Severe Fatigue and Memory Impairment Are Associated with Lower Serum Level of Anti-SARS-CoV-2 Antibodies in Patients with Post-COVID Symptoms
Abstract
:1. Introduction
2. Methods
2.1. Study Design and Population
2.2. Fatigue and Symptom Severity Assessment
2.3. Blood Sampling and Assay
2.4. Data Collection
2.5. Ethical Considerations
2.6. Statistical Analysis
3. Results
3.1. Participants Characteristics
3.2. Serum Level of Anti-SARS-CoV-2 Antibodies in Patients with Different Phase of Long COVID Disease and Its Relation to Fatigue Status
3.3. Relationship between Demography, Laboratory and Clinical Features at Baseline and Anti-SARS-CoV-2 Antibodies
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO Coronavirus (COVID-19) Dashboard|WHO Coronavirus (COVID-19) Dashboard with Vaccination Data. Available online: https://covid19.who.int/ (accessed on 4 August 2021).
- Shi, Y.; Wang, G.; Cai, X.P.; Deng, J.W.; Zheng, L.; Zhu, H.H.; Zheng, M.; Yang, B.; Chen, Z. An overview of COVID-19. J. Zhejiang Univ. Sci. B. 2020, 21, 343–360. [Google Scholar] [CrossRef]
- Magadum, A.; Kishore, R. Cardiovascular Manifestations of COVID-19 Infection. Cells 2020, 19, 2508. [Google Scholar] [CrossRef]
- Madjunkov, M.; Dviri, M.; Librach, C. A comprehensive review of the impact of COVID-19 on human reproductive biology, assisted reproduction care and pregnancy: A Canadian perspective. J. Ovarian Res. 2020, 13, 140. [Google Scholar] [CrossRef]
- Amruta, N.; Chastain, W.H.; Paz, M.; Solch, R.J.; Murray-Brown, I.C.; Befeler, J.B.; Gressett, T.E.; Longo, M.T.; Engler-Chiurazzi, E.B.; Bix, G. SARS-CoV-2 mediated neuroinflammation and the impact of COVID-19 in neurological disorders. Cytokine Growth Factor Rev. 2021, 58, 1–15. [Google Scholar] [CrossRef]
- Case Definition for Coronavirus Disease 2019 (COVID-19), as of 3 December 2020. Available online: https://www.ecdc.europa.eu/en/covid-19/surveillance/case-definition (accessed on 4 August 2021).
- Public Health Surveillance for COVID-19: Interim Guidance. Available online: https://www.who.int/publications/i/item/who-2019-nCoV-surveillanceguidance-2020.8 (accessed on 4 August 2021).
- Coronavirus Post-COVID Syndrome (Long COVID). Available online: https://www.england.nhs.uk/coronavirus/post-covid-syndrome-longcovid/ (accessed on 4 August 2021).
- Venkatesan, P. NICE guideline on long COVID. Lancet Respir. Med. 2021, 9, 129. [Google Scholar] [CrossRef]
- Crook, H.; Raza, S.; Nowell, J.; Young, M.; Edison, P. Long covid-mechanisms, risk factors, and management. BMJ 2021, 374, n1648. [Google Scholar] [CrossRef]
- Chalder, T.; Berelowitz, G.; Pawlikowska, T.; Watts, L.; Wessely, S.; Wright, D.; Wallace, E.P. Development of a fatigue scale. J. Psychosom. Res. 1993, 37, 147–153. [Google Scholar] [CrossRef] [Green Version]
- Jackson, C. The Chalder Fatigue Scale (CFQ 11). Occup. Med. 2016, 65, 86. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morriss, R.K.; Wearden, A.J.; Mullis, R. Exploring the validity of the Chalder Fatigue scale in chronic fatigue syndrome. J. Psychosom. Res. 1998, 45, 411–417. [Google Scholar] [CrossRef]
