Inflammatory Bowel Disease and Neutrophil–Lymphocyte Ratio: A Systematic Scoping Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Protocol
2.2. Inclusion Criteria
2.3. Search Strategy
2.4. Article Review and Data Extraction
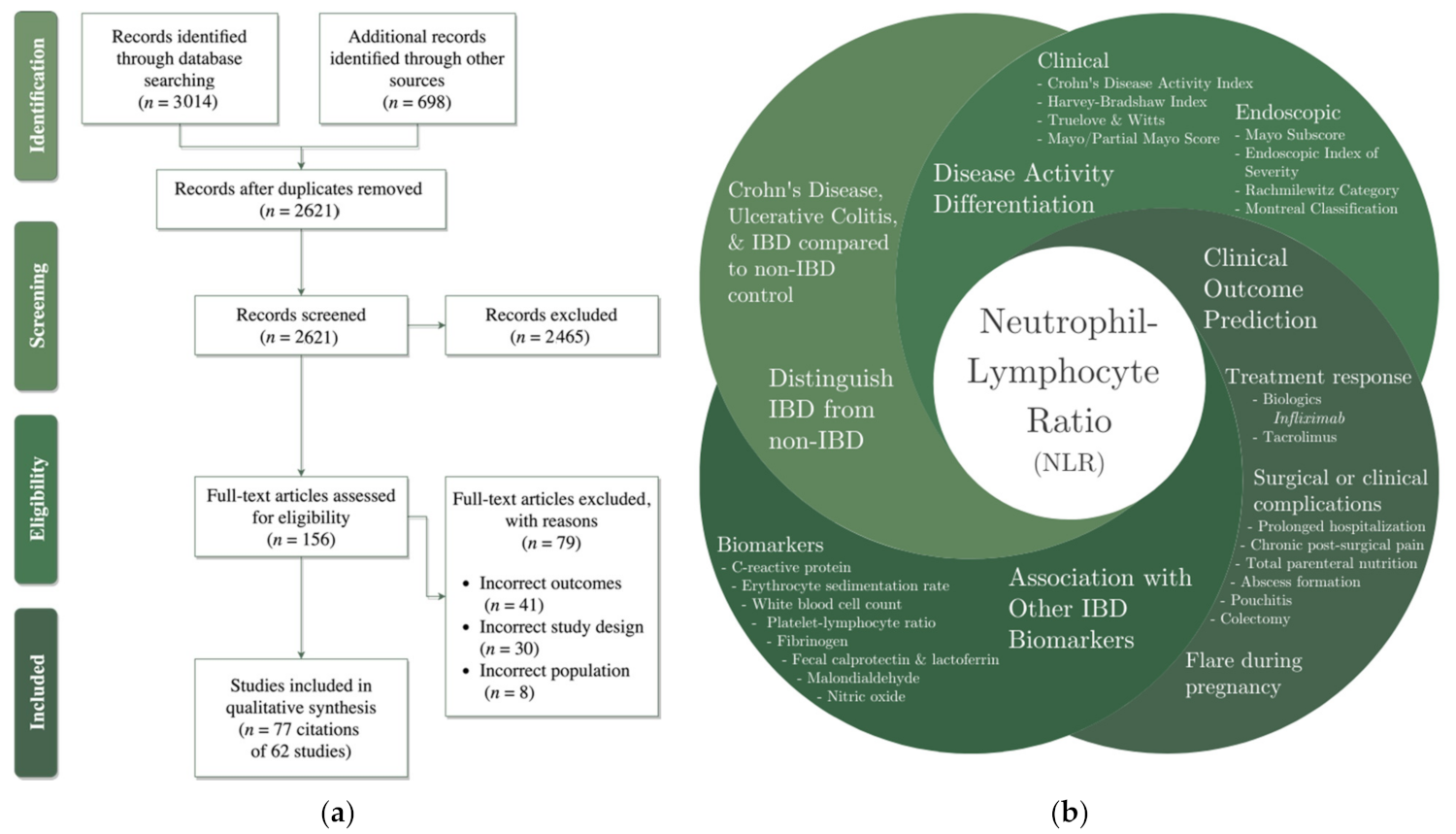
3. Results
3.1. Description of Studies
3.2. NLR to Distinguish IBD from Non-IBD
3.2.1. NLR Differences in IBD vs. Non-IBD
NLR Cutpoints to Distinguish IBD from Non-IBD
3.3. NLR to Differentiate Disease Activity in IBD
3.3.1. Relationship between NLR and Clinical Disease Activity
NLR Cutpoints to Differentiate Clinical Disease Activity
- Ben Jeddi et al. [73]—cutpoint at 1.57
- Zhang et al. (2017) [56]—cutpoint at 1.95 (95.5% sensitivity, 56.1% specificity)
- Eraldemir et al. (2016) [53]—cutpoint at 2.58 (69.6% sensitivity, 76.0% specificity)
- Acarturk et al. [35]—cutpoint at 3.2 (81.0% sensitivity, 59.0% specificity, p < 0.001)
- Chen et al. (2020) [37]—cutpoint at 3.32 (65.9% sensitivity, 75.9% specificity)
- Zhang et al. (2017) [56]—cutpoint at 5.35 to discriminate between mild-to-moderate and severe disease (75.0% sensitivity, 92.9% specificity, p = 0.02)
- Xu et al. [75] found no significance in any NLR cutpoint value to discriminate between active and inactive CD (AUC = 0.631)
- Hanafy et al. [82]—cutpoint at 2.35 (74.0% sensitivity, 86.0% specificity)
- Demir et al. [62]—cutpoint at 2.39 (48.6% sensitivity, 77.5% specificity)
- Chen et al. (2020) [37]—cutpoint at 2.40 (76.2% sensitivity, 84.5% specificity)
- Celikbilek et al. [60]—cutpoint at 2.47 (53.9% sensitivity, 63.2% specificity)
- Acarturk et al. [35]—cutpoint at 3.1 (78.0% sensitivity, 69.0% specificity, p < 0.001)
- Zhang et al. (2017) [56]—cutpoint at 3.29 (47.4% sensitivity, 93.9% specificity)
- Dong et al. [63]—cutpoint at 4.70 (61.0% sensitivity, 86.0% specificity)
3.3.2. Relationship between NLR and Endoscopic Disease Activity
NLR Cutpoints to Differentiate Endoscopic Disease Activity
3.4. NLR to Predict Clinical Outcomes
3.4.1. NLR and IBD Treatment
Shift in NLR and Prediction of Response to Biologics (Including Anti-TNF)
Glucocorticoids and Steroids
Other Treatments
3.4.2. NLR to Predict Length of Post-Operative Hospital Stay and IBD Complications
3.4.3. NLR to Predict Flare during Pregnancy
3.5. NLR and Other Biomarkers
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
References
- Alatab, S.; Sepanlou, S.G.; Ikuta, K.; Vahedi, H.; Bisignano, C.; Safiri, S.; Sadeghi, A.; Nixon, M.R.; Abdoli, A.; Abolhassani, H.; et al. The global, regional, and national burden of inflammatory bowel disease in 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study. Lancet Gastroenterol. Hepatol. 2019, 5, 17–30. [Google Scholar] [CrossRef]
- Beard, J.A.; Click, B.H. The burden of cost in inflammatory bowel disease: A medical economic perspective. Curr. Opin. Gastroenterol. 2020, 36, 310–316. [Google Scholar] [CrossRef]
- Park, K.T.; Ehrlich, O.G.; Allen, J.; Meadows, P.; Szigethy, E.M.; Henrichsen, K.; Kim, S.C.; Lawton, R.C.; Murphy, S.M.; Regueiro, M.; et al. The cost of inflammatory bowel disease: An initiative from the Crohn’s & Colitis Foundation. Inflamm. Bowel Dis. 2019, 26, 1–10. [Google Scholar] [CrossRef]
- Sewell, J.L.; Velayos, F.S. Systematic review: The role of race and socioeconomic factors on IBD healthcare delivery and effectiveness. Inflamm. Bowel Dis. 2013, 19, 627–643. [Google Scholar] [CrossRef]
- Park, M.D.; Bhattacharya, J.; Park, K. Differences in healthcare expenditures for inflammatory bowel disease by insurance status, income, and clinical care setting. PeerJ 2014, 2, e587. [Google Scholar] [CrossRef]
- Rajbhandari, R.; Blakemore, S.; Gupta, N.; Adler, A.J.; Noble, C.A.; Mannan, S.; Nikolli, K.; Yih, A.; Joshi, S.; Bukhman, G. Crohn’s disease in low and lower-middle income countries: A scoping review. World J. Gastroenterol. 2020, 26, 6891–6908. [Google Scholar] [CrossRef] [PubMed]
- Beilman, C.L.; Thanh, N.; Ung, V.; Ma, C.; Wong, K.; Kroeker, K.I.; Lee, T.; Wang, H.; Ohinmaa, A.; Jacobs, P.; et al. Real-life treatment paradigms show adalimumab is cost-effective for the management of ulcerative colitis. Can. J. Gastroenterol. Hepatol. 2016, 2016, 1–11. [Google Scholar] [CrossRef]
- Yu, H.; MacIsaac, D.; Wong, J.J.; Sellers, Z.M.; Wren, A.A.; Bensen, R.; Kin, C.; Park, K.T. Market share and costs of biologic therapies for inflammatory bowel disease in the USA. Aliment. Pharmacol. Ther. 2017, 47, 364–370. [Google Scholar] [CrossRef] [PubMed]
- Orlando, A.; Guglielmi, F.W.; Cottone, M.; Orlando, E.; Romano, C.; Sinagra, E. Clinical implications of mucosal healing in the management of patients with inflammatory bowel disease. Dig. Liver Dis. 2013, 45, 986–991. [Google Scholar] [CrossRef] [PubMed]
- Guthrie, G.J.; Charles, K.A.; Roxburgh, C.S.; Horgan, P.G.; McMillan, D.; Clarke, S.J. The systemic inflammation-based neutrophil–lymphocyte ratio: Experience in patients with cancer. Crit. Rev. Oncol. 2013, 88, 218–230. [Google Scholar] [CrossRef] [PubMed]
- Weissman, S.; Sinh, P.; Mehta, T.; Thaker, R.K.; Derman, A.; Heiberger, C.; Qureshi, N.; Amrutiya, V.; Atoot, A.; Dave, M.; et al. Atherosclerotic cardiovascular disease in inflammatory bowel disease: The role of chronic inflammation. World J. Gastrointest. Pathophysiol. 2020, 11, 104–113. [Google Scholar] [CrossRef]
- Chin, A.C.; Parkos, C.A. Neutrophil transepithelial migration and epithelial barrier function in IBD: Potential targets for inhibiting neutrophil trafficking. Ann. N. Y. Acad. Sci. 2006, 1072, 276–287. [Google Scholar] [CrossRef]
- Zhou, G.X.; Liu, Z.J. Potential roles of neutrophils in regulating intestinal mucosal inflammation of inflammatory bowel disease. J. Dig. Dis. 2017, 18, 495–503. [Google Scholar] [CrossRef]
- Moran, C.P.; Neary, B.; Doherty, G. Endoscopic evaluation in diagnosis and management of inflammatory bowel disease. World J. Gastrointest. Endosc. 2016, 8, 723–732. [Google Scholar] [CrossRef] [PubMed]
