Validation of a Prospective Urinalysis-Based Prediction Model for ICU Resources and Outcome of COVID-19 Disease: A Multicenter Cohort Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Data Quality, Quality Assurance and Study Population
2.2. Urinalysis and Patient Stratification
2.3. Study Endpoints and Statistical Analyses
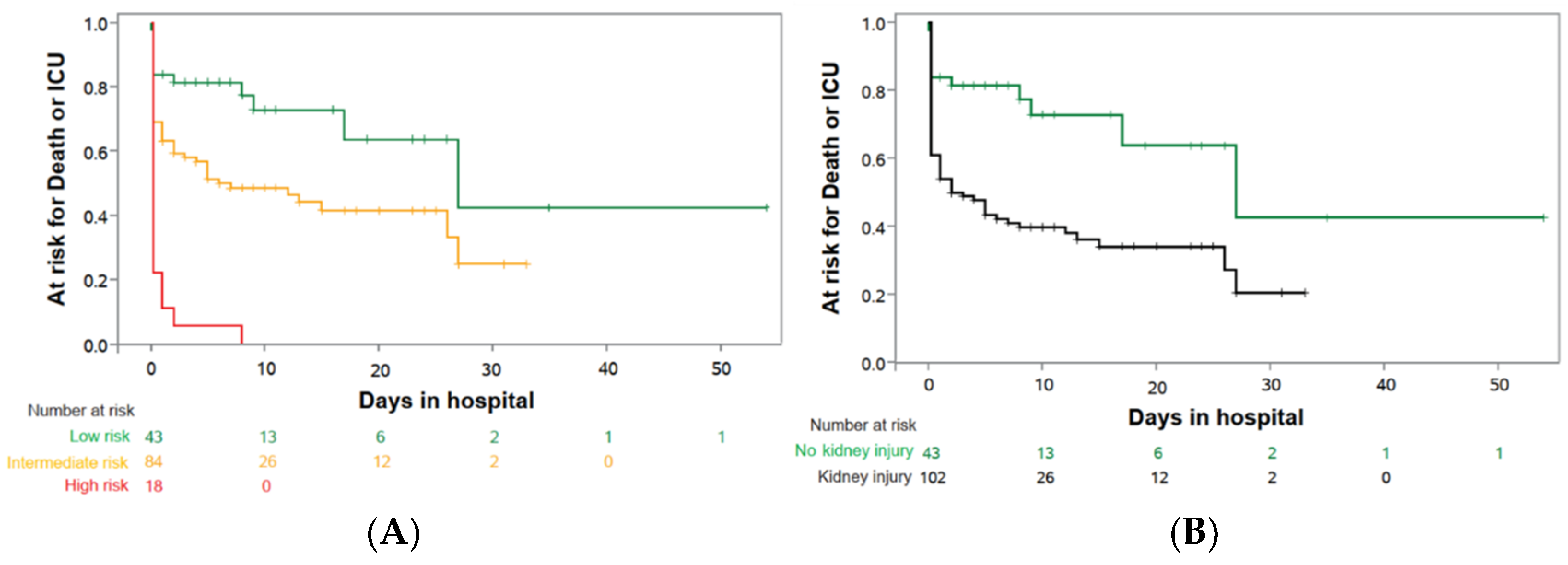
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.; Zhou, M.; Dong, X.; Qu, J.; Gong, F.; Han, Y.; Qiu, Y.; Wang, J.; Liu, Y.; Wie, Y.; et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study. Lancet 2020, 395, 507–513. [Google Scholar] [CrossRef] [Green Version]
- Puelles, V.G.; Lütgehetmann, M.; Lindenmeyer, M.T.; Sperhake, J.P.; Wong, M.N.; Allweiss, L.; Chilla, S.; Heinemann, A.; Wanner, N.; Liu, S.; et al. Multiorgan and Renal Tropism of SARS-CoV-2. N. Engl. J. Med. 2020, 383, 590–592. [Google Scholar] [CrossRef] [PubMed]
- Mehta, P.; McAuley, D.F.; Brown, M.; Sanchez, E.; Tattersall, R.S.; Manson, J.J. COVID-19: Consider cytokine storm syndromes and immunosuppression. Lancet 2020, 395, 1033–1034. [Google Scholar] [CrossRef]
- Ackermann, M.; Verleden, S.E.; Kuehnel, M.; Haverich, A.; Welte, T.; Laenger, F.; Vanstapel, A.; Werlein, C.; Stark, H.; Tzankov, A.; et al. Pulmonary Vascular Endothelialitis, Thrombosis, and Angiogenesis in Covid-19. N. Engl. J. Med. 2020, 383, 120–128. [Google Scholar] [CrossRef]
- Liu, P.P.; Blet, A.; Smyth, D.; Li, H. The science underlying COVID-19: Implications for the cardiovascular system. Circulation 2020, 142, 68–78. [Google Scholar] [CrossRef] [Green Version]
- Helms, J.; Tacquard, C.; Severac, F.; Leonard-Lorant, I.; Ohana, M.; Delabranche, X.; Merdji, H.; Clere-Jehl, R.; Schenck, M.; Fagot Gandet, F.; et al. High risk of thrombosis in patients with severe SARS-CoV-2 infection: A multicenter prospective cohort study. Intens. Care Med. 2020, 46, 1089–1098. [Google Scholar] [CrossRef]
- Varga, Z.; Flammer, A.J.; Steiger, P.; Haberecker, M.; Andermatt, R.; Zinkernagel, A.S.; Mehra, M.R.; Schuepbach, R.A.; Ruschitzka, F.; Moch, H. Endothelial cell infection and endotheliitis in COVID-19. Lancet 2020, 395, 1417–1418. [Google Scholar] [CrossRef]
- Pei, G.; Zhang, Z.; Peng, J.; Liu, L.; Zhang, C.; Yu, C.; Ma, Z.; Huang, Y.; Liu, W.; Yao, Y.; et al. Renal involvement and early prognosis in patients with COVID-19 pneumonia. J. Am. Soc. Nephrol. 2020, 31, 1157–1165. [Google Scholar] [CrossRef]
