How the Gut Microbiome Links to Menopause and Obesity, with Possible Implications for Endometrial Cancer Development
Abstract
:1. Introduction
2. Data Selection and Extraction
2.1. Literature Search
- “Estrogen”, “Estradiol”, “Sex steroid hormones” and varieties of “Gut microbiome”.
- “Menopause”, “Postmenopause, “Postmenopausal women”, “Postmenopausal” and varieties of “Gut microbiome”.
- “Obesity”, “Obese”, “obese women” “overweight”, “Overweight women”, and varieties of “gut microbiome”.
2.2. Eligibility Criteria
2.3. Data Extraction
2.4. Quality Assessment and Data Synthesis
3. Outcome
3.1. Estrogen, Menopausal Status and Gut Microbiome
3.1.1. Literature Search
3.1.2. Quality and Risk of Bias of Selected Studies
3.1.3. Main Outcomes
Alpha Diversity
Firmicutes to Bacteroidetes Ratio
Family and Genus
Estrogen-Gut Axis
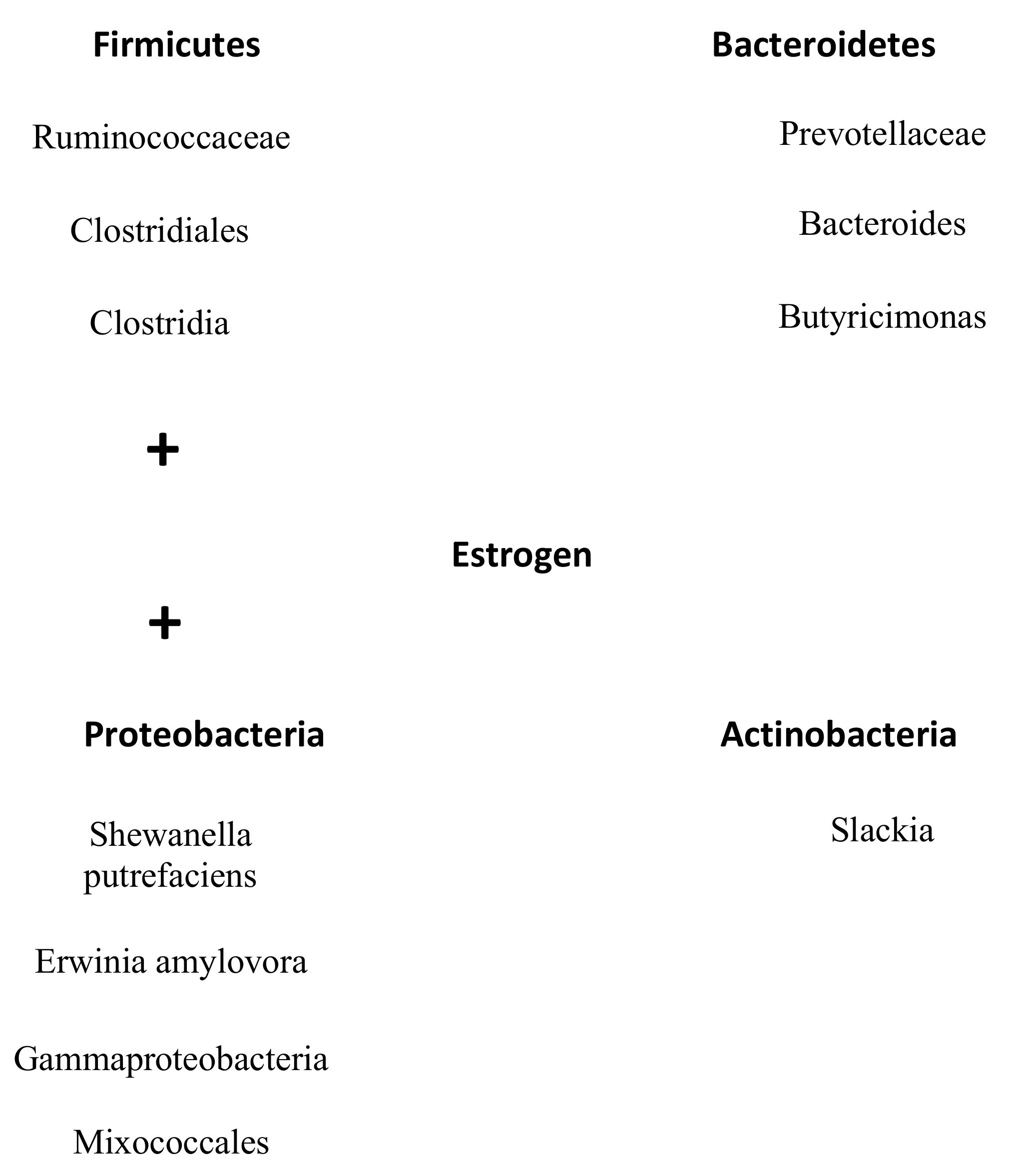
3.2. Obesity in Women and Gut Microbiome
3.2.1. Literature Search
3.2.2. Quality and Risk of Bias of Selected Studies
3.2.3. Main Outcomes
Alpha Diversity
Firmicutes to Bacteroidetes Ratio
Family and Genus
4. Discussion
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pischon, T.; Nimptsch, K. Obesity and Risk of Cancer: An Introductory Overview. Recent Results Cancer Res. 2016, 208, 1–15. [Google Scholar]
- Passarello, K.; Kurian, S.; Villanueva, V. Endometrial Cancer: An Overview of Pathophysiology, Management, and Care. Semin. Oncol. Nurs. 2019, 35, 157–165. [Google Scholar] [CrossRef]
- Stavropoulos, A.; Varras, M.; Philippou, A.; Vasilakaki, T.; Varra, V.K.; Varra, F.N.; Tsavari, A.; Lazaris, A.C.; Koutsilieris, M. Immunohistochemical expression of insulin-like growth factor-1Ec in primary endometrial carcinoma: Association with PTEN, p53 and survivin expression. Oncol. Lett. 2020, 20, 395. [Google Scholar] [CrossRef]
- Byrd, V.; Getz, T.; Padmanabhan, R.; Arora, H.; Eng, C. The microbiome in PTEN hamartoma tumor syndrome. Endocr. Relat. Cancer 2018, 25, 233–243. [Google Scholar] [CrossRef] [Green Version]
- Molina, N.M.; Sola-Leyva, A.; Saez-Lara, M.J.; Plaza-Diaz, J.; Tubić-Pavlović, A.; Romero, B.; Clavero, A.; Mozas-Moreno, J.; Fontes, J.; Altmäe, S. New Opportunities for Endometrial Health by Modifying Uterine Microbial Composition: Present or Future? Biomolecules 2020, 10, 593. [Google Scholar] [CrossRef] [Green Version]
- Heintz-Buschart, A.; Wilmes, P. Human Gut Microbiome: Function Matters. Trends Microbiol. 2018, 26, 563–574. [Google Scholar] [CrossRef]
- Brown, C.T.; Sharon, I.; Thomas, C.B.; Castelle, J.C.; Morowitz, M.J.; Banfield, J.F. Genome resolved analysis of a premature infant gut microbial community reveals a Varibaculum cambriense genome and a shift towards fermentation-based metabolism during the third week of life. Microbiome 2013, 1, 30. [Google Scholar] [CrossRef] [Green Version]
- Clemente, J.C.; Ursell, L.K.; Parfrey, L.W.; Knight, R. The impact of the gut microbiota on human health: An integrative view. Cell 2012, 148, 1258–1270. [Google Scholar] [CrossRef] [Green Version]
- Jackson, D.N.; Theiss, A.L. Gut bacteria signaling to mitochondria in intestinal inflammation and cancer. Gut Microbes 2020, 11, 285–304. [Google Scholar] [CrossRef] [Green Version]
- Rinninella, E.; Raoul, P.; Cintoni, M.; Franceschi, F.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. What is the Healthy Gut Microbiota Composition? A Changing Ecosystem across Age, Environment, Diet, and Diseases. Microorganisms 2019, 7, 14. [Google Scholar] [CrossRef] [Green Version]
- Tomasello, G.; Mazzola, M.; Leone, A.; Sinagra, E.; Zummo, G.; Farina, F.; Damiani, P.; Cappello, F.; Gerges Geagea, A.; Jurjus, A.; et al. Nutrition, oxidative stress and intestinal dysbiosis: Influence of diet on gut microbiota in inflammatory bowel diseases. Biomed. Pap. Med. Fac. Univ. Palacky Olomouc Czech Repub. 2016, 160, 461–466. [Google Scholar] [CrossRef] [Green Version]
- Weiss, G.A.; Hennet, T. Mechanisms and consequences of intestinal dysbiosis. Cell Mol. Life Sci. 2017, 74, 2959–2977. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Belizário, J.E.; Napolitano, M. Human microbiomes and their roles in dysbiosis, common diseases, and novel therapeutic approaches. Front. Microbiol. 2015, 6, 1050. [Google Scholar] [CrossRef] [Green Version]
