Abstract
The gut microbiota is well known to exert multiple benefits on human health including protection from disease causing pathobiont microbes. It has been recognized that healthy intestinal microbiota is of great importance in the pathogenesis of COVID-19. Gut dysbiosis caused by various reasons is associated with severe COVID-19. Therefore, the modulation of gut microbiota and supplementation of commensal bacterial metabolites could reduce the severity of COVID-19. Many approaches have been studied to improve gut microbiota in COVID-19 including probiotics, bacterial metabolites, and prebiotics, as well as nutraceuticals and trace elements. So far, 19 clinical trials for testing the efficacy of probiotics and synbiotics in COVID-19 prevention and treatment are ongoing. In this narrative review, we summarize the effects of various approaches on the prevention and treatment of COVID-19 and discuss associated mechanisms.
1. Introduction
Severe COVID-19 is characterized by both hyperinflammation and lymphopenia [1]. As such, it could affect multiple organs, causing acute respiratory distress syndrome (ARD), multi-organ failure, and a high rate of mortality. Multiple risk factors for severe COVID-19 have been identified including advanced age, underlying chronic diseases, and use of antibiotics. Dysregulation of the gut microbiota (gut dysbiosis) is associated with the hyperinflammation caused by risk factors in severe COVID-19 disease. Gut dysbiosis can cause chronic inflammation and decreased anti-inflammatory mechanisms to facilitate the formation of the hyperinflammation observed in SARS-CoV-2 infections [1]. It has recently been postulated that intestinal dysbiosis in patients with COVID-19 may describe an enhanced pro-inflammatory tone in the gut with the cytokine storm [2]. This could be attributed to reduced production of commensal bacterial metabolites. Therefore, the modulation of gut microbiota and supplementation of commensal bacterial metabolites could reduce the hyperinflammatory response in severe COVID-19.
The modulation of the intestinal microbiota can be achieved by probiotics, prebiotics, and synbiotics (Figure 1). Supplementation with nutraceuticals and trace elements could also reduce the severity of COVID-19 by improving gut microbiota abundance and diversity. In addition, commensal bacterial metabolites could be supplied to patients with COVID-19, particularly in cases with gut dysbiosis caused by gut SARS-CoV-2 infections and the administration of antibiotics [1].
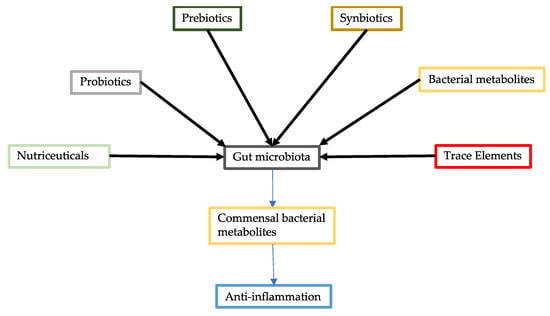
Figure 1.
Modulation of gut microbiota. Gut microbiota can be improved through various approaches including administration of probiotics, prebiotics, synbiotics, commensal bacterial metabolites, nutraceuticals, and trace elements. Improved gut microbiota can increase production of commensal bacterial metabolites, which have an anti-inflammatory effect.
In this narrative review, we summarize preclinical studies of various approaches to improve the gut microbiota, discuss associated mechanisms, and posit possible effects on the prevention and treatment of COVID-19 infections.
2. Probiotics
The rationale for the use of probiotics is from the association of gut dysbiosis and the severity of COVID-19. Several studies have shown the dysbiosis in COVID-19 with decreased beneficial bacteria Lactobacilli and Bifidobacteria as well as butyrate-producing bacteria Faecalibacterium prausnitzii and Eubacterium rectale [3,4]. Alternatively, opportunistic pathogenic bacteria such as Clostrium ramosum and Clostridium hathewayi are increased in abundance [4]. Gut dysbiosis is correlated with an increased cytokine storm and disease severity. The abundance of Faecalibacterium prausnitzii is negatively correlated with the severity of COVID-19, whereas Clostridium ramosum and Clostridium hathewayi are positively correlated with the severity of COVID-19 [4]. Therefore, restoration of these adverse microbiome shifts should be helpful in reducing the severity of COVID-19, and as such, the intestinal dysbiosis that mimics that observed in other clinical scenarios can be relieved by supplementation with probiotics [5]. Indeed, so far, 19 clinical trials are ongoing to test the efficacy of probiotics and synbiotics on the prevention and treatment of COVID-19 (ClinicalTrials.gov, accessed on 15 May 2021) (Table 1).

Table 1.
Clinical trials of probiotics for COVID-19 (ClinicalTrials.gov, accessed on 15 May 2021).
In these clinical trials, the most commonly employed strains were from the Lactobacilli and Bifidobacteria genera which are decreased in COVID-19. Bifidobacteria, belonging to the Actinobacteria phylum, are Gram positive, anaerobic bacteria, which produce acetate, secrete exopolysaccharides, and protect hosts from increased pathobiont proliferation and assault [6]. Bifidobacteria are abundant in infants but decrease with aging. The decreased abundance of Bifidobacteria in advanced age is associated with many chronic diseases [7]. Lactobacilli are Gram-positive, anaerobic, rod-like bacteria which can secrete lactic acid to inhibit pathogenic bacteria [8]. Lactobacilli contain more than 250 member species, a large number of which are employed in probiotic formulation development. Indeed, various Lactobacillus-based probiotics are available which show beneficial effects on many chronic inflammatory diseases such as obesity, diabetes, and hypertension [8].
Probiotics could exert their anti-viral and anti-inflammatory effects to facilitate COVID-19 prevention and treatment. Peptides produced by some probiotics such as Lactobacilli and Paenibacillus have been demonstrated to bind to ACE2, blocking the binding of SARS-CoV-2 to targeted cells [9,10]. Probiotics have also been shown to elicit anti-viral immunity to facilitate the elimination of viruses. In a mouse influenza model, Lactobacillus rhamnosus GG stimulated TLR4 signal to increase anti-viral response [11]. Another study showed that Lactobacillus gasseri increased IFN type I and II secretion, reducing respiratory syncytial viral infections in a mouse model [12]. In thirty heathy elderly patients, administration of Bifidobacterium lactis increased phagocytic and lytic activities of monocytes and killer cells [13]. Sophisticatedly, probiotics have been engineered to carry anti-microbial peptides such as cathelicidin-encoding Lactococcus lactis [14]. Therefore, the use of probiotics in COVID-19 could facilitate blocking the virus from binding to the host cells, reduce viral replication, and increase host immunity against the virus.
The anti-inflammatory effects of probiotics could reduce the hyperinflammation in COVID-19. Probiotics have been demonstrated to reduce inflammation in in vitro and in vivo studies in various inflammation-inducing models [15]. In a high-fat diet-induced obese mouse model, Lactobacillus fermentum CECT5716 reduced inflammation markers and improved epithelial cell functionality [16]. In an intestinal inflammation mouse model, Lactobacilli reduced the levels of TNF-α and increased production of IL-10 [17]. Lactobacilli also increased gut barrier. Bifidobacterium bifidum expressing IL-10 reduced IL-6 production in RAW 264.7 stimulated by LPS and IL-8 production in HT-29 cells stimulated by LPS or TNF-α [18].
The improvement of gut microbiota by probiotics could increase the efficacy of vaccines against SARS-CoV-2 infections. A healthy gut microbiota is necessary for vaccine efficacy [19,20]. Studies have demonstrated that vaccines are unable to elicit robust immune responses in germ-free mice or antibiotics-administered mice [21]. A clinical trial is currently investigating a yeast-based probiotic called ABBC1 to enhance a vaccine against COVID-19 [22]. ABBC1 includes β1,3/β1,6-glucans and inactivated Saccharomyces cerevisae as well as trace elements selenium and zinc. It is expected that supplementation could change gut microbiota to increase COVID-19 vaccine efficacy. The trace elements included in this trial can also modulate gut microbiota (discussed in forthcoming sections). Currently, many vaccines against SARS-CoV-2 have been developed for the prevention of COVID-19. Further enhancement of their efficacies by modulation of the gut microbiota warrants additional focused studies. Furthermore, commensal bacteria such as Enterococcus faecium have been reported to markedly reduce the replication of coronavirus transmissible gastroenteritis virus in a swine testicle cell culture system [23].