- Emanuel, E.J.; Persad, G.; Upshur, R.; Thome, B.; Parker, M.; Glickman, A.; Zhang, C.; Boyle, C.; Smith, M.; Phillips, J.P. Fair Allocation of Scarce Medical Resources in the Time of Covid-19. N. Engl. J. Med. 2020, 382, 2049–2055. [Google Scholar] [CrossRef]
- Richardson, S.; Hirsch, J.S.; Narasimhan, M.; Crawford, J.M.; McGinn, T.; Davidson, K.W.; The Northwell COVID-19 Research Consortium; Barnaby, D.P.; Becker, L.B.; Chelico, J.D.; et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA 2020, 323, 2052–2059. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef] [Green Version]
- Wu, Z.; McGoogan, J.M. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: Summary of a report of 72,314 cases from the Chinese Center for Disease Control and Prevention. JAMA 2020, 323, 1239–1242. [Google Scholar] [CrossRef]
- Goyal, P.; Choi, J.J.; Pinheiro, L.C.; Schenck, E.J.; Chen, R.; Jabri, A.; Satlin, M.J.; Campion, T.R., Jr.; Nahid, M.; Ringel, J.B.; et al. Clinical characteristics of Covid-19 in New York City. N. Engl. J. Med. 2020, 382, 2372–2374. [Google Scholar] [CrossRef]
- Guan, W.; Ni, Z.; Hu, Y.; Liang, W.H.; Ou, C.Q.; He, J.X.; Liu, L.; Shan, H.; Lei, C.L.; Hui, D.S.C.; et al. Clinical characteristics of coronavirus disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef]
- Wu, C.; Chen, X.; Cai, Y.; Xia, J.; Zhou, X.; Xu, S.; Huang, H.; Zhang, L.; Zhou, X.; Du, C.; et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern. Med. 2020, 180, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Fagiuoli, S.; Lorini, F.L.; Remuzzi, G. Covid-19 Bergamo Hospital Crisis Unit. Adaptations and Lessons in the Province of Bergamo. N. Engl. J. Med. 2020, 382, e71. [Google Scholar]
- Gross, O.; Moerer, O.; Weber, M.; Huber, T.B.; Scheithauer, S. COVID-19-associated nephritis: Early warning for disease severity and complications? Lancet 2020, 395, e87–e88. [Google Scholar] [CrossRef]
- Braun, F.; Lütgehetmann, M.; Pfefferle, S.; Wong, M.N.; Carsten, A.; Lindenmeyer, M.T.; Nörz, D.; Heinrich, F.; Meißner, K.; Wichmann, D.; et al. SARS-CoV-2 renal tropism associates with acute kidney injury. Lancet 2020, 396, 597–598. [Google Scholar] [CrossRef]
