Properties of Uncommon Indirect Immunofluorescence Staining Patterns Determined during Antinuclear Antibody Detection on HEp-2 Cells
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. Demographic Characteristics
3.2. Specific Autoantibodies
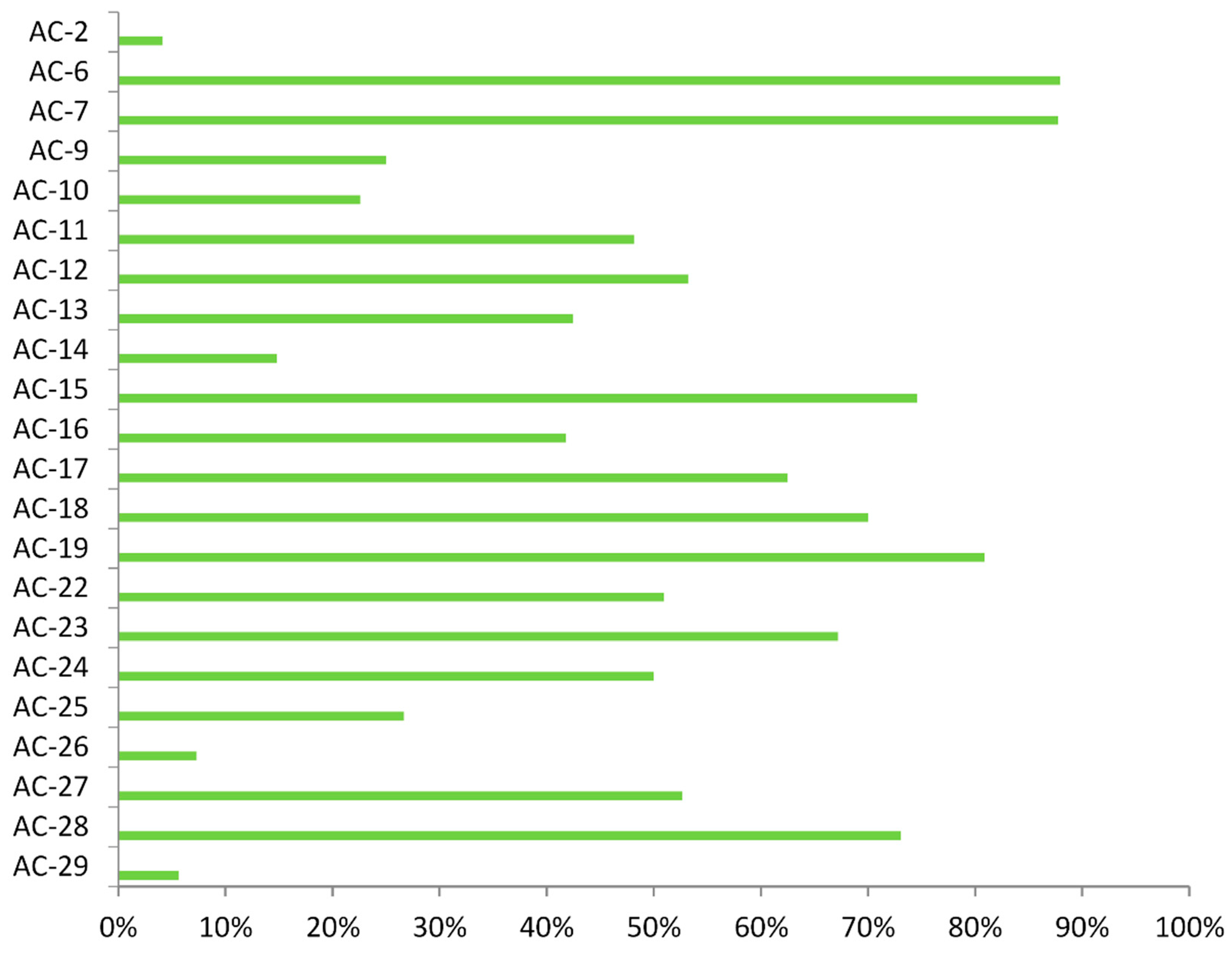
3.3. Clinical Diagnoses
3.4. Simultaneous Presence of Common Immunofluorescence Patterns
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Meroni, P.L.; Schur, P.H. ANA screening: An old test with new recommendations. Ann. Rheum. Dis. 2010, 69, 1420–1422. [Google Scholar] [CrossRef]
- Sur, L.M.; Floca, E.; Sur, D.G.; Colceriu, M.C.; Samasca, G.; Sur, G. Antinuclear Antibodies: Marker of Diagnosis and Evolution in Autoimmune Diseases. Lab. Med. 2018, 49, 62–73. [Google Scholar] [CrossRef] [Green Version]
- Tamirou, F.; Arnaud, L.; Talarico, R.; Scire, C.A.; Alexander, T.; Amoura, Z.; Avcin, T.; Bortoluzzi, A.; Cervera, R.; Conti, F.; et al. Systemic lupus erythematosus: State of the art on clinical practice guidelines. RMD Open 2018, 4, e000793. [Google Scholar] [CrossRef] [PubMed]
- Kavanaugh, A.F.; Solomon, D.H.; American College of Rheumatology Ad Hoc Committee on Immunologic Testing Guidelines. Guidelines for immunologic laboratory testing in the rheumatic diseases: Anti-DNA antibody tests. Arthritis Rheum. 2002, 47, 546. [Google Scholar] [CrossRef] [PubMed]
- Pisetsky, D.S.; Bossuyt, X.; Meroni, P.L. ANA as an entry criterion for the classification of SLE. Autoimmun. Rev. 2019, 18, 102400. [Google Scholar] [CrossRef] [PubMed]
- Vitali, C.; Bombardieri, S.; Jonsson, R.; Moutsopoulos, H.M.; Alexander, E.L.; Carsons, S.E.; Daniels, T.E.; Fox, P.C.; Fox, R.I.; Kassan, S.S.; et al. Classification criteria for Sjögren’s syndrome: A revised version of the European criteria proposed by the American-European Consensus Group. Ann. Rheum. Dis. 2002, 61, 554–558. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Retamozo, S.; Akasbi, M.; Brito-Zerón, P.; Bosch, X.; Bove, A.; Perez-de-Lis, M.; Jimenez, I.; Soto-Cardenas, M.J.; Gandia, M.; Diaz-Lagares, C.; et al. Anti-Ro52 antibody testing influences the classification and clinical characterisation of primary Sjögren’s syndrome. Clin. Exp. Rheumatol. 2012, 30, 686–692. [Google Scholar] [PubMed]
- Allanore, Y.; Simms, R.; Distler, O.; Trojanowska, M.; Pope, J.; Denton, C.P.; Varga, J. Systemic sclerosis. Nat. Rev. Dis. Primers 2015, 1, 15002. [Google Scholar] [CrossRef] [PubMed]