- Klok, F.A.; Boon, G.; Barco, S.; Endres, M.; Geelhoed, J.; Knauss, S.; Rezek, S.A.; Spruit, M.A.; Vehreschild, J.; Siegerink, B. The Post-COVID-19 Functional Status scale: A tool to measure functional status over time after COVID-19. Eur. Respir. J. 2020, 56, 2001494. [Google Scholar] [CrossRef]
- Abdulrab, K.; Heun, R. Subjective Memory Impairment. A review of its definitions indicates the need for a comprehensive set of standardised and validated criteria. Eur. Psychiatry 2020, 23, 321–330. [Google Scholar] [CrossRef] [PubMed]
- Mahmud, R.; Rahman, M.M.; Rassel, M.A.; Monayem, F.B.; Sayeed, S.; Islam, M.S.; Islam, M.M. Post-COVID-19 syndrome among symptomatic COVID-19 patients: A prospective cohort study in a tertiary care center of Bangladesh. PLoS ONE 2021, 16, e0249644. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Pérez, O.; Merino, E.; Leon-Ramirez, J.M.; Andres, M.; Ramos, J.M.; Arenas-Jiménez, J.; Asensio, S.; Sanchez, R.; Ruiz-Torregrosa, P.; Galan, I.; et al. Post-acute COVID-19 syndrome. Incidence and risk factors: A Mediterranean cohort study. J. Infect. 2021, 82, 378–383. [Google Scholar] [CrossRef] [PubMed]
- Townsend, L.; Dyer, A.H.; Jones, K.; Dunne, J.; Mooney, A.; Gaffney, F.; O’Connor, L.; Leavy, D.; O’Brien, K.; Dowds, J.; et al. Persistent fatigue following SARS-CoV-2 infection is common and independent of severity of initial infection. PLoS ONE 2020, 15, e0240784. [Google Scholar] [CrossRef]
- Faro, M.; Sàez-Francás, N.; Castro-Marrero, J.; Aliste, L.; Fernández de Sevilla, T.; Alegre, J. Gender differences in chronic fatigue syndrome. Reumatol. Clin. 2016, 12, 72–77. [Google Scholar] [CrossRef]
- Wang, X.; Guo, X.; Xin, Q.; Pan, Y.; Hu, Y.; Li, J.; Chu, Y.; Feng, Y.; Wang, Q. Neutralizing Antibody Responses to Severe Acute Respiratory Syndrome Coronavirus 2 in Coronavirus Disease 2019 Inpatients and Convalescent Patients. Clin. Infect. Dis. 2020, 71, 2688–2694. [Google Scholar] [CrossRef]
- Wu, F.; Liu, M.; Wang, A.; Lu, L.; Wang, Q.; Gu, C.; Chen, J.; Wu, Y.; Xia, S.; Ling, Y.; et al. Evaluating the Association of Clinical Characteristics With Neutralizing Antibody Levels in Patients Who Have Recovered From Mild COVID-19 in Shanghai, China. JAMA Intern. Med. 2020, 180, 1356–1362. [Google Scholar] [CrossRef]
- Terpos, E.; Politou, M.; Sergentanis, T.N.; Mentis, A.; Rosati, M.; Stellas, D.; Bear, J.; Hu, X.; Felber, B.K.; Pappa, V.; et al. Anti-SARS-CoV-2 Antibody Responses in Convalescent Plasma Donors Are Increased in Hospitalized Patients; Subanalyses of a Phase 2 Clinical Study. Microorganisms 2020, 8, 1885. [Google Scholar] [CrossRef]
- Wu, F.; Wang, A.; Liu, M.; Wang, Q.; Chen, J.; Xia, S.; Ling, Y.; Zhang, Y.; Xun, J.; Lu, L.; et al. Neutralizing Antibody Responses to SARS-CoV-2 in a COVID-19 Recovered Patient Cohort and Their Implications. Available online: https://www.medrxiv.org/content/10.1101/2020.03.30.20047365v2 (accessed on 4 August 2021).
