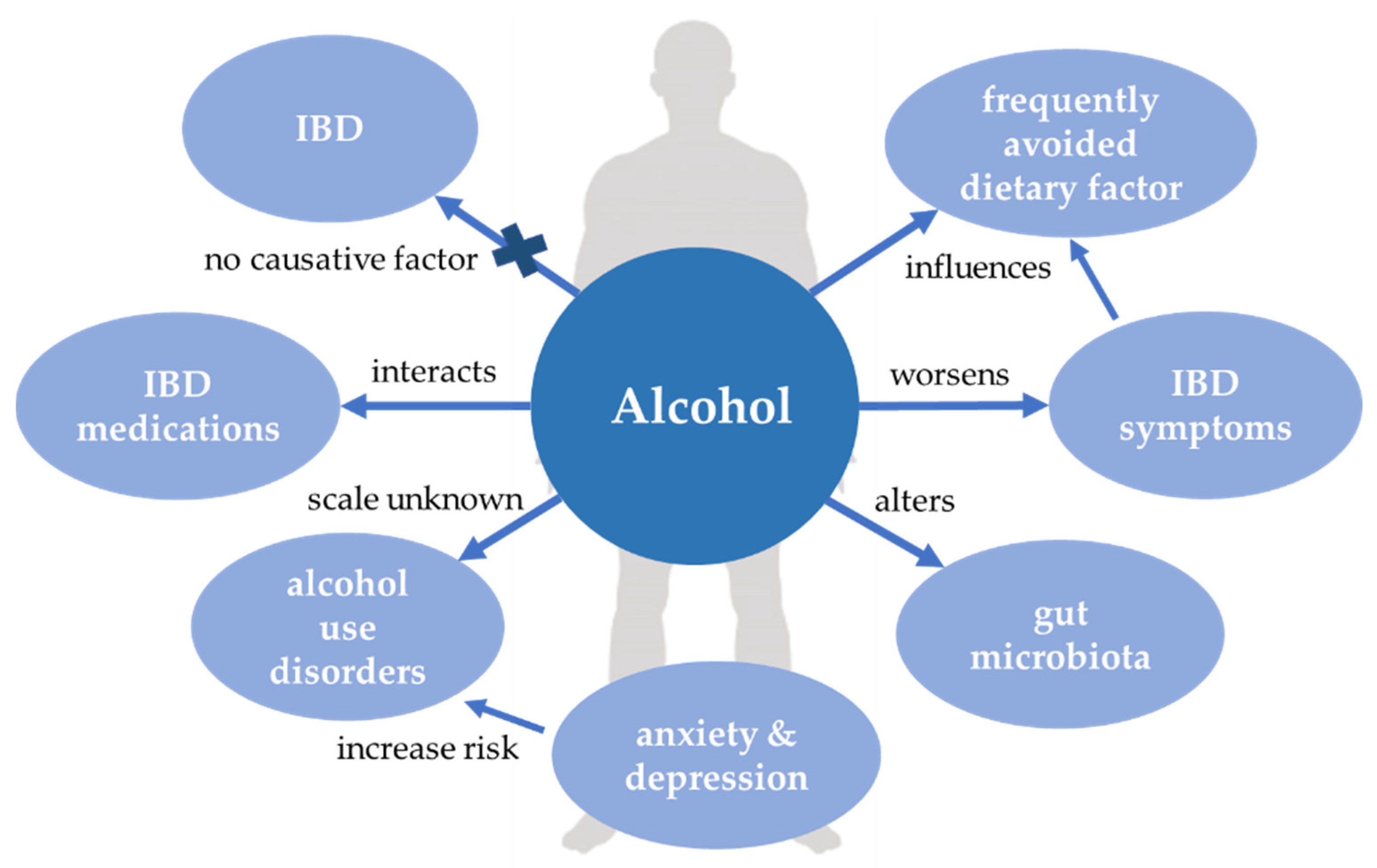
4. Alcohol
4.1. Alcohol and IBD
4.1.1. Alcohol and Pathogenesis of IBD
4.1.2. Alcohol and IBD Outcome
4.1.3. Alcohol and Complications of IBD
4.1.4. Alcohol Consumption in IBD Patients
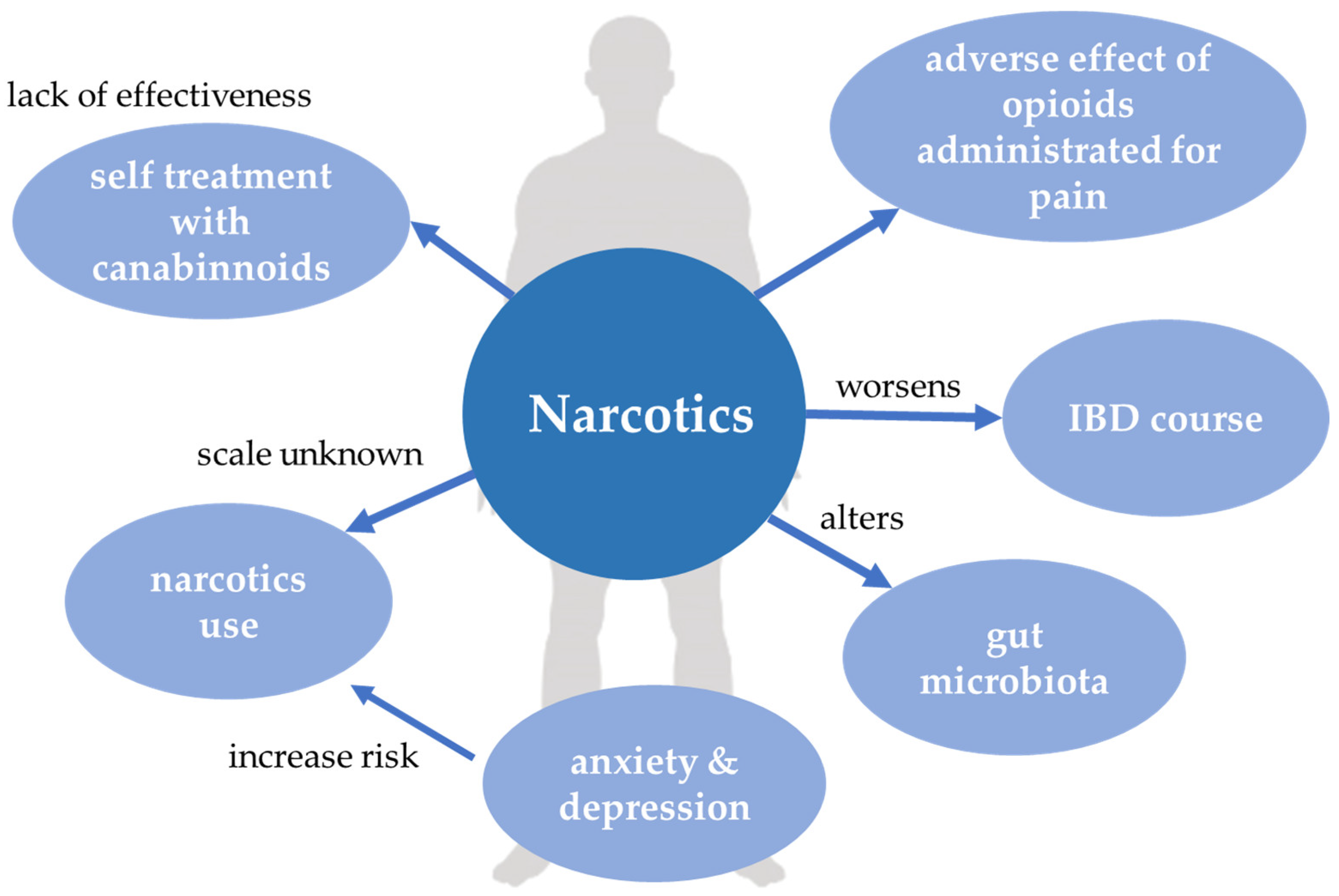
5. Narcotics
5.1. Narcotics Use in IBD Patients
5.2. Opioids
5.3. Cannabinoids
6. Psychiatric Disorders and SUDs
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Magro, F.; Gionchetti, P.; Eliakim, R.; Ardizzone, S.; Armuzzi, A.; Barreiro-de Acosta, M.; Burisch, J.; Gecse, K.B.; Hart, A.L.; Hindryckx, P.; et al. Third European evidence-based consensus on diagnosis and management of ulcerative colitis. Part 1: Definitions, diagnosis, extra-intestinal manifestations, pregnancy, cancer surveillance, surgery, and ileo-anal pouch disorders. J. Crohn’s Colitis 2017, 11, 649–670. [Google Scholar] [CrossRef]
- Gomollón, F.; Dignass, A.; Annese, V.; Tilg, H.; van Assche, G.; Lindsay, J.O.; Peyrin-Biroulet, L.; Cullen, G.J.; Daperno, M.; Kucharzik, T.; et al. 3rd European evidence-based consensus on the diagnosis and management of Crohn’s disease 2016: Part 1: Diagnosis and medical management. J. Crohn’s Colitis 2017, 11, 3–25. [Google Scholar] [CrossRef] [Green Version]
- De Souza, H.S.P. Etiopathogenesis of inflammatory bowel disease: Today and tomorrow. Curr. Opin. Gastroenterol. 2017, 33, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Jiménez Treviño, S.; Pujol Muncunill, G.; Martín-Masot, R.; Rodríguez Martínez, A.; Segarra Cantón, O.; Peña Quintana, L.; Armas Ramos, H.; Eizaguirre Arocena, F.J.; Barrio Torres, J.; García Burriel, J.I.; et al. Spanish Pediatric Inflammatory Bowel Disease Diagnostic Delay Registry: SPIDER Study From Sociedad Española de Gastroenterología, Hepatología y Nutrición Pediátrica. Front. Pediatr. 2020, 8, 584278. [Google Scholar] [CrossRef] [PubMed]
- Cantoro, L.; Di Sabatino, A.; Papi, C.; Margagnoni, G.; Ardizzone, S.; Giuffrida, P.; Giannarelli, D.; Massari, A.; Monterubbianesi, R.; Lenti, M.V.; et al. The time course of diagnostic delay in inflammatory bowel disease over the last sixty years: An Italian multicentre study. J. Crohn’s Colitis 2017, 11, 975–980. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barnes, E.L.; Loftus, E.V.; Kappelman, M.D. Effects of Race and Ethnicity on Diagnosis and Management of Inflammatory Bowel Diseases. Gastroenterology 2021, 160, 677–689. [Google Scholar] [CrossRef]
- Alatab, S.; Sepanlou, S.G.; Ikuta, K.; Vahedi, H.; Bisignano, C.; Safiri, S.; Sadeghi, A.; Nixon, M.R.; Abdoli, A.; Abolhassani, H.; et al. The global, regional, and national burden of inflammatory bowel disease in 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol. Hepatol. 2020, 5, 17–30. [Google Scholar] [CrossRef] [Green Version]
- Ling, F.; Tu, L.; Li, J.; Chen, Y.; Xu, M.; Zhu, L. Psychological aspect of patients with inflammatory bowel disease, which may be related to the quality of life, sleep and disease characteristics. Scand. J. Gastroenterol. 2021, 1–9. [Google Scholar] [CrossRef]
- Larussa, T.; Flauti, D.; Abenavoli, L.; Boccuto, L.; Suraci, E.; Marasco, R.; Imeneo, M.; Luzza, F. The Reality of Patient-Reported Outcomes of Health-Related Quality of Life in an Italian Cohort of Patients with Inflammatory Bowel Disease: Results from a Cross-Sectional Study. J. Clin. Med. 2020, 9, 2416. [Google Scholar] [CrossRef]
- Jones, J.L.; Nguyen, G.C.; Benchimol, E.I.; Bernstein, C.N.; Bitton, A.; Kaplan, G.G.; Murthy, S.K.; Lee, K.; Cooke-Lauder, J.; Otley, A.R. The Impact of Inflammatory Bowel Disease in Canada 2018: Quality of Life. J. Can. Assoc. Gastroenterol. 2019, 2. [Google Scholar] [CrossRef]
- Paulides, E.; Boukema, I.; van der Woude, C.J.; de Boer, N.K.H. The Effect of Psychotherapy on Quality of Life in IBD Patients: A Systematic Review. Inflamm. Bowel Dis. 2021, 27, 711–724. [Google Scholar] [CrossRef]
- Leso, V.; Gervetti, P.; Macrini, M.C.; Russo, F.; Iavicoli, I. Inflammatory bowel diseases and work disability: A systematic review of predictive factors. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 165–181. [Google Scholar] [CrossRef] [PubMed]
- Kuenzig, M.E.; Lee, L.; El-Matary, W.; Weizman, A.V.; Benchimol, E.I.; Kaplan, G.G.; Nguyen, G.C.; Bernstein, C.N.; Bitton, A.; Lee, K.; et al. The Impact of Inflammatory Bowel Disease in Canada 2018: Indirect Costs of IBD Care. J. Can. Assoc. Gastroenterol. 2019, 2, S34–S41. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van Linschoten, R.C.A.; Visser, E.; Niehot, C.D.; van der Woude, C.J.; Hazelzet, J.A.; van Noord, D.; West, R.L. Systematic review: Societal cost of illness of inflammatory bowel disease is increasing due to biologics and varies between continents. Aliment. Pharmacol. Ther. 2021, 54, 234–248. [Google Scholar] [CrossRef] [PubMed]
- Kuenzig, M.E.; Benchimol, E.I.; Lee, L.; Targownik, L.E.; Singh, H.; Kaplan, G.G.; Bernstein, C.N.; Bitton, A.; Nguyen, G.C.; Lee, K.; et al. The Impact of Inflammatory Bowel Disease in Canada 2018: Direct Costs and Health Services Utilization. J. Can. Assoc. Gastroenterol. 2019, 2, S17–S33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rogler, G.; Zeitz, J.; Biedermann, L. The Search for Causative Environmental Factors in Inflammatory Bowel Disease. Dig. Dis. 2016, 34, 48–55. [Google Scholar] [CrossRef] [Green Version]
- Kellermayer, R.; Zilbauer, M. The Gut Microbiome and the Triple Environmental Hit Concept of Inflammatory Bowel Disease Pathogenesis. J. Pediatr. Gastroenterol. Nutr. 2020, 71, 589–595. [Google Scholar] [CrossRef]