- Cani, P.D. Human gut microbiome: Hopes, threats and promises. Gut 2018, 67, 1716–1725. [Google Scholar] [CrossRef] [PubMed]
- Cresci, G.A.; Bawden, E. Gut Microbiome: What We Do and Don’t Know. Nutr. Clin. Pract. 2015, 30, 734–746. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thursby, E.; Juge, N. Introduction to the human gut microbiota. Biochem. J. 2017, 474, 1823–1836. [Google Scholar] [CrossRef] [PubMed]
- Eckburg, P.B.; Bik, E.M.; Bernstein, C.N.; Purdom, E.; Dethlefsen, L.; Sargent, M.; Gill, S.R.; Nelson, K.E.; Relman, D.A. Diversity of the human intestinal microbial flora. Science 2005, 308, 1635–1638. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Segata, N.; Segata, N.; Haake, S.K.; Mannon, P.; Lemon, K.P.; Waldron, L.; Gevers, D.; Huttenhower, C.; Izard, J. Composition of the adult digestive tract bacterial microbiome based on seven mouth surfaces, tonsils, throat and stool samples. Genome Biol. 2012, 13, R42. [Google Scholar] [CrossRef] [Green Version]
- Zoetendal, E.G.; Rajilic-Stojanovic, M.; de Vos, W.M. High-throughput diversity and functionality analysis of the gastrointestinal tract microbiota. Gut 2008, 57, 1605–1615. [Google Scholar] [CrossRef]
- Pushpanathan, P.; Mathew, G.S.; Selvarajan, S.; Seshadri, K.G.; Srikanth, P. Gut microbiota and its mysteries. Indian J. Med. Microbiol. 2019, 37, 268–277. [Google Scholar] [CrossRef]
- Koliada, A.; Syzenko, G.; Moseiko, V.; Budovska, L.; Puchkov, K.; Perederiy, V.; Gavalko, Y.; Dorofeyev, A.; Romanenko, M.; Tkach, S.; et al. Association between body mass index and Firmicutes/Bacteroidetes ratio in an adult Ukrainian population. BMC Microbiol. 2017, 17, 120. [Google Scholar] [CrossRef] [Green Version]
- Baker, J.M.; Al-Nakkash, L.; Herbst-Kralovetz, M.M. Estrogen-gut microbiome axis: Physiological and clinical implications. Maturitas 2017, 103, 45–53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fernández, M.F.; Reina-Pérez, I.; Astorga, J.M.; Rodríguez-Carrillo, A.; Plaza-Díaz, J.; Fontana, L. Breast Cancer and Its Relationship with the Microbiota. Int. J. Environ. Res. Public Health 2018, 15, 1747. [Google Scholar] [CrossRef] [Green Version]
- Kwa, M.; Plottel, C.S.; Blaser, M.J.; Adams, S. The Intestinal Microbiome and Estrogen Receptor-Positive Female Breast Cancer. J. Natl. Cancer Inst. 2016, 108, 8. [Google Scholar]
- Laborda-Illanes, A.; Sanchez-Alcoholado, L.; Dominguez-Recio, M.E.; Jimenez-Rodriguez, B.; Lavado, R.; Comino-Méndez, I.; Alba, E.; Queipo-Ortuño, M.I. Breast and Gut Microbiota Action Mechanisms in Breast Cancer Pathogenesis and Treatment. Cancers 2020, 12, 2465. [Google Scholar] [CrossRef]
- Magne, F.; Gotteland, M.; Gauthier, L.; Zazueta, A.; Pesoa, S.; Navarrete, P.; Balamurugan, R. The Firmicutes/Bacteroidetes Ratio: A Relevant Marker of Gut Dysbiosis in Obese Patients? Nutrients 2020, 12, 1474. [Google Scholar] [CrossRef]
- Lu, W.; He, F.; Lin, Z.; Liu, S.; Tang, L.; Huang, Y.; Hu, Z. Dysbiosis of the endometrial microbiota and its association with inflammatory cytokines in endometrial cancer. Int. J. Cancer 2021, 148, 1708–1716. [Google Scholar] [CrossRef]
- Walther-António, M.R.; Chen, J.; Multinu, F.; Hokenstad, A.; Distad, T.J.; Cheek, E.H.; Keeney, G.L.; Creedon, D.J.; Nelson, H.; Mariani, A.; et al. Potential contribution of the uterine microbiome in the development of endometrial cancer. Genome Med. 2016, 8, 122. [Google Scholar] [CrossRef] [Green Version]
- Łaniewski, P.; ZIlhan, E.; Herbst-Kralovetz, M.M. The microbiome and gynaecological cancer development, prevention and therapy. Nat. Rev. Urol. 2020, 17, 232–250. [Google Scholar] [CrossRef]
- GA Wells, B.S.; Connel, D.; Peterson, J.; Welch, V.; Lolos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses, in Scale. 2010, pp. 2–4. Available online: http://www3.med.unipmn.it/dispense_ebm/2009-2010/Corso%20Perfezionamento%20EBM_Faggiano/NOS_oxford.pdf (accessed on 29 June 2021).