- Soubières, A.A.; Poullis, A. Emerging biomarkers for the diagnosis and monitoring of inflammatory bowel diseases. Inflamm. Bowel Dis. 2016, 22, 2016–2022. [Google Scholar] [CrossRef] [PubMed]
- Baron, J.H.; Connell, A.M.; Lennard-Jones, J.E. Variation between observers in describing mucosal appearances in proctocolitis. BMJ 1964, 1, 89–92. [Google Scholar] [CrossRef] [PubMed]
- Marlicz, W.; Skonieczna-Żydecka, K.; Dabos, K.J.; Łoniewski, I.; Koulaouzidis, A. Emerging concepts in non-invasive monitoring of Crohn’s disease. Ther. Adv. Gastroenterol. 2018, 11, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Chebli, J.M.F.; Queiroz, N.S.F.; Damião, A.O.M.C.; Chebli, L.A.; Costa, M.H.D.M.; Parra, R.S. How to manage inflammatory bowel disease during the COVID-19 pandemic: A guide for the practicing clinician. World J. Gastroenterol. 2021, 27, 1022–1042. [Google Scholar] [CrossRef]
- Lewis, J.D. The utility of biomarkers in the diagnosis and therapy of inflammatory bowel disease. Gastroenterology 2011, 140, 1817–1826.e2. [Google Scholar] [CrossRef]
- Bromke, M.; Neubauer, K.; Kempiński, R.; Krzystek-Korpacka, M. Faecal calprotectin in assessment of mucosal healing in adults with inflammatory bowel disease: A meta-analysis. J. Clin. Med. 2021, 10, 2203. [Google Scholar] [CrossRef]
- Dignass, A.U.; Gasche, C.; Bettenworth, D.; Birgegård, G.; Danese, S.; Gisbert, J.P.; Gomollon, F.; Iqbal, T.; Katsanos, K.; Koutroubakis, I.; et al. European consensus on the diagnosis and management of iron deficiency and anaemia in inflammatory bowel diseases. J. Crohn’s Colitis 2015, 9, 211–222. [Google Scholar] [CrossRef]
- Mosli, M.; Zou, G.; Garg, S.K.; Feagan, S.G.; MacDonald, J.K.; Chande, N.; Sandborn, W.J.; Feagan, B.G. C-Reactive protein, fecal calprotectin, and stool lactoferrin for detection of endoscopic activity in symptomatic inflammatory bowel disease patients: A systematic review and meta-analysis. Am. J. Gastroenterol. 2015, 110, 802–819. [Google Scholar] [CrossRef]
- Fengming, Y.; Jianbing, W. Biomarkers of inflammatory bowel disease. Dis. Markers 2014, 2014, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, F.S.; Burri, E.; Beglinger, C. The role and utility of faecal markers in inflammatory bowel disease. Ther. Adv. Gastroenterol. 2014, 8, 23–36. [Google Scholar] [CrossRef] [PubMed]
- Titz, B.; Gadaleta, R.M.; Sasso, G.L.; Elamin, A.; Ekroos, K.; Ivanov, N.V.; Peitsch, M.C.; Hoeng, J. Proteomics and lipidomics in inflammatory bowel disease research: From mechanistic insights to biomarker identification. Int. J. Mol. Sci. 2018, 19, 2775. [Google Scholar] [CrossRef] [PubMed]
- Denson, L.A.; Curran, M.; McGovern, D.P.B.; Koltun, W.A.; Duerr, R.H.; Kim, S.C.; Sartor, R.B.; Sylvester, F.A.; Abraham, C.; De Zoeten, E.F.; et al. Challenges in IBD research: Precision medicine. Inflamm. Bowel Dis. 2019, 25, S31–S39. [Google Scholar] [CrossRef]
- Erre, G.L.; Paliogiannis, P.; Castagna, F.; Mangoni, A.A.; Carru, C.; Passiu, G.; Zinellu, A. Meta-analysis of neutrophil-to-lymphocyte and platelet-to-lymphocyte ratio in rheumatoid arthritis. Eur. J. Clin. Investig. 2018, 49, e13037. [Google Scholar] [CrossRef]
- Angkananard, T.; Anothaisintawee, T.; McEvoy, M.; Attia, J.; Thakkinstian, A. Neutrophil lymphocyte ratio and cardiovascular disease risk: A systematic review and meta-analysis. BioMed Res. Int. 2018, 2018, 1–11. [Google Scholar] [CrossRef]
- Tan, T.P.; Arekapudi, A.; Metha, J.; Prasad, A.; Venkatraghavan, L. Neutrophil-lymphocyte ratio as predictor of mortality and morbidity in cardiovascular surgery: A systematic review. ANZ J. Surg. 2015, 85, 414–419. [Google Scholar] [CrossRef]
- Wang, L.; Song, Q.; Wang, C.; Wu, S.; Deng, L.; Li, Y.; Zheng, L.; Liu, M. Neutrophil to lymphocyte ratio predicts poor outcomes after acute ischemic stroke: A cohort study and systematic review. J. Neurol. Sci. 2019, 406, 116445. [Google Scholar] [CrossRef]
- Liu, C.-C.; Ko, H.-J.; Liu, W.-S.; Hung, C.-L.; Hu, K.-C.; Yu, L.-Y.; Shih, S.-C. Neutrophil-to-lymphocyte ratio as a predictive marker of metabolic syndrome. Medicine 2019, 98, e17537. [Google Scholar] [CrossRef] [PubMed]
- Yin, X.; Wu, L.; Yang, H.; Yang, H. Prognostic significance of neutrophil–lymphocyte ratio (NLR) in patients with ovarian cancer. Medicine 2019, 98, e17475. [Google Scholar] [CrossRef]
- Mellor, K.L.; Powell, A.G.M.T.; Lewis, W.G. Systematic review and meta-analysis of the prognostic significance of neutrophil-lymphocyte ratio (NLR) after R0 gastrectomy for cancer. J. Gastrointest. Cancer 2018, 49, 237–244. [Google Scholar] [CrossRef]
- Li, X.; Liu, C.; Mao, Z.; Xiao, M.; Wang, L.; Qi, S.; Zhou, F. Predictive values of neutrophil-to-lymphocyte ratio on disease severity and mortality in COVID-19 patients: A systematic review and meta-analysis. Crit. Care 2020, 24, 1–10. [Google Scholar] [CrossRef]
- Acarturk, G.; Acay, A.; Demir, K.; Ulu, M.S.; Ahsen, A.; Yuksel, S. Neutrophil-to-lymphocyte ratio in inflammatory bowel disease—As a new predictor of disease severity. Bratisl. Lek. Listy 2015, 116, 213–217. [Google Scholar] [CrossRef]
- Ahmad, S.M. Adenosine deaminase activity and peripheral immune cells ratios in a sample of inflammatory bowel disease patients. Mustansiriya Med. J. 2018, 14, 4. [Google Scholar]
- Chen, Y.-H.; Wang, L.; Feng, S.-Y.; Cai, W.-M.; Chen, X.-F.; Huang, Z.-M. The relationship between C-reactive Protein/Albumin ratio and disease activity in patients with inflammatory bowel disease. Gastroenterol. Res. Pract. 2020, 2020, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Munn, Z.; Peters, M.D.J.; Stern, C.; Tufanaru, C.; McArthur, A.; Aromataris, E. Systematic review or scoping review? Guidance for authors when choosing between a systematic or scoping review approach. BMC Med. Res. Methodol. 2018, 18, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Arksey, H.; O’Malley, L. Scoping studies: Towards a methodological framework. Int. J. Soc. Res. Methodol. 2005, 8, 19–32. [Google Scholar] [CrossRef]
- Ricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.; Horsley, T.; Weeks, L.; et al. PRISMA extension for scoping reviews (PRISMA-ScR): Checklist and explanation. Ann. Intern. Med. 2018, 169, 467–473. [Google Scholar] [CrossRef]
- Clarivate Analytics. EndNote; Clarivate Analytics: Philadelphia, PA, USA, 2013. [Google Scholar]
- Veritas Health Innovation. Covidence Systematic Review Software; Veritas Health Innovation: Melbourne, Australia. Available online: www.covidence.org.
- Microsoft. Microsoft Excel; Microsoft: Redmond, DC, USA, 2021. [Google Scholar]
- Mary, J.Y.; Modigliani, R. Development and validation of an endoscopic index of the severity for Crohn’s disease: A prospective multicentre study. Groupe d’Etudes Therapeutiques des Affections Inflammatoires du Tube Digestif (GETAID). Gut 1989, 30, 983–989. [Google Scholar] [CrossRef]
- Bernstein, C.N.; Eliakim, A.; Fedail, S.; Fried, M.; Gearry, R.; Goh, K.-L.; Hamid, S.; Khan, A.G.; Khalif, I.; Ng, S.C.; et al. World gastroenterology organisation global guidelines inflammatory bowel disease. J. Clin. Gastroenterol. 2016, 50, 803–818. [Google Scholar] [CrossRef]
- Naber, A.H.J.; De Jong, D.J. Assessment of disease activity in inflammatory bowel disease; relevance for clinical trials. Neth. J. Med. 2003, 61, 105–110. [Google Scholar]
- Pabla, B.S.; Schwartz, D.A. Assessing severity of disease in patients with ulcerative colitis. Gastroenterol. Clin. N. Am. 2020, 49, 671–688. [Google Scholar] [CrossRef]
- Spekhorst, L.M.; Visschedijk, M.C.; Alberts, R.; Festen, E.A.; Van Der Wouden, E.-J.; Dijkstra, G.; Weersma, R.K. Performance of the Montreal classification for inflammatory bowel diseases. World J. Gastroenterol. 2014, 20, 153–174. [Google Scholar] [CrossRef] [PubMed]
- Mandrekar, J.N. Receiver operating characteristic curve in diagnostic test assessment. J. Thorac. Oncol. 2010, 5, 1315–1316. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.-S.; Gao, N.; Sun, X.; Jiang, X.-Y.; Chen, J.-J.; Mao, Q.-Q.; Zhong, L. C-reactive protein/abumin ratio is a useful biomarker for predicting the mucosal healing in the Crohn disease: A retrospective study. Medicine 2021, 100, e24925. [Google Scholar] [CrossRef] [PubMed]
- Jaoude, J.B.; Bakouny, Z.; Hallit, R.; Honein, K.; Ghorra, C.; El Rassy, E. Platelet-to-lymphocyte and neutrophil-to-lymphocyte ratios in Crohn’s disease: The controversy remains. Clin. Res. Hepatol. Gastroenterol. 2018, 42, e16–e18. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Xiong, D.; Jiang, Z.; Zhang, C.; Hu, Q. Diagnostic Value of Platelet-to-Lymphocyte Ratio and Neutrophilto-Lymphocyte Ratio in Crohn’s Disease; Baishideng Publishing Group Inc.: Pleasanton, CA, USA, 2018; pp. 93–98. [Google Scholar]
- Eraldemir, F.C.; Musul, M.; Duman, A.; Oztas, B.; Baydemir, C.; Hulagu, S. The relationship between neutrophil/lymphocyte and platelet/lymphocyte ratios with oxidative stress in active Crohn’s disease patients. Hippokratia 2016, 20, 368. [Google Scholar] [PubMed]
- Feng, J.-R.; Qiu, X.; Wang, F.; Chen, P.-F.; Gao, Q.; Peng, Y.-N.; Lin, X.; Liu, Q.; Liu, J.; Zhao, Q.; et al. Diagnostic value of neutrophil-to-lymphocyte ratio and platelet-to-lymphocyte ratio in Crohns disease. Gastroenterol. Res. Pract. 2017, 2017, 1–5. [Google Scholar] [CrossRef]
- Gao, S.-Q.; Huang, L.-D.; Dai, R.-J.; Chen, N.-D.; Hu, W.-J.; Shan, Y.-F. Neutrophil-lymphocyte ratio: A controversial marker in predicting Crohn’s disease severity. Int. J. Clin. Exp. Pathol. 2015, 8, 14779–14785. [Google Scholar]
- Zhang, S.; Wu, Z.; Li, J.; Wen, X.; Li, L.; Liu, C.; Zhang, F.; Qian, J.; Li, Y. Peripheral differentials by Cytodiff flow cytometric system predict disease activity in Chinese patients with inflammatory bowel disease. Clin. Chim. Acta 2017, 471, 17–22. [Google Scholar] [CrossRef]
- Zhang, M.-H.; Wang, H.; Wang, H.-G.; Wen, X.; Yang, X.-Z. Effective immune-inflammation index for ulcerative colitis and activity assessments. World J. Clin. Cases 2021, 9, 334–343. [Google Scholar] [CrossRef] [PubMed]
- Michalak, A.; Radwan, P.; Kasztelan-Szczerbińska, B.; Laskowska, K.; Cichoż-Lach, H. Neutrophil-to-platelet ratio, platelet-to-lymphocyte ratio and mean platelet volume-to platelet ratio as potential hematological indices in the evaluation of inflammation in ulcerative colitis patients. United Eur. Gastroenterol. J. 2019, 7, 608–609. [Google Scholar]
- Akpinar, M.Y.; Ozin, Y.O.; Kaplan, M.; Ates, I.; Kalkan, I.H.; Kilic, Z.M.Y.; Yuksel, M.; Kayacetin, E. Platelet-to-lymphocyte ratio and neutrophil-to-lymphocyte ratio predict mucosal disease severity in ulcerative colitis. J. Med Biochem. 2018, 37, 155–162. [Google Scholar] [CrossRef]
- Celikbilek, M.; Dogan, S.; Ozbakır, O.; Zararsız, G.; Kücük, H.; Gürsoy, S.; Yurci, A.; Güven, K.; Yücesoy, M. Neutrophil-lymphocyte ratio as a predictor of disease severity in ulcerative colitis. J. Clin. Lab. Anal. 2013, 27, 72–76. [Google Scholar] [CrossRef] [PubMed]
- Cherfane, C.E.; Gessel, L.; Cirillo, D.; Zimmerman, M.B.; Polyak, S. Monocytosis and a low lymphocyte to monocyte ratio are effective biomarkers of ulcerative colitis disease activity. inflamm. Bowel Dis. 2015, 21, 1769–1775. [Google Scholar] [CrossRef] [PubMed]
- Demir, A.K.; Demirtas, A.; Kaya, S.U.; Tastan, I.; Butun, I.; Sagcan, M.; Sahin, S.; Tasliyurt, T.; Yilmaz, A. The relationship between the neutrophil–lymphocyte ratio and disease activity in patients with ulcerative colitis. Kaohsiung J. Med Sci. 2015, 31, 585–590. [Google Scholar] [CrossRef]
- Dong, Y.; Zhang, M.; Zhang, F.; Wang, B.; Feng, B. Clinical Significance of NLR and MLR in Ulcerative Colitis. Chin. J. Gastroenterol. 2019, 24, 729–733. [Google Scholar]
- Eraldemir, C. Neutrophil-lymphocyte and platelet-lymphocyte ratios, MDA, NO and SOD levels in ulcerative colitis; Is there any relationship? Clin. Chem. Lab. Med. 2014, 52, S996. [Google Scholar]
- Hanai, H.; Takeuchi, K.; Iida, T.; Kashiwagi, N.; Saniabadi, A.R.; Matsushita, I.; Sato, Y.; Kasuga, N.; Nakamura, T. Relationship between fecal calprotectin, intestinal inflammation, and peripheral blood neutrophils in patients with active ulcerative colitis. Dig. Dis. Sci. 2004, 49, 1438–1443. [Google Scholar] [CrossRef] [PubMed]
