Seroprevalence of SARS-CoV-2 Antibodies and Factors Associated with Seropositivity at the University of Salamanca: The DIANCUSAL Study
Abstract
:1. Introduction
2. Material and Methods
2.1. Study Design and Population
2.2. Data Collection
2.3. Serological Testing
2.4. Statistical Analysis
2.5. Ethics Statement
2.6. Role of the Funding Source
3. Results
3.1. Demographic Data
3.2. Seroprevalence
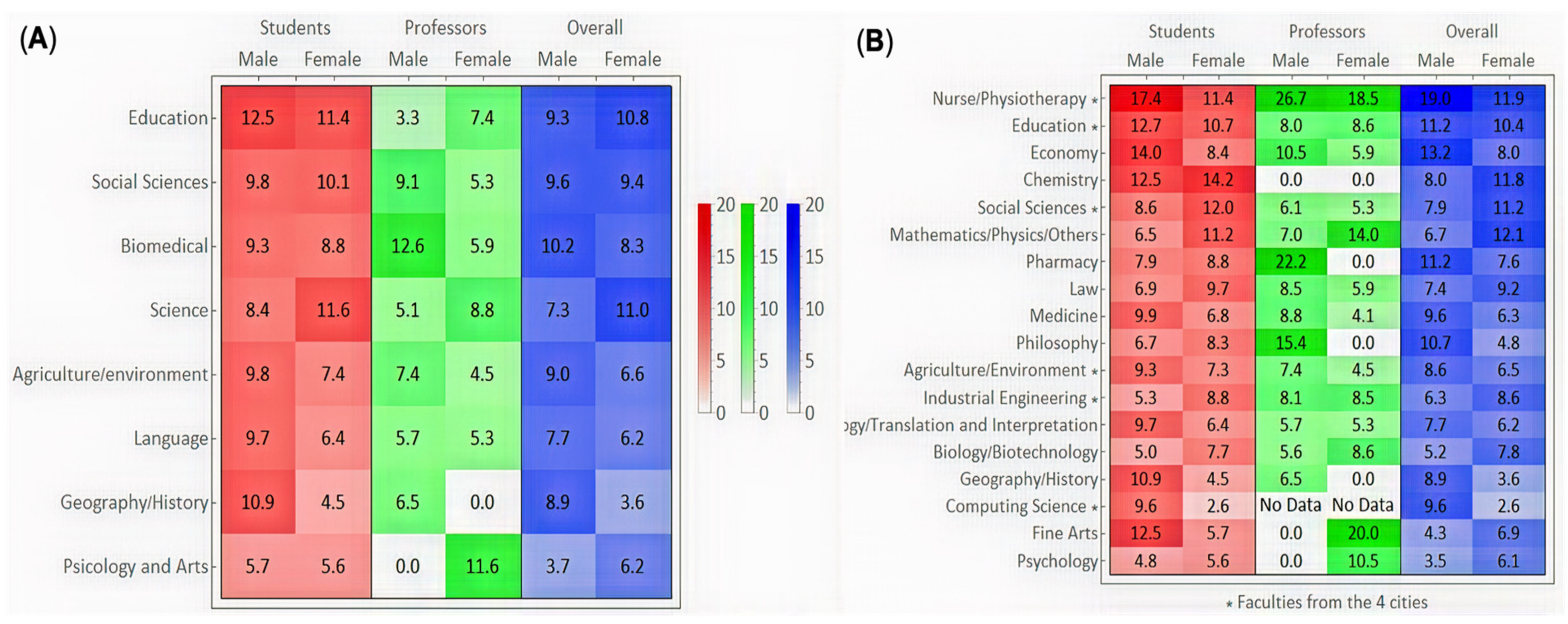
3.3. Associations of Academic Factors with Seropositivity
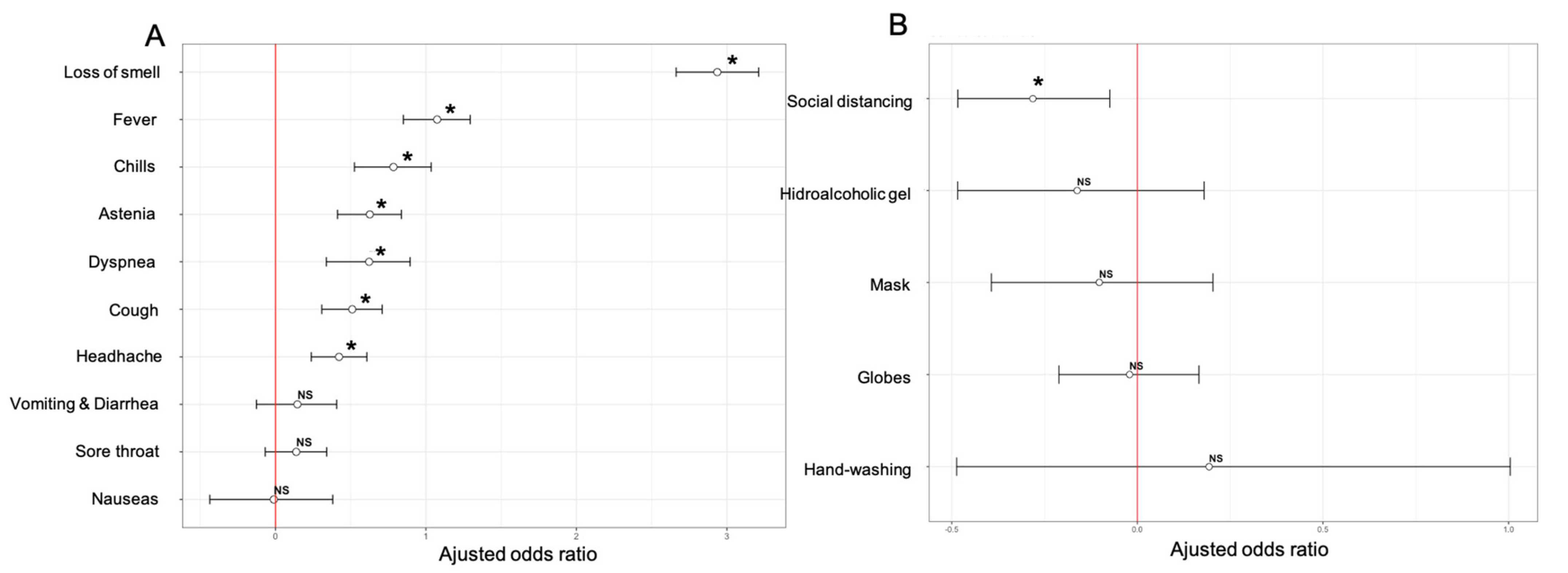
3.4. Associations of Clinical Factors with Seropositivity
3.5. Associations of Lifestyle and Behavioral Factors with Seropositivity
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A. DIANCUSAL Team (Alphabetical Order)
| 1. | Judy | Aala |
| 2. | Elisa | Acosta de la Vega |
| 3. | Álvaro | Aguado Muñiz |
| 4. | Alberto | Alén Andrés |
| 5. | Raquel | Álvarez Lozano |
| 6. | Luis Manuel | Álvarez Oricheta |
| 7. | Roberto | Arévalo Pérez |
| 8. | Elisabeth | Arias Gómez |
| 9. | Samuel | Barbero Garrote |
| 10. | Beatriz María | Bermejo Gil |
| 11. | Enrique | Blanco Peláez |
| 12. | Noelia | Bullón |
| 13. | Esther | Caballero Salvador |
| 14. | Laura | Cabrero |
| 15. | Juan Carlos | Calderón |
| 16. | Rubén | Cañizares Sanchez |
| 17. | Cristina | Carbonell Muñoz |
| 18. | Elena | Carnicero Antón |
| 19. | Raquel | Carnicero Izquierdo |
| 20. | Ana | Carranza de Frutos |
| 21. | Carlos | Carrera Tomás |
| 22. | Laura | Cid Mendes |
| 23. | Sonia | Clavero Sánchez |
| 24. | Clara Isabel | Colino Gandarilla |
| 25. | Victoria | Coral Orbes |
| 26. | Diego | Cotobal García |
| 27. | Beatriz | Crego Vicente |
| 28. | Luis | de Alfonso Vazquez |
| 29. | Manuel | De la Cruz Garcinuño |
| 30. | María Teresa | De la Puente Sanz |
| 31. | Francisco Javier | Derteano Ortiz de Artiñano |
| 32. | David | Eguiluz López |
| 33. | Daniel | Encinas Sanchez |
| 34. | Carlos | Estévez Colmenero |
| 35. | Begoña | Febrer Sendra |
| 36. | Helena | Fernández Cabrera |
| 37. | Adolfo | Fernández Sánchez |
| 38. | Pedro | Fernández Soto |
| 39. | Javier | Flores Fraile |
| 40. | Manuel | Fuentes García |
| 41. | Andrea | Fuentes Gordillo |
