Vaccination for Respiratory Infections in Patients with Heart Failure
Abstract
:1. The Interrelationship between Inflammation, Infection, Thrombosis, and Heart Failure
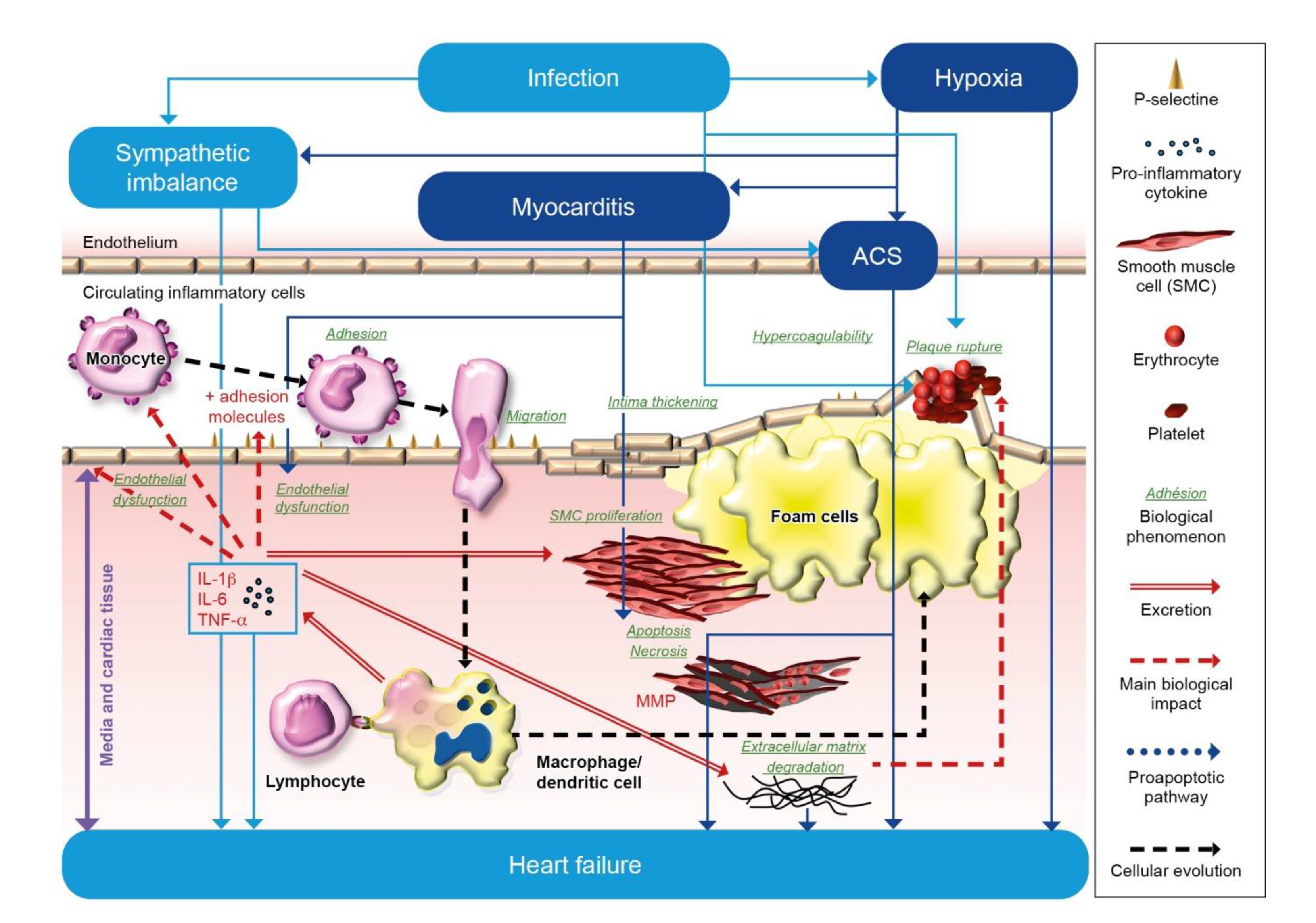
1.1. Inflammation and Heart Failure
1.2. Respiratory Infections and Heart Failure
1.3. Risk of Thrombosis in Respiratory Infections
2. Impact of Respiratory Infections on Cardiovascular Diseases
2.1. Respiratory Infection with Streptococcus Pneumoniae or Influenza Virus
2.2. Respiratory Infection with SARS-CoV-2
3. Current Knowledge about Vaccination and Heart Failure
3.1. Influenza Vaccine
3.2. Pneumococcal Vaccine
3.3. COVID-19 Vaccine
4. Current Recommendations for Vaccination
4.1. Influenza Vaccination
4.2. Pneumococcal Vaccination
4.3. COVID-19 Vaccination
5. Suggestions for Improvement of Vaccination
6. Respiratory Vaccinations from the SARS-CoV2 Pandemic Perspective
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Murphy, S.P.; Kakkar, R.; McCarthy, C.P.; Januzzi, J.L., Jr. Inflammation in heart failure: JACC state-of-the-art review. J. Am. Coll. Cardiol. 2020, 75, 1324–1340. [Google Scholar] [CrossRef]
- Dubrock, H.M.; AbouEzzeddine, O.F.; Redfield, M.M. High-sensitivity C-reactive protein in heart failure with preserved ejection fraction. PLoS ONE 2018, 13, e0201836. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cohen-Solal, A.; Leclercq, C.; Deray, G.; Lasocki, S.; Zambrowski, J.-J.; Mebazaa, A.; de Groote, P.; Damy, T.; Galinier, M. Iron deficiency: An emerging therapeutic target in heart failure. Heart 2014, 100, 1414–1420. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huet, F.; Akodad, M.; Fauconnier, J.; Lacampagne, A.; Roubille, F. Anti-inflammatory drugs as promising cardiovascular treatments. Expert Rev. Cardiovasc. Ther. 2016, 15, 109–125. [Google Scholar] [CrossRef]
- Bhatt, A.S.; DeVore, A.D.; Hernandez, A.F.; Mentz, R.J. Can vaccinations improve heart failure outcomes? Contemporary data and future directions. JACC Heart Fail. 2017, 5, 194–203. [Google Scholar] [CrossRef]
- Clerkin, K.J.; Fried, J.A.; Raikhelkar, J.; Sayer, G.; Griffin, J.M.; Masoumi, A.; Jain, S.S.; Burkhoff, D.; Kumaraiah, D.; Rabbani, L.; et al. COVID-19 and cardiovascular disease. Circulation 2020, 141, 1648–1655. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dean, S.M.; Abraham, W. Venous thromboembolic disease in congestive heart failure. Congest. Heart Fail. 2010, 16, 164–169. [Google Scholar] [CrossRef]
