The Impact of COVID-19 on Kidney Transplant Recipients in Pre-Vaccination and Delta Strain Era: A Systematic Review and Meta-Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Inclusion and Exclusion Criteria
2.3. Data Extraction
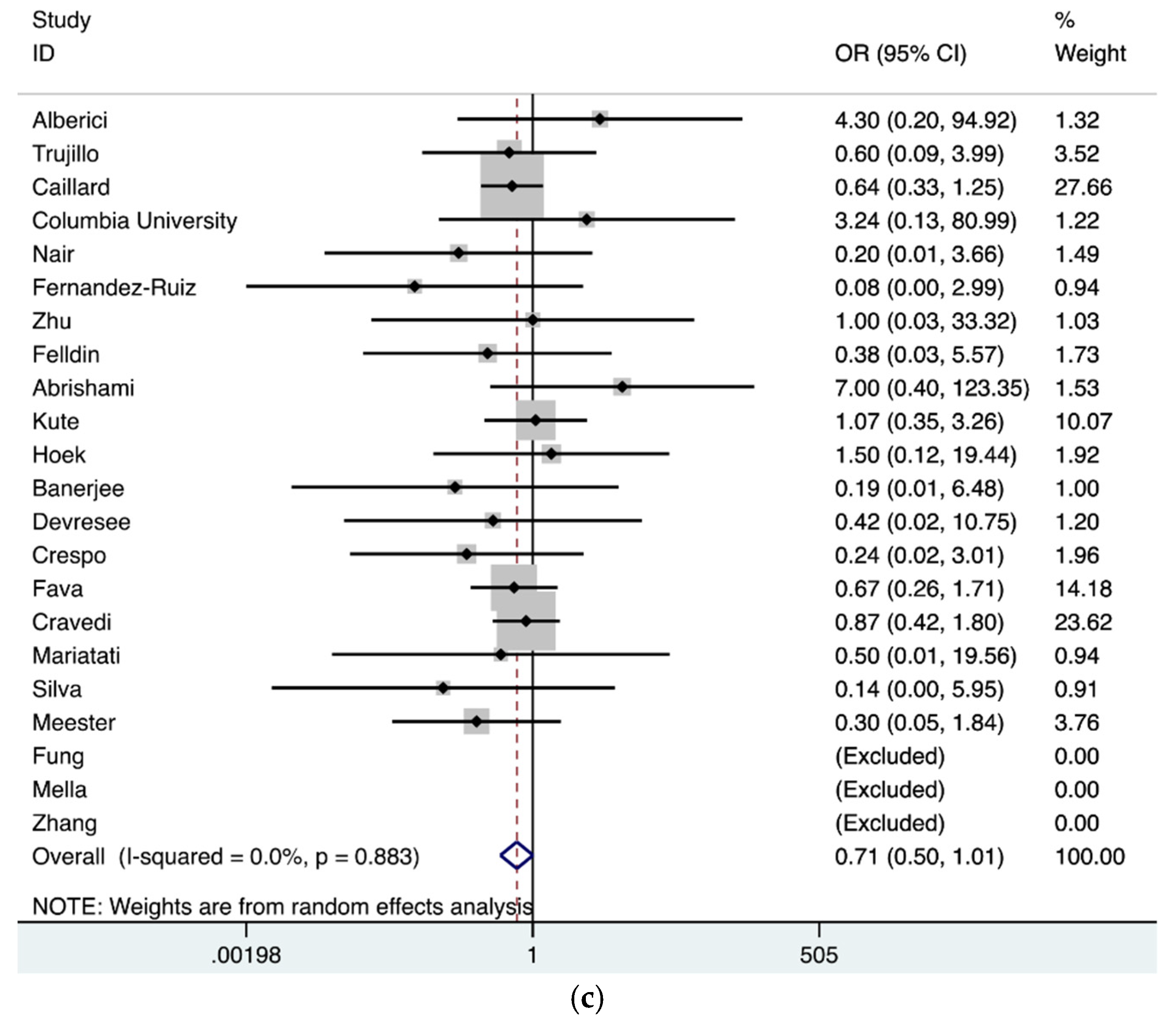
3. Results
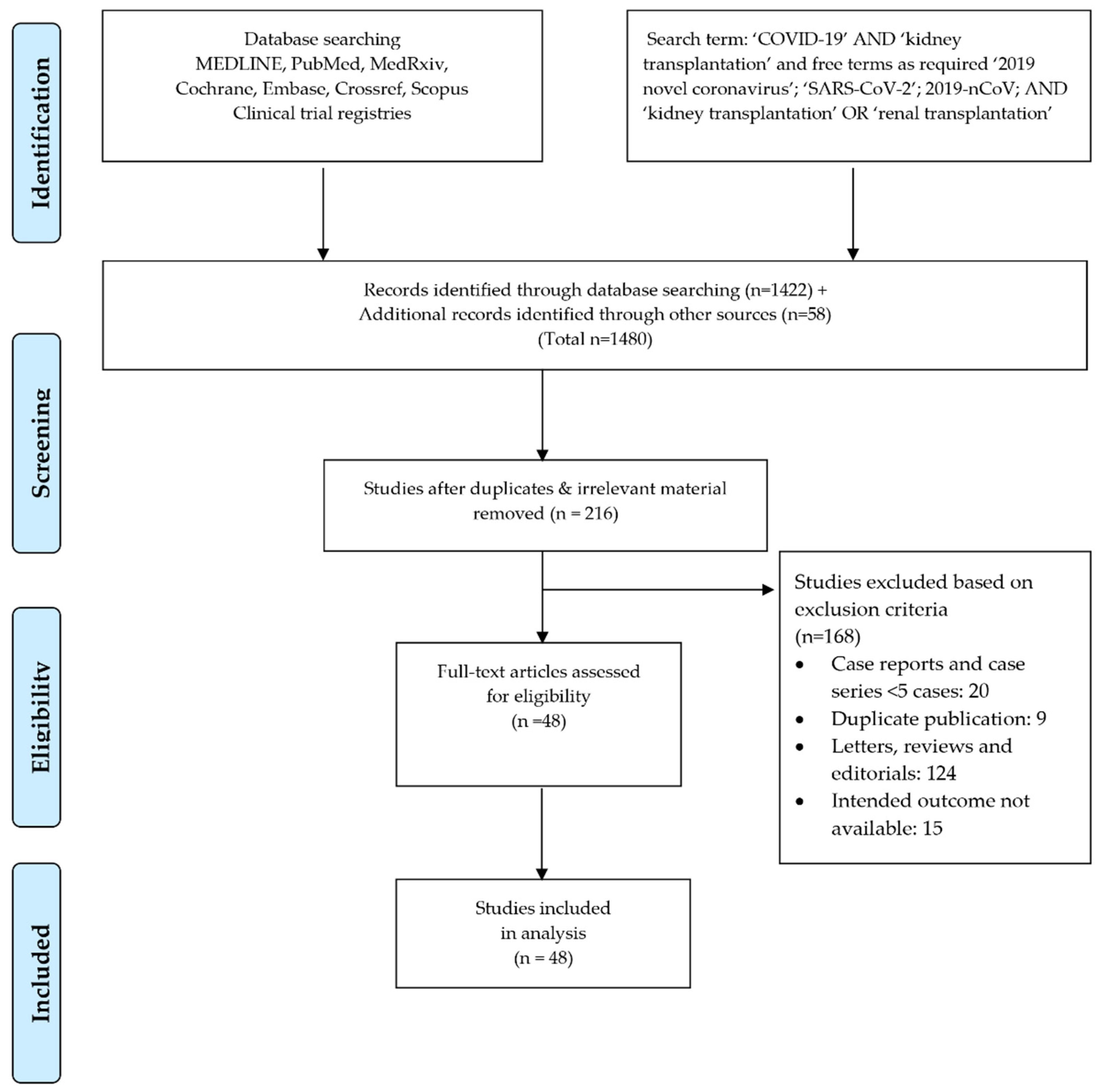
3.1. Search Results
3.2. Demographic Data
3.3. Comorbidities
3.4. Clinical Characteristics and Laboratory Results
3.5. Immunosuppression Modulation and COVID-19 Drug Treatment
3.6. Disease Severity and Mortality
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Study | Country | Number of Patients in the Study | Sex (M/F) | Age (Years) | Ethnicity | Donor Type | Comorbidities | Medications (Immunosuppression + Others) | Time Since Renal-Transplant, Median (Range) or Mean ± SD | Baseline Serum Cr, Median (Range) mg/dL |
|---|---|---|---|---|---|---|---|---|---|---|
| Akalin et al. [19] | United States | 36 | 26 (72%)/10(28%) | 57.25 ± 12.9 | African American: 14 (39%) Hispanics: 15 (42%) Others: 7 (19%) | Deceased: 27 (75%) Living: 9 (25%) | HTN: 34 (94%) DM: 25 (69%) CAD: 6 (17%) Lung disease: 4 (11%) Cancer: 2 (6%) Smoking: 6 (17%) | Tacrolimus: 35 (97%) Prednisone: 34 (94%) MMF/MPA: 31 (86%) ARB: 8 (22%) | NA | 2.4 ± 1.63 |
| Bossini et al. [20] | Italy | 53 | 42 (79%)/11(21%) | 59.25 ± 4.9 | NA | Deceased: 48 (91%) Living: 5 (9%) | HTN: 42 (79%) DM: 11 (21%) CAD: 10 (19%) Previous DVT: 4 (8%) | CNIs: 48 (90%) Prednisone: 30 (57%) MMF/MPA: 32 (60%) mTORi: 6 (11%) | 9.6 ± 3.5 years | 1.89 ± 0.33 |
| Trujillo et al. [21] | Spain | 26 | 12 (46%)/14 (54%) | 61 ± 14 | Caucasian: 22 (85%) Hispanics: 4 (15%) | NA | HTN: 24 (92%) DM: 6 (23%) CAD: 0 (0%) Lung disease: 2 (8%) | CNIs: 24 (92%) Prednisone: 22 (84%) MMF/MPA: 14 (54%) mTORi: 7 (27%) | 7 (4–15) years | 1.9 ± 0.33 |
| Columbia University KT program [22] | United States | 15 | 10 (67%)/5 (33%) | 50.25 ± 16.5 | NA | Deceased: 12 (80%) Living: 3 (20%) | NA | Tacrolimus: 14 (93%) Prednisone: 10 (67%) MMF/MPA: 12 (80%) Azathioprine: 1 (7%) Leflunomide: 1 (7%) Belatacept: 2 (13%) | 49 (0–232) months (one recent transplant < 3 months) | NA |
| Nair et al. [23] | United States | 10 | 6 (60%/4 (40%) | 56.05 ± 14.3 | African American: 3 (30%) Caucasian: 4 (40%) Asian: 1 (10%) Others: 2 (20%) | Deceased: 4 (40%) Living: 5 (50%) Unknown: 1 (10%) | HTN: 10 (100%) DM: 8 (80%) CAD: 2 (20%) Cancer: 1 (10%) Smoking history: 2 (20%) | Tacrolimus: 9 (90%) Prednisone: 7 (70%) MMF/MPA: 9 (90%) mTORi: 2 (20%) | 94.0 (4.1–248.0) months | 2.25 ± 1.15 |
| Fernandez-Ruiz et al. [24] | Spain | 8 | 7 (87%)/1 (13%) | 65.75 ± 11.8 | NA | NA | HTN: 8 (100%) DM: 4 (50%) CAD: 1 (12%) PAD: 1 (12%) Obesity: 1 (12%) | Tacrolimus: 7 (90%) Prednisone: 7 (90%) MMF/MPA: 5 (62%) mTORi: 1 (12%) ARB: 2 (25%) ACEI: 1 (12%) | 7.4 (1.8–30.1) years | NA |
| Zhu et al. [25] | China | 10 | 8 (80%)/2(20%) | 47 ± 11.8 | NA | NA | HTN: 5 (50%) CAD: 3 (12%) PAD: 1 (12%) Obesity: 1 (12%) | Tacrolimus: 9 (90%) Cyclosporine: 1(10%) Prednisone: 7 (70%) MMF/MPA: 9 (90%) Mizoribine: 1 (10%) | NA | NA |
| Cavagna et al. [26] | Italy | 6 | 5 (83%)/1 (17%) | 57.5 ± 3.8 | NA | NA | NA | Tacrolimus: 3 (50%) Cyclosporine: 3 (50%) | NA | NA |
| Fung et al. [27] | United States | 7 | 5 (71%)/2 (29%) | 60.75 ± 9.5 | African American: 3 (43%) Asian: 2 (29%) Hispanics: 1 (14%) Others: 1 (14%) | Deceased: 4 (57%) Living: 3 (43%) | HTN: 4 (57%) CAD: 4 (57%) DM: 1 (14%) | Tacrolimus: 7 (100%) Prednisone: 6 (86%) MMF/MPA: 5 (100%) | 2.9 (0.3–10.8) years 4.2 ± 3.0 | 154 ± 0.75 |
| Abrishami et al. [28] | Iran | 12 | 9 (75%)/3 (25%) | 47.5 ± 10.7 | NA | NA | HTN: 2 (57%) CAD: 4 (57%) DM: 1 (14%) | NA | 13 (3.0–18) years 11.8 ± 4.3 | 2.14 (0.9–4.6) |
| Hoek et al. [29] | Netherland | 16 (1 kidney with heart | 11(69%)/5 | 56.0 ± 17.3 | NA | Deceased: 5 (31%) Living: 11 (69%) | NA | CNI: 16 (100%) Steroid: 3 (18%) MMF/MPA: 15 (94%) | 6 (1.5–13) years | NA |
| Mella et al. [30] | Italy | 6 | 6 (100%)/0 (0%) | 55.5 ± 6.9 | NA | Deceased: 2 (33%) Living: 0 (0%) Unknown: 4 (67%) | HTN: 4 (67%) CAD: 5 (83%) DM: 1 (17%) | CNI: 6 (100%) Steroid: 6 (100%) MMF: 3 (50%) | 10.7 (0.02–14.7) years | NA |
| Montagud-Marrahi et al. [31] | Spain | 33 (2 KP) | 19 (57.6%)/14 (42.4%) | 57.3 ± 17 | NA | NA | NA | CNIs: 29 (87.8%) Prednisone: 26 (78.8%) MMF/MPA: 21 (62.5%) mTORi: 14 (42.4%) | 10.7 (4–14.7) years | NA |
| Banerjee et al. [32] | UK | 7 | 4 (57%)/3 (43%) | 55.5 ± 6.9 | NA | Deceased: 5 (71%) Living: NA (No details available in 2 cases, 1 has second transplant) | HTN: 6 (86%) DM: 3 (43%) | Tacrolimus: 6 (86%) Prednisone: 6 (86%) MMF/MPA: 5 (68%) Azathioprine: 2 (21%) ACEI: 2 (29%) | (01–360) months NA (2 in ≤ 3 months) | 2.04 (1.5–5.1) |
| Malberti et al. [33] | Italy | 11 | 10 (91%)/1 (9%) | 58.5 ± 11 | NA | NA | HTN: NA DM: 1 (9.1%) CAD: 0 (0%) Cancer: 0 (0%) | Tacrolimus: 11 (100%) MMF: 11 (100%) | NA | NA |
| Lubetzky et al. [34] | United States | 39 | 31 (79%)/8 (21%) | 57.5 ± 15.5 | African American: 10 (26%) Hispanics: 14 (36%) Caucasian: 11 (28%) Asian: 4 (10%) | Deceased: 13 (33%) Living: 26 (67%) | HTN: 37 (95%) DM: 12 (31%) CAD: 16 (41%) Lung disease: 7 (18%) Smoking: 8 (21%) | Tacrolimus: 37 (95%) Prednisone: 36 (92%) MMF/MPA: 38 (97%) ACEI: 12 (32%) | 4.7 (0.3–14.4) years | 1.58 ± 0.74 |
| Devresee et al. [35] | Belgium | 18 | 8 (44%)/ 10 (56%) | 54.5 ± 6.3 | African American: 6 (33%) Caucasian: 11 (61%) Asian: 1 (6%) | NA | HTN: 14 (78%) DM: 4 (22%) CAD: 4 (22%) Cancer: 1 (6%) Obesity: 4 (22%) | Tacrolimus: 10 (55%) Cyclosporine: 5 (27%) Prednisone: 18 (100%) MMF/MPA: 12 (67%) mTORi: 3 (17%) Azathioprine: 3 (17%) ACEI/ARB: 10 (56%) | 89 (1–402) months (3 in ≤ 3 months) | NA |
| Crespo et al. [36] | Spain | 414 | 265 (64%)/149 (36%) | 62 ± 3.17 | NA | NA | NA | CNIs: 338 (82%) Prednisone: 310 (75%) MMF/MPA: 297 (50%) mTORi: 94 (23%) | 72 (31–145) months | NA |
| Fava et al. [37] | Spain | 89 | 56 (63%)/33 (37%) | 59.7 ± 13.4 | African American: 4 (5%) Hispanic: 6 (7%) Caucasian: 78 (88%)Asian: 1 (1%) | Deceased: 75 (84%) Living: 14 (16%) | HTN: 77 (87%) DM: 25 (28%) CAD: 25 (28%) Lung disease: 14 (16%) Obesity: 24 (27%) Cancer: 7 (8%) Smoking: 8 (50%) | CNIs: 78 (88%) Prednisone: 81 (91%) MMF/MPA: 73 (82%) mTORi: 18 (20%) ACEI/ARB: 33 (37%) | 4.7 (NA) years (11 in ≤6 months) | 1.8 ± 0.8 |
| Zhang et al. [38] | China | 5 | 4 (80%)/1 (20%) | 45.0 ± 11 | NA | NA | HTN: 2 (40%) DM: 1 (20%) Cancer: 1 (20%) | CNIs: 5 (100%) Prednisone: 5 (100%) MMF/MPA: 5 (100%) | 1.53 ± 1.2 years | NA |
| Cravedi et al. [39] | Multicentric | 144 | 94 (65.3%)/50 (34.7%) | 61.25 ± 4.90 | African American: 24 (25%) Hispanic: 56 (40%) Caucasian: 43 (31%) Others: 7 (5%) | Deceased: 112 (78%) Living: 32 (22%) | HTN: 137 (95%) DM: 75 (52%) CAD: 41 (29%) Lung disease: 27 (19%) Obesity: 71 (50%) Cancer: 22 (15%) Smoking: 39 (29%) | Tacrolimus: 131 (91%) Prednisone: 125 (87%) mTORi: 11 (7.6%)MMF/MPA: 111 (77%) ARB: 24 (17%) ACEI: 20 (14%) | 5.31 ± 2.1 years | 1.5 ± 0.30 |
| Maritati et al. [40] | Italy | 5 | 3 (60%)/2 (40%) | 66.00 ± 9.27 | NA | Deceased: 4 (80%) Living: 1 (20%) | HTN: 5 (100%) DM: 1 (20%) CAD: 2 (40%) Obesity: 2 (40%) | Tacrolimus: 5 (100%) Prednisone: 5 (100%) mTORi: 1 (20%) MMF/MPA: 4 (80%) | 4.20 ± 3.1 years | 1.85 ± 0.80 |
| Elias et al. [41] | France | 66 | 37 (56%)/29 (44%) | 56.40 ± 12.5 | Caucasian: 42 (64%) Others: 24 (36%) | Deceased: 64 (97%) Living: 2 (3%) | NA | Tacrolimus: 57 (86%) Prednisone: 55 (83%) MMF/MPA: 61 (92%) Belatacept: 6 (9%) | 4.81 ± 3.9 years | NA |
