First Evaluation of a New Dynamic Scoring System Intended to Support Prescription of Adjuvant CytoSorb Hemoadsorption Therapy in Patients with Septic Shock
Abstract
:1. Background
2. Material and Methods
2.1. Ethics Approval, Legal Considerations
2.2. Study Design
2.3. Objectives
2.4. Assessed Parameters
2.5. Data Collection
2.6. Procedure
2.7. Statistics
3. Results
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.-D.; Coopersmith, C.M.; et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 801–810. [Google Scholar] [CrossRef]
- Seymour, C.W.; Rosengart, M.R. Septic Shock: Advances in Diagnosis and Treatment. JAMA 2015, 314, 708–717. [Google Scholar] [CrossRef] [Green Version]
- Fleischmann, C.; Thomas–Rueddel, D.O.; Hartmann, M.; Hartog, C.S.; Welte, T.; Heublein, S.; Dennler, U.; Reinhart, K. Hospital Incidence and Mortality Rates of Sepsis. Dtsch. Aerzteblatt Int. 2016, 113, 159–166. [Google Scholar] [CrossRef] [Green Version]
- Kogelmann, K.; Jarczak, D.; Scheller, M.; Druner, M. Hemoadsorption by CytoSorb in septic patients—A case series. Crit. Care 2017, 21, 74. [Google Scholar] [CrossRef] [Green Version]
- Friesecke, S.; Stecher, S.; Gross, S.; Felix, S.B.; Nierhaus, A. Extracorporeal Cytokine Elimination as Rescue Therapy in Refractory Septic Shock—A Prospective Single-Center Study. J. Artif. Organs 2017, 20, 252–259. [Google Scholar] [CrossRef] [PubMed]
- Bassi, E.; Park, M.; Azevedo, L.C.P. Therapeutic Strategies for High-Dose Vasopressor-Dependent Shock. Crit. Care Res. Pract. 2013, 2013, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Hawchar, F.; László, I.; Öveges, N.; Trásy, D.; Ondrik, Z.; Molnar, Z. Extracorporeal cytokine adsorption in septic shock: A proof of concept randomized, controlled pilot study. J. Crit. Care 2019, 49, 172–178. [Google Scholar] [CrossRef] [Green Version]
- Mehta, Y.; Mehta, C.; Kumar, A.; George, J.V.; Gupta, A.; Nanda, S.; Kochhar, G.; Raizada, A. Experience with hemoadsorption (Cy-toSorb®) in the management of septic shock patients. World J. Crit. Care Med. 2020, 9, 1–12. [Google Scholar] [CrossRef]
- Träger, K.; Skrabal, C.; Fischer, G.; Datzmann, T.; Schroeder, J.; Fritzler, D.; Hartmann, J.; Liebold, A.; Reinelt, H. Hemoadsorption Treatment of Patients with Acute Infective Endocarditis during Surgery with Cardiopulmonary Bypass—A Case Series. Int. J. Artif. Organs 2017, 40, 240–249. [Google Scholar] [CrossRef] [Green Version]