- Batlle, D.; Soler, M.J.; Sparks, M.A.; Hiremath, S.; South, A.M.; Welling, P.A.; Swaminathan, S.; COVID-19 and ACE2 in Cardiovascular, Lung, and Kidney Working Group. Acute Kidney Injury in COVID-19: Emerging Evidence of a Distinct Pathophysiology. J. Am. Soc. Nephrol. 2020, 31, 1380–1383. [Google Scholar] [CrossRef]
- Sungurlu, S.; Kuppy, J.; Balk, R.A. Role of Antithrombin III and Tissue Factor Pathway in the Pathogenesis of Sepsis. Crit. Care Clin. 2020, 36, 255–265. [Google Scholar] [CrossRef]
- Noone, D.G.; Iijima, K.; Parekh, R. Idiopathic nephrotic syndrome in children. Lancet 2018, 392, 61–74. [Google Scholar] [CrossRef]
- Kluge, S.; Janssens, U.; Spinner, C.D.; Pfeifer, M.; Marx, G.; Karagiannidis, C. Clinical practice guideline: Recommendations on in-hospital treatment of patients with COVID-19. Dtsch. Arztebl. Int. 2021, 118, 1–7. [Google Scholar]
- Wynants, L.; Van Calster, B.; Collins, G.S.; Riley, R.D.; Heinze, G.; Schuit, E.; Bonten, M.M.J.; Dahly, D.L.; Damen, J.A.A.; Debray, T.P.A.; et al. Prediction models for diagnosis and prognosis of COVID-19 infection: Systematic review and critical appraisal. BMJ 2020, 369, m1328. [Google Scholar] [CrossRef] [Green Version]
- Sun, Q.; Qiu, H.; Huang, M.; Yang, Y. Lower mortality of COVID-19 by early recognition and intervention: Experience from Jiangsu Province. Ann. Intens. Care 2020, 10, 33. [Google Scholar] [CrossRef] [Green Version]
- Edler, C.; Schröder, A.S.; Aepfelbacher, M.; Fitzek, A.; Heinemann, A.; Heinrich, F.; Klein, A.; Langenwalder, F.; Lütgehetmann, M.; Meißner, K.; et al. Dying with SARS-CoV-2 infection-an autopsy study of the first consecutive 80 cases in Hamburg, Germany. Int. J. Leg. Med. 2020, 134, 1275–1284. [Google Scholar] [CrossRef] [PubMed]
- Tang, N.; Bai, H.; Chen, X.; Gong, J.; Li, D.; Sun, Z. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J. Thromb. Haemost. 2020, 18, 1094–1099. [Google Scholar] [CrossRef] [PubMed]
- Karagiannidis, C.; Mostert, C.; Hentschker, C.; Voshaar, T.; Malzahn, J.; Schillinger, G.; Klauber, J.; Janssens, U.; Marx, G.; Weber-Carstens, S.; et al. Case characteristics, resource use, and outcomes of 10 021 patients with COVID-19 admitted to 920 German hospitals: An observational study. Lancet Respir. Med. 2020, 8, 853–862. [Google Scholar] [CrossRef]
- Vijayan, A.; Humphreys, B.D. SARS-CoV-2 in the kidney: Bystander or culprit? Nat. Rev. Nephrol. 2020, 14, 1–2. [Google Scholar] [CrossRef]