- Downes, M.J.; Brennan, M.L.; Williams, H.C.; Dean, R.S. Development of a critical appraisal tool to assess the quality of cross-sectional studies (AXIS). BMJ Open 2016, 6, e011458. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hooijmans, C.R.; Rovers, M.M.; de Vries, R.B.; Leenaars, M.; Ritskes-Hoitinga, M.; Langendam, M.W. SYRCLE’s risk of bias tool for animal studies. BMC Med. Res. Methodol. 2014, 14, 43. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.; Zolnik, C.P.; Qiu, Y.; Usyk, M.; Wang, T.; Strickler, H.D.; Isasi, C.R.; Kaplan, R.C.; Kurland, I.J.; Qi, Q.; et al. Comparison of Fecal Collection Methods for Microbiome and Metabolomics Studies. Front. Cell Infect. Microbiol. 2018, 8, 301. [Google Scholar] [CrossRef]
- Fuhrman, B.J.; Feigelson, H.S.; Flores, R.; Gail, M.H.; Xu, X.; Ravel, J.; Goedert, J.J. Associations of the fecal microbiome with urinary estrogens and estrogen metabolites in postmenopausal women. J. Clin. Endocrinol. Metab. 2014, 99, 4632–4640. [Google Scholar] [CrossRef]
- Shin, J.H.; Park, Y.H.; Sim, M.; Kim, S.A.; Joung, H.; Shin, D.M. Serum level of sex steroid hormone is associated with diversity and profiles of human gut microbiome. Res. Microbiol. 2019, 170, 192–201. [Google Scholar] [CrossRef]
- Zhang, Z.; Chen, Y.; Xiang, L.; Wang, Z.; Xiao, G.G.; Hu, J. Effect of Curcumin on the Diversity of Gut Microbiota in Ovariectomized Rats. Nutrients 2017, 9, 1146. [Google Scholar] [CrossRef] [Green Version]
- Zhao, H.; Chen, J.; Li, X.; Sun, Q.; Qin, P.; Wang, Q. Compositional and functional features of the female premenopausal and postmenopausal gut microbiota. FEBS Lett. 2019, 593, 2655–2664. [Google Scholar] [CrossRef]
- Zhu, J.; Liao, M.; Yao, Z.; Liang, W.; Li, Q.; Liu, J.; Yang, H.; Ji, Y.; Wei, W.; Tan, A.; et al. Breast cancer in postmenopausal women is associated with an altered gut metagenome. Microbiome 2018, 6, 136. [Google Scholar] [CrossRef] [Green Version]
- Santos-Marcos, J.A.; Rangel-Zuñiga, O.A.; Jimenez-Lucena, R.; Quintana-Navarro, G.M.; Garcia-Carpintero, S.; Malagon, M.M.; Landa, B.B.; Tena-Sempere, M.; Perez-Martinez, P.; Lopez-Miranda, J.; et al. Influence of gender and menopausal status on gut microbiota. Maturitas 2018, 116, 43–53. [Google Scholar] [CrossRef]
- Choi, S.; Hwang, Y.J.; Shin, M.J.; Yi, H. Difference in the Gut Microbiome between Ovariectomy-Induced Obesity and Diet-Induced Obesity. J. Microbiol. Biotechnol. 2017, 27, 2228–2236. [Google Scholar] [CrossRef]
- Portune, K.J.; Benítez-Páez, A.; Del Pulgar, E.M.; Cerrudo, V.; Sanz, Y. Gut microbiota, diet, and obesity-related disorders-The good, the bad, and the future challenges. Mol. Nutr. Food Res. 2017, 61, 1600252. [Google Scholar] [CrossRef] [Green Version]
- Flores, R.; Shi, J.; Fuhrman, B.; Xu, X.; Veenstra, T.D.; Gail, M.H.; Gajer, P.; Ravel, J.; Goedert, J.J. Fecal microbial determinants of fecal and systemic estrogens and estrogen metabolites: A cross-sectional study. J. Transl. Med. 2012, 10, 253. [Google Scholar] [CrossRef] [Green Version]
- Alpuim Costa, D.; Nobre, J.G.; Batista, M.V.; Ribeiro, C.; Calle, C.; Cortes, A.; Marhold, M.; Negreiros, I.; Borralho, P.; Brito, M.; et al. Human Microbiota and Breast Cancer-Is There Any Relevant Link?-A Literature Review and New Horizons Toward Personalised Medicine. Front. Microbiol. 2021, 12, 584332. [Google Scholar] [CrossRef] [PubMed]
- Menni, C.; Jackson, M.A.; Pallister, T.; Steves, C.J.; Spector, T.D.; Valdes, A.M. Gut microbiome diversity and high-fibre intake are related to lower long-term weight gain. Int. J. Obes. 2017, 41, 1099–1105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Min, Y.; Ma, X.; Sankaran, K.; Ru, Y.; Chen, L.; Baiocchi, M.; Zhu, S. Sex-specific association between gut microbiome and fat distribution. Nat. Commun. 2019, 10, 2408. [Google Scholar] [CrossRef]
- Chávez-Carbajal, A.; Nirmalkar, K.; Pérez-Lizaur, A.; Hernández-Quiroz, F.; Ramírez-Del-Alto, S.; García-Mena, J.; Hernández-Guerrero, C. Gut Microbiota and Predicted Metabolic Pathways in a Sample of Mexican Women Affected by Obesity and Obesity Plus Metabolic Syndrome. Int. J. Mol. Sci. 2019, 20, 438. [Google Scholar] [CrossRef] [Green Version]
- Miranda, V.P.N.; Dos Santos Amorim, P.R.; Bastos, R.R.; de Faria, E.R.; de Castro Moreira, M.E.; do Carmo Castro Franceschini, S.; do Carmo Gouveia Peluzio, M.; de Luces Fortes Ferreira, C.L.; Priore, S.E. Abundance of Gut Microbiota, Concentration of Short-Chain Fatty Acids, and Inflammatory Markers Associated with Elevated Body Fat, Overweight, and Obesity in Female Adolescents. Mediat. Inflamm. 2019, 2019, 7346863. [Google Scholar] [CrossRef] [Green Version]