3. Commensal Bacterial Metabolites
Commensal intestinal bacteria can produce various metabolites to mediate health benefits. The metabolites include short-chain fatty acids (SCFAs), bile acid derivatives, and amino acids [24]. SCFAs, particularly butyrate has been extensively studied as a mediator of commensal bacteria. In COVID-19, these beneficial metabolites could be in deficit due to dysbiosis. Several reports have proposed the use of butyrate for reducing the severity of COVID-19 infections [1,25,26]. Explicitly, butyrate could be administered in those cases with severe intestinal SARS-CoV-2 infections and/or the extensive use of antibiotics or other therapeutic agents that affect gut microbiota, such as proton pump inhibitors, antidepressants, laxatives, and statins [27,28], which makes it difficult for probiotics to help restore the abundance and diversity of the gut microbiota.
Butyrate could increase immune responses to protect hosts from SARS-CoV-2 infections. Butyrate has direct anti-viral effects through increased secretion of mucins and anti-microbial peptide defensins [1]. A study has shown that administration of SCFAs to animals can increase expression of IFN-γ and granzyme B in cytotoxic lymphocytes through inhibition of histone deacetylases (HDACs) [29]. All three SCFAs have such an effect but butyrate is strongest, propionate second, and acetate weakest. Another study revealed that HDAC inhibitors butyrate and panobinostat could suppress ACE2 [30]. In a gut organoid model, butyrate treatment decreased the expression of both Ace2 and Tmprss2 genes [31].
Butyrate can reduce the severity of COVID-19 through its well-known anti-inflammatory effect. It reduces gut inflammation by activating regulatory T cells and reduces systemic inflammation through increasing the gut barrier to prevent the translocation of endotoxins and bacteria to extra-intestinal organs [28]. Butyrate also inhibits multiple pro-inflammatory signaling pathways to reduce cytokine storms [32].
4. Prebiotics
Prebiotics are substances that can stimulate the growth of gut commensal bacteria and produce microbial metabolites [33]. There are a wide range of prebiotics, which have been extensively studied for health benefits through gut microbiota modulation and stimulation for the production of commensal bacterial metabolites. Common prebiotics include dietary fibers, inulin, fructan, β-glucans, and arabinoxylan [33]. Unfermented dietary fibers such as cellulose cannot be considered prebiotics. Although these prebiotics have not been tested in patients with COVID-19, they are potentially effective for reducing the severity of COVID-19 through characteristic properties that modulate the intestinal microbiota. Several clinical trials in COVID-19 have included prebiotics. The beneficial effects of common prebiotics are introduced below.
4.1. Inulin, FOS (Fructo-Oligosaccharides), and GOS (Galacto-Oligosaccharides)
Inulin is a natural polymer of fructose linked by β (2,1) glycosidic bond, which exists in many plants such as Jerusalem artichoke, onion, garlic, wheat, and asparagus as energy storage [34]. Inulin can have as many as 200 fructose residues while it refers to fructo-oligosaccharide if the fructose residue number is less than 10. An intervention study in healthy subjects showed that administration of inulin (20 g daily for 6 weeks) increased Bifidobacterium and Lachnispiraceae with increased butyrate, isobutyrate, and isovalerate productions in the gut [35]. Inulin was also reported to have anti-influenza effects through the activation of CD8+ T cells [36]. Inulin-fed mice had increased systemic Ly6c− patrolling monocytes, which in turn increased activated macrophages with decreased production of CXCL1 in the respiratory tract. Decreased CXCL1 led to decreased neutrophil recruitment to the respiratory tract and thus limited inflammation. Dietary fiber also increased butyrate production, which boosted effector CD8+ T cells to increase virus clearance. In a feline experiment, feeding of 1-kestose (the simplest fructo-oligosaccharides with two fructose residues) increased the abundance of butyrate-producing bacteria as well as Bifidobacterium [37]. In a mouse obese model established by subcutaneous injection of monosodium glutamate, administration of 5% FOS reduced chronic inflammation, steatohepatitis, and visceral adiposity with increased fecal concentrations of SCFAs [38]. FOS increased not only cecal butyrate but also portal and aortic butyrate concentrations [39]. A clinical trial showed that FOS increased probiotic bacteria Bifidobacterium and Lactobacillus as well as butyrate-producing bacteria Faecalibacterium, Ruminococcus, and Oscillospira [40]. Therefore, the use of inulin and FOS in COVID-19 could restore gut microbiota abundance and diversity with increased butyrate production.
GOS is a polymer of galactose linked by β (1,4) glycosidic bond, which cannot be digested by humans but can be fermented by commensal bacteria in the intestines [34]. It is the substrate of commensal bacteria Lactobacilli and Bifidobacterium. Thus, GOS can increase the abundance of Lactobacilli and Bifidobacteria [41,42,43,44]. Not surprisingly, GOS is able to reduce infections by pathobionts. Searle et al. (2009) showed that GOS reduced Salmonella infections in mice [45]. Zou et al. (2020) demonstrated that GOS reduced infections of pathogenic E. Coli through the modification of commensal bacteria and the gut barrier [46]. GOS also decreased the inflammatory markers IL-6, TNF-α, and IL-1β. The anti-inflammatory effect may be related to butyrate production as an in vitro study showed GOS increased butyrate production [47]. In a rotavirus infection suckling rat model, FOS and GOS reduced infectivity of the virus and symptoms in rats [48].
FOS and GOS could plausibly increase the efficacy of vaccines against COVID-19. A study showed that maternal FOS supplementation increased offspring responses to an oral vaccine with increased IFN-γ and IgA as well as butyrate levels [49]. In a mouse model of influenza vaccination, a combination of 2-fucosyllactose, FOS, and GOS increased immune responses, including increased vaccine-specific delayed type hypersensitivity, increased IG1/IG2a in serum and activated B cells, regulatory T cells, and Th1 cells in mesenteric lymph nodes [50]. Supplementation of FOS also reduced the allergy of mice to bovine milk whey protein through the activation of regulatory T cells [51], indicating that FOS may be able to reduce the side effects of vaccines against COVID-19.
4.2. β-Glucans
β-glucans are glucose polymers linked by β(1,3)-glycosidic bonds as backbones branched with β(1,6) bonds, which are mainly derived from the cell walls of yeast, mushroom, and oats [52]. β-glucans are well studied to have immune modulation effects to promote both innate and adaptive immune responses [52]. Studies have shown that β-glucans are also designated as prebiotics, which promote beneficial bacterial growth and increase production of SCFAs. Dong et al. (2020) showed that oat β-glucan promoted the growth of Lactobacillus, Bifidobacterium, and butyrate-producing bacteria Blautia and Dialister with increased production of butyrate in an in vitro human fecal microbiota fermentation system [53]. In weaned pigs, feeding of oat β-glucan promoted the growth of Lactobacilli and Bifidobacteria with increased production of butyrate [54]. In rats, feeding of oat β-glucan together with resistant starch increased acetate, propionate, and butyrate levels [55]. A clinical trial showed that 3% barley β-glucans in diet increased beneficial bacteria Clostridium orbiscindens, Clostridium sp., Roseburia hominis, and Ruminococcus sp. with markedly increased SCFAs including 2-methyl-propanoic, acetic, butyric, and propionic acids. β-glucan hydroxylates have a higher ability to increase the production of butyrate [56], which warrants further studies and clinical trials.
4.3. Resistant Starches
Resistant starches cannot be digested by human enzymes amylases and amyloglucosidases but can be fermented by gut microbiota in the intestines [57]. There are five types of resistant starches, namely, type 1 (RS1)—in food matrix or seed; type 2 (RS2)—in compact granules; type 3 (RS3)—retrogradation by cooking/cooling; type 4 (RS4)—chemical modification by food manufacture; and type 5 (RS5)—forming complex with lipid [53]. In a mouse model fed with a high-fat diet, addition of RS2 in the diet increased the colonic butyric acid concentrations by 2.6-fold with decreased inflammation, weight gain, and hepatic steatosis [58]. Several clinical trials showed that RS2 increased abundance of Bifidobacteria and increased production of SCFAs [59]. RS3 produced large amounts of butyric acid after fermentation in vitro [60]. A recent study in an in vitro fermentation system showed that RS5 was superior in the production of butyrate while RS3 produced more lactic acid [61]. These studies indicate that resistant starches could modulate gut microbiota and improve butyrate levels in COVID-19.
