Increased Risks of Death and Hospitalization in Influenza/Pneumonia and Sepsis for Individuals Affected by Psychotic Disorders, Bipolar Disorders, and Single Manic Episodes: A Retrospective Cross-Sectional Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Data Sources
2.3. Study Population
2.4. Variable Definitions
2.4.1. Outcomes
2.4.2. Exposures
2.5. Statistical Methods
3. Results
3.1. Baseline Characteristics
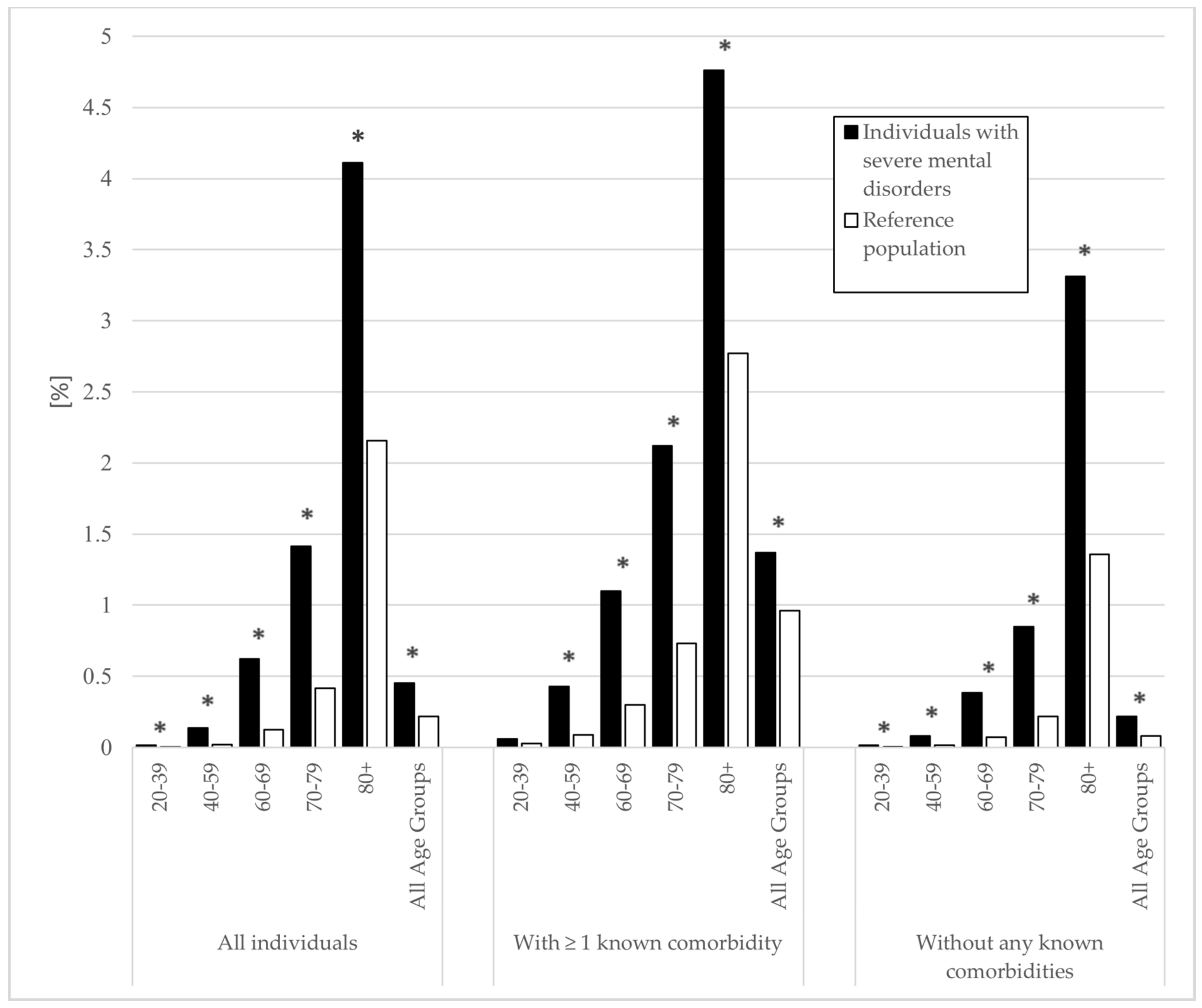
3.2. Death and Hospitalization Associated with Influenza/Pneumonia
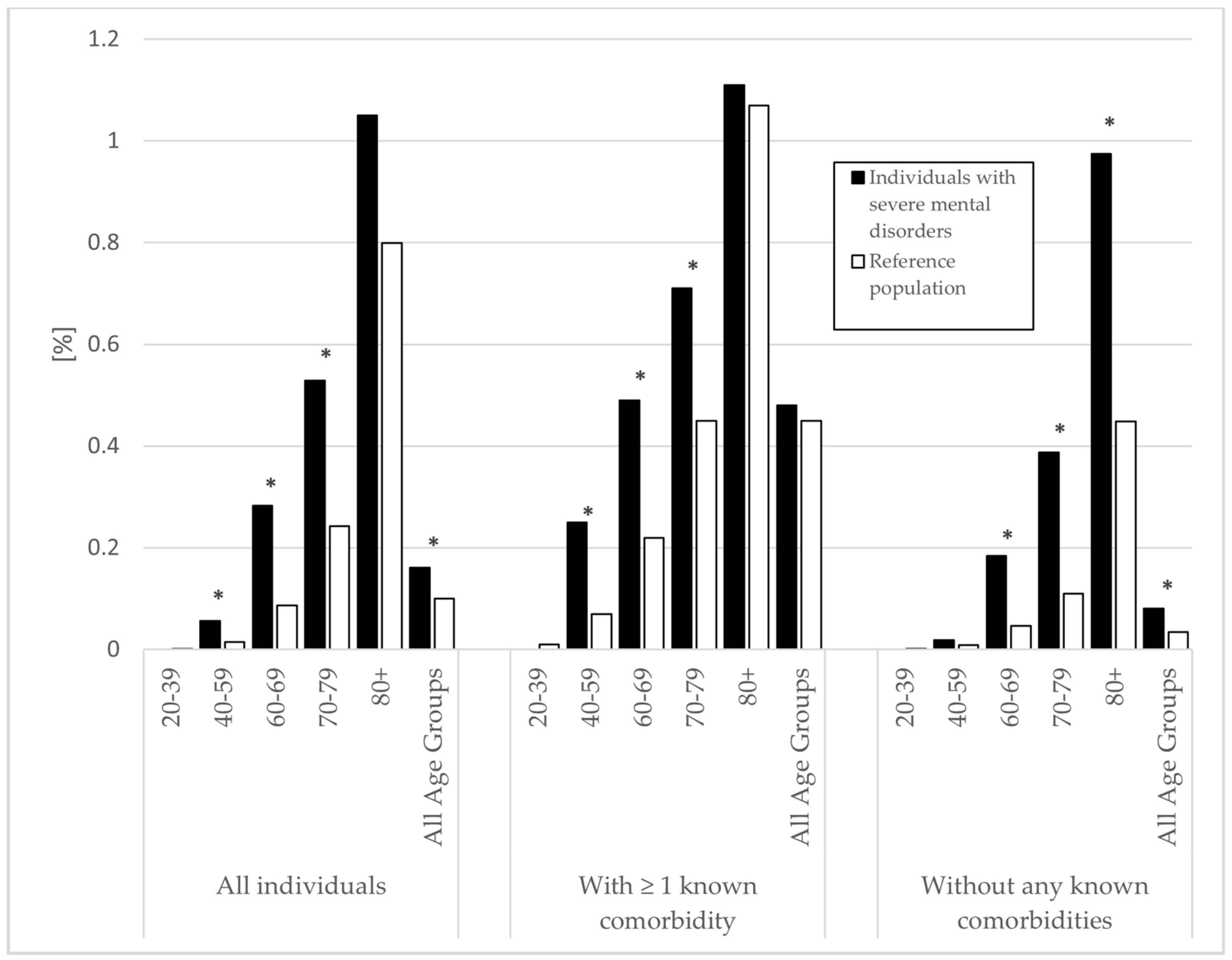
3.3. Death and Hospitalization Associated with Sepsis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Perälä, J.; Suvisaari, J.; Saarni, S.I.; Kuoppasalmi, K.; Isometsä, E.; Pirkola, S.; Partonen, T.; Tuulio-Henriksson, A.; Hintikka, J.; Kieseppä, T.; et al. Lifetime prevalence of psychotic and bipolar I disorders in a general population. Arch. Gen. Psychiatry 2007, 64, 19–28. [Google Scholar] [CrossRef] [Green Version]
- Maripuu, M.; Bendix, M.; Öhlund, L.; Widerström, M.; Werneke, U. Death Associated With Coronavirus (COVID-19) Infection in Individuals With Severe Mental Disorders in Sweden During the Early Months of the Outbreak-An Exploratory Cross-Sectional Analysis of a Population-Based Register Study. Front Psychiatry 2020, 11, 609579. [Google Scholar] [CrossRef]
- Nordentoft, M.; Wahlbeck, K.; Hällgren, J.; Westman, J.; Osby, U.; Alinaghizadeh, H.; Gissler, M.; Laursen, T.M. Excess mortality, causes of death and life expectancy in 270,770 patients with recent onset of mental disorders in Denmark, Finland and Sweden. PLoS ONE 2013, 8, e55176. [Google Scholar]
- Laursen, T.M. Life expectancy among persons with schizophrenia or bipolar affective disorder. Schizophr. Res. 2011, 131, 101–104. [Google Scholar] [CrossRef] [PubMed]