- Dalakas, M.C.; Hohlfeld, R. Polymyositis and dermatomyositis. Lancet 2003, 362, 971. [Google Scholar] [CrossRef]
- Hiepe, F.; Dörner, T.; Burmester, G. Antinuclear antibody- and extractable nuclear antigen-related diseases. Int. Arch. Allergy Immunol. 2000, 123, 5–9. [Google Scholar] [CrossRef] [PubMed]
- Smeenk, R.J. Antinuclear antibodies: Cause of disease or caused by disease? Rheumatology 2000, 39, 581–584. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Claessens, J.; Belmondo, T.; De Langhe, E.; Westhovens, R.; Poesen, K.; Hüe, S.; Blockmans, D.; Mahler, M.; Fritzler, M.J.; Bossuyt, X. Solid phase assays versus automated indirect immunofluorescence for detection of antinuclear antibodies. Autoimmun. Rev. 2018, 17, 533–540. [Google Scholar] [CrossRef]
- Willems, P.; De Langhe, E.; Claessens, J.; Westhovens, R.; van Hoeyveld, E.; Poesen, K.; Vanderschueren, S.; Blockmans, D.; Bossuyt, X. Screening for connective tissue disease-associated antibodies by automated immunoassay. Clin. Chem. Lab. Med. 2018, 56, 909–918. [Google Scholar] [CrossRef]
- Op De Beéck, K.; Vermeersch, P.; Verschueren, P.; Westhovens, R.; Mariën, G.; Blockmans, D.; Bossuyt, X. Antinuclear antibody detection by automated multiplex immunoassay in untreated patients at the time of diagnosis. Autoimmun. Rev. 2012, 12, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Agmon-Levin, N.; Damoiseaux, J.; Kallenberg, C.; Sack, U.; Witte, T.; Herold, M.; Bossuyt, X.; Musset, L.; Cervera, R.; Plaza-Lopez, A.; et al. International recommendations for the assessment of autoantibodies to cellular antigens referred to as anti-nuclear antibodies. Ann. Rheum. Dis. 2014, 73, 17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pisetsky, D.S. Antinuclear antibody testing—Misunderstood or misbegotten? Nat. Rev. Rheumatol. 2017, 13, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Meroni, P.L.; Bizzaro, N.; Cavazzana, I.; Borghi, M.O.; Tincani, A. Automated tests of ANA immunofluorescence as throughput autoantibody detection technology: Strengths and limitations. BMC Med. 2014, 12, 38. [Google Scholar] [CrossRef] [Green Version]
- Haugbro, K.; Nossent, J.C.; Winkler, T.; Figenschau, Y.; Rekvig, O.P. Anti-dsDNA antibodies and disease classification in antinuclear antibody positive patients: The role of analytical diversity. Ann. Rheum. Dis. 2004, 63, 386. [Google Scholar] [CrossRef] [Green Version]