- Jeong, Y.; Jeon, S.R.; Kim, H.G.; Moon, J.R.; Lee, T.H.; Jang, J.Y.; Cho, J.-H.; Park, J.S.; Park, H.; Lee, K.-H.; et al. The role of platelet to lymphocyte ratio and neutrophil to lymphocyte ratio in ulcerative colitis. Intest. Res. 2021, 19, 62–70. [Google Scholar] [CrossRef]
- Okba, A.M.; Amin, M.M.; Abdelmoaty, A.S.; Ebada, H.; Kamel, A.H.; Allam, A.S.; Sobhy, O.M. Neutrophil/lymphocyte ratio and lymphocyte/monocyte ratio in ulcerative colitis as non-invasive biomarkers of disease activity and severity. Autoimmun. Highlights 2019, 10, 4–9. [Google Scholar] [CrossRef]
- Torun, S.; Tunc, B.D.; Suvak, B.; Yildiz, H.; Tas, A.; Sayilir, A.; Ozderin, Y.O.; Beyazit, Y.; Kayacetin, E. Assessment of neutrophil-lymphocyte ratio in ulcerative colitis: A promising marker in predicting disease severity. Clin. Res. Hepatol. Gastroenterol. 2012, 36, 491–497. [Google Scholar] [CrossRef]
- Chalmers, I.; Wilson, D.; Henderson, P. P167 Diagnostic accuracy of neutrophil-lymphocyte ratio in suspected paediatric inflammatory bowel disease. J. Crohn’s Colitis 2017, 11, S161–S162. [Google Scholar] [CrossRef][Green Version]
- Guthrie, G.; McMillan, D.; Foulis, A.; Horgan, P.; Roxburgh, C. Cancer-associated inflammation in a cohort of colorectal cancer patients with inflammatory bowel disease. Gastroenterology 2013, 144, S1070. [Google Scholar] [CrossRef]
- Ndulue, C.; Obiajunwa, C.; Ndulue, J. Leukocyte ratios in adults 20–70 years old with inflammatory bowel disease in the United States population: Nhanes 2009–2010. Am. J. Gastroenterol. 2019, 114, S431–S432. [Google Scholar] [CrossRef]
- Jeong, S. The usefulness of neutrophil to lymphocyte ratio, platelet to lymphocyte ratio, and fecal calprotectin as disease activity markers in inflammatory bowel disease. J. Gastroenterol. Hepatol. 2018, 33, 475. [Google Scholar]
- Ben Jeddi, H.; Kchir, H.; Hassine, A.; Issaoui, D.; Chaabouni, H.; Maamouri, N. P215 The mean platelet volume compared with other serum biomarkers: Is it predictive of activity of Crohn’s disease? J. Crohn’s Colitis 2019, 13, S203. [Google Scholar] [CrossRef]
- Gold, S.L.; Gordon, B.L.; Schneider, Y.; Dixon, R.E.; Scherl, E.J.; Steinlauf, A.F. Su1922 neutrophil to lymphocyte ratio (NLR) and platelet to lymphocyte ratio (PLR): Novel predictors of disease activity in patients with inflammatory bowel disease. Gastroenterology 2020, 158, S703. [Google Scholar] [CrossRef]
- Xu, M.; Cen, M.; Chen, X.; Chen, H.; Liu, X.; Cao, Q. Correlation between serological biomarkers and disease activity in patients with inflammatory bowel disease. BioMed Res. Int. 2019, 2019, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Posul, E.; Yilmaz, B.; Aktas, G.; Kurt, A.P.M. Does neutrophil-to-lymphocyte ratio predict active ulcerative colitis? Wien. Klin. Wochenschr. 2015, 127, 262–265. [Google Scholar] [CrossRef]
- Fidan, K.; Kocak, M.Z. Assessment of platelet-to-lymphocyte ratio and neutrophil-to-lymphocyte ratio in ulcerative colitis: A retrospective study. Eurasian J. Med. Oncol. 2017, 1, 224–227. [Google Scholar]
- Yamamoto-Furusho, J.K.; Mendieta-Escalante, E.A. Diagnostic utility of the neutrophil-platelet ratio as a novel marker of activity in patients with Ulcerative Colitis. PLoS ONE 2020, 15, e0231988. [Google Scholar] [CrossRef] [PubMed]
- Ovidiu, F.; Gratiela, A.; Alexandru, G.; Ilias, T. Neutrophil to lymphocyte ratio: Predictive marker for assessing the severity of ulcerative colitis? J. Gastrointest. Liver Dis. 2017, 26, 131. [Google Scholar]
- Nishida, Y.; Hosomi, S.; Yamagami, H.; Yukawa, T.; Otani, K.; Nagami, Y.; Tanaka, F.; Taira, K.; Kamata, N.; Tanigawa, T.; et al. Neutrophil-to-lymphocyte ratio for predicting loss of response to infliximab in ulcerative colitis. PLoS ONE 2017, 12, e0169845. [Google Scholar] [CrossRef]
- Abedi, M.; anesh, N.; Ostadrahimi, A.; Somi, M.; Abedi Manesh, S. Effect of vitamin a supplementation on inflammatory cells in patients with ulcerative colitis. Inflamm. Res. 2011, 60, S244. [Google Scholar]
- Hanafy, A.S.; Monir, M.H.; Abdel Malak, H.; Desoky Aiad, M. A simple noninvasive score predicts disease activity and deep remission in ulcerative colitis. Inflamm. Intest. Dis. 2018, 3, 16–24. [Google Scholar] [CrossRef]
- Crispino, F.; Grova, M.; Maida, M.; Renna, S.; Mocciaro, F.; Casà, A.; Rizzuto, G.; Tesè, L.; Scimeca, D.; Di Mitri, R.; et al. Blood-based prognostic biomarkers in Crohn’s disease patients on biologics: A promising tool to predict endoscopic outcomes. Expert Opin. Biol. Ther. 2021, 21, 1133–1141. [Google Scholar] [CrossRef]
- Kang, W.-M.; Zhu, C.-Z.; Yang, X.-X.; Yu, J.-C.; Ma, Z.-Q.; Ye, X.; Li, K.; Liu, D. Application of the Onodera prognostic nutrition index and neutrophil-to-lymphocyte ratio in risk evaluation of postoperative complications in Crohn’s disease. Sci. Rep. 2017, 7, 1–11. [Google Scholar] [CrossRef]
- Nassri, A.B.; Muftah, M.; Nassri, R.; Fialho, A.; Fialho, A.; Ribeiro, B.; Ghali, P. Novel inflammatory-nutritional biomarkers as predictors of histological activity in Crohn’s disease. Clin. Lab. 2020, 66. [Google Scholar] [CrossRef]
- Dorobăţ, A.G. Can the neutrophil-to-lymphocyte and platelet-tolymphocyte ratios predict disease activity in patients with ulcerative colitis? J. Gastrointest. Liver Dis. 2018, 27, 9. [Google Scholar]
- Bertani, L.; Rossari, F.; Barberio, B.; Demarzo, M.G.; Tapete, G.; Albano, E.; Svizzero, G.B.; Ceccarelli, L.; Mumolo, M.G.; Brombin, C.; et al. Novel prognostic biomarkers of mucosal healing in ulcerative colitis patients treated with anti-TNF: Neutrophil-to-Lymphocyte ratio and platelet-to-lymphocyte ratio. Inflamm. Bowel Dis. 2020, 26, 1579–1587. [Google Scholar] [CrossRef] [PubMed]