5. Synbiotics
Synbiotics have greater advantages in modulating the gut microbiota when compared with probiotics and prebiotics. The gut fermentation production of commensal bacteria requires both commensal bacteria and substrates for fermentation. Synbiotics provide both needs to facilitate the production of commensal bacterial metabolites. In a mouse colitis model, Son et al. (2019) showed that a synbiotic composed of Lactobaccillus GG and tagatose was superior to Lactobaccillus GG and tagatose alone in inhibiting inflammatory responses [62]. Fuhren et al. (2021) showed that inulin increased the intestinal persistence of inulin consuming bacterium L. plantarum Lp900 contained in the synbiotic [63]. Studies also showed that the synbiotic Bacillus coagulans MTCC5856 and sugarcane fiber produced more profound effects than B. coagulans and sugarcane fiber alone in inhibiting inflammation and producing SCFAs in a colitis model [64,65].
In a clinical trial, a synbiotic called Omni-Biotic® 10 AAD was used for COVID-19 treatment. It contained two Bifidobacterial strains, one Enterococcus strain, and seven Lactobacillus strains [66]. The prebiotics used were maize starch, inulin, and FOS. The synbiotic also contained trace elements magnesium sulphate, manganese sulphate, and potassium chloride. The effectiveness of Omni-Biotic®® 10 AAD was demonstrated in a previous clinical trial for the treatment of sepsis [67]. It improved gut microbiota diversity and the gut barrier, and reduced inflammation. A similar synbiotic (Omni-Biotic® stress repair) was shown to have anti-inflammatory effect in inflammatory bowel disease [68] with increased butyrate production, reduced CD4+ T cells, and increased gut barrier. The anti-inflammatory effect of the synbiotic could be mediated by regulatory T cells, which are activated by butyrate [69].
6. Nutraceuticals
Nutrition status has been associated with severity of COVID-19 infections. Moreover, in the elderly hospitalized with COVID-19, the Geriatric Nutritional Risk Index was shown to be a significant predictor of survival [70]. Deficiencies in nutraceuticals can result in severe COVID-19. Therefore, supplementation with nutraceuticals that have immune-boosting capabilities (e.g., lactoferrin, selenium, quercetin) [71] could be adjunct therapies to relieve COVID-19 infection severity and reduce the rate of mortality. Certainly, additional studies are required to fully understand the putative effects of nutraceuticals against COVID-19 severity of infections [72].
6.1. Vitamin C
Vitamin C was found to have anti-viral effects during the early 1970s, decreasing the severity of the common cold [73,74]. In a mouse model, supplementation of vitamin C increased anti-viral effects through increased production of IFNα and IFNβ [75]. These findings lead to suggestions of vitamin C for the treatment of COVID-19. Indeed, a study showed that vitamin C can reduce mortality rate and oxygen support in COVID-19 in a high dose [76]. So far, vitamin C has been included in more than 50 clinical trials for the treatment of COVD-19 infections (ClinicalTrials.gov, accessed on 15 May 2021).
Vitamin C supplementation can improve gut microbiota and butyrate production [77]. In an animal model, vitamin C improved hypertension, which is a risk factor for severe COVID-19, through improvements in the gut microbiota, gut integrity, and reducing inflammatory sequelae [78,79].
6.2. Vitamin D
Gavioli et al. (2021) [80] and others have reported that deficiency of vitamin D is associated with susceptibility to SARS-CoV-2 infections, severity of COVID-19, and mortality rate [1,79,81]. Katz et al. (2021) examined the strength of such an association and found that vitamin D deficiency increased the likelihood of positive COVID-19 by 4.6 times [81]. A recent meta-analysis study showed that deficiency of vitamin D was correlated with the severity of COVID-19; patients with poor prognosis had much lower levels of vitamin D [82]. In a retrospective study, Infante et al. (2021) reported that total serum 25-hydroxy-vitamin D levels were much higher in survivors than non-survivors (12 ng/mL vs. 8 ng/mL). Moreover, a multivariate analysis showed an inverse correlation between 25-hydroxy-vitamin D and risk of mortality [82]. Gavioli et al. (2021) also showed that 25-hydroxy-vitamin D levels less than 10 ng/mL had high hospitalization rates (98%) and mortality rates (49%) [80]. The role of vitamin D in COVID-19 could be related with its anti-inflammatory effect. Vitamin D is metabolized into 25-hydroxy vitamin D and active form 1,25-dihydroxy-vitamin D. The binding of 1,25 dihydroxy vitamin D to its intracellular receptor VDR has been postulated to regulate immune responses. As VDR exists in many cells, vitamin D can regulate many physiological processes to exert anti-inflammatory effects (Figure 2) [83,84].
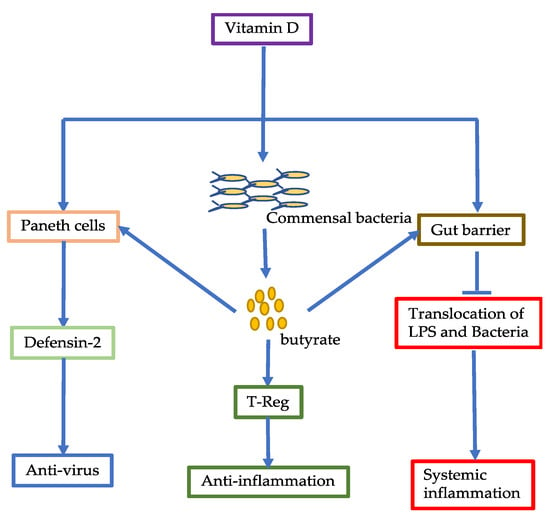
Figure 2.
Effects of vitamin D on the intestinal cells, gut barrier, and gut microbiota. Vitamin D can improve gut microbiota, leading to increased production of butyrate. Butyrate can exert anti-inflammatory effects, increase gut barrier, and promote Paneth cells to secret defensin-2. Vitamin D can also directly act on gut barrier to decrease translocation of LPS and bacteria to prevent systemic inflammation. Vitamin D can also promote defensin-2 secretion by Paneth cells to increase anti-viral effect.
There are bidirectional interactions between vitamin D and the gut microbiota, in terms of significantly influencing the abundance and diversity of the gut microbiota. In a recent study, Charoenngam et al. (2020) showed that administration of 25(OH)D increased the abundance of Bacteroides and Parabacteroides in a dose-dependent manner [85]. Alternatively, the gut microbiota can increase vitamin D levels through inducing increases in the levels of VDR receptors. In a zebrafish model, probiotic Lactobacillus casae BL23 increased the expression of VDR [86]. Lu et al. (2020) showed that conditional medium from five probiotic lactic acid bacteria increased VDR expression in HCT116 cells [87]. Probiotics Lactobacillus rhamnosus GG and Lactobacillus plantarum increased VDR proteins in mouse and human epithelial cells as well as expression of the target gene cathelicidin [88]. Knockout of VDR resulted in the loss of the protective effect of these probiotics on Salmonella-induced colitis [89].
6.3. Lactoferrin
Lactoferrin is an iron chelating milk bioactive peptide. Due to its beneficial effects on the development of the gut as well as disease prevention and therapy through anti-inflammatory and anti-viral effects, lactoferrin has been suggested as a therapy for COVID-19 [90,91]. An in vitro study in Caco-2 cells showed that lactoferrin inhibited SARS-CoV-2 replication through promoting anti-viral gene expression and reducing the expression of genes necessary for viral replication including RNA-dependent RNA polymerase (RdRp) and E gene (CoVE) [92]. Another in vitro study showed that bovine lactoferrin bound to heparin sulfate proteoglycan, an attachment factor for the binding of SARS-CoV-2 to ACE2, reducing the viral entry into cells [93].