| 42. | Raúl | Fuentes Martín |
| 43. | Ana Isabel | Galán Hernández |
| 44. | Juan | García-Bernalt Diego |
| 45. | Lucía | García Aparicio |
| 46. | Carlos | García Cabezas |
| 47. | Vega | Garcia Cirilo |
| 48. | Raquel | Garcia López |
| 49. | María de los Ángeles | García Pascua |
| 50. | José Ángel | García Pedraza |
| 51. | Paula | García Vallés |
| 52. | Nerea | Gestoso Uzal |
| 53. | Mariona | Gil Llagostera |
| 54. | Sonia | Gómez Gaspar |
| 55. | María Isabel | González Flores |
| 56. | Susana | González Manzano |
| 57. | José | Gordo Gonzalo |
| 58. | Oscar | Gorgojo Galindo |
| 59. | Carlos | Gutiérrez Cerrajero |
| 60. | Rosa | Hermosa Prieto |
| 61. | Reyes | Hernández |
| 62. | Isabel María | Hernández de la Fuente |
| 63. | Luis Manuel | Hernández Medina |
| 64. | Nieves | Hernández San Antonio |
| 65. | Santiago | Herrero González |
| 66. | Luis | Jiménez Jurado |
| 67. | Rosa | Juana Tejera Pérez |
| 68. | Paula | Linde Leiva |
| 69. | Inés | Llamas Ramos |
| 70. | Julio | López Abán |
| 71. | Amparo | López Bernus |
| 72. | Joaquín F | López Marcos |
| 73. | Noelia | López Velázquez |
| 74. | Antonio | López-Valverde Centeno |
| 75. | Nansi | López-Valverde Hernández |
| 76. | María | Lorenzo Santiago |
| 77. | Paloma | Malmierca Román |
| 78. | Elvira | Manjón Pérez |
| 79. | Sergio | Manso Hierro |
| 80. | Laura | Márquez Arcos |
| 81. | Abel Jesús | Martel Martel |
| 82. | Juan Carlos | Martín Corral |
| 83. | Alba | Martín del Rey |
| 84. | Raquel | Martín Fernández |
| 85. | Elena | Martín González |
| 86. | Alba | Martín Hernández |
| 87. | Daniel | Martín Hidalgo |
| 88. | Manuel | Martín Morales |
| 89. | Ana María | Martín Nogueras |
| 90. | Ana | Martín Suarez |
| 91. | Andrea | Martín Tomé |
| 92. | María | Martínez Ferradal |
| 93. | Alba | Mata Caballero |
| 94. | Diego | Matellán Alonso |
| 95. | Laura | Mateos Sánchez |
| 96. | Francisco José | Matos |
| 97. | Marta | Mayo Caballero |
| 98. | Isabel | Méndez Hernández |
| 99. | Roberto | Méndez Sánchez |
| 100. | Alba María | Merino Expósito |
| 101. | Adrián | Miguélez Martínez |
| 102. | Elly | Mondolis |
| 103. | Cristina | Mora González |
| 104. | Carlos | Moreno Dorado |
