Persistent Resistant Hypertension Has Worse Renal Outcomes in Chronic Kidney Disease than that Resolved in Two Years: Results from the KNOW-CKD Study
Abstract
:1. Introduction
2. Materials and Methods
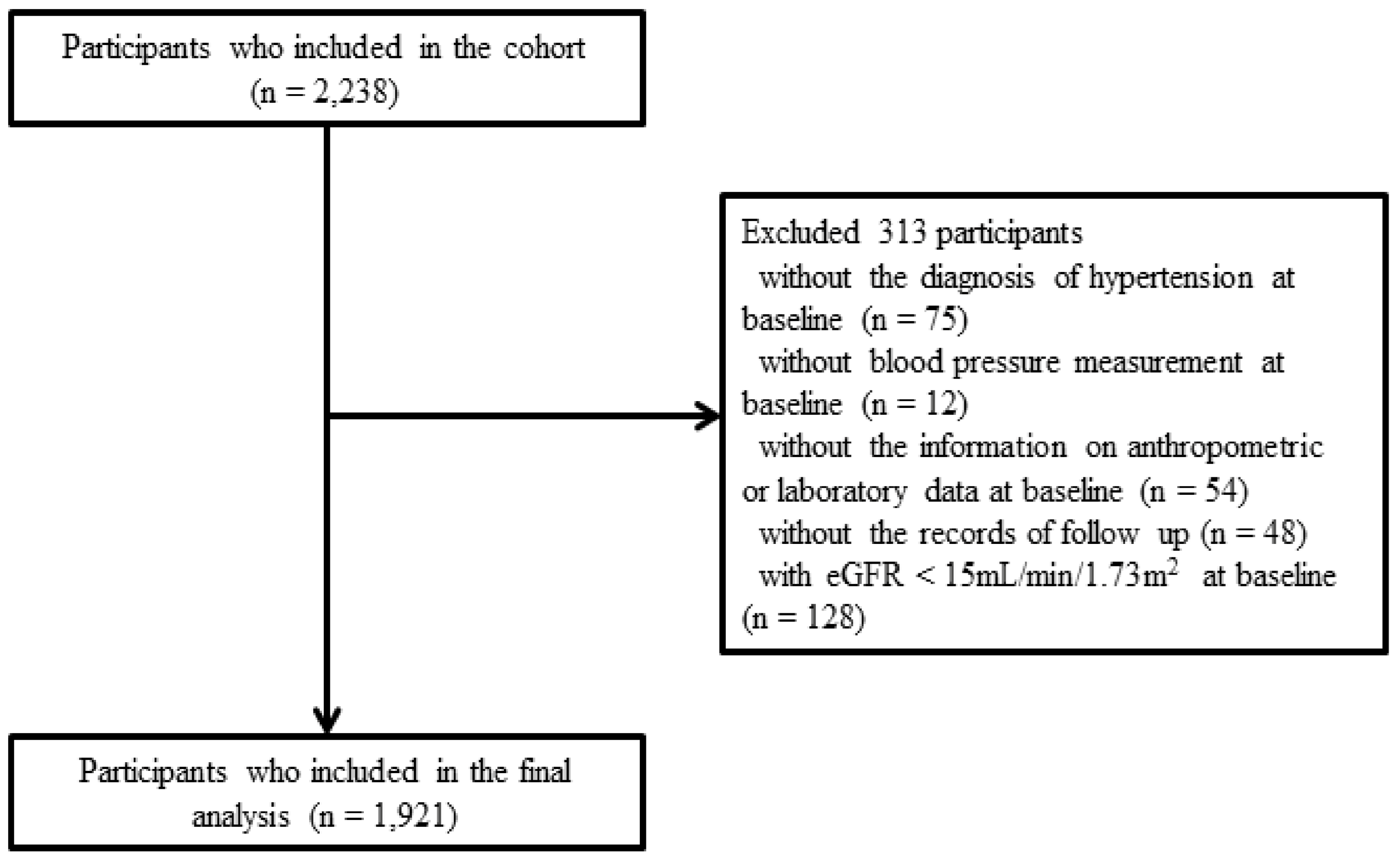
2.1. Study Participants
2.2. Data Collection
2.3. Definition of ATRH and BP Measurement
2.4. Study End Point and Definition of the ATRH Subgroups
2.5. Statistical Methods
3. Results