- Op De Beeck, K.; Vermeersch, P.; Verschueren, P.; Westhovens, R.; Mariën, G.; Blockmans, D.; Bossuyt, X. Detection of antinuclear antibodies by indirect immunofluorescence and by solid phase assay. Autoimmun. Rev. 2011, 10, 801–808. [Google Scholar] [CrossRef]
- Olsen, N.J.; Choi, M.Y.; Fritzler, M.J. Emerging technologies in autoantibody testing for rheumatic diseases. Arthritis Res. Ther. 2017, 19, 172. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chan, E.K.; Damoiseaux, J.; Carballo, O.G.; Conrad, K.; de Melo, C.W.; Francescantonio, P.L.; Fritzler, M.J.; la Torre, I.G.; Herold, M.; Mimori, T.; et al. Report of the First International Consensus on Standardized Nomenclature of Antinuclear Antibody HEp-2 Cell Patterns 2014–2015. Front. Immunol. 2015, 6, 412. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chan, E.K.; Damoiseaux, J.; De Melo, C.W.; Carballo, O.G.; Conrad, K.; Francescantonio, P.L.; Fritzler, M.J.; la Torre, I.G.; Herold, M.; Mimori, T.; et al. Report on the second International Consensus on ANA Pattern (ICAP) workshop in Dresden 2015. Lupus 2016, 25, 797–804. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rigon, A.; Infantino, M.; Merone, M.; Iannello, G.; Tincani, A.; Cavazzana, A.; Carabellese, N.; Radice, A.; Manfredi, M.; Soda, P.; et al. The inter-observer reading variability in anti-nuclear antibodies indirect (ANA) immunofluorescence test: A multicenter evaluation and a review of the literature. Autoimmun. Rev. 2017, 16, 1224–1229. [Google Scholar] [CrossRef]
- Pham, B.-N.; Albarede, S.; Guyard, A.; Burg, E.; Maisonneuve, P. Impact of external quality assessment on antinuclear antibody detection performance. Lupus 2005, 14, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Keppeke, G.D.; Calise, S.J.; Chan, E.K.; Andrade, L.E. Anti-rods/rings autoantibody generation in hepatitis C patients during interferon-α/ribavirin therapy. World J. Gastroenterol. 2016, 22, 1966–1974. [Google Scholar] [CrossRef]
- Mahler, M.; Andrade, L.E.; Casiano, C.A.; Malyavantham, K.; Fritzler, M.J. Anti-DFS70 antibodies: An update on our current understanding and their clinical usefulness. Expert Rev. Clin. Immunol. 2019, 15, 241–250. [Google Scholar] [CrossRef]
- World Health Organization. ICD-10 Version 2010. 2010. Available online: https://icd.who.int/browse10/2010/en (accessed on 14 May 2021).