- Seymour, C.W. A cooperation of the doves. Intens. Care Med. 2020, 18, 1–2. [Google Scholar] [CrossRef]
- Poston, J.T.; Patel, B.K.; Davis, A.M. Management of critically ill adults with COVID-19. JAMA 2020, 323, 1839–1841. [Google Scholar] [CrossRef]
- Cao, B.; Wang, Y.; Wen, D.; Liu, W.; Wang, J.; Fan, G.; Ruan, L.; Song, B.; Cai, Y.; Wei, M.; et al. A trial of lopinavir–ritonavir in adults hospitalized with severe Covid-19. N. Engl. J. Med. 2020, 382, 1787–1799. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, D.; Du, G.; Du, R.; Zhao, J.; Jin, Y.; Fu, S.; Gao, L.; Cheng, Z.; Lu, Q.; et al. Remdesivir in adults with severe Covid-19: A randomized, double-blind, placebo-controlled, multicentre trial. Lancet 2020, 395, 1569–1578. [Google Scholar] [CrossRef]
- RECOVERY Collaborative Group; Horby, P.; Lim, W.S.; Emberson, J.R.; Mafham, M.; Bell, J.L.; Linsell, L.; Staplin, N.; Brightling, C.; Ustianowski, A.; et al. Dexamethasone in Hospitalized Patients with Covid-19—Preliminary Report. N. Engl. J. Med. 2021, 384, 693–704. [Google Scholar] [PubMed]
- Duan, K.; Liu, B.; Li, C.; Zhang, H.; Yu, T.; Qu, J.; Zhou, M.; Chen, L.; Meng, S.; Hu, Y.; et al. Effectiveness of convalescent plasma therapy in severe COVID-19 patients. Proc. Natl. Acad. Sci. USA 2020, 117, 9490–9496. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Characteristic | Association: Urine Status in Relation to Disease Severity (N = 223) | ||||
|---|---|---|---|---|---|
| Green (N = 58) | Non-Green (N = 165) | Green vs. Non-Green | |||
| Male sex—no. (%) | 40/58 | (69) | 114/165 | (69.1) | 0.98 |
| Median age (IQR)—year | 60 | (25) | 66 | (19) | <0.01 |
| Median body mass index BMI (IQR) | 27.2 | (7.5) | 27.4 | (6.9) | 0.778 |
| Chronic disease PRIOR to COVID-19—no. (%) | 44/58 | (75.9) | 122/165 | (73.9) | |
| Chronic heart disease—no. (%) | 16/58 | (27.6) | 46/165 | (27.9) | |
| Chronic lung disease—no. (%) | 16/58 | (27.6) | 28/165 | (17) | |
| Chronic kidney disease—no. (%) | 6/58 | (10.3) | 17/165 | (10.3) | |
| Renal replacement therapy—no. (%) | 1/58 | (1.7) | 4/165 | (2.4) | |
| Malignant tumor disease—no. (%) | 13/58 | (22.4) | 14/165 | (8.5) | |
| Diabetes mellitus—no. (%) | 8/58 | (13.8) | 35/165 | (21.2) | |
| Immunosuppressive therapy—no. (%) | 19/58 | (32.8) | 29/165 | (17.6) | |