- Ibarrondo, F.J.; Fulcher, J.A.; Goodman-Meza, D.; Elliott, J.; Hofmann, C.; Hausner, M.A.; Ferbas, K.G.; Tobin, N.H.; Aldrovandi, G.M.; Yang, O.O. Rapid Decay of Anti-SARS-CoV-2 Antibodies in Persons with Mild Covid-19. N. Engl. J. Med. 2020, 383, 1085–1087. [Google Scholar] [CrossRef]
- Long, Q.X.; Tang, X.J.; Shi, Q.L.; Li, Q.; Deng, H.J.; Yuan, J.; Hu, J.L.; Xu, W.; Zhang, Y.; Lv, F.J.; et al. Clinical and immunological assessment of asymptomatic SARS-CoV-2 infections. Nat. Med. 2020, 26, 1200–1204. [Google Scholar] [CrossRef]
- Marklund, E.; Leach, S.; Axelsson, H.; Nyström, K.; Norder, H.; Bemark, M.; Angeletti, D.; Lundgren, A.; Nilsson, S.; Andersson, L.-M.; et al. Serum-IgG responses to SARS-CoV-2 after mild and severe COVID-19 infection and analysis of IgG non-responders. PLoS ONE 2020, 15, e0241104. [Google Scholar] [CrossRef]
- Guthmiller, J.J.; Stovicek, O.; Wang, J.; Changrob, S.; Li, L.; Halfmann, P.; Zheng, N.Y.; Utset, H.; Stamper, C.T.; Dugan, H.L.; et al. SARS-CoV-2 Infection Severity Is Linked to Superior Humoral Immunity against the Spike. mBio 2021, 12, e02940-20. [Google Scholar] [CrossRef]
- McAndrews, K.M.; Dowlatshahi, D.P.; Dai, J.; Becker, L.M.; Hensel, J.; Snowden, L.M.; Leveille, J.M.; Brunner, M.R.; Holden, K.W.; Hopkins, N.S.; et al. Heterogeneous antibodies against SARS-CoV-2 spike receptor binding domain and nucleocapsid with implications for COVID-19 immunity. JCI Insight 2020, 5, e142386. [Google Scholar] [CrossRef]
- Schmidt, F.; Weisblum, Y.; Muecksch, F.; Hoffmann, H.H.; Michailidis, E.; Lorenzi, J.; Mendoza, P.; Rutkowska, M.; Bednarski, E.; Gaebler, C.; et al. Measuring SARS-CoV-2 neutralizing antibody activity using pseudotyped and chimeric viruses. J. Exp. Med. 2020, 217, e20201181. [Google Scholar] [CrossRef]
- Cao, W.C.; Liu, W.; Zhang, P.H.; Zhang, F.; Richardus, J.H. Disappearance of antibodies to SARS-associated coronavirus after recovery. N. Engl. J. Med. 2007, 357, 1162–1163. [Google Scholar] [CrossRef]
- Chia, W.N.; Zhu, F.; Ong, S.; Young, B.E.; Fong, S.W.; Le Bert, N.; Tan, C.W.; Tiu, C.; Zhang, J.; Tan, S.Y.; et al. Dynamics of SARS-CoV-2 neutralising antibody responses and duration of immunity: A longitudinal study. Lancet Microbe 2021, 2, e240–e249. [Google Scholar] [CrossRef]
- Bansal, A.S.; Bradley, A.S.; Bishop, K.N.; Kiani-Alikhan, S.; Ford, B. Chronic fatigue syndrome, the immune system and viral infection. Brain Behav. Immun. 2012, 26, 24–31. [Google Scholar] [CrossRef]
- Loebel, M.; Strohschein, K.; Giannini, C.; Koelsch, U.; Bauer, S.; Doebis, C.; Thomas, S.; Unterwalder, N.; von Baehr, V.; Reinke, P.; et al. Deficient EBV-specific B- and T-cell response in patients with chronic fatigue syndrome. PLoS ONE 2014, 9, e85387. [Google Scholar] [CrossRef] [Green Version]
- Raijmakers, R.; Koeken, V.; Jansen, A.; Keijmel, S.P.; Roerink, M.E.; Joosten, L.; Netea, M.G.; van der Meer, J.; Bleeker-Rovers, C.P. Cytokine profiles in patients with Q fever fatigue syndrome. J. Infect. 2019, 78, 349–357. [Google Scholar] [CrossRef]