- Pekkala, S.; Munukka, E.; Kong, L.; Pöllänen, E.; Autio, R.; Roos, C.; Wiklund, P.; Fischer-Posovszky, P.; Wabitsch, M.; Alen, M.; et al. Toll-like receptor 5 in obesity: The role of gut microbiota and adipose tissue inflammation. Obesity 2015, 23, 581–590. [Google Scholar] [CrossRef]
- Haro, C.; Rangel-Zúñiga, O.A.; Alcalá-Díaz, J.F.; Gómez-Delgado, F.; Pérez-Martínez, P.; Delgado-Lista, J.; Quintana-Navarro, G.M.; Landa, B.B.; Navas-Cortés, J.A.; Tena-Sempere, M.; et al. Intestinal Microbiota Is Influenced by Gender and Body Mass Index. PLoS ONE 2016, 11, e0154090. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ott, B.; Skurk, T.; Hastreiter, L.; Lagkouvardos, I.; Fischer, S.; Buttner, J.; Kellerer, T.; Clavel, T.; Rychlik, M.; Haller, D.; et al. Effect of caloric restriction on gut permeability, inflammation markers, and fecal microbiota in obese women. Sci. Rep. 2017, 7, 11955. [Google Scholar] [CrossRef]
- Tamanai-Shacoori, Z.; Smida, I.; Bousarghin, L.; Loreal, O.; Meuric, V.; Fong, S.B.; Bonnaure-Mallet, M.; Jolivet-Gougeon, A. Roseburia spp.: A marker of health? Future Microbiol. 2017, 12, 157–170. [Google Scholar] [CrossRef]
- Amabebe, E.; Anumba, D.O.C. Female Gut and Genital Tract Microbiota-Induced Crosstalk and Differential Effects of Short-Chain Fatty Acids on Immune Sequelae. Front. Immunol. 2020, 11, 2184. [Google Scholar] [CrossRef]
- Adlercreutz, H.; Pulkkinen, M.O.; Hämäläinen, E.K.; Korpela, J.T. Studies on the role of intestinal bacteria in metabolism of synthetic and natural steroid hormones. J. Steroid. Biochem. 1984, 20, 217–229. [Google Scholar] [CrossRef]
- Guadamuro, L.; Dohrmann, A.B.; Tebbe, C.C.; Mayo, B.; Delgado, S. Bacterial communities and metabolic activity of faecal cultures from equol producer and non-producer menopausal women under treatment with soy isoflavones. BMC Microbiol. 2017, 17, 93. [Google Scholar] [CrossRef] [Green Version]
- Iino, C.; Shimoyama, T.; Iino, K.; Yokoyama, Y.; Chinda, D.; Sakuraba, H.; Fukuda, S.; Nakaji, S. Daidzein Intake Is Associated with Equol Producing Status through an Increase in the Intestinal Bacteria Responsible for Equol Production. Nutrients 2019, 11, 433. [Google Scholar] [CrossRef] [Green Version]
- Brown, L.M.; Clegg, D.J. Central effects of estradiol in the regulation of food intake, body weight, and adiposity. J. Steroid. Biochem. Mol. Biol. 2010, 122, 65–73. [Google Scholar] [CrossRef] [Green Version]
- Song, C.H.; Kim, N.; Nam, R.H.; Choi, S.I.; Lee, H.N.; Surh, Y.J. 17β-Estradiol supplementation changes gut microbiota diversity in intact and colorectal cancer-induced ICR male mice. Sci. Rep. 2020, 10, 12283. [Google Scholar] [CrossRef]
- Sorbara, M.T.; Littmann, E.R.; Fontana, E.; Moody, T.U.; Kohout, C.E.; Gjonbalaj, M.; Eaton, V.; Seok, R.; Leiner, I.M.; Pamer, E.G. Functional and Genomic Variation between Human-Derived Isolates of Lachnospiraceae Reveals Inter- and Intra-Species Diversity. Cell Host. Microbe 2020, 28, 134–146. [Google Scholar] [CrossRef]
- Meisinger, C.; Ezzati, M.; Di Cesare, M. Trends in adult body-mass index in 200 countries from 1975 to 2014: A pooled analysis of 1698 population-based measurement studies with 19.2 million participants. Lancet 2016, 387, 1377–1396. [Google Scholar]
- Onstad, M.A.; Schmandt, R.E.; Lu, K.H. Addressing the Role of Obesity in Endometrial Cancer Risk, Prevention, and Treatment. J. Clin. Oncol. 2016, 34, 4225–4230. [Google Scholar] [CrossRef]
- Santos-Marcos, J.A.; Haro, C.; Vega-Rojas, A.; Alcala-Diaz, J.F.; Molina-Abril, H.; Leon-Acuña, A.; Lopez-Moreno, J.; Landa, B.B.; Tena-Sempere, M.; Perez-Martinez, P.; et al. Sex Differences in the Gut Microbiota as Potential Determinants of Gender Predisposition to Disease. Mol. Nutr. Food Res. 2019, 63, e1800870. [Google Scholar] [CrossRef]
- Calle, E.E.; Rodriguez, C.; Walker-Thurmond, K.; Thun, M.J. Overweight, obesity, and mortality from cancer in a prospectively studied cohort of U.S. adults. N. Engl. J. Med. 2003, 348, 1625–1638. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meldrum, D.R.; Morris, M.A.; Gambone, J.C. Obesity pandemic: Causes, consequences, and solutions-but do we have the will? Fertil. Steril. 2017, 107, 833–839. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Torres-Fuentes, C.; Schellekens, H.; Dinan, T.G.; Cryan, J.F. The microbiota-gut-brain axis in obesity. Lancet. Gastroenterol. Hepatol. 2017, 2, 747–756. [Google Scholar] [CrossRef]
