Understanding the Patterns of Serological Testing for COVID-19 Pre- and Post-Vaccination Rollout in Michigan
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Sample
2.2. Statistical Analysis
2.2.1. Frequency and Pattern of Serologic Testing
2.2.2. Factors Associated with Serologic Tested Individuals and Test Positivity in the Pre-Vaccine Launch Period
2.2.3. Patterns of Pre-Vaccine Launch Test Results for RT-PCR versus Serologic Testing
2.2.4. Factors Associated with Serological Testing in the Post-Vaccine Launch Period
2.2.5. Vaccination Timing and Estimation of Seropositivity
3. Results
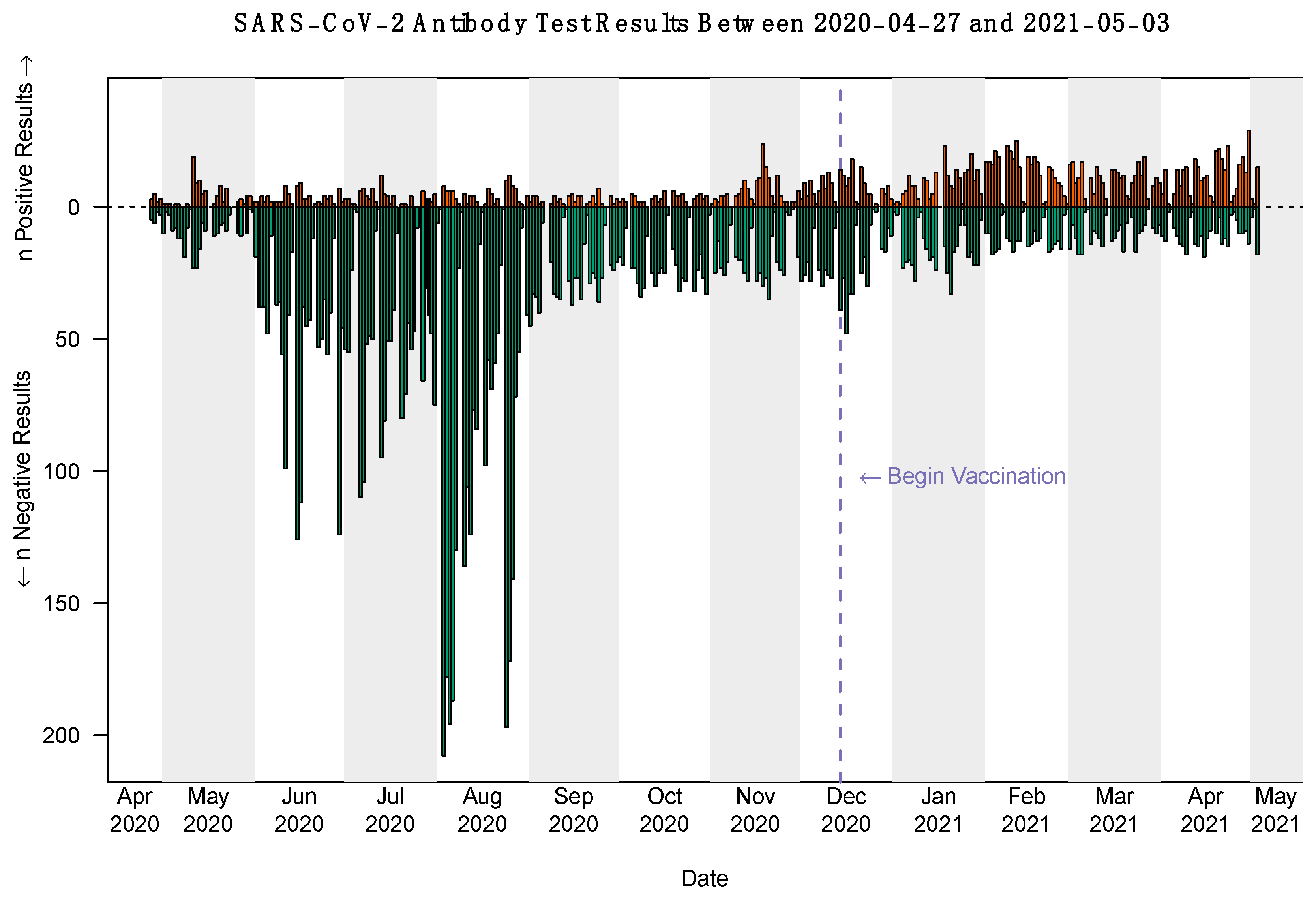
3.1. Frequency and Pattern of Serologic Testing
3.2. Factors Associated with Serologically Tested Individuals and Test Positivity in the Pre-Vaccine Launch Period
3.3. Factors Associated with Serologic Tested Individuals and Test Positivity in the Post-Vaccine Launch Period
3.4. Patterns of Pre-Vaccine Launch Test Results for RT-PCR versus Serologic Testing
3.5. Estimating the COVID-19 Vaccine-Mediated Immunity
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bastos, M.L.; Tavaziva, G.; Abidi, S.K.; Campbell, J.R.; Haraoui, L.P.; Johnston, J.C.; Lan, Z.; Law, S.; MacLean, E.; Khan, F.A.; et al. Diagnostic accuracy of serological tests for covid-19: Systematic review and meta-analysis. BMJ 2020, 370, m2516. [Google Scholar] [CrossRef]
- Winter, K.A.; Hegde, S.T. The important role of serology for COVID-19 control. Lancet Infect. Dis. 2020, 20, 758–759. [Google Scholar] [CrossRef]
- Peeling, R.W.; Wedderburn, C.J.; Garcia, P.J.; Boeras, D.; Fongwen, N.; Nkengasong, J.; Sall, A.; Tanuri, A.; Heymann, D.L. Serology testing in the COVID-19 pandemic response. Lancet Infect. Dis. 2020, 20, e245–e249. [Google Scholar] [CrossRef]
- Clarke, C.; Prendecki, M.; Dhutia, A.; Ali, M.A.; Sajjad, H.; Shivakumar, O.; Lightstone, L.; Kelleher, P.; Pickering, M.C.; Willicombe, M.; et al. High prevalence of asymptomatic COVID-19 infection in hemodialysis patients detected using serologic screening. J. Am. Soc. Nephrol. 2020, 31, 1969–1975. [Google Scholar] [CrossRef] [PubMed]