Lactoferrin can modulate the intestinal microbiota by stimulating the growth of beneficial bacteria and inhibiting pathogenic bacteria. Lactoferrin and its fragment Lactoferricin B were tested for their effects on anaerobic opportunistic pathogens Bacteroides fragilis and Bacteroides thetaiotaomicron [94]. Lactoferrin but not Lactoferricin B was able to inhibit in vitro biofilm formation and binding to the laminins of these two bacteria. Other studies also reported the properties of lactoferrin and the inhibition of biofilm formation by Bacteroides fragilis [95,96]. In suckling piglets, feeding of lactoferrin increased abundance of Roseburia and decreased Escherichia-Shigella [96]. The colon butyrate concentrations were also increased with increased gut barrier integrity and increased secretion of mucin. In addition, the concentration of anti-inflammatory cytokine IL-10 in the colonic mucosa was increased while the concentrations of pro-inflammatory cytokines IL-1α and IL-1β were decreased. This study suggests that lactoferrin can increase gut barrier functional integrity and decrease inflammatory triggers through the modulation of gut bacteria and the secretion of bacterial metabolites such as butyrate. Therefore, supplementation of lactoferrin could reduce infectivity of SARS-CoV-2 and improve the overall gut microbiota, reducing the severity of COVID-19. At present, several clinical trials are ongoing to examine the efficacy of lactoferrin in the prevention and treatment of COVID-19 (ClinicalTrials.gov, accessed on 15 May 2021).
6.4. Omega-3 Fatty Acids
Omega-3 polyunsaturated fatty acids (PUFA) are well-known micronutrients benefiting human health with anti-inflammatory and anti-thrombotic effects [97,98]. However, omega-6 PUFA is pro-inflammatory and thus, the ratio of omega-3 to omega-6 has been used to indicate an adverse inflammatory state. In severe COVID-19, it was found that metabolites of omega-6, such as eicosanoids including prostanoids and leukotrienes, are increased [99]. What Hammock and colleagues regarded as an eicosanoid storm in COVID-19 promoted hyperinflammation [100]. A recent study showed that omega-3 PUFA could bind to protein spikes in its close conformation, thus preventing SARS-CoV-2 viral entry into cells [101]. Numerous clinical trials investigating supplementation with omega-3 PUFA in COVID-19 are ongoing (ClinicalTrials.gov, accessed on 15 May 2021).
The effect of omega-3 PUFA on SARS-CoV infections could be indirectly associated with the gut microbiota. Studies have shown that supplementation with omega-3 PUFA and its metabolites docosahexaenoic acid improves the composition of the gut microbiota [102,103,104,105]. Indeed, supplementation of omega-3 to healthy subjects increased beneficial Bifidobacterium, Roseburia, and Lactobacillus [106]. Another study also showed that dietary intervention of human subjects with 500 mg of omega-3 increased gut beneficial Coprococcus spp. and Bacteroides spp. and decreased fatty liver associated Colinsella spp. with increased SCFAs [35]. In a rat model, Zhu et al. (2020) showed that omega 3 (flaxseed oil) reduced diabetes patients’ blood glucose through decreasing blood levels of LPS and inflammatory markers IL-6, IL-17A, IL-1β, and TNF-α, as well as increasing SCFAs [107].
7. Trace Elements
Trace elements are essential for numerous normal physiological processes, and deficiencies of trace elements can cause various diseases and weaken immune network defenses against pathogenic insults. Several trace element deficiencies have been associated with the severity of COVID-19 [108]. Thus, supplementation with trace elements could reduce the severity of the COVID-19 infection. Alternatively, excessive trace elements could be deleterious to health; therefore, a prudent supplementation approach is important in order to maintain these elements in proportionate and adequate levels.
7.1. Zinc
Zinc is a trace element required for many physiological processes such as activation of signaling pathways involved in innate and adaptive immune responses, many enzyme functionalities, and cell membrane permeability. Thus, zinc deficiency (less than 60 mcg/dL) can cause dysfunctions of some physiological processes, leading to disease status [100]. Indeed, many risk factors for severe COVID-19 have been associated with deficiency of zinc such as advanced age, chronic cardiac or pulmonary disease, hypertension, and diabetes [109]. Weakened anti-viral immunity due to the deficiency of zinc could also be a mechanism for these risk factors-causing severe COVID-19 infections. Zinc has been shown to have direct anti-viral effects [109,110].
Studies have shown that zinc is important for the maintenance of gut microbiota homeostasis. In a chick model, chronic zinc depletion reduced species diversity with decreased the levels of SCFAs [111]. With supplementation of zinc to animals challenged with pathogenic bacteria Salmonella and E. Coli, the number of their pathobionts was reduced with increased abundance of Lactobacilli [112,113]. Butyrate concentration was increased with increased IL-10 and decreased IL-6 [113]. However, a study showed that excessive zinc (12-fold of normal mouse chow) caused dysbiosis which increased the severity of Clostridium difficile infection [114]. Therefore, adequate dosage of zinc supplementation is critical for its role in gut microbiota homeostasis.
7.2. Selenium
Selenium is necessary for normal physiological processes and lack of selenium causes poor immune function [115]. Deficiency of selenium can impair immune cell functionalities and cause inadequate inflammatory responses [116]. Adequate levels of selenium have been associated with good outcomes of COVID-19 while sub-optimal selenium has been found in hospitalized patients [117]. Therefore, supplementation of selenium of patients with COVID-19 to maintain suitable selenium levels could be helpful to avoid severe COVID-19 and reduce the mortality rate [117].
Selenium is closely associated with the homeostasis of gut microbiota and commensal bacterial metabolites. Administration of selenium nanoparticles into chicks increased beneficial bacteria Lactobacillus and Faecalibacterium with increased production of butyrate [118]. Wei et al. (2019) showed that a hydroxy-analog of selenomethionine was better that sodium selenite, which produced dose-dependent increases of volatile fatty acids, propionate, and butyrate [119]. However, excessive selenium is also a disturbing factor for the gut microbiota. Therefore, it is important to maintain selenium at optimal levels in patients with COVID-19.
7.3. Magnesium
Magnesium deficiency has been shown to increase the cytokine storm in COVID-19 [120]. Indeed, magnesium deficiency is common in patients with risk factors of severe COVID-19 such as aging, type 2 diabetes, metabolic syndrome, cardiovascular disease, depression, and hypertension [121,122]. Deficiency of magnesium causes intestinal and systemic inflammation with leukocyte and macrophage activation and increased production of pro-inflammatory cytokines [123,124]. Therefore, supplementation with magnesium may be an important requisite for patients with COVID-19 infections.
Magnesium deficiency as well as oversupply can cause gut dysbiosis. In a mouse model, a magnesium deficient-diet caused altered gut microbiota such as decreased Bifidobacterum [124,125,126]. Magnesium deficiency in mice caused increases in both intestinal and systemic inflammatory tone, indicated by increased blood levels of TNF-α and IL6 [126]. In addition, a study showed that oversupply with high dose magnesium to rats which were not deficient in magnesium progressed intestinal dysbiosis [127].
8. Conclusions
As intestinal dysbiosis is a common occurrence in the pathogenesis of COVID-19, it is essential that improvement in the health of the gut microbiota is addressed. Various approaches could be helpful in modulating the gut microbiota. Probiotics, prebiotics, and synbiotics could directly and beneficially alter the gut microbiota. However, commensal bacterial metabolites may be more effective in those cases with severe intestinal SARS-CoV-2 infections and/or with the extensive use of antibiotics and other therapeutic agents such as proton pump inhibitors, antidepressants, laxatives, and statins. Supplementation of nutraceuticals and trace elements could facilitate the improvement of the gut microbiota, particularly in those patients proven to have trace element deficits. However, particular attention should be paid to the dosage of trace elements that are administered, given that excessive trace elements can trigger gut dysbiosis. A healthy, diverse, and abundant gut microbiota not only reduces the severity of COVID-19 but also increases the efficacy of vaccines against COVID-19.
Author Contributions
Conceptualization, J.C. and L.V; writing—original draft preparation, J.C.; writing—review and editing, L.V.; supervision, L.V.; All authors have read and agreed to the published version of the manuscript.
Funding
The preparation of this article received no external funding.
Institutional Review Board Statement
Not Applicable.
Informed Consent Statement
Not Applicable.
Data Availability Statement
www.clinicaltrials.gov, accessed on 15 May 2021.
Conflicts of Interest
We declare that we have no conflicts of interest.
