Paracetamol: A Review of Guideline Recommendations
Abstract
1. Introduction
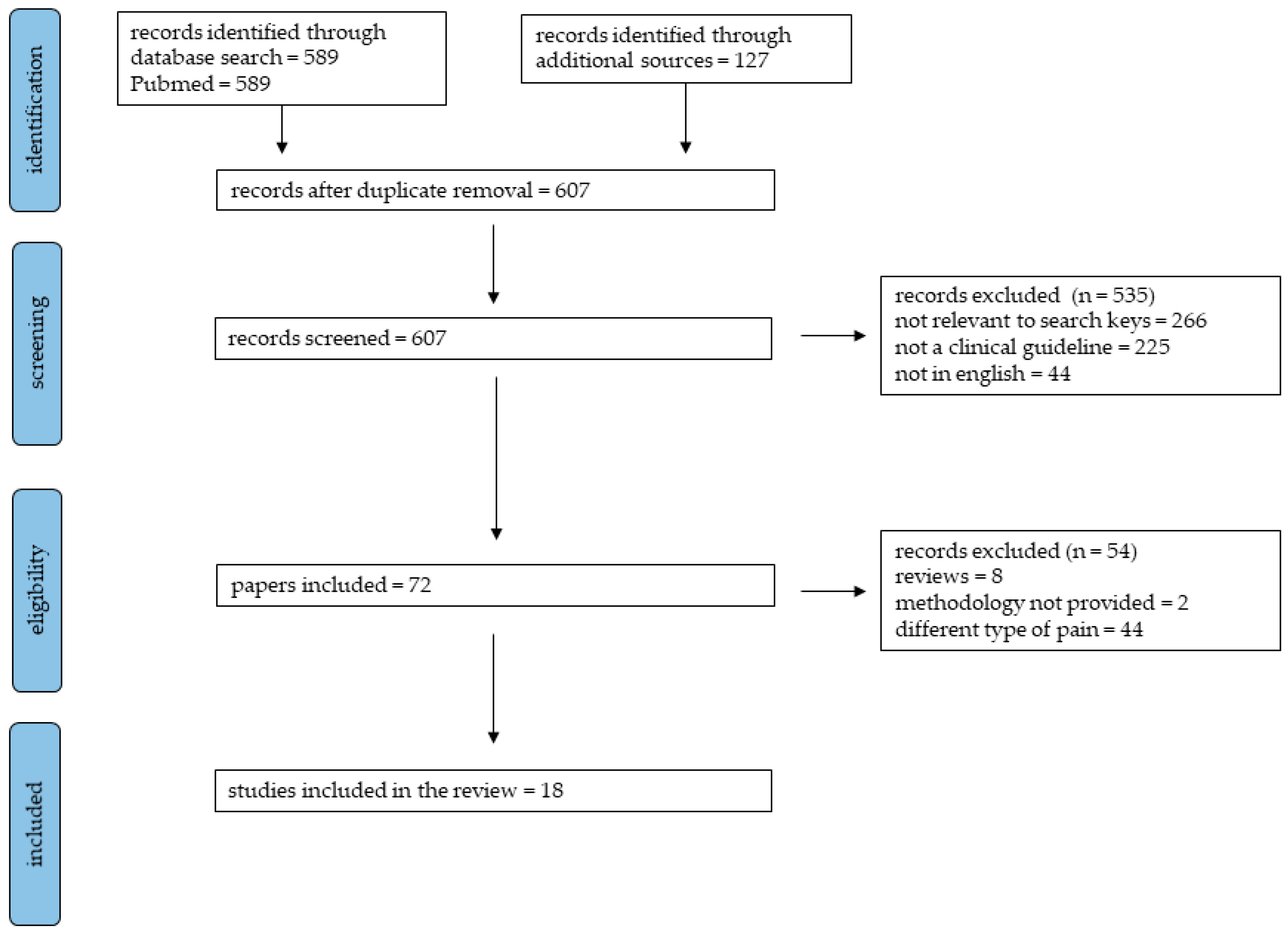
2. Methods
3. Paracetamol
3.1. Acetaminophen or Paracetamol
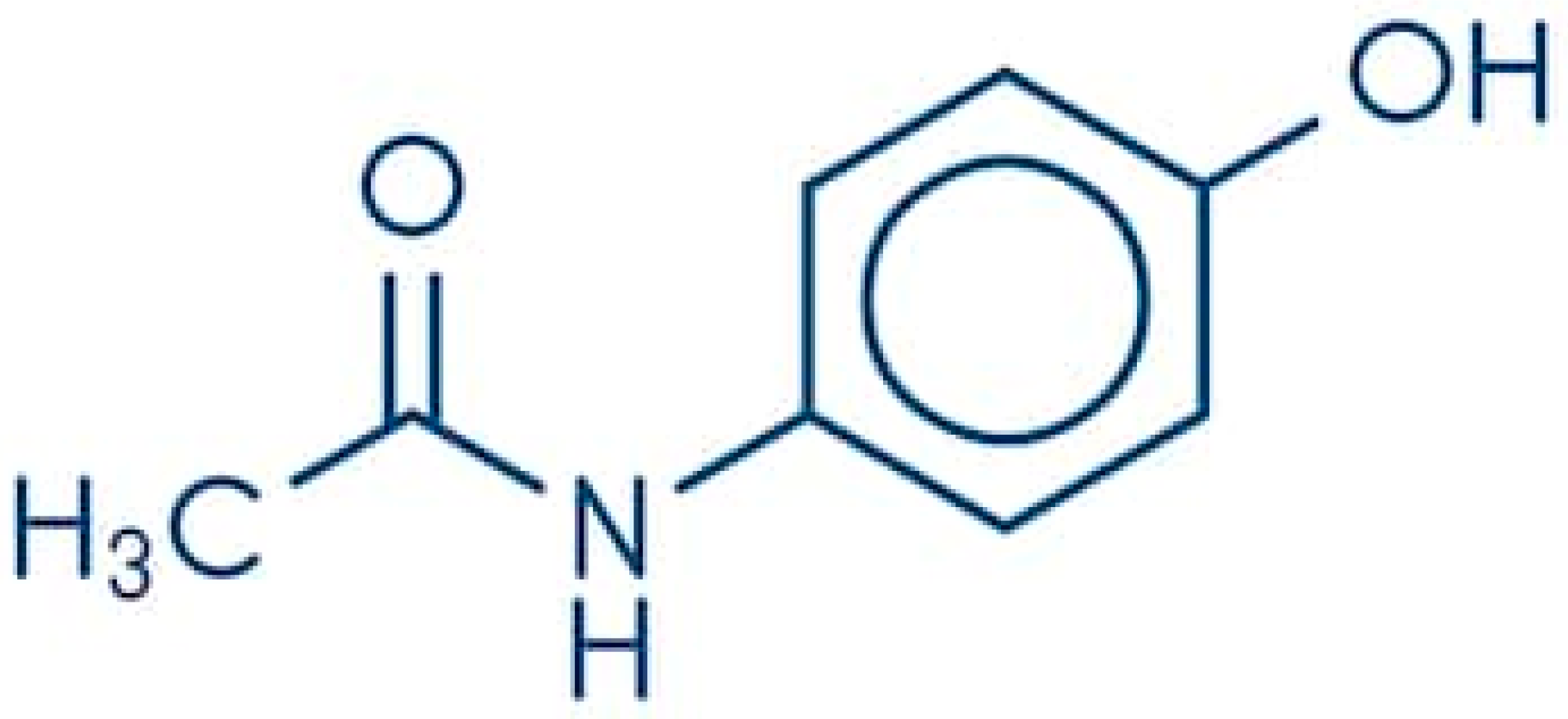
3.2. Chemistry
3.3. Mechanisms of Actions
3.4. Pharmacokinetics
3.5. Oral vs. Intravenous Formulations
4. Pain
5. Clinical Guidelines
5.1. Acute Pain
5.2. Chronic Pain
5.3. Cancer Pain
6. Safety and Toxicity
6.1. Pharmacoepidemiology
6.2. Potential Mechanisms of Paracetamol Toxicity
6.3. Treatments of Paracetamol Toxicity
7. Recent Findings and Future Directions
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Blyth, F.M.; Briggs, A.M.; Schneider, C.H.; Hoy, D.G.; March, L.M. The Global Burden of Musculoskeletal Pain-Where to From Here? Am. J. Public Health 2019, 109, 35–40. [Google Scholar] [CrossRef]
- Sprangers, M.A.; de Regt, E.B.; Andries, F.; van Agt, H.M.; Bijl, R.V.; de Boer, J.B.; Foets, M.; Hoeymans, N.; Jacobs, A.E.; Kempen, I.J.M.; et al. Which chronic conditions are associated with better or poorer quality of life? J. Clin. Epidemiol. 2000, 53, 895–907. [Google Scholar] [CrossRef]
- Thakral, M.; Shi, L.; Foust, J.B.; Patel, K.V.; Shmerling, R.H.; Bean, J.F.; Leveille, S.G. Persistent pain quality as a novel approach to assessing risk for disability in community-dwelling elders with chronic pain. J. Gerontol. A Biol. Sci. Med. Sci. 2018, 39, 450–456. [Google Scholar] [CrossRef]
- Wongrakpanich, S.; Wongrakpanich, A.; Melhado, K.; Rangaswami, J.A. comprehensive review of non-steroidal anti-inflammatory drug use in the elderly. Aging Dis. 2018, 9, 143–150. [Google Scholar] [CrossRef]
- Schjerning Olsen, A.M.; Fosbol, E.L.; Lindhardsen, J.; Folke, F.; Charlot, M.; Selmer, C.; Lamberts, M.; Bjerring Olesen, J.; Køber, L.; Hansen, P.R. Duration of treatment with nonsteroidal anti-inflammatory drugs and impact on risk of death and recurrent myocardial infarction in patients with prior myocardial infarction: A nationwide cohort study. Circulation 2011, 123, 2226–2235. [Google Scholar] [CrossRef]
- Freo, U.; Furnari, M.; Ori, C. Effects of tapentadol on pain, motor symptoms and cognitive functions in Parkinson’s disease. J. Pain Res. 2018, 11, 1849–1856. [Google Scholar] [CrossRef]
- Freo, U.; Furnari, M.; Ambrosio, F.; Navalesi, P. Efficacy and tolerability of tapentadol for the treatment of chronic low back pain in elderly patients. Aging Clin. Exp. Res. 2020, 33, 973–982. [Google Scholar] [CrossRef]
- Pergolizzi, J.; Böger, R.H.; Budd, K.; Dahan, A.; Erdine, S.; Hans, G.; Kress, H.G.; Langford, R.; Likar, R.; Raffa, R.B.; et al. Opioids and the management of chronic severe pain in the elderly: Consensus statement of an international expert panel with focus on the six clinically most often used world health organization step III opioids (Buprenorphine, Fentanyl, Hydromorphone, Methadone, Morphine, Oxycodone). Pain Pract. 2008, 8, 287–313. [Google Scholar] [CrossRef]
- Available online: https://www.drugabuse.gov/drug-topics/trends-statistics/overdose-death-rates (accessed on 5 June 2021).
- Qaseem, A.; McLean, R.M.; O’Gurek, D.; Batur, P.; Lin, K.; Kansagara, D.L. Nonpharmacologic and Pharmacologic Management of Acute Pain From Non-Low Back, Musculoskeletal Injuries in Adults: A Clinical Guideline From the American College of Physicians and American Academy of Family Physicians. Ann. Int. Med. 2020, 173, 739–748. [Google Scholar] [CrossRef]
- Moore, R.A.; Moore, N. Paracetamol and pain: The kiloton problem. Eur. J. Hosp. Pharm. 2016, 23, 187–188. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://wiki.jbi.global (accessed on 2 July 2021).
- Available online: http://www.prisma-statement.org/Extensions/ScopingReviews (accessed on 2 July 2021).
- Available online: https://www.agreetrust.org/agree-ii (accessed on 2 July 2021).
- Kis, B.; Snipes, J.A.; Busija, D.W. Acetaminophen and the cyclooxygenase-3 puzzle: Sorting out facts, fictions, and uncertainties. J. Pharmacol. Exp. Ther. 2005, 315, 1–7. [Google Scholar] [CrossRef]
- Aronoff, D.M.; Oates, J.A.; Boutaud, O. New insights into the mechanism of action of acetaminophen: Its clinical pharmacologic characteristics reflect its inhibition of the two prostaglandin H2 synthases. Clin. Pharmacol. Ther. 2006, 79, 9–19. [Google Scholar] [CrossRef]
- Bertolini, A.; Ferrari, A.; Ottani, A.; Guerzoni, S.; Tacchi, R.; Leone, S. Paracetamol: New vistas of an old drug. CNS Drug Rev. Fall-Winter 2006, 12, 250–275. [Google Scholar] [CrossRef] [PubMed]
- Graham, G.G.; Scott, K.F. Mechanism of action of paracetamol. Am. J. Ther. 2005, 12, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Roberts, L.J., II; Marrow, J.D. Analgesic-antipyretic and antiinflammatory agents and drugs employed in the treatment of gout. In Goodman Gilman’s the Pharmacological Basis of Therapeutics, 10th ed.; Hardman, J.G., Limbird, L.E., Gilman, A.G., Eds.; McGraw Hill: New York, NY, USA, 2001; pp. 687–731. [Google Scholar]