3.1. Baseline Characteristics of Study Participants
3.2. Risk Factors Associated with ATRH
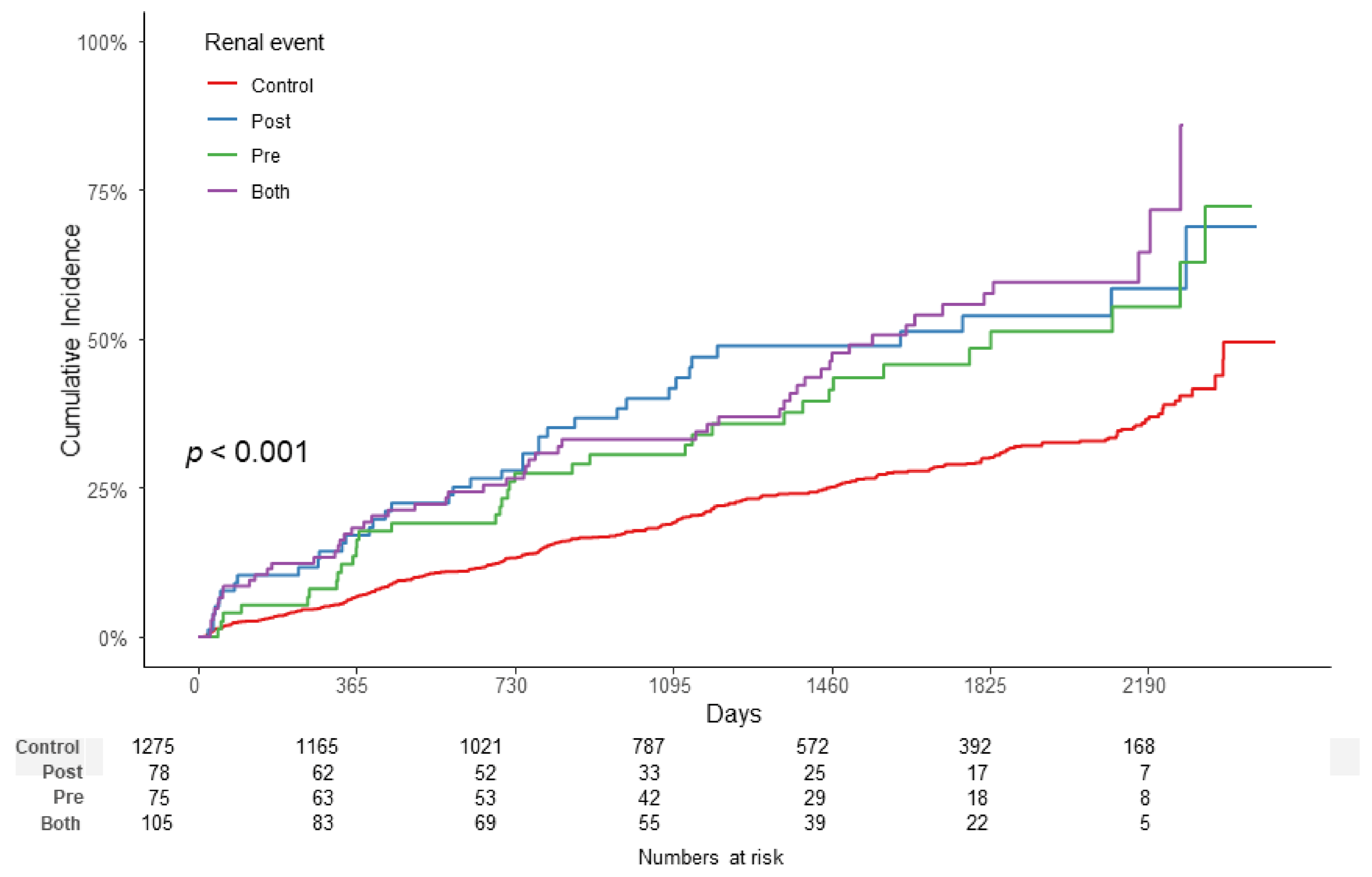
3.3. Association between ATRH Subgroups and Renal Outcome
3.4. Subgroup Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Kidney Disease: Improving Global Outcomes (KDIGO) Blood Pressure Work Group. KDIGO clinical practice guideline for the management of blood pressure in chronic kidney disease. Kidney Int. Supply 2012, 2, 337–414.