- Crump, C.; Sundquist, K.; Winkleby, M.A.; Sundquist, J. Comorbidities and mortality in bipolar disorder: A Swedish national cohort study. JAMA Psychiatry 2013, 70, 931–939. [Google Scholar] [CrossRef]
- Crump, C.; Winkleby, M.A.; Sundquist, K.; Sundquist, J. Comorbidities and mortality in persons with schizophrenia: A Swedish national cohort study. Am. J. Psychiatry 2013, 170, 324–333. [Google Scholar] [CrossRef]
- Jayatilleke, N.; Hayes, R.D.; Dutta, R.; Shetty, H.; Hotopf, M.; Chang, C.K.; Stewart, R. Contributions of specific causes of death to lost life expectancy in severe mental illness. Eur. Psychiatry 2017, 43, 109–115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Birkenaes, A.B.; Opjordsmoen, S.; Brunborg, C.; Engh, J.A.; Jonsdottir, H.; Ringen, P.A.; Simonsen, C.; Vaskinn, A.; Birkeland, K.I.; Friis, S.; et al. The level of cardiovascular risk factors in bipolar disorder equals that of schizophrenia: A comparative study. J. Clin. Psychiatry 2007, 68, 917–923. [Google Scholar] [CrossRef] [PubMed]
- Fagiolini, A.; Goracci, A. The effects of undertreated chronic medical illnesses in patients with severe mental disorders. J. Clin. Psychiatry 2009, 70 (Suppl. 3), 22–29. [Google Scholar] [CrossRef]
- Nielsen, R.E.; Kugathasan, P.; Straszek, S.; Jensen, S.E.; Licht, R.W. Why are somatic diseases in bipolar disorder insufficiently treated? Int. J. Bipolar. Disord. 2019, 7, 12. [Google Scholar] [CrossRef] [PubMed]
- Cradock-O’Leary, J.; Young, A.S.; Yano, E.M.; Wang, M.; Lee, M.L. Use of general medical services by VA patients with psychiatric disorders. Psychiatr. Serv. 2002, 53, 874–878. [Google Scholar] [CrossRef]
- The World Health Organization Coronavirus Disease (covid-19) Dashboard. Available online: https://covid19.who.int/ (accessed on 4 March 2021).
- Wang, Q.; Xu, R.; Volkow, N.D. Increased risk of COVID-19 infection and mortality in people with mental disorders: Analysis from electronic health records in the United States. World Psychiatry 2021, 20, 124–130. [Google Scholar] [CrossRef]