- Greuter, T.; Rieder, F.; Kucharzik, T.; Peyrin-Biroulet, L.; Schoepfer, A.M.; Rubin, D.T.; Vavricka, S.R. Emerging treatment options for extraintestinal manifestations in IBD. Gut 2021, 70, 796–802. [Google Scholar] [CrossRef]
- Argollo, M.; Gilardi, D.; Peyrin-Biroulet, C.; Chabot, J.F.; Peyrin-Biroulet, L.; Danese, S. Comorbidities in inflammatory bowel disease: A call for action. Lancet Gastroenterol. Hepatol. 2019, 4, 643–654. [Google Scholar] [CrossRef]
- Hruz, P.; Juillerat, P.; Kullak-Ublick, G.A.; Schoepfer, A.M.; Mantzaris, G.J.; Rogler, G. Management of the Elderly Inflammatory Bowel Disease Patient. Digestion 2020, 101, 105–119. [Google Scholar] [CrossRef] [PubMed]
- Kuźnicki, P.; Kempiński, R.; Neubauer, K. The emerging role of mood disorders in inflammatory bowel diseases. Adv. Clin. Exp. Med. 2021, 29, 1505–1510. [Google Scholar] [CrossRef]
- Lai, H.M.X.; Cleary, M.; Sitharthan, T.; Hunt, G.E. Prevalence of comorbid substance use, anxiety and mood disorders in epidemiological surveys, 1990-2014: A systematic review and meta-analysis. Drug Alcohol Depend. 2015, 154, 1–13. [Google Scholar] [CrossRef]
- Wark, G.; Samocha-Bonet, D.; Ghaly, S.; Danta, M. The role of diet in the pathogenesis and management of inflammatory bowel disease: A review. Nutrients 2021, 13, 135. [Google Scholar] [CrossRef]
- De Souza, H.S.P.; Fiocchi, C.; Iliopoulos, D. The IBD interactome: An integrated view of aetiology, pathogenesis and therapy. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 739–749. [Google Scholar] [CrossRef]
- Lee, M.; Chang, E.B. Inflammatory Bowel Diseases (IBD) and the Microbiome—Searching the Crime Scene for Clues. Gastroenterology 2021, 160, 524–537. [Google Scholar] [CrossRef]
- Treasure, J.; Duarte, T.A.; Schmidt, U. Eating disorders. Lancet 2020, 395, 899–911. [Google Scholar] [CrossRef]
- Sarmiento, C.; Lau, C. Diagnostic and Statistical Manual of Mental Disorders. In The Wiley Encyclopedia of Personality and Individual Differences, 5th ed.; DSM-5; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 2020. [Google Scholar] [CrossRef]
- Li, S.; Ney, M.; Eslamparast, T.; Vandermeer, B.; Ismond, K.P.; Kroeker, K.; Halloran, B.; Raman, M.; Tandon, P. Systematic review of nutrition screening and assessment in inflammatory bowel disease. World J. Gastroenterol. 2019, 25, 3823–3837. [Google Scholar] [CrossRef] [PubMed]
- Marcil, V.; Levy, E.; Amre, D.; Bitton, A.; Sant’Anna, A.M.G.D.A.; Szilagy, A.; Sinnett, D.; Seidman, E.G. Erratum: A Cross-Sectional Study on Malnutrition in Inflammatory Bowel Disease: Is There a Difference Based on Pediatric or Adult Age Grouping? Inflamm. Bowel Dis. 2020, 37, 884–889. [Google Scholar] [CrossRef] [PubMed]
- Bergeron, F.; Bouin, M.; D’Aoust, L.; Lemoyne, M.; Presse, N. Food avoidance in patients with inflammatory bowel disease: What, when and who? Clin. Nutr. 2018, 37, 884–889. [Google Scholar] [CrossRef]
- Ng, Q.X.; Soh, A.Y.S.; Loke, W.; Lim, D.Y.; Yeo, W.S. The role of inflammation in irritable bowel syndrome (IBS). J. Inflamm. Res. 2018, 11, 345–349. [Google Scholar] [CrossRef] [Green Version]
- Butler, M.J.; Perrini, A.A.; Eckel, L.A. The role of the gut microbiome, immunity, and neuroinflammation in the pathophysiology of eating disorders. Nutrients 2021, 13, 500. [Google Scholar] [CrossRef]
- Mack, I.; Cuntz, U.; Grmer, C.; Niedermaier, S.; Pohl, C.; Schwiertz, A.; Zimmermann, K.; Zipfel, S.; Enck, P.; Penders, J. Weight gain in anorexia nervosa does not ameliorate the faecal microbiota, branched chain fatty acid profiles, and gastroin-testinal complaints. Sci. Rep. 2016, 6, 1–16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moran, G.W.; Thapaliya, G. The gut–brain axis and its role in controlling eating behavior in intestinal inflammation. Nutrients 2021, 13, 981. [Google Scholar] [CrossRef] [PubMed]
- Cao, Q.; Huang, Y.H.; Jiang, M.; Dai, C. The prevalence and risk factors of psychological disorders, malnutrition and quality of life in IBD patients. Scand. J. Gastroenterol. 2019, 54, 1458–1466. [Google Scholar] [CrossRef] [PubMed]
- Swanson, S.A.; Crow, S.J.; Le Grange, D.; Swendsen, J.; Merikangas, K.R. Prevalence and correlates of eating disorders in adolescents: Results from the national comorbidity survey replication adolescent supplement. Arch. Gen. Psychiatry 2011, 68, 714–723. [Google Scholar] [CrossRef] [Green Version]
- Werlang, M.E.; Sim, L.A.; Lebow, J.R.; Lacy, B.E. Assessing for Eating Disorders: A Primer for Gastroenterologists. Am. J. Gastroenterol. 2021, 116, 68–76. [Google Scholar] [CrossRef]
- Avila, J.T.; Park, K.T.; Golden, N.H. Eating disorders in adolescents with chronic gastrointestinal and endocrine diseases. Lancet Child Adolesc. Health 2019, 3, 181–189. [Google Scholar] [CrossRef]
- Baylé, F.J.; Bouvard, M.P. Anorexia nervosa and Crohn’s disease dual diagnosis: A case study. Eur. Psychiatry 2003, 18, 421–422. [Google Scholar] [CrossRef]
- Ilzarbe, L.; Fàbrega, M.; Quintero, R.; Bastidas, A.; Pintor, L.; García-Campayo, J.; Gomollón, F.; Ilzarbe, D. Inflammatory Bowel Disease and Eating Disorders: A systematized review of comorbidity. J. Psychosom. Res. 2017, 102, 47–53. [Google Scholar] [CrossRef] [Green Version]
- Butwicka, A.; Olén, O.; Larsson, H.; Halfvarson, J.; Almqvist, C.; Lichtenstein, P.; Serlachius, E.; Frisén, L.; Ludvigsson, J.F. Association of Childhood-Onset Inflammatory Bowel Disease with Risk of Psychiatric Disorders and Suicide Attempt. JAMA Pediatr. 2019, 173, 969–978. [Google Scholar] [CrossRef]