- Bertani, L.; De Bortoli, N.; Tapete, G.; Albano, E.; Mazzanti, R.; Mumolo, M.; Ceccarelli, L.; Di Fluri, G.; Svizzero, G.B.; Bellini, M.; et al. Two new prognostic markers of mucosal healing in patients treated with anti-TNF for ulcerative colitis: The neutrophil-to-lymphocyte ratio and the platelet-to-lymphocyte ratio. Dig. Liver Dis. 2019, 51, e216. [Google Scholar] [CrossRef]
- Jardak, S.; Medhioub, M.; Agar, K.; Hamzaoui, L.; Khsiba, A.; Azouz, M. Ulcerative colitis: Role of neutrophil-to-lymphocyte and platelet-to-lymphocyte ratios in predicting disease severity. J. Crohn’s Colitis 2018, 12, S263. [Google Scholar] [CrossRef]
- Wlodarczyk, M.K.; Sobolewska, A.E.; Stec-Michalska, K.; Fichna, J.J.; Wisniewska-Jarosinska, M.E. Neutrophil-lymphocyte ratio in Crohn’s disease patients predicts sustained response to infliximab 52-week therapy. J. Gastrointest. Liver Dis. 2015, 24, 127–128. [Google Scholar]
- Ben Mustapha, N.; Mahmoudi, M.; Bejaoui, M.; Laabidi, A.; Serghini, M.; Kallel, L. Mean platelet volume and neutrophil-to-lymphocyte ratio as new biomarkers of predicting response to infliximab therapy in Crohn’s disease patients. J. Crohn’s Colitis 2015, 9, S399. [Google Scholar]
- Gao, Q.; Dong, H.; Li, J. Neutrophil-lymphocyte ratio at 14th week predicts loss of response to 52-week infliximab therapy in patients with Crohn’s disease. Nan Fang Yi Ke Da Xue Xue Bao 2020, 40, 453–458. [Google Scholar] [CrossRef]
- Andrew, B.; Con, D.; Van Langenberg, D.; Vasudevan, A. C -reactive protein-albumin ratio, but not neutrophil-lymphocyte or platelet-lymphocyte ratios, predicts outcomes after infliximab salvage therapy for acute severe ulcerative colitis. J. Gastroenterol. Hepatol. 2020, 35, 148. [Google Scholar]
- Stefanidis, E.; Katsinelos, P.; Papanikolaou, N.; Anastasiadou, K.; Kountouras, J. The effect of infliximab therapy on the neutrophil-lymphocyte and platelet-lymphocyte ratios in inflammatory bowel disease patients. J. Crohn’s Colitis 2015, 9, S376. [Google Scholar]
- Gordon, B.; Gold, S.; Scherl, E.; Steinlauf, A. Neutrohil-to-lymphocyte ratio is associated with disease activity in inflammatory bowel disease patients regardless of corticosteroid use. Gastroenterology 2020, 158, S697. [Google Scholar] [CrossRef]
- Nishida, Y.; Hosomi, S.; Yamagami, H.; Sugita, N.; Itani, S.; Yukawa, T.; Otani, K.; Nagami, Y.; Tanaka, F.; Taira, K.; et al. Pretreatment neutrophil-to-lymphocyte ratio predicts clinical relapse of ulcerative colitis after tacrolimus induction. PLoS ONE 2019, 14, e0213505. [Google Scholar] [CrossRef]
- Ryan, J.J.; A Hanes, D.; Bradley, R.D.; Contractor, N. Effect of a nutrition support formula in adults with inflammatory bowel disease: A pilot study. Glob. Adv. Health Med. 2019, 8, 1–8. [Google Scholar] [CrossRef]
- Khoury, T.; Daher, S.; Massarwa, M.; Hakimian, D.; A Benson, A.; Viener, E.; Farah, R.; Mari, A.; Hazou, W.; Kadah, A.; et al. A validated score assessing the risk of an intra-abdominal abscess in patients with Crohn’s disease presenting at the emergency department. J. Crohn’s Colitis 2019, 13, 1131–1137. [Google Scholar] [CrossRef] [PubMed]
- Argeny, S.; Stift, A.; Bergmann, M.; Mittlböck, M.; Maschke, S.; Yang, Y.; Chitsabesan, P.; Riss, S. Prognostic value of preoperative neutrophil-to-lymphocyte ratio in Crohn’s disease. Wien. Klin. Wochenschr. 2018, 130, 398–403. [Google Scholar] [CrossRef] [PubMed]
- Gur, E.O.; Karaisli, S.; Dilek, O.N.; Acar, T.; Haciyanli, S.; Unsal, B.; Haciyanli, M. Reliability of blood tests, neutrophil lymphocyte ratio (NLR) and Glasgow prognostic index (GPI) to evaluate the severity of the Crohn’s disease. J. Crohn’s Colitis 2018, 12, S440. [Google Scholar] [CrossRef]
- Gur, E.; Yuksel, E.; Haciyanli, S.; Cekic, C.; Dilek, O.; Haciyanli, M. The predictive value of laboratory factors in evaluating the necessity of surgicaltreatment in crohn’s disease. Ann. Med. Res. 2020, 27, 105–109. [Google Scholar] [CrossRef]
- Abotaga, S.A.; Oliner, C.; Goodman, A.J.; Azab, B.; Weiserbs, K.; Agarwal, M.; Koczka, C.P.; Purow, E.; Gress, F.G. Value of the neutrophil to lymphocyte ratio (NLR) as a predictive tool of disease severity and length of hospitalizations in ulcerative colitis. Gastroenterology 2009, 136, A667–A668. [Google Scholar] [CrossRef]
- Con, D.; Andrew, B.; Nicolaides, S.; van Langenberg, D.R.; Vasudevan, A. Biomarker dynamics during infliximab salvage for acute severe ulcerative colitis: C-reactive protein (CRP)-lymphocyte ratio and CRP-albumin ratio are useful in predicting colectomy. Intest. Res. 2021, 1–22. [Google Scholar] [CrossRef]
- Nishida, Y.; Hosomi, S.; Yamagami, H.; Fujimoto, K.; Nakata, R.; Itani, S.; Nadatani, Y.; Fukunaga, S.; Otani, K.; Tanaka, F.; et al. Novel prognostic biomarkers of pouchitis after ileal pouch-anal anastomosis for ulcerative colitis: Neutrophil-to-lymphocyte ratio. PLoS ONE 2020, 15, e0241322. [Google Scholar] [CrossRef]
- Fleshner, P.; Zaghiyan, K.; Kamdjou, T. Preoperative neutrophil to lymphocyte ratio in ulcerative colitis is associated with denovo Crohn’s disease after ileal pouch-anal anastomosis. Am. J. Gastroenterol. 2019, 114, S24. [Google Scholar]
- Parisi, N. Impact of pro-inflammatory states on chronic post-surgical pain after fast-track laparoscopic colectomy. Reg. Anesth. Pain Med. 2013, 38, E129. [Google Scholar]
- Yarur, A.J.; Sussman, D.A.; Deshpande, A.R.; Pechman, D.M.; Czul, F.; Rueda, S.; Abreu, M.T. Neutrophil-to-lymphocyte ratio as a predictor of surgical complications in inflammatory bowel disease. Gastroenterology 2011, 140. [Google Scholar] [CrossRef]
- El-Sadek, H. Could neutrophil-to-lymphocyte ratio predict ulcerative colitis flare during pregnancy? United Eur. Gastroenterol. J. 2019, 7, 738–739. [Google Scholar]
- Messner, Z.; Emminger, R.; Kobleder, I.; Auzinger, C.; Renner, F. Fecal calprotectin in relation to established and putative systemic inflammatory biomarkers in inflammatory bowel disease—A single center experience. Z. Für Gastroenterol. 2016, 54, P05. [Google Scholar] [CrossRef]
- Forget, P.; Khalifa, C.; Defour, J.-P.; Latinne, D.; Van Pel, M.-C.; De Kock, M. What is the normal value of the neutrophil-to-lymphocyte ratio? BMC Res. Notes 2017, 10, 1–4. [Google Scholar] [CrossRef]