- James, P.A.; Oparil, S.; Carter, B.L.; Cushman, W.C.; Dennison-Himmelfarb, C.; Handler, J.; Lackland, D.T.; LeFevre, M.L.; MacKenzie, T.D.; Ogedegbe, O.; et al. 2014 evidence-based guideline for the management of high blood pressure in adults: Report from the panel members appointed to the Eighth Joint National Committee (JNC 8). JAMA 2014, 5, 507–520. [Google Scholar] [CrossRef] [Green Version]
- Carey, R.M.; Calhoun, D.A.; Bakris, G.L.; Brook, R.D.; Daugherty, S.L.; Dennison-Himmelfarb, C.R.; Egan, B.M.; Flack, J.M.; Gidding, S.S.; Judd, E.; et al. Resistant Hypertension: Detection, Evaluation, and Management: A Scientific Statement from the American Heart Association. Hypertension 2018, 72, e53–e90. [Google Scholar] [CrossRef] [PubMed]
- Williams, B.; Mancia, G.; Spiering, W.; Agabiti Rosei, E.; Azizi, M.; Burnier, M.; Clement, D.L.; Coca, A.; de Simone, G.; Dominiczak, A.; et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension. Eur. Heart J. 2018, 39, 3021–3104. [Google Scholar] [CrossRef] [PubMed]
- Calhoun, D.A.; Jones, D.; Textor, S.; Goff, D.C.; Murphy, T.P.; Toto, R.D.; White, A.; Cushman, W.C.; White, W.; Sica, D.; et al. Resistant hypertension: Diagnosis, evaluation, and treatment. A scientific statement from the American Heart Association Professional Education Committee of the Council for High Blood Pressure Research. Hypertension 2018, 51, 1403–1419. [Google Scholar] [CrossRef] [Green Version]
- Noubiap, J.J.; Nansseu, J.R.; Nyaga, U.F.; Sime, P.S.; Francis, I.; Bigna, J.J. Global prevalence of resistant hypertension: A meta-analysis of data from 3.2 million patients. Heart 2019, 105, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Rossignol, P.; Massy, Z.A.; Azizi, M.; Bakris, G.; Ritz, E.; Covic, A.; Goldsmith, D.; Heine, G.H.; Jager, K.J.; Kanbay, M.; et al. The double challenge of resistant hypertension and chronic kidney disease. Lancet 2015, 386, 1588–1598. [Google Scholar] [CrossRef]
- Bangalore, S.; Fayyad, R.; Laskey, R.; Demicco, D.A.; Deedwania, P.; Kostis, J.B.; Messerli, F.H.; Treating to New Targets Steering Committee and Investigators. Prevalence, predictors, and outcomes in treatment-resistant hypertension in patients with coronary disease. Am. J. Med. 2014, 127, 71–81.e1. [Google Scholar] [CrossRef] [PubMed]
- Chia, R.; Pandey, A.; Vongpatanasin, W. Resistant hypertension-defining the scope of the problem. Prog. Cardiovasc. Dis. 2020, 63, 46–50. [Google Scholar] [CrossRef]