- Leonardis, F.; De Angelis, V.; Frisardi, F.; Pietrafitta, C.; Riva, I.; Valetti, T.M.; Broletti, V.; Marchesi, G.; Menato, L.; Nani, R.; et al. Effect of Hemoadsorption for Cytokine Removal in Pneumococcal and Meningococcal Sepsis. Case Rep. Crit. Care 2018, 2018, 1–7. [Google Scholar] [CrossRef]
- Rhodes, A.A.; Evans, L.E.; Alhazzani, W.; Levy, M.M.; Antonelli, M.; Ferrer, R.; Kumar, A.; Sevransky, J.E.; Sprung, C.L.; Nunnally, M.E.; et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Crit. Care Med. 2017, 45, 486–552. [Google Scholar] [CrossRef]
- Vincent, J.L.; Moreno, R.; Takala, J.; Willatts, S.; De Mendonça, A.; Bruining, H.; Reinhart, C.K.; Suter, P.M.; Thijs, L.G. The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med. 1996, 22, 707–710. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, F.L.; Bota, D.P.; Bross, A.; Melot, C.; Vincent, J.-L. Serial Evaluation of the SOFA Score to Predict Outcome in Critically Ill Patients. JAMA 2001, 286, 1754–1758. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lewandowski, K. Scoring-Systeme auf der Intensivtherapiestation. Der Anaesthesist 2003, 52, 965–990. [Google Scholar] [CrossRef]
- Goldhill, D.R.; Withington, P.S. Mortality predicted by APACHE II. The effect of changes in physiological values and post-ICU hospital mortality. Anaesthesia 1996, 51, 719–723. [Google Scholar]
- Knaus, W.A.; Draper, E.A.; Wagner, D.P.; Zimmerman, J.E. APACHE II: A severity of disease classification system. Crit. Care Med. 1985, 13, 818–829. [Google Scholar] [CrossRef]
- Friesecke, S.; Träger, K.; Schittek, G.; Molnar, Z.; Bach, F.; Kogelmann, K.; Bogdanski, R.; Weyland, A.; Nierhaus, A.; Nestler, F.; et al. International registry on the use of the CytoSorb® adsorber in ICU patients: Study protocol and preliminary results. Med. Klin. Intensivmed. Notfallmed. 2019, 114, 699–707. [Google Scholar] [CrossRef] [PubMed]
- Rugg, C.; Klose, R.; Hornung, R.; Innerhofer, N.; Bachler, M.; Schmid, S.; Fries, D.; Ströhle, M. Hemoadsorption with CytoSorb in Septic Shock reduces Catecholamine Requirements and In-Hospital Mortality: A Single-Center Retrospective ‘Genetic’ Matched Analysis. Biomedicines 2020, 8, 539. [Google Scholar] [CrossRef] [PubMed]