- Turnbaugh, P.J.; Hamady, M.; Yatsunenko, T.; Cantarel, B.L.; Duncan, A.; Ley, R.E.; Sogin, M.L.; Jones, W.J.; Roe, B.A.; Affourtit, J.P.; et al. A core gut microbiome in obese and lean twins. Nature 2009, 457, 480–484. [Google Scholar] [CrossRef] [Green Version]
- Le Chatelier, E.; Nielsen, T.; Qin, J.; Prifti, E.; Hildebrand, F.; Falony, G.; Almeida, M.; Arumugam, M.; Batto, J.M.; Kennedy, S.; et al. Richness of human gut microbiome correlates with metabolic markers. Nature 2013, 500, 541–546. [Google Scholar] [CrossRef]
- Hills, R.D., Jr.; Pontefract, B.A.; Mishcon, H.R.; Black, C.A.; Sutton, S.C.; Theberge, C.R. Gut Microbiome: Profound Implications for Diet and Disease. Nutrients 2019, 11, 1613. [Google Scholar] [CrossRef] [Green Version]
- Federico, A.; Dallio, M.; Tolone, S.; Gravina, A.G.; Patrone, V.; Romano, M.; Tuccillo, C.; Mozzillo, A.L.; Amoroso, V.; Misso, G.; et al. Gastrointestinal Hormones, Intestinal Microbiota and Metabolic Homeostasis in Obese Patients: Effect of Bariatric Surgery. In Vivo 2016, 30, 321–330. [Google Scholar]
- Aron-Wisnewsky, J.; Prifti, E.; Belda, E.; Ichou, F.; Kayser, B.D.; Dao, M.C.; Verger, E.O.; Hedjazi, L.; Bouillot, J.L.; Chevallier, J.M.; et al. Major microbiota dysbiosis in severe obesity: Fate after bariatric surgery. Gut 2019, 68, 70–82. [Google Scholar] [CrossRef] [PubMed]
- Sims, T.T.; Colbert, L.E.; Zheng, J.; Delgado Medrano, A.Y.; Hoffman, K.L.; Ramondetta, L.; Jazaeri, A.; Jhingran, A.; Schmeler, K.M.; Daniel, C.R.; et al. Gut microbial diversity and genus-level differences identified in cervical cancer patients versus healthy controls. Gynecol. Oncol. 2019, 155, 237–244. [Google Scholar] [CrossRef]
- Tong, J.; Zhang, X.; Fan, Y.; Chen, L.; Ma, X.; Yu, H.; Li, J.; Guan, X.; Zhao, P.; Yang, J. Changes of Intestinal Microbiota in Ovarian Cancer Patients Treated with Surgery and Chemotherapy. Cancer Manag. Res. 2020, 12, 8125–8135. [Google Scholar] [CrossRef]
- Avilés-Jiménez, F.; Yu, G.; Torres-Poveda, K.; Madrid-Marina, V.; Torres, J. On the Search to Elucidate the Role of Microbiota in the Genesis of Cancer: The Cases of Gastrointestinal and Cervical Cancer. Arch. Med. Res. 2017, 48, 754–765. [Google Scholar] [CrossRef]
- Terrisse, S.; Derosa, L.; Iebba, V.; Ghiringhelli, F.; Vaz-Luis, I.; Kroemer, G.; Fidelle, M.; Christodoulidis, S.; Segata, N.; Thomas, A.M.; et al. Intestinal microbiota influences clinical outcome and side effects of early breast cancer treatment. Cell Death Diff. 2021, 16, 1–19. [Google Scholar]
- Mantovani, A.; Allavena, P.; Sica, A.; Balkwill, F. Cancer-related inflammation. Nature 2008, 454, 436–444. [Google Scholar] [CrossRef]
- Rao, V.P.; Poutahidis, T.; Ge, Z.; Nambiar, P.R.; Boussahmain, C.; Wang, Y.Y.; Horwitz, B.H.; Fox, J.G.; Erdman, S.E. Innate immune inflammatory response against enteric bacteria Helicobacter hepaticus induces mammary adenocarcinoma in mice. Cancer Res. 2006, 66, 7395–7400. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caesar, R.; Tremaroli, V.; Kovatcheva-Datchary, P.; Cani, P.D.; Bäckhed, F. Crosstalk between Gut Microbiota and Dietary Lipids Aggravates WAT Inflammation through TLR Signaling. Cell Metab. 2015, 22, 658–668. [Google Scholar] [CrossRef] [Green Version]
- Wallace, A.E.; Gibson, D.A.; Saunders, P.T.; Jabbour, H.N. Inflammatory events in endometrial adenocarcinoma. J. Endocrinol. 2010, 206, 141–157. [Google Scholar] [CrossRef] [Green Version]
- Fortuny, J.; Sima, C.; Bayuga, S.; Wilcox, H.; Pulick, K.; Faulkner, S.; Zauber, A.G.; Olson, S.H. Risk of endometrial cancer in relation to medical conditions and medication use. Cancer Epidemiol. Biomark. Prev. 2009, 18, 1448–1456. [Google Scholar] [CrossRef] [Green Version]
- Viswanathan, A.N.; Feskanich, D.; Schernhammer, E.S.; Hankinson, S.E. Aspirin, NSAID, and acetaminophen use and the risk of endometrial cancer. Cancer Res. 2008, 68, 2507–2513. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Webb, P.M.; Na, R.; Weiderpass, E.; Adami, H.O.; Anderson, K.E.; Bertrand, K.A.; Botteri, E.; Brasky, T.M.; Brinton, L.A.; Chen, C.; et al. Use of aspirin, other nonsteroidal anti-inflammatory drugs and acetaminophen and risk of endometrial cancer: The Epidemiology of Endometrial Cancer Consortium. Ann. Oncol. 2019, 30, 310–316. [Google Scholar] [CrossRef] [Green Version]
- McAlpine, J.; Leon-Castillo, A.; Bosse, T. The rise of a novel classification system for endometrial carcinoma; integration of molecular subclasses. J. Pathol. 2018, 244, 538–549. [Google Scholar] [CrossRef]