- Högestätt, E.D.; Jönsson, B.A.; Ermund, A.; Andersson, D.A.; Björk, H.; Alexander, J.P.; Cravatt, B.F.; Basbaum, A.I.; Zygmunt, P.M. Conversion of acetaminophen to the bioactive N-acylphenolamine AM404 via fatty acid amide hydrolase-dependent arachidonic acid conjugation in the nervous system. J. Biol. Chem. 2005, 280, 31405–31412. [Google Scholar] [CrossRef]
- Köfalvi, A. Alternative interacting sites and novel receptors for cannabinoid ligands. In Cannabinoids and the Brain; Köfalvi, A., Ed.; Springer: New York, NY, USA, 2008; pp. 131–160. [Google Scholar]
- Ottani, A.; Leone, S.; Sandrini, M.; Ferrari, A.; Bertolini, A. The analgesic activity of paracetamol is prevented by the blockade of cannabinoid CB1 receptors. Eur. J. Pharmacol. 2006, 531, 280–281. [Google Scholar] [CrossRef]
- Bandschapp, O.; Filitz, J.; Urwyler, A.; Koppert, W.; Ruppen, W. Tropisetron blocks analgesic action of acetaminophen: A human pain model study. Pain 2011, 152, 1304–1310. [Google Scholar] [CrossRef]
- Pickering, G.; Faure, M.; Commun, F.; de Boissy, E.C.; Roche, G.; Mom, T.; Simen, E.; Dubray, C.; Eschalier, A.; Gilain, L. Tropisetron and paracetamol association in post-operative patients. Fundam. Clin. Pharmacol. 2012, 26, 432–437. [Google Scholar] [CrossRef]
- Pini, L.A.; Vitale, G.; Ottani, A.; Sandrini, M. Naloxone-reversible antinociception by paracetamol in the rat. J. Pharmacol. Exp. Ther. 1997, 280, 934–940. [Google Scholar]
- Ruggieri, V.; Vitale, G.; Pini, L.A.; Sandrini, M. Differential involvement of opioidergic and serotonergic systems in the antinociceptive activity of N-arachidonoyl-phenolamine (AM404) in the rat: Comparison with paracetamol. Naunyn Schmiedebergs Arch. Pharmacol. 2008, 377, 219–229. [Google Scholar] [CrossRef]
- Pickering, G.; Kastler, A.; Macian, N.; Pereira, B.; Valabrègue, R.; Lehericy, S.; Boyer, L.; Dubray, C.; Jean, B. The brain signature of paracetamol in healthy volunteers: A double-blind randomized trial. Drug Des. Dev. Ther. 2015, 9, 3853–3862. [Google Scholar] [CrossRef]
- Pickering, G.; Macian, N.; Dubray, C.; Pereira, B. Paracetamol sharpens reflection and spatial memory: A double-blind randomized controlled study in healthy volunteers. Drug Des. Dev. Ther. 2016, 10, 3969–3976. [Google Scholar] [CrossRef]
- Borne, R.F. Nonsteroidal Anti-inflammatory Drugs. In Principles of Medicinal Chemistry, 4th ed.; Foye, W.O., Lempke, T.L., Williams, D.A., Eds.; Williams and Wilkins: Baltimore, MD, USA, 1995; pp. 544–545. [Google Scholar]
- Dong, H.; Haining, R.L.; Thummel, K.E.; Rettie, A.E.; Nelson, S.D. Involvement of human cytochrome P450 2D6 in the bioactivation of acetaminophen. Drug Metab. Dispos. 2000, 28, 1397–1400. [Google Scholar]
- Jibril, F.; Sharaby, S.; Mohamed, A.; Wilby, K.J. Intravenous versus oral acetaminophen for pain: Systematic review of current evidence to support clinical decision-making. Can. J. Hosp. Pharm. 2015, 68, 238–247. [Google Scholar] [CrossRef]
- White, P.F. Cost-effective multimodal analgesia in the perioperative period: Use of intravenous vs. oral acetaminophen. J. Clin. Anesth. 2020, 61, 109625. [Google Scholar] [CrossRef]
- Furyk, J.; Levas, D.; Close, B.; Laspina, K.; Fitzpatrick, M.; Robinson, K.; Vangaveti, V.N.; Ray, R. Intravenous versus oral paracetamol for acute pain in adults in the emergency department setting: A prospective, double-blind, double-dummy, randomised controlled trial. Emerg. Med. J. 2018, 35, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Westrich, G.H.; Birch, G.A.; Muskat, A.R.; Padgett, D.E.; Goytizolo, E.A.; Bostrom, M.P.; Mayman, D.J.; Lin, Y.; YaDeau, J.T. Intravenous vs oral acetaminophen as a component of multimodal analgesia after total hip arthroplasty: A randomized, blinded trial. J. Arthroplast. 2019, 34, S215–S220. [Google Scholar] [CrossRef]
- Fenlon, S.; Collyer, J.; Giles, J.; Bidd, H.; Lees, M.; Nicholson, J.; Dulai, R.; Hankins, M.; Edelman, N. Oral vs intravenous paracetamol for lower third molar extractions under general anaesthesia: Is oral administration inferior? Br. J. Anaesth. 2013, 110, 432–437. [Google Scholar] [CrossRef]
- Mallama, M.; Valencia, A.; Rijs, K.; Rietdijk, W.J.R.; Klimek, M.; Calvache, J.A. A systematic review and trial sequential analysis of intravenous vs. oral peri-operative paracetamol. Anaesthesia 2021, 76, 270–276. [Google Scholar] [CrossRef]
- Volkow, N.D. America’s Addiction to Opioids: Heroin and Prescription Drug Abuse. 2014. Available online: https://www.drugabuse.gov/about-nida/legislative-activities/testimony-to-congress/2016/americas-addiction-to-opioids-heroin-prescription-drug-abuse (accessed on 1 July 2020).
- Anson, P. AMA Drops the Pain as the Fifth Vital Sign. 2016. Available online: https://www.painnewsnetwork.org/stories/2016/6/16/ama-drops-pain-as-vital-sign (accessed on 1 July 2020).