- Bellomo, R.; Kellum, J.A.; Ronco, C.; Wald, R.; Martensson, J.; Maiden, M.; Bagshaw, S.M.; Glassford, N.J.; Lankadeva, Y.; Vaara, S.T.; et al. Acute kidney injury in sepsis. Intensive Care Med. 2017, 43, 816–828. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Uchino, S.; Kellum, J.A.; Bellomo, R.; Doig, G.S.; Morimatsu, H.; Morgera, S.; Schetz, M.; Tan, I.; Bouman, C.; Macedo, E.; et al. Acute renal failure in critically ill patients: A multi-national, multicenter study. JAMA 2005, 294, 813–818. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khwaja, A. KDIGO Clinical Practice Guidelines for Acute Kidney Injury. Nephron 2012, 120, c179–c184. [Google Scholar] [CrossRef] [PubMed]
- Barbar, S.D.; Clere-Jehl, R.; Bourredjem, A.; Hernu, R.; Montini, F.; Bruyère, R.; Lebert, C.; Bohé, J.; Badie, J.; Eraldi, J.-P.; et al. Timing of Renal-Replacement Therapy in Patients with Acute Kidney Injury and Sepsis. N. Engl. J. Med. 2018, 379, 1431–1442. [Google Scholar] [CrossRef] [PubMed]
- Liaño, F.; Junco, E.; Pascual, J.; Madero, R.; Verde, E. The spectrum of acute renal failure in the intensive care unit compared with that seen in other settings. The Madrid Acute Renal Failure Study Group. Kidney Int. Suppl. 1998, 66, 16–24. [Google Scholar]
- Chertow, G.M.; Burdick, E.; Honour, M.; Bonventre, J.V.; Bates, D.W. Acute kidney injury, mortality, length of stay, and costs in hos-pitalized patients. J. Am. Soc. Nephrol. 2005, 16, 3365–3370. [Google Scholar] [CrossRef] [Green Version]
- Oppert, M.; Engel, C.; Brunkhorst, F.-M.; Bogatsch, H.; Reinhart, K.; Frei, U.; Eckardt, K.-U.; Loeffler, M.; John, S. German Competence Network Sepsis (Sepnet) Acute renal failure in patients with severe sepsis and septic shock--a significant independent risk factor for mortality: Results from the German Prevalence Study. Nephrol. Dial. Transplant. 2007, 23, 904–909. [Google Scholar] [CrossRef]
- Brouwer, W.P.; Duran, S.; Kuijper, M.; Ince, C. Hemoadsorption with CytoSorb shows a decreased observed versus expected 28-day all-cause mortality in ICU patients with septic shock: A propensity-score-weighted retrospective study. Crit. Care 2019, 23, 1–9. [Google Scholar] [CrossRef] [Green Version]