| Chen et al. [42] | United States | 30 | 16 (53%)/14 (37%) | 56 ± 12 | African American: 22 (73%) Hispanic: 5 (17%) Caucasian: 2 (7%) Asian: 1 (3%) | Deceased: 18 (60%) Living: 12 (40%) | HTN: 29 (97%) DM: 14 (47%) CAD: 11 (37%) Lung disease: 0 (0%) Obesity: 10 (33%) | CNIs: 29 (97%) Prednisone: 30 (100%) MMF/MPA: 12 (40%) | 8.4 ± 2.9 years | 1.9 ± 0.42 |
| Mehta et al. [43] | United States | 34 | 22 (65%)/12 (45%) | 58.6 ± 3.25 | African American: 15 (44%) Hispanic: 8 (23%) Caucasian: 7 (21%) Asian: 2 (7%) | Deceased: 27 (79%) Living: 7 (21%) | NA | Tacrolimus: 30 (88%) Prednisone: 34 (100%) Belatacept: 6 (18%) MMF: 33 (97%) mTORi: 1 (3%) | 1.21 ± 0.56 years | NA |
| Silva et al. [44] | Portugal | 5 | 3 (75%)/2 (25%) | 50.4 ± 14.4 | NA | Deceased: 4 (80%) Living: 1 (20%) | HTN: 5 (100%) DM: 2 (40%) CAD: 1 (20%) Lung disease: 1 (20%) Obesity: 3 (60%) Smoking: 1 (20%) | CNI: 5 (100%) Prednisone: 5 (100%) MMF: 3 (60%) Azathioprine: 2 (40%) | 5.1 ± 5.9 years | 1.6 ± 0.42 |
| Chavarot et al. [45] | France | 100 | 64 (64%)/36 (36%) | 64.45 ± 5.13 | NA | NA | HTN: 85 (85%) DM: 48 (48%) CAD: 18 (18%) Lung disease: 89 (89%) | CNI: 78/94 (83%) Prednisone: 91/94 (97%) MMF: 69/94 (73%) mTORi: 8/94 (9%) Azathioprine: 7/94 (7.4%) | 6.4 ± 2.9 years | 1.3 ± 0.26 |
| Meester et al. [46] | US and European countries | 46 | 26 (56%)/20 (44%) | NA | NA | NA | NA | NA | NA | NA |
| Abolghasemi et al. [47] | Iran | 24 | 15 (62.5%)/9 (37.5%) | 47.75 ± 10.09 | NA | Deceased: 18 (75%) Living: 6 (25%) | HTN: 15 (62.5%) DM: 5 (20%) CAD: 4 (17%) Lung disease: 1 (4%) | CNI: 22 (92%) Prednisone: 24 (100%) MMF: 20 (83%) mTORi: 1 (4%) Azathioprine: 2 (8%) | 10.3 (1-20) years | NA |
| Cucchiari et al. [48] | Spain | 28 | 18 (64%)/10 (36%) | 54.81 ± 6.3 | NA | NA | NA | CNI: 23 (82%) MPA: 14 (50%) mTORi: 9 (32%) | 5.4 ± 2.5 years | 1.60 ± 0.36 |
| Kute et al. [49] | India | 250 | 215 (86%)/35 (14%) | 43.00 ± 4.6 | NA | Deceased: 24 (9.6%) Living: 226 (90%) | HTN: 210 (84%) DM: 80 (32%) CAD: 30 (12%) Obesity: 53 (24%) | CNI: 236 (94%) Prednisone: 250 (100%) MMF: 250 (100%) mTORi: 14 (5.6%) | 3.75 ± 1.29 years | 1.88 ± 0.42 |
| Caillard et al. [50] | France | 243 | 162 (67%)/81 (33%) | 60.75 ± 5.25 | NA | NA | HTN: 201 (83%) DM: 92 (39%) CAD: 81 (33%) Cancer: 35 (14%) Smoking: 30 (12%) | CNI: 202 (83%) Prednisone: 177 (73%) MMF: 183 (75%) mTORi: 29(12%) Belatacept: 15(6.1%) | 6.08 ± 2.26 years | 2.05 ± 0.42 |
| Kutzler et al. [51] | United States | 10 | NA | 62.60 ± 7.00 | African American: 2 (20%) Hispanic: 3 (30%) Caucasian: 5 (50%) | NA | HTN: 8 (80%) DM: 5 (50%) CAD: 6 (60%) Cancer: 2 (20%) Obesity: 2 (20%) | CNI: 5 (50%) Prednisone: 7 (70%) MMF: 5 (50%) mTORi: 1(10%) Belatacept: 10(10%) | 10.98 ± 8.7 years | NA |
| Mairons et al. [52] | Spain | 30 | NA | NA | NA | NA | NA | NA | NA | NA |
| Sharma et al. [53] | United States | 16 | NA | NA | NA | NA | NA | NA | NA | NA |
| Mohamed et al. [54] | United Kingdom | 35 | 16 (57%)/11 (43%) | 52.75 ± 13.55 | NA | Deceased: 22 (79%) Living: 13 (21%) | HTN: 23 (85%) DM: 10 (37%) CAD: 5 (18%) Obesity: 21 (75%) Lung disease: 4 (14%) | CNI: 27/27 (100%) Prednisone: 27/27 (100%) MMF: 21/27 (78%) Azathioprine: 3/27 (10%) | 6.38 ± 5.43 years | 2.08 ± 0.97 |
| Felldin et al. [55] | Sweden | 22 | 18 (82%)/4 (18%) | 54.11 ± 11.26 | NA | NA | HTN: 16 (73%) DM: 7 (32%) CAD: 2 (9%) Lung disease: 2 (9%) Obesity: 10 (45%) | CNI: 22 (100%) Prednisone: 23 (57.5%) MMF: 12 (55%) Azathioprine: 2 (9%) | 10.99 ± 1.74 years | NA |
| Kates et al. [56] | United States | 318 | NA | NA | NA | NA | NA | NA | NA | NA |
| Benotmane et al. [57] | France | 40 | 31 (77.5%)/9 (22.5%) | 63.8 ± 3.4 | NA | NA | HTN: 33 (82.5%) DM: 19 (47.5%) CAD: 16 (40%) Lung disease: 9 (22.5%) Obesity: 20 (50%) | CNI: 35 (87.5%) Prednisone: 23 (57.5%) MMF: 34 (85%) Azathioprine: 1 (2.5%) | NA | |
| Chaudhry et al. [58] | United States | 38 | 26 (68%)/12 (32%) | 62 ± 5.75 | African American: 31 (81.5%) | NA | HTN: 37 (97%) DM: 27 (71%) CAD: 11 (29%) Lung disease: 5 (13%) Smoking: 7 (18%) | NA | NA | NA |
| Craig-Schapiro et al. [59] | United States | 80 | 56 (70%)/24 (30%) | 57 ± 9.16 | African American: 21 (26%) Hispanic: 24 (30%) Caucasian: 25 (31%) Asian 9 (11%) | NA | HTN: 20 (25%) DM: 22 (27%) CAD: 26 (62%) Lung disease: 11 (14%) Smoking: 16 (20%) | Prednisone: 22 (51%) MMF: 52 (47%) | NA | 1.3 ± 0.22 |
| Demir et al. [60] | Turkey | 40 | 20 (50%)/20 (50%) | 44.9 ± 14.8 | NA | Deceased: 5 (12.5%) Living: 35 (87.5%) | HTN: 26 (65%) CAD: 3 (7.5%) Lung disease: 3 (7.5%) | CNI: 36 (90%) Prednisone: 40 (100%) MMF: 36 (90%) mTORi: 4 (10%) | 74.5 (31.5–128.3) months | 1.6 ± 0.25 |
| Hilbrands et al. [61] | ERACODA Collaborators, Europe | 271 | 118 (43.5%)/153 (56.5%) | 60 ± 12 | African American: 15 (0.06%) Caucasian: 151 (56%) Asian 7 (3%) | NA | HTN: 172 (63%) DM: 64 (24%) CAD: 37 (14%) Lung disease: 19 (7%) Cancer: 16 (6%) Obesity: 55 (20%) | CNI: 175 (65%) Prednisone: 173 (64%) MMF: 145 (54%) mTORi: 33 (12%) | NA | NA |
| Pascual et al. [62] | Spain | 24 | 9 (37.5%)/15 (62.5%) | 62 ± 8.75 | NA | Deceased: 23 (96%) Living: 1 (4%) | HTN: 22 (92%) DM: 12 (50%) | CNI: 24 (100%) Prednisone: 24 (100%) MMF: 21 (88%) mTORi: 2 (8%) | NA | NA |
| Rodriguez-Cubillo et al. [63] | Spain | 29 | 17 (58.6%)/12 (41.4%) | 66 ± 3.25 | NA | NA | HTN: 28 (97%) DM: 2 (40%) Obesity: 15 (52%) | CNI: 24 (83%) Prednisone: 23 (79%) MMF: 23 (79%) | NA | NA |
| Sanchez-Alvarez et al. [64] | Spain | 470 | 299 (63.6%)/171 (36.4%) | 60 ± 13 | NA | NA | NA | NA | NA | NA |
| Oto et al. [65] | Turkey | 109 | 63 (57.8%)/46 (42.2%) | 48.4 ± 12.4 | NA | Deceased: 17 (16%) Living: 92 (84%) | HTN: 81/106 (76%) DM: 25/107 (23%) CAD: 18/103 (17%) Lung disease: 5/105 (5%) Cancer: 6/105 (6%) Smoking: 23 (21%) | CNI: 95 (87%) Prednisone: 106 (97%) MMF: 94 (86%) mTORi: 12 (11%) | 5.0 (3.0–9.0) years | 1.5 (1.10–2.19) |
| Elhadedy et al. [66] | United Kingdom | 8 | 4 (50%)/4 (50%) | 50.14 ± 17.5 | NA | NA | HTN: 4/8 (50%) DM: 1/8 (13%) Lung disease: 1/8 (13%) | CNI: 8/8 (100%) Prednisone: 8/8 (100%) MMF: 7/8 (88%) | 4.6 ± 4.8 years | 3.23 ± 163.60 |
| Study | Clinical Presentation | Respiratory Disease Severity | Intensive Care Unit Admission & Organ Failure (Respiratory or Renal) | Hematological Data | Serum Markers | ||
|---|---|---|---|---|---|---|---|
| Median (Range) | Significant Decline n (%) | Median (Range) | Significant Change n (%) | ||||
| Akalin et al. [19] | Fever: 21 (58%) Dyspnea: 16 (44%) Cough: 19 (53%) GIs: 8 (22%) | Major: 28 (78%) Minor: 8 (22%) Outpatient Radiographic evidence of pneumonia: 27/28 (96%) | ICU: 11/28 (39%) MV: 11/28 (39%) AKI: NA RRT: NA | WBC: 5300 (2100–14,700)/mm3 Platelets: 146 × 103 (78 × 103–450 × 103)/mm3 Lymphocyte: 600 (100–1900)/mm3 CD3+ T cells: 319 (34–1049) CD4+ T cells: 173 (6–507) CD8+ T cells: 132 (39–654) | Thrombocytopenia (<150,000/mm3): 12/28 (43%) Lymphocytopenia (<1000/mm3): 22/28 (79%) CD3+ T cells (<706/mm3): 19/28 (68%) CD4+ T cells (<344/mm3): 20/28 (71%) CD8+ T cells (<104/mm3): 8/28 (29%) | CRP: 7.9 (0.5–48.7) mg/dL Ferritin: 1230 (191–9259) ng/mL Procalcitonin: 0.2 (0.1–5.1) ng/mL D-dimer: 1.02 (0.32–5.19) µg/mL | CRP (>5 mg/dL): 13/28 (46%) Ferritin (>900 ng/mL): 10/28 (36%) Procalcitonin (>0.2 ng/mL): 12/28 (43%) D-dimer (>0.5 µg/mL): 16/28 (57%) |
| Bossini et al. [20] | Fever: 51 (96%) Dyspnea: 15 (28%) Cough: 26 (49%) Pharyngitis: 7 (13%) Diarrhea: 9 (17%) | Hospitalized: 45 (85%) Radiological evidence of pneumonia: 38/39 (97%) | ICU: 10/45 (22%) ARDS: 27/45 (60%) MV: 9/45 (20%) AKI: 15/45 (33%) RRT: 3/15 (20%) | WBC: 5560 (4140–7400)/mm3 Platelets: 162 × 103 (129 × 103–219 × 103) Lymphocyte: 590 (430–1092)/mm3 | NA | CRP: 3.9 (1.6–10.3) mg/dL Ferritin: 433 (284–872) ng/mL D-dimer: 0.41 (0.10–0.68) µg/mL | NA |