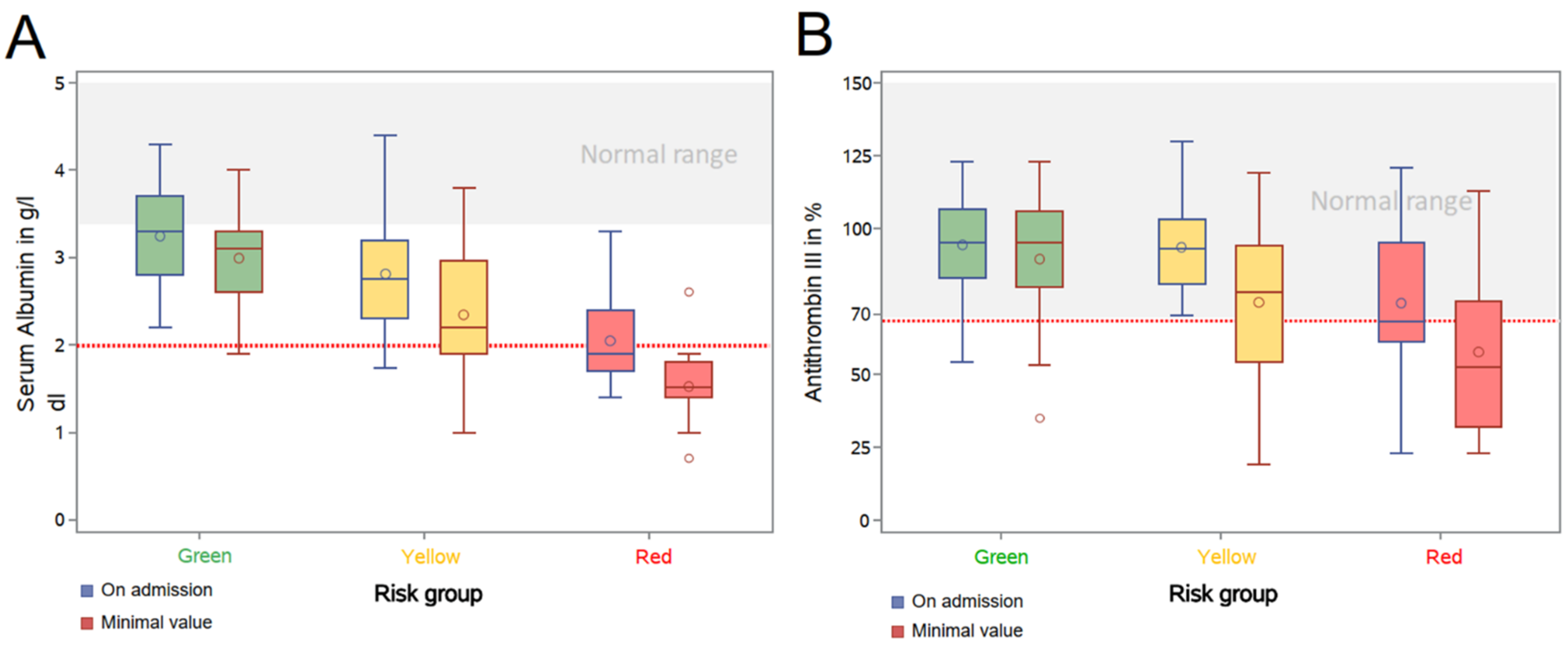
| Median serum albumin (IQR)—g/dL | |||||
| First value in hospital (IQR)—g/dL | 3.3 | (0.9) | 2.4 | (0.9) | |
| Lowest value (IQR)—g/dL | 3 | (0.9) | 2 | (0.8) | |
| <2.0 g/dL during hospital stay—no. (%) | 3/47 | (6.4) | 70/148 | (47.3) | |
| Median antithrombin III (IQR)—% | |||||
| First value in hospital (IQR)—% | 92.5 | (23) | 85 | 28 | |
| Lowest value (IQR)—% | 90 | (30) | 63 | 37 | |
| <70% during hospital stay—no. (%) | 7/28 | (25) | 67/118 | (56.8) | |
| ICU or death until day 10—no. (%) | 14/58 | (24.1) | 109/164 | (66.5) | <0.01 |
| Death during hospital stay—no. (%) | 7/58 | (12.1) | 47/165 | (28.5) | <0.01 |
| Still at risk of death in hospital—no. (%) | 2/58 | (3.4) | 30/165 | (18.2) | |
| ICU level 3 during hospital stay—no. (%) | 13/58 | (22.4) | 106/165 | (64.2) | |
| Mechanical ventilation—no. (%) | 9/58 | (15.5) | 95/165 | (57.6) | <0.01 |
| ECMO therapy—no. (%) | 1/58 | (1.7) | 31/164 | (18.9) | <0.01 |
| Renal replacement therapy RRT—no. (%) | 7/58 | (12.1) | 65/164 | (39.6) | <0.01 |
| Baseline Characteristic | Green (N = 43) | Yellow (N = 84) | Red (N = 18) | Non-Green (N = 102) | ||||
|---|---|---|---|---|---|---|---|---|
| Male sex—no. (%) | 29/43 | (67.4) | 53/84 | (63.1) | 12/18 | (66.7) | 65/102 | (63.7) |
| Median serum creatinine (IQR)—mg/dL | 0.97 | (0.97–1.18) | 1.0 | (0.76-1.63) | ||||
| Median age (IQR)—year | 61 | (49-75) | 71 | (60-79) | 67 | (58-74) | 70 | (59-78) |
| 20 to 39 years of age | 8/43 | (18.6) | 4/84 | (4.8) | 0/18 | (0) | 4/102 | (3.9) |
| 40 to 59 years of age | 12/43 | (27.9) | 17/84 | (20.2) | 5/18 | (27.8) | 22/102 | (21.6) |
| 60 to 79 years of age | 20/43 | (46.5) | 44/84 | (52.4) | 11/18 | (61.1) | 55/102 | (53.9) |
| 80 and older years of age | 3/43 | (7) | 19/84 | (22.6) | 2/18 | (11.1) | 21/102 | (20.6) |
| Median body mass index BMI (IQR) | 27 | (24-31) | 26 | (24-31) | 27 | (26-33) | 26 | (24-31) |
| Chronic disease PRIOR COVID-19—no. (%) | 31/43 | (72.1) | 73/84 | (86.9) | 12/18 | (66.7) | 85/102 | (83.3) |
| Chronic heart disease—no. (%) | 10/43 | (23.3) | 28/84 | (33.3) | 6/18 | (33.3) | 34/102 | (33.3) |
| Chronic lung disease—no. (%) | 13/43 | (30.2) | 15/84 | (17.9) | 5/18 | (27.8) | 20/102 | (19.6) |