- Moosazadeh, M.; Maleki, I.; Alizadeh-Navaei, R.; Kheradmand, M.; Hedayatizadeh-Omran, A.; Shamshirian, A.; Barzegar, A. Normal values of neutrophil-to-lymphocyte ratio, lymphocyte-to-monocyte ratio and platelet-to-lymphocyte ratio among Iranian population: Results of Tabari cohort. Aspian J. Intern. Med. 2019, 10, 320–325. [Google Scholar]
- Solem, C.A.; Loftus, E.V.; Tremaine, W.J.; Harmsen, W.S.; Zinsmeister, A.R.; Sandborn, W.J. Correlation of C-reactive protein with clinical, endoscopic, histologic, and radiographic activity in inflammatory bowel disease. Inflamm. Bowel Dis. 2005, 11, 707–712. [Google Scholar] [CrossRef]
- Rodrigues, B.L.; Mazzaro, M.C.; Nagasako, C.K.; Ayrizono, M.D.L.S.; Fagundes, J.J.; Leal, R.F. Assessment of disease activity in inflammatory bowel diseases: Non-invasive biomarkers and endoscopic scores. World J. Gastrointest. Endosc. 2020, 12, 504–520. [Google Scholar] [CrossRef]
- D’Incà, R.; Pont, E.D.; Di Leo, V.; Ferronato, A.; Fries, W.; Vettorato, M.G.; Martines, D.; Sturniolo, G.C. Calprotectin and lactoferrin in the assessment of intestinal inflammation and organic disease. Int. J. Color. Dis. 2006, 22, 429–437. [Google Scholar] [CrossRef]
- Privitera, G.; Pugliese, D.; Lopetuso, L.R.; Scaldaferri, F.; Neri, M.; Guidi, L.; Gasbarrini, A.; Armuzzi, A. Novel trends with biologics in inflammatory bowel disease: Sequential and combined approaches. Ther. Adv. Gastroenterol. 2021, 14, 1–19. [Google Scholar] [CrossRef]
- Roda, G.; Jharap, B.; Neeraj, N.; Colombel, J.-F. Loss of response to anti-TNFs: Definition, epidemiology, and management. Clin. Transl. Gastroenterol. 2016, 7, e135. [Google Scholar] [CrossRef] [PubMed]
- Hanauer, S.B.; Feagan, B.G.; Lichtenstein, G.R.; Mayer, L.F.; Schreiber, S.; Colombel, J.F.; Rachmilewitz, D.; Wolf, D.C.; Olson, A.; Bao, W.; et al. Maintenance infliximab for Crohn’s disease: The ACCENT I randomised trial. Lancet 2002, 359, 1541–1549. [Google Scholar] [CrossRef]
- Stevens, T.W.; Matheeuwsen, M.; Lönnkvist, M.H.; Parker, C.E.; Wildenberg, M.E.; Gecse, K.B.; D’Haens, G.R. Systematic review: Predictive biomarkers of therapeutic response in inflammatory bowel disease-personalised medicine in its infancy. Aliment. Pharmacol. Ther. 2018, 48, 1213–1231. [Google Scholar] [CrossRef] [PubMed]
- Pham, M.T.; Rajić, A.; Greig, J.D.; Sargeant, J.M.; Papadopoulos, A.; McEwen, S.A. A scoping review of scoping reviews: Advancing the approach and enhancing the consistency. Res. Synth. Methods 2014, 5, 371–385. [Google Scholar] [CrossRef]
- Iskandar, H.N.; Ciorba, M.A. Biomarkers in inflammatory bowel disease: Current practices and recent advances. Transl. Res. 2012, 159, 313–325. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; Kim, N.Y.; Na, S.H.; Youn, Y.H.; Shin, C.S. Reference values of neutrophil-lymphocyte ratio, lymphocyte-monocyte ratio, platelet-lymphocyte ratio, and mean platelet volume in healthy adults in South Korea. Medicine 2018, 97, e11138. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Chen, Q.; Luo, X.; Hong, J.; Pan, K.; Lin, X.; Liu, X.; Zhou, L.; Wang, H.; Xu, Y.; et al. Neutrophil-to-lymphocyte ratio positively correlates to age in healthy population. J. Clin. Lab. Anal. 2014, 29, 437–443. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Graubard, B.I.; Rabkin, C.S.; Engels, E.A. Neutrophil-to-lymphocyte ratio and mortality in the United States general population. Sci. Rep. 2021, 11, 1–9. [Google Scholar] [CrossRef]
- Gumus, F.; Solak, I.; Eryilmaz, M.A. The effects of smoking on neutrophil/lymphocyte, platelet/ /lymphocyte ratios. Bratisl. Lek. List. 2018, 119, 116–119. [Google Scholar] [CrossRef]
| First Author, Year of Publication | Country | Study Type | Study Sample (n) | Condition | Use of Control | Study Aims Pertaining to NLR | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Differentiate Diagnoses | Differentiate Clinical Activity | Differentiate Endoscopic Activity | Predict Treatment Response | Predict Other Clinical Outcomes | Generate Optimal Cutpoint | Association with Other Biomarkers | ||||||
| AbediManesh, 2011 | Iran | Uncontrolled, non-randomized trial | 43 | UC | No | X | X | |||||
| Abotaga, 2009 | USA | Retrospective cohort | 62 | UC | No | X | ||||||
| Acarturk, 2015 | Turkey | Retrospective case-control | 83 | UC, CD | Yes | X | X | X | X | |||
| Ahmad, 2015 | Iraq | Prospective case-control | 90 | UC, CD | Yes | X | ||||||
| Akpinar, 2018 | Turkey | Retrospective cross-sectional | 313 | UC | Yes | X | X | X | ||||
| Andrew, 2020 | Australia | Retrospective cohort | 72 | UC | No | X | X | |||||
| Argeny, 2018 | Austria | Retrospective cohort | 373 | CD | No | X | ||||||
| Ben Jeddi, 2019 | Tunisia | Retrospective cohort | 74 | CD | No | X | X | |||||
| Ben Mustapha, 2015 | Tunisia | Prospective case-control | 47 | CD | Yes | X | X | |||||
| Bertani, 2019 | Italy | Retrospective cohort | 46 | UC | No | X | X | X | X | |||
| Bertani, 2020 | Italy | Prospective cohort | 88 | UC | No | X | X | X | X | X | ||
| BouJaoude, 2018 | France/ Lebanon | Prospective case-control | 116 | CD | Yes | X | X | X | ||||
| Celikbilek, 2013 | Turkey | Prospective case-control | 54 | UC | No | X | X | X | ||||
| Chalmers, 2017 | Scotland | Prospective case-control | 182 | IBD | Yes | X | X | |||||
| Chen, 2018 | China | Retrospective case-control | 120 | CD | Yes | X | X | |||||
| Chen, 2020 | China | Retrospective cohort | 876 | UC, CD | Yes | X | X | |||||
| Cherfane, 2013 | USA | Retrospective cohort | 185 | UC | Yes | X | X | X | X | |||
| Con, 2021 | Australia | Retrospective cohort | 94 | UC | No | X | X | |||||
| Crispino, 2021 | Italy | Retrospective cohort | 107 | CD | No | X | X | X | ||||
| Demir, 2015 | Turkey | Retrospective cohort | 211 | UC | Yes | X | X | X | X | |||
| Dong, 2019 | China | Prospective case-control | 104 | UC | No | X | X | X | X | X | ||