- Robelin, K.; Senada, P.A.; Ghoz, H.; Sim, L.A.; Lebow, J.R.; Farraye, F.A.; Werlang, M.E. Sa1812 eating disorders in an out-patient academic inflammatory bowel disease practice: Prevalence and provider diagnostic accuracy. Gastroenterology 2020, 158, S-435. [Google Scholar] [CrossRef]
- Wabich, J.; Bellaguarda, E.; Joyce, C.; Keefer, L.; Kinsinger, S. Disordered Eating, Body Dissatisfaction, and Psychological Distress in Patients with Inflammatory Bowel Disease (IBD). J. Clin. Psychol. Med. Settings 2020, 27. [Google Scholar] [CrossRef]
- Marafini, I.; Longo, L.; Lavasani, D.M.; Rossi, R.; Salvatori, S.; Pianigiani, F.; Calabrese, E.; Siracusano, A.; Di Lorenzo, G.; Monteleone, G. High Frequency of Undiagnosed Psychiatric Disorders in Inflammatory Bowel Diseases. J. Clin. Med. 2020, 9, 1387. [Google Scholar] [CrossRef]
- Jonathan, S.; Ghassan, W. P055 Restrictive Eating Symptoms May Persist in Children & Adolescents With Treated IBD: Case Series. Am. J. Gastroenterol. 2020, 115, S14–S15. [Google Scholar] [CrossRef]
- Erdur, L.; Kallenbach-Dermutz, B.; Lehmann, V.; Zimmermann-Viehoff, F.; Köpp, W.; Weber, C.; Deter, H.C. Somatic comorbidity in anorexia nervosa: First results of a 21-year follow-up study on female inpatients. Biopsychosoc. Med. 2012, 6, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Raevuori, A.; Haukka, J.; Vaarala, O.; Suvisaari, J.M.; Gissler, M.; Grainger, M.; Linna, M.S.; Suokas, J.T. The increased risk for autoimmune diseases in patients with eating disorders. PLoS ONE 2014, 9, e104845. [Google Scholar] [CrossRef]
- Wotton, C.J.; James, A.; Goldacre, M.J. Coexistence of eating disorders and autoimmune diseases: Record linkage cohort study, UK. Int. J. Eat. Disord. 2016, 49, 663–672. [Google Scholar] [CrossRef] [PubMed]
- WHO. Global Status Report on Alcohol and Health 2018; World Health Organization: Geneva, Switzerland, 2018. [Google Scholar]
- Richter, L.; Pugh, B.S.; Peters, E.A.; Vaughan, R.D.; Foster, S.E. Underage drinking: Prevalence and correlates of risky drinking measures among youth aged 12–20. Am. J. Drug Alcohol Abuse 2016, 42, 385–394. [Google Scholar] [CrossRef] [PubMed]
- IARC. Personal Habits and Indoor Combustions. In IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; IARC: Lyon, France, 2012. [Google Scholar]
- LoConte, N.K.; Brewster, A.M.; Kaur, J.S.; Merrill, J.K.; Alberg, A.J. Alcohol and cancer: A statement of the American society of clinical oncology. J. Clin. Oncol. 2018, 36, 83–93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rao, R. Endotoxemia and gut barrier dysfunction in alcoholic liver disease. Hepatology 2009, 50, 638–644. [Google Scholar] [CrossRef] [PubMed]
- Bishehsari, F.; Magno, E.; Swanson, G.; Desai, V.; Voigt, R.M.; Forsyth, C.B.; Keshavarzian, A. Alcohol and gut-derived inflammation. Alcohol Res. Curr. Rev. 2017, 38, 163–171. [Google Scholar]
- Molina, P.E.; Happel, K.I.; Zhang, P.; Kolls, J.K.; Nelson, S. Focus on: Alcohol and the immune system. Alcohol Res. Health 2010, 33, 97–108. [Google Scholar]
- Cannon, A.R.; Kuprys, P.V.; Cobb, A.N.; Ding, X.; Kothari, A.N.; Kuo, P.C.; Eberhardt, J.M.; Hammer, A.M.; Morris, N.L.; Li, X.; et al. Alcohol enhances symptoms and propensity for infection in inflammatory bowel disease patients and a murine model of DSS-induced colitis. J. Leukoc. Biol. 2018, 104, 543–555. [Google Scholar] [CrossRef] [PubMed]
- Khoruts, A.; Stahnke, L.; McClain, C.J.; Logan, G.; Allen, J.I. Circulating tumor necrosis factor, interleukin-1 and interleukin-6 concentrations in chronic alcoholic patients. Hepatology 1991, 13, 267–276. [Google Scholar] [CrossRef] [PubMed]
- Mandrekar, P.; Bala, S.; Catalano, D.; Kodys, K.; Szabo, G. The Opposite Effects of Acute and Chronic Alcohol on Lipopolysaccharide-Induced Inflammation Are Linked to IRAK-M in Human Monocytes. J. Immunol. 2009, 183, 1320–1327. [Google Scholar] [CrossRef] [PubMed]
- Swanson, G.R.; Tieu, V.; Shaikh, M.; Forsyth, C.; Keshavarzian, A. Is moderate red wine consumption safe in inactive inflammatory bowel disease? Digestion 2011, 84, 238–244. [Google Scholar] [CrossRef] [Green Version]
- Jiang, L.; Xia, B.; Li, J.; Ye, M.; Deng, C.; Ding, Y.; Luo, H.; Ren, H.; Hou, X.; Liu, H.; et al. Risk factors for ulcerative colitis in a Chinese population: An age-matched and sex-matched case-control study. J. Clin. Gastroenterol. 2007, 41, 280–284. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.C.; Rampertab, S.D.; Mullin, G.E. Existing dietary guidelines for Crohns disease and ulcerative colitis. Expert Rev. Gastroenterol. Hepatol. 2011, 5, 411–425. [Google Scholar] [CrossRef]
- Hsu, T.Y.; Shih, H.M.; Wang, Y.C.; Lin, L.C.; He, G.Y.; Chen, C.Y.; Kao, C.H.; Chen, C.H.; Chen, W.K.; Yang, T.Y. Effect of alcoholic intoxication on the risk of inflammatory bowel disease: A nationwide retrospective cohort study. PLoS ONE 2016, 11, e0165411. [Google Scholar] [CrossRef] [Green Version]
- Bergmann, M.M.; Hernandez, V.; Bernigau, W.; Boeing, H.; Chan, S.S.M.; Luben, R.; Khaw, K.T.; van Schaik, F.; Oldenburg, B.; Bueno-De-Mesquita, B.; et al. No association of alcohol use and the risk of ulcerative colitis or Crohn’s disease: Data from a European Prospective cohort study (EPIC). Eur. J. Clin. Nutr. 2017, 71, 512–518. [Google Scholar] [CrossRef] [Green Version]