- Whitaker, H.J.; Elgohari, S.; Rowe, C.; Otter, A.D.; Brooks, T.; Linley, E.; Hayden, I.; Ribeiro, S.; Hewson, J.; Lakhani, A. Impact of COVID-19 vaccination program on seroprevalence in blood donors in England, 2021. J. Infect. 2021, 83, 237–279. [Google Scholar] [CrossRef]
- Gu, T.; Mack, J.A.; Salvatore, M.; Sankar, S.P.; Valley, T.S.; Singh, K.; Nallamothu, B.K.; Kheterpal, S.; Lisabeth, L.; Fritsche, L.G. COVID-19 outcomes, risk factors and associations by race: A comprehensive analysis using electronic health records data in Michigan Medicine. medRxiv 2020. [Google Scholar] [CrossRef]
- Salerno, S.; Zhao, Z.; Prabhu Sankar, S.; Salvatore, M.; Gu, T.; Fritsche, L.G.; Lee, S.; Lisabeth, L.D.; Valley, T.S.; Mukherjee, B. Patterns of repeated diagnostic testing for COVID19 in relation to patient characteristics and outcomes. J. Intern. Med. 2021, 289, 726–737. [Google Scholar] [CrossRef]
- Von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. Ann. Intern. Med. 2007, 147, 573–577. [Google Scholar] [CrossRef] [Green Version]
- Ludvigsson, J.F. Systematic review of COVID-19 in children shows milder cases and a better prognosis than adults. Acta Paediatr. 2020, 109, 1088–1095. [Google Scholar] [CrossRef]
- Ludvigsson, J.F. Children are unlikely to be the main drivers of the COVID-19 pandemic—A systematic review. Acta Paediatr. 2020, 109, 1525–1530. [Google Scholar] [CrossRef]
- Review, W.P. Michigan Population 2021. Available online: https://worldpopulationreview.com/states/michigan-population (accessed on 9 September 2021).
- Michigan.gov. Michigan Data. Available online: https://www.michigan.gov/coronavirus/0,9753,7-406-98163_98173---,00.html (accessed on 9 September 2021).
- Centers for Disease Control and Prevention. Estimated COVID-19 Burden. Available online: https://www.cdc.gov/coronavirus/2019-ncov/cases-updates/burden.html (accessed on 9 September 2021).
- Michigan COVID-19 Vaccine Tracker. Available online: https://data.thetimesherald.com/covid-19-vaccine-tracker/michigan/26/ (accessed on 9 September 2021).
- Washtenaw County, MI COVID-19 Vaccine Tracker. Available online: https://data.thetimesherald.com/covid-19-vaccine-tracker/michigan/washtenaw-county-mi/26161/ (accessed on 9 September 2021).