- Hsu, H.C.; Mountz, J.D. Origin of late-onset autoimmune disease. Immunol. Allergy Clin. N. Am. 2003, 23, 65–66. [Google Scholar] [CrossRef]
- Ray, D.; Yung, R. Immune senescence, epigenetics and autoimmunity. Clin. Immunol. 2018, 196, 59–63. [Google Scholar] [CrossRef]
- Mahler, M.; Parker, T.; Peebles, C.L.; Andrade, L.E.; Swart, A.; Carbone, Y.; Ferguson, D.J.; Villalta, D.; Bizzaro, N.; Hanly, J.G.; et al. Anti-DFS70/LEDGF antibodies are more prevalent in healthy individuals compared to patients with systemic autoimmune rheumatic diseases. J. Rheumatol. 2012, 39, 2104–2110. [Google Scholar] [CrossRef]
- Benito-Garcia, E.; Schur, P.H.; Lahita, R.; American College of Rheumatology Ad Hoc Committee on Immunologic Testing Guidelines. Guidelines for immunologic laboratory testing in the rheumatic diseases: Anti-Sm and anti-RNP antibody tests. Arthritis Rheum. 2004, 51, 1030. [Google Scholar] [CrossRef]
- Vermeersch, P.; De Beeck, K.O.; Lauwerys, B.R.; Van den Bergh, K.; Develter, M.; Mariën, G.; Houssiau, F.A.; Bossuyt, X. Antinuclear antibodies directed against proliferating cell nuclear antigen are not specifically associated with systemic lupus erythematosus. Ann. Rheum. Dis. 2009, 68, 1791–1793. [Google Scholar] [CrossRef]
- Ben-Chetrit, E.; Fox, R.I.; Tan, E.M. Dissociation of immune responses to the ss-a (ro) 52-kd and 60-kd polypeptides in systemic lupus erythematosus and Sjögren’s syndrome. Arthritis Rheum. 1990, 33, 349–355. [Google Scholar] [CrossRef]
- Mahler, M.; Miyachi, K.; Peebles, C.; Fritzler, M.J. The clinical significance of autoantibodies to the proliferating cell nuclear antigen (PCNA). Autoimmun. Rev. 2012, 11, 771–775. [Google Scholar] [CrossRef]
- Satoh, M.; Tanaka, S.; Ceribelli, A.; Calise, S.J.; Chan, E.K. A comprehensive overview on myositis-specific antibodies: New and old biomarkers in idiopathic inflammatory myopathy. Clin. Rev. Allergy Immunol. 2017, 52, 1–19. [Google Scholar] [CrossRef] [Green Version]
- Elkon, K.B.; Parnassa, A.P.; Foster, C.L. Lupus autoantibodies target ribosomal P proteins. J. Exp. Med. 1985, 162, 459. [Google Scholar] [CrossRef]
