Prevalence and Prognostic Value of Myocardial Injury in the Initial Presentation of SARS-CoV-2 Infection among Older Adults
Abstract
:1. Introduction
2. Patients and Methods
2.1. Clinical Variables, Data Source, and Study Outcomes
2.2. Ethics Considerations
2.3. Statistical Analysis
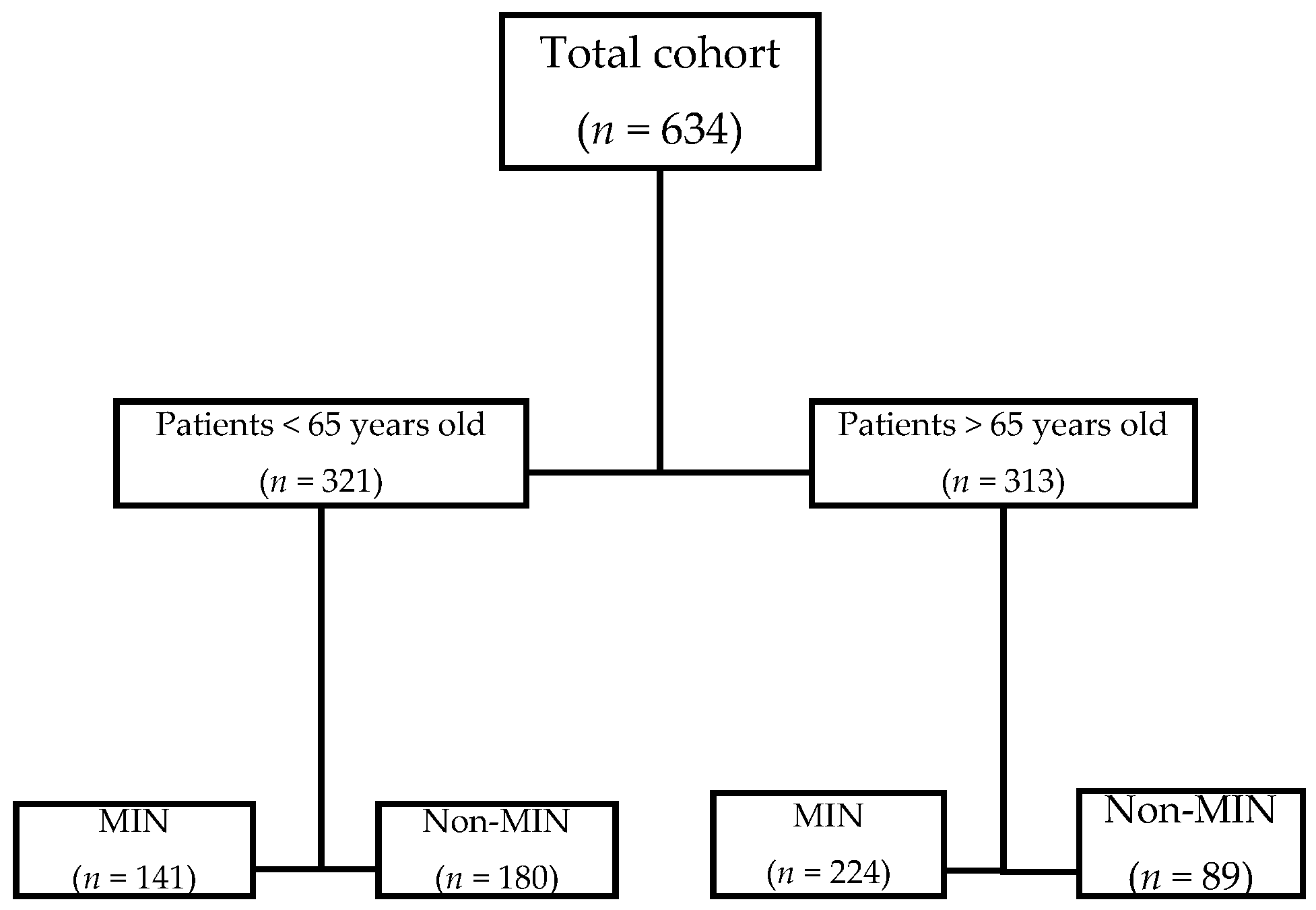
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lippi, G.; Lavie, C.J.; Sanchis-Gomar, F. Cardiac troponin I in patients with coronavirus disease 2019 (COVID-19): Evidence from a meta-analysis. Prog. Cardiovasc. Dis. 2020, 63, 390–391. [Google Scholar] [CrossRef] [PubMed]
- Zou, F.; Qian, Z.; Wang, Y.; Zhao, Y.; Bai, J. Cardiac Injury and COVID-19: A Systematic Review and Meta-analysis. CJC Open 2020, 2, 386–394. [Google Scholar] [CrossRef] [PubMed]
- Napoli, C.; Tritto, I.; Benincasa, G.; Mansueto, G.; Ambrosio, G. Cardiovascular involvement during COVID-19 and clinical implications in elderly patients. A review. Ann. Med. Surg. 2020, 57, 236–243. [Google Scholar] [CrossRef]
- Cao, J.; Tu, W.-J.; Cheng, W.; Yu, L.; Liu, Y.-K.; Hu, X.; Liu, Q. Clinical Features and Short-term Outcomes of 102 Patients with Coronavirus Disease 2019 in Wuhan, China. Clin. Infect. Dis. 2020, 71, 748–755. [Google Scholar] [CrossRef] [PubMed]
- Thygesen, K.; Alpert, J.S.; Jaffe, A.S.; Chaitman, B.R.; Bax, J.J.; Morrow, D.A.; White, H.D.; ESC Scientific Document Group. Fourth universal definition of myocardial infarction. Eur. Heart J. 2019, 40, 237–269. [Google Scholar] [CrossRef] [Green Version]
- Lala, A.; Johnson, K.; Januzzi, J.L.; Russak, A.J.; Paranjpe, I.; Richter, F.; Zhao, S.; Somani, S.; Van Vleck, T.; Vaid, A.; et al. Prevalence and Impact of Myocardial Injury in Patients Hospitalized with COVID-19 Infection. J. Am. Coll. Cardiol. 2020, 76, 533–546. [Google Scholar] [CrossRef]
- Case, B.C.; Yerasi, C.; Forrestal, B.J.; Shea, C.; Rappaport, H.; Medranda, G.A.; Zhang, C.; Abramowitz, J.; Satler, L.F.; Ben-Dor, I.; et al. Clinical impact and predictors of troponin elevation in patients with COVID-19. Cardiovasc. Revasc. Med. 2021. [Google Scholar] [CrossRef]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Cheng, R.; Leedy, D. COVID-19 and acute myocardial injury: The heart of the matter or an innocent bystander? Heart 2020, 106, 1122–1124. [Google Scholar] [CrossRef]