| Dorobăţ, 2018 | Romania | Retrospective cohort | 63 | UC | No | X | ||||||
| El-Sadek, 2019 | Egypt | Retrospective cohort | 27 | UC | No | X | X | X | ||||
| Eraldemir, 2014 | Turkey | Prospective case-control | 65 | UC | Yes | X | X | |||||
| Eraldemir, 2016 | Turkey | Prospective case-control | 87 | CD | Yes | X | X | X | X | |||
| Feng, 2017 | China | Retrospective case-control | 206 | CD | Yes | X | X | X | ||||
| Fidan, 2017 | Turkey | Retrospective cohort | 67 | UC | No | X | X | X | ||||
| Fleshner, 2019 | USA | Prospective cohort | No data | UC | No | X | ||||||
| Gao, 2015 | China | Prospective case-control | 110 | CD | Yes | X | X | X | X | |||
| Gao, 2020 | China | Retrospective cohort | 54 | CD | No | X | X | |||||
| Gold, 2020 | USA | Retrospective cohort | 107 | UC, CD | No | X | ||||||
| Gur, 2018 | Turkey | Retrospective cohort | 43 | CD | No | X | ||||||
| Gur, 2020 | Turkey | Retrospective case-control | 104 | CD | Yes | X | ||||||
| Guthrie, 2013 | Scotland | Retrospective case-control | 57 | IBD | Yes | X | ||||||
| Hanafy, 2018 | Egypt | Prospective case-control | 168 | UC | Yes | X | X | X | X | |||
| Hanai, 2004 | Japan | Prospective case-control | 100 | UC | Yes | X | X | |||||
| Jardak, 2018 | Tunisia | Retrospective cohort | 87 | UC | No | X | ||||||
| Jeong, 2018 | South Korea | Retrospective case-control | 174 | IBD | Yes | X | X | X | X | |||
| Jeong, 2021 | South Korea | Retrospective case-control | 144 | UC | Yes | X | X | X | ||||
| Kang, 2017 | China | Retrospective cohort | 108 | CD | No | X | X | X | ||||
| Khoury, 2019 | Israel | Retrospective case-control | 436 | CD | Yes | X | X | |||||
| Messner, 2016 | Austria | Retrospective cohort | 206 | IBD | No | X | ||||||
| Michalak, 2019 | Poland | Prospective case-control | 112 | UC | Yes | X | X | |||||
| Nassri, 2020 | USA | Retrospective cohort | No data | CD | No | X | ||||||
| Ndulue, 2019 | USA | Retrospective case-control | 4739 | IBD | Yes | X | ||||||
| Nishida, 2017 | Japan | Retrospective cohort | 59 | UC | No | X | X | X | ||||
| Nishida, 2019 | Japan | Retrospective cohort | 45 | UC | No | X | X | |||||
| Nishida, 2020 | Japan | Retrospective cohort | 49 | UC | No | X | X | |||||
| Okba, 2019 | Egypt | Prospective case-control | 80 | UC | Yes | X | X | X | X | X | ||
| Ovidiu, 2017 | Romania | Retrospective cohort | 86 | UC | No | X | ||||||
| Parisi, 2013 | Belgium | Retrospective cross-sectional | 139 | IBD | No | X | ||||||
| Posul, 2015 | Turkey | Prospective cohort | 49 | UC | No | X | X | |||||
| Ryan, 2019 | USA | Uncontrolled, non-randomized trial | 9 | IBD | No | X | ||||||
| Stefanidis, 2015 | Greece | Retrospective cohort | 35 | IBD | No | X | ||||||
| Torun, 2012 | Turkey | Retrospective case-control | 255 | UC | Yes | X | X | X | X | |||
| Wlodarczyk, 2015 | Poland | Retrospective case-control | 45 | CD | No | X | X | |||||
| Xu, 2019 | China | Prospective cohort | 214 | UC, CD | No | X | X | |||||
| Yamamoto-Furusho, 2020 | Japan | Retrospective cohort | 158 | UC | No | X | X | X | X | |||
| Yarur, 2011 | USA | Retrospective cohort | 68 | IBD | No | X | ||||||
| Zhang, 2017 | China | Prospective case-control | 34 | UC, CD | Yes | X | X | X | ||||
| Zhang, 2021 | China | Retrospective case-control | 344 | UC | Yes | X | X | X | ||||
| Zhou, 2021 | China | Retrospective case-control | 112 | CD | Yes | X | X | X | ||||
| Cutpoint Properties | Calculated Likelihood Ratios | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Author, Year of Publication | Purpose | AUC | Cutpoint | SEN | SPE | PPV | NPV | OA | p-Value * | LR+ | LR− | |
| Crohn’s Disease | Bou Jaoude, 2018 | Differentiate CD from non-CD | 0.522 | >1.98 | 0.684 | 0.431 | >0.05 | 1.202 | 0.733 | |||
| Chen, 2018 | 0.828 | 2.85 | 0.692 | 0.762 | 2.908 | 0.404 | ||||||
| Gao, 2015 | 0.850 | 2.13 | 0.827 | 0.769 | 3.580 | 0.225 | ||||||
| Feng, 2017 | 0.740 | 2.72 | 0.683 | 0.759 | 0.701 | 2.834 | 0.418 | |||||
| Acarturk, 2015 | Differentiate active CD and remission (clinical) | 0.830 | 3.20 | 0.810 | 0.590 | 0.930 | 0.740 | <0.001 | 1.976 | 0.322 | ||
| Ben Jeddi, 2019 | -- | 1.57 | -- | -- | ||||||||
| Chen, 2020 | 0.764 | 3.32 | 0.659 | 0.759 | 2.734 | 0.449 | ||||||
| Eraldemir, 2016 | 0.703 | 2.58 | 0.696 | 0.760 | 0.727 | 0.731 | 2.900 | 0.400 | ||||
| Xu, 2019 | 0.631 | NR | NS | NS | NS | NS | NS | -- | -- | |||
| Zhang, 2017 | 0.812 | 1.95 | 0.955 | 0.571 | 0.778 | 0.889 | 0.806 | 2.226 | 0.079 | |||
| Zhang, 2017 | Differentiate severe and mild-to-moderate CD (clinical) | 0.880 | 5.35 | 0.75 | 0.929 | 0.857 | 0.867 | 0.864 | 0.02 | 10.563 | 0.269 | |
| Khoury, 2019 | Part of a new clinical score to predict intra-abdominal masses | 0.747 | 11.75 5.60 | 0.530 0.850 | 0.850 0.480 | 3.533 1.635 | 0.283 0.612 | |||||
| Crispino, 2021 | Predict endoscopic remission from biologic therapy at baseline | 0.640 | 1.55 | 0.400 | 0.860 | 0.640 | 0.707 | 0.003 | 2.857 | 0.698 | ||
| Ben Mustapha, 2015 | Predict sustained response to IFX therapy at baseline | -- | <4.00 | 0.800 | 0.800 | <0.05 | 4.000 | 0.250 | ||||
| Wlodarczyk, 2015 | 0.850 | 4.07 | 0.800 | 0.870 | 0.860 | 0.810 | 6.154 | 0.230 | ||||
| Ben Mustapha, 2015 | Predict sustained response to IFX therapy at week 14 | -- | <3.50 | 0.720 | 0.700 | <0.05 | 2.400 | 0.400 | ||||
| Wlodarczyk, 2015 | 0.760 | 3.670 | 0.670 | 0.800 | 0.770 | 0.710 | 3.350 | 0.413 | ||||
| Gao, 2020 | Predict loss of response to IFX therapy at week 14 | 0.903 | 2.75 | 0.933 | 0.846 | <0.00 | 6.058 | 0.079 | ||||
| Kang, 2017 | Predict postoperative complications | 0.675 | 4.10 | 0.700 | 0.564 | 1.606 | 0.532 | |||||
| Cherfane, 2013 | Differentiate UC from non-UC | 0.735 | 2.60 | 0.700 | 0.630 | 1.892 | 0.476 | |||||
| Dong, 2019 | 0.731 | 4.70 * | 0.613 | 0.857 | 4.287 | 0.452 | ||||||
| Ulcerative Colitis | Jeong, 2021 | 0.774 | 2.26 | 0.542 | 0.906 | 0.578 | 5.766 | 0.506 | ||||
| Zhang, 2021 | 0.858 | 2.66 | 0.750 | 0.826 | <0.001 | 4.310 | 0.303 | |||||