- The Public Health Agency of Sweden COVID-19: Groups at Risk of Serious Illness. Available online: https://www.folkhalsomyndigheten.se/the-public-health-agency-of-sweden/communicable-disease-control/covid-19/covid-19-and-at-risk-groups/ (accessed on 3 May 2021).
- Swedish National Board of Health and Welfare (Socialstyrelsen) Risk Att Drabbas Allvarligt Av Covid-19. Available online: https://www.socialstyrelsen.se/globalassets/sharepoint-dokument/dokument-webb/ovrigt/riskgrupper-risk-att-drabbas-allvarligt-covid19.pdf (accessed on 3 May 2021).
- Joint Committee on Vaccination and Immunisation. Department of Health and Social Security Priority Groups for Coronavirus (COVID-19) Vaccination: Advice from the JCVI. Available online: https://www.gov.uk/government/publications/priority-groups-for-coronavirus-covid-19-vaccination-advice-from-the-jcvi-30-december-2020 (accessed on 3 May 2021).
- Miller, B.J.; Paschall, C.B., 3rd; Svendsen, D.P. Mortality and medical comorbidity among patients with serious mental illness. Psychiatr. Serv. 2006, 57, 1482–1487. [Google Scholar] [CrossRef] [PubMed]
- Grohskopf, L.A.; Alyanak, E.; Broder, K.R.; Blanton, L.H.; Fry, A.M.; Jernigan, D.B.; Atmar, R.L. Prevention and Control of Seasonal Influenza with Vaccines: Recommendations of the Advisory Committee on Immunization Practices—United States, 2020–2021 Influenza Season. MMWR Recomm. Rep. 2020, 69, 1–24. [Google Scholar] [CrossRef]
- World Health Organization. WHO Regional Office for Europe Recommendations on Influenza Vaccination for the 2020/2021 Season during the Ongoing COVID-19 Pandemic; World Health Organization, Regional Office for Europe: Copenhagen, Denmark, 2020; Available online: https://apps.who.int/iris/handle/10665/335721 (accessed on 3 May 2021).