- Lee, K.N.; Na, J.O.; Choi, C.U.; Lim, H.E.; Kim, J.W.; Kim, E.J.; Rha, S.W.; Seo, H.S.; Oh, D.J.; Park, C.G. Prevalence and characteristics of resistant hypertension at primary clinics in Korea: A nationwide cross-sectional study. Clin. Hypertens. 2016, 22, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chun, K.H.; Lee, C.J.; Oh, J.; Lee, S.H.; Kang, S.M.; Kario, K.; Park, S. Prevalence and prognosis of the 2018 vs. 2008 AHA definitions of apparent treatment-resistant hypertension in high-risk hypertension patients. J. Clin. Hypertens. 2020, 22, 2093–2102. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.I.; Kim, S.K.; Park, S.; Kim, J.H.; Ihm, S.H.; Kim, G.I.; Kim, W.S.; Pyun, W.B.; Kim, Y.M.; Shin, J. Prevalence of resistant hypertension and associated factors for blood pressure control status with optimal medical therapy using Korean ambulatory blood pressure monitoring registry data. Clin. Hypertens. 2016, 22, 8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsioufis, C.; Kasiakogias, A.; Kordalis, A.; Dimitriadis, K.; Thomopoulos, C.; Tsiachris, D.; Vasileiou, P.; Doumas, M.; Makris, T.; Papademetriou, V.; et al. Dynamic resistant hypertension patterns as predictors of cardiovascular morbidity: A 4-year prospective study. J. Hypertens. 2014, 32, 415–422. [Google Scholar] [CrossRef]
- Viazzi, F.; Greco, E.; Ceriello, A.; Fioretto, P.; Giorda, C.; Guida, P.; Russo, G.; De Cosmo, S.; Pontremoli, R.; AMD-Annals Study Group. Apparent Treatment Resistant Hypertension, Blood Pressure Control and the Progression of Chronic Kidney Disease in Patients with Type 2 Diabetes. Kidney Blood Press. Res. 2018, 43, 422–438. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaboré, J.; Metzger, M.; Helmer, C.; Berr, C.; Tzourio, C.; Massy, Z.A.; Stengel, B. Kidney Function Decline and Apparent Treatment-Resistant Hypertension in the Elderly. PLoS ONE 2016, 11, e0146056. [Google Scholar] [CrossRef] [Green Version]
- De Nicola, L.; Gabbai, F.B.; Agarwal, R.; Chiodini, P.; Borrelli, S.; Bellizzi, V.; Nappi, F.; Conte, G.; Minutolo, R. Prevalence and prognostic role of resistant hypertension in chronic kidney disease patients. J. Am. Coll. Cardiol. 2013, 61, 2461–2467. [Google Scholar] [CrossRef] [Green Version]
- Oh, K.H.; Park, S.K.; Park, H.C.; Chin, H.J.; Chae, D.W.; Choi, K.H.; Han, S.H.; Yoo, T.H.; Lee, K.; Kim, Y.S.; et al. KNOW-CKD (KoreaN cohort study for Outcome in patients With Chronic Kidney Disease): Design and methods. BMC Nephrol. 2014, 15, 80. [Google Scholar] [CrossRef] [Green Version]
- Kidney Disease: Improving Global Outcomes (KDIGO). KDIGO clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int. Suppl. 2013, 3, 5–14.
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.L.; Castro, A.F., 3rd; Feldman, H.I.; Kusek, J.W.; Eggers, P.; Van Lente, F.; Greene, T.; et al. A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 2009, 5, 604–612. [Google Scholar] [CrossRef]
- Alencar de Pinho, N.; Levin, A.; Fukagawa, M.; Hoy, W.E.; Pecoits-Filho, R.; Reichel, H.; Robinson, B.; Kitiyakara, C.; Wang, J.; Eckardt, K.U.; et al. Considerable international variation exists in blood pressure control and antihypertensive prescription patterns in chronic kidney disease. Kidney Int. 2019, 96, 983–994. [Google Scholar] [CrossRef] [Green Version]