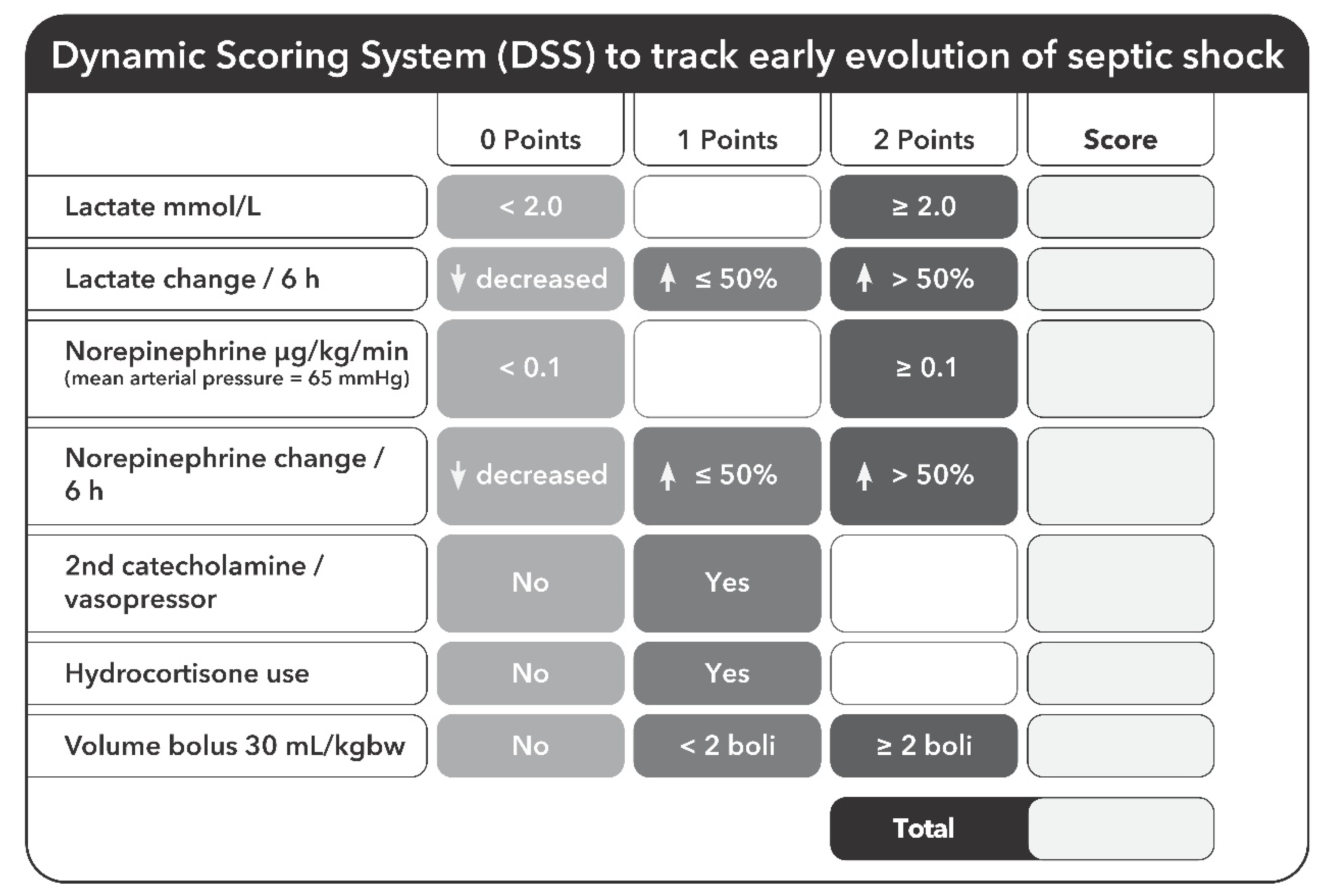
| DSS < 6 (n = 98) | DSS 6–8 (n = 294) | DSS > 8 (n = 110) | p-Value (DSS < 6 vs. DSS 6–8) | p-Value (DSS < 6 vs. DSS > 8) | p-Value (DSS 6–8 vs. DSS > 8) | |
|---|---|---|---|---|---|---|
| Age (years) | 68.5 (±12.4) | 65.11 (±14.7) | 66.6 (±14.4) | 0.027 | 0.325 | 0.335 |
| APACHE II (points) | 35.6 (±9.2) | 36.1 (±10.2) | 36.9 (±9.5) | 0.617 | 0.321 | 0.515 |
| SAPS II (points) | 50.2 (±13.2) | 53.7 (±14.8) | 62.3 (±18.6) | 0.033 | <0.001 * | <0.001 * |
| Ventilator days | 7.8 (±10.1) | 11.1 (±11.1) | 11.7 (±16.1) | 0.016 * | 0.038 | 0.776 |
| ICU stay (days) | 13.2 (±11.9) | 17.2 (±21.0) | 16.4 (±19.8) | 0.073 | 0.173 | 0.695 |
| Hospital stay (days) | 24.1 (±24.1) | 26.3 (±31.0) | 23.1 (±34.0) | 0.457 | 0.804 | 0.379 |
| ICU mortality (%) | 42 (42.9%) | 165 (56.1%) | 71 (64.5%) | 0.023 | 0.002 * | 0.127 |
| Hospital mortality (%) | 48 (49.0%) | 173 (58.8%) | 79 (71.8%) | 0.089 | <0.001 * | 0.017 * |
| Lactate T0 (mmol/L) | 3.90 (±3.61) | 4.65 (±3.50) | 4.93 (±3.21) | 0.074 | 0.032 | 0.450 |
| Lactate T6 (mmol/L) | 2.65 (±2.89) | 3.98 (±3.25) | 6.74 (±3.85) | <0.001 * | <0.001 * | <0.001 * |
| Norepinephrine T0 (µg/kg/min) | 0.21 (±0.25) | 0.39 (±0.41) | 0.43 (±0.42) | <0.001 * | <0.001 * | 0.314 |
| Norepinephrine T6 (µg/kg/min) | 0.20 (±0.19) | 0.49 (±0.40) | 0.72 (±0.36) | <0.001 * | <0.001 * | <0.001 * |
| Second catecholamine T0 (%) | 3 (3.1%) | 81 (27.6%) | 68 (61.8%) | <0.001 * | <0.001 * | <0.001 * |
| Hydrocortisone T0 (%) | 5 (5.1%) | 135 (45.9%) | 89 (80.9%) | <0.001 * | <0.001 * | <0.001 * |
| Volume bolus used (mL/kg) | 62.4 (±20.4) | 80.4 (±28.8) | 90.0 (±31.8) | <0.001 * | <0.001 * | 0.006 * |
| Dynamic Scoring System (points) | 4.41 (±0.94) | 7.04 (±0.81) | 9.63 (±0.77) | <0.001 * | <0.001 * | <0.001 * |
| DSS < 6 (n = 17) | DSS 6–8 (n = 118) | DSS > 8 (n = 63) | p-Value (DSS < 6 vs. DSS 6–8) | p-Value (DSS < 6 vs. DSS > 8) | p-Value (DSS 6–8 vs. DSS > 8) | |
|---|---|---|---|---|---|---|
| Age (years) | 66.8 (±10.43) | 60.5 (±14.80) | 64.4 (±15.76) | 0.037 | 0.462 | 0.097 |
| APACHE II (points) | 34.0 (±9.29) | 33.4 (±10.23) | 34.6 (±10.03) | 0.811 | 0.838 | 0.473 |
| SAPS II (points) | 56.2 (±18.81) | 56.6 (±15.30) | 65.8 (±19.85) | 0.947 | 0.079 | <0.001 * |