| Trujillo et al. [21] | Fever: 12 (46%) Dyspnea: 15 (58%) Cough: 23 (89%) GIs: 10 (38%) | Hospitalized: 26 (100%) Radiological evidence of pneumonia: 17 (85%) Ground glass opacities: 16 (62%) Alveolar consolidation: 14 (54%) Bilateral infiltrates: 17 (65%) | ICU: NA ARDS: 10/26 (39%) MV: NA AKI: NA | Lymphocyte: 700 (400–1100) | NA | CRP: 13 (6–23) mg/dL D-dimer: 0.82 (0.51–0.12) µg/mL | NA |
| Columbia University KT program [22] | Fever: 13 (87%) Dyspnea: 4 (27%) Cough: 9 (60%) Diarrhea: 3 (20%) | Hospitalized: 15 (100%) Radiological evidence of pneumonia: 10 (67%) Multifocal/bilateral patchy opacities: 7 (47%) | ICU: 4/15 (27%) MV: 4/15 (27%) AKI: 6/15 (40%) RRT: 2/6 (33%) | WBC: 4800 (2100–12,700)/mm3 Lymphocyte: 800 (110–1410)/mm3 | NA | CRP: 10.4 (0.03–23.2) mg/dL Ferritin: 471 (93–1963) ng/mL Procalcitonin: 0.46 (0.08–18.7) ng/mL IL-6: 24 (<5–120) pg/mL | NA |
| Nair et al. [23] | Fever: 7 (70%) Cough: 8 (80%) Dyspnea: 3 (30%) GIs: 2 (20%) | Hospitalized: 10 (100%) Radiological evidence of pneumonia: 9 (90%) | ICU: 4/10 (40%) MV: 4/10 (40%) AKI: 5/10 (50%) RRT: 1/5 (20%) | WBC: 4300 (2380–9410)/mm3 Lymphocyte: 970 (269–1410)/mm3 | Lymphocytopenia (<1000/mm3): 5/10 (50%) | CRP: 13.3 (3.4–30.6) mg/dL Ferritin: 787.5 (101–2871) ng/mL | CRP (>5 mg/dL): 6/8 (75%) Ferritin (>900 ng/mL): 3/8 (37%) |
| Fernandez-Ruiz et al. [24] | Fever: 7 (87%) Dyspnea: 5 (62%) Cough: 4 (50%) GIs: 1 (12%) | Hospitalized: 8 (100%) Radiological evidence of pneumonia: 5/8 (62%) | ICU: 1/8 (12%) ARDS: 3/8 (38%) CPAP: 3/8 (37%) AKI: 1/8 (12%) RRT: 1/1 (100%) | NA | NA | NA | NA |
| Zhu et al. [25] | Fever: 9 (90%) Dyspnea: 9 (90%) Cough: 9 (90%) GIs: 3 (30%) | Hospitalized: 10 (100%) Radiological evidence of pneumonia: 10 (100%) B/L multiple consolidation: 8 | ICU: 1/10 (10%) Oxygen requirement: NIV: 3/10 (30%) AKI: 6/10 (60%) RRT: 1/6 (16%) | Lymphocyte: 450 (190–990)/mm3 | Lymphocytopenia (<1000/mm3): 10/10 (1000%) | CRP: 4.6 (3–11.8) mg/dL | CRP (>5 mg/dL): 4/10 (40%) |
| Cavagna et al. [26] | NA | Hospitalized: 5 Radiological evidence of pneumonia: 5 | NA | NA | NA | NA | NA |
| Fung et al. [27] | Fever: 7 (100%) Cough: 6 (86%) Dyspnea: 3 (43%) | Hospitalized: 5 (72%) Radiological evidence of pneumonia: 3 (60%) Outpatient: 2 (28%) | ICU: 2/5 (40%) ARDS: 2/5 (40%) MV: 2/5 (40%) AKI: 3/5 (60%) RRT: 1/3 (33%) | WBC: 4300 (2800–5800)/mm3 Lymphocyte: 280 (80–1480)/mm3 | Lymphocytopenia (<1000/mm3): 6/7 (86%) | CRP: 4.9 (2.3–17.7) mg/dL Procalcitonin: 0.09 (0.03–0.31) ng/mL | CRP (>5 mg/dL): 2/5 (40%) Procalcitonin (>0.2 ng/mL): 1/7 (14%) |
| Abrishami et al. [28] | Fever: 9 (75%) Dyspnea: 5 (42%) Cough: 9 (75%) GIs: 3 (25%) | Hospitalized: 12 (100%) Radiological evidence of pneumonia: 12 (100%) Bilateral infiltrates: 8 (67%) Unilateral infiltrates 4 (33%) | ICU: 10/12 (83%) ARDS: 9/12 (75%) MV: 9/12 (75%) AKI: NA RRT: NA | WBC: 5550 (2000–12,200)/mm3 Lymphocyte: 819 (80–1480)/mm3 Platelets: 154 × 103 (54 × 103–485 × 103) | Lymphocytopenia (<1000/mm3): 7/12 (58%) | CRP: 3.0 (0.3–6.6) mg/dL | CRP (>5 mg/dL): 2/12 (17%) |
| Hoek et al. [29] | NA | Hospitalized: 16 Radiological evidence of pneumonia: 16 (100%) | ICU: 1/16 (6.2%) MV: 1/16 (6.2%) AKI: NA RRT: NA | NA | NA | NA | NA |
| Mella et al. [30] | Fever: 6 (100%) Dyspnea: 2 (33%) Cough: 3 (50%) GIs: 2 (33%) | Hospitalized: 6 (100%) | ICU: 4/6 (67%) MV: 4/6 (67%) AKI: 2/6 (33%) RRT: 2/6 (33%) | WBC: 7565 (4130–13,660) Lymphocyte: 385 (230–610) | Lymphocytopenia (<1000/mm3): 6/6 (100%) | CRP: 8.2 (3.2–17.4) mg/dL Procalcitonin: 0.29 (0.12–24.1) ng/mL | CRP (>5 mg/dL): 4/6 (67%) Procalcitonin (>0.2 ng/mL): 3/6 (50%) |
| Montagud-Marrahi et al. [31] | NA | Major: 26 (79%)—Hospitalized Minor: 7 (21%)—OutpatientRadiological evidence of pneumonia: 24 (73%) | ICU: 13/26 (50%) MV: 2/26 (7.7%) RRT: NA | NA | NA | NA | NA |
| Banerjee et al. [32] | Fever: 6 (86%) Dyspnea: 6 (86%) Cough: 4 (57%) Chest pain: 1 (14%) Diarrhea: 1 (14%) | Hospitalized: 5 (71%) Outpatient: 2 (29%) Radiographic evidence of viral pneumonia: 5 (71%) | ICU: 4/5 (80%) MV: 2/5 (40%) AKI: 4/5 (80%) RRT: 3/4 (75%) | WBC: 9400 (5500–11,250) Platelets: NA Lymphocyte: 500 (300–4000) | Lymphocytopenia (<1000/mm3): 4/7 (57%) | CRP: 19.8 (8.3–32.9) mg/dL Ferritin: 6919 (NA) ng/mL Procalcitonin: NA D-dimer: 1.97 (1.91–2.03) µg/mL | CRP (>5 mg/dL): 3/7 (43%) |
| Malberti et al. [33] | NA | Hospitalized: 11 (100%) | ICU: 3/11 NIV: 4/11 (36%) (27%) MV: 3/11 (27%) AKI: 5/11 (45%) RRT: 2/5 (40%) | NA | NA | NA | NA |
| Lubetzky et al. [34] | Fever: 30 (77%) Dyspnea: 21 (54%)Cough: 22 (56%) GIs: 19 (48%) | Hospitalized: 39 (100%) Radiological evidence of pneumonia: 32/37 (86%) | ICU: 12/39 (31%) MV: 9/39 (23%) AKI: 20/39 (51%) RRT: 5/20 (25%) | WBC: NA (2500–9900) Platelets: NA Lymphocyte: 600 (300–1000) | NA | CRP: NA (5.2–35) mg/dL Ferritin: NA (307–2646) ng/mL Procalcitonin: NA (0.10–1.0) ng/mL IL-6: NA (4.5–268) pg/mL | NA |
| Devresee et al. [35] | Fever: 14 (78%) Dyspnea: 7 (39%) Cough: 12 (67%) GIs: 5 (28%) | Hospitalized: 18 (100%) Radiological evidence of pneumonia: 15 (83%) | ICU: 2/18 (31%) MV: 2/18 (23%) AKI: 5/18 (51%) RRT: 0/5 (0%) | NA | NA | NA | NA |
| Crespo et al. [36] | Fever: 340 (82%) Dyspnea: 181 (43.7%) Cough: 62 (70%) GIs: 152 (36.7%) | Hospitalized: 380 (92%) Radiological evidence of pneumonia: 337 (89%) | ICU: 50/380 (13%) MV: 73/380 (19%) | NA | NA | NA | NA |
| Fava et al. [37] | Fever: 72 (81%) Dyspnea: 34 (38%) Cough: 62 (70%) GIs: 27 (30%) | Hospitalized: 89 (100%)Radiological evidence of pneumonia: 67/89 (75%) | ICU: 17/89 (19%) ARDS: 47/89 (53%) MV: 8/89 (9%) | NA | Lymphocytopenia (<1000/mm3): 37/89 (17%) | NA | NA |
| Zhang et al. [38] | Fever: 5 (100%) Dyspnea: 0 (0%) Cough: 5 (100%) GIs: 0 (0%) | Hospitalized: 5 (100%) Radiological evidence of pneumonia: 5/5 (100%) | ICU: 0/5 (0%) ARDS: NA MV: 0/5 (0%) AKI: 1/5 (20%) RRT: 0/1 (0%) | WBC: 6.42 ± 1.09/mm3 Platelets: 186 ± 40.0/mm3 Lymphocyte: 0.58 ± 0.22/mm3 | Lymphocytopenia (<1000/mm3): 5/5 (100%) | NA | NA |
| Cravedi et al. [39] | Fever: 96 (67%) Dyspnea: 97 (68%) GIs: 55 (38%) | Hospitalized: 144 (100%) Radiological evidence of pneumonia: NA | ICU: NA ARDS: NA MV: 42/144 (29%) AKI: 74/144 (51%) RRT: NA | WBC: 7.70 ± 5.65/mm3 Platelets: 181.5 ± 29.0/mm3 Lymphocyte: 1.36 ± 0.78/mm3 | NA | CRP: 4.1 (1.1–12.5) mg/dL Ferritin: 6919 (NA) ng/mL Procalcitonin: NA D-dimer: 1.97 (1.91–2.03) µg/mL | NA |
| Maritati et al. [40] | Fever: 4 (80%) Dyspnea: 4 (80%) Cough: 4 (80%) | Hospitalized: 5 (100%) Radiological evidence of pneumonia: 5 (100%) | ICU: 3/5 (60%) ARDS: 4/5 (80%) MV: 3/5 (60%) AKI: 3/5 (60%) RRT: 1/3 (33%) | NA | NA | NA | NA |
| Elias et al. [41] | Fever: 51 (77%) Dyspnea: 26 (39%) Cough: 38 (58%) GIs: 17 (26%) | Hospitalized: 60 (91%) Radiological evidence of pneumonia: 41/56 (73.2%) | ICU: 15/60 (25%) ARDS: 38/56 (68%) MV: 15/38 (39%) AKI: 28/60 (42%) RRT: 7/28 (35%) | WBC: 6.90 ± 3.64/mm3 Lymphocyte: 0.60 ± 0.35/mm3 | NA | CRP: 11.5 (0.3–33.5) mg/dL | NA |
| Chen et al. [42] | Fever: 22 (73%) Dyspnea: 20 (67%) Cough: 22 (72%) GIs: 13 (43%) | Hospitalized: 30 (100%) Radiological evidence of pneumonia: NA | ICU: NA ARDS: NA MV: 7/30(23%) AKI: 23/30 (77%) RRT: 4/23 (17%) | WBC: 6.75 ± 1.28/mm3 Lymphocyte: 0.73 ± 0.25/mm3 | Lymphocytopenia (<1000/mm3): 30/30 (100%) | CRP: 7.6 (4.4–14.7) mg/dL Ferritin: 979 (422–1977) ng/mL Procalcitonin: 0.2 (0.1–1.3) ng/mL | NA |
| Mehta et al. [43] | Fever: 19 (56%) Dyspnea: 18 (53%) GIs: 5 (15%) | Hospitalized: 34 (100%)Radiological evidence of pneumonia: 32/34 (94.11) | ICU: NAARDS: NAMV: 13/34(38%) AKI: 18/34 (53%) RRT: 0/18 (0%) | Lymphocyte: 0.43 ± 0.22/mm3 | Lymphocytopenia (<1000/mm3): 32/34 (94%) | NA | NA |