- Goode, A.P.; Carey, T.S.; Jordan, J.M. Low back pain and lumbar spine osteoarthritis: How are they related? Curr. Rheumatol. Rep. 2013, 15, 305. [Google Scholar] [CrossRef]
- Hunter, D.J.; Bierma-Zeinstra, S. Osteoarthritis. Lancet 2019, 393, 1745–1759. [Google Scholar] [CrossRef]
- Gignac, M.A.M.; Irvin, E.; Cullen, K.; Eerd, D.V.; Beaton, D.E.; Mahood, Q.; McLeod, C.; Backman, C.L. Men and women’s occupational activities and the risk of developing osteoarthritis of the knee, hip, or hands: A systematic review and recommendations for future research. Arthritis Care Res. 2020, 72, 378–396. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Hong, J.Y.; Han, K.; Suh, S.W.; Park, S.Y.; Yang, J.H.; Han, S.W. Prevalence of symptomatic hip, knee, and spine osteoarthritis nationwide health survey analysis of an elderly Korean population. Medicine 2017, 96, e6372. [Google Scholar] [CrossRef] [PubMed]
- Thakur, M.; Dickenson, A.; Baron, R. Osteoarthritis pain: Nociceptive or neuropathic? Nat. Rev. Rheumatol. 2014, 10, 374–380. [Google Scholar] [CrossRef] [PubMed]
- Minetto, M.A.; Giannini, A.; McConnell, R.; Busso, C.; Torre, G.; Massazza, G. Common musculoskeletal disorders in the elderly: The star triad. J. Clin. Med. 2020, 9, 1216. [Google Scholar] [CrossRef]
- Ryan, C.G.; McDonough, S.; Kirwan, J.P.; Leveille, S.; Martin, D.J. An investigation of association between chronic musculoskeletal pain and cardiovascular disease in the Health Survey for England (2008). Eur. J. Pain 2014, 18, 740–750. [Google Scholar] [CrossRef]
- Wilkie, R.; Parmar, S.S.; Blagojevic-Bucknall, M.; Smith, D.; Thomas, M.J.; Seale, B.J.; Mansell, G.; Peat, G. Reasons why osteoarthritis predicts mortality: Path analysis within a Cox proportional hazards model. BMJ RMD Open 2019, 5, e001048. [Google Scholar] [CrossRef] [PubMed]
- Dixon, J.; Ashton, F.; Baker, P.; Charlton, K.; Bates, C.; Eardley, W. Assessment and early management of pain in hip fractures: The impact of paracetamol. Geriatr. Orthop. Surg. Rehabil. 2018, 9, 2151459318806443. [Google Scholar] [CrossRef]
- Schneider, J.; Algharably, E.; Budnick, A.; Wenzel, A.; Dräger, D.; Kreutz, R. Deficits in pain medication in older adults with chronic pain receiving home care: A cross-sectional study in Germany. PLoS ONE 2020, 15, e0229229. [Google Scholar] [CrossRef]
- Schofield, P. The assessment of pain in older people: UK National guidelines. Age Ageing 2018, 47 (Suppl. 1), i1–i22. [Google Scholar] [CrossRef]
- Rochon, P.A.; Fortin, P.R.; Dear, K.B.G.; Minaker, K.L.; Chalmers, T.C. Reporting of age data in clinical trials of arthritis: Deficiencies and solutions. Arch. Intern. Med. 1993, 153, 243–248. [Google Scholar] [CrossRef]
- McLachlan, A.J.; Bath, S.; Naganathan, V.; Hilmer, S.N.; Le Couteur, D.G.; Gibson, S.J.; Blyth, F.M. Clinical pharmacology of analgesic medicines in older people: Impact of frailty and cognitive impairment. Br. J. Clin. Pharmacol. 2011, 71, 351–364. [Google Scholar] [CrossRef]
- Arahata, M.; Asakura, H. Antithrombotic therapies for elderly patients: Handling problems originating from their comorbidities. Clin. Interv. Aging 2018, 13, 1675–1690. [Google Scholar] [CrossRef] [PubMed]
- Head, T.; Daunert, S.; Goldschmidt-Clermont, P.J. The aging risk and atherosclerosis: A fresh look at arterial homeostasis. Front. Genet. 2017, 8, 216. [Google Scholar] [CrossRef]
- Kadam, U.T.; Jordan, K.; Croft, P.R. Clinical comorbidity in patients with osteoarthritis: A case-control study of general practice consulters in England and Wales. Ann. Rheum. Dis. 2004, 63, 408–414. [Google Scholar] [CrossRef]
- Hunter, D.J. Osteoarthritis: Time for us all to shift the needle. Rheumatology 2018, 57 (Suppl. 4), iv1–iv2. [Google Scholar] [CrossRef]
- Miller, S.R.; Littenberg, B.; MacLean, C.D. Prevalence of antiplatelet therapy in patients with diabetes. Cardiovasc. Diabetol. 2005, 4, 18. [Google Scholar] [CrossRef]
- Caraceni, A.; Shkodra, M. Cancer Pain Assessment and Classification. Cancers 2019, 11, 510. [Google Scholar] [CrossRef]
- Temel, J.S.; Greer, J.A.; Muzikansky, A.; Gallagher, E.R.; Admane, S.; Jackson, V.A.; Dahlin, C.M.; Blinderman, C.D.; Jacobsen, J.; Pirl, W.F.; et al. Early palliative care for patients with metastatic non-small-cell lung cancer. N. Eng. J. Med. 2010, 363, 733–742. [Google Scholar] [CrossRef]
- Guyatt, G.H.; Oxman, A.D.; Kunz, R.; Vist, G.E.; Falck-Ytter, Y.; Schünemann, H.J.; GRADE Working Group. What is “quality of evidence” and why is it important to clinicians? BMJ 2008, 336, 995–998. [Google Scholar] [CrossRef]
- Hung, K.; Graham, C.A.; Lo, R.; Leung, Y.K.; Leung, L.Y.; Man, S.Y.; Woo, W.K.; Cattermole, G.N.; Rainer, T.H. Oral paracetamol and/or ibuprofen for treating pain after soft tissue injuries: Single centre double-blind, randomised controlled clinical trial. PLoS ONE 2018, 13, e0192043. [Google Scholar] [CrossRef]