- Berenguer, J.; Ryan, P.; Rodríguez-Baño, J.; Jarrín, I.; Carratala, J.; Pachón, J.; Yllescas, M.; Arriba, J.R.; Muñoz, E.A.; Gil Divasson, P.; et al. Characteristics and predictors of death among 4035 consecutively hospitalized patients with COVID-19 in Spain. Clin. Microbiol. Infect. 2020, 26, 1525–1536. [Google Scholar] [CrossRef] [PubMed]
- Subbe, C.P.; Davies, R.G.; Williams, E.; Rutherford, P.; Gemmell, L. Effect of introducing the Modified Early Warning score on clinical outcomes, cardio-pulmonary arrests and intensive care utilisation in acute medical admissions. Anaesthesia 2003, 58, 797–802. [Google Scholar] [CrossRef]
- Subbe, C.P.; Williams, E.; Fligelstone, L.; Gemmell, L. Does earlier detection of critically ill patients on surgical wards lead to better outcomes? Ann. R. Coll. Surg. Engl. 2005, 87, 226–232. [Google Scholar] [CrossRef]
- Nuzzi, V.; Merlo, M.; Specchia, C.; Lombardi, C.M.; Carubelli, V.; Iorio, A.; Inciardi, R.M.; Bellasi, A.; Canale, C.; Camporotondo, R.; et al. The prognostic value of serial troponin measurements in patients admitted for COVID-19. ESC Heart Fail. 2021. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.J.; Zhang, Y.M.; Yang, M.; Huang, X. Predictors of mortality for patients with COVID-19 pneumonia caused by SARS-CoV-2. Eur. Respir. J. 2020, 56, 2000524. [Google Scholar]
- Shi, S.; Qin, M.; Cai, Y.; Liu, T.; Shen, B.; Yang, F.; Cao, S.; Liu, X.; Xiang, Y.; Zhao, Q.; et al. Characteristics and clinical significance of myocardial injury in patients with severe coronavirus disease 2019. Eur. Heart J. 2020, 41, 2070–2079. [Google Scholar] [CrossRef] [PubMed]
- Zou, L.; Ruan, F.; Huang, M.; Liang, L.; Huang, H.; Hong, Z.; Yu, J.; Kang, M.; Song, Y.; Xia, J.; et al. SARS-CoV-2 Viral Load in Upper Respiratory Specimens of Infected Patients. N. Engl. J. Med. 2020, 382, 1177–1179. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Lu, L.; Cao, W.; Li, T. Hypothesis for potential pathogenesis of SARS-CoV-2 infection—A review of immune changes in patients with viral pneumonia. Emerg. Microbes Infect. 2020, 9, 727–732. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tang, O.; Matsushita, K.; Coresh, J.; Hoogeveen, R.C.; Windham, B.G.; Ballantyne, C.M.; Selvin, E. High-Sensitivity Cardiac Troponin I for Risk Stratification in Older Adults. J. Am. Geriatr. Soc. 2021, 69, 986–994. [Google Scholar] [CrossRef]