| 105. | Raquel | Moreno García |
| 106. | Mirian | Moreno Ramos |
| 107. | Jorge | Moreno Teniente |
| 108. | Daniel | Muñoz Reyes |
| 109. | Elena | Naranjo Bueno |
| 110. | Verónica | Navarro Santamaría |
| 111. | Cecilia | Oliva Mangas |
| 112. | Paula | Oramas Padrón |
| 113. | Olga | Ortuño López |
| 114. | María | Ovejero Sánchez |
| 115. | María | Oviedo Madrid |
| 116. | Josué | Pendones Ulerio |
| 117. | Sara | Peral Garrido |
| 118. | Ana | Perera Gregorio |
| 119. | Leyre | Pérez Hernández |
| 120. | Laura | Pérez Huerga |
| 121. | Sade | Pérez López |
| 122. | Daniel | Pérez Martin |
| 123. | Daniela | Pérez Ramos |
| 124. | Fátima | Pérez Robledo |
| 125. | Ángel | Pindado Pérez |
| 126. | Roció | Pindado Saez |
| 127. | Carlos Rafael | Pires Baltazar |
| 128. | Olga | Pozas Flores |
| 129. | Isabel | Redero Sanchón |
| 130. | María José | Rodrigo Gonzalo |
| 131. | Clara | Rodrigo Pérez |
| 132. | Beatriz | Rodríguez Alonso |
| 133. | Carlos | Rodríguez Carneiro |
| 134. | Celia | Rodríguez Tudero |
| 135. | Melanie | Ruiz Navarro |
| 136. | Enrique | Sánchez Carrasco |
| 137. | Myriam | Sánchez Díaz |
| 138. | Ana M | Sánchez Fernández |
| 139. | Daniel | Sánchez González |
| 140. | Javier | Sánchez Montejo |
| 141. | Carmen | Sánchez Sánchez |
| 142. | María | Sánchez Tabernero |
| 143. | Alicia | Sanjosé Crespo |
| 144. | Guillermo | Santabrígida Oreja |
| 145. | Laura | Santos Gómez |
| 146. | Elena | Santos Hernandez |
| 147. | Carmen | Santos Marcos |
| 148. | María | Santos Plaza |
| 149. | Cristina | Sanz Cuesta |
| 150. | Rosa | Sepúlveda Correa |
| 151. | Teresa | Sereno Mateos |
| 152. | Susana | Sudon Pollo |
| 153. | Vladut Alexandru | Tanase Iosub |
| 154. | Javier | Tascón Romero |
| 155. | José | Tortosa Cámara |
| 156. | Elena | Varas Martín |
| 157. | Ana | Vicente García |
| 158. | Lidia | Vicente Medina |
| 159. | Laura | Vicente Vicente |
| 160. | Carmen | Vieira Lista |
| 161. | Paula | Vigario Calaco |
| 162. | Elena | Villanueva Sánchez |
| 163. | Cristina | Villaoslada Fuentes |
| 164. | Aranzazu | Zarzuelo Castañeda |
| 165. | Pilar | González Arrieta |
| 166. | Rosa Isabel | Sánchez Alonso |