References
- Chen, J.; Hall, S.; Vitetta, L. Altered gut microbial metabolites could mediate the effects of risk factors in Covid-19. Rev. Med. Virol. 2021. [Google Scholar] [CrossRef]
- Marasco, G.; Lenti, M.V.; Cremon, C.; Barbaro, M.R.; Stanghellini, V.; Di Sabatino, A.; Barbara, G. Implications of SARS-CoV-2 infection for neurogastroenterology. Neurogastroenterol. Motil. 2021, 33, e14104. [Google Scholar] [CrossRef]
- Yeoh, Y.K.; Zuo, T.; Lui, G.C.; Zhang, F.; Liu, Q.; Li, A.Y.; Chung, A.C.; Cheung, C.P.; Tso, E.Y.; Fung, K.S.; et al. Gut microbiota composition reflects disease severity and dysfunctional immune responses in patients with COVID-19. Gut 2021, 70, 698–706. [Google Scholar] [CrossRef]
- Zuo, T.; Zhang, F.; Lui, G.C.Y.; Yeoh, Y.K.; Li, A.Y.L.; Zhan, H.; Wan, Y.; Chung, A.C.K.; Cheung, C.P.; Chen, N.; et al. Alterations in Gut Microbiota of Patients With COVID-19 During Time of Hospitalization. Gastroenterology 2020, 159, 944–955. [Google Scholar] [CrossRef]
- Marasco, G.; Cirota, G.G.; Rossini, B.; Lungaro, L.; Di Biase, A.R.; Colecchia, A.; Volta, U.; De Giorgio, R.; Festi, D.; Caio, G. Probiotics, Prebiotics and Other Dietary Supplements for Gut Microbiota Modulation in Celiac Disease Patients. Nutrients 2020, 12, 2674. [Google Scholar] [CrossRef] [PubMed]
- Schiavi, E.; Gleinser, M.; Molloy, E.; Groeger, D.; Frei, R.; Ferstl, R.; Rodriguez-Perez, N.; Ziegler, M.; Grant, R.; Moriarty, T.F.; et al. The Surface-Associated Exopolysaccharide of Bifidobacterium longum 35624 Plays an Essential Role in Dampening Host Proinflammatory Responses and Repressing Local TH17 Responses. Appl. Environ. Microbiol. 2016, 82, 7185–7196. [Google Scholar] [CrossRef] [Green Version]
- Kelly, S.M.; Munoz-Munoz, J.; van Sinderen, D. Plant Glycan Metabolism by Bifidobacteria. Front. Microbiol. 2021, 12, 609418. [Google Scholar] [CrossRef]
- Hutchinson, A.N.; Tingö, L.; Brummer, R.J. The Potential Effects of Probiotics and ω-3 Fatty Acids on Chronic Low-Grade Inflammation. Nutrients 2020, 12, 2402. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhao, J.; Wang, X.; Qayum, A.; Hussain, M.A.; Liang, G.; Hou, J.; Jiang, Z.; Li, A. Novel Angiotensin-Converting Enzyme-Inhibitory Peptides From Fermented Bovine Milk Started by Lactobacillus helveticus KLDS.31 and Lactobacillus casei KLDS.105: Purification, Identification, and Interaction Mechanisms. Front. Microbiol. 2019, 10, 2643. [Google Scholar] [CrossRef] [PubMed]
- Minato, T.; Nirasawa, S.; Sato, T.; Yamaguchi, T.; Hoshizaki, M.; Inagaki, T.; Nakahara, K.; Yoshihashi, T.; Ozawa, R.; Yokota, S.; et al. B38-CAP is a bacteria-derived ACE2-like enzyme that suppresses hypertension and cardiac dysfunction. Nat. Commun. 2020, 11, 1058. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumova, O.K.; Fike, A.J.; Thayer, J.L.; Nguyen, L.T.; Mell, J.C.; Pascasio, J.; Stairiker, C.; Leon, L.G.; Katsikis, P.D.; Carey, A.J. Lung transcriptional unresponsiveness and loss of early influenza virus control in infected neonates is prevented by intranasal Lactobacillus rhamnosus GG. PLoS Pathog. 2019, 15, e1008072. [Google Scholar] [CrossRef] [Green Version]
- Eguchi, K.; Fujitani, N.; Nakagawa, H.; Miyazaki, T. Prevention of respiratory syncytial virus infection with probiotic lactic acid bacterium Lactobacillus gasseri SBT2055. Sci. Rep. 2019, 9, 4812. [Google Scholar] [CrossRef] [PubMed]
- Gill, H.S.; Rutherfurd, K.J.; Cross, M.L.; Gopal, P.K. Enhancement of immunity in the elderly by dietary supplementation with the probiotic Bifidobacterium lactis HN019. Am. J. Clin. Nutr. 2001, 74, 833–839. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.C.; Zhang, L.; Li, Z.J.; Wu, W.K.; Ren, S.X.; Chen, Y.C.; Ng, T.B.; Cho, C.H. Protective effects of cathelicidin-encoding Lactococcus lactis in murine ulcerative colitis. J. Gastroenterol. Hepatol. 2012, 27, 1205–1212. [Google Scholar] [CrossRef] [PubMed]
- Plaza-Díaz, J.; Ruiz-Ojeda, F.J.; Vilchez-Padial, L.M.; Gil, A. Evidence of the Anti-Inflammatory Effects of Probiotics and Synbiotics in Intestinal Chronic Diseases. Nutrients 2017, 9, 555. [Google Scholar] [CrossRef] [Green Version]
- Molina-Tijeras, J.A.; Diez-Echave, P.; Vezza, T.; Hidalgo-García, L.; Ruiz-Malagón, A.J.; Rodríguez-Sojo, M.J.; Romero, M.; Robles-Vera, I.; García, F.; Plaza-Diaz, J.; et al. Lactobacillus fermentum CECT5716 ameliorates high fat diet-induced obesity in mice through modulation of gut microbiota dysbiosis. Pharmacol. Res. 2021, 167, 105471. [Google Scholar] [CrossRef]
- Monteros, M.J.M.; Galdeano, C.M.; Balcells, M.F.; Weill, R.; De Paula, J.A.; Perdigón, G.; Cazorla, S.I. Probiotic lactobacilli as a promising strategy to ameliorate disorders associated with intestinal inflammation induced by a non-steroidal anti-inflammatory drug. Sci. Rep. 2021, 11, 571. [Google Scholar] [CrossRef]
- Hong, N.; Ku, S.; Yuk, K.; Johnston, T.V.; Ji, G.E.; Park, M.S. Production of biologically active human interleukin-10 by Bifidobacterium bifidum BGN4. Microb. Cell Factories 2021, 20, 16. [Google Scholar] [CrossRef] [PubMed]
- Vitetta, L.; Vitetta, G.; Hall, S. Immunological Tolerance and Function: Associations Between Intestinal Bacteria, Probiotics, Prebiotics, and Phages. Front. Immunol. 2018, 9, 2240. [Google Scholar] [CrossRef] [Green Version]
- Vitetta, L.; Saltzman, E.T.; Thomsen, M.; Nikov, T.; Hall, S. Adjuvant Probiotics and the Intestinal Microbiome: Enhancing Vaccines and Immunotherapy Outcomes. Vaccines 2017, 5, 50. [Google Scholar] [CrossRef] [Green Version]
- Sommer, F.; Bäckhed, F. The gut microbiota—Masters of host development and physiology. Nat. Rev. Microbiol. 2013, 11, 227–238. [Google Scholar] [CrossRef]
- Matues Rodriguez, J.A. Efficacy and Tolerability of ABBC1 in Volunteers Receiving the Influenza or Covid-19 Vaccine. Available online: https://clinicaltrials.gov/ct2/show/NCT04798677 (accessed on 15 May 2021).