- Lau, H.; Khosrawipour, T.; Kocbach, P.; Ichii, H.; Bania, J.; Khosrawipour, V. Evaluating the massive underreporting and undertesting of COVID-19 cases in multiple global epicenters. Pulmonology 2021, 27, 110–115. [Google Scholar] [CrossRef]
- Jagodnik, K.M.; Ray, F.; Giorgi, F.M.; Lachmann, A. Correcting under-reported COVID-19 case numbers: Estimating the true scale of the pandemic. medRxiv 2020. [Google Scholar] [CrossRef] [Green Version]
- De Oliveira, A.C.S.; Morita, L.H.M.; Da Silva, E.B.; Zardo, L.A.R.; Fontes, C.J.F.; Granzotto, D.C.T. Bayesian modeling of COVID-19 cases with a correction to account for under-reported cases. Infect. Dis. Model. 2020, 5, 699–713. [Google Scholar] [CrossRef]
- Israel, A.; Feldhamer, I.; Lahad, A.; Levin-Zamir, D.; Lavie, G. Smoking and the risk of COVID-19 in a large observational population study. Medrxiv 2020. [Google Scholar] [CrossRef]
- Ivanov, A.; Annabathula, R.V.; Hammoud, A.; Zhao, D.X.; Correa, A.; Weintraub, W.S.; Herrington, D.M. Association Of Cardiovascular Comorbidities With Sars-cov-2 Seropositivity In A Population-based Cohort. Circulation 2021, 143 (Suppl. 1), A044. [Google Scholar] [CrossRef]
- Lustig, Y.; Sapir, E.; Regev-Yochay, G.; Cohen, C.; Fluss, R.; Olmer, L.; Indenbaum, V.; Mandelboim, M.; Doolman, R.; Amit, S. BNT162b2 COVID-19 vaccine and correlates of humoral immune responses and dynamics: A prospective, single-centre, longitudinal cohort study in health-care workers. Lancet Respir. Med. 2021, 9, 999–1009. [Google Scholar] [CrossRef]
- Xie, J.; Ding, C.; Li, J.; Wang, Y.; Guo, H.; Lu, Z.; Wang, J.; Zheng, C.; Jin, T.; Gao, Y. Characteristics of patients with coronavirus disease (COVID-19) confirmed using an IgM-IgG antibody test. J. Med Virol. 2020, 92, 2004–2010. [Google Scholar] [CrossRef]
- Huang, Z.; Chen, H.; Xue, M.; Huang, H.; Zheng, P.; Luo, W.; Liang, X.; Sun, B.; Zhong, N. Characteristics and roles of severe acute respiratory syndrome coronavirus 2-specific antibodies in patients with different severities of coronavirus 19. Clin. Exp. Immunol. 2020, 202, 210–219. [Google Scholar] [CrossRef] [PubMed]
- Hall, V.J.; Foulkes, S.; Charlett, A.; Atti, A.; Monk, E.J.; Simmons, R.; Wellington, E.; Cole, M.; Saei, A.; Oguti, B. Do antibody positive healthcare workers have lower SARS-CoV-2 infection rates than antibody negative healthcare workers? Large multi-centre prospective cohort study (the SIREN study), England: June to November 2020. Medrxiv 2021. [Google Scholar] [CrossRef]
- Hansen, C.H.; Michlmayr, D.; Gubbels, S.M.; Mølbak, K.; Ethelberg, S. Assessment of protection against reinfection with SARS-CoV-2 among 4 million PCR-tested individuals in Denmark in 2020: A population-level observational study. Lancet 2021, 397, 1204–1212. [Google Scholar] [CrossRef]
- Cox, R.J.; Brokstad, K.A. Not just antibodies: B cells and T cells mediate immunity to COVID-19. Nat. Rev. Immunol. 2020, 20, 581–582. [Google Scholar] [CrossRef]
- An, J.; Liao, X.; Xiao, T.; Qian, S.; Yuan, J.; Ye, H.; Qi, F.; Shen, C.; Wang, L.; Liu, Y. Clinical characteristics of recovered COVID-19 patients with re-detectable positive RNA test. Ann. Transl. Med. 2020, 8, 1084. [Google Scholar] [CrossRef] [PubMed]
- Lechien, J.R.; Chiesa-Estomba, C.M.; Radulesco, T.; Michel, J.; Vaira, L.; Le Bon, S.D.; Horoi, M.; Falanga, C.; Barillari, M.R.; Hans, S. Clinical features of patients who had two COVID-19 episodes: A European multicentre case series. J. Intern. Med. 2021, 290, 421–429. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Facts about COVID-19 Vaccines. Available online: https://www.cdc.gov/coronavirus/2019-ncov/vaccines/vaccine-benefits/facts.html (accessed on 13 December 2020).