- Nie, J.Y.; Zhao, Q. Beverage consumption and risk of ulcerative colitis: Systematic review and meta-analysis of epidemiological studies. Medicine 2017, 96, e9070. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Xiang, L.; He, J. Beverage intake and risk of Crohn disease: A meta-analysis of 16 epidemiological studies. Medicine 2019, 98, e15795. [Google Scholar] [CrossRef]
- Khasawneh, M.; Spence, A.D.; Addley, J.; Allen, P.B. The role of smoking and alcohol behaviour in the management of inflammatory bowel disease. Best Pract. Res. Clin. Gastroenterol. 2017, 31, 553–559. [Google Scholar] [CrossRef] [PubMed]
- Han, D.Y.; Fraser, A.G.; Dryland, P.; Ferguson, L.R. Environmental factors in the development of chronic inflammation: A case-control study on risk factors for Crohn’s disease within New Zealand. Mutat. Res. Fundam. Mol. Mech. Mutagen. 2010, 690, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Sonnenberg, G.F.; Artis, D. Innate lymphoid cells in the initiation, regulation and resolution of inflammation. Nat. Med. 2015, 21, 698–708. [Google Scholar] [CrossRef] [PubMed]
- Xavier, R.J.; Podolsky, D.K. Unravelling the pathogenesis of inflammatory bowel disease. Nature 2007, 448, 427–434. [Google Scholar] [CrossRef]
- Magee, E.A.; Edmond, L.M.; Tasker, S.M.; Kong, S.C.; Curno, R.; Cummings, J.H. Associations between diet and disease activity in ulcerative colitis patients using a novel method of data analysis. Nutr. J. 2005, 4, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Christl, S.U.; Eisner, H.D.; Dusel, G.; Kasper, H.; Scheppach, W. Antagonistic Effects of Sulfide and Butyrate on Proliferation of Colonic Mucosa: A Potential Role for These Agents in the Pathogenesis of Ulcerative Colitis. Dig. Dis. Sci. 1996, 41, 2477–2481. [Google Scholar] [CrossRef]
- Hey, H.; Schmedes, A.; Nielsen, A.A.; Winding, P.; Grønbæk, H. Effects of five different alcoholic drinks on patients with Crohn’s disease. Scand. J. Gastroenterol. 2007, 42, 968–972. [Google Scholar] [CrossRef]
- Mantzouranis, G.; Fafliora, E.; Saridi, M.; Tatsioni, A.; Glanztounis, G.; Albani, E.; Katsanos, K.H.; Christodoulou, D.K. Alcohol and narcotics use in inflammatory bowel disease. Ann. Gastroenterol. 2018, 31, 649. [Google Scholar] [CrossRef]
- Zutshi, M.; Hull, T.L.; Hammel, J. Crohn’s disease: A patient’s perspective. Int. J. Colorectal Dis. 2007, 22, 1437–1444. [Google Scholar] [CrossRef]
- Guida, L.; Di Giorgio, F.M.; Busacca, A.; Carrozza, L.; Ciminnisi, S.; Almasio, P.L.; Di Marco, V.; Cappello, M. Perception of the role of food and dietary modifications in patients with inflammatory bowel disease: Impact on lifestyle. Nutrients 2021, 13, 759. [Google Scholar] [CrossRef]
- Swanson, G.R.; Sedghi, S.; Farhadi, A.; Keshavarzian, A. Pattern of alcohol consumption and its effect on gastrointestinal symptoms in inflammatory bowel disease. Alcohol 2010, 44, 223–228. [Google Scholar] [CrossRef] [Green Version]
- Triggs, C.M.; Munday, K.; Hu, R.; Fraser, A.G.; Gearry, R.B.; Barclay, M.L.; Ferguson, L.R. Dietary factors in chronic inflammation: Food tolerances and intolerances of a New Zealand Caucasian Crohn’s disease population. Mutat. Res. Fundam. Mol. Mech. Mutagen. 2010, 690, 123–138. [Google Scholar] [CrossRef] [PubMed]
- Zallot, C.; Quilliot, D.; Chevaux, J.B.; Peyrin-Biroulet, C.; Guéant-Rodriguez, R.M.; Freling, E.; Collet-Fenetrier, B.; Williet, N.; Ziegler, O.; Bigard, M.A.; et al. Dietary beliefs and behavior among inflammatory bowel disease patients. Inflamm. Bowel Dis. 2013, 19. [Google Scholar] [CrossRef] [Green Version]
- Cohen, A.B.; Lee, D.; Long, M.D.; Kappelman, M.D.; Martin, C.F.; Sandler, R.S.; Lewis, J.D. Dietary patterns and self-reported associations of diet with symptoms of inflammatory bowel disease. Dig. Dis. Sci. 2013, 58, 1322–1328. [Google Scholar] [CrossRef] [PubMed]
- Limdi, J.K.; Aggarwal, D.; McLaughlin, J.T. Dietary Practices and Beliefs in Patients with Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2016, 22, 164–170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Crooks, B.; Misra, R.; Arebi, N.; Kok, K.; Brookes, M.J.; McLaughlin, J.; Limdi, J.K. The dietary practices and beliefs of people living with older-onset inflammatory bowel disease. Eur. J. Gastroenterol. Hepatol. 2021. [Google Scholar] [CrossRef]
- Crooks, B.; Misra, R.; Arebi, N.; Kok, K.; Brookes, M.J.; McLaughlin, J.; Limdi, J.K. The dietary practices and beliefs of British South Asian people living with inflammatory bowel disease: A multicenter study from the United Kingdom. Intest. Res. 2021. [Google Scholar] [CrossRef]
- Crooks, B.; McLaughlin, J.; Matsuoka, K.; Kobayashi, T.; Yamazaki, H.; Limdi, J.K. The dietary practices and beliefs of people living with inactive ulcerative colitis. Eur. J. Gastroenterol. Hepatol. 2021, 33, 372–379. [Google Scholar] [CrossRef]
- Vagianos, K.; Clara, I.; Carr, R.; Graff, L.A.; Walker, J.R.; Targownik, L.E.; Lix, L.M.; Rogala, L.; Miller, N.; Bernstein, C.N. What are adults with inflammatory bowel disease (IBD) eating? A closer look at the dietary habits of a population-based Canadian IBD cohort. J. Parenter. Enter. Nutr. 2016, 40, 405–411. [Google Scholar] [CrossRef]
- Chiba, T.; Phillips, S.F. Alcohol-related diarrhea. Addict. Biol. 2000, 5, 117–125. [Google Scholar] [CrossRef]
- Djordjevic, D.; Nikolic, J.; Stefanovic, V. Ethanol interactions with other cytochrome P450 substrates including drugs, xenobiotics, and carcinogens. Pathol. Biol. 1998, 46, 760–770. [Google Scholar] [PubMed]
- Ramos, G.P.; Kane, S. Alcohol use in patients with inflammatory bowel disease. Gastroenterol. Hepatol. 2021, 17, 221. [Google Scholar]