| Ventilator days | 7.6 (±8.17) | 13.9 (±19.05) | 13.0 (±18.53) | 0.022 | 0.247 | 0.773 |
| ICU stay (days) | 12.4 (±8.20) | 21.4 (±25.20) | 17.9 (±22.85) | 0.147 | 0.123 | 0.355 |
| Hospital stay (days) | 26.1 (±33.70) | 30.9 (±37.39) | 25.35 (±40.39) | 0.599 | 0.939 | 0.356 |
| ICU mortality (%) | 11 (64.7%) | 66 (55.9%) | 41 (65.1%) | 0.501 | 0.978 | 0.235 |
| Hospital mortality (%) | 13 (76.5%) | 67 (56.8%) | 46 (73.0%) | 0.122 | 0.774 | 0.032 |
| CytoSorb therapy delay (hours) | 52.6 (±30.50) | 23.0 (±21.50) | 18.20 (±20.57) | <0.001 * | <0.001 * | 0.138 |
| Number of CytoSorb adsorbers used (n) | 2.2 (±0.77) | 2.7 (±1.57) | 2.7 (±1.58) | 0.230 | 0.117 | 0.895 |
| Lactate T0 (mmol/L) | 3.12 (±3.49) | 4.87 (±3.81) | 5.01 (±3.26) | 0.076 | 0.041 | 0.805 |
| Lactate T6 (mmol/L) | 2.40 (±3.02) | 4.09 (±3.34) | 6.60 (±3.17) | 0.051 | <0.001 * | <0.001 * |
| Norepinephrine T0 (µg/kg/min) | 0.31 (±0.26) | 0.48 (±0.50) | 0.47 (±0.46) | 0.036 | 0.169 | 0.906 |
| Norepinephrine T6 (µg/kg/min) | 0.31 (±0.25) | 0.50 (±0.42) | 0.75 (±0.35) | 0.012 * | <0.001 * | <0.001 * |
| Second catecholamine T0 (%) | 3 (17.6%) | 57 (48.3%) | 51 (81.0%) | 0.017 * | <0.001 * | <0.001 * |
| Hydrocortisone T0 (%) | 4 (23.5%) | 78 (66.1%) | 54 (85.7%) | <0.001 * | <0.001 * | 0.003 * |
| Volume bolus used (mL/kg) | 63.0 (±14.4) | 77.4 (±27.0) | 82.8 (±28.8) | 0.038 | 0.011 * | 0.261 |
| Dynamic Scoring System (points) | 4.23 (±0.97) | 7.22 (±0.82) | 9.84 (±0.86) | <0.001 * | <0.001 * | <0.001 * |
| All Patients, DSS < 6 | All Patients, DSS 6–8 | All Patients, DSS > 8 | p-Value (<6 vs. 6–8) | p-Value (<6 vs. >8) | p-Value (6–8 vs. >8) | |
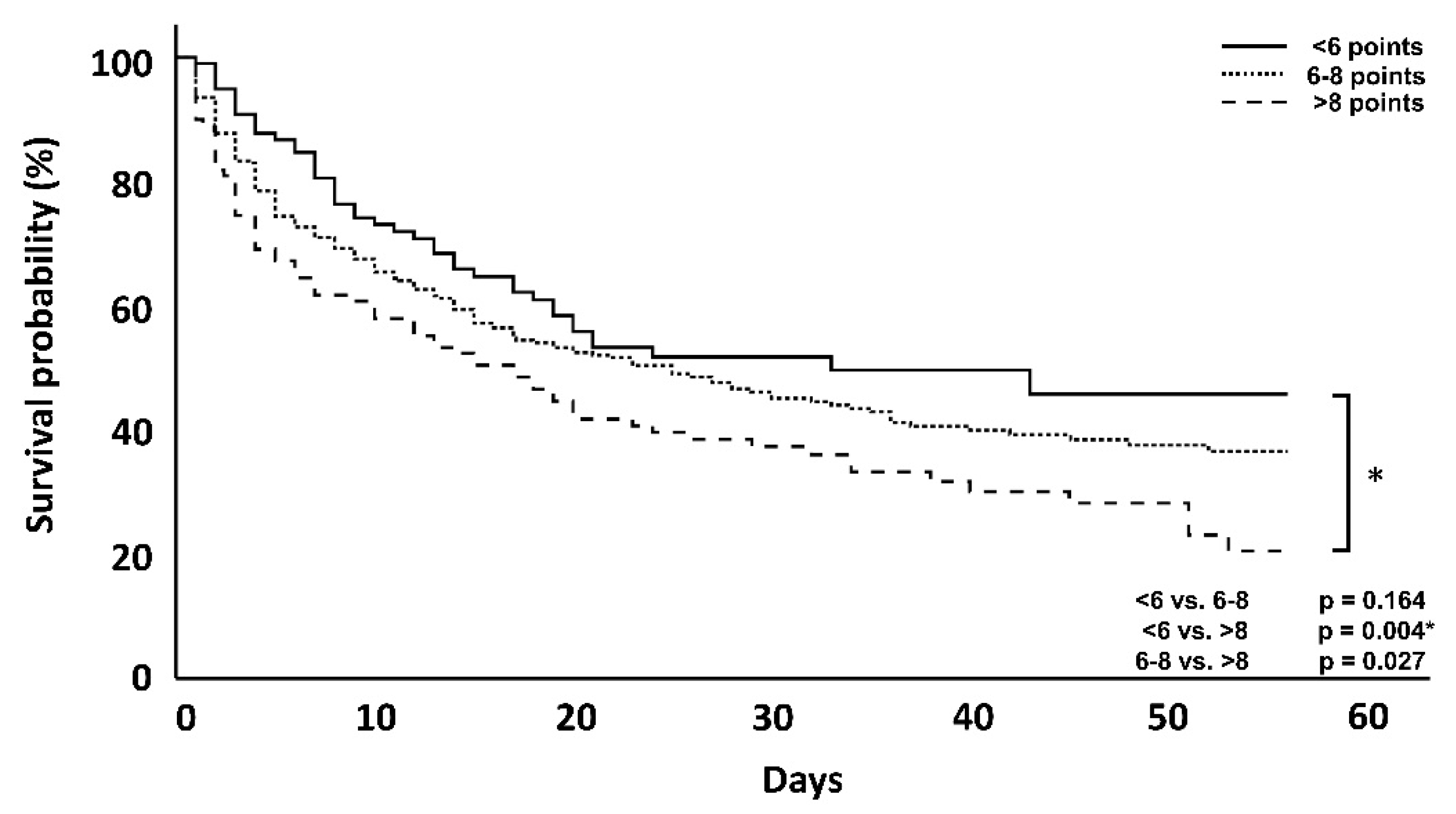
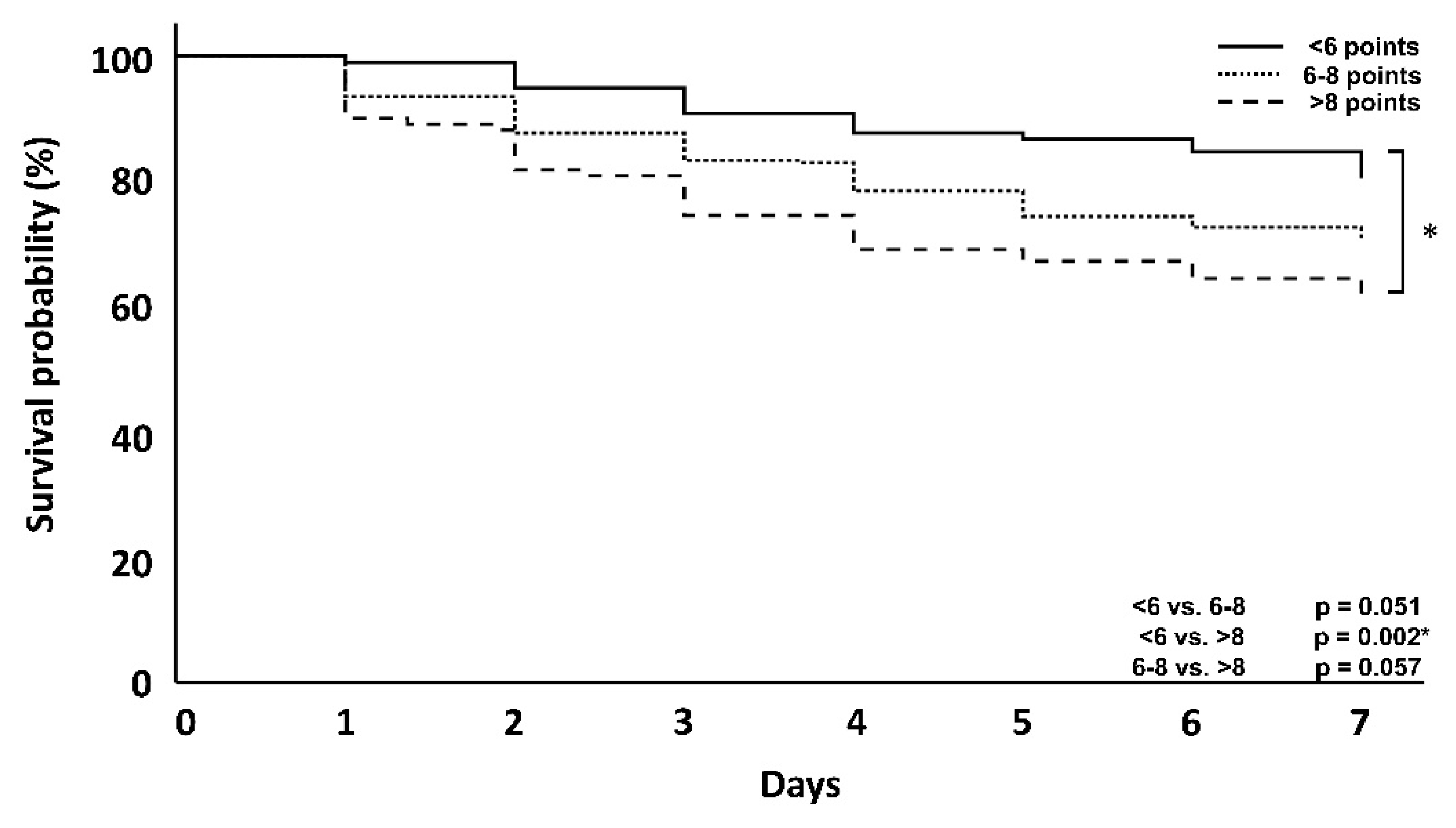
| 7-day mortality | 19 (19.4%) | 85 (28.9%) | 42 (38.2%) | 0.051 | 0.002 * | 0.057 |
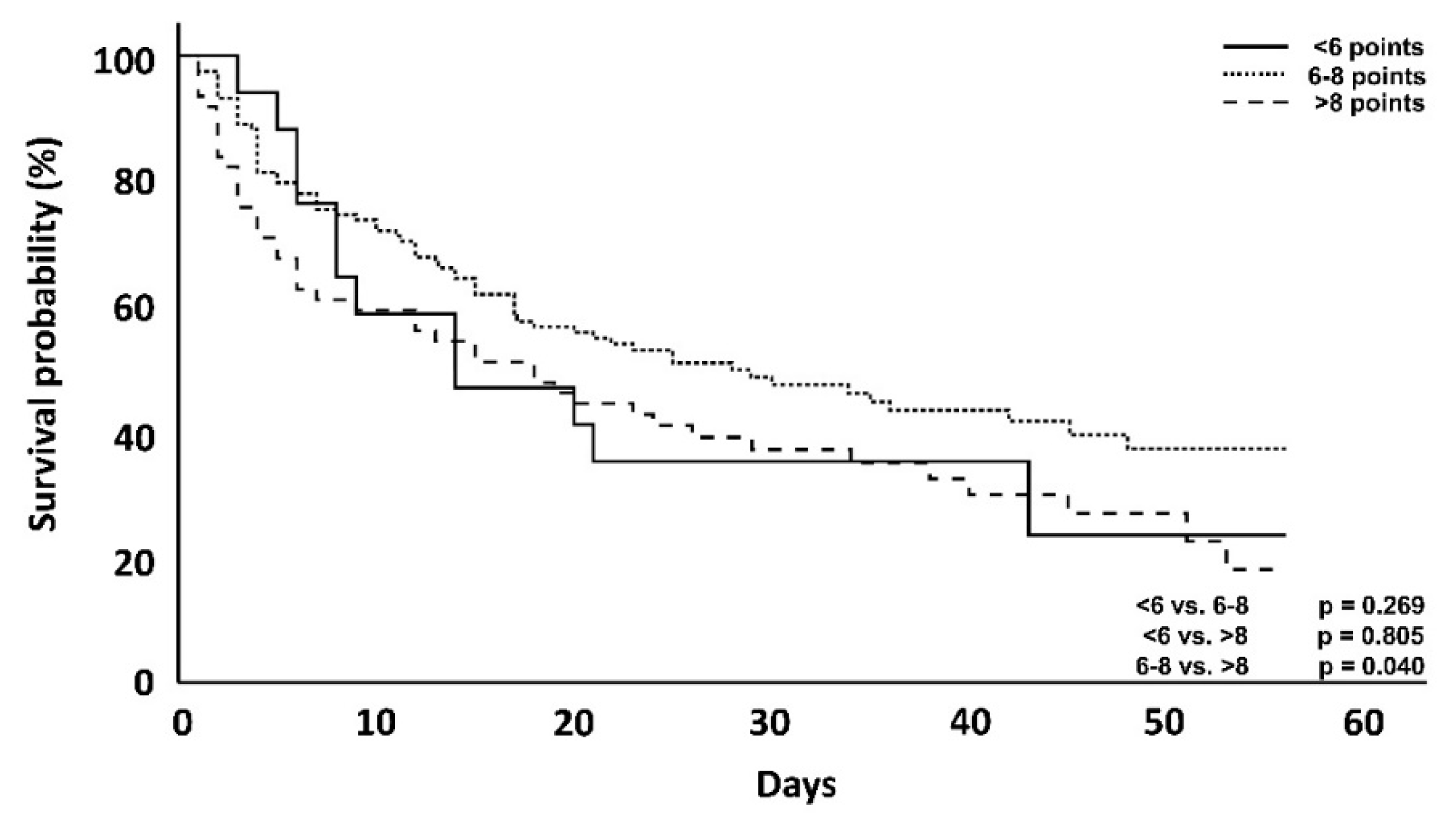
| 56-day mortality | 45 (45.9%) | 165 (56.1%) | 76 (69.1%) | 0.164 | 0.004 * | 0.027 |
| ICU mortality | 42 (42.8%) | 165 (56.1%) | 71 (64.5%) | 0.023 | 0.002 * | 0.127 |
| Hospital mortality | 48 (48.9%) | 173 (58.8%) | 80 (72.7%) | 0.089 | <0.001 * | 0.017 * |
| CytoSorb Patients, DSS < 6 | CytoSorb Patients, DSS 6–8 | CytoSorb Patients, DSS > 8 | p-Value (<6 vs. 6–8) | p-Value (<6 vs. >8) | p-Value (6–8 vs. >8) | |
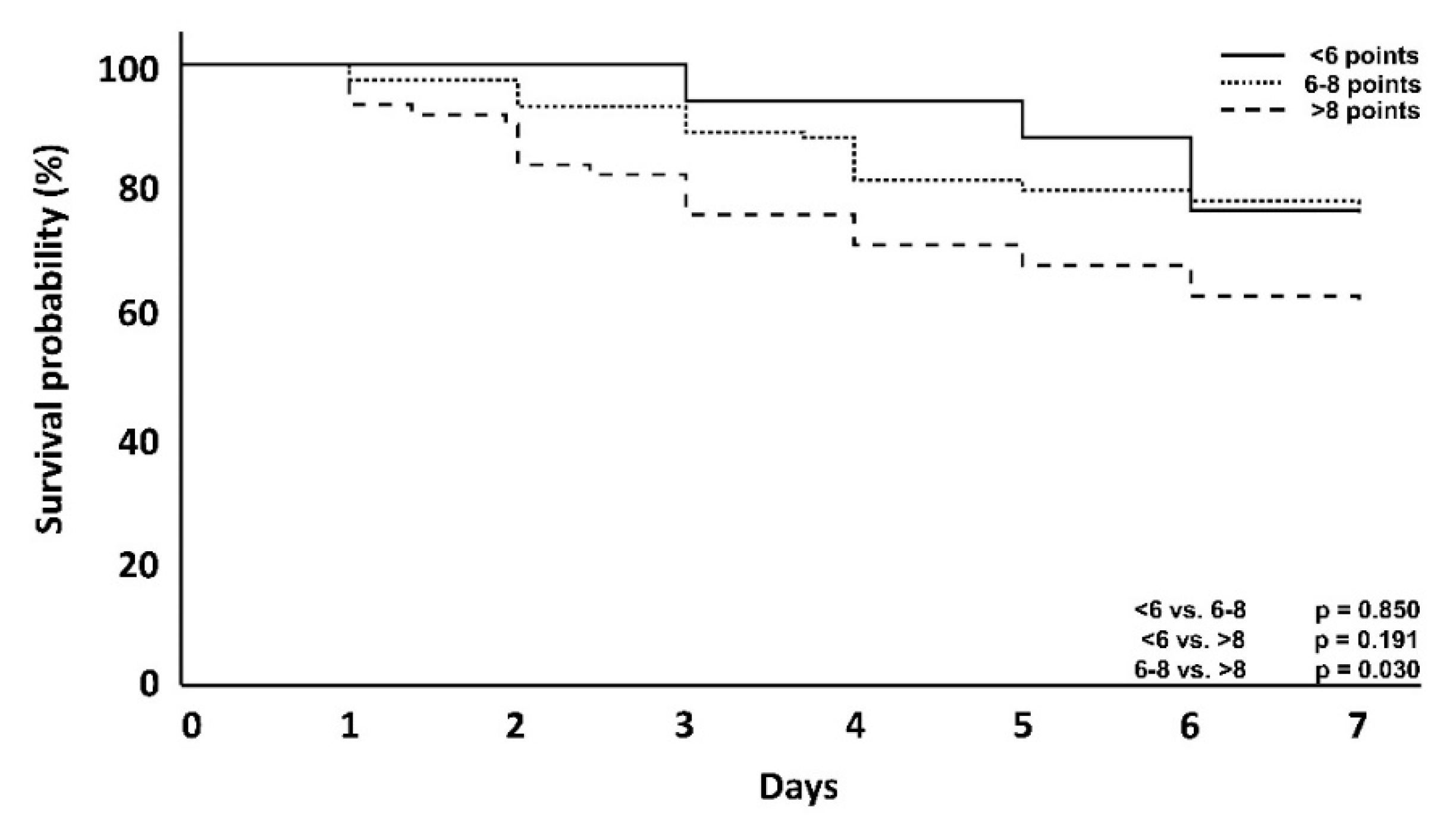