| Silva et al. [44] | Fever: 5 (100%) Dyspnea: 1 (20%) Cough: 2 (40%) GIs: 3 (60%) | Hospitalized: 5 (100%) Radiological evidence of pneumonia: 4/5 (80%) | ICU: 1/5 (20%) ARDS: 3/5 (60%) MV: 1/5 (20%) AKI: 3/5 (60%) RRT: 0/3 (0%) | WBC: 5.57 ± 1.77/mm3 Lymphocyte: 1.87 ± 1.10/mm3 | Lymphocytopenia (<1000/mm3): 1/5 (20%) | CRP: 5.6 (0.6–12.4) mg/dL | CRP (>5 mg/dL): 2/5 (40%) |
| Chavarot et al. [45] | Fever: 67/93 (72%) Dyspnea: 41/89 (46%)Cough: 55/87 (63%) GIs: 32/89 (36%) | Hospitalized: 100 (100%) Radiological evidence of pneumonia: 40/83 (48%) | ICU: 34 (34%) MV: 29 (29%) | NA | NA | NA | NA |
| Meester et al. [46] | NA | NA | NA | NA | NA | NA | NA |
| Abolghasemi et al. [47] | Fever: 15 (62.5%) Dyspnea: 17 (71%) Cough: 11 (46%) GIs: 6 (25%) | Hospitalized: 24 (100%) Radiological evidence of pneumonia: 23/24 (96%) | ICU: 12/24 (50%) | NA | Lymphocytopenia (<1100/mm3): 19/24 (79%) | NA | CRP (>10 mg/dL): 24/24 (100%) |
| Cucchiari et al. [48] | Fever: 26 (93%) Dyspnea: 9 (32%) Cough: 18 (64%) GIs: 7 (25%) | Hospitalized: 28 (100%) | ICU: 8/28 (29%) ARDS: 25/28 (89%) AKI: 19/28 (68%) RRT: 3/19 (0%) | NA | NA | NA | NA |
| Kute et al. [49] | Fever: 220 (88%) Dyspnea: 55 (22%) Cough: 180 (72%) GIs: 60 (24%) | Hospitalized: 200 (80%) Radiological evidence of pneumonia: 120/250 (48%) | ICU: 53/200 (26%) MV: 30/200 (15%) AKI: 121/200 (60%) RRT: 24/121 (20%) | NA | NA | NA | NA |
| Caillard et al. [50] | Fever: 180 (74%) Dyspnea: 98 (40%) Cough: 145 (60%) GIs: 97 (25%) | Hospitalized: 243 (100%) | ICU: 88/243 (36%) MV: 72 (30%) AKI: 106/243 (43.6%) RRT: 27/106 (25%) | Lymphocyte: 1.87 ± 1.10/mm3 | NA | CRP: 6.2(2.7–11.4) mg/dL Procalcitonin: 0.2 (0.1–0.48) ng/mL | NA |
| Kutzler et al. [51] | Fever: 5(50%) Dyspnea: 5 (50%) Cough: 6 (60%) GIs: 2 (20%) | Hospitalized: 10 (100%) | NA | NA | NA | NA | NA |
| Mairons et al. [52] | NA | NA | NA | NA | NA | NA | NA |
| Sharma et al. [53] | NA | NA | NA | NA | NA | NA | NA |
| Mohamed et al. [54] | Fever: 13/25(52%) Dyspnea: 17/25 (68%) Cough: 13/24 (54%) GIs: 7/25 (28%) | Hospitalized: 27/28 (96%) Radiological evidence of pneumonia: 19/28 (76%) | ICU: 5/28 (18%) AKI: 14/25 (56%) RRT: 2/14 (14.3%) | WBC: 8.65 ± 4.34/mm3 Lymphocyte: 0.77 ± 0.47/mm3 | NA | CRP: 9.4 (0.7–44.6) mg/dL Ferritin: 1101 (49–5959) ng/mL | NA |
| Felldin et al. [55] | NA | Hospitalized: 22/34 (65%) | ICU: 7/22 (36%) MV: 6/22 (27%) AKI: 14/22 (64%) RRT: 5/14 (36%) | NA | NA | NA | NA |
| Kates et al. [56] | NA | NA | NA | NA | NA | NA | NA |
| Benotmane et al. [57] | Fever: 38/40 (95%) Dyspnea: 28/40 (70%) Cough: 31/40 (77.5%) GIs: 31/40 (77.5%) | Hospitalized: 40/40 (100%) | NA | NA | NA | NA | NA |
| Chaudhry et al. [58] | NA | Hospitalized: 26/40 (68%) | ICU: 12/26 (46.1%) ARDS: 11/26 (42.3%) MV: 11/26 (42.3%) AKI: 5/26 (19%) | NA | NA | NA | NA |
| Craig-Schapiro et al. [59] | Fever: 53/75 (71%) Dyspnea: 36/74 (49%) Cough: 44/74 (59%) GIs: 25/73 (34%) | Hospitalized: 52/80 (68%) Radiological evidence of pneumonia: 39/45 (87%) | ICU: 16/52 (30.7%) MV: 18/51 (35.3%) AKI: 26/46 (56.5%) RRT: 4/26 (15.3%) | WBC: 5.70 ± 0.85/mm3 Lymphocyte: 0.60 ± 0.17/mm3 | NA | CRP: 12.3 (7.2–30.2) mg/dL Ferritin: 1424 (503–2428) ng/mL Procalcitonin: 0.3 (0.1–0.6) ng/mL | NA |
| Demir et al. [60] | Fever: 25/40 (62.5%) Dyspnea: 21/40 (52.5%) Cough: 30/40 (75%) GIs: 10/25 (40%) | Hospitalized: 40/40 (100%) | ICU: 7/40 (18%) MV: 6/40 (15%) | WBC: 5.15 ± 0.75/mm3 Lymphocyte: 0.72 ± 0.14/mm3 | NA | CRP: 4.5 (2.4–8.8) mg/dL Ferritin: 358 (173–1374) ng/mL Procalcitonin: 0.2 (0.1–0.3) ng/mL D-dimer: 0.36 (0.17–0.99) µg/mL | NA |
| Hilbrands et al. [61] | Fever: 155/271 (57%) Dyspnea: 107/271 (39%) Cough: 136/271 (50%) GIs: 65/271 (24%) | Hospitalized: 271/305 (89%) | ICU: 58/271 (40%) | NA | NA | NA | NA |
| Pascual et al. [62] | Fever: 15/24 (63%) Dyspnea: 14/24 (58%) Cough: 14/24 (58%) GIs: 3/24 (13%) | Hospitalized: 24/24 (100%) | ICU: 4/24 (17%) MV: 9/24 (38%) AKI: 13/24 (54%) | NA | Lymphocytopenia (<1100/mm3): 24/24 (100%) | NA | NA |
| Rodriguez-Cubillo et al. [63] | Fever: 23/29 (79%) Dyspnea: 12/29 (41%) Cough: 18/29 (62%) GIs: 14/29 (48%) | Hospitalized: 29/29 (100%) Radiological evidence of pneumonia: 18/29 (62%) | ICU: 5/29 (17%) MV: 4/29 (14%) AKI: 14/29 (48%) RRT: 3/14 (21%) | NA | NA | NA | NA |
| Sanchez-Alvarez et al. [64] | NA | Hospitalized: 437/470 (93%) | NA | NA | NA | NA | NA |
| Oto et al. [65] | Fever: 70/109 (64%) Dyspnea: 53/109 (41%) Cough: 72/109 (66%) GIs: 12/109 (11%) | Hospitalized: 109/109 (100%) | ICU: 22/109 (20%) MV: 19/109 (17%) AKI: 46/109 (42%) RRT: 4/46 (9%) | NA | Lymphocytopenia (<1100/mm3): 77/108 (71%) | NA | CRP (>5 mg/dL): 47/109 (43%) |
| Elhadedy et al. [66] | Fever: 5/8 (63%) Dyspnea: 2/8 (25%) Cough: 5/8 (63%) | Hospitalized: 8/8 (100%) Radiological evidence of pneumonia: 5/8 (63%) | ICU: 1/8 (13%) MV: 1/8 (13%) AKI: 2/8 (25%) RRT: 1/2 (50%) | NA | Lymphocytopenia (<1100/mm3): 8/8 (100%) | CRP: 12.74 ± 10.0 mg/dL | CRP (>5 mg/dL): 6/8 (75%) |
| Study | Modification of Immunosuppression | Treatments/Therapies | Outcomes | Remarks |
|---|---|---|---|---|
| Akalin et al. [19] |
| Hospitalized-
|
|
|
| Bossini et al. [20] |
| Hospitalized-
|
|
|
| Trujillo et al. [21] |
| Hospitalized-
|
|
|
| Columbia University KT program [22] |
| Hospitalized-
|
|
|
| Nair et al. [23] |
| Hospitalized-
|
|
|
| Fernandez-Ruiz et al. [24] |
| Hospitalized-
|
|
|
| Zhu et al. [25] |
|
|
|
|
| Cavagna et al. [26] | NA | NA |
|
|
| Fung et al. [27] |
|
|
|
|
| Abrishami et al. [28] |
|
|
|
|
| Hoek et al. [29] |
| Hospitalized-
|
|
|
| Mella et al. [30] |
|
|
|
|
| Montagud-Marrahi et al. [31] |
| Hospitalized-
|
|
|
| Banerjee et al. [32] |
| NA |
|
|
| Malberti et al. [33] |
| Hospitalized-
|
|
|
| Lubetzky et al. [34] |
| Hospitalized-
|
|
|
| Devresee et al. [35] |
| Hospitalized-
|
|
|
| Crespo et al. [36] |
|
|
|
|
| Fava et al. [37] |
|
|
|
|
| Zhang et al. [38] |
|
|
|
|
| Cravedi et al. [39] |
|
|
|
|
| Maritati et al. [40] |
|
|
|
|
| Elias et al. [41] |
|
|
|
|
| Chen et al. [42] |
|
|
|
|
| Mehta et al. [43] |
|
|
|
|
| Silva et al. [44] |
|
|
|
|
| Chavarot et al. [45] |
|
|
|
|
| Meester et al. [46] | NA | NA |
|
|
| Abolghasemi et al. [47] | NA |
|
|
|
| Cucchiari et al. [48] |
|
|
|
|
| Kute et al. [49] |
|
|
|
|
| Caillard et al. [50] |
|
|
|
|
| Kutzler et al. [51] |
|
|
|
|
| Mairons et al. [52] | NA | NA |
|
|
| Sharma et al. [53] | NA | NA |
|
|
| Mohamed et al. [54] |
| NA |
|
|
| Felldin et al. [55] |
| NA |
|
|
| Kates et al. [56] | NA | NA |
|
|
| Benotmane et al. [57] |
|
|
|
|
| Chaudhry et al. [58] | NA | NA |
|
|
| Craig-Schapiro et al. [59] |
|
|
|
|
| Demir et al. [60] |
|
|
|
|
| Hilbrands et al. [61] |
|
|
|
|
| Pascual et al. [62] |
|
|
|
|
| Rodriguez-Cubillo et al. [63] |
|
|
|
|
| Sanchez-Alvarez et al. [64] |
|
|
|
|
| Oto et al. [65] |
|
|
|
|
| Elhadedy et al. [66] |
| NA |
|
|
| Attributes | Events | Total | Studies | Single Group Summary (95% CI) |
|---|---|---|---|---|
| Random-Effects Model | ||||
| Demographic variables | ||||
| Age (years) | NA | 2971 | 44 | 57.08 (54.55–59.03) |
| Male | 1911 | 2985 | 44 | 0.66 (0.61–0.70) |
| Caucasians | 401 | 860 | 14 | 0.40 (0.21–0.58) |
| African-American | 170 | 898 | 15 | 0.27 (0.19–0.36) |
| Hispanics | 136 | 589 | 13 | 0.19 (0.10–0.28) |
| Deceased donor transplant | 492 | 996 | 20 | 0.58 (0.40–0.77) |
| Living donor transplant | 497 | 996 | 18 | 0.41 (0.21–0.61) |
| Comorbidities | ||||
| T2DM | 621 | 1785 | 33 | 0.34 (0.29–0.40) |
| Hypertension | 1404 | 1733 | 32 | 0.83 (0.78–0.88) |
| Heart disease | 398 | 1738 | 28 | 0.23 (0.17–0.28) |
| Chronic lung disease | 233 | 1368 | 19 | 0.15 (0.05–0.24) |
| Malignancy | 95 | 1010 | 12 | 0.08 (0.06–0.11) |