- Sun, X.Y.; Shi, J.; Han, L.; Su, Y.; Li, Z.G. Anti-histones antibodies in systemic lupus erythematosus: Prevalence and frequency in neuropsychiatric lupus. J. Clin. Lab. Anal. 2008, 2, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Andrade, L.E.C.; Klotz, W.; Herold, M.; Conrad, K.; Ronnelid, J.; Fritzler, M.J.; von Mühlen, C.A.; Satoh, M.; Damoiseaux, J.; de Melo Cruvinel, W.; et al. International consensus on antinuclear antibody patterns: Definition of the AC-29 pattern associated with antibodies to DNA topoisomerase I. Clin. Chem. Lab. Med. 2018, 56, 1783–1788. [Google Scholar] [CrossRef] [PubMed]
- van den Hoogen, F.; Khanna, D.; Fransen, J.; Johnson, S.R.; Baron, M.; Tyndall, A.; Matucci-cerinic, M.; Naden, R.P.; Medsger, T.A.; Carreira, P.E.; et al. 2013 classification criteria for systemic sclerosis: An American College of rheumatology/European League against rheumatism collaborative initiative. Ann. Rheum. Dis. 2013, 72, 1747–1755. [Google Scholar] [CrossRef] [Green Version]
- Watanabe, A.; Kodera, M.; Sugiura, K.; Usuda, T.; Tan, E.M.; Takasaki, Y.; Tomita, Y.; Muro, Y. Anti-DFS70 antibodies in 597 healthy hospital workers. Arthritis Rheum. 2004, 50, 892–900. [Google Scholar] [CrossRef]
- Infantino, M.; Bizzaro, N.; Grossi, V.; Manfredi, M. The long-awaited ‘pseudo-DFS pattern’. Expert Rev. Clin. Immunol. 2019, 15, 445. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bhanji, R.A.; Eystathioy, T.; Chan, E.K.; Bloch, D.B.; Fritzler, M.J. Clinical and serological features of patients with autoantibodies to GW/P bodies. Clin. Immunol. 2007, 125, 247–256. [Google Scholar] [CrossRef] [Green Version]
- Miyachi, K.; Hankins, R.W.; Matsushima, H.; Kikuchi, F.; Inomata, T.; Horigome, T.; Shibata, M.; Onozuka, Y.; Ueno, Y.; Hashimoto, E.; et al. Profile and clinical significance of anti-nuclear envelope antibodies found in patients with primary biliary cirrhosis: A multicenter study. J. Autoimmun. 2003, 20, 247–254. [Google Scholar] [CrossRef]
- Conrad, K.; Schössler, W.; Hiepe, F.; Fritzler, M.J. Autoantigens Autoantibodies Autoimmunity, 2nd ed.; Pabst Science: Lengerich, Germany, 2017. [Google Scholar]
- Hong, H.S.; Chung, W.H.; Hung, S.I.; Chen, M.J.; Lee, S.H.; Yang, L.C. Clinical association of anti-golgi autoantibodies and their autoantigens. Scand. J. Immunol. 2004, 59, 79–87. [Google Scholar] [CrossRef]