- López-Collazo, E.; Avendaño-Ortiz, J.; Martín-Quirós, A.; Aguirre, L.A. Immune Response and COVID-19: A mirror image of Sepsis. Int. J. Biol. Sci. 2020, 16, 2479–2489. [Google Scholar] [CrossRef] [PubMed]
- Ludvigsson, J.F.; Håberg, S.E.; Knudsen, G.P.; Lafolie, P.; Zoega, H.; Sarkkola, C.; von Kraemer, S.; Weiderpass, E.; Nørgaard, M. Ethical aspects of registry-based research in the Nordic countries. Clin. Epidemiol. 2015, 7, 491–508. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cuschieri, S. The STROBE guidelines. Saudi. J. Anaesth. 2019, 13 (Suppl. 1), S31–S34. [Google Scholar] [CrossRef] [PubMed]
- Ludvigsson, J.F.; Andersson, E.; Ekbom, A.; Feychting, M.; Kim, J.L.; Reuterwall, C.; Heurgren, M.; Olausson, P.O. External review and validation of the Swedish national inpatient register. BMC Public Health 2011, 11, 450. [Google Scholar] [CrossRef] [Green Version]
- World Health Organization. ICD-10, International Statistical Classification of diseases and Related Health Problems. World Health Organization: Geneva, Switzerland, 2004. [Google Scholar]
- Brooke, H.L.; Talbäck, M.; Hörnblad, J.; Johansson, L.A.; Ludvigsson, J.F.; Druid, H.; Feychting, M.; Ljung, R. The Swedish cause of death register. Eur. J. Epidemiol. 2017, 32, 765–773. [Google Scholar] [CrossRef] [PubMed]
- Wettermark, B.; Hammar, N.; Fored, C.M.; Leimanis, A.; Otterblad Olausson, P.; Bergman, U.; Persson, I.; Sundström, A.; Westerholm, B.; Rosén, M. The new Swedish Prescribed Drug Register—Opportunities for pharmacoepidemiological research and experience from the first six months. Pharmacoepidemiol. Drug Saf. 2007, 16, 726–735. [Google Scholar] [CrossRef]
- World Health Organization, Collaborating Centre for Drug Statistics Methodology. Guidelines for ATC Classification and DDD Assignment 2021; Norwegian Institute of Public Health: Oslo, Norway, 2021; pp. 23–31. [Google Scholar]
- Altman, D.G. Practical Statistics for Medical Research, 1st ed.; Chapman and Hall: London: New York, NY, USA, 1991. [Google Scholar]
- Altman, D.G.; Bland, J.M. How to obtain the P value from a confidence interval. BMJ 2011, 343, d2304. [Google Scholar] [CrossRef] [Green Version]
- John, A.; McGregor, J.; Jones, I.; Lee, S.C.; Walters, J.T.R.; Owen, M.J.; O’Donovan, M.; DelPozo-Banos, M.; Berridge, D.; Lloyd, K. Premature mortality among people with severe mental illness—New evidence from linked primary care data. Schizophr. Res. 2018, 199, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Daumit, G.L.; Pronovost, P.J.; Anthony, C.B.; Guallar, E.; Steinwachs, D.M.; Ford, D.E. Adverse events during medical and surgical hospitalizations for persons with schizophrenia. Arch. Gen. Psychiatry 2006, 63, 267–272. [Google Scholar] [CrossRef]
- Ribe, A.R.; Vestergaard, M.; Katon, W.; Charles, M.; Benros, M.E.; Vanderlip, E.; Nordentoft, M.; Laursen, T.M. Thirty-Day Mortality After Infection Among Persons With Severe Mental Illness: A Population-Based Cohort Study in Denmark. Am. J. Psychiatry 2015, 172, 776–783. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.H.; Lin, H.C. Poor clinical outcomes among pneumonia patients with schizophrenia. Schizophr. Bull. 2011, 37, 1088–1094. [Google Scholar] [CrossRef] [Green Version]