- Kang, Y.; Chen, T.; Mui, D.; Ferrari, V.; Jagasia, D.; Scherrer-Crosbie, M.; Chen, Y.; Han, Y. Cardiovascular manifestations and treatment considerations in COVID-19. Heart 2020, 106, 1132–1141. [Google Scholar] [CrossRef]
- Bavishi, C.; Bonow, R.O.; Trivedi, V.; Abbott, J.D.; Messerli, F.H.; Bhatt, D.L. Special Article—Acute myocardial injury in patients hospitalized with COVID-19 infection: A review. Prog. Cardiovasc. Dis. 2020, 63, 682–689. [Google Scholar] [CrossRef]
- Zhou, B.; She, J.; Wang, Y.; Ma, X. The clinical characteristics of myocardial injury in severe and very severe patients with 2019 novel coronavirus disease. J. Infect. 2020, 81, 147–178. [Google Scholar] [CrossRef] [PubMed]
- Shi, S.; Qin, M.; Shen, B.; Cai, Y.; Liu, T.; Yang, F.; Gong, W.; Liu, X.; Liang, J.; Zhao, Q.; et al. Association of Cardiac Injury with Mortality in Hospitalized Patients With COVID-19 in Wuhan, China. JAMA Cardiol. 2020, 5, 802. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Imazio, M.; Klingel, K.; Kindermann, I.; Brucato, A.; De Rosa, F.G.; Adler, Y.; De Ferrari, G.M. COVID-19 pandemic and troponin: Indirect myocardial injury, myocardial inflammation or myocarditis? Heart 2020, 106, 1127–1131. [Google Scholar] [CrossRef]
- Zheng, Y.-Y.; Ma, Y.-T.; Zhang, J.-Y.; Xie, X. COVID-19 and the cardiovascular system. Nat. Rev. Cardiol. 2020, 17, 259–260. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Overall | Myocardial Injury | Without Myocardial Injury | p-Value | ||||
|---|---|---|---|---|---|---|---|
| Cohort Characteristics | n = 313 | n = 224 | n = 89 | ||||
| Median age, years (IQR) | 79 | (73–87) * | 83 | (75–88) | 74 | (68–78) | <0.001 |
| Male sex (%) | 134 | (44%) | 105 | (48%) | 29 | (33%) | 0.022 |
| Long Term Care Facility (%) | 59 | (19%) | 47 | (21%) | 12 | (13%) | 0.231 |
| Dependent for life activities (%) | 42 | (14%) | 39 | (18%) | 3 | (3%) | 0.002 |
| Comorbidities | |||||||
| Current smoker (%) | 11 | (3.5%) | 9 | (4%) | 2 | (2.2%) | 0.442 |
| Hypertension (%) | 230 | (74%) | 175 | (78%) | 55 | (62%) | 0.033 |
| Diabetes Mellitus (%) | 87 | (28%) | 75 | (33%) | 12 | (13%) | <0.001 |