- Kwong, J.C.; Schwartz, K.L.; Campitelli, M.A.; Chung, H.; Crowcroft, N.S.; Karnauchow, T.; Katz, K.; Ko, D.T.; McGeer, A.J.; McNally, D.; et al. Acute myocardial infarction after laboratory-confirmed influenza infection. N. Engl. J. Med. 2018, 378, 345–353. [Google Scholar] [CrossRef]
- Rothnie, K.J.; Connell, O.; Mullerova, H.; Smeeth, L.; Pearce, N.; Douglas, I.; Quint, J.K. Myocardial infarction and ischemic stroke after exacerbations of chronic obstructive pulmonary disease. Ann. Am. Thorac. Soc. 2018, 15, 935–946. [Google Scholar] [CrossRef]
- De Caterina, R.; D’Ugo, E.; Libby, P. Inflammation and thrombosis—Testing the hypothesis with anti-inflammatory drug trials. Thromb. Haemost. 2016, 116, 1012–1021. [Google Scholar]
- Levi, M.; Van Der Poll, T.; Schultz, M. Infection and inflammation as risk factors for thrombosis and atherosclerosis. Semin. Thromb. Hemost. 2012, 38, 506–514. [Google Scholar] [CrossRef] [PubMed]
- Iba, T.; Levy, J.H. Inflammation and thrombosis: Roles of neutrophils, platelets and endothelial cells and their interactions in thrombus formation during sepsis. J. Thromb. Haemost. 2018, 16, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Laridan, E.; Martinod, K.; De Meyer, S.F. Neutrophil extracellular traps in arterial and venous thrombosis. Semin. Thromb. Hemost. 2019, 45, 086–093. [Google Scholar] [CrossRef] [PubMed]
- Alon, D.; Stein, G.Y.; Korenfeld, R.; Fuchs, S. Predictors and outcomes of infection-related hospital admissions of heart failure patients. PLoS ONE 2013, 8, e72476. [Google Scholar] [CrossRef]
- Shen, L.; Jhund, P.S.; Anand, I.S.; Bhatt, A.S.; Desai, A.S.; Maggioni, A.P.; Martinez, F.A.; Pfeffer, M.A.; Rizkala, A.R.; Rouleau, J.L.; et al. Incidence and outcomes of pneumonia in patients with heart failure. J. Am. Coll. Cardiol. 2021, 77, 1961–1973. [Google Scholar] [CrossRef]
- Fonarow, G.C.; Abraham, W.T.; Albert, N.M.; Stough, W.G.; Gheorghiade, M.; Greenberg, B.H.; O’Connor, C.M.; Pieper, K.; Sun, J.L.; Yancy, C.W.; et al. Factors identified as precipitating hospital admissions for heart failure and clinical outcomes: Findings from OPTIMIZE-HF. Arch. Intern. Med. 2008, 168, 847–854. [Google Scholar] [CrossRef] [Green Version]
- Narukawa, M.; Minezaki, K.K.; Okubo, M.; Kario, K. Impact of an influenza pandemic on the mortality of congestive heart failure in older Japanese: The 1998 Japanese influenza pandemic. J. Am. Geriatr. Soc. 2001, 49, 689–690. [Google Scholar] [CrossRef]