| Chronic kidney disease—no. (%) | 4/43 | (9.3) | 16/84 | (19) | 0/18 | (0) | 16/102 | (15.7) |
| Renal replacement therapy—no. (%) | 1/43 | (2.3) | 4/84 | (4.8) | 0/18 | (0) | 4/102 | (3.9) |
| Malignant tumor disease—no. (%) | 8/43 | (18.6) | 12/84 | (14.3) | 1/18 | (5.6) | 13/102 | (12.7) |
| Diabetes mellitus—no. (%) | 4/43 | (9.3) | 23/84 | (27.4) | 6/18 | (33.3) | 29/102 | (28.4) |
| Immunosuppressive therapy—no. (%) | 13/43 | (30.2) | 22/84 | (26.2) | 3/18 | (33.3) | 25/102 | (24.5) |
| Outcome | Green (N = 43) | Yellow (N = 84) | Red (N = 18) | Non-Green (N = 102) | ||||
|---|---|---|---|---|---|---|---|---|
| Primary endpoint | ||||||||
| ICU or death until day 10—no. (%) | 10/43 | (23.3) | 43/84 | (51.2) | 18/18 | (100.0) | 61/102 | (59.8) |
| Secondary endpoints—Complications | ||||||||
| Death during hospital stay—no. (%) | 5/43 | (11.6) | 23/84 | (27.4) | 7/18 | (38.8) | 30/102 | (29.4) |
| Still at risk of death in hospital—no. (%) | 1/43 | (2.3) | 8/84 | (9.5) | 3/18 | (16.7) | 11/102 | (10.8) |
| ICU level 3 during hospital stay—no. (%) | 9/43 | (20.9) | 38/84 | (45.2) | 17/18 | (94.4) | 55/102 | (53.9) |
| Mechanical ventilation—no. (%) | 6/43 | (14) | 28/84 | (33.3) | 17/18 | (94.4) | 45/102 | (44.1) |
| ECMO therapy—no. (%) | 1/43 | (2.3) | 5/84 | (6.0) | 6/18 | (33.3) | 11/102 | (10.8) |
| Renal replacement therapy RRT—no. (%) | 5/43 | (11.6) | 20/83 | (24.1) | 11/18 | (61.1) | 31/101 | (30.7) |
| Secondary endpoints—Resources | ||||||||
| Time on mechanical ventilation—days (DPP) | 23 | (2.6) | 23 | (7.7) | 10 | (9.4) | 19 | (8.4) |
| Time on ECMO therapy—days (DPP) | 11 | (0.3) | 20 | (1.2) | 23 | (7.7) | 22 | (2.3) |
| Time on RRT—days (DPP) | 23 | (2.7) | 17 | (4.1) | 13 | (7.9) | 15 | (4.6) |
| Secondary endpoints—Blood values | ||||||||
| Median serum albumin (IQR)—g/dL | ||||||||
| First value in hospital (IQR)—g/dL | 3.3 | (0.9) | 2.8 | (0.9) | 1.9 | (0.7) | 2.7 | (1) |
| Lowest value (IQR)—g/dL | 3.1 | (0.7) | 2.2 | (1.1) | 1.5 | (0.4) | 2.1 | (1.1) |
| <2.0 g/dL during hospital stay—no. (%) | 1/33 | (3.3) | 21/70 | (30) | 17/18 | (94.4) | 38/88 | (43.2) |
| Median antithrombin III (IQR)—% | ||||||||
| First value in hospital (IQR)—% | 95 | (23.5) | 93 | (22) | 68 | (34) | 89 | (24.5) |
| Lowest value (IQR)—% | 95 | (26) | 78 | (40) | 52.5 | (43) | 71 | (42) |