| 167. | Mª del Pino | Mendez Arroyo |
| 168. | David | Martín Fernandez |
| 169. | Laura | del Rio Sanz |
| 170. | Pilar | González Barez |
| 171. | Jesús | Martín González |
| 172. | Jorge | García Pindado |
| 173. | Vega | Angulo Sánchez |
References
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef]
- World Health Organization 2020. Available online: https://www.who.int/ (accessed on 1 May 2021).
- Pollan, M.; Perez-Gomez, B.; Pastor-Barriuso, R.; Oteo, J.; Hernán, M.A.; Pérez-Olmeda, M.; Sanmartín, J.L.; Fernández-García, A.; Cruz, I.; Fernández de Larrea, N.; et al. A Population-Based Seroepidemiological Study of SARS-CoV-2 in Spain (ENE-COVID). SSRN Electron. J. 2020, 22, 535–544. [Google Scholar] [CrossRef]
- Gudbjartsson, D.F.; Norddahl, G.L.; Melsted, P.; Gunnarsdottir, K.; Holm, H.; Eythorsson, E.; Arnthorsson, A.O.; Helgason, D.; Bjarnadottir, K.; Ingvarsson, R.F.; et al. Humoral Immune Response to SARS-CoV-2 in Iceland. N. Engl. J. Med. 2020, 383, 1724–1734. [Google Scholar] [CrossRef] [PubMed]
- Long, Q.-X.; Tang, X.-J.; Shi, Q.-L.; Li, Q.; Deng, H.-J.; Yuan, J.; Hu, J.-L.; Xu, W.; Zhang, Y.; Lv, F.-J.; et al. Clinical and immunological assessment of asymptomatic SARS-CoV-2 infections. Nat. Med. 2020, 26, 1200–1204. [Google Scholar] [CrossRef] [PubMed]
- Iversen, K.; Bundgaard, H.; Hasselbalch, R.B.; Kristensen, J.H.; Nielsen, P.B.; Pries-Heje, M.; Knudsen, A.D.; Christensen, C.E.; Fogh, K.; Norsk, J.B.; et al. Risk of COVID-19 in health-care workers in Denmark: An observational cohort study. Lancet Infect. Dis. 2020, 20, 1401–1408. [Google Scholar] [CrossRef]
- Slot, E.; Hogema, B.M.; Reusken, C.B.; Reimerink, J.H.; Molier, M.; Karregat, J.H.; IJlst, J.; Novotný, V.M.; van Lier, R.A.; Zaaijer, H.L. Low SARS-CoV-2 seroprevalence in blood donors in the early COVID-19 epidemic in the Netherlands. Nat. Commun. 2020, 11, 5744. [Google Scholar] [CrossRef] [PubMed]
- Tuells, J.; Egoavil, C.M.; Pardo, M.A.P.; Montagud, A.C.; Montagud, E.; Caballero, P.; Zapater, P.; Puig-Barberá, J.; Hurtado-Sanchez, J.A. Seroprevalence Study and Cross-Sectional Survey on COVID-19 for a Plan to Reopen the University of Alicante (Spain). Int. J. Environ. Res. Public Health 2021, 18, 1908. [Google Scholar] [CrossRef] [PubMed]