- Raffone, A.; Travaglino, A.; Raimondo, D.; Neola, D.; Renzulli, F.; Santoro, A.; Insabato, L.; Casadio, P.; Zannoni, G.F.; Zullo, F.; et al. Prognostic value of myometrial invasion and TCGA groups of endometrial carcinoma. Gynecol. Oncol. 2021, in press. [Google Scholar]


| Study | Sample Size | Patient Characteristics | Gut Microbiota Analysis | Main Findings |
|---|---|---|---|---|
| Zhao et al. (2019) | n = 24 Premenopausal women n = 24 Postmenopausal women | Premenopausal: Age (yrs) 52.6 ± 6 BMI (kg/m2) 23.1 ± 4.5 LDL (mM) 3.0 ± 0.8 Postmenopausal Age (yrs) 53.9 ± 3.8 BMI (kg/m2) 23.0 ± 3.2 LDL (mM) 2.89 ± 0.83 No statistical differences. | Single-end metagenomic sequencing on BGISEQ-500 platform. Relative abundance calculation by Metaphlann2 (used by the NIH Human Microbiome Project part 2). Alpha-diversity → Shannon-index. | Alpha diversity (Shannon index): Premenopausal 1.8 Postmenopausal 1.3 (p 0.000005) Phyla: Firmicutes:
Postmenopausal state:
When ↑ Eubacterium rectale (stimulated by isoflavones → ability prevention dysbiosis) |
| Shin et al. (2019) | n = 9 high estrogen women (premenopausal) n = 8 medium estrogen women (premenopausal) n = 9 low estrogen women (postmenopausal) | High estrogen (>60 pg/mL) Age (yrs) 39.3 ± 3.2 BMI 28.9 ± 0.2 Medium estrogen: (5–60 pg/mL) Age (yrs) 44 ± 2 BMI (kg/m2) 26.9 ± 0.9 Low estrogen: (<5 pg/mL). Age (yrs) 54.9 ± 1.0 BMI (kg/m2) 24.9 ± 0.5 BMI not statistically different | 16S V6 rRNA amplicon sequencing using QIIME. Taxonomy assigned against the Greengenes 16S rRNA gene database. Alfa diversity → Chao1 richness, Simpson evenness, Good’s coverage and Shannon diversity. | 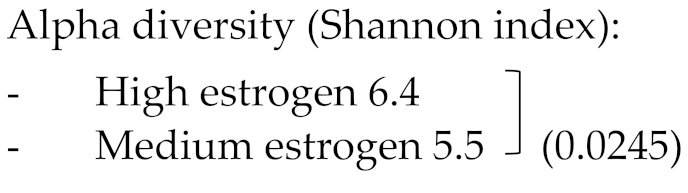
Firmicutes:
Dominant: Bacteroidaceae (61.2%), Prevotellaceae (28.6%), Rikenellaceae (3.6%) Postmenopausal vs. premenopausal
Dominant: Ruminococcaceae (42.3%), Lachnospiraceae (39.9%), Veillonellaceae (11.7%). Postmenopausal vs. premenopausal.
Butyricimonas (r = −0.4; p 0.046) |
| Zhu et al. (2018) | n = 25 premenopausal women n = 46 postmenopausal women (breast cancer patients excluded) | Premenopausal Age (yrs) 35.5 ± 6.0 BMI (kg/m2) 23.0 ± 2.0 Postmenopausal Age (yrs) 56.9 ± 6.4 BMI (kg/m2) 24.0 ± 2.5 BMI not statistically different | Illumina DNA sequencing. Taxonomy calculated against the integrated reference catalog of the human gut microbiome (IGC) by bowtie2 Alfa-diversity → Shannon index, Chao index |
Alpha diversity (Shannon index) Premenopausal 3.1 Postmenopausal 3.2 (p-value not calculated) Alpha diversity (Chao1 index) Premenopausal −430 Postmenopausal −415 (p-value not calculated) Alpha diversity (OTU) Premenopausal −400 Postmenopausal −390 (p-value not calculated) Link genera and serum estradiol levels.
|
| Santos-Marcos et al. (2018) | n = 17 premenopausal women n = 20 postmenopausal women | Premenopausal Age (yrs) 46.1 ± 0.8 BMI (kg/m2) 26.3 ± 1.5 LDL (mg/dL) 119 ± 7 Postmenopausal Age (yrs) 55.6 ± 0.6 BMI (kg/m2) 28.9 ± 1.3 LDL (mg/dL) 137 ± 7 | Sequencing the V1–V2 microbial 16S rRNA gene on the Illumina MiSeq Taxonomy assigned against Greengenes v13-8 database | Phyla: Firmicutes
Premenopausal
Positively correlated:
TNF-alfa (pg/mL) Premenopausal 0.26 (±0.05) Postmenopausal 0.38 (±0.06; NS) IL-6 (pg/mL) Premenopausal 1.25 (±0.15) Postmenopausal 1.75 (±0.25; p 0.036) MCP-1 (pg/mL) Premenopausal 72 (±4) Postmenopausal 94 (±7; p 0.045) |
| Choi et al. (2017) Animal study | n = 3 SHAM mice n = 5 ovariectomized mice (OVX) | SHAM Weight (g) 29.96 ± 2.13 LDL (mg/dL) 30.9 ± 5.1 OVX Weight (g) 41.44 ± 1.52 LDL (mg/dL) 45.1 ± 9.1 Weight significantly different | V3-V4 16S rRNA amplification following the 16S Metagenomic Sequencing Library Preparation guide by Illumina. Gene-enrichment and functional annotation analysis performed using gene ontology and KEGG pathway analysis. | Alpha diversity (Shannon index)
Firmicutes
SHAM
|
| Zhang et al. (2017) Animal study | n = 6 SHAM rats n = 12 OVX
| All groups: Virgin Wistar rats Age (yrs) 0.5 Weight: 310 ± 20.0 g (OVX rats significantly higher weight) | The estradiol concentration in the serum detected through electrochemiluminescence immunoassay (ECLIA) |
Alpha diversity (Shannon index)
Incertae_Sedis
|
| Fuhrman et al. (2014) | n = 6 postmenopausal women (acting as their own controls) | Postmenopausal Age (yrs) 60.2 ± 3.2 BMI (kg/m2) 27.3 ± 5.4 | Pyrosequencing V1–V2 16S rRNA amplicons, QIIME: Ribosomal Data Project Bayesian classifier. | Alpha diversity (Shannon index)
Firmicutes
Positive correlation ratio of estrogen metabolites to parent estrogen:
Postmenopausal women 28.1 (±17.8) Parent estrogen (estrone and estradiol 32 % of total EM’s) 2-, 4- and 16-hydroxilated metabolites represented 29%, 3% and 35%) |
| Flores et al. (2012) | n = 19 premenopausal women n = 7 postmenopausal women n = 22 age matched men (55 yrs and older) | Average BMI 26 | In feces, β-glucuronidase and β-glucosidase activities were determined by real-time kinetics, and microbiome diversity and taxonomy were estimated by pyrosequencing 16S rRNA amplicons. | Urinary estrogen (pM/mg creatinine): men 82.6 premenopausal women 68.7 postmenopausal women 155.1 Levels non-ovarian estrogens Premenopausal
Postmenopausal
Postmenopausal
|
| (a) Obesity in Women and Gut Microbiome | ||||
|---|---|---|---|---|
| Study | Sample Size | Patient Characteristics | Gut Microbiota Analysis | Main Findings |
| Menni et al. (2016) | n = 544 women with weight loss: BMI from 25.4 to 24.4 (group 1) n = 544 women with little weight gain: BMI from 24 to 25.2 (group 2) n = 544 women with heavy weight gain BMI from 25.4 to 28.8 (group 3) | Group 1 Age (yrs) 49.91 ± 9.49 Group 2 Age (yrs) 50.11 ± 5.54 Group 3 Age (yrs) 49.25 ± 8.48 All groups 15% smokers, further no exclusions. | V4 region of the 16S ribosomal RNA gene was amplified and sequenced on Illumina. De novo OTU clustering was carried across all reads using Sumaclust within QIIME 1.9.0. Alpha diversities → Shannon index, OTU counts. | Alpha diversity (Shannon index): Group 1 (weight loss) 5.21 Group 2 (weight gain) 5.19 Group 3 (heavy weight gain) 5.07 (p < 0.05) Alpha diversity (OTU): Group 1 346.3 Group 2 348 Group 3 331.8 (p < 0.05) Family Bacteriodes
|
| Chavez-Carbajal et al. (2019) | n = 25 control women n = 17 obese women n = 25 obese women with metabolic syndrome | Controls Age (yrs) 23.3 ± 3.1 BMI (kg/m2) 21.4 ± 1.9 Obesity Age (yrs) 28.8 ± 8.4 BMI (kg/m2) 34.8 ± 6.1 Obesity + metabolic syndrome (ms) Age (yrs) 40.5 ±10.3 BMI (kg/m2) 35.8 ± 5.1 Only women to avoid gender bias Controls significant different in age and bmi from other 2 groups | V3 region of the 16S rDNA Amplicon PCR amplification using PCR GeneAmp System 2700 Thermal Cycler. Determine with an open reference the OTUs and using a 97% similarity using QIIME pipeline (v1.9.0) and Geengenes database v13.8. Alpha diversity → Observed Species, Chao1, Shannon, Simpson. | Alpha diversity (Shannon index) Controls 4.9 Obesity 5.23 Obesity + MS 5.15 Dominant phyla in all groups: Firmicutes, Bacteroidetes, Proteobacteria, and Actinobacteria Phyla Frimicutes
Obesity and obesity + MS
Faecalibacterium (phyla firmicutes) Controls 0.55%
Controls 0.89%
Controls 0.99%
Controls 2.18%
Controls 1.74%
|
| Miranda er al. (2017) Observational study | n = 31 controls n = 32 normal BMI but high body fat percentage. n = 33 obesity | Controls Age (yrs) 16.3 ± 0.8 Gynoid fat (%) 34.5 (30.6–36.7) High body fat Age (yrs) 16.5 ± 0.9 Gynoid fat (%) 39.7 (37.9–46.9) Obesity Age (yrs) 16.2 ±1.3 Gynoid fat (%) 48.0 (45.5–54.1) | RT-qPCR to analyze microbiota CFX96 Touch™ detection system (Bio-Rad, Hercules, CA, USA) Alfa diversity → Shannon index |  |
| Pekkala et al. (2015) | n = 4 women with high TLR gene expression (BMI 31) n = 4 women with low TLR gene expression (BMI 28) | High TLR gene expression Age (yrs) 35.5 ± 6.0 BMI (kg/m2) 31 ± 2.0 Low TLR gene expression Age (yrs) 56.9 ± 6.4 BMI (kg/m2) 28 ± 2.5 BMI significantly higher in High TLR group. | Real-time PCR analysis was performed using in-house designed primers, iQ SYBR Supermix and CFX96 TM Real-time PCR Detection System (Bio-Rad Laboratories) Real-time PCR analysis was performed using in-house designed primers, iQ SYBR Supermix and CFX96 TM Real-time PCR Detection System (Bio-Rad Laboratories) Real-time PCR analysis was performed using in-house designed primers, iQ SYBR Supermix and CFX96 TM Real-time PCR Detection System (Bio-Rad Laboratories) RNA extraction and rt-PCR analysis using in-house designed primers. |
Alpha diversity High TLR group: significant dysbiosis. Phyla Firmicutes to Bacteroidetes ratio
|
| Ott et al. (2018) | n = 20 women (own controls) n = 20 after diet n = 20 14 days after diet | Women Age (yrs) 46.8 ± 11.5 Before diet BMI (kg/m2) 34.9 ± 3.8 After diet BMI (kg/m2) 32.5 ± 3.5 14 dys after diet BMI (kg/m2) 32.6 ± 3.8 | 16 S rRNA gene amplicons were sequenced in paired-end modus (PE275) using a MiSeq system (Illumina) | Alpha diversity No differences Phyla Protobacteria
|