- Sandoval, C.; Walter, S.D.; Krueger, P.; Smieja, M.; Smith, A.; Yusuf, S.; Loeb, M.B. Risk of hospitalization during influenza season among a cohort of patients with congestive heart failure. Epidemiol. Infect. 2006, 135, 574–582. [Google Scholar] [CrossRef]
- Kytomaa, S.; Hegde, S.; Claggett, B.; Udell, J.A.; Rosamond, W.; Temte, J.; Nichol, K.; Wright, J.D.; Solomon, S.D.; Vardeny, O. Association of Influenza-like illness activity with hospitalizations for heart failure: The atherosclerosis risk in communities study. JAMA Cardiol. 2019, 4, 363–369. [Google Scholar] [CrossRef] [Green Version]
- Panhwar, M.S.; Kalra, A.; Gupta, T.; Kolte, D.; Khera, S.; Bhatt, D.L.; Ginwalla, M. Effect of influenza on outcomes in patients with heart failure. JACC Heart Fail. 2019, 7, 112–117. [Google Scholar] [CrossRef]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Li, B.; Yang, J.; Zhao, F.; Zhi, L.; Wang, X.; Liu, L.; Bi, Z.; Zhao, Y. Prevalence and impact of cardiovascular metabolic diseases on COVID-19 in China. Clin. Res. Cardiol. 2020, 109, 531–538. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Garcia, J.; Lee, S.; Gupta, A.; Cagliostro, M.; Joshi, A.A.; Rivas-Lasarte, M.; Contreras, J.; Mitter, S.S.; LaRocca, G.; Tlachi, P.; et al. Prognostic impact of prior heart failure in patients hospitalized with COVID-19. J. Am. Coll. Cardiol. 2020, 76, 2334–2348. [Google Scholar] [CrossRef] [PubMed]
- Soulat-Dufour, L.; Lang, S.; Ederhy, S.; Adavane-Scheuble, S.; Chauvet-Droit, M.; Nhan, P.; Jean, M.-L.; Ben Said, R.; Issaurat, P.; Boccara, F.; et al. Left ventricular ejection fraction: An additional risk marker in COVID-19. Arch. Cardiovasc. Dis. 2020, 113, 760–762. [Google Scholar] [CrossRef]
- Bhatt, A.S.; Jering, K.S.; Vaduganathan, M.; Claggett, B.L.; Cunningham, J.W.; Rosenthal, N.; Signorovitch, J.; Thune, J.J.; Vardeny, O.; Solomon, S.D. Clinical outcomes in patients with heart failure hospitalized with COVID-19. JACC Heart Fail. 2020, 9, 65–73. [Google Scholar] [CrossRef]
- Rey, J.R.; Caro-Codon, J.; Rosillo, S.O.; Iniesta, A.M.; Castrejon-Castrejon, S.; Marco-Clement, I.; Martin-Polo, L.; Merino-Argos, C.; Rodriguez-Sotelo, L.; Garcia-Veas, J.M.; et al. Heart failure in COVID-19 patients: Prevalence, incidence and prognostic implications. Eur. J. Heart Fail. 2020, 22, 2205–2215. [Google Scholar] [CrossRef]
- Vardeny, O.; Claggett, B.; Udell, J.A.; Packer, M.; Zile, M.; Rouleau, J.; Swedberg, K.; Desai, A.S.; Lefkowitz, M.; Shi, V.; et al. Influenza vaccination in patients with chronic heart failure: The PARADIGM-HF trial. JACC Heart Fail. 2016, 4, 152–158. [Google Scholar] [CrossRef]