- Hernández Ceruelos, A.; Romero-Quezada, L.C.; Ruvalcaba Ledezma, J.C.; López Contreras, L. Therapeutic uses of metronidazole and its side effects: An update. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 397–401. [Google Scholar] [CrossRef] [PubMed]
- Fadda, H.M.; Mohamed, M.A.M.; Basit, A.W. Impairment of the in vitro drug release behaviour of oral modified release preparations in the presence of alcohol. Int. J. Pharm. 2008, 360, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Vena, G.A.; Cassano, N. The effects of alcohol on the metabolism and toxicology of anti-psoriasis drugs. Expert Opin. Drug Metab. Toxicol. 2012, 8, 959–972. [Google Scholar] [CrossRef] [PubMed]
- Elsing, C.; Placke, J.; Herrmann, T. Alcohol binging causes peliosis hepatis during azathioprine therapy in crohn’s disease. World J. Gastroenterol. 2007, 13, 4646. [Google Scholar] [CrossRef] [PubMed]
- Humphreys, J.H.; Warner, A.; Costello, R.; Lunt, M.; Verstappen, S.M.M.; Dixon, W.G. Quantifying the hepatotoxic risk of alcohol consumption in patients with rheumatoid arthritis taking methotrexate. Ann. Rheum. Dis. 2017, 76, 1509–1514. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Whiting-O’Keefe, Q.E.; Fye, K.H.; Sack, K.D. Methotrexate and histologic hepatic abnormalities: A meta-analysis. Am. J. Med. 1991, 90, 711–716. [Google Scholar] [CrossRef]
- Kalsi, S.S.; Wood, D.M.; Waring, W.S.; Dargan, P.I. Does cytochrome P450 liver isoenzyme induction increase the risk of liver toxicity after paracetamol overdose? Open Access Emerg. Med. 2011, 3, 69–76. [Google Scholar] [CrossRef] [Green Version]
- Thavamani, A.; Umapathi, K.K.; Khatana, J.; Gulati, R. Burden of psychiatric disorders among pediatric and young adults with inflammatory bowel disease: A population-based analysis. Pediatr. Gastroenterol. Hepatol. Nutr. 2019, 22, 527–535. [Google Scholar] [CrossRef]
- Plevinsky, J.M.; Maddux, M.H.; Greenley, R.N. Substance Use in Adolescents and Young Adults with Inflammatory Bowel Diseases: An Exploratory Cluster Analysis. J. Pediatr. Gastroenterol. Nutr. 2019, 69, 324–329. [Google Scholar] [CrossRef]
- Ludvigsson, J.F.; Olén, O.; Larsson, H.; Halfvarson, J.; Almqvist, C.; Lichtenstein, P.; Butwicka, A. Association Between Inflammatory Bowel Disease and Psychiatric Morbidity and Suicide: A Swedish Nationwide Population-Based Cohort Study With Sibling Comparisons. J. Crohn’s Colitis 2021. [Google Scholar] [CrossRef]
- Vigod, S.N.; Kurdyak, P.; Brown, H.K.; Nguyen, G.C.; Targownik, L.E.; Seow, C.H.; Kuenzig, M.E.; Benchimol, E.I. Inflammatory bowel disease and new-onset psychiatric disorders in pregnancy and post partum: A population-based cohort study. Gut 2019, 68, 1597–1605. [Google Scholar] [CrossRef]
- Carney, H.; Marrie, R.A.; Bolton, J.M.; Patten, S.B.; Graff, L.A.; Bernstein, C.N.; Kowalec, K. Prevalence and Risk Factors of Substance Use Disorder in Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2021, 27, 58–64. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brooks, A.; Rowse, G.; Peach, E.; Ryder, A.; Narula, P.; Corfe, B.; Norman, P.; Lobo, A. OC-087 Frequency of Health Risk Behaviours in Young People With Inflammatory Bowel Disease. Gut 2016. [Google Scholar] [CrossRef]
- Cross, R.K.; Wilson, K.T.; Binion, D.G. Narcotic use in patients with Crohn’s disease. Am. J. Gastroenterol. 2005. [Google Scholar] [CrossRef]
- Zeitz, J.; Ak, M.; Muller-Mottet, S.; Scharl, S.; Biedermann, L.; Fournier, N.; Frei, P.; Pittet, V.; Scharl, M.; Fried, M.; et al. Pain in IBD patients: Very frequent and frequently insufficiently taken into account. PLoS ONE 2016, 11, e0156666. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Noureldin, M.; Higgins, P.D.R.; Govani, S.M.; Cohen-Mekelburg, S.; Kenney, B.C.; Stidham, R.W.; Waljee, J.F.; Waljee, A.K. Incidence and predictors of new persistent opioid use following inflammatory bowel disease flares treated with oral corticosteroids. Aliment. Pharmacol. Ther. 2019, 49, 74–83. [Google Scholar] [CrossRef]
- Targownik, L.E.; Nugent, Z.; Singh, H.; Bugden, S.; Bernstein, C.N. The prevalence and predictors of opioid use in inflammatory bowel disease: A population-based analysis. Am. J. Gastroenterol. 2014, 109, 1613–1620. [Google Scholar] [CrossRef]
- Philippe, D.; Chakass, D.; Thuru, X.; Zerbib, P.; Tsicopoulos, A.; Geboes, K.; Bulois, P.; Breisse, M.; Vorng, H.; Gay, J.; et al. Mu opioid receptor expression is increased in inflammatory bowel diseases: Implications for homeostatic intestinal inflammation. Gut 2006, 55, 815–823. [Google Scholar] [CrossRef] [PubMed]
- Raknes, G.; Simonsen, P.; Småbrekke, L. The effect of low-dose naltrexone on medication in inflammatory bowel disease: A quasi experimental before-and-after prescription database study. J. Crohn’s Colitis 2018, 12, 677–686. [Google Scholar] [CrossRef] [Green Version]
- Lie, M.R.K.L.; van der Giessen, J.; Fuhler, G.M.; de Lima, A.; Peppelenbosch, M.P.; van der Ent, C.; van der Woude, C.J. Low dose Naltrexone for induction of remission in inflammatory bowel disease patients. J. Transl. Med. 2018, 16, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, G.C.; Boland, K.; Afif, W.; Bressler, B.; Jones, J.L.; Weizman, A.V.; Fowler, S.; Halder, S.; Huang, V.W.; Kaplan, G.G.; et al. Modified Delphi Process for the Development of Choosing Wisely for Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2017, 23, 858–865. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Volz, M.S.; Siegmund, B.; Häuser, W. Efficacy, tolerability, and safety of cannabinoids in gastroenterology: A systematic review. Schmerz 2016, 30, 37–46. [Google Scholar] [CrossRef]