- Sahin, U.; Muik, A.; Derhovanessian, E.; Vogler, I.; Kranz, L.M.; Vormehr, M.; Baum, A.; Pascal, K.; Quandt, J.; Maurus, D. COVID-19 vaccine BNT162b1 elicits human antibody and TH 1 T cell responses. Nature 2020, 586, 594–599. [Google Scholar] [CrossRef]
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Pérez Marc, G.; Moreira, E.D.; Zerbini, C. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef]
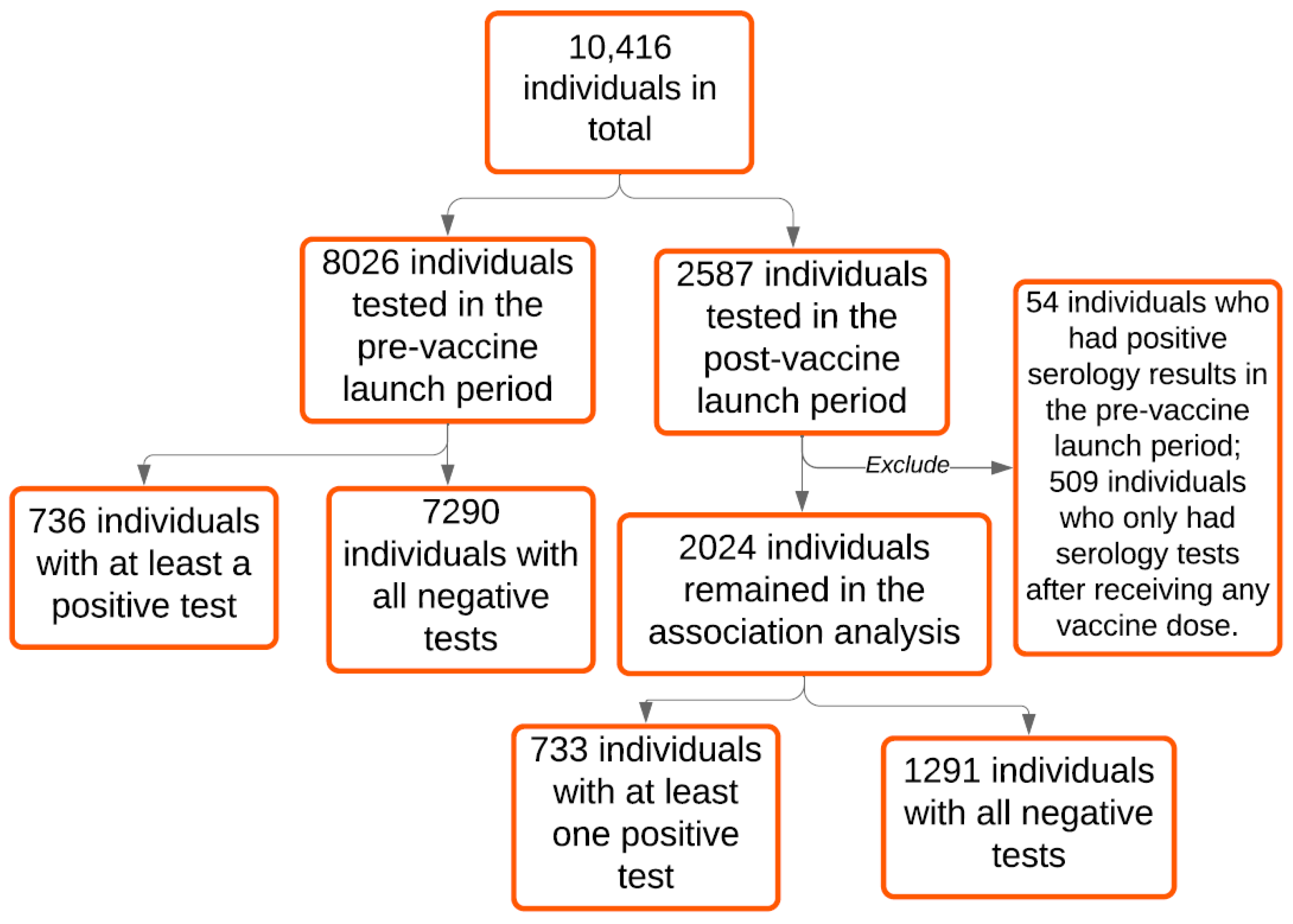
| Overall | Test Period | ||
|---|---|---|---|
| Before 14 December 2020 * | On/After 14 December 2020 | ||
| Number of Individuals | 10,416 | 8026 | 2587 |
| Mean Age at First Test (S.D.) | 43.41 (18.78) | 43.28 (18.29) | 43.71 (20.21) |
| Age Category in Years (%) | |||
| Below 12 | 240 (2.3) | 140 (1.7) | 106 (4.1) |
| Adolescent (>12 and <18) | 215 (2.1) | 131 (1.6) | 87 (3.4) |
| Adult (≥18) | 9961 (95.6) | 7755 (96.6) | 2394 (92.5) |
| Female (%) | 6388 (61.3) | 4926 (61.4) | 1598 (61.8) |
| Race/Ethnicity (%) | |||
| Non-Hispanic White | 7941 (76.2) | 6116 (76.2) | 1987 (76.8) |
| Non-Hispanic Black | 663 (6.4) | 543 (6.8) | 133 (5.1) |
| Other/Unknown | 1812 (17.4) | 1367 (17.0) | 467 (18.1) |
| COVID-19 Research Study Participation | |||
| Yes | 2128 (20.4) | 1955 (24.4) | 200 (7.7) |
| No | 8288 (79.6) | 6071 (75.6) | 2387 (92.3) |
| COVID-19 Test Result ** | |||
| Before 1. Vaccination Dose | |||
| Positive | 1017 (11.5) | 736 (9.2) | 281 (30.8) |
| Negative | 7864 (88.5) | 7290 (90.8) | 630 (69.2) |
| After 1. Before 2. Vaccination Dose | |||
| Positive | n/a | n/a | 46 (50.0) |
| Negative | 46 (50.0) | ||
| After 2. Vaccination Dose | |||
| Positive | n/a | n/a | 337 (86.7) |
| Negative | 52 (13.3) | ||
| Unknown Vaccination Status | |||
| Positive | n/a | n/a | 455 (40.9) |
| Negative | 658 (59.1) | ||
| * Assuming no vaccinations before 14 December 2020. ** Some individuals were tested multiple times at various time points. | |||
| Tested Positive/Tested (%) | |||
| Overall | Test Period | ||
| Before 14 December 2020 * | On/After 14 December 2020 ** | ||
| Overall | 1469/9947 (14.8) | 736/8026 (9.2) | 733/2024 (36.2) |
| Age Category in Years | |||
| Below 12 | 39/240 (16.2) | 12/140 (8.6) | 27/103 (26.2) |
| Adolescent (>12 and <18) | 42/214 (19.6) | 17/131 (13.0) | 25/85 (29.4) |
| Adult (≥18) | 1388/9493 (14.6) | 707/7755 (9.1) | 681/1836 (37.1) |
| Sex | |||
| Female | 855/6073 (14.1) | 420/4926 (8.5) | 435/1217 (35.7) |