- De Souza Araújo, A.A.; Quintans-Júnior, L.J.; Schimieguel, D.M.; Corrêa, C.B.; de Moura, T.R.; Cavalcante, R.C.M.; Grespan, R.; de Vasconcelos Cerqueira-Meneses, D.; Barreto-Alves, J.A.; Martins-Filho, P.R. Seroprevalence of SARS-CoV-2 antibodies in low-income university students. EXCLI J. 2021, 20, 276–280. [Google Scholar] [CrossRef]
- Tilley, K.; Ayvazyan, V.; Martinez, L.; Nanda, N.; Kawaguchi, E.S.; O’Gorman, M.; Conti, D.; Gauderman, W.J.; Orman, S.V. A Cross-Sectional Study Examining the Seroprevalence of Severe Acute Respiratory Syndrome Coronavirus 2 Antibodies in a University Student Population. J. Adolesc. Health 2020, 67, 763–768. [Google Scholar] [CrossRef]
- Arnold, C.R.K.; Srinivasan, S.; Herzog, C.M.; Gontu, A.; Bharti, N.; Small, M.; Rogers, C.J.; Schade, M.M.; Kuchipudi, S.V.; Kapur, V.; et al. SARS-CoV-2 Seroprevalence in a University Community: A Longitudinal Study of the Impact of Student Return to Campus on Infection Risk Among Community Members. medRxiv 2021. [Google Scholar] [CrossRef]
- Tsitsilonis, O.E.; Paraskevis, D.; Lianidou, E.; Pierros, V.; Akalestos, A.; Kastritis, E.; Moutsatsou, P.; Scorilas, A.; Sphicopoulos, T.; Terpos, E.; et al. Seroprevalence of Antibodies against SARS-CoV-2 among the Personnel and Students of the National and Kapodistrian University of Athens, Greece: A Preliminary Report. Life 2020, 10, 214. [Google Scholar] [CrossRef]
- World Bank Group. The COVID-19 Crisis Reponse: Supporting Tertiary Education for Continuity, Adaptation, and Innovation; World Bank: Washington, DC, USA, 2020. [Google Scholar]
- Rostami, A.; Sepidarkish, M.; Leeflang, M.; Riahi, S.M.; Shiadeh, M.N.; Esfandyari, S.; Mokdad, A.H.; Hotez, P.J.; Gasser, R.B. SARS-CoV-2 seroprevalence worldwide: A systematic review and meta-analysis. Clin. Microbiol. Infect. 2020, 27, 331–340. [Google Scholar] [CrossRef]
- Zhou, Y.; Chi, J.; Lv, W.; Wang, Y. Obesity and diabetes as high-risk factors for severe coronavirus disease 2019 (Covid-19). Diabetes Metab. Res. Rev. 2021, 37, e3377. [Google Scholar] [CrossRef]