- Schneider, M.P.; Hilgers, K.F.; Schmid, M.; Hübner, S.; Nadal, J.; Seitz, D.; Busch, M.; Haller, H.; Köttgen, A.; Kronenberg, F.; et al. Blood pressure control in chronic kidney disease: A cross-sectional analysis from the German Chronic Kidney Disease (GCKD) study. PLoS ONE 2018, 13, e0202604. [Google Scholar] [CrossRef] [PubMed]
- Egan, B.M.; Zhao, Y.; Axon, R.N.; Brzezinski, W.A.; Ferdinand, K.C. Uncontrolled and apparent treatment resistant hypertension in the United States, 1988 to 2008. Circulation 2011, 124, 1046–1058. [Google Scholar] [CrossRef] [Green Version]
- de la Sierra, A.; Banegas, J.R.; Oliveras, A.; Gorostidi, M.; Segura, J.; de la Cruz, J.J.; Armario, P.; Ruilope, L.M. Clinical differences between resistant hypertensives and patients treated and controlled with three or less drugs. J. Hypertens. 2012, 30, 1211–1216. [Google Scholar] [CrossRef] [PubMed]
- Sim, J.J.; Bhandari, S.K.; Shi, J.; Reynolds, K.; Calhoun, D.A.; Kalantar-Zadeh, K.; Jacobsen, S.J. Comparative risk of renal, cardiovascular, and mortality outcomes in controlled, uncontrolled resistant, and nonresistant hypertension. Kidney Int. 2015, 88, 622–632. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ku, E.; Lee, B.J.; Wei, J.; Weir, M.R. Hypertension in CKD: Core Curriculum 2019. Am. J. Kidney Dis. 2019, 74, 120–131. [Google Scholar] [CrossRef] [Green Version]
- Bakris, G.L.; Williams, M.; Dworkin, L.; Elliott, W.J.; Epstein, M.; Toto, R.; Tuttle, K.; Douglas, J.; Hsueh, W.; Sowers, J. Preserving renal function in adults with hypertension and diabetes: A consensus approach. National Kidney Foundation Hypertension and Diabetes Executive Committees Working Group. Am. J. Kidney Dis. 2000, 36, 646–661. [Google Scholar] [CrossRef]
- Yiannakopoulou, E.C.; Papadopulos, J.S.; Cokkinos, D.V.; Mountokalakis, T.D. Adherence to antihypertensive treatment: A critical factor for blood pressure control. Eur. J. Cardiovasc. Prev. Rehabil. 2005, 12, 243–249. [Google Scholar] [CrossRef]

| Variable | Total Population (n = 1921) | p-Value | |
|---|---|---|---|
| No ATRH (n = 1653) | ATRH † (n = 268) | ||
| Age, year (average ± SD) | 53.4 ± 12.3 | 56.3 ± 12.4 | <0.001 |
| Male gender, n (%) | 983 (59.5%) | 194 (72.4%) | <0.001 |
| Education: | 0.791 | ||
| No education or illiteracy | 25 (1.5%) | 5 (1.9%) | |
| Elementary school graduate | 173 (10.5%) | 32 (11.9%) | |
| Middle school graduate | 206 (12.5%) | 28 (10.4%) | |
| High school graduate | 565 (34.2%) | 89 (33.2%) | |
| More than college graduate | 684 (41.4%) | 114 (42.5%) | |
| Employment status, n (%) | 1223 (74.0%) | 190 (70.9%) | 0.322 |
| Smoking status: | <0.001 | ||
| Never | 909 (55.5%) | 112 (41.8%) | |
| Ex-smoker | 481 (29.1%) | 117 (43.7%) | |