- Man, S.Y.; Woo, W.K.; Lam, P.K.W.; Reiner, T.H. Feasibility study comparing oral paracetamol and oral non-steroidal anti-inflammatory drugs for treating pain after musculoskeletal injury: A randomised, double blind, controlled trial. Hong Kong J. Emerg. Med. 2004, 11, 78–84. [Google Scholar] [CrossRef]
- Ridderikhof, M.L.; Lirk, P.; Goddijn, H.; Vandewalle, E.; Schinkel, E.; Van Dieren, S.; Kemper, E.M.; Hollmann, M.W.; Goslings, J.C. Acetaminophen or Nonsteroidal Anti-Inflammatory Drugs in Acute Musculoskeletal Trauma: A Multicenter, Double-Blind, Randomized, Clinical Trial. Ann. Emerg. Med. 2018, 71, 357–368.e8. [Google Scholar] [CrossRef] [PubMed]
- Woo, W.W.; Man, S.Y.; Lam, P.K.; Rainer, T.H. Randomized double-blind trial comparing oral paracetamol and oral nonsteroidal antiinflammatory drugs for treating pain after musculoskeletal injury. Ann. Emerg. Med. 2005, 46, 352–361. [Google Scholar] [CrossRef]
- Jones, P.; Dalziel, S.R.; Lamdin, R.; Miles-Chan, J.L.; Frampton, C. Oral non-steroidal anti-inflammatory drugs versus other oral analgesic agents for acute soft tissue injury. Cochrane Database Syst. Rev. 2015, 7, CD007789. [Google Scholar] [CrossRef] [PubMed]
- Hyllested, M.; Jones, S.; Pedersen, J.L.; Kehlet, H. Comparative effect of paracetamol, NSAIDs or their combination in postoperative pain management: A qualitative review. Br. J. Anaesth. 2002, 88, 199–214. [Google Scholar] [CrossRef] [PubMed]
- Pincus, T.; Koch, G.G.; Sokka, T.; Lefkowith, J.; Wolfe, F.; Jordan, J.M.; Luta, G.; Callahan, L.F.; Wang, X.; Schwartz, T. A randomized, double-blind, crossover clinical trial of diclofenac plus misoprostol versus acetaminophen in patients with osteoarthritis of the hip or knee. Arthritis Rheum. 2001, 44, 1587–1598. [Google Scholar] [CrossRef]
- Pathan, S.A.; Mitra, B.; Straney, L.D.; Afzal, M.S.; Anjum, S.; Shukla, D.; Morley, K.; Al Hilli, S.A.; Al Rumaihi, K.; Thomas, S.H.; et al. Delivering safe and effective analgesia for management of renal colic in the emergency department: A double-blind, multigroup, randomised controlled trial. Lancet 2016, 387, 1999–2007. [Google Scholar] [CrossRef]
- Blok, Z.; Ridderikhof, M.L.; Goddijn, H.; Berendsen, M.; Hollmann, M.W. Intravenous acetaminophen does not have an opioid sparing effect in Emergency Department patients with painful conditions. Am. J. Emerg. Med. 2021, 39, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Ridderikhof, M.L.; Saanen, J.; Goddijn, H.; Van Dieren, S.; Van Etten-Jamaludin, F.; Lirk, P.; Goslings, J.C.; Hollmann, M.W. Paracetamol versus other analgesia in adult patients with minor musculoskeletal injuries: A systematic review. Emerg. Med. J. 2019, 36, 493–500. [Google Scholar] [CrossRef]
- Oliveira, C.B.; Maher, C.G.; Pinto, R.Z.; Traeger, A.C.; Lin, C.C.; Chenot, J.F.; van Tulder, M.; Koes, B.W. Clinical practice guidelines for the management of non-specific low back pain in primary care: An updated overview. Eur. Spine J. 2018, 27, 2791–2803. [Google Scholar] [CrossRef]
- Wick, E.C.; Grant, M.C.; Wu, C.L. Postoperative multimodal analgesia pain management with nonopioid analgesics and techniques: A review. JAMA Surg. 2017, 152, 691–697. [Google Scholar] [CrossRef]
- Miranda, H.F.; Puig, M.M.; Prieto, J.C.; Pinardi, G. Synergism between paracetamol and nonsteroidal anti-inflammatory drugs in experimental acute pain. Pain 2006, 121, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Zeidan, A.; Mazoit, J.X.; Ali Abdullah, M.; Maaliki, H.; Ghattas, T.; Saifan, A. Median effective dose (ED₅₀) of paracetamol and morphine for postoperative pain: A study of interaction. Br. J. Anaesth. 2014, 112, 118–123. [Google Scholar] [CrossRef]
- Naples, J.G.; Gellad, W.F.; Hanlon, J.T. The role of opioid analgesics in geriatric pain management. Clin. Geriatr. Med. 2016, 32, 725–735. [Google Scholar] [CrossRef] [PubMed]
- Moore, R.A.; Derry, S.; Aldington, D.; Wiffen, P.J. Single dose oral analgesics for acute postoperative pain in adults—An overview of Cochrane reviews. Cochrane Database Syst. Rev. 2015, 9, CD008659. [Google Scholar] [CrossRef]
- Moore, R.A.; Wiffen, P.J.; Derry, S.; Maguire, T.; Roy, Y.M.; Tyrrell, L. Non-prescription (OTC) oral analgesics for acute pain—An overview of Cochrane reviews. Cochrane Database Syst. Rev. 2015, 11, CD010794. [Google Scholar] [CrossRef]
- Caughey, G.E.; James, M.J.; Proudman, S.M.; Cleland, L.G. Fish oil supplementation increases the cyclooxygenase inhibitory activity of paracetamol in rheumatoid arthritis patients. Complement. Ther. Med. 2010, 18, 171–174. [Google Scholar] [CrossRef]
- Marmura, M.J.; Silberstein, S.D.; Schwedt, T.J. The acute treatment of migraine in adults: The American Headache Society evidence assessment of migraine pharmacotherapies. Headache 2015, 55, 3–20. [Google Scholar] [CrossRef] [PubMed]