| Male | 614/3874 (15.8) | 316/3100 (10.2) | 298/807 (36.9) |
| Race/Ethnicity | |||
| Non-Hispanic White | 1051/7570 (13.9) | 497/6116 (8.1) | 554/1541 (36.0) |
| Non-Hispanic Black | 142/648 (21.9) | 104/543 (19.2) | 38/113 (33.6) |
| Other/Unknown | 276/1729 (16.0) | 135/1367 (9.9) | 141/370 (38.1) |
| COVID-19 Research Study Participation | |||
| Yes | 95/2046 (4.6) | 67/1955 (3.4) | 28/104 (26.9) |
| No | 1374/7901 (17.4) | 669/6071 (11.0) | 705/1920 (36.7) |
| * Assuming no vaccinations before 14 December 2020. ** Excluding 54 individuals who tested positive before 14 December and 509 individuals who were only tested after vaccination; excluding results from tests after vaccination. | |||
| Unadjusted Comparisons | Adjusted Comparisons | ||||||
|---|---|---|---|---|---|---|---|
| Characteristic | Overall, n = 8026 1 | Individuals with All Negative Serologic Results, n = 7290 1 | Individuals with at Least One Positive Serologic Result, n = 736 1 | p-Value 2 | OR 3 | 95% CI 3 | p-Value |
| Age, per 10 years | 4.4 (2.8, 5.9) | 4.4 (2.9, 5.9) | 4.0 (2.2, 5.5) | <0.001 | 0.80 | 0.73, 0.89 | <0.001 |
| Body Mass Index | 27 (23, 31) | 27 (23, 31) | 26 (23, 31) | >0.9 | 1.00 | 0.98, 1.03 | 0.668 |
| Sex | 0.012 | ||||||
| Male | 3100 (39%) | 2784 (38%) | 316 (43%) | — | — | ||
| Female | 4926 (61%) | 4506 (62%) | 420 (57%) | 1.31 | 0.99, 1.74 | 0.061 | |
| Race/Ethnicity | <0.001 | ||||||
| Non-Hispanic White | 6116 (76%) | 5619 (77%) | 497 (68%) | — | — | ||
| Non-Hispanic Black | 543 (6.8%) | 439 (6.0%) | 104 (14%) | 2.18 | 1.33, 3.58 | 0.003 | |
| Other/Unknown | 1367 (17%) | 1232 (17%) | 135 (18%) | 1.04 | 0.7, 1.54 | 0.855 | |
| Smoking Status | <0.001 | ||||||
| Never | 1898 (24%) | 1774 (24%) | 124 (17%) | — | — | ||
| Current/Former | 5427 (68%) | 4882 (67%) | 545 (74%) | 0.49 | 0.35, 0.67 | <0.001 | |
| Unknown | 701 (8.7%) | 634 (8.7%) | 67 (9.1%) | 0.42 | 0.18, 1 | 0.052 | |
| Neighborhood Unemployment 4 | 5.10 (3.76, 7.09) | 5.01 (3.76, 7.03) | 5.64 (4.02, 8.15) | <0.001 | 0.98 | 0.92, 1.04 | 0.533 |
| Neighborhood Poverty 4 | 6 (4, 12) | 6 (4, 12) | 7 (4, 17) | 0.001 | 0.98 | 0.97, 1 | 0.052 |
| Neighborhood Education 4 | 4.7 (2.7, 8.2) | 4.6 (2.7, 8.0) | 5.5 (3.1, 9.3) | <0.001 | 0.97 | 0.94, 1 | 0.085 |
| Population Density, 1000 persons per square mile | 1.84 (0.50, 3.43) | 1.79 (0.48, 3.38) | 2.46 (0.86, 3.86) | <0.001 | 1.10 | 1.03, 1.18 | 0.007 |