- Ieven, M.; Coenen, S.; Loens, K.; Lammens, C.; Coenjaerts, F.; Vanderstraeten, A.; Henriques-Normark, B.; Crook, D.; Huygen, K.; Butler, C.C.; et al. Aetiology of lower respiratory tract infection in adults in primary care: A prospective study in 11 European countries. Clin. Microbiol. Infect. 2018, 24, 1158–1163. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seminog, O.O.; Goldacre, M.J. Risk of pneumonia and pneumococcal disease in people with severe mental illness: English record linkage studies. Thorax 2013, 68, 171–176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rizzo, C.; Rezza, G.; Ricciardi, W. Strategies in recommending influenza vaccination in Europe and US. Hum. Vaccin. Immunother. 2018, 14, 693–698. [Google Scholar] [CrossRef] [Green Version]
- Grau, I.; Ardanuy, C.; Calatayud, L.; Schulze, M.H.; Liñares, J.; Pallares, R. Smoking and alcohol abuse are the most preventable risk factors for invasive pneumonia and other pneumococcal infections. Int. J. Infect. Dis. 2014, 25, 59–64. [Google Scholar] [CrossRef] [Green Version]
- O’Brien, J.M., Jr.; Lu, B.; Ali, N.A.; Martin, G.S.; Aberegg, S.K.; Marsh, C.B.; Lemeshow, S.; Douglas, I.S. Alcohol dependence is independently associated with sepsis, septic shock, and hospital mortality among adult intensive care unit patients. Crit. Care Med. 2007, 35, 345–350. [Google Scholar] [CrossRef]
- Greenbaum, A.; Chaves, S.S.; Perez, A.; Aragon, D.; Bandyopadhyay, A.; Bennett, N.; Fowler, B.; Hancock, E.; Lynfield, R.; McDonald-Hamm, C.; et al. Heavy alcohol use as a risk factor for severe outcomes among adults hospitalized with laboratory-confirmed influenza, 2005–2012. Infection 2014, 42, 165–170. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.; Chaves, S.S. Obesity and influenza. Clin. Infect. Dis. 2011, 53, 422–424. [Google Scholar] [CrossRef]
- Falagas, M.E.; Kompoti, M. Obesity and infection. Lancet Infect. Dis. 2006, 6, 438–446. [Google Scholar] [CrossRef]
- Coodin, S. Body mass index in persons with schizophrenia. Can. J. Psychiatry 2001, 46, 549–555. [Google Scholar] [CrossRef] [Green Version]
- McElroy, S.L.; Keck, P.E., Jr. Obesity in bipolar disorder: An overview. Curr. Psychiatry Rep. 2012, 14, 650–658. [Google Scholar] [CrossRef]
- Dickerson, F.; Stallings, C.R.; Origoni, A.E.; Vaughan, C.; Khushalani, S.; Schroeder, J.; Yolken, R.H. Cigarette smoking among persons with schizophrenia or bipolar disorder in routine clinical settings, 1999–2011. Psychiatr. Serv. 2013, 64, 44–50. [Google Scholar] [CrossRef]
- Nesvåg, R.; Knudsen, G.P.; Bakken, I.J.; Høye, A.; Ystrom, E.; Surén, P.; Reneflot, A.; Stoltenberg, C.; Reichborn-Kjennerud, T. Substance use disorders in schizophrenia, bipolar disorder, and depressive illness: A registry-based study. Soc. Psychiatry Psychiatr. Epidemiol. 2015, 50, 1267–1276. [Google Scholar] [CrossRef] [PubMed]
- Stroup, T.S.; Olfson, M.; Huang, C.; Wall, M.M.; Goldberg, T.; Devanand, D.P.; Gerhard, T. Age-Specific Prevalence and Incidence of Dementia Diagnoses Among Older US Adults With Schizophrenia. JAMA Psychiatry 2021, 78, 632–641. [Google Scholar] [CrossRef]
- Manabe, T.; Fujikura, Y.; Mizukami, K.; Akatsu, H.; Kudo, K. Pneumonia-associated death in patients with dementia: A systematic review and meta-analysis. PLoS ONE 2019, 14, e0213825. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wiemken, T.L.; Carrico, R.M.; Furmanek, S.P.; Guinn, B.E.; Mattingly, W.A.; Peyrani, P.; Ramirez, J.A. Socioeconomic Position and the Incidence, Severity, and Clinical Outcomes of Hospitalized Patients With Community-Acquired Pneumonia. Public Health Rep. 2020, 135, 364–371. [Google Scholar] [CrossRef]