- Cornejo-Pareja, I.M.; Gómez-Pérez, A.M.; Fernández-García, J.C.; Millan, R.B.S.; Luque, A.A.; Hollanda, A.; Jiménez, A.; Jimenez-Murcia, S.; Munguia, L.; Ortega, E.; et al. Coronavirus disease 2019 (COVID-19) and obesity. Impact of obesity and its main comorbidities in the evolution of the disease. Eur. Eat. Disord. Rev. 2020, 28, 799–815. [Google Scholar] [CrossRef]
- Shastri, M.D.; Shukla, S.D.; Chong, W.C.; Kc, R.; Dua, K.; Patel, R.P.; Peterson, G.M.; O’Toole, R.F. Smoking and COVID-19: What we know so far. Respir. Med. 2020, 176, 106237. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Yang, Y.; Huang, H.; Li, D.; Gu, D.; Lu, X.; Zhang, Z.; Liu, L.; Liu, T.; Liu, Y.; et al. Relationship between the ABO Blood Group and the COVID-19 Susceptibility. medRxiv 2020. [Google Scholar] [CrossRef] [Green Version]
- Lechien, J.R.; Chiesa-Estomba, C.M.; Place, S.; Laethem, Y.V.; Cabaraux, P.; Mat, Q.; Huet, K.; Plzak, J.; Horoi, M.; Hans, S.; et al. Clinical and epidemiological characteristics of 1420 European patients with mild-to-moderate coronavirus disease 2019. J. Intern. Med. 2020, 288, 335–344. [Google Scholar] [CrossRef]
- Kontou, P.I.; Braliou, G.G.; Dimou, N.L.; Nikolopoulos, G.; Bagos, P.G. Antibody Tests in Detecting SARS-CoV-2 Infection: A Meta-Analysis. Diagnostics 2020, 10, 319. [Google Scholar] [CrossRef]
- Brauner, J.M.; Mindermann, S.; Sharma, M.; Johnston, D.; Salvatier, J.; Gavenčiak, T.; Stephenson, A.B.; Leech, G.; Altman, G.; Mikulik, V.; et al. Inferring the effectiveness of government interventions against COVID-19. Science 2021, 371, eabd9338. [Google Scholar] [CrossRef]
- Fisher, K.A.; Tenforde, M.W.; Feldstein, L.R.; Lindsell, C.J.; Shapiro, N.I.; Files, D.C.; Gibbs, K.W.; Erickson, H.L.; Prekker, M.E.; Steingrub, J.S.; et al. Community and Close Contact Exposures Associated with COVID-19 Among Symptomatic Adults ≥18 Years in 11 Outpatient Health Care Facilities—United States, July 2020. Morb. Mortal. Wkly. Rep. 2020, 69, 1258–1264. [Google Scholar] [CrossRef]