| Respiratory Diseases | 0.14 | ||||||
| No | 1450 (20%) | 1333 (20%) | 117 (18%) | — | — | ||
| Yes | 5920 (80%) | 5369 (80%) | 551 (82%) | 4.38 | 3.13, 6.12 | <0.001 | |
| Circulatory Diseases | 0.014 | ||||||
| No | 2518 (34%) | 2261 (34%) | 257 (38%) | — | — | ||
| Yes | 4852 (66%) | 4441 (66%) | 411 (62%) | 2.09 | 1.48, 2.96 | <0.001 | |
| Any Cancer | 0.4 | ||||||
| No | 5673 (77%) | 5150 (77%) | 523 (78%) | — | — | ||
| Yes | 1697 (23%) | 1552 (23%) | 145 (22%) | 1.20 | 0.83, 1.72 | 0.328 | |
| Type 2 Diabetes | 0.2 | ||||||
| No | 6499 (88%) | 5921 (88%) | 578 (87%) | — | — | ||
| Yes | 871 (12%) | 781 (12%) | 90 (13%) | 1.25 | 0.79, 1.98 | 0.349 | |
| Kidney Diseases | <0.001 | ||||||
| No | 6811 (92%) | 6219 (93%) | 592 (89%) | — | — | ||
| Yes | 559 (7.6%) | 483 (7.2%) | 76 (11%) | 1.56 | 0.92, 2.64 | 0.1 | |
| Liver Diseases | >0.9 | ||||||
| No | 6776 (92%) | 6161 (92%) | 615 (92%) | — | — | ||
| Yes | 594 (8.1%) | 541 (8.1%) | 53 (7.9%) | 2.06 | 1.11, 3.84 | 0.024 | |
| Autoimmune Diseases | 0.6 | ||||||
| No | 6122 (83%) | 5562 (83%) | 560 (84%) | — | — | ||
| Yes | 1248 (17%) | 1140 (17%) | 108 (16%) | 1.33 | 0.84,2.12 | 0.227 | |
| Enrolled in a COVID-19 Research study | <0.001 | ||||||
| No | 6071 (76%) | 5402 (74%) | 669 (91%) | — | — | ||
| Yes | 1955 (24%) | 1888 (26%) | 67 (9.1%) | — | — | ||
| Unadjusted Comparisons | Adjusted Comparisons | ||||||
|---|---|---|---|---|---|---|---|
| Characteristic | Overall, n = 2587 1 | Individuals with all Negative Serologic Results, n = 1379 1 | Individuals with at Least One Positive Serologic Result, n = 1208 1 | p-Value 2 | OR 3 | 95% CI 3 | p-Value |
| Age, per 10 years | 4.6 (2.4, 6.1) | 4.5 (2.6, 6.0) | 4.6 (2.2, 6.1) | 0.5 | 0.82 | 0.75, 0.91 | <0.001 |
| Body Mass Index | 27 (23, 31) | 27 (23, 31) | 27 (23, 31) | 0.4 | 1.00 | 0.98, 1.03 | 0.678 |
| Sex | 0.2 | ||||||
| Male | 989 (38%) | 542 (39%) | 447 (37%) | — | — | ||
| Female | 1598 (62%) | 837 (61%) | 761 (63%) | 1.20 | 0.90, 1.61 | 0.215 | |
| Race/Ethnicity | 0.14 | ||||||
| Non-Hispanic White | 1987 (77%) | 1059 (77%) | 928 (77%) | — | — | ||
| Non-Hispanic Black | 133 (5.1%) | 81 (5.9%) | 52 (4.3%) | 0.92 | 0.5, 1.69 | 0.787 | |
| Other/Unknown | 467 (18%) | 239 (17%) | 228 (19%) | 0.88 | 0.59, 1.31 | 0.526 | |
| Smoking Status | 0.003 | ||||||
| Never | 663 (26%) | 391 (28%) | 272 (23%) | — | — | ||