| Obesity | 239 | 713 | 14 | 0.36 (0.26–0.46) |
| Smoking | 155 | 976 | 11 | 0.15 (0.10–0.20) |
| Maintenance immunosuppression | ||||
| CNI | 1942 | 2307 | 39 | 0.91 (0.88–0.94) |
| MMF | 1784 | 2325 | 38 | 0.79 (0.73–0.85) |
| Steroid | 1876 | 2313 | 37 | 0.86 (0.82–0.90) |
| mTOR inhibitors | 267 | 1934 | 20 | 0.12 (0.09–0.16) |
| Presenting parameters and symptoms | ||||
| Fever | 1716 | 2308 | 37 | 0.77 (0.72–0.81) |
| Cough | 1362 | 2122 | 35 | 0.65 (0.61–0.69) |
| Dyspnea | 1003 | 2303 | 37 | 0.48 (0.42–0.53) |
| Gastrointestinal symptoms | 711 | 2282 | 34 | 0.29 (0.25–0.34) |
| Investigations | ||||
| Lymphocytopenia | 607 | 802 | 15 | 0.79 (0.70–0.89) |
| High CRP | 109 | 205 | 9 | 0.60 (0.35–0.84) |
| Radiological evidence of pneumonia | 982 | 1312 | 29 | 0.81 (0.74–0.88) |
| Clinical management & outcome | ||||
| ARDS | 197 | 344 | 13 | 0.58 (0.48–0.69) |
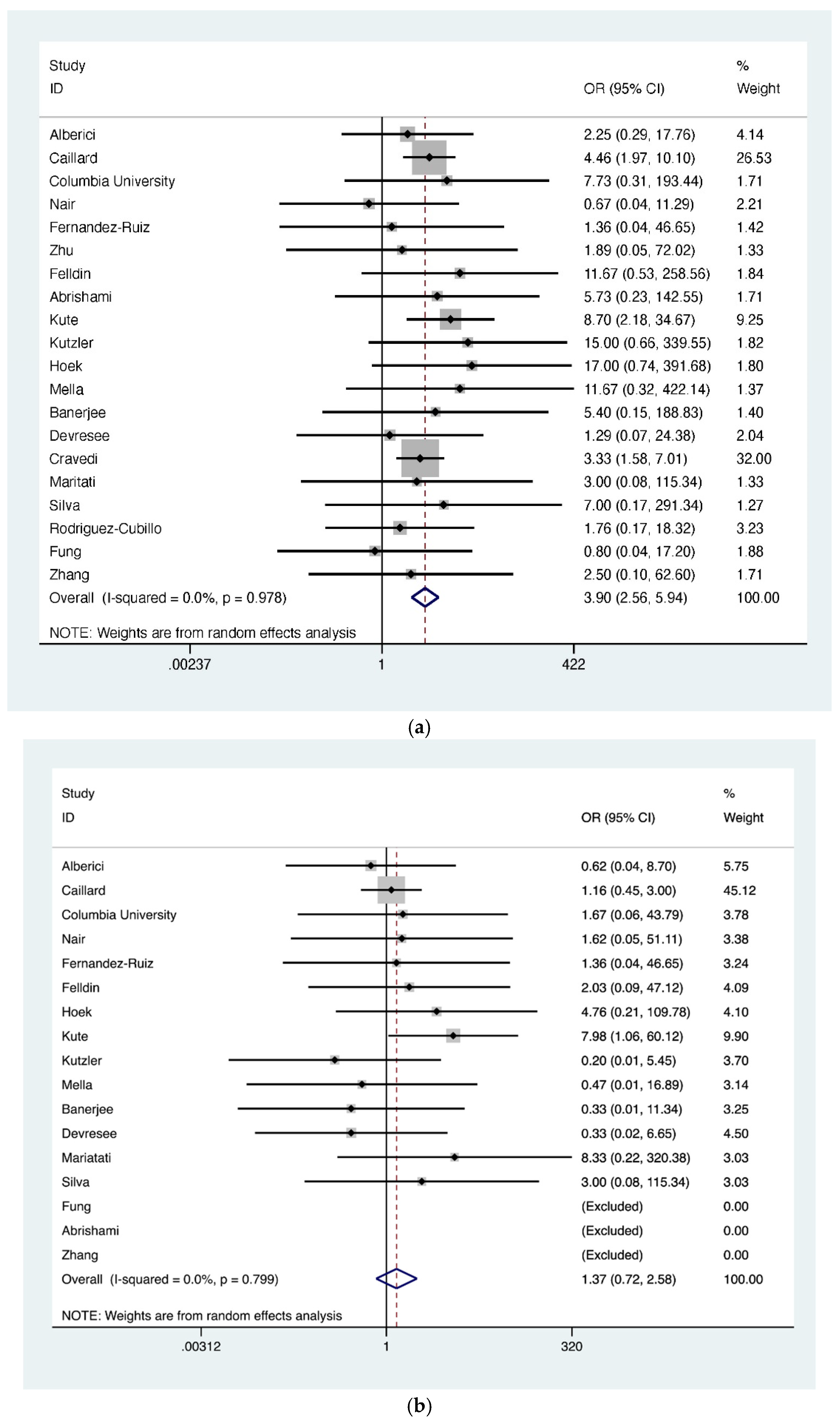
| CNI withheld/reduced | 527 | 1498 | 33 | 0.43 (0.30–0.56) |
| MMF withheld/reduced | 1181 | 1393 | 35 | 0.91 (0.88–0.94) |
| mTOR inhibitor withheld/reduced | 31 | 47 | 9 | 0.72 (0.51–0.94) |
| Increase/pulse steroid | 940 | 2172 | 31 | 0.40 (0.30–0.50) |
| HCQ | 1945 | 2623 | 34 | 0.73 (0.66–0.80) |
| Azithromycin | 739 | 1302 | 17 | 0.62 (0.47–0.76) |
| Lopinavir/ritonavir | 554 | 1861 | 18 | 0.41 (0.29–0.53) |
| Tocilizumab | 386 | 2477 | 26 | 0.21 (0.16–0.26) |
| Remdesivir | 28 | 977 | 8 | 0.03 (0.01–0.04) |
| ICU admissions | 570 | 2439 | 37 | 0.26 (0.22–0.30) |
| Mechanical ventilation | 433 | 1848 | 33 | 0.24 (0.20–0.28) |
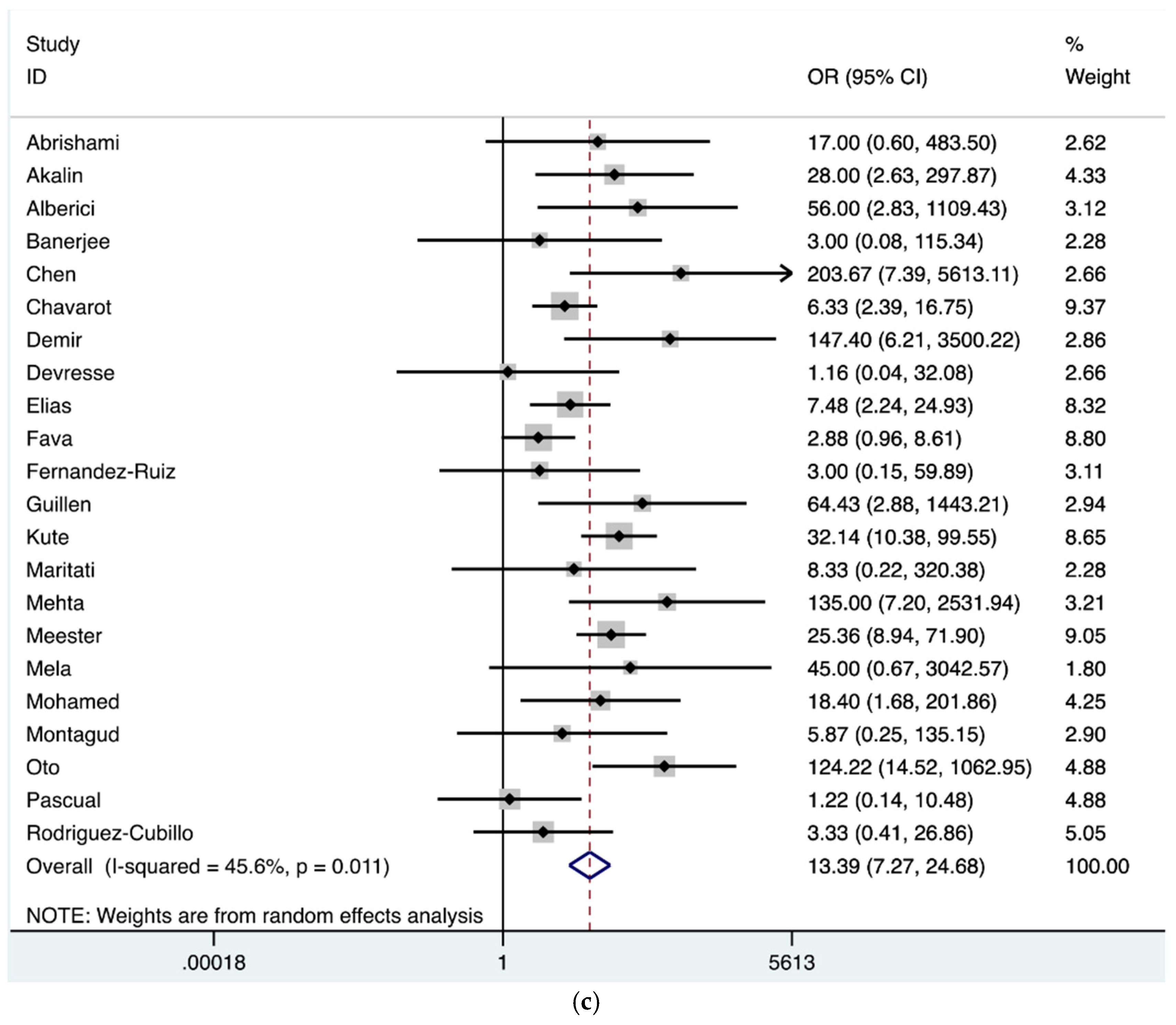
| AKI | 626 | 1260 | 30 | 0.48 (0.43–0.53) |
| Dialysis | 118 | 534 | 27 | 0.22 (0.16–0.29) |
| ICU death | 184 | 355 | 27 | 0.53 (0.44–0.63) |
| Death on mechanical ventilator | 206 | 285 | 24 | 0.68 (0.58–0.79) |
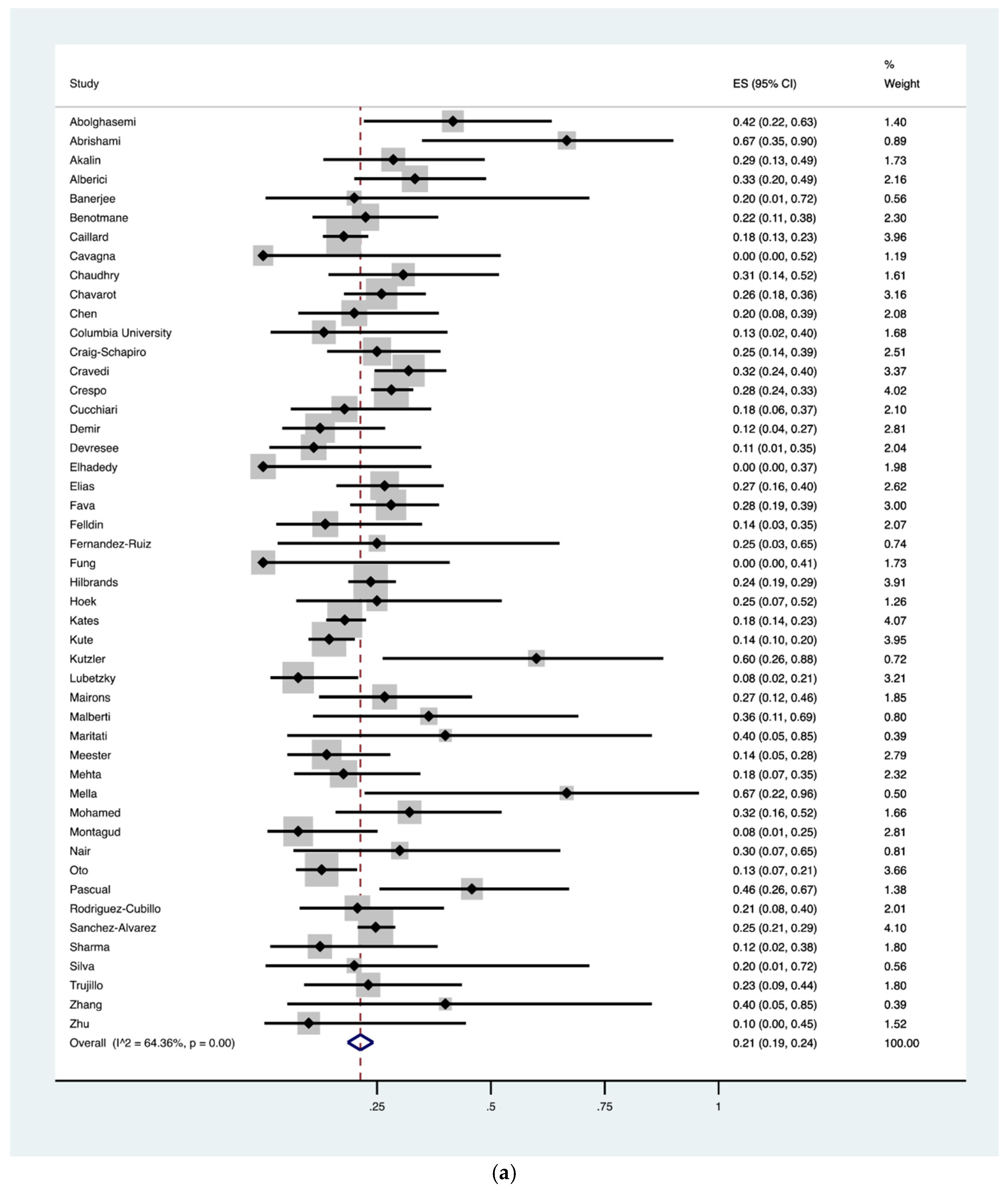
| Hospital death | 718 | 3137 | 48 | 0.21 (0.19–0.24) |
References
- Wang, L.; He, W.; Yu, X.; Hu, D.; Bao, M.; Liu, H.; Zhou, J.; Jiang, H. Coronavirus Disease 2019 in Elderly Patients: Characteristics and Prognostic Factors Based on 4-Week Follow-Up. J. Infect. 2020, 80, 639–645. [Google Scholar] [CrossRef]
- Zheng, K.I.; Gao, F.; Wang, X.-B.; Sun, Q.-F.; Pan, K.-H.; Wang, T.-Y.; Ma, H.-L.; Chen, Y.-P.; Liu, W.-Y.; George, J.; et al. Letter to the Editor: Obesity as a Risk Factor for Greater Severity of COVID-19 in Patients with Metabolic Associated Fatty Liver Disease. Metabolism 2020, 108, 154244. [Google Scholar] [CrossRef]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical Course and Risk Factors for Mortality of Adult Inpatients with COVID-19 in Wuhan, China: A Retrospective Cohort Study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Laupacis, A.; Keown, P.; Pus, N.; Krueger, H.; Ferguson, B.; Wong, C.; Muirhead, N. A Study of the Quality of Life and Cost-Utility of Renal Transplantation. Kidney Int. 1996, 50, 235–242. [Google Scholar] [CrossRef] [Green Version]
- Kostro, J.Z.; Hellmann, A.; Kobiela, J.; Skóra, I.; Lichodziejewska-Niemierko, M.; Dębska-Ślizień, A.; Śledziński, Z. Quality of Life After Kidney Transplantation: A Prospective Study. Transplant. Proc. 2016, 48, 50–54. [Google Scholar] [CrossRef]
- Kumar, D.; Manuel, O.; Natori, Y.; Egawa, H.; Grossi, P.; Han, S.; Fernández-Ruiz, M.; Humar, A. COVID-19: A Global Transplant Perspective on Successfully Navigating a Pandemic. Am. J. Transplant. 2020, 20, 1773–1779. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thaunat, O.; Legeai, C.; Anglicheau, D.; Couzi, L.; Blancho, G.; Hazzan, M.; Pastural, M.; Savoye, E.; Bayer, F.; Morelon, E.; et al. IMPact of the COVID-19 Epidemic on the MoRTAlity of Kidney Transplant Recipients and Candidates in a French Nationwide Registry STudy (IMPORTANT). Kidney Int. 2020, 98, 1568–1577. [Google Scholar] [CrossRef]
- de Jager, D.J. Cardiovascular and Noncardiovascular Mortality Among Patients Starting Dialysis. JAMA 2009, 302, 1782. [Google Scholar] [CrossRef] [Green Version]
- Carrero, J.J.; de Jager, D.J.; Verduijn, M.; Ravani, P.; De Meester, J.; Heaf, J.G.; Finne, P.; Hoitsma, A.J.; Pascual, J.; Jarraya, F.; et al. Cardiovascular and Noncardiovascular Mortality among Men and Women Starting Dialysis. Clin. J. Am. Soc. Nephrol. 2011, 6, 1722–1730. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boyarsky, B.J.; Werbel, W.A.; Durand, C.M.; Avery, R.K.; Jackson, K.R.; Kernodle, A.B.; Snyder, J.; Hirose, R.; Massie, I.M.; Garonzik-Wang, J.M.; et al. Early National and Center-level Changes to Kidney Transplantation in the United States during the COVID-19 Epidemic. Am. J. Transplant. 2020, 20, 3131–3139. [Google Scholar] [CrossRef]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D. MOOSE Guidelines for Meta-Analyses and Systematic Reviews of Observational Studies. JAMA 2000, 283, 2008–2012. [Google Scholar] [CrossRef]
- Bonanad, C.; García-Blas, S.; Tarazona-Santabalbina, F.; Sanchis, J.; Bertomeu-González, V.; Fácila, L.; Ariza, A.; Núñez, J.; Cordero, A. The Effect of Age on Mortality in Patients with COVID-19: A Meta-Analysis with 611,583 Subjects. J. Am. Med. Dir. Assoc. 2020, 21, 915–918. [Google Scholar] [CrossRef] [PubMed]
- Parohan, M.; Yaghoubi, S.; Seraji, A.; Javanbakht, M.H.; Sarraf, P.; Djalali, M. Risk Factors for Mortality in Patients with Coronavirus Disease 2019 (COVID-19) Infection: A Systematic Review and Meta-Analysis of Observational Studies. Aging Male 2020, 23, 1416–1424. [Google Scholar] [CrossRef]
- Wan, X.; Wang, W.; Liu, J.; Tong, T. Estimating the Sample Mean and Standard Deviation from the Sample Size, Median, Range and/or Interquartile Range. BMC Med Res. Methodol. 2014, 14, 135. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hozo, S.P.; Djulbegovic, B.; Hozo, I. Estimating the Mean and Variance from the Median, Range, and the Size of a Sample. BMC Med Res. Methodol. 2005, 5, 13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Higgins, J.P.T.; Thompson, S.G. Quantifying Heterogeneity in a Meta-Analysis. Stat. Med. 2002, 21, 1539–1558. [Google Scholar] [CrossRef]
- National Heart Lung and Blood Institute Quality Assessment Tool for Observational Cohort and Cross-Sectional Studies; National Institutes of Health, Department of Health and Human Services: Bethesda, MD, USA, 2014.
- Farrah, K.; Young, K.; Tunis, M.C.; Zhao, L. Risk of Bias Tools in Systematic Reviews of Health Interventions: An Analysis of PROSPERO-Registered Protocols. Syst. Rev. 2019, 8, 280. [Google Scholar] [CrossRef]
- Akalin, E.; Azzi, Y.; Bartash, R.; Seethamraju, H.; Parides, M.; Hemmige, V.; Ross, M.; Forest, S.; Goldstein, Y.D.; Ajaimy, M.; et al. COVID -19 and Kidney Transplantation. N. Engl. J. Med. 2020, 382, 2475–2477. [Google Scholar] [CrossRef]
- Bossini, N.; Alberici, F.; Delbarba, E.; Valerio, F.; Manenti, C.; Possenti, S.; Econimo, L.; Maffei, C.; Pola, A.; Terlizzi, V.; et al. Kidney Transplant Patients with SARS-CoV-2 Infection: The Brescia Renal COVID Task Force Experience. Am. J. Transplant. 2020, 20, 3019–3029. [Google Scholar] [CrossRef]
- Trujillo, H.; Caravaca-Fontán, F.; Sevillano, Á.; Gutiérrez, E.; Caro, J.; Gutiérrez, E.; Yuste, C.; Andrés, A.; Praga, M. SARS-CoV-2 Infection in Hospitalized Patients with Kidney Disease. Kidney Int. Rep. 2020, 5, 905–909. [Google Scholar] [CrossRef]
- Mohan, S. Early Description of Coronavirus 2019 Disease in Kidney Transplant Recipients in New York. J. Am. Soc. Nephrol. 2020, 31, 1150–1156. [Google Scholar] [CrossRef]
- Nair, V.; Jandovitz, N.; Hirsch, J.S.; Nair, G.; Abate, M.; Bhaskaran, M.; Grodstein, E.; Berlinrut, I.; Hirschwerk, D.; Cohen, S.L.; et al. COVID-19 in Kidney Transplant Recipients. Am. J. Transplant. 2020, 20, 1819–1825. [Google Scholar] [CrossRef]