- Madewell, Z.J.; Yang, Y.; Longini, I.M.; Halloran, M.E.; Dean, N.E. Household Transmission of SARS-CoV-2. JAMA Netw. Open 2020, 3, e2031756. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo-Campos, P.; Blankenhaus, B.; Mota, C.; Gomes, A.; Serrano, M.; Ariotti, S.; Costa, C.; Nunes-Cabaço, H.; Mendes, A.M.; Gaspar, P.; et al. Seroprevalence of anti-SARS-CoV-2 antibodies in COVID-19 patients and healthy volunteers up to 6 months post disease onset. Eur. J. Immunol. 2020, 50, 2025–2040. [Google Scholar] [CrossRef] [PubMed]
- Lijia, S.; Lihong, S.; Huabin, W.; Xiaoping, X.; Xiaodong, L.; Yixuan, Z.; Pin, H.; Yina, X.; Xiaoyun, S.; Junqi, W. Serological chemiluminescence immunoassay for the diagnosis of SARS-CoV-2 infection. J. Clin. Lab. Anal. 2020, 34, e23466. [Google Scholar] [CrossRef]
- Hooper, M.W.; Nápoles, A.M.; Pérez-Stable, E.J. COVID-19 and Racial/Ethnic Disparities. JAMA 2020, 323, 2466–2467. [Google Scholar] [CrossRef] [PubMed]
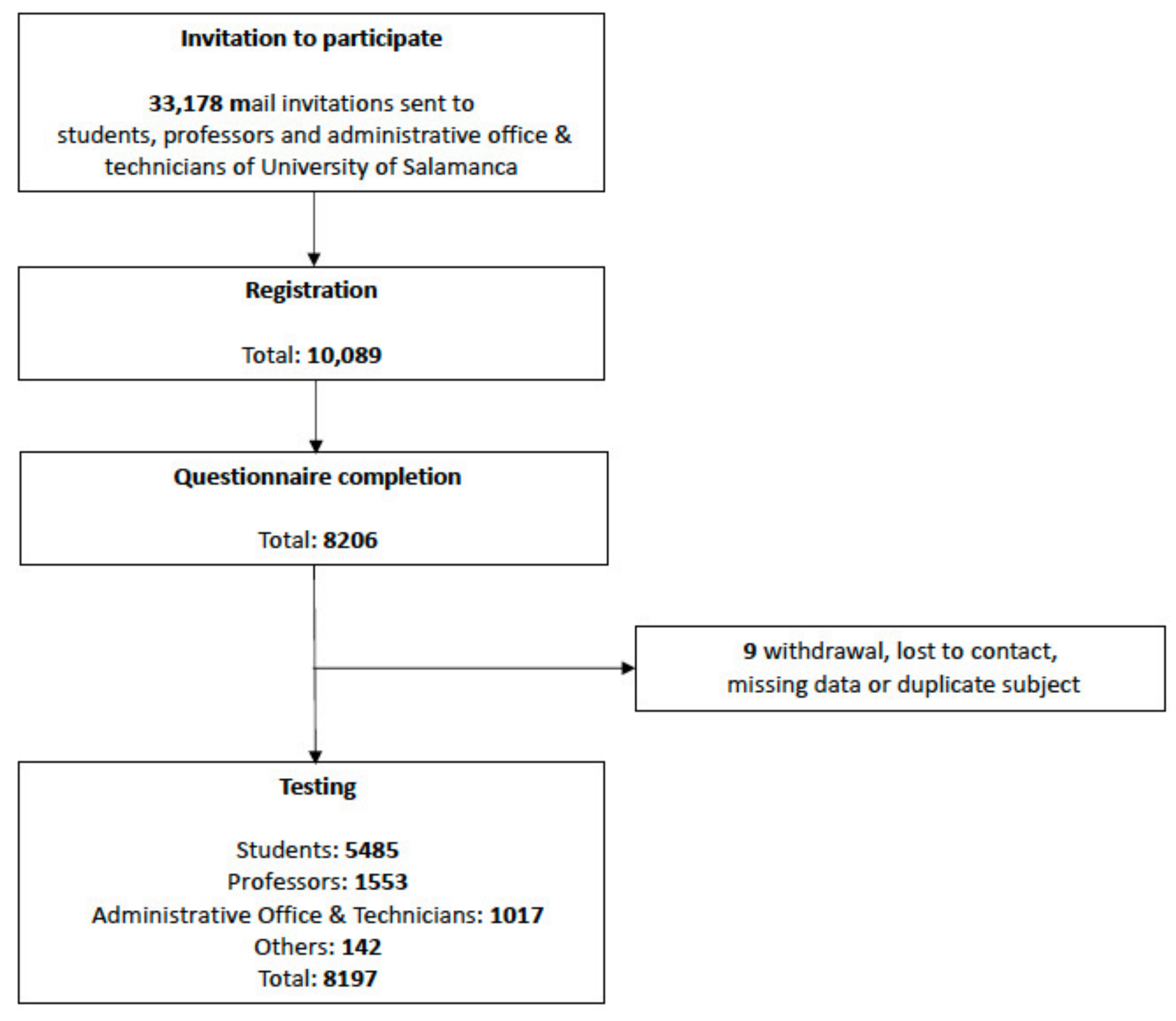
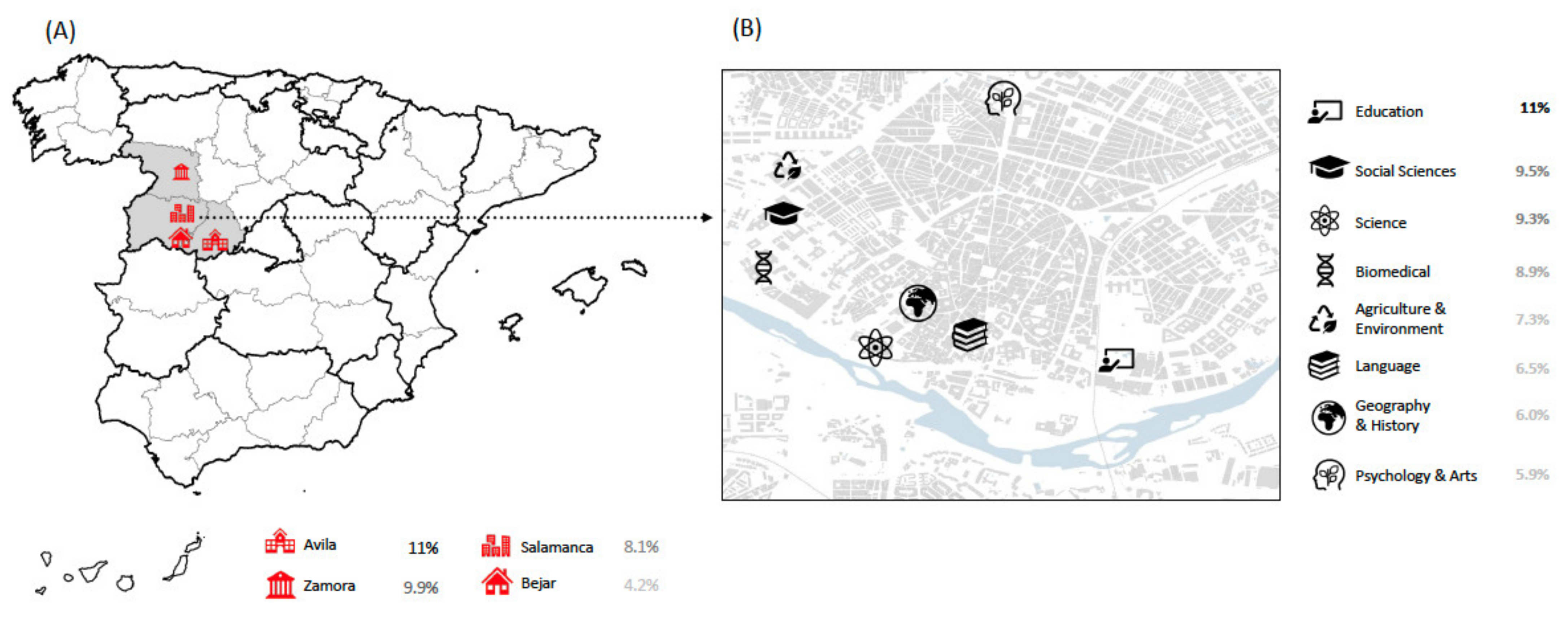
| Variables | n = 8197 (%) | |
|---|---|---|
| Age | ||
| Mean ± SD; years | 31.4 ± 14.5 | |
| Population | 17–28 | 5503 (68.1%) |
| 29–39 | 486 (6.0%) | |
| 40–49 | 606 (7.5%) | |
| 50–59 | 1039 (12.9%) | |
| 60–76 | 441 (5.5%) | |
| Sex | ||
| Male | 2709 (34.0%) | |
| Female | 5248 (66.0%) | |
| Position | ||
| Students | Undergraduate | 5093 (62.1%) |
| Postgraduate (Master and PhD) | 392 (4.8%) | |
| Professors | 1553 (18.9%) | |
| Technicians and Administrative Officers | 1017 (12.4%) | |
| Others | 142 (1.7%) | |
| Salamanca University Campus Map | ||
| Salamanca | 7390 (90.2%) | |
| Zamora | 355 (4.3%) | |
| Avila | 334 (4.1%) | |
| Bejar | 118 (1.4%) | |
| Salamanca University Campus | ||
| Agriculture and Environment | 160 (2.2%) | |
| Biomedical | 1791 (24.2%) | |
| Education | 456 (6.2%) | |
| Geography and History | 215 (2.9%) | |
| Language | 601 (8.1%) | |
| Psychology and Arts | 580 (7.8%) | |
| Science | 687 (9.3%) | |
| Social Sciences | 1408 (19.1%) | |
| Others | 1492 (20.2%) | |
| Dwelling and Exposure | n = 7034 (%) | Seropositivity | ||
|---|---|---|---|---|
| n (%) | p-Value | |||
| Residence | Professors | 1549 (22.0%) | 114 (7.4%) | 0.133 |
| Student private house | 5039 (71.6%) | 449 (8.9%) | ||
| Student colleges | 446 (6.3%) | 42 (9.4%) | ||
| Life with animals | Yes | 1867 (22.8%) | 138 (7.4%) | 0.127 |
| No | 6330 (77.2%) | 538 (8.5%) | ||