| Current smoker | 263 (15.9%) | 39 (14.6%) | |
| Primary renal disease: | <0.001 | ||
| Diabetic kidney disease | 378 (22.9%) | 130 (48.5%) | |
| Hypertensive renal disease | 313 (18.9%) | 72 (26.9%) | |
| Glomerulonephropathy | 535 (32.4%) | 37 (13.8%) | |
| Tubulointerstitial nephropathy | 12 (0.7%) | 0 (0.0%) | |
| Polycystic kidney disease | 302 (18.3%) | 19 (7.1%) | |
| Other kidney disease | 113 (6.8%) | 10 (3.7%) | |
| Coronary artery disease, n (%) | 97 (5.9%) | 32 (11.9%) | <0.001 |
| DM, n (%) | 516 (31.2%) | 159 (59.3%) | <0.001 |
| BMI, Kg/m2 (average ± SD) | 24.4 ± 3.3 | 26.2 ± 3.7 | <0.001 |
| Serum creatinine, mg/dL (average ± SD) | 1.7 ± 1.1 | 2.5 ± 1.4 | <0.001 |
| eGFR, mL/min/1.73 m2 (average ± SD) | 54.9 ± 30.8 | 37.0 ± 24.0 | <0.001 |
| BUN, mg/dL (average ± SD) | 27.0 ± 15.0 | 38.2 ± 18.8 | <0.001 |
| Random urine ACR, mg/gCr (average ± SD) | 781.3 ± 1276.0 | 1621.5 ± 2008.3 | <0.001 |
| Random urine PCR, g/gCr | 1.1 ±1.9 | 2.4 ± 3.1 | <0.001 |
| Serum hemoglobin, g/dL (average ± SD) | 12.9 ± 2.0 | 12.3 ± 2.1 | <0.001 |
| Serum albumin, g/dL (average ± SD) | 4.2 ± 0.4 | 4.1 ± 0.5 | <0.001 |
| Serum uric acid, mg/dL (average ± SD) | 6.9 ± 1.9 | 7.9 ± 2.0 | <0.001 |
| Na, mmol/L (average ± SD) | 140.8 ± 2.4 | 140.8 ± 2.8 | 0.702 |
| K, mmol/L (average ± SD) | 4.6 ± 0.6 | 4.8 ± 0.6 | <0.001 |
| Total cholesterol, mg/dL (average ± SD) | 173.6 ± 38.4 | 176.3 ± 42.7 | 0.330 |
| LDL, mg/dL (average ± SD) | 96.8 ± 31.2 | 96.0 ± 31.5 | 0.706 |
| HDL, mg/dL (average ± SD) | 49.9 ± 15.4 | 44.4 ± 14.2 | <0.001 |
| Triglyceride, mg/dL (average ± SD) | 151.4 ± 92.1 | 189.5 ± 119.8 | <0.001 |
| Systolic blood pressure, mmHg (average ± SD) | 126.5 ± 15.2 | 136.7 ± 18.6 | <0.001 |
| Diastolic blood pressure, mmHg (average ± SD) | 76.4 ± 10.6 | 80.0 ± 13.4 | <0.001 |
| Taking aspirin, n (%) | 418 (25.3%) | 117 (43.7%) | <0.001 |
| Number of anti-hypertensive agents | 1.7 ± 0.9 | 4.1 ± 0.7 | <0.001 |
| ACE inhibitor | 160 (9.7%) | 53 (19.8%) | <0.001 |
| Angiotensin receptor blocker | 1295 (78.3%) | 251 (93.7%) | <0.001 |
| Diuretics | 392 (23.7%) | 216 (80.6%) | <0.001 |
| Thiazide | 163 (9.9%) | 66 (24.6%) | <0.001 |
| Loop diuretics | 207 (12.5%) | 144 (53.7%) | |
| Potassium sparing diuretics | 22 (1.3%) | 6 (2.2%) | |
| Beta blocker | 277 (16.8%) | 225 (84.0%) | <0.001 |
| Nondihydropyridine CCB | 28 (1.7%) | 17 (6.3%) | <0.001 |
| Dihydropyridine CCB | 558 (33.8%) | 244(91.0%) | <0.001 |
| Nitrate | 25 (1.5%) | 27 (10.1%) | <0.001 |
| Alpha blocker | 44 (2.7%) | 57 (21.3%) | <0.001 |
| Minoxidil | 2 (0.1%) | 11 (4.1%) | <0.001 |
| Variable | Unadjusted Odds Ratio (95% CI) | p-Value | Adjusted Odds Ratio (95% CI) | p-Value |
|---|---|---|---|---|
| Age (per 1 year change) | 1.02 (1.01–1.03) | <0.001 | 1.00 (0.99, 1.01) | 0.838 |