- Lemay, J.A.; Ricketson, L.J.; Zwicker, L.; Kellner, J.D. Homelessness in Adults with Invasive Pneumococcal Disease (IPD) in Calgary, Canada. Open Forum Infect. Dis. 2019, 6, ofz362. [Google Scholar] [CrossRef]
- Shariatzadeh, M.R.; Huang, J.Q.; Tyrrell, G.J.; Johnson, M.M.; Marrie, T.J. Bacteremic pneumococcal pneumonia: A prospective study in Edmonton and neighboring municipalities. Medicine 2005, 84, 147–161. [Google Scholar] [CrossRef]
- Druss, B.G.; Zhao, L.; Von Esenwein, S.; Morrato, E.H.; Marcus, S.C. Understanding excess mortality in persons with mental illness: 17-year follow up of a nationally representative US survey. Med. Care 2011, 49, 599–604. [Google Scholar] [CrossRef]
- Mazereel, V.; Van Assche, K.; Detraux, J.; De Hert, M. COVID-19 vaccination for people with severe mental illness: Why, what, and how? Lancet Psychiatry 2021. [Google Scholar] [CrossRef]
- Bradford, D.W.; Kim, M.M.; Braxton, L.E.; Marx, C.E.; Butterfield, M.; Elbogen, E.B. Access to medical care among persons with psychotic and major affective disorders. Psychiatr. Serv. 2008, 59, 847–852. [Google Scholar] [CrossRef] [PubMed]
- De Hert, M.; Cohen, D.; Bobes, J.; Cetkovich-Bakmas, M.; Leucht, S.; Ndetei, D.M.; Newcomer, J.W.; Uwakwe, R.; Asai, I.; Möller, H.J.; et al. Physical illness in patients with severe mental disorders. II. Barriers to care, monitoring and treatment guidelines, plus recommendations at the system and individual level. World Psychiatry 2011, 10, 138–151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heiberg, I.H.; Jacobsen, B.K.; Balteskard, L.; Bramness, J.G.; Naess, Ø.; Ystrom, E.; Reichborn-Kjennerud, T.; Hultman, C.M.; Nesvåg, R.; Høye, A. Undiagnosed cardiovascular disease prior to cardiovascular death in individuals with severe mental illness. Acta Psychiatr. Scand. 2019, 139, 558–571. [Google Scholar] [CrossRef] [PubMed]
- Holt, R.I.; Mitchell, A.J. Diabetes mellitus and severe mental illness: Mechanisms and clinical implications. Nat. Rev. Endocrinol. 2015, 11, 79–89. [Google Scholar] [CrossRef]
- Grøn, A.O.; Dalsgaard, E.M.; Ribe, A.R.; Seidu, S.; Mora, G.; Cebrián-Cuenca, A.M.; Charles, M. Improving diabetes care among patients with severe mental illness: A systematic review of the effect of interventions. Prim. Care Diabetes 2018, 12, 289–304. [Google Scholar] [CrossRef]
- Zimmermann, U.; Kraus, T.; Himmerich, H.; Schuld, A.; Pollmächer, T. Epidemiology, implications and mechanisms underlying drug-induced weight gain in psychiatric patients. J. Psychiatr. Res. 2003, 37, 193–220. [Google Scholar] [CrossRef]
- Govind, R.; Fonseca de Freitas, D.; Pritchard, M.; Hayes, R.D.; MacCabe, J.H. Clozapine treatment and risk of COVID-19 infection: Retrospective cohort study. Br. J. Psychiatry 2020. [Google Scholar] [CrossRef] [PubMed]
- Himmerich, H.; Bartsch, S.; Hamer, H.; Mergl, R.; Schönherr, J.; Petersein, C.; Munzer, A.; Kirkby, K.C.; Bauer, K.; Sack, U. Impact of mood stabilizers and antiepileptic drugs on cytokine production in-vitro. J. Psychiatr. Res. 2013, 47, 1751–1759. [Google Scholar] [CrossRef] [PubMed]
- De Hert, M.; Mazereel, V.; Detraux, J.; Van Assche, K. Prioritizing COVID-19 vaccination for people with severe mental illness. World Psychiatry 2021, 20, 54–55. [Google Scholar] [CrossRef] [PubMed]