| 7-day mortality | 4 (23.5%) | 29 (24.4%) | 24 (38.7%) | 0.850 | 0.030 | 0.191 |
| 56-day mortality | 12 (70.5%) | 66 (55.5%) | 44 (71.0%) | 0.269 | 0.040 | 0.805 |
| ICU mortality | 11 (64.7%) | 66 (55.9%) | 41 (65.0%) | 0.501 | 0.978 | 0.235 |
| Hospital mortality | 13 (76.4%) | 67 (56.7%) | 47 (74.6%) | 0.102 | 0.877 | 0.018 |
| CytoSorb Therapy Delay ≤ 12 h | CytoSorb Therapy Delay > 12–24 h | CytoSorb Therapy Delay > 24 h | p-Value (≤12 vs. >12–24 h) | p-Value (≤12 vs. >24 h) | p-Value (>12–24 vs. >24 h) | |
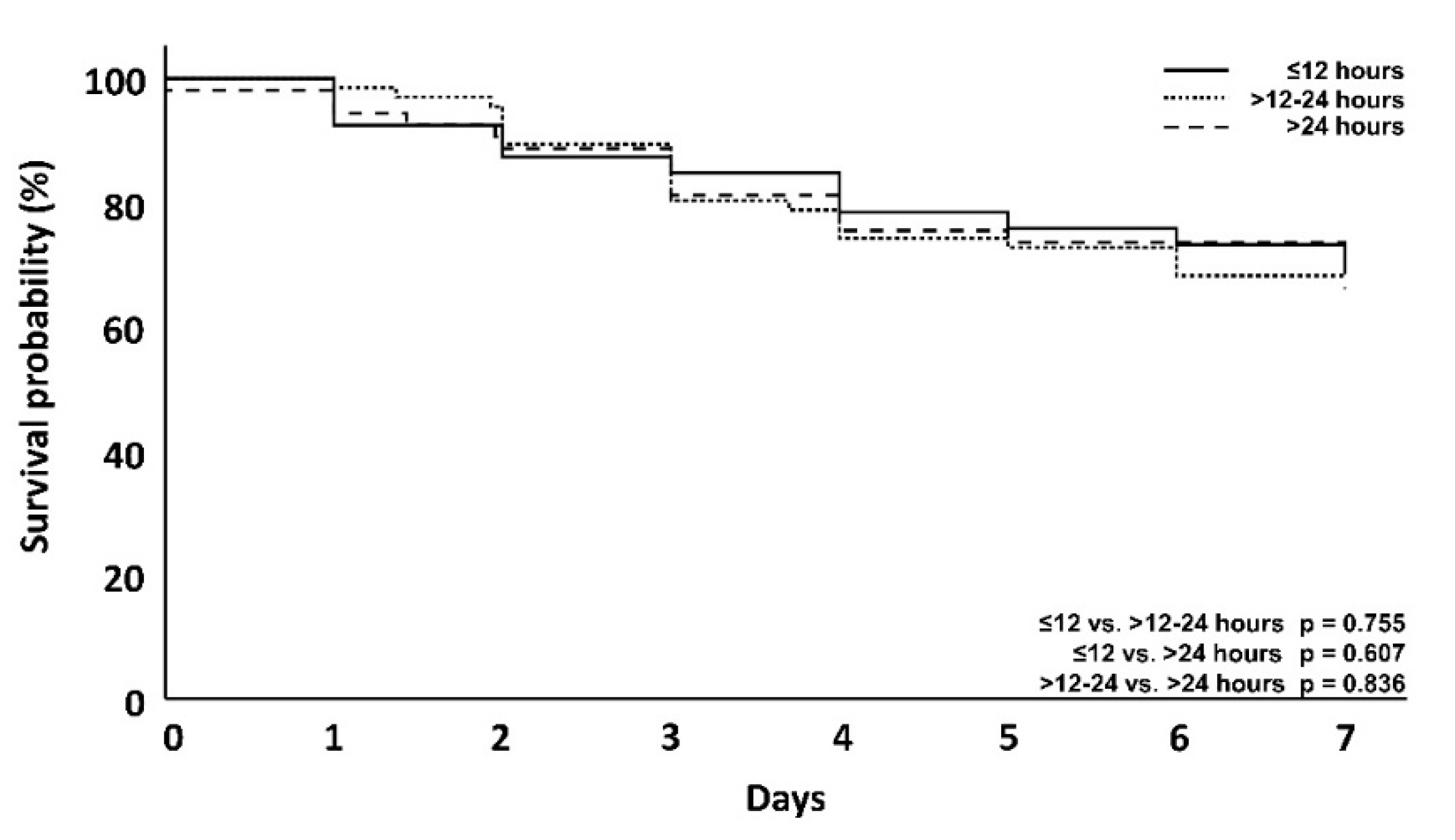
| 7-day mortality | 23 (29.1%) | 21 (31.8%) | 18 (34.0%) | 0.755 | 0.607 | 0.836 |
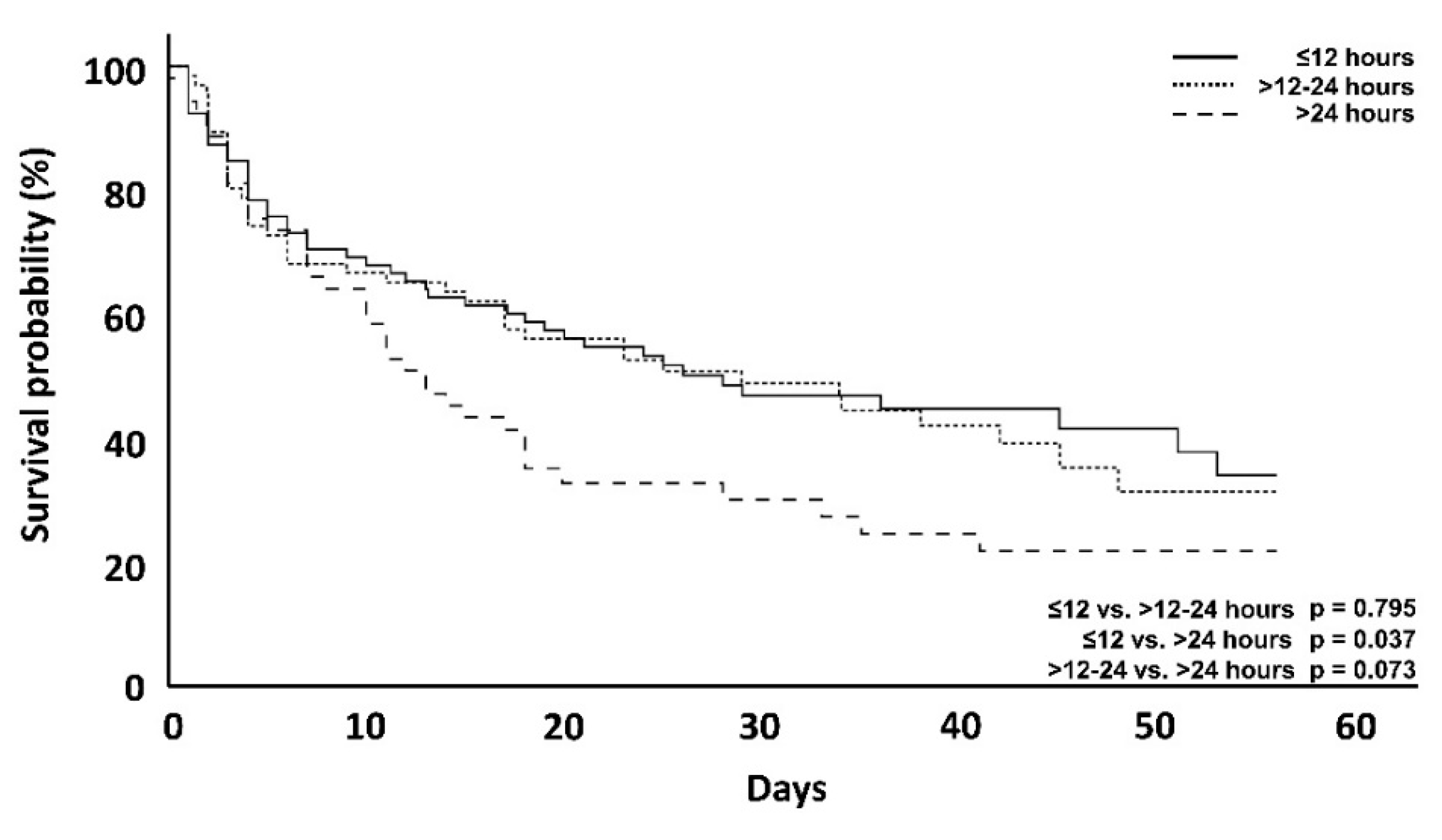
| 56-day mortality | 44 (55.7%) | 39 (59.1%) | 39 (73.6%) | 0.795 | 0.037 | 0.073 |
| ICU mortality | 40 (50.0%) | 39 (60.0%) | 39 (73.5%) | 0.231 | 0.006 * | 0.123 |
| Hospital mortality | 45 (56.2%) | 40 (61.5%) | 42 (79.2%) | 0.430 | 0.006 * | 0.038 |
| Therapy Delay ≤12 h (n = 80) | Therapy Delay >12–24 h (n = 65) | Therapy Delay >24 h (n = 53) | p-Value (<12 vs. 12–24 h) | p-Value (<12 vs. >24 h) | p-Value (12–24 vs. >24 h) | |
|---|---|---|---|---|---|---|
| Age (years) | 62.0 (±13.72) | 61.2 (±16.06) | 64.1 (±15.28) | 0.670 | 0.410 | 0.310 |
| APACHE II (points) | 32.7 (±9.38) | 36.2 (±9.31) | 32.7 (±11.53) | 0.028 | 0.994 | 0.078 |
| SAPS II (points) | 64.2 (±19.26) | 56.1 (±14.93) | 56.7 (±16.96) | 0.007 * | 0.021 | 0.850 |
| Ventilator days | 12.9 (±18.50) | 13.2 (±19.11) | 13.0 (±16.93) | 0.930 | 0.996 | 0.937 |
| ICU stay (days) | 20.2 (±25.80) | 19.9 (±23.95) | 18.0 (±19.49) | 0.930 | 0.578 | 0.643 |
| Hospital stay (days) | 28.1 (±39.99) | 32.1 (±42.71) | 25.0 (±29.98) | 0.560 | 0.609 | 0.293 |
| ICU mortality (%) | 40 (50.0%) | 39 (60.0%) | 39 (73.5%) | 0.231 | 0.006 * | 0.123 |
| Hospital mortality (%) | 45 (56.2%) | 40 (61.5%) | 42 (79.2%) | 0.430 | 0.006 * | 0.038 |
| CytoSorb therapy delay (hours) | 6.8 (±4.50) | 20.4 (±3.99) | 54.6 (±25.92) | <0.001 * | <0.001 * | <0.001 * |
| Number of CytoSorb adsorbers used (n) | 3.03 (±1.66) | 2.69 (±1.62) | 2.24 (±0.97) | 0.211 | 0.002 * | 0.082 |
| Lactate T0 (mmol/L) | 4.73 (±3.48) | 5.11 (±3.90) | 4.38 (±3.55) | 0.540 | 0.574 | 0.292 |
| Lactate T6 (mmol/L) | 5.09 (±3.63) | 5.05 (±3.67) | 3.80 (±3.00) | 0.940 | 0.034 | 0.046 |