| Male sex (reference: female) | 1.79 (1.34–2.38) | <0.001 | 1.70 (1.24–2.32) | 0.001 |
| eGFR (per 1 mL/min/1.73 m2 change) | 0.98 (0.97–0.98) | <0.001 | 0.98 (0.98–0.99) | <0.001 |
| BMI (per 1 kg/m2 change) | 1.15 (1.11–1.20) | <0.001 | 1.16 (1.11–1.20) | <0.001 |
| Diabetes | 3.21 (2.47–4.19) | <0.001 | 1.82 (1.33–2.49) | <0.001 |
| Coronary artery disease | 2.18 (1.43–3.32) | <0.001 | 1.24 (0.78–1.97) | 0.764 |
| Uric acid (per 1 mg/dL change) | 1.28 (1.20–1.37) | <0.001 | 1.15 (1.06–1.24) | <0.001 |
| LDL (per 1 mg/dL change) | 1 (1.00–1.00) | 0.703 | 1 (1.00–1.01) | 0.485 |
| Na (per 1 mmol/L change) | 0.99 (0.94–1.04) | 0.665 | 1.01 (0.96–1.07) | 0.635 |
| Random urine PCR (per 1 g/gCr) | 1.21 (1.16–1.27) | <0.001 | 1.11 (1.05–1.17) | <0.001 |
| PRE Group | POST Group | BOTH Group | ||
|---|---|---|---|---|
| Unadjusted | HR (95% CI) | 1.85 (1.31–2.62) | 2.11 (1.50–2.98) | 2.30 (1.72–3.08) |
| p-Value | < 0.001 | < 0.001 | < 0.001 | |
| Model 1 | HR (95% CI) | 1.87 (1.32–2.65) | 2.11 (1.49–2.98) | 2.32 (1.73–3.12) |
| p-Value | < 0.001 | < 0.001 | < 0.001 | |
| Model 2 | HR (95% CI) | 1.44 (1.01–2.06) | 1.70 (1.19–2.42) | 2.08 (1.53–2.83) |
| p-Value | 0.043 | 0.003 | <0.001 | |
| Model 3 | HR (95% CI) | 1.04 (0.72–1.52) | 1.23 (0.86–1.75) | 1.41 (1.04–1.92) |
| p-Value | 0.831 | 0.254 | 0.027 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, S.-H.; Kim, Y.-J.; Choi, H.-S.; Kim, C.-S.; Bae, E.-H.; Ahn, C.; Oh, K.-H.; Park, S.-K.; Lee, K.-B.; Sung, S.; et al. Persistent Resistant Hypertension Has Worse Renal Outcomes in Chronic Kidney Disease than that Resolved in Two Years: Results from the KNOW-CKD Study. J. Clin. Med. 2021, 10, 3998. https://doi.org/10.3390/jcm10173998
Song S-H, Kim Y-J, Choi H-S, Kim C-S, Bae E-H, Ahn C, Oh K-H, Park S-K, Lee K-B, Sung S, et al. Persistent Resistant Hypertension Has Worse Renal Outcomes in Chronic Kidney Disease than that Resolved in Two Years: Results from the KNOW-CKD Study. Journal of Clinical Medicine. 2021; 10(17):3998. https://doi.org/10.3390/jcm10173998
Chicago/Turabian StyleSong, Su-Hyun, Young-Jin Kim, Hong-Sang Choi, Chang-Seong Kim, Eun-Hui Bae, Curie Ahn, Kook-Hwan Oh, Sue-Kyung Park, Kyu-Beck Lee, Suah Sung, and et al. 2021. "Persistent Resistant Hypertension Has Worse Renal Outcomes in Chronic Kidney Disease than that Resolved in Two Years: Results from the KNOW-CKD Study" Journal of Clinical Medicine 10, no. 17: 3998. https://doi.org/10.3390/jcm10173998
APA StyleSong, S.-H., Kim, Y.-J., Choi, H.-S., Kim, C.-S., Bae, E.-H., Ahn, C., Oh, K.-H., Park, S.-K., Lee, K.-B., Sung, S., Han, S.-H., Ma, S.-K., & Kim, S.-W. (2021). Persistent Resistant Hypertension Has Worse Renal Outcomes in Chronic Kidney Disease than that Resolved in Two Years: Results from the KNOW-CKD Study. Journal of Clinical Medicine, 10(17), 3998. https://doi.org/10.3390/jcm10173998