- Bhatt, A.S.; Liang, L.; DeVore, A.D.; Fonarow, G.C.; Solomon, S.D.; Vardeny, O.; Yancy, C.W.; Mentz, R.J.; Khariton, Y.; Chan, P.S. Vaccination trends in patients with heart failure: Insights from get with the guidelines—Heart failure. JACC Heart Fail. 2018, 6, 844–855. [Google Scholar] [CrossRef]
- Udell, J.A.; Zawi, R.; Bhatt, D.L.; Keshtkar-Jahromi, M.; Gaughran, F.; Phrommintikul, A.; Ciszewski, A.; Vakili, H.; Hoffman, E.B.; Farkouh, M.E.; et al. Association between influenza vaccination and cardiovascular outcomes in high-risk patients: A meta-analysis. JAMA 2013, 310, 1711–1720. [Google Scholar] [CrossRef]
- Loeb, M.; Dokainish, H.; Dans, A.; Palileo-Villanueva, L.M.; Roy, A.; Karaye, K.; Zhu, J.; Liang, Y.; Goma, F.; Damasceno, A.; et al. Randomized controlled trial of influenza vaccine in patients with heart failure to reduce adverse vascular events (IVVE): Rationale and design. Am. Heart J. 2019, 212, 36–44. [Google Scholar] [CrossRef]
- Vardeny, O.; Moran, J.J.M.; Sweitzer, N.K.; Johnson, M.R.; Hayney, M.S. Decreased T-cell responses to influenza vaccination in patients with heart failure. Pharmacotherapy 2010, 30, 10–16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vardeny, O.; Kim, K.; Udell, J.A.; Joseph, J.; Desai, A.S.; Farkouh, M.E.; Hegde, S.M.; Hernandez, A.F.; McGeer, A.; Talbot, H.K.; et al. Effect of high-dose trivalent vs standard-dose quadrivalent influenza vaccine on mortality or cardiopulmonary hospitalization in patients with high-risk cardiovascular disease: A randomized clinical trial. JAMA 2021, 325, 39–49. [Google Scholar] [CrossRef]
- Frøbert, O.; Götberg, M.; Erlinge, D.; Akhtar, Z.; Christiansen, E.H.; MacIntyre, C.R.; Oldroyd, K.G.; Motovska, Z.; Erglis, A.; Moer, R.; et al. Influenza vaccination after myocardial infarction: A randomized, double-blind, placebo-controlled, multicenter trial. Circulation 2021. [Google Scholar] [CrossRef] [PubMed]
- Marra, F.; Zhang, A.; Gillman, E.; Bessai, K.; Parhar, K.; Vadlamudi, N.K. The protective effect of pneumococcal vaccination on cardiovascular disease in adults: A systematic review and meta-analysis. Int. J. Infect. Dis. 2020, 99, 204–213. [Google Scholar] [CrossRef] [PubMed]