- Fernández-Ruiz, M.; Andrés, A.; Loinaz, C.; Delgado, J.F.; López-Medrano, F.; San Juan, R.; González, E.; Polanco, N.; Folgueira, M.D.; Lalueza, A.; et al. COVID-19 in Solid Organ Transplant Recipients: A Single-center Case Series from Spain. Am. J. Transplant. 2020, 20, 1849–1858. [Google Scholar] [CrossRef]
- Zhu, L.; Gong, N.; Liu, B.; Lu, X.; Chen, D.; Chen, S.; Shu, H.; Ma, K.; Xu, X.; Guo, Z.; et al. Coronavirus Disease 2019 Pneumonia in Immunosuppressed Renal Transplant Recipients: A Summary of 10 Confirmed Cases in Wuhan, China. Eur. Urol. 2020, 77, 748–754. [Google Scholar] [CrossRef] [PubMed]
- Cavagna, L.; Bruno, R.; Zanframundo, G.; Gregorini, M.; Seminari, E.; Di Matteo, A.; Rampino, T.; Montecucco, C.; Pelenghi, S.; Cattadori, B.; et al. Clinical Presentation and Evolution of COVID-19 in Immunosuppressed Patients. Preliminary Evaluation in a North Italian Cohort on Calcineurin-Inhibitors Based Therapy. medRxiv 2020. [Google Scholar] [CrossRef]
- Fung, M.; Chiu, C.Y.; DeVoe, C.; Doernberg, S.B.; Schwartz, B.S.; Langelier, C.; Henrich, T.J.; Yokoe, D.; Davis, J.; Hays, S.R.; et al. Clinical Outcomes and Serologic Response in Solid Organ Transplant Recipients with COVID-19: A Case Series from the United States. Am. J. Transplant. 2020, 20, 3225–3233. [Google Scholar] [CrossRef]
- Abrishami, A.; Samavat, S.; Behnam, B.; Arab-Ahmadi, M.; Nafar, M.; Sanei Taheri, M. Clinical Course, Imaging Features, and Outcomes of COVID-19 in Kidney Transplant Recipients. Eur. Urol. 2020, 78, 281–286. [Google Scholar] [CrossRef]
- Hoek, R.A.S.; Manintveld, O.C.; Betjes, M.G.H.; Hellemons, M.E.; Seghers, L.; Van Kampen, J.A.A.; Caliskan, K.; Wetering, J.; Hoogen, M.; Metselaar, H.J.; et al. COVID-19 in Solid Organ Transplant Recipients: A Single-center Experience. Transpl. Int. 2020, 33, 1099–1105. [Google Scholar] [CrossRef] [PubMed]
- Mella, A.; Mingozzi, S.; Gallo, E.; Lavacca, A.; Rossetti, M.; Clari, R.; Randone, O.; Maffei, S.; Salomone, M.; Imperiale, D.; et al. Case Series of Six Kidney Transplanted Patients with COVID-19 Pneumonia Treated with Tocilizumab. Transpl. Infect. Dis. 2020, 22, e13348. [Google Scholar] [CrossRef] [PubMed]
- Montagud-Marrahi, E.; Cofan, F.; Torregrosa, J.; Cucchiari, D.; Ventura-Aguiar, P.; Revuelta, I.; Bodro, M.; Piñeiro, G.J.; Esforzado, N.; Ugalde, J.; et al. Preliminary Data on Outcomes of SARS-CoV-2 Infection in a Spanish Single Center Cohort of Kidney Recipients. Am. J. Transplant. 2020, 20, 2958–2959. [Google Scholar] [CrossRef]
- Banerjee, D.; Popoola, J.; Shah, S.; Ster, I.C.; Quan, V.; Phanish, M. COVID-19 Infection in Kidney Transplant Recipients. Kidney Int. 2020, 97, 1076–1082. [Google Scholar] [CrossRef]
- Malberti, F.; Pecchini, P.; Marchi, G.; Foramitti, M. When a Nephrology Ward Becomes a COVID-19 Ward: The Cremona Experience. J. Nephrol. 2020, 33, 625–628. [Google Scholar] [CrossRef] [PubMed]
- Lubetzky, M.; Aull, M.; Craig-Shapiro, R.; Lee, J.; Lee, J.; Sultan, S.; Marku-Podvorica, J.; Gingras, L.; Kodiyanplakkal, R.P.; Hartono, C.; et al. Kidney Allograft Recipients Diagnosed with Coronavirus Disease-2019: A Single Center Report. medRxiv 2020. [Google Scholar] [CrossRef]
- Devresse, A.; Belkhir, L.; Vo, B.; Ghaye, B.; Scohy, A.; Kabamba, B.; Goffin, E.; De Greef, J.; Mourad, M.; De Meyer, M.; et al. COVID-19 Infection in Kidney Transplant Recipients: A Single-Center Case Series of 22 Cases From Belgium. Kidney Med. 2020, 2, 459–466. [Google Scholar] [CrossRef] [PubMed]
- Crespo, M.; Pérez-Sáez, M.J.; Redondo-Pachón, D.; Llinàs-Mallol, L.; Montero, M.M.; Villar-García, J.; Arias-Cabrales, C.; Buxeda, A.; Burballa, C.; Vázquez, S.; et al. COVID-19 in Elderly Kidney Transplant Recipients. Am. J. Transplant. 2020, 20, 2883–2889. [Google Scholar] [CrossRef] [PubMed]
- Favà, A.; Montero, N.; Cucchiari, D.; Toapanta, N.; Centellas, J.; Vila-Santandreu, A.; Coloma, A.; Meneghini, M.; Manonelles, A.; Sellarès, J.; et al. SARS-CoV-2 in Kidney Transplant Recipients: A Multicentric Prospective Cohort Study. SSRN Electron. J. 2020. Available online: https://ssrn.com/abstract=3594582 (accessed on 10 August 2021). [CrossRef]
- Zhang, H.; Chen, Y.; Yuan, Q.; Xia, Q.-X.; Zeng, X.-P.; Peng, J.-T.; Liu, J.; Xiao, X.-Y.; Jiang, G.-S.; Xiao, H.-Y.; et al. Identification of Kidney Transplant Recipients with Coronavirus Disease 2019. Eur. Urol. 2020, 77, 742–747. [Google Scholar] [CrossRef]
- Cravedi, P.; Mothi, S.S.; Azzi, Y.; Haverly, M.; Farouk, S.S.; Pérez-Sáez, M.J.; Redondo-Pachón, M.D.; Murphy, B.; Florman, S.; Cyrino, L.G.; et al. COVID-19 and Kidney Transplantation: Results from the TANGO International Transplant Consortium. Am. J. Transplant. 2020, 20, 3140–3148. [Google Scholar] [CrossRef]
- Maritati, F.; Cerutti, E.; Zuccatosta, L.; Fiorentini, A.; Finale, C.; Ficosecco, M.; Cristiano, F.; Capestro, A.; Balestra, E.; Taruscia, D.; et al. SARS-CoV-2 Infection in Kidney Transplant Recipients: Experience of the Italian Marche Region. Transpl. Infect. Dis. 2020, 22, e13377. [Google Scholar] [CrossRef]
- Elias, M.; Pievani, D.; Randoux, C.; Louis, K.; Denis, B.; Delion, A.; Le Goff, O.; Antoine, C.; Greze, C.; Pillebout, E.; et al. COVID-19 Infection in Kidney Transplant Recipients: Disease Incidence and Clinical Outcomes. J. Am. Soc. Nephrol. 2020, 31, 2413–2423. [Google Scholar] [CrossRef]
- Chen, T.Y.; Farghaly, S.; Cham, S.; Tatem, L.L.; Sin, J.H.; Rauda, R.; Ribisi, M.; Sumrani, N. COVID-19 Pneumonia in Kidney Transplant Recipients: Focus on Immunosuppression Management. Transpl. Infect. Dis. 2020, 22, e13378. [Google Scholar] [CrossRef]
- Mehta, S.A.; Leonard, J.; Labella, P.; Cartiera, K.; Soomro, I.; Neumann, H.; Montgomery, R.A.; Ali, N.M. Outpatient Management of Kidney Transplant Recipients with Suspected COVID-19—Single-center Experience during the New York City Surge. Transpl. Infect. Dis. 2020, 22, e13383. [Google Scholar] [CrossRef]
- Silva, F.; Cipriano, A.; Cruz, H.; Tavares, J.; Fragoso, J.; Malheiro, J.; Almeida, M.; Martins, L.S.; Abreu, M.; Pedroso, S.; et al. SARS-CoV-2 Infection in Kidney Transplant Recipients: Early Report of Five Cases. Transpl. Infect. Dis. 2021, 23, e13394. [Google Scholar] [CrossRef]
- Chavarot, N.; Gueguen, J.; Bonnet, G.; Jdidou, M.; Trimaille, A.; Burger, C.; Amrouche, L.; Weizman, O.; Pommier, T.; Aubert, O.; et al. COVID-19 Severity in Kidney Transplant Recipients Is Similar to Nontransplant Patients with Similar Comorbidities. Am. J. Transplant. 2021, 21, 1285–1294. [Google Scholar] [CrossRef]
- De Meester, J.; De Bacquer, D.; Naesens, M.; Meijers, B.; Couttenye, M.M.; De Vriese, A.S. Incidence, Characteristics, and Outcome of COVID-19 in Adults on Kidney Replacement Therapy: A Regionwide Registry Study. J. Am. Soc. Nephrol. 2021, 32, 385–396. [Google Scholar] [CrossRef]
- Abolghasemi, S.; Mardani, M.; Sali, S.; Honarvar, N.; Baziboroun, M. COVID-19 and Kidney Transplant Recipients. Transpl. Infect. Dis. 2020, 22, e13413. [Google Scholar] [CrossRef]
- Cucchiari, D.; Guillén, E.; Cofan, F.; Torregrosa, J.; Esforzado, N.; Revuelta, I.; Ventura-Aguiar, P.; Oppenheimer, F.; Bayés, B.; Marcos, M.Á.; et al. Taking Care of Kidney Transplant Recipients during the COVID-19 Pandemic: Experience from a Medicalized Hotel. Clin. Transplant. 2021, 35, e14132. [Google Scholar] [CrossRef] [PubMed]
- Kute, V.B.; Bhalla, A.K.; Guleria, S.; Ray, D.S.; Bahadur, M.M.; Shingare, A.; Hegde, U.; Gang, S.; Raju, S.; Patel, H.V.; et al. Clinical Profile and Outcome of COVID-19 in 250 Kidney Transplant Recipients: A Multicenter Cohort Study From India. Transplantation 2021, 105, 851–860. [Google Scholar] [CrossRef] [PubMed]