| Current/Former | 1762 (68%) | 908 (66%) | 854 (71%) | 0.70 | 0.52, 0.96 | 0.025 | |
| Unknown | 162 (6.3%) | 80 (5.8%) | 82 (6.8%) | 0.22 | 0.07, 0.71 | 0.012 | |
| Neighborhood Unemployment 4 | 5.25 (3.84, 7.19) | 5.22 (3.80, 7.00) | 5.34 (3.90, 7.45) | 0.044 | 1.00 | 0.94, 1.06 | 1 |
| Neighborhood Poverty 4 | 27 (23, 31) | 27 (23, 31) | 27 (23, 31) | 0.4 | 0.98 | 0.96, 1 | 0.06 |
| Neighborhood Education 4 | 0.2 | 0.96 | 0.93, 0.99 | 0.018 | |||
| Population Density, 1000 persons per square mile | 989 (38%) | 542 (39%) | 447 (37%) | 1.02 | 0.95, 1.1 | 0.599 | |
| Respiratory Diseases | 1598 (62%) | 837 (61%) | 761 (63%) | ||||
| No | 0.14 | — | — | ||||
| Yes | 1987 (77%) | 1059 (77%) | 928 (77%) | 3.09 | 2.24, 4.26 | <0.001 | |
| Circulatory Diseases | 133 (5.1%) | 81 (5.9%) | 52 (4.3%) | ||||
| No | 467 (18%) | 239 (17%) | 228 (19%) | — | — | ||
| Yes | 0.003 | 2.02 | 1.44, 2.84 | <0.001 | |||
| Any Cancer | 663 (26%) | 391 (28%) | 272 (23%) | ||||
| No | 1762 (68%) | 908 (66%) | 854 (71%) | — | — | ||
| Yes | 162 (6.3%) | 80 (5.8%) | 82 (6.8%) | 1.21 | 0.85, 1.72 | 0.288 | |
| Type 2 Diabetes | 0.4 | ||||||
| No | 2082 (87%) | 1113 (86%) | 969 (88%) | — | — | ||
| Yes | 314 (13%) | 176 (14%) | 138 (12%) | 1.23 | 0.78, 1.94 | 0.383 | |
| Kidney Diseases | 0.090 | ||||||
| No | 2107 (88%) | 1147 (89%) | 960 (87%) | — | — | ||
| Yes | 289 (12%) | 142 (11%) | 147 (13%) | 1.64 | 0.98, 2.73 | 0.059 | |
| Liver Diseases | 0.3 | ||||||
| No | 2186 (91%) | 1183 (92%) | 1003 (91%) | — | — | ||
| Yes | 210 (8.8%) | 106 (8.2%) | 104 (9.4%) | 2.05 | 1.08, 3.89 | 0.029 | |
| Autoimmune Diseases | 0.4 | ||||||
| No | 1882 (79%) | 1004 (78%) | 878 (79%) | — | — | ||
| Yes | 514 (21%) | 285 (22%) | 229 (21%) | 2.53 | 1.61, 3.96 | <0.001 | |
| Received at least one vaccine dose | <0.001 | ||||||
| No | 1131 (44%) | 661 (48%) | 470 (39%) | — | — | ||
| Yes | 1456 (56%) | 718 (52%) | 738 (61%) | — | — | ||
| Fully vaccinated | <0.001 | ||||||
| No | 1634 (63%) | 948 (69%) | 686 (57%) | — | — | ||
| Yes | 953 (37%) | 431 (31%) | 522 (43%) | — | — | ||
| Enrolled in a COVID-19 research study | 0.002 | ||||||
| No | 2387 (92%) | 1293 (94%) | 1094 (91%) | — | — | ||
| Yes | 200 (7.7%) | 86 (6.2%) | 114 (9.4%) | — | — | ||
| Subsequent Serologic Test Results | |||
|---|---|---|---|
| Positive at Least Once | Always Negative | ||
| Prior PCR Test Result | Positive | 522 | 371 |
| Negative | 169 | 2587 | |
| Vaccine | |||