- Marques Antunes, M.; Duarte, G.S.; Brito, D.; Borges, M.; Costa, J.; Ferreira, J.J.; Pinto, F.J.; Caldeira, D. Pneumococcal vaccination in adults at very high risk or with established cardiovascular disease: Systematic review and meta-analysis. Eur. Heart J. Qual. Care Clin. Outcomes 2021, 7, 97–106. [Google Scholar] [CrossRef]
- Christenson, B.; Hedlund, J.; Lundbergh, P.; Örtqvist, A. Additive preventive effect of influenza and pneumococcal vaccines in elderly persons. Eur. Respir. J. 2004, 23, 363–368. [Google Scholar] [CrossRef] [PubMed]
- Grohskopf, L.A.; Alyanak, E.; Broder, K.R.; Blanton, L.H.; Fry, A.M.; Jernigan, D.B.; Atmar, R.L. Prevention and control of seasonal influenza with vaccines: Recommendations of the Advisory Committee on Immunization Practices—United States, 2020–21 influenza season. MMWR Recomm Rep. 2020, 69, 1–24. [Google Scholar] [CrossRef] [PubMed]
- National Immunization Advisory Committee Technical Working Group Influenza Vaccination TWG. Technical guidelines for seasonal influenza vaccination in China (2020–2021). Zhonghua Liu Xing Bing Xue Za Zhi 2020, 41, 1555–1576. [Google Scholar]
- European Centre for Disease Prevention and Control Influenza Vaccination. Prevention and Control of Seasonal Influenza. Available online: http://ecdc.europa.eu/en/healthtopics/seasonal_influenza/vaccines/Pages/influenza_vaccination.aspx#vaccinationstrategies (accessed on 21 September 2021).
- Perk, J.; De Backer, G.; Gohlke, H.; Graham, I.; Reiner, Z.; Verschuren, M.; Albus, C.; Benlian, P.; Boysen, G.; Cifkova, R.; et al. European Guidelines on cardiovascular disease prevention in clinical practice (version 2012). The Fifth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice (constituted by representatives of nine societies and by invited experts). Eur. Heart J. 2012, 33, 1635–1701. [Google Scholar]
- Ponikowski, P.; Voors, A.A.; Anker, S.D.; Bueno, H.; Cleland, J.G.F.; Coats, A.J.S.; Falk, V.; Gonzalez-Juanatey, J.R.; Harjola, V.P.; Jankowska, E.A.; et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. Heart J. 2016, 37, 2129–2200. [Google Scholar]
- Cereda, A.; Conca, C.; Barbieri, L.; Ferrante, G.; Tumminello, G.; Lucreziotti, S.; Guazzi, M.; Mafrici, A. Acute myocarditis after the second dose of SARS-CoV-2 vaccine: Serendipity or atypical causal relationship? Anatol. J. Cardiol. 2021, 25, 522–523. [Google Scholar] [CrossRef]
- Ministère des Solidarités et de la Santé. Calendrier des Vaccinations et Recommandations Vaccinales 2021. Avril 2021. Available online: https://solidarites-sante.gouv.fr/IMG/pdf/calendrier_vaccinal_090721.pdf (accessed on 21 September 2021).