- Raijmakers, R.; Jansen, A.; Keijmel, S.P.; Ter Horst, R.; Roerink, M.E.; Novakovic, B.; Joosten, L.; van der Meer, J.; Netea, M.G.; Bleeker-Rovers, C.P. A possible role for mitochondrial-derived peptides humanin and MOTS-c in patients with Q fever fatigue syndrome and chronic fatigue syndrome. J. Transl. Med. 2019, 17, 157. [Google Scholar] [CrossRef]
- Raijmakers, R.; Roerink, M.E.; Jansen, A.; Keijmel, S.P.; Gacesa, R.; Li, Y.; Joosten, L.; van der Meer, J.; Netea, M.G.; Bleeker-Rovers, C.P.; et al. Multi-omics examination of Q fever fatigue syndrome identifies similarities with chronic fatigue syndrome. J. Transl. Med. 2020, 18, 448. [Google Scholar] [CrossRef]
- Villapol, S. Gastrointestinal symptoms associated with COVID-19: Impact on the gut microbiome. Transl. Res. 2020, 226, 57–69. [Google Scholar] [CrossRef] [PubMed]
- Zuo, T.; Liu, Q.; Zhang, F.; Lui, G.C.; Tso, E.Y.; Yeoh, Y.K.; Chen, Z.; Boon, S.S.; Chan, F.K.; Chan, P.K.; et al. Depicting SARS-CoV-2 faecal viral activity in association with gut microbiota composition in patients with COVID-19. Gut 2021, 70, 276–284. [Google Scholar] [CrossRef] [PubMed]
- Ye, Q.; Wang, B.; Mao, J. The pathogenesis and treatment of the ‘Cytokine Storm’ in COVID-19. J. Infect. 2020, 80, 607–613. [Google Scholar] [CrossRef]
- Gardoni, F.; Boraso, M.; Zianni, E.; Corsini, E.; Galli, C.L.; Cattabeni, F.; Marinovich, M.; Di Luca, M.; Viviani, B. Distribution of interleukin-1 receptor complex at the synaptic membrane driven by interleukin-1β and NMDA stimulation. J. Neuroinflamm. 2011, 8, 14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prieto, G.A.; Tong, L.; Smith, E.D.; Cotman, C.W. TNFα and IL-1β but not IL-18 Suppresses Hippocampal Long-Term Potentiation Directly at the Synapse. Neurochem. Res. 2019, 44, 49–60. [Google Scholar] [CrossRef] [PubMed]
- Motaghinejad, M.; Gholami, M. Possible Neurological and Mental Outcomes of COVID-19 Infection: A Hypothetical Role of ACE-2\Mas\BDNF Signaling Pathway. Int. J. Prev. Med. 2020, 9, 84. [Google Scholar] [CrossRef]
- Zheng, J.L.; Li, G.Z.; Chen, S.Z.; Wang, J.J.; Olson, J.E.; Xia, H.J.; Lazartigues, E.; Zhu, Y.L.; Chen, Y.F. Angiotensin converting enzyme 2/Ang-(1-7)/mas axis protects brain from ischemic injury with a tendency of age-dependence. CNS Neurosci. Ther. 2014, 20, 452–459. [Google Scholar] [CrossRef] [Green Version]
- Ng, T.K.S.; Ho, C.S.H.; Tam, W.W.S.; Kua, E.H.; Ho, R.C. Decreased Serum Brain-Derived Neurotrophic Factor (BDNF) Levels in Patients with Alzheimer’s Disease (AD): A Systematic Review and Meta-Analysis. Int. J. Mol. Sci. 2019, 20, 257. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arnold, D.T.; Milne, A.; Samms, E.; Stadon, L.; Maskell, N.A.; Hamilton, F.W. Are Vaccines Safe in Patients with Long COVID? A Prospective Observational Study. Available online: https://www.medrxiv.org/content/10.1101/2021.03.11.21253225v3 (accessed on 8 August 2021).