| Chronic lung disease (%) | 31 | (10%) | 25 | (11%) | 6 | (7%) | 0.238 |
| Chronic heart disease (%) | 78 | (25%) | 71 | (32%) | 7 | (8%) | <0.001 |
| Chronic renal disease (%) | 88 | (28%) | 66 | (29%) | 22 | (24%) | 0.400 |
| Chronic liver disease (%) | 17 | (%) | 13 | (6%) | 4 | (4.5%) | 0.645 |
| Dementia (%) | 54 | (17%) | 45 | (20%) | 9 | (3%) | 0.035 |
| ARB-2 (%) | 57 | (18%) | 47 | (21%) | 10 | (11%) | 0.050 |
| ACE inhibitors (%) | 98 | (31%) | 70 | (31%) | 28 | (31%) | 0.971 |
| Estatins (%) | 116 | (37%) | 83 | (37%) | 33 | (37%) | 0.997 |
| Charlson Comorbidy Index, median (IQR) | |||||||
| No comorbidity, n (%) | 81 | (25%) | 41 | (21%) | 40 | (45%) | <0.001 |
| Medium-low (1–2), n (%) | 104 | (33%) | 76 | (34%) | 28 | (31%) | 0.416 |
| High (≥3), n (%) | 128 | (42%) | 107 | (45%) | 21 | (24%) | <0.001 |
| Onset symptoms | |||||||
| Dyspnoea (%) | 158 | (50%) | 119 | (53%) | 39 | (44%) | 0.137 |
| Fever (%) | 222 | (71%) | 146 | (65%) | 76 | (85%) | <0.001 |
| Cough (%) | 203 | (65%) | 131 | (58%) | 72 | (81%) | <0.001 |
| Consciousness impairment (%) | 58 | (18%) | 52 | (23%) | 6 | (7%) | 0.001 |
| Confirmed Pulmonary Emboslism (%) | 15 | (5%) | 9 | (4%) | 6 | (7%) | 0.309 |
| Acute Abnormalities in the EKG (%) | 13 | (4%) | 12 | (5%) | 1 | (1%) | 0.090 |
| Clinical markers at onset | |||||||
| Median C-Reactive Protein mg/dL (IQR) | 7.3 | (3.3–15.4) | 8.68 | (3.8–18.6) | 5.3 | (2.3–11.2) | 0.02 |
| Procalcitonin mg/dL (IQR) | 0.152 | (0.09–0.36) | 0.21 | (0.10–0.54) | 0.10 | (0.07–0.17) | 0.02 |
| Median lymphocyte count /mL (IQR) | 0.955 | (0.65–1.4) | 0.88 | (0.62–1.29) | 1.07 | (0.82–1.63) | 0.001 |
| Median IL-6 pg/mL (IQR) | 49 | (19–103) | 45 | (12–131) | 57 | (25–85) | 0.112 |
| Median Lactate Dehydrogenase UI/l (IQR) | 285 | (232–386) | 288 | (232–411) | 271 | (236–344) | 0.038 |
| Median D-Dimer UI/l (IQR) | 1000 | (620–2200) | 1215 | (680–2540) | 780 | (450–1330) | <0.001 |
| Median Pro-BNP UI/l (IQRS) | 487 | (222–1391) | 861 | (344–3316) | 235 | (101–349) | 0.001 |
| Median Creatinin mg/dl(IQR) | 0.99 | (0.77–1.26) | 1.08 | (0.85–1.47) | 0.79 | (0.67–0.95) | <0.001 |
| Median PaFi (IQR) | 180 | (95–289) | 166 | (91–281) | 219 | (101–310) | 0.213 |