- Kienzl, M.; Storr, M.; Schicho, R. Cannabinoids and Opioids in the Treatment of Inflammatory Bowel Diseases. Clin. Transl. Gastroenterol. 2020, 11. [Google Scholar] [CrossRef]
- Li, K.; Fichna, J.; Schicho, R.; Saur, D.; Bashashati, M.; MacKie, K.; Li, Y.; Zimmer, A.; Göke, B.; Sharkey, K.A.; et al. A role for O-1602 and G protein-coupled receptor GPR55 in the control of colonic motility in mice. Neuropharmacology 2013, 71, 255–263. [Google Scholar] [CrossRef] [Green Version]
- Everard, A.; Plovier, H.; Rastelli, M.; van Hul, M.; de Wouters d’Oplinter, A.; Geurts, L.; Druart, C.; Robine, S.; Delzenne, N.M.; Muccioli, G.G.; et al. Intestinal epithelial N-acylphosphatidylethanolamine phospholipase D links dietary fat to metabolic adaptations in obesity and steatosis. Nat. Commun. 2019, 10, 1–17. [Google Scholar] [CrossRef]
- Santiago, A.; Torres, E. 2907 Knowledge and Perception of Gastroenterologists Regarding Medical Marijuana Use in Patients With Inflammatory Bowel Disease. Am. J. Gastroenterol. 2019, 114, S1588. [Google Scholar] [CrossRef]
- Naftali, T.; Bar-Lev Schleider, L.; Dotan, I.; Lansky, E.P.; Sklerovsky Benjaminov, F.; Konikoff, F.M. Cannabis induces a clinical response in patients with crohn’s disease: A prospective placebo-controlled study. Clin. Gastroenterol. Hepatol. 2013, 11, 1276–1280. [Google Scholar] [CrossRef]
- Naftali, T.; Schleider, L.B.L.; Benjaminov, F.S.; Konikoff, F.M.; Matalon, S.T.; Ringel, Y. Cannabis is associated with clinical but not endoscopic remission in ulcerative colitis: A randomized controlled trial. PLoS ONE 2021, 16, e0246871. [Google Scholar] [CrossRef] [PubMed]
- García-Planella, E.; Marín, L.; Domènech Morral, E.; Bernal, I.; Mañosa, M.; Zabana, Y.; Gassull, M.A. Utilización de medicinas alternativas y consumo de drogas por pacientes con enfermedad inflamatoria intestinal. Med. Clin. 2007, 128, 45–48. [Google Scholar] [CrossRef] [PubMed]
- Lal, S.; Prasad, N.; Ryan, M.; Tangri, S.; Silverberg, M.S.; Gordon, A.; Steinhart, H. Cannabis use amongst patients with inflammatory bowel disease. Eur. J. Gastroenterol. Hepatol. 2011, 23, 891–896. [Google Scholar] [CrossRef]
- Allegretti, J.R.; Courtwright, A.; Lucci, M.; Korzenik, J.R.; Levine, J. Marijuana use patterns among patients with inflammatory bowel disease. Inflamm. Bowel Dis. 2013, 19, 2809–2814. [Google Scholar] [CrossRef] [Green Version]
- Storr, M.; Devlin, S.; Kaplan, G.G.; Panaccione, R.; Andrews, C.N. Cannabis use provides symptom relief in patients with inflammatory bowel disease but is associated with worse disease prognosis in patients with Crohn’s disease. Inflamm. Bowel Dis. 2014, 20, 472–480. [Google Scholar] [CrossRef] [PubMed]
- Weiss, A.; Friedenberg, F. Patterns of cannabis use in patients with Inflammatory Bowel Disease: A population based analysis. Drug Alcohol Depend. 2015, 156, 84–89. [Google Scholar] [CrossRef]
- Phatak, U.P.; Rojas-Velasquez, D.; Porto, A.; Pashankar, D.S. Prevalence and Patterns of Marijuana Use in Young Adults with Inflammatory Bowel Disease. J. Pediatr. Gastroenterol. Nutr. 2017, 64, 261–264. [Google Scholar] [CrossRef]
- Hoffenberg, E.J.; McWilliams, S.; Mikulich-Gilbertson, S.; Murphy, B.; Hoffenberg, A.; Hopfer, C.J. Cannabis oil use by adolescents and young adults with inflammatory bowel disease. J. Pediatr. Gastroenterol. Nutr. 2019, 68, 348–352. [Google Scholar] [CrossRef]
- Benson, M.J.; Abelev, S.V.; Connor, S.J.; Corte, C.J.; Martin, L.J.; Gold, L.K.; Suraev, A.S.; McGregor, I.S. Medicinal cannabis for inflammatory bowel disease: A survey of perspectives, experiences, and current use in australian patients. Crohn’s Colitis 2020, 360, otaa015. [Google Scholar] [CrossRef] [Green Version]
- Hansen, T.M.; Sabourin, B.C.; Oketola, B.; Bernstein, C.N.; Singh, H.; Targownik, L.E. Cannabis use in persons with inflammatory bowel disease and vulnerability to substance misuse. Inflamm. Bowel Dis. 2020, 26, 1401–1406. [Google Scholar] [CrossRef] [PubMed]
- Secades-Villa, R.; Garcia-Rodríguez, O.; Jin, C.J.; Wang, S.; Blanco, C. Probability and predictors of the cannabis gateway effect: A national study. Int. J. Drug Policy 2015, 26, 135–142. [Google Scholar] [CrossRef] [Green Version]
- Sharkey, K.A.; Wiley, J.W. The Role of the Endocannabinoid System in the Brain–Gut Axis. Gastroenterology 2016, 151, 252–266. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jacobs, J.D.; Boden, E.K.; Lord, J.D.; Chiorean, M.V. Tu1906 prevalence and association of cannabis use with disease activity in patients with inflammatory bowel disease. Gastroenterology 2020, 158. [Google Scholar] [CrossRef]
- Naftali, T.; Mechulam, R.; Marii, A.; Gabay, G.; Stein, A.; Bronshtain, M.; Laish, I.; Benjaminov, F.; Konikoff, F.M. Low-Dose Cannabidiol Is Safe but Not Effective in the Treatment for Crohn’s Disease, a Randomized Controlled Trial. Dig. Dis. Sci. 2017, 62, 1615–1620. [Google Scholar] [CrossRef]
- Kaplan, M.A.; Korelitz, B.I. Narcotic dependence in inflammatory bowel disease. J. Clin. Gastroenterol. 1988, 10, 275–278. [Google Scholar] [CrossRef]
- Edwards, J.T.; Radford-Smith, G.L.; Florin, T.H.J. Chronic narcotic use in inflammatory bowel disease patients: Prevalence and clinical characteristics. J. Gastroenterol. Hepatol. 2001, 16, 1235–1238. [Google Scholar] [CrossRef]