- Lipton, R.B.; Baggish, J.S.; Stewart, W.F.; Codispoti, M. Efficacy and safety of acetaminophen in the treatment of migraine: Results of a randomized, double-blind, placebo-controlled, population-based study. Arch. Intern. Med. 2000, 160, 3486–3492. [Google Scholar] [CrossRef] [PubMed]
- Leinisch, E.; Evers, S.; Kaempfe, N.; Kraemer, C.; Sostak, P.; Jürgens, T.; Straube, A.; May, A. Evaluation of the efficacy of intravenous acetaminophen in the treatment of acute migraine attacks: A double-blind, placebo-controlled parallel group multicenter study. Pain 2005, 117, 396–400. [Google Scholar] [CrossRef] [PubMed]
- Sarchielli, P.; Granella, F.; Prudenzano, M.P.; Pini, L.A.; Guidetti, V.; Bono, G.; Pinessi, L.; Alessandri, M.; Antonaci, F.; Fanciullacci, M.; et al. Italian guidelines for primary headaches: 2012 revised version. J. Headache Pain 2012, 13 (Suppl. 2), S31–S70. [Google Scholar] [CrossRef] [PubMed]
- Snow, V.; Weiss, K.; Wall, E.M.; Mottur-Pilson, C. American Academy of Family Physicians; American College of Physicians-American Society of Internal Medicine. Pharmacologic management of acute attacks of migraine and prevention of migraine headache. Ann. Intern. Med. 2002, 137, 840–849. [Google Scholar] [CrossRef] [PubMed]
- Bendtsen, L.; Evers, S.; Linde, M.; Mitsikostas, D.D.; Sandrini, G.; Schoenen, J. EFNS guideline on the treatment of tension-type headache—Report of an EFNS task force. Eur. J. Neurol. 2010, 17, 1318–1325. [Google Scholar] [CrossRef] [PubMed]
- Marcum, Z.A.; Duncan, N.A.; Makris, U.E. Pharmacotherapies in geriatric chronic pain management. Clin. Geriatr. Med. 2016, 32, 705–724. [Google Scholar] [CrossRef]
- Temple, A.R.; Benson, G.D.; Zinsenheim, J.R.; Schweinle, J.E. Multicenter, randomized, double-blind, active-controlled, parallel-group trial of the long-term (6-12 months) safety of acetaminophen in adult patients with osteoarthritis. Clin. Ther. 2006, 28, 222–235. [Google Scholar] [CrossRef]
- Towheed, T.E.; Maxwell, L.; Judd, M.G.; Catton, M.; Hochberg, M.C.; Wells, G. Acetaminophen for osteoarthritis. Cochrane Database Syst. Rev. 2006, 1, CD004257. [Google Scholar] [CrossRef]
- Ambrosio, F.; Finco, G.; Mattia, C.; Mediati, R.; Paoletti, F.; Coluzzi, F.; Piacevoli, Q.; Savoia, G.; Amantea, B.; Aurilio, C. SIAARTI Chronic Non-Cancer Pain Study Group. SIAARTI recommendations for chronic noncancer pain. Minerva Anestesiol. 2006, 72, 859–880. [Google Scholar] [PubMed]
- Verkleij, S.P.; Luijsterburg, P.A.; Bohnen, A.M.; Koes, B.W.; Bierma-Zeinstra, S.M. NSAIDs vs acetaminophen in knee and hip osteoarthritis: A systematic review regarding heterogeneity influencing the outcomes. Osteoarthr. Cartil. 2011, 19, 921–929. [Google Scholar] [CrossRef]
- Zhang, W.; Jones, A.; Doherty, M. Does paracetamol (acetaminophen) reduce the pain of osteoarthritis? A meta-analysis of randomised controlled trials. Ann. Rheum. Dis. 2004, 63, 901–907. [Google Scholar] [CrossRef] [PubMed]
- Nelson, A.E.; Allen, K.D.; Golightly, Y.M.; Goode, A.P.; Jordan, J.M. A systematic review of recommendations and guidelines for the management of osteoarthritis: The chronic osteoarthritis management initiative of the U.S. bone and joint initiative. Semin. Arthritis Rheum. 2014, 43, 701–712. [Google Scholar] [CrossRef]
- Nikles, C.J.; Yelland, M.; Del Mar, C.; Wilkinson, D. The role of paracetamol in chronic pain: An evidence-based approach. Am. J. Ther. 2005, 12, 80–91. [Google Scholar] [CrossRef]
- Pendleton, A.; Arden, N.; Dougados, M.; Doherty, M.; Bannwarth, B.; Bijlsma, J.W.; Cluzeau, F.; Cooper, C.; Dieppe, P.A.; Günther, K.P.; et al. EULAR recommendations for the management of knee osteoarthritis: Report of a task force of the Standing Committee for International Clinical Studies Including Therapeutic Trials (ESCISIT). Ann. Rheum. Dis. 2000, 59, 936–944. [Google Scholar] [CrossRef]
- Zhang, W.; Doherty, M.; Arden, N.; Bannwarth, B.; Bijlsma, J.; Gunther, K.P.; Hauselmann, H.J.; Herrero-Beaumont, G.; Jordan, K.; Kaklamanis, P.; et al. EULAR evidence based recommendations for the management of hip osteoarthritis: Report of a task force of the EULAR Standing Committee for International Clinical Studies Including Therapeutics (ESCISIT). Ann. Rheum. Dis. 2005, 64, 669–861. [Google Scholar] [CrossRef]
- Hochberg, M.C.; Altman, R.D.; April, K.T.; Benkhalti, M.; Guyatt, G.; McGowan, J.; Towheed, T.; Welch, V.; Wells, G.; Tugwell, P. American College of Rheumatology Recommendations for the use of nonpharmacologic and pharmacologic therapies in osteoarthritis of the hand, hip, and knee. Arthritis Care Res. 2012, 64, 465–474. [Google Scholar] [CrossRef]
- Available online: https://www.nice.org.uk/guidance/cg177/resources/osteoarthritis-care-and-management-pdf-35109757272517 (accessed on 5 June 2021).