| Exposure | Household | 637 (8.0%) | 189 (29.7%) | 0.000 |
| No household | 7049 (88.2%) | 436 (6.2%) | ||
| Not know | 302 (3.8%) | 31 (10.3%) | ||
| Household Exposure | Colleges | 38 (6.0%) | 13 (34.2%) | 0.583 |
| Private house | 599 (94.0%) | 176 (29.4%) | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muro, A.; Belhassen-García, M.; Muñoz Bellido, J.L.; Lorenzo Juanes, H.; Vicente, B.; Pendones, J.; Adserias, J.; Sánchez Hernández, G.; Rodríguez Rosa, M.; Vicente Villardón, J.L.; et al. Seroprevalence of SARS-CoV-2 Antibodies and Factors Associated with Seropositivity at the University of Salamanca: The DIANCUSAL Study. J. Clin. Med. 2021, 10, 3214. https://doi.org/10.3390/jcm10153214
Muro A, Belhassen-García M, Muñoz Bellido JL, Lorenzo Juanes H, Vicente B, Pendones J, Adserias J, Sánchez Hernández G, Rodríguez Rosa M, Vicente Villardón JL, et al. Seroprevalence of SARS-CoV-2 Antibodies and Factors Associated with Seropositivity at the University of Salamanca: The DIANCUSAL Study. Journal of Clinical Medicine. 2021; 10(15):3214. https://doi.org/10.3390/jcm10153214
Chicago/Turabian StyleMuro, Antonio, Moncef Belhassen-García, Juan Luís Muñoz Bellido, Helena Lorenzo Juanes, Belén Vicente, Josué Pendones, José Adserias, Gonzalo Sánchez Hernández, Miguel Rodríguez Rosa, José Luis Vicente Villardón, and et al. 2021. "Seroprevalence of SARS-CoV-2 Antibodies and Factors Associated with Seropositivity at the University of Salamanca: The DIANCUSAL Study" Journal of Clinical Medicine 10, no. 15: 3214. https://doi.org/10.3390/jcm10153214
APA StyleMuro, A., Belhassen-García, M., Muñoz Bellido, J. L., Lorenzo Juanes, H., Vicente, B., Pendones, J., Adserias, J., Sánchez Hernández, G., Rodríguez Rosa, M., Vicente Villardón, J. L., Burguillo, J., López Andaluz, J., Martín Oterino, J. A., García Criado, F. J., Barbero, F., Morales, A. I., Galindo Villardón, P., González Sarmiento, R., & on behalf of the DIANCUSAL Team. (2021). Seroprevalence of SARS-CoV-2 Antibodies and Factors Associated with Seropositivity at the University of Salamanca: The DIANCUSAL Study. Journal of Clinical Medicine, 10(15), 3214. https://doi.org/10.3390/jcm10153214