| Acarturk, 2015 | Differentiate active UC and remission (clinical) | 0.740 | 3.10 | 0.780 | 0.690 | 0.840 | 0.640 | <0.001 | 2.516 | 0.319 | ||
| Celikbilek, 2013 | -- | 2.47 | 0.539 | 0.632 | 0.667 | 0.500 | 0.578 | 1.465 | 0.729 | |||
| Chen, 2020 | 0.828 | 2.85 | 0.762 | 0.845 | 4.916 | 0.282 | ||||||
| Demir, 2015 | 0.640 | 2.39 | 0.486 | 0.775 | 0.680 | 0.604 | 2.160 | 0.663 | ||||
| Fidan, 2017 | 0.722 | 2.20 | 0.620 | 0.700 | <0.05 | 2.067 | 0.543 | |||||
| Hanafy, 2018 | 0.810 | 2.35 | 0.740 | 0.860 | 5.286 | 0.302 | ||||||
| Okba, 2019 | -- | 1.91 | 0.900 | 0.900 | 9.000 | 0.111 | ||||||
| Posul, 2015 | 0.650 | 2.30 | 0.612 | 0.667 | 1.838 | 0.582 | ||||||
| Torun, 2012 | 0.850 | 2.16 | 0.818 | 0.805 | 0.868 | 0.738 | 4.195 | 0.226 | ||||
| Xu, 2019 | 0.625 | NR | NS | NS | NS | NS | NS | -- | -- | |||
| Yamamoto-Furosho, 2020 | -- | 2.00 | 0.750 | 0.635 | 2.055 | 0.394 | ||||||
| Zhang, 2017 | 0.726 | 3.29 | 0.474 | 0.939 | 0.900 | 0.583 | 0.676 | 7.770 | 0.560 | |||
| Jeong, 2021 | Differentiate severe and mild-to-moderate UC (clinical) | 0.714 | 3.44 | 0.636 | 0.811 | 3.365 | 0.449 | |||||
| Zhang, 2017 | 0.560 | 3.92 | 0.375 | 1.000 | 1.000 | 0.231 | 0.474 | 0.517 | 0.625 | |||
| Akpinar, 2018 | Differentiate active UC and remission (endoscopic) | 0.718 | 2.42 | 0.760 | 0.702 | 0.003 | 2.550 | 0.342 | ||||
| Zhou, 2021 | 0.680 | 4.45 | 0.839 | 0.469 | 0.522 | 0.809 | 0.62 | < 0.001 | 1.580 | 0.343 | ||
| Yamamoto-Furosho, 2020 | -- | 2.09 | 0.639 | 0.588 | 1.551 | 0.614 | ||||||
| Cherfane, 2013 | Differentiate active UC from C. difficile infection | 0.693 | 3.10 | 0.700 | 0.650 | 2.000 | 0.462 | |||||
| El-Sadek, 2021 | Predict UC flare during pregnancy | 0.915 | 2.85 | 0.900 | 0.882 | 0.001 | -- | -- | ||||
| Nishida, 2021 | Predict development of pouchitis after ileal pouch-anal anastomosis | 0.680 | 2.15 | 0.722 | 0.677 | -- | -- | |||||
| Bertani, 2019 | Predict clinical remission with anti-TNF medications at baseline | 0.889 | 2.33 | 0.900 | 0.650 | 2.571 | 0.154 | |||||
| Bertani, 2019 | Predict mucosal healing with anti-TNF medications at baseline | 0.853 | 2.33 | 0.800 | 0.6700 | 2.424 | 0.299 | |||||
| Bertani, 2020 | -- | 2.06 | 0.600 | 0.792 | 2.885 | 0.505 | ||||||
| Nishida, 2017 | Predict response to IFX therapy at baseline | 0.702 | 4.49 | 0.786 | 0.783 | 3.622 | 0.273 | |||||
| Nishida, 2019 | Predict risk of relapse with tacrolimus therapy at baseline | -- | 5.84 | 0.625 | 0.667 | 1.877 | 0.562 | |||||
| IBD | Jeong, 2018 | Differentiate IBD from non-IBD | 0.802 | 1.80 | 0.707 | 0.733 | 2.648 | 0.400 | ||||
| Chalmers, 2017 | Differentiate PIBD from non-IBD | 0.810 | 2.37 | 0.67 | 0.85 | 4.467 | 0.388 | |||||
| Study Population | NLR Associations & Correlations | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Author, Year of Publication | CRP | ESR | WBC | PLR | Fibrinogen | Fecal Calprotectin | Fecal Lactoferrin | Malondialdehyde | Nitric Oxide | |
| Crohn’s Disease | Acarturk, 2015 | * rs = –0.61, p = 0.793 ** rs = −0.022, p = 0.924 | * rs = 0.242, p = 0.291 ** rs = −0.042, p = 0.856 | * rs = 0.242, p ≤ 0.001 ** rs = −0.135, p = 0.561 | ||||||
| Eraldemir, 2016 | * B = −0.044, 95% CI −0.205–0.116, p = 0.573 | * B = 0.174, 95% CI −0.044–0.393, p = 0.112 | * B = 0.422, 95% CI 0.0480.796, p = 0.029 | |||||||
| Feng, 2017 | rs = 0.39, p < 0.01 | rs = 0.43, p < 0.01 | ||||||||
| Gao, 2015 | rs = 0.327, p < 0.001 | rs = 0.137, p = 0.082 | rs = 0.493, p < 0.001 | |||||||
| Ulcerative Colitis | Acarturk, 2015 | * rs = 0.116, p = 0.463 ** rs = −0.198, p = 0.208 | * rs = 0.051, p = 0.750 ** rs = 0.200, p = 0.203 | * rs = 0.260, p = 0.096 ** rs = 0.266, p = 0.089 | ||||||
| Bertani, 2019 | NS | NS | ||||||||
| Bertani, 2020 | rs = 0.11 (baseline), p > 0.05 rs = 0.21 (week 8), p > 0.05 | |||||||||
| Demir, 2015 | rs = 0.185, p = 0.059 * rs = 0.141, p = 0.246 ** rs = 0.020, p = 0.911 | rs = 0.170, p = 0.043 * rs = 0.121, p = 0.319 ** rs = 0.088, p = 0.468 | rs = 0.282, p = 0.001 * rs = 0.360, p = 0.002 ** rs = 0.097, p = 0.420 | |||||||
| Dong, 2019 | * p < 0.05 | * p < 0.05 | ||||||||
| El-Sadek, 2019 | rs = 0.418, p = 0.03 | rs = 0.522, p = 0.005 | ||||||||
| Eraldemir, 2014 | NS | r2 = 0.593, p < 0.001 | ||||||||
| Fidan, 2017 | * rs = 0.370, p < 0.05 | * rs = 0.944, p < 0.05 | ||||||||
| Hanafy, 2018 | p < 0.001 | |||||||||
| Michalak, 2019 | p < 0.001 | |||||||||
| Okba, 2019 | rs = 0.789, p < 0.001 * rs = 0.490, p = 0.028 ** rs = 0.146, p = 0.538 | rs = 0.556, p < 0.001 * rs = 0.597, p = 0.005 ** rs = −0.139, p = 0.558 | rs = 0.324, p = 0.012 * rs = 0.184, p = 0.437 ** rs = 0.088, p = 0.712 | |||||||
| Torun, 2012 | rs = 0.102, p = 0.153 | rs = 0.217, p = 0.002 | rs = 0.416, p < 0.001 | rs = 0.095, p = 0.187 | ||||||
| Yamamoto-Furosho, 2020 | rs = 0.347, p < 0.001 | |||||||||
| IBD | Jeong, 2018 | r2 = 0.348, p = 0.008 | ||||||||
| Messner, 2016 | r2 = 0.210, p ≤ 0.05 | |||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Langley, B.O.; Guedry, S.E.; Goldenberg, J.Z.; Hanes, D.A.; Beardsley, J.A.; Ryan, J.J. Inflammatory Bowel Disease and Neutrophil–Lymphocyte Ratio: A Systematic Scoping Review. J. Clin. Med. 2021, 10, 4219. https://doi.org/10.3390/jcm10184219
Langley BO, Guedry SE, Goldenberg JZ, Hanes DA, Beardsley JA, Ryan JJ. Inflammatory Bowel Disease and Neutrophil–Lymphocyte Ratio: A Systematic Scoping Review. Journal of Clinical Medicine. 2021; 10(18):4219. https://doi.org/10.3390/jcm10184219
Chicago/Turabian StyleLangley, Blake O., Sara E. Guedry, Joshua Z. Goldenberg, Douglas A. Hanes, Jennifer A. Beardsley, and Jennifer Joan Ryan. 2021. "Inflammatory Bowel Disease and Neutrophil–Lymphocyte Ratio: A Systematic Scoping Review" Journal of Clinical Medicine 10, no. 18: 4219. https://doi.org/10.3390/jcm10184219
APA StyleLangley, B. O., Guedry, S. E., Goldenberg, J. Z., Hanes, D. A., Beardsley, J. A., & Ryan, J. J. (2021). Inflammatory Bowel Disease and Neutrophil–Lymphocyte Ratio: A Systematic Scoping Review. Journal of Clinical Medicine, 10(18), 4219. https://doi.org/10.3390/jcm10184219