- Fillingham, Y.A.; Hannon, C.P.; Erens, G.A.; Mullen, K.; Casambre, F.; Visvabharathy, V.; Hamilton, W.G.; Della Valle, C.J. The Efficacy and Safety of Acetaminophen in Total Joint Arthroplasty: Systematic Review and Direct Meta-Analysis. J. Arthroplast. 2020, 35, 2715–2729. [Google Scholar] [CrossRef]
- Zhang, W.; Nuki, G.; Moskowitz, R.W.; Abramson, S.; Altman, R.D.; Arden, N.K.; Bierma-Zeinstra, S.; Brandt, K.D.; Croft, P.; Doherty, M.; et al. OARSI recommendations for the management of hip and knee osteoarthritis: Part III: Changes in evidence following systematic cumulative update of research published through January 2009. Osteoarthr. Cartil. 2010, 18, 476–499. [Google Scholar] [CrossRef]
- Arden, N.K.; Perry, T.A.; Bannuru, R.R.; Bruyère, O.; Cooper, C.; Haugen, I.K.; Hochberg, M.C.; McAlindon, T.E.; Mobasheri, A.; Reginster, J.Y. Non-surgical management of knee osteoarthritis: Comparison of ESCEO and OARSI 2019 guidelines. Nat. Rev. Rheumatol. 2021, 17, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Leopoldino, A.O.; Machado, G.C.; Ferreira, P.H.; Pinheiro, M.B.; Day, R.; McLachlan, A.J.; Hunter, D.J.; Ferreira, M.L. Paracetamol versus placebo for knee and hip osteoarthritis. Cochrane Database Syst. Rev. 2019, 2, CD013273. [Google Scholar] [CrossRef]
- Kolasinski, S.L.; Neogi, T.; Hochberg, M.C.; Oatis, C.; Guyatt, G.; Block, J.; Callahan, L.; Copenhaver, C.; Dodge, C.; Felson, D.; et al. 2019 American College of Rheumatology/Arthritis Foundation Guideline for the Management of Osteoarthritis of the Hand, Hip, and Knee. Arthritis Rheumatol. 2020, 72, 220–233. [Google Scholar] [CrossRef]
- O’Neil, C.K.; Hanlon, J.T.; Marcum, Z.A. Adverse effects of analgesics commonly used by older adults with osteoarthritis: Focus on non-opioid and opioid analgesics. Am. J. Geriatr. Pharmacother. 2012, 10, 331–342. [Google Scholar] [CrossRef] [PubMed]
- Wise, J. NICE keeps paracetamol in UK guidelines on osteoarthritis. BMJ 2014, 348, g1545. [Google Scholar] [CrossRef]
- Makris, U.E.; Abrams, R.C.; Gurland, B.; Reid, M.C. Management of persistent pain in the older patient: A clinical review. JAMA 2014, 312, 825–836. [Google Scholar] [CrossRef]
- Reid, M.C.; Shengelia, R.; Parker, S.-J. Pharmacologic management of osteoarthritis-related pain in older adults: A review shows that many drug therapies provide small-to-modest pain relief. HSS J. 2012, 8, 159–164. [Google Scholar] [CrossRef]
- American Geriatrics Society Panel on Pharmacological Management of Persistent Pain in Older Persons. Pharmacological management of persistent pain in older persons. J. Am. Geriatr. Soc. 2009, 57, 1331–1346. [Google Scholar] [CrossRef]
- Abdulla, A.; Adams, N.; Bone, M.; Elliott, A.M.; Gaffin, J.; Jones, D.; Knaggs, R.; Martin, D.; Sampson, L.; Schofield, P. Geriatric Society on the management of pain in older people. Age Ageing 2013, 42S1, i1–i57. [Google Scholar] [CrossRef]
- Motter, F.R.; Hilmer, S.N.; Paniz, V.M.V. Pain and inflammation management in older adults: A brazilian consensus of potentially inappropriate medication and their alternative therapies. Front. Pharmacol. 2019, 10, 1408. [Google Scholar] [CrossRef] [PubMed]
- Cravello, L.; Di Santo, S.; Varrassi, G.; Benincasa, D.; Marchettini, P.; de Tommaso, M.; Shofany, J.; Assogna, F.; Perotta, D.; Palmer, K.; et al. Chronic pain in the elderly with cognitive decline: A narrative review. Pain Ther. 2019, 8, 53–65. [Google Scholar] [CrossRef]
- Malotte, K.L.; McPherson, M.L. Identification, assessment, and management of pain in patients with advanced dementia. Ment. Health Clin. 2016, 6, 89–94. [Google Scholar] [CrossRef]
- Flo, E.; Gulla, C.; Husebo, B.S. Effective pain management in patients with dementia: Benefits beyond pain? Drugs Aging 2014, 31, 863–871. [Google Scholar] [CrossRef] [PubMed]
- Sandvik, R.K.; Selbaek, G.; Seifert, R.; Aarsland, D.; Ballard, C.; Corbett, A.; Husebo, B.S. Impact of a stepwise protocol for treating pain on pain intensity in nursing home patients with dementia: A cluster randomized trial. Eur. J. Pain 2014, 18, 1490–1500. [Google Scholar] [CrossRef]
- Major-Monfried, H.; DeCherrie, L.V.; Wajnberg, A.; Zhang, M.; Kelley, A.S.; Ornstein, K.A. Managing pain in chronically ill homebound patients through home-based primary and palliative care. Am. J. Hosp. Palliat. Care 2019, 36, 333–338. [Google Scholar] [CrossRef]
- Shorter, E.; Sannicandro, A.J.; Poulet, B.; Goljanek-Whysall, K. Skeletal muscle wasting and its relationship with osteoarthritis: A mini-review of mechanisms and current interventions. Curr. Rheumatol. Rep. 2019, 21, 40. [Google Scholar] [CrossRef]
- Carroll, C.C.; Dickinson, J.M.; LeMoine, J.K.; Haus, J.M.; Weinheimer, E.M.; Hollon, C.J.; Aagaard, P.; Magnusson, S.P.; Trappe, T.A. Influence of acetaminophen and ibuprofen on in vivo patellar tendon adaptations to knee extensor resistance exercise in older adults. J. Appl. Physiol. 2011, 111, 508–515. [Google Scholar] [CrossRef]
- Trappe, T.A.; Carroll, C.C.; Dickinson, J.M.; LeMoine, J.K.; Haus, J.M.; Sullivan, B.E.; Lee, J.D.; Jemiolo, B.; Weinheimer, E.M.; Hollon, C.J. Influence of acetaminophen and ibuprofen on skeletal muscle adaptations to resistance exercise in older adults. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2011, 300, R655–R662. [Google Scholar] [CrossRef]
- Koes, B.; Schreijenberg, M.; Tkachev, A. Paracetamol for low back pain: The state of the research field. Exp. Rev. Clin. Pharmacol. 2020, 13, 1059–1066. [Google Scholar] [CrossRef]
- Morlion, B. Chronic low back pain: Pharmacological, interventional and surgical strategies. Nat. Rev. Neurol. 2013, 9, 462–473. [Google Scholar] [CrossRef] [PubMed]
- Saragiotto, B.T.; Machado, G.C.; Ferreira, M.L.; Pinheiro, M.B.; Abdel Shaheed, C.; Maher, C.G. Paracetamol for low back pain. Cochrane Database Syst Rev. 2016, 6, CD012230. [Google Scholar] [CrossRef]
- Low Back Pain EBM Guidelines 18 September 2019. Available online: https://www.ebmguidelines.com/ebmg/ltk.free?p_artikkeli=ebm00435 (accessed on 2 July 2020).
- van der Heijde, D.; Ramiro, S.; Landewé, R.; Baraliakos, X.; Van den Bosch, F.; Sepriano, A.; Regel, A.; Ciurea, A.; Dagfinrud, H.; Dougados, M.; et al. 2016 update of the ASAS-EULAR management recommendations for axial spondyloarthritis. Ann. Rheum. Dis. 2017, 76, 978–991. [Google Scholar] [CrossRef]
- World Health Organization Model List of Essential Medicines, 21st List. 2019. Available online: https://apps.who.int/iris/bitstream/handle/10665/325771/WHO-MVP-EMP-IAU-2019.06-eng.pdf?ua=1 (accessed on 1 July 2020).
- Portenoy, R.K.; Ahmed, E.; Keilson, Y.Y. Cancer Pain Management: Use of Acetaminophen and Nonsteroidal Antiinflammatory Drugs. Available online: https://www.uptodate.com/contents/cancer-pain-management-use-of-acetaminophen-and-nonsteroidal-antiinflammatory-drugs?source=related_link (accessed on 2 July 2020).