- Chai, W.; Burwinkel, M.; Wang, Z.; Palissa, C.; Esch, B.; Twardziok, S.; Rieger, J.; Wrede, P.; Schmidt, M.F. Antiviral effects of a probiotic Enterococcus faecium strain against transmissible gastroenteritis coronavirus. Arch. Virol. 2013, 158, 799–807. [Google Scholar] [CrossRef] [Green Version]
- Chen, J.; Vitetta, L. Gut Microbiota Metabolites in NAFLD Pathogenesis and Therapeutic Implications. Int. J. Mol. Sci. 2020, 21, 5214. [Google Scholar] [CrossRef]
- Anderson, G.; Reiter, R.J. Melatonin: Roles in influenza, Covid-19, and other viral infections. Rev. Med. Virol. 2020, 30, e2109. [Google Scholar] [CrossRef] [PubMed]
- Archer, D.L.; Kramer, D.C. The Use of Microbial Accessible and Fermentable Carbohydrates and/or Butyrate as Supportive Treatment for Patients With Coronavirus SARS-CoV-2 Infection. Front. Med. 2020, 7, 292. [Google Scholar] [CrossRef]
- Vich Vila, A.; Collij, V.; Sanna, S.; Sinha, T.; Imhann, F.; Bourgonje, A.R.; Mujagic, Z.; Jonkers, D.; Masclee, A.A.M.; Fu, J.; et al. Impact of commonly used drugs on the composition and metabolic function of the gut microbiota. Nat. Commun. 2020, 11, 362. [Google Scholar] [CrossRef]
- Imhann, F.; Vich Vila, A.; Bonder, M.J.; Lopez Manosalva, A.G.; Koonen, D.P.Y.; Fu, J.; Wijmenga, C.; Zhernakova, A.; Weersma, R.K. The influence of proton pump inhibitors and other commonly used medication on the gut microbiota. Gut Microbes 2017, 8, 351–358. [Google Scholar] [CrossRef] [Green Version]
- Luu, M.; Weigand, K.; Wedi, F.; Breidenbend, C.; Leister, H.; Pautz, S.; Adhikary, T.; Visekruna, A. Regulation of the effector function of CD8(+) T cells by gut microbiota-derived metabolite butyrate. Sci. Rep. 2018, 8, 14430. [Google Scholar] [CrossRef] [Green Version]
- Takahashi, Y.; Hayakawa, A.; Sano, R.; Fukuda, H.; Harada, M.; Kubo, R.; Okawa, T.; Kominato, Y. Histone deacetylase inhibitors suppress ACE2 and ABO simultaneously, suggesting a preventive potential against COVID-19. Sci. Rep. 2021, 11, 3379. [Google Scholar] [CrossRef] [PubMed]
- Chemudupati, M.; Kenney, A.D.; Smith, A.C.; Fillinger, R.J.; Zhang, L.; Zani, A.; Liu, S.L.; Anderson, M.Z.; Sharma, A.; Yount, J.S. Butyrate Reprograms Expression of Specific Interferon-Stimulated Genes. J. Virol. 2020, 94. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Zhao, K.N.; Vitetta, L. Effects of Intestinal Microbial–Elaborated Butyrate on Oncogenic Signaling Pathways. Nutrients 2019, 11, 1026. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Quigley, E.M.M. Prebiotics and Probiotics in Digestive Health. Clin. Gastroenterol. Hepatol. 2019, 17, 333–344. [Google Scholar] [CrossRef]
- Wilson, B.; Whelan, K. Prebiotic inulin-type fructans and galacto-oligosaccharides: Definition, specificity, function, and application in gastrointestinal disorders. J. Gastroenterol. Hepatol. 2017, 32 (Suppl. 1), 64–68. [Google Scholar] [CrossRef] [Green Version]
- Vijay, A.; Astbury, S.; Le Roy, C.; Spector, T.D.; Valdes, A.M. The prebiotic effects of omega-3 fatty acid supplementation: A six-week randomised intervention trial. Gut Microbes 2021, 13, 1–11. [Google Scholar] [CrossRef]
- Trompette, A.; Gollwitzer, E.S.; Pattaroni, C.; Lopez-Mejia, I.C.; Riva, E.; Pernot, J.; Ubags, N.; Fajas, L.; Nicod, L.P.; Marsland, B.J. Dietary Fiber Confers Protection against Flu by Shaping Ly6c(-) Patrolling Monocyte Hematopoiesis and CD8(+) T Cell Metabolism. Immunity 2018, 48, 992–1005. [Google Scholar] [CrossRef] [Green Version]
- Shinohara, M.; Kiyosue, M.; Tochio, T.; Kimura, S.; Koga, Y. Activation of butyrate-producing bacteria as well as bifidobacteria in the cat intestinal microbiota by the administration of 1-kestose, the smallest component of fructo-oligosaccharide. J. Veter Med. Sci. 2020, 82, 866–874. [Google Scholar] [CrossRef]
- Takai, A.; Kikuchi, K.; Ichimura, M.; Tsuneyama, K.; Moritoki, Y.; Matsumoto, K.; Tsunashima, H.; Onda, T.; Kuniyoshi, N.; Nariyama, T.; et al. Fructo-oligosaccharides ameliorate steatohepatitis, visceral adiposity, and associated chronic inflammation via increased production of short-chain fatty acids in a mouse model of non-alcoholic steatohepatitis. BMC Gastroenterol. 2020, 20, 46. [Google Scholar] [CrossRef] [PubMed]
- Jakobsdottir, G.; Jädert, C.; Holm, L.; Nyman, M.E. Propionic and butyric acids, formed in the caecum of rats fed highly fermentable dietary fibre, are reflected in portal and aortic serum. Br. J. Nutr. 2013, 110, 1565–1572. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tandon, D.; Haque, M.M.; Gote, M.; Jain, M.; Bhaduri, A.; Dubey, A.K.; Mande, S.S. A prospective randomized, double-blind, placebo-controlled, dose-response relationship study to investigate efficacy of fructo-oligosaccharides (FOS) on human gut microflora. Sci. Rep. 2019, 9, 5473. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Garrido, D.; Ruiz-Moyano, S.; Jimenez-Espinoza, R.; Eom, H.J.; Block, D.E.; Mills, D.A. Utilization of galactooligosaccharides by Bifidobacterium longum subsp. infantis isolates. Food Microbiol. 2013, 33, 262–270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giovannini, M.; Verduci, E.; Gregori, D.; Ballali, S.; Soldi, S.; Ghisleni, D.; Riva, E. Prebiotic effect of an infant formula supplemented with galacto-oligosaccharides: Randomized multicenter trial. J. Am. Coll. Nutr. 2014, 33, 385–393. [Google Scholar] [CrossRef]
- Gonai, M.; Shigehisa, A.; Kigawa, I.; Kurasaki, K.; Chonan, O.; Matsuki, T.; Yoshida, Y.; Aida, M.; Hamano, K.; Terauchi, Y. Galacto-oligosaccharides ameliorate dysbiotic Bifidobacteriaceae decline in Japanese patients with type 2 diabetes. Benef. Microbes 2017, 8, 705–716. [Google Scholar] [CrossRef]
- Fukuda, S.; Toh, H.; Hase, K.; Oshima, K.; Nakanishi, Y.; Yoshimura, K.; Tobe, T.; Clarke, J.M.; Topping, D.L.; Suzuki, T.; et al. Bifidobacteria can protect from enteropathogenic infection through production of acetate. Nature 2011, 469, 543–547. [Google Scholar] [CrossRef] [PubMed]
- Searle, L.E.J.; Best, A.; Nunez, A.; Salguero, F.J.; Johnson, L.; Weyer, U.; Dugdale, A.H.; Cooley, W.A.; Carter, B.; Jones, G.; et al. A mixture containing galactooligosaccharide, produced by the enzymic activity of Bifidobacterium bifidum, reduces Salmonella enterica serovar Typhimurium infection in mice. J. Med. Microbiol. 2009, 58, 37–48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zou, Y.; Wang, J.; Wang, Y.; Peng, B.; Liu, J.; Zhang, B.; Lv, H.; Wang, S. Protection of Galacto-Oligosaccharide against E. coli O157 Colonization through Enhancing Gut Barrier Function and Modulating Gut Microbiota. Foods 2020, 9, 1710. [Google Scholar] [CrossRef]