- De Picker, L.J.; Dias, M.C.; Benros, M.E.; Vai, B.; Branchi, I.; Benedetti, F.; Borsini, A.; Leza, J.C.; Kärkkäinen, H.; Männikkö, M.; et al. Severe mental illness and European COVID-19 vaccination strategies. Lancet Psychiatry 2021. [Google Scholar] [CrossRef]
- Nicoll, A.; Ciancio, B.; Tsolova, S.; Blank, P.; Yilmaz, C. The scientific basis for offering seasonal influenza immunisation to risk groups in Europe. Euro Surveill 2008, 13. [Google Scholar] [CrossRef] [Green Version]
- Walker, T.A.; Waite, B.; Thompson, M.G.; McArthur, C.; Wong, C.; Baker, M.G.; Wood, T.; Haubrock, J.; Roberts, S.; Gross, D.K.; et al. Risk of Severe Influenza Among Adults With Chronic Medical Conditions. J. Infect. Dis. 2020, 221, 183–190. [Google Scholar] [CrossRef]
| Population with Severe Mental Disorder a n = 97,034 | Reference Population n = 7,683,693 | ||||
|---|---|---|---|---|---|
| n | % | n | % | p-Value | |
| Age Groups (years) | |||||
| 20–39 | 28,725 | 29.6 | 2,612,883 | 34.0 | <0.001 |
| 40–59 | 38,813 | 40.0 | 2,529,453 | 32.9 | <0.001 |
| 60–69 | 16,243 | 16.7 | 1,108,856 | 14.4 | <0.001 |
| 70–79 | 9823 | 10.1 | 923,261 | 12.0 | <0.001 |
| 80+ | 3430 | 3.5 | 509,240 | 6.6 | <0.001 |
| Comorbidities across All Age Groups b,c | |||||
| Diabetes | 7467 | 7.7 | 315,521 | 4.1 | <0.001 |
| Cardiovascular disease | 6780 | 7.0 | 560,754 | 7.3 | <0.001 |
| Hypertension | 10,048 | 10.4 | 769,321 | 10.0 | <0.001 |
| Chronic lung disease | 5335 | 5.5 | 234,227 | 3.0 | <0.001 |
| None of the above | 77,302 | 79.7 | 6,474,647 | 84.3 | <0.001 |
| Influenza/Pneumonia | |||||
|---|---|---|---|---|---|
| Death | Hospitalization | ||||
| OR (95% CI) | p Value | OR (95% CI) | p Value | ||
| Across all Age groups | All individuals | 2.06 (1.87–2.27) | <0.001 | 2.12 (2.03–2.20) | <0.001 |
| Individuals with ≥ 1 known comorbidity b | 1.43 (1.27–1.61) | <0.001 | 1.50 (1.42–1.59) | <0.001 | |
| Individuals without any known comorbidity | 2.68 (2.30–3.13) | <0.001 | 2.56 (2.41–2.72) | <0.001 | |
| 20–39 Years | All individuals | 4.10 (1.67–10.04) | 0.002 | 3.05 (2.60–3.57) | <0.001 |
| Individuals with ≥ 1 known comorbidity | 2.16 (0.29–16.11) | 0.461 | 2.09 (1.47–2.98) | <0.001 | |
| Individuals without any known comorbidity | 4.14 (1.52–11.27) | 0.005 | 2.91 (2.43–3.49) | <0.001 | |
| 40–59 Years | All individuals | 6.25 (4.72–8.29) | <0.001 | 3.75 (3.45–4.08) | <0.001 |
| Individuals with ≥ 1 known comorbidity | 4.53 (3.03–6.77) | <0.001 | 2.90 (2.56–3.28) | <0.001 | |
| Individuals without any known comorbidity | 5.39 (3.61–8.02) | <0.001 | 3.21 (2.86–3.61) | <0.001 | |
| 60–69 Years | All individuals | 4.95 (4.04–6.06) | <0.001 | 3.64 (3.37–3.93) | <0.001 |
| Individuals with ≥ 1 known comorbidity | 3.71 (2.84–4.84) | <0.001 | 2.81 (2.54–3.12) | <0.001 | |
| Individuals without any known comorbidity | 5.32 (3.89–7.27) | <0.001 | 3.82 (3.41–4.29) | <0.001 | |
| 70–79 Years | All individuals | 3.43 (2.89–4.06) | <0.001 | 2.46 (2.27–2.66) | <0.001 |
| Individuals with ≥ 1 known comorbidity | 2.95 (2.40–3.64) | <0.001 | 2.07 (1.87–2.28) | <0.001 | |
| Individuals without any known comorbidity | 3.88 (2.89–5.22) | <0.001 | 2.92 (2.57–3.33) | <0.001 | |
| 80+ Years | All individuals | 1.94 (1.64–2.30) | <0.001 | 1.42 (1.28–1.59) | <0.001 |
| Individuals with ≥ 1 known comorbidity | 1.76 (1.42–2.17) | <0.001 | 1.34 (1.17–1.53) | <0.001 | |
| Individuals without any known comorbidity | 2.49 (1.88–3.30) | <0.001 | 1.72 (1.42–2.08) | <0.001 | |
| Sepsis | |||||