- Caillard, S.; Anglicheau, D.; Matignon, M.; Durrbach, A.; Greze, C.; Frimat, L.; Thaunat, O.; Legris, T.; Moal, V.; Westeel, P.F.; et al. An Initial Report from the French SOT COVID Registry Suggests High Mortality Due to COVID-19 in Recipients of Kidney Transplants. Kidney Int. 2020, 98, 1549–1558. [Google Scholar] [CrossRef] [PubMed]
- Kutzler, H.L.; Poulos, C.M.; Cheema, F.; O’Sullivan, D.M.; Ali, A.; Ebcioglu, Z.; Einstein, M.; Feingold, A.D.; Gluck, J.; Hammond, J.A.; et al. COVID-19 in Solid Organ Transplant Recipients: Observations From Connecticut. Transplantation 2021, 105, e6–e8. [Google Scholar] [CrossRef] [PubMed]
- Miarons, M.; Larrosa-García, M.; García-García, S.; Los-Arcos, I.; Moreso, F.; Berastegui, C.; Castells, L.; Pérez-Hoyos, S.; Varela, J.; Pau-Parra, A.; et al. COVID-19 in Solid Organ Transplantation: A Matched Retrospective Cohort Study and Evaluation of Immunosuppression Management. Transplantation 2021, 105, 138–150. [Google Scholar] [CrossRef]
- Sharma, P.; Chen, V.; Fung, C.M.; Troost, J.P.; Patel, V.N.; Combs, M.; Norman, S.; Garg, P.; Colvin, M.; Aaronson, K.; et al. COVID-19 Outcomes Among Solid Organ Transplant Recipients: A Case-Control Study. Transplantation 2021, 105, 128–137. [Google Scholar] [CrossRef]
- Mohamed, I.H.; Chowdary, P.B.; Shetty, S.; Sammartino, C.; Sivaprakasam, R.; Lindsey, B.; Thuraisingham, R.; Yaqoob, M.M.; Khurram, M.A. Outcomes of Renal Transplant Recipients with SARS-CoV-2 Infection in the Eye of the Storm: A Comparative Study with Waitlisted Patients. Transplantation 2021, 105, 115–120. [Google Scholar] [CrossRef]
- Felldin, M.; Søfteland, J.M.; Magnusson, J.; Ekberg, J.; Karason, K.; Schult, A.; Larsson, H.; Oltean, M.; Friman, V. Initial Report From a Swedish High-Volume Transplant Center After the First Wave of the COVID-19 Pandemic. Transplantation 2021, 105, 108–114. [Google Scholar] [CrossRef]
- Kates, O.S.; Haydel, B.M.; Florman, S.S.; Rana, M.M.; Chaudhry, Z.S.; Ramesh, M.S.; Safa, K.; Kotton, C.N.; Blumberg, E.A.; Besharatian, B.D.; et al. Coronavirus Disease 2019 in Solid Organ Transplant: A Multicenter Cohort Study. Clin. Infect. Dis. 2020, ciaa1097. [Google Scholar] [CrossRef] [PubMed]
- Benotmane, I.; Gautier-Vargas, G.; Wendling, M.; Perrin, P.; Velay, A.; Bassand, X.; Bedo, D.; Baldacini, C.; Sagnard, M.; Bozman, D.; et al. In-depth Virological Assessment of Kidney Transplant Recipients with COVID-19. Am. J. Transplant. 2020, 20, 3162–3172. [Google Scholar] [CrossRef] [PubMed]
- Chaudhry, Z.S.; Williams, J.D.; Vahia, A.; Fadel, R.; Parraga Acosta, T.; Prashar, R.; Shrivastava, P.; Khoury, N.; Pinto Corrales, J.; Williams, C.; et al. Clinical Characteristics and Outcomes of COVID-19 in Solid Organ Transplant Recipients: A Cohort Study. Am. J. Transplant. 2020, 20, 3051–3060. [Google Scholar] [CrossRef] [PubMed]
- Craig-Schapiro, R.; Salinas, T.; Lubetzky, M.; Abel, B.T.; Sultan, S.; Lee, J.R.; Kapur, S.; Aull, M.J.; Dadhania, D.M. COVID-19 Outcomes in Patients Waitlisted for Kidney Transplantation and Kidney Transplant Recipients. Am. J. Transplant. 2021, 21, 1576–1585. [Google Scholar] [CrossRef]
- Demir, E.; Uyar, M.; Parmaksiz, E.; Sinangil, A.; Yelken, B.; Dirim, A.B.; Merhametsiz, O.; Yadigar, S.; Atan Ucar, Z.; Ucar, A.R.; et al. COVID-19 in Kidney Transplant Recipients: A Multicenter Experience in Istanbul. Transpl. Infect. Dis. 2020, 22, e13371. [Google Scholar] [CrossRef] [PubMed]
- Hilbrands, L.B.; Duivenvoorden, R.; Vart, P.; Franssen, C.F.M.; Hemmelder, M.H.; Jager, K.J.; Kieneker, L.M.; Noordzij, M.; Pena, M.J.; de Vries, H.; et al. COVID-19-Related Mortality in Kidney Transplant and Dialysis Patients: Results of the ERACODA Collaboration. Nephrol. Dial. Transplant. 2020, 35, 1973–1983. [Google Scholar] [CrossRef] [PubMed]
- Pascual, J.; Melilli, E.; Jiménez-Martín, C.; González-Monte, E.; Zárraga, S.; Gutiérrez-Dalmau, A.; López-Jiménez, V.; Juega, J.; Muñoz-Cepeda, M.; Lorenzo, I.; et al. COVID-19–Related Mortality During the First 60 Days After Kidney Transplantation. Eur. Urol. 2020, 78, 641–643. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Cubillo, B.; Higuera, M.A.M.; Lucena, R.; Franci, E.V.; Hurtado, M.; Romero, N.C.; Moreno, A.R.; Valencia, D.; Velo, M.; Fornie, I.S.; et al. Should Cyclosporine Be Useful in Renal Transplant Recipients Affected by SARS-CoV-2? Am. J. Transplant. 2020, 20, 3173–3181. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Alvarez, E.; Macía, M.; de Sequera Ortiz, P. Management of Hemodialysis Patients with Suspected or Confirmed COVID-19 Infection: Perspective from the Spanish Nephrology. Kidney360 2020, 1, 1254–1258. [Google Scholar] [CrossRef]
- Oto, O.A.; Ozturk, S.; Turgutalp, K.; Arici, M.; Alpay, N.; Merhametsiz, O.; Sipahi, S.; Ogutmen, M.B.; Yelken, B.; Altiparmak, M.R.; et al. Predicting the Outcome of COVID-19 Infection in Kidney Transplant Recipients. BMC Nephrol. 2021, 22, 100. [Google Scholar] [CrossRef] [PubMed]
- Elhadedy, M.A.; Marie, Y.; Halawa, A. COVID-19 in Renal Transplant Recipients: Case Series and a Brief Review of Current Evidence. Nephron 2020, 145, 192–198. [Google Scholar] [CrossRef]
- Zeng, F.; Huang, Y.; Guo, Y.; Yin, M.; Chen, X.; Xiao, L.; Deng, G. Association of Inflammatory Markers with the Severity of COVID-19: A Meta-Analysis. Int. J. Infect. Dis. 2020, 96, 467–474. [Google Scholar] [CrossRef]
- Schnuelle, P.; Lorenz, D.; Trede, M.; Van Der Woude, F.J. Impact of Renal Cadaveric Transplantation on Survival in End-Stage Renal Failure: Evidence for Reduced Mortality Risk Compared with Hemodialysis during Long-Term Follow-Up. J. Am. Soc. Nephrol. 1998, 9, 2135–2141. [Google Scholar] [CrossRef]
- Port, F.K.; Wolfe, R.A.; Mauger, E.A.; Berling, D.P.; Jiang, K. Comparison of Survival Probabilities for Dialysis Patients vs Cadaveric Renal Transplant Recipients. JAMA: J. Am. Med Assoc. 1993, 270, 1339–1343. [Google Scholar] [CrossRef]
- Ojo, A.O.; Port, F.K.; Wolfe, R.A.; Mauger, E.A.; Williams, L.; Berling, D.P. Comparative Mortality Risks of Chronic Dialysis and Cadaveric Transplantation in Black End-Stage Renal Disease Patients. Am. J. Kidney Dis. 1994, 24, 59–64. [Google Scholar] [CrossRef]
- Su, H.; Yang, M.; Wan, C.; Yi, L.-X.; Tang, F.; Zhu, H.-Y.; Yi, F.; Yang, H.-C.; Fogo, A.B.; Nie, X.; et al. Renal Histopathological Analysis of 26 Postmortem Findings of Patients with COVID-19 in China. Kidney Int. 2020, 98, 219–227. [Google Scholar] [CrossRef]
- Larsen, C.P.; Bourne, T.D.; Wilson, J.D.; Saqqa, O.; Sharshir, M.A. Collapsing Glomerulopathy in a Patient with Coronavirus Disease 2019 (COVID-19). Kidney Int. Rep. 2020, 5, 935. [Google Scholar] [CrossRef] [PubMed]
- Varga, Z.; Flammer, A.J.; Steiger, P.; Haberecker, M.; Andermatt, R.; Zinkernagel, A.S.; Mehra, M.R.; Schuepbach, R.A.; Ruschitzka, F.; Moch, H. Endothelial Cell Infection and Endotheliitis in COVID-19. Lancet 2020, 395, 1417–1418. [Google Scholar] [CrossRef]
- Ronco, C.; Reis, T. Kidney Involvement in COVID-19 and Rationale for Extracorporeal Therapies. Nat. Rev. Nephrol. 2020, 16, 308–310. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Xiao, M.; Zhang, S.; Xia, P.; Cao, W.; Jiang, W.; Chen, H.; Ding, X.; Zhao, H.; Zhang, H.; et al. Coagulopathy and Antiphospholipid Antibodies in Patients with COVID -19. N. Engl. J. Med. 2020, 382, e38. [Google Scholar] [CrossRef] [PubMed]
- Report of the WHO-China Joint Mission on Coronavirus Disease 2019 (COVID-19). 16–24 February 2020. Available online: https://www.who.int/docs/default-source/coronaviruse/who-china-joint-mission-on-covid-19-final-report.pdf (accessed on 10 August 2021).