- Konstantinov, K.; Foisner, R.; Byrd, D.; Liu, F.T.; Tsai, W.M.; Wiik, A.; Gerace, L. Integral membrane proteins associated with the nuclear lamina are novel autoimmune antigens of the nuclear envelope. Clin. Immunol. Immunopathol. 1995, 74, 89–99. [Google Scholar] [CrossRef]
- Murota, M.; Nishioka, M.; Fujita, J.; Dobashi, N.; Wu, F.; Ohtsuki, Y.; Hojo, S.; Takahara, J.; Kuriyama, S. Anti-cytokeratin antibodies in sera of the patients with autoimmune hepatitis. Clin. Exp. Immunol. 2001, 125, 291–299. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.L.; Zhao, F.R.; Hu, Q.; Chen, W.X. Meta-analysis assessment of gp210 and Sp100 for the diagnosis of primary biliary cirrhosis. PLoS ONE 2014, 9, e101916. [Google Scholar] [CrossRef] [Green Version]
- Calise, S.J.; Keppeke, G.D.; Andrade, L.E.; Chan, E.K.L. Anti-rods/rings: A human model of drug-induced autoantibody generation. Front. Immunol. 2015, 6, 41. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wandstrat, A.; Carr-Johnson, F.; Branch, V.; Gray, H.; Fairhurst, A.M.; Reimold, A.; Karp, D.; Wakeland, E.K.; Olsen, N.J. Autoantibody profiling to identify individuals at risk for systemic lupus erythematosus. J. Autoimmun. 2006, 27, 153–160. [Google Scholar] [CrossRef] [PubMed]
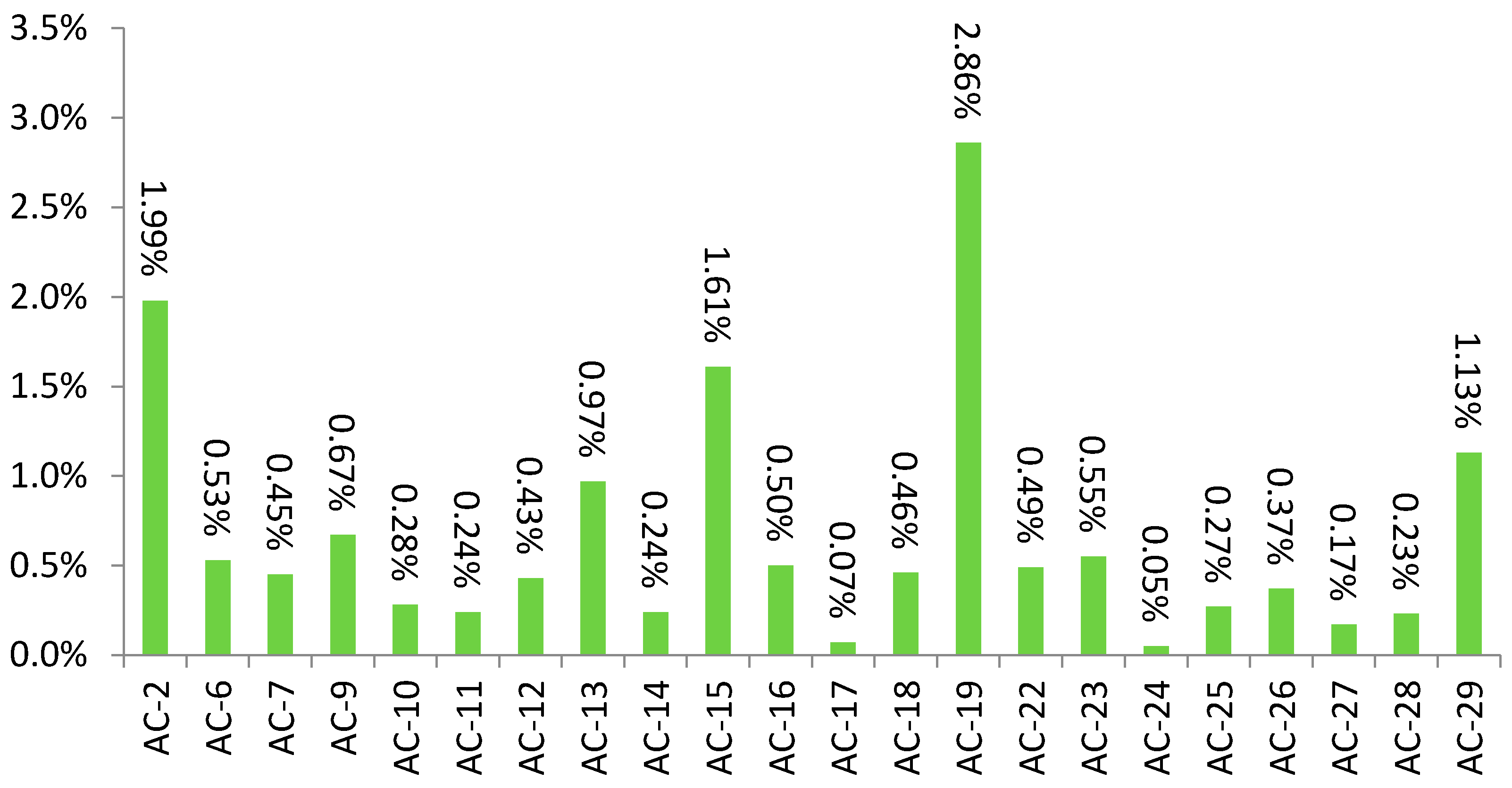


| Category | Subdivision | 2nd Subdivision | AC Designation |
|---|---|---|---|
| Negative | AC-0 | ||
| Nuclear | Homogeneous | AC-1 | |
| Speckled | Dense Fine Speckled | AC-2 | |
| Fine Speckled | AC-4 | ||
| Large/Coarse Speckled | AC-5 | ||
| Topo I | AC-29 | ||
| Centromere | AC-3 | ||
| Discrete Nuclear Dots | Multiple | AC-6 | |
| Few | AC-7 | ||
| Nucleolar | Homogenous | AC-8 | |
| Clumpy | AC-9 | ||
| Punctate | AC-10 | ||
| Nuclear Envelope | Smooth | AC-11 | |
| Punctuate | AC-12 | ||
| Pleomorphic | PCNA | AC-13 | |
| CENP-F | AC-14 | ||
| Cytoplasmic | Fibrillar | Linear | AC-15 |
| Filamentous | AC-16 | ||
| Segmental | AC-17 | ||
| Speckled | Discrete Dots | AC-18 | |
| Dense Fine Speckled | AC-19 | ||
| Fine Speckled | AC-20 | ||
| AMA | AC-21 | ||
| Golgi | AC-22 | ||
| Rods and Rings | AC-23 | ||
| Mitotic | Centrosome | AC-24 | |
| Spindle Fibers | AC-25 | ||
| NuMA | AC-26 | ||
| Intercellular Bridge | AC-27 | ||