|---|---|---|---|
| Pfizer-BioNTech | Moderna | Johnson&Johnson/Jannsen | |
| n | 5129 | 1304 | 161 |
| Vaccination Status (%) | |||
| Received 1 dose | 1197 (23.3) | 583 (44.7) | 161 (100) |
| Received 2 doses | 3932 (76.7) | 721 (55.3) | n/a |
| Timing 2nd Vaccination (%) | |||
| Early a | 4 (0.1) | 3 (0.2) | n/a |
| Recommended b | 3778 (73.7) | 697 (53.5) | n/a |
| Late/Allowable c | 140 (2.7) | 20 (1.5) | n/a |
| Late d | 10 (0.2) | 1 (0.1) | n/a |
| Pending, Late e | 228 (4.4) | 309 (23.7) | n/a |
| Pending f | 969 (18.9) | 274 (21.0) | n/a |
| Positive Tested Individuals Among Vaccinated (%) | ||||
|---|---|---|---|---|
| Overall | Pfizer-BioNTech | Moderna | Johnson&Johnson/Janssen a | |
| n | 472 | 355 | 103 | 14 |
| Between 1st and 2nd Dose | 78/139 (56.1) | 52/98 (53.1) | 26/41 (63.4) | n/a |
| Fully vaccinated b | 296/347 (85.3) | 233/268 (86.9) | 52/65 (80.0) | 11/14 (78.6) |
| 2–4 Weeks after Final Dose | 125/153 (81.7) | 99/119 (83.2) | 21/27 (77.8) | 5/7 (71.4) |
| 4–8 Weeks after Final Dose | 105/125 (84.0) | 76/90 (84.4) | 24/29 (82.8) | 5/6 (83.3) |
| 8–12 Weeks after Final Dose | 55/60 (91.7) | 47/50 (94.0) | 7/9 (77.8) | 1/1 (100.0) |
| >12 Weeks after Final Dose | 19/21 (90.5) | 18/20 (90.0) | 1/1 (100.0) | n/a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, Z.; Salerno, S.; Shi, X.; Lee, S.; Mukherjee, B.; Fritsche, L.G. Understanding the Patterns of Serological Testing for COVID-19 Pre- and Post-Vaccination Rollout in Michigan. J. Clin. Med. 2021, 10, 4341. https://doi.org/10.3390/jcm10194341
Zhao Z, Salerno S, Shi X, Lee S, Mukherjee B, Fritsche LG. Understanding the Patterns of Serological Testing for COVID-19 Pre- and Post-Vaccination Rollout in Michigan. Journal of Clinical Medicine. 2021; 10(19):4341. https://doi.org/10.3390/jcm10194341
Chicago/Turabian StyleZhao, Zhangchen, Stephen Salerno, Xu Shi, Seunggeun Lee, Bhramar Mukherjee, and Lars G. Fritsche. 2021. "Understanding the Patterns of Serological Testing for COVID-19 Pre- and Post-Vaccination Rollout in Michigan" Journal of Clinical Medicine 10, no. 19: 4341. https://doi.org/10.3390/jcm10194341
APA StyleZhao, Z., Salerno, S., Shi, X., Lee, S., Mukherjee, B., & Fritsche, L. G. (2021). Understanding the Patterns of Serological Testing for COVID-19 Pre- and Post-Vaccination Rollout in Michigan. Journal of Clinical Medicine, 10(19), 4341. https://doi.org/10.3390/jcm10194341