- Tran, N.T.; Tang, Y.; Li, Z.; Zhang, M.; Wen, X.; Ma, H.; Li, S. Galactooligosaccharides and Resistant Starch Altered Microbiota and Short-Chain Fatty Acids in an in vitro Fermentation Study Using Gut Contents of Mud Crab (Scylla paramamosain). Front. Microbiol. 2020, 11, 1352. [Google Scholar] [CrossRef] [PubMed]
- Rigo-Adrover, M.; Pérez-Berezo, T.; Ramos-Romero, S.; van Limpt, K.; Knipping, K.; Garssen, J.; Knol, J.; Franch, À.; Castell, M.; Pérez-Cano, F.J. A fermented milk concentrate and a combination of short-chain galacto-oligosaccharides/long-chain fructo-oligosaccharides/pectin-derived acidic oligosaccharides protect suckling rats from rotavirus gastroenteritis. Br. J. Nutr. 2017, 117, 209–217. [Google Scholar] [CrossRef] [Green Version]
- Le Bourgot, C.; Le Normand, L.; Formal, M.; Respondek, F.; Blat, S.; Apper, E.; Ferret-Bernard, S.; Le Huërou-Luron, I. Maternal short-chain fructo-oligosaccharide supplementation increases intestinal cytokine secretion, goblet cell number, butyrate concentration and Lawsonia intracellularis humoral vaccine response in weaned pigs. Br. J. Nutr. 2017, 117, 83–92. [Google Scholar] [CrossRef] [Green Version]
- Xiao, L.; Engen, P.A.; Leusink-Muis, T.; van Ark, I.; Stahl, B.; Overbeek, S.A.; Garssen, J.; Naqib, A.; Green, S.J.; Keshavarzian, A.; et al. The Combination of 2’-Fucosyllactose with Short-Chain Galacto-Oligosaccharides and Long-Chain Fructo-Oligosaccharides that Enhance Influenza Vaccine Responses Is Associated with Mucosal Immune Regulation in Mice. J. Nutr. 2019, 149, 856–869. [Google Scholar] [CrossRef] [PubMed]
- Vonk, M.M.; Diks, M.A.P.; Wagenaar, L.; Smit, J.J.; Pieters, R.H.H.; Garssen, J.; van Esch, B.; Knippels, L.M.J. Improved Efficacy of Oral Immunotherapy Using Non-Digestible Oligosaccharides in a Murine Cow’s Milk Allergy Model: A Potential Role for Foxp3+ Regulatory T Cells. Front. Immunol. 2017, 8, 1230. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, J.; Seviour, R. Medicinal importance of fungal beta-(1→3), (1→6)-glucans. Mycol. Res. 2007, 111, 635–652. [Google Scholar] [CrossRef]
- Dong, J.L.; Yang, M.; Zhu, Y.Y.; Shen, R.L.; Zhang, K.Y. Comparative study of thermal processing on the physicochemical properties and prebiotic effects of the oat β-glucan by in vitro human fecal microbiota fermentation. Food Res. Int. 2020, 138, 109818. [Google Scholar] [CrossRef] [PubMed]
- Metzler-Zebeli, B.U.; Zijlstra, R.T.; Mosenthin, R.; Gänzle, M.G. Dietary calcium phosphate content and oat β-glucan influence gastrointestinal microbiota, butyrate-producing bacteria and butyrate fermentation in weaned pigs. FEMS Microbiol. Ecol. 2011, 75, 402–413. [Google Scholar] [CrossRef] [PubMed]
- Drzikova, B.; Dongowski, G.; Gebhardt, E. Dietary fibre-rich oat-based products affect serum lipids, microbiota, formation of short-chain fatty acids and steroids in rats. Br. J. Nutr. 2005, 94, 1012–1025. [Google Scholar] [CrossRef]
- Dong, J.L.; Yu, X.; Dong, L.E.; Shen, R.L. In vitro fermentation of oat β-glucan and hydrolysates by fecal microbiota and selected probiotic strains. J. Sci. Food Agric. 2017, 97, 4198–4203. [Google Scholar] [CrossRef]
- DeMartino, P.; Cockburn, D.W. Resistant starch: Impact on the gut microbiome and health. Curr. Opin. Biotechnol. 2020, 61, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Chen, L.; Hu, M.; Kim, J.J.; Lin, R.; Xu, J.; Fan, L.; Qi, Y.; Wang, L.; Liu, W.; et al. Dietary type 2 resistant starch improves systemic inflammation and intestinal permeability by modulating microbiota and metabolites in aged mice on high-fat diet. Aging 2020, 12, 9173–9187. [Google Scholar] [CrossRef]
- Bendiks, Z.A.; Knudsen, K.E.B.; Keenan, M.J.; Marco, M.L. Conserved and variable responses of the gut microbiome to resistant starch type 2. Nutr. Res. 2020, 77, 12–28. [Google Scholar] [CrossRef]
- Chang, R.; Jin, Z.; Lu, H.; Qiu, L.; Sun, C.; Tian, Y. Type III Resistant Starch Prepared from Debranched Starch: Structural Changes under Simulated Saliva, Gastric, and Intestinal Conditions and the Impact on Short-Chain Fatty Acid Production. J. Agric. Food Chem. 2021, 69, 2595–2602. [Google Scholar] [CrossRef]
- Qin, R.; Wang, J.; Chao, C.; Yu, J.; Copeland, L.; Wang, S.; Wang, S. RS5 Produced More Butyric Acid through Regulating the Microbial Community of Human Gut Microbiota. J. Agric. Food Chem. 2021, 69, 3209–3218. [Google Scholar] [CrossRef]
- Son, S.J.; Koh, J.H.; Park, M.R.; Ryu, S.; Lee, W.J.; Yun, B.; Lee, J.H.; Oh, S.; Kim, Y. Effect of the Lactobacillus rhamnosus strain GG and tagatose as a synbiotic combination in a dextran sulfate sodium-induced colitis murine model. J. Dairy Sci. 2019, 102, 2844–2853. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fuhren, J.; Schwalbe, M.; Rösch, C.; Nijland, R.; Wels, M.; Schols, H.A.; Kleerebezem, M. Dietary inulin increases Lactiplantibacillus plantarum strain Lp900 persistence in rats depending on the dietary-calcium level. Appl. Environ. Microbiol. 2021, 87, e00122-21. [Google Scholar] [CrossRef] [PubMed]
- Shinde, T.; Vemuri, R.; Shastri, S.; Perera, A.P.; Gondalia, S.V.; Beale, D.J.; Karpe, A.V.; Eri, R.; Stanley, R. Modulating the Microbiome and Immune Responses Using Whole Plant Fibre in Synbiotic Combination with Fibre-Digesting Probiotic Attenuates Chronic Colonic Inflammation in Spontaneous Colitic Mice Model of IBD. Nutrients 2020, 12, 2380. [Google Scholar] [CrossRef] [PubMed]
- Shinde, T.; Perera, A.P.; Vemuri, R.; Gondalia, S.V.; Karpe, A.V.; Beale, D.J.; Shastri, S.; Southam, B.; Eri, R.; Stanley, R. Synbiotic Supplementation Containing Whole Plant Sugar Cane Fibre and Probiotic Spores Potentiates Protective Synergistic Effects in Mouse Model of IBD. Nutrients 2019, 11, 818. [Google Scholar] [CrossRef] [Green Version]
- Medical University of Graz. Synbiotic Therapy of Gastrointestinal Symptoms During Covid-19 Infection (SynCov). Available online: https://clinicaltrials.gov/ct2/show/record/NCT04420676 (accessed on 15 May 2021).