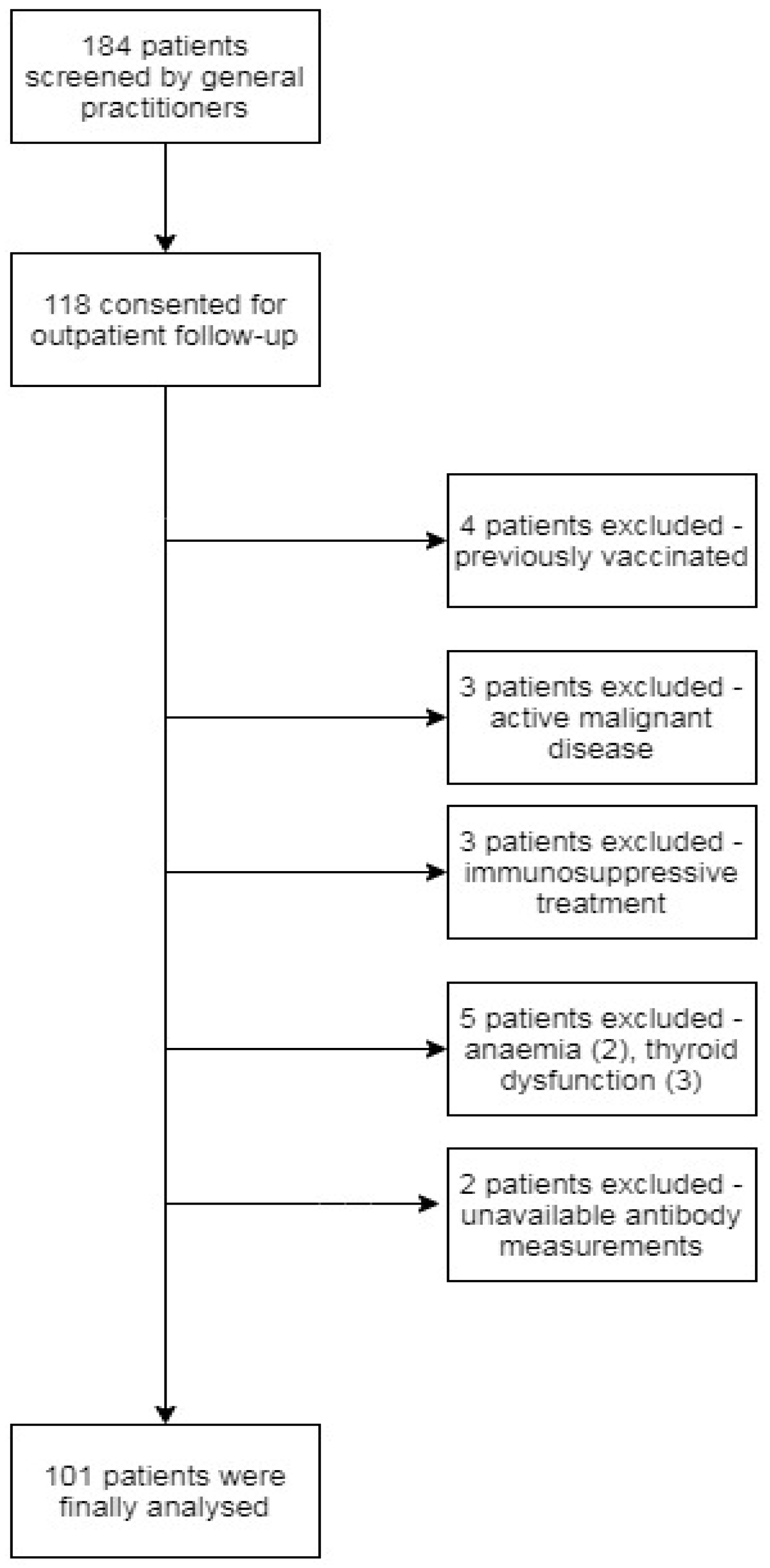
| Total Population (N = 101) | 4–12 Weeks (N = 68) | >12 Weeks (N = 33) | p-Value | |
|---|---|---|---|---|
| Age (mean ± SD) | 50 ± 12 | 51.1 ± 11.5 | 47.7 ± 12.6 | 0.118 |
| Female (N (%)) | 60 (59.4) | 41 (60.3) | 21 (63.6) | 0.746 |
| BMI (mean ± SD) | 27.8 ± 6.4 | 27.5 ± 5 | 28.3 ± 9 | 0.476 |
| Symptom onset to baseline visit (days; mean ± SD) | 70.1 ± 30 | 52.7 ± 18 | 106 ± 13 | <0.001 |
| Hospitalization (N (%)) | 39 (38.6) | 34 (50) | 5 (15.2) | 0.001 |
| PCFS (median (IQR)) | 2 (1–2) | 2 (1–2) | 1 (1–2) | 0.766 |
| Total value of post-COVID symptoms on a VAS scale * (median (IQR)) | 16 (9–19) | 16 (9–19) | 16 (10–21) | 0.204 |
| Total CFQ-11 score (mean ± SD) (Likert scoring) | 15.7 ± 5.9 | 15.8 ± 5.5 | 15.6 ± 6.7 | 0.951 |
| Physical fatigue (mean ± SD) (CFQ-11 items 1–7) | 11.7 ± 4.2 | 11.7 ± 3.8 | 11.8 ± 4.9 | 0.668 |
| Psychological fatigue (mean ± SD) (CFQ-11 items 8–11) | 4.1 ± 2.6 | 4.2 ± 2.5 | 3.9 ± 2.8 | 0.619 |
| Total CFQ-11 score (mean ± SD) (bimodal scoring) | 4.9 ± 3.2 | 4.9 ± 3 | 5 ± 3 | 0.773 |
| Anti-SARS-CoV-2 S-Ig (U/mL; median (IQR)) | 125 (40–289) | 169.5 (58–308) | 70.8 (31–131) | 0.037 |
| Anti-SARS-CoV-2 NC-Ig (U/mL; median (IQR)) | 53.9 (18–105) | 54.7 (21–107) | 41.8 (9–97) | 0.201 |
| Non-Severe Fatigue (N = 38) | Severe Fatigue (N = 63) | p-Value | |
|---|---|---|---|
| Age (mean ± SD) | 52.2 ± 13 | 48.7 ± 11 | 0.064 |
| Female (N (%)) | 18 (47.4) | 44 (69.8) | 0.025 * |
| BMI (mean ± SD) | 28.5 ± 8 | 27.3 ± 5 | 0.468 |
| Symptom onset to baseline (day; mean ± SD) | 71.2 ± 26 | 69 ± 32 | 0.641 |
| Hospitalization (N (%)) | 16 (42.1) | 23 (36.5) | 0.576 |