| Mitotic Chromosomal | AC-28 |
| N | dsDNA | SS-A 60 | TRIM21 | SS-B | Sm | Sm/RNP | Scl-70 | Jo-1 | Ribosomal | CENP-B | PmScl | PCNA | Histone | U1-RNP | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AC-2 | 218 | 3 1.4% | 5 2.7% | ||||||||||||
| AC-6 | 58 | ||||||||||||||
| AC-7 | 49 | 0% | |||||||||||||
| AC-9 | 73 | 0% | |||||||||||||
| AC-10 | 31 | 0% | |||||||||||||
| AC-11 | 27 | 0% | |||||||||||||
| AC-12 | 47 | ||||||||||||||
| AC-13 | 106 | 82 77.4% | 75 67.9% | 42 39.6% | |||||||||||
| AC-14 | 27 | ||||||||||||||
| AC-15 | 177 | 2 1.1% | |||||||||||||
| AC-16 | 55 | ||||||||||||||
| AC-17 | 8 | ||||||||||||||
| AC-18 | 50 | 9 18% | |||||||||||||
| AC-19 | 314 | 88 28.0% | 63 20.1% | 101 32.2% | 21 6.7% | 37 11.8% | |||||||||
| AC-22 | 54 | ||||||||||||||
| AC-23 | 61 | 2 3.3% | |||||||||||||
| AC-24 | 6 | ||||||||||||||
| AC-25 | 30 | ||||||||||||||
| AC-26 | 41 | ||||||||||||||
| AC-27 | 19 | ||||||||||||||
| AC-28 | 26 | ||||||||||||||
| AC-29 | 142 | 46 32.4% | 23 16.2% | 0% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tomić Sremec, N.; Kozmar, A.; Sremec, J.; Anić, B.; Batinić, D. Properties of Uncommon Indirect Immunofluorescence Staining Patterns Determined during Antinuclear Antibody Detection on HEp-2 Cells. J. Clin. Med. 2021, 10, 3866. https://doi.org/10.3390/jcm10173866
Tomić Sremec N, Kozmar A, Sremec J, Anić B, Batinić D. Properties of Uncommon Indirect Immunofluorescence Staining Patterns Determined during Antinuclear Antibody Detection on HEp-2 Cells. Journal of Clinical Medicine. 2021; 10(17):3866. https://doi.org/10.3390/jcm10173866
Chicago/Turabian StyleTomić Sremec, Nada, Ana Kozmar, Josip Sremec, Branimir Anić, and Drago Batinić. 2021. "Properties of Uncommon Indirect Immunofluorescence Staining Patterns Determined during Antinuclear Antibody Detection on HEp-2 Cells" Journal of Clinical Medicine 10, no. 17: 3866. https://doi.org/10.3390/jcm10173866
APA StyleTomić Sremec, N., Kozmar, A., Sremec, J., Anić, B., & Batinić, D. (2021). Properties of Uncommon Indirect Immunofluorescence Staining Patterns Determined during Antinuclear Antibody Detection on HEp-2 Cells. Journal of Clinical Medicine, 10(17), 3866. https://doi.org/10.3390/jcm10173866