- Roncalli, J.; Roubille, F.; Lamblin, N.; Girerd, N.; Mouquet, F.; Chapet, N.; Roubille, C.; Berthelot, E.; Galois, K.; Battistella, P.; et al. Coronavirus disease vaccination in heart failure: No time to waste. Arch. Cardiovasc. Dis. 2021, 114, 434–438. [Google Scholar] [CrossRef] [PubMed]
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Marc, G.P.; Moreira, E.D.; Zerbini, C.; et al. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef]
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Diemert, D.; Spector, S.A.; Rouphael, N.; Creech, C.B.; et al. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N. Engl. J. Med. 2021, 384, 403–416. [Google Scholar] [CrossRef]
- Meylan, S.; Livio, F.; Foerster, M.; Genoud, P.J.; Marguet, F.; Wuerzner, G. Stage III hypertension in patients after mRNA-based SARS-CoV-2 vaccination. Hypertension 2021, 77, e56–e57. [Google Scholar] [CrossRef] [PubMed]
- Wicke, F.S.; Ditscheid, B.; Breitkreuz, T.; Glushan, A.; Lehmann, T.; Karimova, K.; Sawicki, O.A.; Vogel, M.; Freytag, A.; Beyer, M. Clinical and economic outcomes of a collaborative cardiology care program. Am. J. Manag. Care 2021, 27, e114–e122. [Google Scholar]
- Dexter, P.R.; Perkins, S.; Overhage, J.M.; Maharry, K.; Kohler, R.B.; McDonald, C.J. A computerized reminder system to increase the use of preventive care for hospitalized patients. N. Engl. J. Med. 2001, 345, 965–970. [Google Scholar] [CrossRef]
- Lawson, F.; Baker, V.; Au, D.; McElhaney, J.E. Standing orders for influenza vaccination increased vaccination rates in inpatient settings compared with community rates. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2000, 55, M522–M526. [Google Scholar] [CrossRef] [Green Version]
- Breuker, C.; Guedj, A.M.; Allan, M.; Coinus, L.; Molinari, N.; Chapet, N.; Roubille, F.; Le Quintrec, M.; Duhalde, V.; Jouglen, J.; et al. The COVID-19 Pandemic Led to a Small Increase in Changed Mentality Regarding Infection Risk without Any Change in Willingness to Be Vaccinated in Chronic Diseases Patients. J. Clin. Med. 2021, 10, 3967. [Google Scholar] [CrossRef]
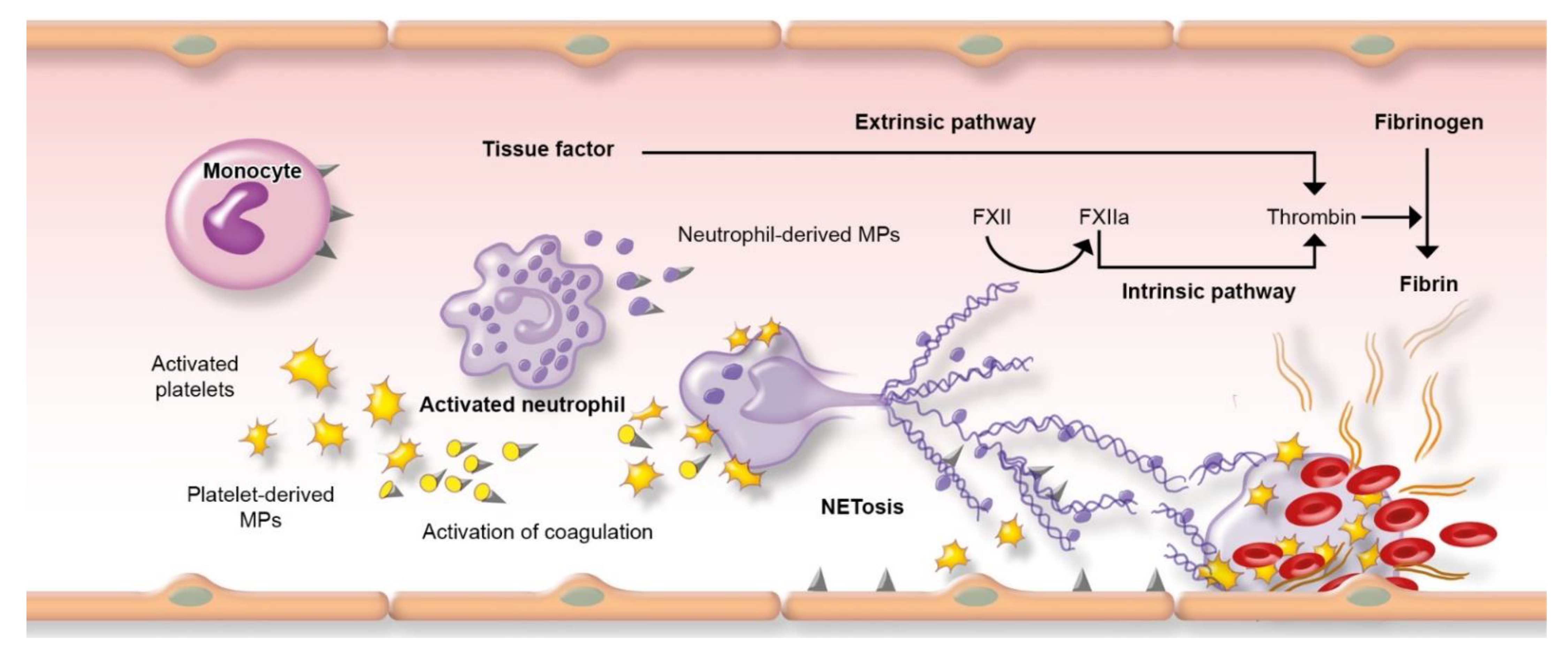
| Endothelial Cell Dysfunction and Activation |
|---|
| Platelet activation |