| Choi et al. (2017) Animal study | n = 3 SHAM mice n = 3 SHAM-HF n = 5 ovariectomized mice (OVX) n = 5 OVX-HF | SHAM Weight (g) 29.96 ± 2.13 LDL (mg/dL) 30.9 ± 5.1 SHAM-HF Weight (g) 53.13 ± 3.88 LDL (mg/dL) 78 ± 4.4 OVX Weight (g) 41.44 ± 1.52 LDL (mg/dL) 45.1 ± 9.1 OVX-HF Weight (g) 57.54 ± 3.84 LDL (mg/dL) 95.7 ± 12.3 Weight significantly different | V3-V4 16S rRNA amplification following the 16S Metagenomic Sequencing Library Preparation guide by Illumina. Gene-enrichment and functional annotation analysis performed using gene ontology and KEGG pathway analysis. | Alpha diversity (Shannon index)
Firmicutes
SHAM
Akkermansia muciniphila related to
|
| (b) Obesity and Gut Microbiome: Sex Differences | ||||
| Study | Sample Size | Patient Characteristics | Gut Microbiota Analysis | Main Findings |
| Haro et al. (2016) | n = 39 men n = 13 men < BMI 30 n = 13 BMI 30–33 n = 13 men BMI > 33 n = 36 women n = 13 BMI < 30 n = 10 BMI 30–33 n = 23 BMI > 33 | Men BMI < 30 Age (yrs) 63.2 ± 2.0 BMI (kg/m2) 27.6 ± 0.6 LDL (mg/dL) 76.6 ± 4.2 BMI 30–33 Age (yrs) 58.9 ± 2.4 BMI (kg/m2) 31.4 ± 0.3 LDL (mg/dL) 95.3 ± 6.0 BMI > 33 Age (yrs) 61.3 ± 2.2 BMI (kg/m2) 35.3 ± 0.7 LDL (mg/dL) 87.8 ± 2.1 Women BMI < 30 Age (yrs) 60.1 ± 2.6 BMI (kg/m2) 27.0 ± 0.8 LDL (mg/dL) 94.2 ± 9.4 BMI 30–33 Age (yrs) 62.4 ±2.3 BMI (kg/m2) 31.4 ± 0.3 LDL (mg/dL) 87.1 ± 7.6 BMI > 33 Age (yrs) 58.9 ± 2.3 BMI (kg/m2) 36.7 ± 1.4 LDL (mg/dL) 80.4 ± 4.4 | Sequencing V4 16S microbial rRNA on the Illumina MiSeq. Taxonomy assigned to OTUs against the Greengenes v13-8 preclustered at 97% identity. Alpha diversities → observed OTU counts, Shannon, Simpson. | Alpha diversity similar men and women and comparing BMI Phyla Firmicutes to Bacteroidetes ratio
Women BMI > 33
Women BMI > 33
|
| Min et al. (2019) | n = 116 women n = 96 men | Women Age (yrs) 50.7 ± 14.1 BMI (kg/m2) 23.0 ± 3.0 Gynoid fat 15.9 ± 3.0 Android fat 12.5 ± 1.2 LDL (mmol/L) 2.7 ± 0.7 Men Age (yrs) 50.7 ± 14.5 BMI (kg/m2) 23.6 ± 3.0 Gynoid fat 17.7 ± 3.0 (p < 0.005) Android fat 9.9 ± 1.4 (p < 0.005) LDL (mmol/L) 2.8 ± 0.7 | 16S rRNA V4 region sequencing The denoised sequences are mapped to the GreenGenes reference database43. Taxonomy is assigned at 97% identity. Alfa diversity → Shannon index | Alpha diversity potential negative association between gynoid fat ratio and microbiome abundance in both sexes. In women compared to men different taxa responsible for relation between fat distribution and diversity. Gynoid fat ratio positive correlation Women:
|
| Case-Control | NOS Scale | |||
|---|---|---|---|---|
| Selection | Comparibilty | Exposure | ||
| Byrd et al. | ** | * | *** | |
| Zhao et al. | ** | ** | *** | |
| Shin et al. | * | ** | *** | |
| Zhu et al. | ** | ** | ** | |
| Santos-Marcos et al. | **** | ** | *** | |
| Menni et al. | *** | ** | *** | |
| Chavez et al. | ** | ** | *** | |
| Miranda et al. | *** | ** | ** | |
| Pekkala et al. | **** | ** | ** | |
| Haro et al. | *** | ** | *** | |
| Min et al. | *** | * | *** | |
| Cohort | NOS Scale | |||
| Selection | Comparability | Outcome | ||
| Ott et al. | ** | ** | *** | |
| Cross-Sectional | AXIS | |||
| Intro | Methods | Results | Discussion/Ethics | |
| Fuhrman et al. | 1/1 | 7/11 | 4/5 | 4/4 |
| Flores et al. | 1/1 | 6/11 | 4/5 | 4/4 |
| Animal | SYRCLE’s | Bias Tool | ||
| Selection/Performance | Detection | Attrition | Reporting | |
| Choi et al. | 2/5 | 0/2 | 1/1 | 1/1 |
| Zhang et al. | 0/5 | 0/2 | 1/1 | 1/1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schreurs, M.P.H.; de Vos van Steenwijk, P.J.; Romano, A.; Dieleman, S.; Werner, H.M.J. How the Gut Microbiome Links to Menopause and Obesity, with Possible Implications for Endometrial Cancer Development. J. Clin. Med. 2021, 10, 2916. https://doi.org/10.3390/jcm10132916
Schreurs MPH, de Vos van Steenwijk PJ, Romano A, Dieleman S, Werner HMJ. How the Gut Microbiome Links to Menopause and Obesity, with Possible Implications for Endometrial Cancer Development. Journal of Clinical Medicine. 2021; 10(13):2916. https://doi.org/10.3390/jcm10132916
Chicago/Turabian StyleSchreurs, Malou P. H., Peggy J. de Vos van Steenwijk, Andrea Romano, Sabine Dieleman, and Henrica M. J. Werner. 2021. "How the Gut Microbiome Links to Menopause and Obesity, with Possible Implications for Endometrial Cancer Development" Journal of Clinical Medicine 10, no. 13: 2916. https://doi.org/10.3390/jcm10132916
APA StyleSchreurs, M. P. H., de Vos van Steenwijk, P. J., Romano, A., Dieleman, S., & Werner, H. M. J. (2021). How the Gut Microbiome Links to Menopause and Obesity, with Possible Implications for Endometrial Cancer Development. Journal of Clinical Medicine, 10(13), 2916. https://doi.org/10.3390/jcm10132916