| Norepinephrine T0 (µg/kg/min) | 0.50 (±0.65) | 0.45 (±0.26) | 0.42 (±0.37) | 0.506 | 0.323 | 0.613 |
| Norepinephrine T6 (µg/kg/min) | 0.64 (±0.46) | 0.54 (±0.35) | 0.50 (±0.40) | 0.153 | 0.083 | 0.647 |
| Second catecholamine T0 (%) | 64 (80.0%) | 25 (38.0%) | 22 (41.5%) | <0.001 * | <0.001 * | 0.740 |
| Hydrocortisone T0 (%) | 63 (78.7%) | 44 (67.6%) | 29 (56.6%) | 0.134 | 0.008 * | 0.235 |
| Volume bolus used (mL/kg) | 75.0 (±25.8) | 81.6 (±28.8) | 78.0 (±27.6) | 0.122 | 0.510 | 0.444 |
| Dynamic Scoring System (points) | 8.48 (±1.53) | 7.60 (±1.58) | 7.01 (±2.19) | 0.001 * | <0.001 * | 0.093 |
| Control + RRT Group (n = 69) | CytoSorb Group (n = 198) | p-Value | |
|---|---|---|---|
| Age (years) | 66.2 (±12.4) | 62.3 (±14.9) | 0.035 |
| APACHE II (points) | 39.8 (±9.6) | 33.8 (±10.0) | <0.001 |
| SAPS II (points) | 56.2 (±14.8) | 59.5 (±17.6) | 0.144 |
| Ventilator days | 12.3 (±13.4) | 13.0 (±18.2) | 0.715 |
| ICU stay (days) | 19.9 (±18.1) | 19.5 (±23.5) | 0.882 |
| Hospital stay (days) | 30.2 (±28.9) | 28.7 (±37.9) | 0.703 |
| ICU mortality (%) | 44 (63.8%) | 118 (59.6%) | 0.540 |
| Hospital mortality (%) | 49 (71.0%) | 123 (63.6%) | 0.268 |
| Lactate T0 (mmol/L) | 4.96 (±4.28) | 4.76 (±3.63) | 0.738 |
| Lactate T6 (mmol/L) | 5.08 (±4.40) | 4.73 (±3.52) | 0.565 |
| Norepinephrine T0 (µg/kg/min) | 0.37 (±0.41) | 0.46 (±0.48) | 0.142 |
| Norepinephrine T6 (µg/kg/min) | 0.55 (±0.44) | 0.57 (±0.41) | 0.783 |
| Second catecholamine T0 (%) | 17 (24.6%) | 111 (56.1%) | <0.001 |
| Hydrocortisone T0 (%) | 37 (53.6%) | 136 (68.7%) | 0.024 |
| Volume bolus used (mL/kg) | 75.6 (±30.0) | 78.0 (±27.6) | 0.537 |
| Dynamic Scoring System (points) | 7.20 (±1.68) | 7.80 (±1.82) | 0.014 |
| ICU Mortality | Hospital Mortality | |||||
|---|---|---|---|---|---|---|
| Control + RRT Group (n = 69) | CytoSorb Group (n = 198) | p-Value | Control + RRT Group (n = 69) | CytoSorb Group (n = 198) | p-Value | |
| DSS <6 points, CytoSorb delay ≤12 h | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| DSS 6–8 points, CytoSorb delay ≤12 h | 27 (58.7%) | 21 (47.7%) | 0.303 | 30 (65.2%) | 21 (47.7%) | 0.096 |
| DSS >8 points, CytoSorb delay ≤12 h | 12 (92.3%) | 17 (51.5%) | 0.009 * | 12 (92.3%) | 21 (63.6%) | 0.053 |