| <70% during hospital stay—no. (%) | 5/22 | (22.7) | 21/52 | (40.4) | 12/18 | (66.7) | 33/70 | (47.1) |
| Parameter | Hazard Ratio | 95% Confidence Intervals | p | |
|---|---|---|---|---|
| Kidney injury | 2.868 | 1.484 | 5.505 | 0.0017 |
| Age (year) | 0.995 | 0.979 | 1.011 | 0.5412 |
| Sex: male | 1.088 | 0.660 | 1.794 | 0.7417 |
| Chronic heart disease | 1.191 | 0.673 | 2.110 | 0.5480 |
| Chronic lung disease | 1.311 | 0.756 | 2.273 | 0.3355 |
| Chronic kidney disease | 0.645 | 0.275 | 1.513 | 0.3138 |
| RRT | 3.132 | 0.897 | 10.938 | 0.0736 |
| Malignant tumor disease | 0.888 | 0.337 | 2.344 | 0.8109 |
| Diabetes mellitus | 0.973 | 0.557 | 1.701 | 0.9247 |
| Immunosuppressive therapy | 0.938 | 0.444 | 1.980 | 0.8660 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gross, O.; Moerer, O.; Rauen, T.; Böckhaus, J.; Hoxha, E.; Jörres, A.; Kamm, M.; Elfanish, A.; Windisch, W.; Dreher, M.; et al. Validation of a Prospective Urinalysis-Based Prediction Model for ICU Resources and Outcome of COVID-19 Disease: A Multicenter Cohort Study. J. Clin. Med. 2021, 10, 3049. https://doi.org/10.3390/jcm10143049
Gross O, Moerer O, Rauen T, Böckhaus J, Hoxha E, Jörres A, Kamm M, Elfanish A, Windisch W, Dreher M, et al. Validation of a Prospective Urinalysis-Based Prediction Model for ICU Resources and Outcome of COVID-19 Disease: A Multicenter Cohort Study. Journal of Clinical Medicine. 2021; 10(14):3049. https://doi.org/10.3390/jcm10143049
Chicago/Turabian StyleGross, Oliver, Onnen Moerer, Thomas Rauen, Jan Böckhaus, Elion Hoxha, Achim Jörres, Matthias Kamm, Amin Elfanish, Wolfram Windisch, Michael Dreher, and et al. 2021. "Validation of a Prospective Urinalysis-Based Prediction Model for ICU Resources and Outcome of COVID-19 Disease: A Multicenter Cohort Study" Journal of Clinical Medicine 10, no. 14: 3049. https://doi.org/10.3390/jcm10143049
APA StyleGross, O., Moerer, O., Rauen, T., Böckhaus, J., Hoxha, E., Jörres, A., Kamm, M., Elfanish, A., Windisch, W., Dreher, M., Floege, J., Kluge, S., Schmidt-Lauber, C., Turner, J.-E., Huber, S., Addo, M. M., Scheithauer, S., Friede, T., Braun, G. S., ... Blaschke, S. (2021). Validation of a Prospective Urinalysis-Based Prediction Model for ICU Resources and Outcome of COVID-19 Disease: A Multicenter Cohort Study. Journal of Clinical Medicine, 10(14), 3049. https://doi.org/10.3390/jcm10143049