| Median MEWS (IQR) | 2 | (1–3) | 2 | (2–3) | 2 | (1–2) | 0.004 |
| Median Cholesterol mg/mL (IQR) | 134 | (118–161) | 142 | (119–162) | 131 | (113–143) | 0.072 |
| Clinical Outcomes | |||||||
| Median Time to clinical recovery days (IQR) | 12 | (6–20) | 13 | (6–21) | 9 | (5–17) | 0.036 |
| ICU admission (%) | 46 | (15%) | 29 | (14%) | 17 | (19%) | 0.230 |
| Death (%) | 82 | (26%) | 71 | (31%) | 11 | (12%) | <0.001 |
| Odds-Ratio | 95% CI | p-Value | |
|---|---|---|---|
| Myocardial Injury | 2.1 | 1.02-4.42 | 0.043 |
| Age | 1.08 | 1.05-1.11 | <0.001 |
| Sex | 1.11 | 0.98-1.32 | 0.093 |
| Charlson Index | 1.11 | 1.03-1.19 | 0.004 |
| MEWS | 1.125 | 1.01-1.31 | 0.019 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arnau-Barrés, I.; Pascual-Dapena, A.; López-Montesinos, I.; Gómez-Zorrilla, S.; Sorlí, L.; Herrero, M.; Nogués, X.; Montero, M.; Vázquez, O.; García-Giralt, N.; et al. Prevalence and Prognostic Value of Myocardial Injury in the Initial Presentation of SARS-CoV-2 Infection among Older Adults. J. Clin. Med. 2021, 10, 3738. https://doi.org/10.3390/jcm10163738
Arnau-Barrés I, Pascual-Dapena A, López-Montesinos I, Gómez-Zorrilla S, Sorlí L, Herrero M, Nogués X, Montero M, Vázquez O, García-Giralt N, et al. Prevalence and Prognostic Value of Myocardial Injury in the Initial Presentation of SARS-CoV-2 Infection among Older Adults. Journal of Clinical Medicine. 2021; 10(16):3738. https://doi.org/10.3390/jcm10163738
Chicago/Turabian StyleArnau-Barrés, Isabel, Ana Pascual-Dapena, Inmaculada López-Montesinos, Silvia Gómez-Zorrilla, Luisa Sorlí, Marta Herrero, Xavier Nogués, Mila Montero, Olga Vázquez, Natalia García-Giralt, and et al. 2021. "Prevalence and Prognostic Value of Myocardial Injury in the Initial Presentation of SARS-CoV-2 Infection among Older Adults" Journal of Clinical Medicine 10, no. 16: 3738. https://doi.org/10.3390/jcm10163738
APA StyleArnau-Barrés, I., Pascual-Dapena, A., López-Montesinos, I., Gómez-Zorrilla, S., Sorlí, L., Herrero, M., Nogués, X., Montero, M., Vázquez, O., García-Giralt, N., Miralles, R., & Güerri-Fernández, R. (2021). Prevalence and Prognostic Value of Myocardial Injury in the Initial Presentation of SARS-CoV-2 Infection among Older Adults. Journal of Clinical Medicine, 10(16), 3738. https://doi.org/10.3390/jcm10163738