- Buckley, J.P.; Cook, S.F.; Allen, J.K.; Kappelman, M.D. Prevalence of chronic narcotic use among children with inflammatory bowel disease. Clin. Gastroenterol. Hepatol. 2015, 13, 310–315. [Google Scholar] [CrossRef]
- Freeman, K.; Ryan, R.; Parsons, N.; Taylor-Phillips, S.; Willis, B.H.; Clarke, A. The incidence and prevalence of inflammatory bowel disease in UK primary care: A retrospective cohort study of the IQVIA Medical Research Database. BMC Gastroenterol. 2021, 21, 1–7. [Google Scholar] [CrossRef]
- Jussila, A.; Virta, L.J.; Salomaa, V.; Mäki, J.; Jula, A.; Färkkilä, M.A. High and increasing prevalence of inflammatory bowel disease in Finland with a clear North-South difference. J. Crohn’s Colitis 2013, 7, e256–e262. [Google Scholar] [CrossRef] [Green Version]
- De Groof, E.J.; Rossen, N.G.M.; van Rhijn, B.D.; Karregat, E.P.M.; Boonstra, K.; Hageman, I.; Bennebroek Evertsz, F.; Kingma, P.J.; Naber, A.H.J.; van den Brande, J.H.M.; et al. Burden of disease and increasing prevalence of inflammatory bowel disease in a population-based cohort in the Netherlands. Eur. J. Gastroenterol. Hepatol. 2016, 28, 1065–1072. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galmiche, M.; Déchelotte, P.; Lambert, G.; Tavolacci, M.P. Prevalence of eating disorders over the 2000-2018 period: A systematic literature review. Am. J. Clin. Nutr. 2019, 109, 1402–1413. [Google Scholar] [CrossRef] [PubMed]
| Study | Author | IBD Patients | EDs Patients n (%) | Tool | Remarks |
|---|---|---|---|---|---|
| Prospective cohort study [44] | Marafini et al. 2020 | 101 UC and 136 CD | 5 (4.9) BN 4 (2.9) BN/136 CD | MINI 5.0.0 * | single university hospital; Italy |
| Retrospective cohort study [41] | Butwicka et al. 2020 | 6464 | 83 (1.3) | ICD codes | early onset IBD; compared to general population and siblings without IBD; Sweden |
| Cross-sectional study [43] | Wabich et al. 2020 | 83 | 13 (15.7) patients scoring above threshold when clinical evaluation is recommended | survey with EAT-26 | abstract |
| Cross-sectional study [42] | Robelin et al. 2020 | 95 | 23 (24.2) | survey with EDDS | academic outpatient IBD practice; abstract; United States |
| Study | Author | IBD | EDs | Patients with EDs and IBD (%) | Remarks |
|---|---|---|---|---|---|
| Retrospective cohort study [46] | Erdur et al. 2012 | 3 | 169 | 1.8 | limited to females with AN; 21 years follow-up; Germany |
| Retrospective cohort study [47] | Raevuori et al. 2014 | 27 CD/2315 24 UC/2318 | 2342 | UC: 1 CD: 1.1 | patients from special care centre; compared to the general population; Finland |
| Retrospective cohort study [48] | Wotton et al. 2016 | 42 CD and 36 UC in AN females as the first admission 41 CD and 26 UC in AID as the first admission 15 CD and 7 UC in BN as first BN admission 17 CD and 15 UC in AID as first admission | 8700 females and 651 males with AN 4783 females and 330 males with BN | AN: 0.83 BN: 0.43 | a study using national administrative statistical data on hospital care and mortality, 1999–2011; limited to hospitalized patients; United Kingdom |
| Study | Author | IBD Patients | Method | Major Findings |
|---|---|---|---|---|
| Cross-sectional study | Jowett et al. 2004 | 183 inactive UC | Food frequency questionnaire Simple clinical colitis activity index | High intake of alcohol as well as high intake of sulfur and sulfate were associated with increased risk of UC relapse. |
| Cross-sectional study | Swanson et al. 2010 [76] | 52 inactive CD 38 inactive UC 39 IBS | Validated questionnaire of National Institute of Alcohol Abuse and Alcoholism Novel questionnaire evaluating the effect of alcohol on symptoms | Worsening of symptoms with alcohol in current drinkers No correlation between the type of alcoholic beverage and symptoms. |
| Cross-sectional study | Triggs et al. 2010 [77] | 446 CD | An extensive dietary questionnaire (257 food items in 15 groups) recorded self-reported dietary tolerances and intolerances | Beer and wine had adverse effects in more than 45% of CD patients. |
| Cross-sectional study | Zallot C. et al. 2012 [78] | 244 IBD | Questionnaire of 14 items | Only 6 patients reported alcohol as avoided factor to prevent relapse. |
| Cross-sectional study | Cohen et al. 2013 [79] | 1121 CD 405 CD ostomy patients 597 UC 206 UC pouch patients | Semi-quantitative food frequency questionnaire to measure dietary consumption patterns and open-ended questions to elicit responses from patients about food items they believe ameliorate or exacerbate IBD | Alcohol made symptoms worse in CD, UC, and UC patients with pouch. |
| Cross-sectional study | Limdi et al. 2016 [80] | 400 IBD patients | A questionnaire assessing demographics, dietary beliefs, and habits in IBD patients | Alcohol was implicated in the worsening of symptoms in 21% of patients. |
| Cross-sectional study | Guida et al. 2021 [75] | 81 CD 86 UC most patients were in remission or had mild disease activity | A semi-structured questionnaire consisting of 48 questions | 18.6% of patients perceived alcoholic drinks as a symptoms trigger. |
| Group of Medications | The Chemical Name of the Active Substance | Mechanism of Interaction | Possible Clinical Implications |
|---|---|---|---|
| Antibiotics | Metronidazole | Disulfiram-like—inhibition of aldehyde dehydrogenase [88] | Facial flushing, diaphoresis, tachycardia, hypotension, abdominal pain, pounding headache |
| Immunomodulator/immunosuppressants | 5-aminosalicylates | Interference with modified-release formulations [89] | Reduced effectiveness |