| Total number of comorbidities (median (IQR)) | 1 (0–2) | 1 (0–1) | 0.658 |
| PCFS (median, IQR) | 1 (0–1) | 2 (1–2) | <0.001 |
| Length of hospitalization (day, mean ± SD) | 3.2 ± 4 | 2.3 ± 3.5 | 0.370 |
| CTss (median (IQR)) | 9 (6–11) | 11 (7–13) | 0.217 |
| O2 supplementation (N (%)) | 8 (21) | 14 (22) | 0.890 |
| Antiviral medication (N (%)) | 15 (39.5) | 19 (30.2) | 0.337 |
| CRP (mg/L; median (IQR)) | 52.9 (14–81) | 29.5 (12–79) | 0.630 |
| High-sensitivity Troponin-T (ng/L; median (IQR)) | 10.4 (4–13) | 6.6 (5–9) | 0.201 |
| NLR (median (IQR)) | 3.2 (2–6) | 4.3 (2–6) | 0.476 |
| IL-6 (pg/mL; median (IQR)) | 29 (12–36) | 28 (20–48) | 0.580 |
| D-dimer (µg/L; median (IQR)) | 738 (573–1009) | 740 (412–1159) | 0.730 |
| Ferritin (µg/L; median (IQR)) | 552 (354–899) | 569 (462–881) | 0.695 |
| Total CFQ-11 score (mean ± SD) (Likert Scoring) | 9.9 ± 3.7 | 19.2 ± 3.7 | <0.001 |
| Physical fatigue (mean ± SD) (CFQ-11 items 1–7) | 7.3 ± 2.6 | 14.3 ± 2.6 | <0.001 |
| Psychological fatigue (mean ± SD) (CFQ-11 items 8–11) | 2.6 ± 1.9 | 5 ± 2.6 | <0.001 |
| Total CFQ-11 score (mean ± SD) (bimodal scoring) | 1.5 ± 1.4 | 7 ± 1.9 | <0.001 |
| Number of post-COVID symptoms (median (IQR)) | 2 (1–3) | 7 (5–10) | <0.001 |
| Total value of post-COVID symptoms on a VAS scale * (median (IQR)) | 6 (0–14) | 18 (15–21) | <0.001 |
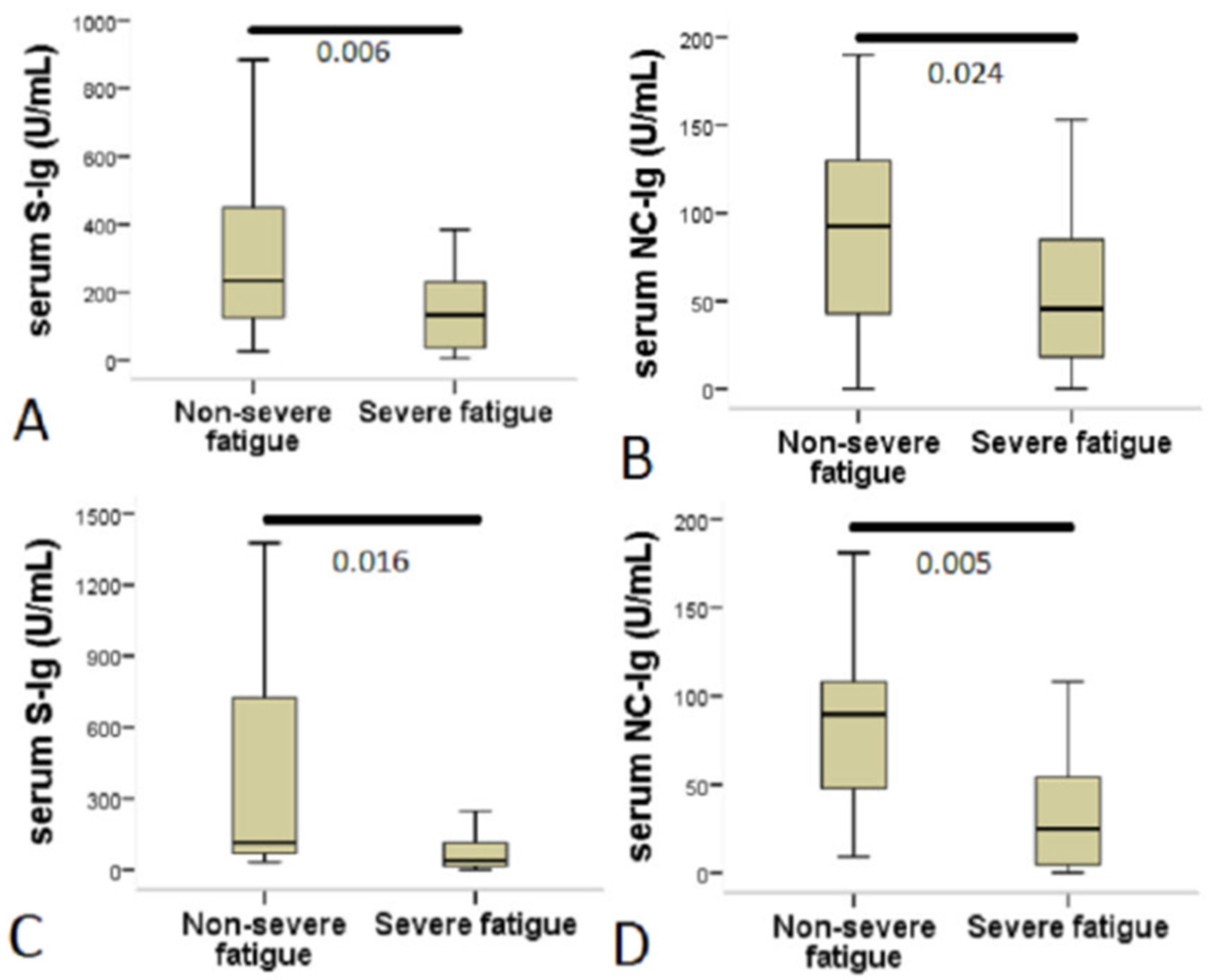
| Anti-SARS-CoV-2 S-Ig (U/mL; median (IQR)) | 211 (103–473) | 72.7 (25–201) | <0.001 |
| Anti-SARS-CoV-2 NC-Ig (U/mL; median (IQR)) | 91.8 (46–125) | 34.5 (10–66) | <0.001 |
| Variables | S-Ig | NC-Ig |
|---|---|---|
| Total number of symptoms at baseline | −0.229 * | −0.237 * |