| DSS <6 points, CytoSorb delay >12–24 h | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| DSS 6–8 points, CytoSorb delay >12–24 h | 27 (58.7%) | 25 (54.3%) | 0.678 | 30 (65.2%) | 26 (56.5%) | 0.398 |
| DSS >8 points, CytoSorb delay >12–24 h | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| DSS <6 points, CytoSorb delay >24 h | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| DSS 6–8 points, CytoSorb delay >24 h | 27 (58.7%) | 19 (70.3%) | 0.318 | 30 (65.2%) | 19 (70.3%) | 0.654 |
| DSS >8 points, CytoSorb delay >24 h | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Predictor Variable | Odds Ratio | 95% Confidence Interval | p-Value |
|---|---|---|---|
| CytoSorb Therapy (Yes/No) | 0.552 | 0.275, 1.108 | 0.095 |
| DSS Score | 1.237 | 1.106, 1.383 | <0.001 |
| Therapy Delay (h) | 1.015 | 1.001, 1.030 | 0.034 |
| RRT (Yes/No) | 1.795 | 0.991, 3.252 | 0.054 |
| Age (Years) | 1.037 | 1.023, 1.052 | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kogelmann, K.; Hübner, T.; Schwameis, F.; Drüner, M.; Scheller, M.; Jarczak, D. First Evaluation of a New Dynamic Scoring System Intended to Support Prescription of Adjuvant CytoSorb Hemoadsorption Therapy in Patients with Septic Shock. J. Clin. Med. 2021, 10, 2939. https://doi.org/10.3390/jcm10132939
Kogelmann K, Hübner T, Schwameis F, Drüner M, Scheller M, Jarczak D. First Evaluation of a New Dynamic Scoring System Intended to Support Prescription of Adjuvant CytoSorb Hemoadsorption Therapy in Patients with Septic Shock. Journal of Clinical Medicine. 2021; 10(13):2939. https://doi.org/10.3390/jcm10132939
Chicago/Turabian StyleKogelmann, Klaus, Tobias Hübner, Franz Schwameis, Matthias Drüner, Morten Scheller, and Dominik Jarczak. 2021. "First Evaluation of a New Dynamic Scoring System Intended to Support Prescription of Adjuvant CytoSorb Hemoadsorption Therapy in Patients with Septic Shock" Journal of Clinical Medicine 10, no. 13: 2939. https://doi.org/10.3390/jcm10132939
APA StyleKogelmann, K., Hübner, T., Schwameis, F., Drüner, M., Scheller, M., & Jarczak, D. (2021). First Evaluation of a New Dynamic Scoring System Intended to Support Prescription of Adjuvant CytoSorb Hemoadsorption Therapy in Patients with Septic Shock. Journal of Clinical Medicine, 10(13), 2939. https://doi.org/10.3390/jcm10132939