- Stockler, M.; Vardy, J.; Pillai, A.; Warr, D. Acetaminophen (paracetamol) improves pain and well-being in people with advanced cancer already receiving a strong opioid regimen: A randomized, double-blind, placebo-controlled cross-over trial. J. Clin. Oncol. 2004, 22, 3389–3394. [Google Scholar] [CrossRef]
- Wiffen, P.J.; Derry, S.; Moore, R.A.; McNicol, E.D.; Bell, R.F.; Carr, D.B.; McIntyre, M.; Wee, B. Oral paracetamol (acetaminophen) for cancer pain. Cochrane Database Syst. Rev. 2017, 7, CD012637. [Google Scholar] [CrossRef] [PubMed]
- Fallon, M.; Giusti, R.; Aielli, F.; Hoskin, P.; Rolke, R.; Sharma, M.; Ripamonti, C.I.; ESMO Guidelines Committee. Management of cancer pain in adult patients: ESMO Clinical Practice Guidelines. Ann. Oncol. 2018, 29 (Suppl. 4), iv166–iv191. [Google Scholar] [CrossRef]
- Huang, R.; Jiang, L.; Cao, Y.; Liu, H.; Ping, M.; Li, W.; Xu, Y.; Ning, J.; Chen, Y.; Wang, X. Comparative Efficacy of Therapeutics for Chronic Cancer Pain: A Bayesian Network Meta-Analysis. J. Clin. Oncol. 2019, 37, 1742–1752. [Google Scholar] [CrossRef]
- Hachem, G.E.; Rocha, F.O.; Pepersack, T.; Jounblat, Y.; Drowart, A.; Lago, L.D. Advances in pain management for older patients with cancer. Ecancermedicalscience 2019, 13, 980. [Google Scholar] [CrossRef]
- Paice, J.A.; Portenoy, R.; Lacchetti, C.; Campbell, T.; Cheville, A.; Citron, M.; Constine, L.S.; Cooper, A.; Glare, P.; Keefe, F.; et al. Management of chronic pain in survivors of adult cancers: American Society of Clinical Oncology Clinical Practice Guideline. J. Clin. Oncol. 2016, 34, 3325–3345. [Google Scholar] [CrossRef]
- Jara, C.; Del Barco, S.; Grávalos, C.; Hoyos, S.; Hernández, B.; Muñoz, M.; Quintanar, T.; Meana, J.A.; Rodriguez, C.; de Las Peñas, R. SEOM clinical guideline for treatment of cancer pain. Clin. Transl. Oncol. 2018, 20, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Shiffman, S.; Battista, D.R.; Kelly, J.P.; Malone, M.K.; Weinstein, R.B.; Kaufman, D.W. Prevalence of exceeding maximum daily dose of paracetamol, and seasonal variations in cold-flu season. Br. J. Clin. Pharmacol. 2018, 84, 1250–1257. [Google Scholar] [CrossRef]
- Conaghan, P.G.; Arden, N.; Avouac, B.; Migliore, A.; Rizzoli, R. Safety of paracetamol in osteoarthritis: What does the literature say? Drugs Aging 2019, 36 (Suppl. 1), 7–14. [Google Scholar] [CrossRef]
- Pezzia, C.; Sanders, C.; Welch, S.; Bowling, A.; Lee, W.M. Acute Liver Failure Study Group. Psychosocial and behavioral factors in acetaminophen-related acute liver failure and liver injury. J. Psychosom. Res. 2017, 101, 51–57. [Google Scholar] [CrossRef]
- Köhler-Forsberg, O.; Sylvia, L.; Thase, M.; Calabrese, J.R.; Deckersbach, T.; Tohen, M.; Bowden, C.L.; McInnis, M.; Kocsis, J.H.; Friedman, E.S.; et al. Nonsteroidal anti-inflammatory drugs (NSAIDs) and paracetamol do not affect 6-month mood-stabilizing treatment outcome among 482 patients with bipolar disorder. Depress. Anxiety 2017, 34, 281–290. [Google Scholar] [CrossRef]
- Kaur, J.; McFaull, S.R.; Bang, F. Trends in emergency department visits for acetaminophen-related poisonings: 2011-2019. Tendances des visites aux services des urgences pour des intoxications liées à l’acétaminophène, 2011 à 2019. Health Promot. Chronic Dis. Prev. Can. Res. Policy Pract. 2020, 40, 130–133. [Google Scholar] [CrossRef] [PubMed]
- Thusius, N.J.; Romanowicz, M.; Bostwick, J.M. Intentional or Inadvertent Acetaminophen Overdose-How Lethal It Really Is? Psychosomatics 2019, 60, 574–581. [Google Scholar] [CrossRef]
- Chauhan, A.; Sheriff, L.; Hussain, M.T.; Webb, G.J.; Patten, D.A.; Shepherd, E.L.; Shaw, R.; Weston, C.J.; Haldar, D.; Bourke, S.; et al. The platelet receptor CLEC-2 blocks neutrophil mediated hepatic recovery in acetaminophen induced acute liver failure. Nat. Commun. 2020, 11, 1939. [Google Scholar] [CrossRef]
- Jaeschke, H.; Murray, F.J.; Monnot, A.D.; Jacobson-Kram, D.; Cohen, S.M.; Hardisty, J.F.; Atillasoy, E.; Hermanowski-Vosatka, A.; Kuffner, E.; Wikoff, D.; et al. Assessment of the biochemical pathways for acetaminophen toxicity: Implications for its carcinogenic hazard potential. Regul. Toxicol. Pharmacol. RTP 2020, 120, 104859. [Google Scholar] [CrossRef]
- McGill, M.R.; Hinson, J.A. The development and hepatotoxicity of acetaminophen: Reviewing over a century of progress. Drug Metab. Rev. 2020, 52, 472–500. [Google Scholar] [CrossRef]
- Chiew, A.L.; Reith, D.; Pomerleau, A.; Wong, A.; Isoardi, K.Z.; Soderstrom, J.; Buckley, N.A. Updated guidelines for the management of paracetamol poisoning in Australia and New Zealand. Med. J. Aust. 2020, 212, 175–183. [Google Scholar] [CrossRef]
- Pickering, G.; Macian, N.; Papet, I.; Dualé, C.; Coudert, C.; Pereira, B. N-acetylcysteine prevents glutathione decrease and does not interfere with paracetamol antinociceptive effect at therapeutic dosage: A randomized double-blind controlled trial in healthy subjects. Fund. Clin. Pharmacol. 2020, 33, 303–311. [Google Scholar] [CrossRef]
- Chowdhury, A.; Nabila, J.; Adelusi Temitope, I.; Wang, S. Current etiological comprehension and therapeutic targets of acetaminophen-induced hepatotoxicity. Pharmacol. Res. 2020, 161, 105102. [Google Scholar] [CrossRef]
- Wong, J.J.; Côté, P.; Sutton, D.A.; Randhawa, K.; Yu, H.; Varatharajan, S.; Goldgrub, R.; Nordin, M.; Gross, D.P.; Shearer, H.M.; et al. Clinical practice guidelines for the noninvasive management of low back pain: A systematic review by the Ontario Protocol for Traffic Injury Management (OPTIMa) Collaboration. Eur. J. Pain 2017, 21, 201–216. [Google Scholar] [CrossRef]
- Lee, W.M. Acetaminophen (APAP) hepatotoxicity-Isn’t it time for APAP to go away? J. Hepatol. 2017, 67, 1324–1331. [Google Scholar] [CrossRef]
- Lee, B.T.; Odin, J.A.; Grewal, P. An Approach to Drug-Induced Liver Injury from the Geriatric Perspective. Curr. Gastroenterol. Rep. 2021, 23, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Abdel Shaheed, C.; Ferreira, G.E.; Dmitritchenko, A.; McLachlan, A.J.; Day, R.O.; Saragiotto, B.; Lin, C.; Langendyk, V.; Stanaway, F.; Latimer, J.; et al. The efficacy and safety of paracetamol for pain relief: An overview of systematic reviews. Med. J. Aust. 2021, 214, 324–331. [Google Scholar] [CrossRef] [PubMed]