| Total number of comorbidities | 0.225 * | 0.108 |
| Body mass index | 0.249 * | 0.175 |
| Length of hospitalization | 0.519 ** | 0.230 |
| Admission GOT ** | 0.233 | −0.476 * |
| Admission GPT ** | 0.159 | −0.334 * |
| Admission platelet ** | 0.344 * | 0.148 |
| Admission hs-Tn-T ** | −0.430 * | 0.065 |
| Baseline lymphocyte count | 0.251 * | 0.133 |
| Baseline monocyte count | 0.337 ** | 0.069 |
| Baseline eosinophile count | 0.147 | 0.233 * |
| Total score on VAS * | −0.162 | −0.292 * |
| Baseline GOT | 0.245 * | 0.062 |
| Baseline GPT | 0.318 ** | 0.006 |
| Baseline GGT | 0.287 * | −0.013 |
| Baseline hs-Tn-T | 0.295 * | 0.148 |
| Baseline LDH | 0.327 ** | 0.124 |
| Value of NC-Ig (U/mL, Median as the Cutoff) § | ||||
| Variables | B | Odds ratio | 95% CI | p-Value |
| Symptom onset to baseline (days) | 0.993 | 0.660 | 0.976–1.010 | 0.417 |
| Total CFQ-11 | 0.883 | 9.037 | 0.814–0.958 | 0.003 |
| Age | 1.031 | 2.512 | 0.993–1.071 | 0.113 |
| Need for hospitalization | 0.908 | 0.030 | 0.308–2.676 | 0.862 |
| Value of S-Ig (U/mL, Median as the Cutoff) § | ||||
| Variables | B | Odds ratio | 95% CI | p-value |
| Symptom onset to baseline (days) | 0.985 | 2.337 | 0.966–1.004 | 0.126 |
| Total CFQ-11 | 0.905 | 4.968 | 0.829–0.988 | 0.026 |
| Age | 1.045 | 3.929 | 1.000–1.091 | 0.047 |
| Need for hospitalization | 0.169 | 8.772 | 0.052–0.548 | 0.003 |
| Severe Fatigue Status | ||||
| Variables | B | Odds ratio | 95% CI | p-value |
| Age | 0.950 | 1.811 | 0.882–1.024 | 0.178 |
| S-Ig | 0.997 | 4.688 | 0.994–1.000 | 0.030 |
| NC-Ig | 0.993 | 1.105 | 0.980–1.006 | 0.293 |
| Total VAS * | 1.096 | 1.078 | 0.922–1.303 | 0.299 |