|---|---|---|---|---|---|
| Death | Hospitalization | ||||
| OR (95% CI) | p Value | OR (95% CI) | p Value | ||
| Across all Age groups | All individuals | 1.61 (1.38–1.89) | <0.001 | 1.89 (1.75–2.03) | <0.001 |
| Individuals with ≥ 1 known comorbidity b | 1.06 (0.86–1.30) | 0.587 | 1.37 (1.24–1.51) | <0.001 | |
| Individuals without any known comorbidity | 2.33 (1.81–3.00) | <0.001 | 2.20 (1.97–2.47) | <0.001 | |
| 20–39 Years | All individuals | - c | - | 2.71 (1.98–3.69) | <0.001 |
| Individuals with ≥ 1 known comorbidity | - | - | 1.38 (0.65–2.93) | 0.414 | |
| Individuals without any known comorbidity | - | - | 2.76 (1.96–3.89) | <0.001 | |
| 40–59 Years | All individuals | 3.98 (2.59–6.13) | <0.001 | 3.29 (2.85–3.80) | <0.001 |
| Individuals with ≥ 1 known comorbidity | 3.59 (2.14–6.00) | <0.001 | 2.51 (2.05–3.07) | <0.001 | |
| Individuals without any known comorbidity | 2.1 (0.93–4.73) | 0.073 | 2.71 (2.20–3.34) | <0.001 | |
| 60–69 Years | All individuals | 3.29 (2.45–4.43) | <0.001 | 2.68 (2.34–3.08) | <0.001 |
| Individuals with ≥ 1 known comorbidity | 2.26 (1.53–3.36) | <0.001 | 2.04 (1.71–2.44) | <0.001 | |
| Individuals without any known comorbidity | 3.98 (2.54–6.24) | <0.001 | 2.82 (2.28–3.5) | <0.001 | |
| 70–79 Years | All individuals | 2.19 (1.66–2.88) | <0.001 | 2.02 (1.76–2.33) | <0.001 |
| Individuals with ≥ 1 known comorbidity | 1.56 (1.10–2.23) | 0.014 | 1.75 (1.48–2.08) | <0.001 | |
| Individuals without any known comorbidity | 3.54 (2.29–5.47) | <0.001 | 2.26 (1.77–2.89) | <0.001 | |
| 80+ Years | All individuals | 1.32 (0.95–1.83) | 0.101 | 1.16 (0.94–1.44) | 0.176 |
| Individuals with ≥ 1 known comorbidity | 1.04 (0.68–1.61) | 0.860 | 1.03 (0.78–1.35) | 0.857 | |
| Individuals without any known comorbidity | 2.18 (1.31–3.64) | 0.003 | 1.58 (1.10–2.28) | 0.013 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nilsson, N.H.; Bendix, M.; Öhlund, L.; Widerström, M.; Werneke, U.; Maripuu, M. Increased Risks of Death and Hospitalization in Influenza/Pneumonia and Sepsis for Individuals Affected by Psychotic Disorders, Bipolar Disorders, and Single Manic Episodes: A Retrospective Cross-Sectional Study. J. Clin. Med. 2021, 10, 4411. https://doi.org/10.3390/jcm10194411
Nilsson NH, Bendix M, Öhlund L, Widerström M, Werneke U, Maripuu M. Increased Risks of Death and Hospitalization in Influenza/Pneumonia and Sepsis for Individuals Affected by Psychotic Disorders, Bipolar Disorders, and Single Manic Episodes: A Retrospective Cross-Sectional Study. Journal of Clinical Medicine. 2021; 10(19):4411. https://doi.org/10.3390/jcm10194411
Chicago/Turabian StyleNilsson, Niklas Harry, Marie Bendix, Louise Öhlund, Micael Widerström, Ursula Werneke, and Martin Maripuu. 2021. "Increased Risks of Death and Hospitalization in Influenza/Pneumonia and Sepsis for Individuals Affected by Psychotic Disorders, Bipolar Disorders, and Single Manic Episodes: A Retrospective Cross-Sectional Study" Journal of Clinical Medicine 10, no. 19: 4411. https://doi.org/10.3390/jcm10194411
APA StyleNilsson, N. H., Bendix, M., Öhlund, L., Widerström, M., Werneke, U., & Maripuu, M. (2021). Increased Risks of Death and Hospitalization in Influenza/Pneumonia and Sepsis for Individuals Affected by Psychotic Disorders, Bipolar Disorders, and Single Manic Episodes: A Retrospective Cross-Sectional Study. Journal of Clinical Medicine, 10(19), 4411. https://doi.org/10.3390/jcm10194411