- Yang, J.; Zheng, Y.; Gou, X.; Pu, K.; Chen, Z.; Guo, Q.; Ji, R.; Wang, H.; Wang, Y.; Zhou, Y. Prevalence of Comorbidities and Its Effects in Patients Infected with SARS-CoV-2: A Systematic Review and Meta-Analysis. Int. J. Infect. Dis. 2020, 94, 91–95. [Google Scholar] [CrossRef]
- Chen, T.; Wu, D.; Chen, H.; Yan, W.; Yang, D.; Chen, G.; Ma, K.; Xu, D.; Yu, H.; Wang, H.; et al. Clinical Characteristics of 113 Deceased Patients with Coronavirus Disease 2019: Retrospective Study. BMJ 2020, 368, m1091. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Xu, S.; Yu, M.; Wang, K.; Tao, Y.; Zhou, Y.; Shi, J.; Zhou, M.; Wu, B.; Yang, Z.; et al. Risk Factors for Severity and Mortality in Adult COVID-19 Inpatients in Wuhan. J. Allergy Clin. Immunol. 2020, 146, 110–118. [Google Scholar] [CrossRef]
- Guan, W.J.; Liang, W.H.; Zhao, Y.; Liang, H.R.; Chen, Z.S.; Li, Y.M.; Liu, X.Q.; Chen, R.C.; Tang, C.L.; Wang, T.; et al. Comorbidity and Its Impact on 1,590 Patients with COVID -19 in China: A Nationwide Analysis. Eur. Respir. J. 2020, 55, 200054. [Google Scholar] [CrossRef] [Green Version]
- Robbins-Juarez, S.Y.; Qian, L.; King, K.L.; Stevens, J.S.; Husain, S.A.; Radhakrishnan, J.; Mohan, S. Outcomes for Patients with COVID-19 and Acute Kidney Injury: A Systematic Review and Meta-Analysis. Kidney Int. Rep. 2020, 5, 1149–1160. [Google Scholar] [CrossRef]
- Pereira, M.R.; Mohan, S.; Cohen, D.J.; Husain, S.A.; Dube, G.K.; Ratner, L.E.; Arcasoy, S.; Aversa, M.M.; Benvenuto, L.J.; Dadhania, D.M.; et al. COVID-19 in Solid Organ Transplant Recipients: Initial Report from the US Epicenter. Am. J. Transplant. 2020, 20, 1800–1808. [Google Scholar] [CrossRef]
- Azzi, Y.; Parides, M.; Alani, O.; Loarte-Campos, P.; Bartash, R.; Forest, S.; Colovai, A.; Ajaimy, M.; Liriano-Ward, L.; Pynadath, C.; et al. COVID-19 Infection in Kidney Transplant Recipients at the Epicenter of Pandemics. Kidney Int. 2020, 98, 1559–1567. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Yu, Y.; Xu, J.; Shu, H.; Xia, J.; Liu, H.; Wu, Y.; Zhang, L.; Yu, Z.; Fang, M.; et al. Clinical Course and Outcomes of Critically Ill Patients with SARS-CoV-2 Pneumonia in Wuhan, China: A Single-Centered, Retrospective, Observational Study. Lancet Respir. Med. 2020, 8, 475–481. [Google Scholar] [CrossRef] [Green Version]
- Wu, C.; Chen, X.; Cai, Y.; Xia, J.; Zhou, X.; Xu, S.; Huang, H.; Zhang, L.; Zhou, X.; Du, C.; et al. Risk Factors Associated with Acute Respiratory Distress Syndrome and Death in Patients with Coronavirus Disease 2019 Pneumonia in Wuhan, China. JAMA Intern. Med. 2020, 180, 934. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arentz, M.; Yim, E.; Klaff, L.; Lokhandwala, S.; Riedo, F.X.; Chong, M.; Lee, M. Characteristics and Outcomes of 21 Critically Ill Patients with COVID-19 in Washington State. JAMA 2020, 323, 1612. [Google Scholar] [CrossRef] [Green Version]
- Grasselli, G.; Greco, M.; Zanella, A.; Albano, G.; Antonelli, M.; Bellani, G.; Bonanomi, E.; Cabrini, L.; Carlesso, E.; Castelli, G.; et al. Risk Factors Associated with Mortality Among Patients with COVID-19 in Intensive Care Units in Lombardy, Italy. JAMA Intern. Med. 2020, 180, 1345. [Google Scholar] [CrossRef]
- Williamson, E.J.; Walker, A.J.; Bhaskaran, K.; Bacon, S.; Bates, C.; Morton, C.E.; Curtis, H.J.; Mehrkar, A.; Evans, D.; Inglesby, P.; et al. Factors Associated with COVID-19-Related Death Using OpenSAFELY. Nature 2020, 584, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, R.A.; Kane, A.D.; Cook, T.M. Outcomes from Intensive Care in Patients with COVID-19: A Systematic Review and Meta-Analysis of Observational Studies. Anaesthesia 2020, 75, 1340–1349. [Google Scholar] [CrossRef]
- Sánchez-Álvarez, J.E.; Fontán, M.P.; Martín, C.J.; Pelícano, M.B.; Reina, C.J.C.; Prieto, Á.M.S.; Melilli, E.; Barrios, M.C.; Heras, M.M.; y Pino, M.D. Status of SARS-CoV-2 Infection in Patients on Renal Replacement Therapy. Report of the COVID-19 Registry of the Spanish Society of Nephrology (SEN). Nefrología 2020, 40, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Felsenstein, S.; Herbert, J.A.; McNamara, P.S.; Hedrich, C.M. COVID-19: Immunology and Treatment Options. Clin. Immunol. 2020, 215, 108448. [Google Scholar] [CrossRef]
- Carsetti, R.; Zaffina, S.; Piano Mortari, E.; Terreri, S.; Corrente, F.; Capponi, C.; Palomba, P.; Mirabella, M.; Cascioli, S.; Palange, P.; et al. Different Innate and Adaptive Immune Responses to SARS-CoV-2 Infection of Asymptomatic, Mild, and Severe Cases. Front. Immunol. 2020, 11, 3365. [Google Scholar] [CrossRef] [PubMed]
- Mathew, D.; Giles, J.R.; Baxter, A.E.; Oldridge, D.A.; Greenplate, A.R.; Wu, J.E.; Alanio, C.; Kuri-Cervantes, L.; Pampena, M.B.; D’Andrea, K.; et al. Deep Immune Profiling of COVID-19 Patients Reveals Distinct Immunotypes with Therapeutic Implications. Science 2020, 369, eabc8511. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Nie, J.; Wang, H.; Zhao, Q.; Xiong, Y.; Deng, L.; Song, S.; Ma, Z.; Mo, P.; Zhang, Y. Characteristics of Peripheral Lymphocyte Subset Alteration in COVID-19 Pneumonia. J. Infect. Dis. 2020, 221, 1762–1769. [Google Scholar] [CrossRef] [Green Version]
- Thevarajan, I.; Nguyen, T.H.O.; Koutsakos, M.; Druce, J.; Caly, L.; van de Sandt, C.E.; Jia, X.; Nicholson, S.; Catton, M.; Cowie, B.; et al. Breadth of Concomitant Immune Responses Prior to Patient Recovery: A Case Report of Non-Severe COVID-19. Nat. Med. 2020, 26, 453–455. [Google Scholar] [CrossRef] [Green Version]
- Johnston, O.; Jaswal, D.; Gill, J.S.; Doucette, S.; Fergusson, D.A.; Knoll, G.A. Treatment of Polyomavirus Infection in Kidney Transplant Recipients: A Systematic Review. Transplantation 2010, 89, 1057–1070. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saad, E.R.; Bresnahan, B.A.; Cohen, E.P.; Lu, N.; Orentas, R.J.; Vasudev, B.; Hariharan, S. Successful Treatment of BK Viremia Using Reduction in Immunosuppression without Antiviral Therapy. Transplantation 2008, 85, 850–854. [Google Scholar] [CrossRef] [PubMed]
- de Mezerville, M.H.; Tellier, R.; Richardson, S.; Hébert, D.; Doyle, J.; Allen, U. Adenoviral Infections in Pediatric Transplant Recipients. Pediatr. Infect. Dis. J. 2006, 25, 815–818. [Google Scholar] [CrossRef]
- Watcharananan, S.P.; Avery, R.; Ingsathit, A.; Malathum, K.; Chantratita, W.; Mavichak, V.; Chalermsanyakorn, P.; Jirasiritham, S.; Sumethkul, V. Adenovirus Disease after Kidney Transplantation: Course of Infection and Outcome in Relation to Blood Viral Load and Immune Recovery. Am. J. Transplant. 2011, 11, 1308–1314. [Google Scholar] [CrossRef]
- Mehta, P.; McAuley, D.F.; Brown, M.; Sanchez, E.; Tattersall, R.S.; Manson, J.J. COVID-19: Consider Cytokine Storm Syndromes and Immunosuppression. Lancet 2020, 395, 1033–1034. [Google Scholar] [CrossRef]
- Ritchie, A.I.; Singanayagam, A. Immunosuppression for Hyperinflammation in COVID-19: A Double-Edged Sword? Lancet 2020, 395, 1111. [Google Scholar] [CrossRef]
- Ye, Z.; Wang, Y.; Colunga-Lozano, L.E.; Prasad, M.; Tangamornsuksan, W.; Rochwerg, B.; Yao, L.; Motaghi, S.; Couban, R.J.; Ghadimi, M.; et al. Efficacy and Safety of Corticosteroids in COVID-19 Based on Evidence for COVID-19, Other Coronavirus Infections, Influenza, Community-Acquired Pneumonia and Acute Respiratory Distress Syndrome: A Systematic Review and Meta-Analysis. Can. Med. Assoc. J. 2020, 192, E756–E767. [Google Scholar] [CrossRef]
- Horby, P.; Lim, W.S.; Emberson, J.R.; Mafham, M.; Bell, J.L.; Linsell, L.; Staplin, N.; Brightling, C.; Ustianowski, A.; Elmahi, E.; et al. Dexamethasone in Hospitalized Patients with COVID -19—Preliminary Report. N. Engl. J. Med. 2021, 384, 693–704. [Google Scholar] [CrossRef] [PubMed]






Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jayant, K.; Reccia, I.; Bachul, P.J.; Al-Salmay, Y.; Pyda, J.S.; Podda, M.; Perez-Gutierrez, A.; Dor, F.J.M.F.; Becker, Y.; di Sabato, D.; et al. The Impact of COVID-19 on Kidney Transplant Recipients in Pre-Vaccination and Delta Strain Era: A Systematic Review and Meta-Analysis. J. Clin. Med. 2021, 10, 4533. https://doi.org/10.3390/jcm10194533
Jayant K, Reccia I, Bachul PJ, Al-Salmay Y, Pyda JS, Podda M, Perez-Gutierrez A, Dor FJMF, Becker Y, di Sabato D, et al. The Impact of COVID-19 on Kidney Transplant Recipients in Pre-Vaccination and Delta Strain Era: A Systematic Review and Meta-Analysis. Journal of Clinical Medicine. 2021; 10(19):4533. https://doi.org/10.3390/jcm10194533
Chicago/Turabian StyleJayant, Kumar, Isabella Reccia, Piotr J. Bachul, Yaser Al-Salmay, Jordan S. Pyda, Mauro Podda, Angelica Perez-Gutierrez, Frank J. M. F. Dor, Yolanda Becker, Diego di Sabato, and et al. 2021. "The Impact of COVID-19 on Kidney Transplant Recipients in Pre-Vaccination and Delta Strain Era: A Systematic Review and Meta-Analysis" Journal of Clinical Medicine 10, no. 19: 4533. https://doi.org/10.3390/jcm10194533
APA StyleJayant, K., Reccia, I., Bachul, P. J., Al-Salmay, Y., Pyda, J. S., Podda, M., Perez-Gutierrez, A., Dor, F. J. M. F., Becker, Y., di Sabato, D., LaMattina, J., Barth, R., Fung, J., & Witkowski, P. (2021). The Impact of COVID-19 on Kidney Transplant Recipients in Pre-Vaccination and Delta Strain Era: A Systematic Review and Meta-Analysis. Journal of Clinical Medicine, 10(19), 4533. https://doi.org/10.3390/jcm10194533