| Total number of symptoms at baseline | 2.021 | 6.917 | 1.196–3.413 | 0.009 |
| Sex | 0.885 | 0.024 | 0.190–4.117 | 0.877 |
| Total number of symptoms at baseline | 1.159 | 0.801 | 0.839–1.601 | 0.371 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Molnar, T.; Varnai, R.; Schranz, D.; Zavori, L.; Peterfi, Z.; Sipos, D.; Tőkés-Füzesi, M.; Illes, Z.; Buki, A.; Csecsei, P. Severe Fatigue and Memory Impairment Are Associated with Lower Serum Level of Anti-SARS-CoV-2 Antibodies in Patients with Post-COVID Symptoms. J. Clin. Med. 2021, 10, 4337. https://doi.org/10.3390/jcm10194337
Molnar T, Varnai R, Schranz D, Zavori L, Peterfi Z, Sipos D, Tőkés-Füzesi M, Illes Z, Buki A, Csecsei P. Severe Fatigue and Memory Impairment Are Associated with Lower Serum Level of Anti-SARS-CoV-2 Antibodies in Patients with Post-COVID Symptoms. Journal of Clinical Medicine. 2021; 10(19):4337. https://doi.org/10.3390/jcm10194337
Chicago/Turabian StyleMolnar, Tihamer, Reka Varnai, Daniel Schranz, Laszlo Zavori, Zoltan Peterfi, David Sipos, Margit Tőkés-Füzesi, Zsolt Illes, Andras Buki, and Peter Csecsei. 2021. "Severe Fatigue and Memory Impairment Are Associated with Lower Serum Level of Anti-SARS-CoV-2 Antibodies in Patients with Post-COVID Symptoms" Journal of Clinical Medicine 10, no. 19: 4337. https://doi.org/10.3390/jcm10194337
APA StyleMolnar, T., Varnai, R., Schranz, D., Zavori, L., Peterfi, Z., Sipos, D., Tőkés-Füzesi, M., Illes, Z., Buki, A., & Csecsei, P. (2021). Severe Fatigue and Memory Impairment Are Associated with Lower Serum Level of Anti-SARS-CoV-2 Antibodies in Patients with Post-COVID Symptoms. Journal of Clinical Medicine, 10(19), 4337. https://doi.org/10.3390/jcm10194337