| Modulation of plasma coagulation |
| Augmented pro-coagulant functions—Tissue Factor-mediated activation of coagulation |
| Reduction of endogenous anticoagulants: Antithrombin, Tissue Factor pathwayinhibitor (TFPI); Protein C pathway |
| Inhibition of fibrinolytic activity |
| Hyperfibrinogenemia |
| Influenza Vaccine | Placebo or Control | Risk Ratio(95% CI) | |||
|---|---|---|---|---|---|
| Study | Events, n | Participants, n | Events, n | Participants, n | |
| Govaerts et al. (1994) | 7 | 927 | 5 | 911 | 1.38 (0.44–4.32) |
| FLUVACS trial (2004) | 32 | 145 | 54 | 147 | 0.60 (0.41–0.87) |
| FLUCAD trial (2008) | 16 | 325 | 30 | 333 | 0.55 (0.30–0.98) |
| De Villiers et al. (2011) | 20 | 1620 | 20 | 1622 | 1.00 (0.54–1.85) |
| Phrommintikul et al. (2011) | 20 | 221 | 42 | 218 | 0.47 (0.29–0.77) |
| Total | 95 | 3238 | 151 | 3231 | 0.64 (0.48–0.86) a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Girerd, N.; Chapet, N.; Roubille, C.; Roncalli, J.; Salvat, M.; Mouquet, F.; Lamblin, N.; Gueffet, J.-P.; Damy, T.; Galinier, M.; et al. Vaccination for Respiratory Infections in Patients with Heart Failure. J. Clin. Med. 2021, 10, 4311. https://doi.org/10.3390/jcm10194311
Girerd N, Chapet N, Roubille C, Roncalli J, Salvat M, Mouquet F, Lamblin N, Gueffet J-P, Damy T, Galinier M, et al. Vaccination for Respiratory Infections in Patients with Heart Failure. Journal of Clinical Medicine. 2021; 10(19):4311. https://doi.org/10.3390/jcm10194311
Chicago/Turabian StyleGirerd, Nicolas, Nicolas Chapet, Camille Roubille, Jérôme Roncalli, Muriel Salvat, Frédéric Mouquet, Nicolas Lamblin, Jean-Pierre Gueffet, Thibaud Damy, Michel Galinier, and et al. 2021. "Vaccination for Respiratory Infections in Patients with Heart Failure" Journal of Clinical Medicine 10, no. 19: 4311. https://doi.org/10.3390/jcm10194311
APA StyleGirerd, N., Chapet, N., Roubille, C., Roncalli, J., Salvat, M., Mouquet, F., Lamblin, N., Gueffet, J.-P., Damy, T., Galinier, M., Tartiere, J.-M., Janssen, C., Berthelot, E., Aguilhon, S., Escamilla, R., & Roubille, F. (2021). Vaccination for Respiratory Infections in Patients with Heart Failure. Journal of Clinical Medicine, 10(19), 4311. https://doi.org/10.3390/jcm10194311