- Ritsmer Stormholt, E.; Steiness, J.; Derby, C.B.; Larsen, M.E.; Maagaard, M.; Mathiesen, O. Paracetamol, NSAIDs, and glucocorticoids for postoperative pain: A protocol for a systematic review with meta-analysis and Trial Sequential Analysis. Acta Anaesthesiol. Scand. 2021, 17. [Google Scholar] [CrossRef]
- Dolati, S.; Tarighat, F.; Pashazadeh, F.; Shahsavarinia, K.; Gholipouri, S.; Soleimanpour, H. The Role of Opioids in Pain Management in Elderly Patients with Chronic Kidney Disease: A Review Article. Anesth. Pain Med. 2020, 10, e105754. [Google Scholar] [CrossRef]
- Anger, M.; Valovska, T.; Beloeil, H.; Lirk, P.; Joshi, G.P.; Van de Velde, M.; Raeder, J.; PROSPECT Working Group and the European Society of Regional Anaesthesia and Pain Therapy. PROSPECT guideline for total hip arthroplasty: A systematic review and procedure-specific postoperative pain management recommendations. Anaesthesia 2021, 20. [Google Scholar] [CrossRef]
- Akbas, S.; Ozkan, A.S.; Durak, M.A.; Yologlu, S. Efficacy of Intravenous Paracetamol and Ibuprofen on Postoperative Pain and Morphine Consumption in Lumbar Disc Surgery: Prospective, Randomized, Double-Blind, Placebo-Controlled Clinical Trial. Neurochirurgie 2021. [Google Scholar] [CrossRef]
- Ramdin, C.; Yu, C.; Colorado, J.; Nelson, L. The impact of adherence to a guideline for minimizing opioid use for treatment of pain in an urban emergency department. Am. J. Emerg. Med. 2021, 49, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Bussières, A.; Cancelliere, C.; Ammendolia, C.; Comer, C.M.; Zoubi, F.A.; Châtillon, C.E.; Chernish, G.; Cox, J.M.; Gliedt, J.A.; Haskett, D.; et al. Non-Surgical Interventions for Lumbar Spinal Stenosis Leading To Neurogenic Claudication: A Clinical Practice Guideline. J. Pain. 2021. [Google Scholar] [CrossRef]
- Roofthooft, E.; Joshi, G.P.; Rawal, N.; Van de Velde, M.; PROSPECT Working Group of the European Society of Regional Anaesthesia; The Pain Therapy and Supported by the Obstetric Anaesthetists’ Association. PROSPECT guideline for elective caesarean section: Updated systematic review and procedure-specific postoperative pain management recommendations. Anaesthesia 2021, 76, 665–680. [Google Scholar] [CrossRef]
- VanderPluym, J.H.; Halker Singh, R.B.; Urtecho, M.; Morrow, A.S.; Nayfeh, T.; Torres Roldan, V.D.; Farah, M.H.; Hasan, B.; Saadi, S.; Shah, S.; et al. Acute Treatments for Episodic Migraine in Adults: A Systematic Review and Meta-analysis. JAMA 2021, 325, 2357–2369. [Google Scholar] [CrossRef]
- Robbins, M.S. Diagnosis and Management of Headache: A Review. JAMA 2021, 325, 1874–1885. [Google Scholar] [CrossRef]
- Mielke, D.; Rohde, V. Randomized controlled trials-a critical re-appraisal. Neurosurg. Rev. 2020. [Google Scholar] [CrossRef]
| analgesic | high activity |
| antipyretic | high activity |
| anti-inflammatory | low activity |
| antiplatelet | low activity |
| antidepressant | anecdotal |
| cognitive-enhancer | anecdotal |
| Organization/Society | Condition | AGREE II Domain Score (%) | ||||||
|---|---|---|---|---|---|---|---|---|
| Scope and Purpose | Stakeholder Involvement | Rigor of Development | Clarity of Presentation | Applicability | Editorial Independence | Overall | ||
| ASAS-EULAR | axSpA | 97 | 65 | 67 | 91 | 30 | 58 | 68 |
| OPTIMa | LBP | 100 | 77 | 73 | 86 | 14 | 67 | 70 |
| NICE | LBP | 100 | 92 | 89 | 68 | 42 | 90 | 80 |
| EBM | LBP | 97 | 49 | 46 | 94 | 35 | 21 | 57 |
| ACR | OA | 97 | 89 | 64 | 89 | 27 | 62 | 61 |
| AAOS/ASRA | OA | 89 | 42 | 34 | 69 | 0 | 52 | 51 |
| ESCEO | OA | 100 | 66 | 64 | 100 | 24 | 47 | 67 |
| NICE | OA | 100 | 99 | 83 | 100 | 86 | 87 | 93 |
| OARSI | OA | 100 | 77 | 54 | 93 | 40 | 92 | 76 |
| ACP/ASIM | MSP | 100 | 86 | 78 | 97 | 12 | 100 | 79 |
| AGS | MSP | 87 | 57 | 34 | 94 | 1 | 48 | 56 |
| BGS, BPS | MSP | 100 | 71 | 60 | 94 | 16 | 33 | 63 |
| AHS | HA | 94 | 71 | 69 | 94 | 12 | 92 | 72 |
| EFNS | HA | 89 | 69 | 39 | 100 | 5 | 50 | 59 |
| NICE | HA | 89 | 89 | 73 | 86 | 42 | 90 | 78 |
| ASCO | CP | 100 | 80 | 86 | 100 | 51 | 75 | 82 |
| ESMO | CP | 94 | 71 | 83 | 86 | 6 | 54 | 59 |
| Organization/Society | First Author, Year | Condition | Recommendation | Comments |
|---|---|---|---|---|
| ASAS-EULAR | van der Heijde, 2016 | axSpA | R | to be considered after NSAIDs failed |
| OPTIMa | Wong, 2016 | LBP | R | recommended in acute LBP |
| NICE | NICE 2020 | LBP | CR | not recommended alone, recommended in association with opioids |
| EBM | EBM 2019 | LBP | R | recommended for acute and chronic LBP |
| ACR | Kolasinski, 2020 | OA | CR | recommended for patients intolerant to NSAIDs, monitor liver function |
| AAOS/ASRA | Fillingham, 2020 | OA | R | |
| ESCEO | Bruyere, 2014 | OA | R | first line for short-term treatment (<3 g/day); not for long-term treatment |
| NICE | NICE 2020 | OA | R | to be considered ahead of NSAIDs |
| OARSI | Bannuru, 2019 | OA | CNR | |
| ACP/AAFP | Qasem, 2020 | MSP | CR | |
| AGS | AGS Panel, 2009 | MSP | R | contraindicated in liver failure; not exceed max 4 g/day dose |
| BGS, BPS | Abdulla, 2013 | MSP | R | elderly population; not to exceed max 4 g/day dose |
| AHS | Marmura, 2018 | HA | R | |
| EFNS | Bendtsen, 2010 | HA | R | 1 g for acute therapy |
| NICE | NICE, 2021 | HA | R | indicated for migraine and tension headache |
| ASCO | Paice, 2016 | CP | R | avoid drug interaction |
| ESMO | Fallon, 2018 | CP | NR | |
| WHO | WHO, 2019 | pain | R |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Freo, U.; Ruocco, C.; Valerio, A.; Scagnol, I.; Nisoli, E. Paracetamol: A Review of Guideline Recommendations. J. Clin. Med. 2021, 10, 3420. https://doi.org/10.3390/jcm10153420
Freo U, Ruocco C, Valerio A, Scagnol I, Nisoli E. Paracetamol: A Review of Guideline Recommendations. Journal of Clinical Medicine. 2021; 10(15):3420. https://doi.org/10.3390/jcm10153420
Chicago/Turabian StyleFreo, Ulderico, Chiara Ruocco, Alessandra Valerio, Irene Scagnol, and Enzo Nisoli. 2021. "Paracetamol: A Review of Guideline Recommendations" Journal of Clinical Medicine 10, no. 15: 3420. https://doi.org/10.3390/jcm10153420
APA StyleFreo, U., Ruocco, C., Valerio, A., Scagnol, I., & Nisoli, E. (2021). Paracetamol: A Review of Guideline Recommendations. Journal of Clinical Medicine, 10(15), 3420. https://doi.org/10.3390/jcm10153420