| Cyclosporine | Decreased or increased drug-circulating level [90] | Reduced effectiveness or toxicity | |
| Azathioprine | Glutathione depletion in hepatic endothelial cells leading to increased azathioprine toxicity [91] | Peliosis hepatis | |
| Methotrexate | Direct hepatotoxicity via inhibition of DNA and RNA synthesis [92,93] | Liver injury | |
| Analgesics | Paracetamol | Increased metabolism of paracetamol by CYP2E1 [94] | Higher risk of hepatotoxicity |
| Study | Author Year | IBD Patients | Major Findings |
|---|---|---|---|
| Cross-sectional study | Swanson et al. 2010 [76] | 129 patients with inactive disease (52 CD and 38 UC) | Abstainers: 38% CD; 37% UC Current drinkers 62% CD; 63% UC Light drinkers: 21% CD; 26% UC Moderate drinkers: 33% CD; 24% UC; Heavy drinkers: 0% CD; 3% UC; Binge drinkers: 19% CD;16% UC |
| Cross-sectional study | Plevinsky et al. 2019 [96] | 132 adolescents and young adults (age 16–25 years) | Substance use (tobacco use, marijuana use, and binge drinking) in the last 30 days: global users (n = 17), marijuana users engaging in binge drinking (n = 18), exclusive binge drinkers (n = 21), and global abstainers (n = 76) Older age, male gender, active disease, at least 1 hospitalization in the past year, low self-efficacy, low HRQoL, and high adherence barriers were significantly more likely for those reporting multi substance use. All those reporting both marijuana use and binge drinking also reported tobacco use. |
| Case control study | Thavamani et al. 2019 [95] | IBD (n = 58,020) Controls (n = 11,258,430) age 5–24 years | Prevalence of psychiatric disorders 21.6% The odds ratio for substance misuse * 2.78 |
| Retrospective cohort study (Canada) | Vigod et al. 2019 [98] | New-onset mental illness from conception to 1-year postpartum was compared between 3721 women with and 798,908 without IBD | Women with IBD were at an increased risk of new-onset psychiatric diagnosis in the postpartum period, but not during pregnancy. The risk was specifically elevated for a new-onset mood or anxiety disorder (aHR 1.14, 95% CI 1.04 to 1.26) and alcohol or substance use disorders (aHR 2.73, 95% CI 1.42 to 5.26). |
| Retrospective cohort study | Butwicka et al. 2020 [41] | 6464 early onset IBD patients | Frequency and Absolute Incidence Rates per 1000 Person-Years (Incidence Rate): IBD patients: 250/70,343 (3.6) compared to population: 11,682/3,531,789 (3.3); IBD patients: 175/49,178 (3.6) compared to siblings: 187/72,500 (2.6) |
| Retrospective cohort study (Sweden; 1973–2013) | Ludwigsson et al. 2021 [97] | 69,865 adult-onset IBD patients (UC: 43,557; CD: n = 21,245; IBD-unclassified: n = 5063) compared to 3,472,913 general population references and 66,292 siblings | Absolute incidence rates per 1000 person-years for substance misuse * in patients with adult-onset IBD (97,232/47,852,723) compared with matched reference individuals (2065/925,841; p = 0.0002 and IBD patients (1202/528,668) compared with their siblings without IBD (1913/964,487; <0.0001). |
| Cross-sectional study | Carney et al. 2021 [99] | 247 IBD | SUD ** 16.6% Factors associated with elevated odds of SUD were ever smoking (adjusted odds ratio [aOR], 2.96; 95% confidence interval [CI], 1.17–7.50), male sex (aOR, 2.44; 95% CI, 1.11–5.36), lifetime anxiety disorder (aOR, 2.41; 95% CI, 1.08–5.37), and higher pain impact (aOR, 1.08; 95% CI, 1.01–1.16). Alcohol abuse was the most common lifetime SUD diagnosis (9.3%), followed by alcohol dependence (7.3%) and drug abuse (7.3%). |
| Study | Author, Year | IBD Patients | Method | The Frequency of Cannabinoids Use (%) |
|---|---|---|---|---|
| Cross-sectional study | García-Planella et al. 2007 [116] | 214 | Anonymous, structured questionnaire, administered to consecutive patients with IBD of at least 2 years of duration, seen in an IBD outpatient clinic | 10 |
| Cross-sectional study | Lal et al. 2011 [117] | UC: 100 CD: 191 | Questionnaire regarding current and previous cannabis use | UC 33 CD 50 |
| Prospective cohort survey study | Ravikoff and Allegretti et al. 2013 [118] | 292 | Survey | 16.4 |
| Cross-sectional study | Storr et al. 2014 [119] | 313 | Questionnaire | 17.6 |
| Retrospective cohort study | Weiss and Friedenberg 2015 [120] | 2,084,895 | Survey | 67.3 |
| Cross-sectional study | Phatak et al. 2017 [121] | 53 | Survey | 70 |
| Descriptive study | Hoffenberg et al. 2018 [122] | UC: 27 CD: 62 unknown colitis: 10 | Questionnaire | 32 |
| Cross-sectional study | Benson et al. 2020 [123] | 838 | Anonymous online survey | 25.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuźnicki, P.; Neubauer, K. Emerging Comorbidities in Inflammatory Bowel Disease: Eating Disorders, Alcohol and Narcotics Misuse. J. Clin. Med. 2021, 10, 4623. https://doi.org/10.3390/jcm10194623
Kuźnicki P, Neubauer K. Emerging Comorbidities in Inflammatory Bowel Disease: Eating Disorders, Alcohol and Narcotics Misuse. Journal of Clinical Medicine. 2021; 10(19):4623. https://doi.org/10.3390/jcm10194623
Chicago/Turabian StyleKuźnicki, Paweł, and Katarzyna Neubauer. 2021. "Emerging Comorbidities in Inflammatory Bowel Disease: Eating Disorders, Alcohol and Narcotics Misuse" Journal of Clinical Medicine 10, no. 19: 4623. https://doi.org/10.3390/jcm10194623
APA StyleKuźnicki, P., & Neubauer, K. (2021). Emerging Comorbidities in Inflammatory Bowel Disease: Eating Disorders, Alcohol and Narcotics Misuse. Journal of Clinical Medicine, 10(19), 4623. https://doi.org/10.3390/jcm10194623