- Stadlbauer, V.; Horvath, A.; Komarova, I.; Schmerboeck, B.; Feldbacher, N.; Klymiuk, I.; Durdevic, M.; Rainer, F.; Blesl, A.; Stiegler, P.; et al. Dysbiosis in early sepsis can be modulated by a multispecies probiotic: A randomised controlled pilot trial. Benef. Microbes 2019, 10, 265–278. [Google Scholar] [CrossRef]
- Moser, A.M.; Spindelboeck, W.; Halwachs, B.; Strohmaier, H.; Kump, P.; Gorkiewicz, G.; Högenauer, C. Effects of an oral synbiotic on the gastrointestinal immune system and microbiota in patients with diarrhea-predominant irritable bowel syndrome. Eur. J. Nutr. 2019, 58, 2767–2778. [Google Scholar] [CrossRef] [Green Version]
- Chen, J.; Vitetta, L. Activation of T-regulatory cells by a synbiotic may be important for its anti-inflammatory effect. Eur. J. Nutr. 2019, 58, 3379–3380. [Google Scholar] [CrossRef]
- Recinella, G.; Marasco, G.; Serafini, G.; Maestri, L.; Bianchi, G.; Forti, P.; Zoli, M. Prognostic role of nutritional status in elderly patients hospitalized for COVID-19: A monocentric study. Aging Clin. Exp. Res. 2020, 32, 2695–2701. [Google Scholar] [CrossRef]
- Mrityunjaya, M.; Pavithra, V.; Neelam, R.; Janhavi, P.; Halami, P.M.; Ravindra, P.V. Immune-Boosting, Antioxidant and Anti-inflammatory Food Supplements Targeting Pathogenesis of COVID-19. Front. Immunol. 2020, 11, 570122. [Google Scholar] [CrossRef]
- Infusino, F.; Marazzato, M.; Mancone, M.; Fedele, F.; Mastroianni, C.M.; Severino, P.; Ceccarelli, G.; Santinelli, L.; Cavarretta, E.; Marullo, A.G.M.; et al. Diet Supplementation, Probiotics, and Nutraceuticals in SARS-CoV-2 Infection: A Scoping Review. Nutrients 2020, 12, 1718. [Google Scholar] [CrossRef]
- Pauling, L. The significance of the evidence about ascorbic acid and the common cold. Proc. Natl. Acad. Sci. USA 1971, 68, 2678–2681. [Google Scholar] [CrossRef] [Green Version]
- Karlowski, T.R.; Chalmers, T.C.; Frenkel, L.D.; Kapikian, A.Z.; Lewis, T.L.; Lynch, J.M. Ascorbic acid for the common cold. A prophylactic and therapeutic trial. JAMA 1975, 231, 1038–1042. [Google Scholar] [CrossRef]
- Kim, Y.; Kim, H.; Bae, S.; Choi, J.; Lim, S.Y.; Lee, N.; Kong, J.M.; Hwang, Y.I.; Kang, J.S.; Lee, W.J. Vitamin C Is an Essential Factor on the Anti-viral Immune Responses through the Production of Interferon-α/β at the Initial Stage of Influenza A Virus (H3N2) Infection. Immune Netw. 2013, 13, 70–74. [Google Scholar] [CrossRef] [Green Version]
- Liu, F.; Ye, S.; Zhu, X.; He, X.; Wang, S.; Li, Y.; Lin, J.; Wang, J.; Lin, Y.; Ren, X.; et al. Gastrointestinal disturbance and effect of fecal microbiota transplantation in discharged COVID-19 patients. J. Med. Case Rep. 2021, 15, 60. [Google Scholar] [CrossRef] [PubMed]
- Million, M.; Armstrong, N.; Khelaifia, S.; Guilhot, E.; Richez, M.; Lagier, J.C.; Dubourg, G.; Chabriere, E.; Raoult, D. The Antioxidants Glutathione, Ascorbic Acid and Uric Acid Maintain Butyrate Production by Human Gut Clostridia in The Presence of Oxygen In Vitro. Sci. Rep. 2020, 10, 7705. [Google Scholar] [CrossRef] [PubMed]
- Xia, W.J.; Xu, M.L.; Yu, X.J.; Du, M.M.; Li, X.H.; Yang, T.; Li, L.; Li, Y.; Kang, K.B.; Su, Q.; et al. Antihypertensive effects of exercise involve reshaping of gut microbiota and improvement of gut-brain axis in spontaneously hypertensive rat. Gut Microbes 2021, 13, 1–24. [Google Scholar] [CrossRef]
- Li, Y.; Salih Ibrahim, R.M.; Chi, H.L.; Xiao, T.; Xia, W.J.; Li, H.B.; Kang, Y.M. Altered Gut Microbiota is Involved in the Anti-Hypertensive Effects of Vitamin C in Spontaneously Hypertensive Rat. Mol. Nutr. food Res. 2021, 65, e2000885. [Google Scholar] [CrossRef] [PubMed]
- Gavioli, E.M.; Miyashita, H.; Hassaneen, O.; Siau, E. An Evaluation of Serum 25-Hydroxy Vitamin D Levels in Patients with COVID-19 in New York City. J. Am. Coll. Nutr. 2021. [Google Scholar] [CrossRef]
- Katz, J.; Yue, S.; Xue, W. Increased risk for COVID-19 in patients with vitamin D deficiency. Nutrition 2021, 84, 111106. [Google Scholar] [CrossRef]
- Munshi, R.; Hussein, M.H.; Toraih, E.A.; Elshazli, R.M.; Jardak, C.; Sultana, N.; Youssef, M.R.; Omar, M.; Attia, A.S.; Fawzy, M.S.; et al. Vitamin D insufficiency as a potential culprit in critical COVID-19 patients. J. Med. Virol. 2021, 93, 733–740. [Google Scholar] [CrossRef]
- Bakke, D.; Sun, J. Ancient Nuclear Receptor VDR With New Functions: Microbiome and Inflammation. Inflamm. Bowel Dis. 2018, 24, 1149–1154. [Google Scholar] [CrossRef] [Green Version]
- Wu, S.; Zhang, Y.G.; Lu, R.; Xia, Y.; Zhou, D.; Petrof, E.O.; Claud, E.C.; Chen, D.; Chang, E.B.; Carmeliet, G.; et al. Intestinal epithelial vitamin D receptor deletion leads to defective autophagy in colitis. Gut 2015, 64, 1082–1094. [Google Scholar] [CrossRef]
- Charoenngam, N.; Shirvani, A.; Kalajian, T.A.; Song, A.; Holick, M.F. The Effect of Various Doses of Oral Vitamin D(3) Supplementation on Gut Microbiota in Healthy Adults: A Randomized, Double-blinded, Dose-response Study. Anticancer Res. 2020, 40, 551–556. [Google Scholar] [CrossRef]
- Qin, C.; Xie, Y.; Wang, Y.; Li, S.; Ran, C.; He, S.; Zhou, Z. Impact of Lactobacillus casei BL23 on the Host Transcriptome, Growth and Disease Resistance in Larval Zebrafish. Front. Physiol. 2018, 9, 1245. [Google Scholar] [CrossRef]
- Lu, R.; Shang, M.; Zhang, Y.G.; Jiao, Y.; Xia, Y.; Garrett, S.; Bakke, D.; Bäuerl, C.; Martinez, G.P.; Kim, C.H.; et al. Lactic Acid Bacteria Isolated From Korean Kimchi Activate the Vitamin D Receptor-autophagy Signaling Pathways. Inflamm. Bowel Dis. 2020, 26, 1199–1211. [Google Scholar] [CrossRef]
- Wu, S.; Yoon, S.; Zhang, Y.G.; Lu, R.; Xia, Y.; Wan, J.; Petrof, E.O.; Claud, E.C.; Chen, D.; Sun, J. Vitamin D receptor pathway is required for probiotic protection in colitis. Am. J. Physiol. Gastrointest. Liver Physiol. 2015, 309, G341–G349. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.G.; Lu, R.; Xia, Y.; Zhou, D.; Petrof, E.; Claud, E.C.; Sun, J. Lack of Vitamin D Receptor Leads to Hyperfunction of Claudin-2 in Intestinal Inflammatory Responses. Inflamm. Bowel Dis. 2019, 25, 97–110. [Google Scholar] [CrossRef]
- Morniroli, D.; Consales, A.; Crippa, B.L.; Vizzari, G.; Ceroni, F.; Cerasani, J.; Colombo, L.; Mosca, F.; Giannì, M.L. The Antiviral Properties of Human Milk: A Multitude of Defence Tools from Mother Nature. Nutrients 2021, 13, 694. [Google Scholar] [CrossRef]
- Chang, R.; Ng, T.B.; Sun, W.Z. Lactoferrin as potential preventative and adjunct treatment for COVID-19. Int. J. Antimicrob. Agents 2020, 56, 106118. [Google Scholar] [CrossRef] [PubMed]
- Salaris, C.; Scarpa, M.; Elli, M.; Bertolini, A.; Guglielmetti, S.; Pregliasco, F.; Blandizzi, C.; Brun, P.; Castagliuolo, I. Protective Effects of Lactoferrin against SARS-CoV-2 Infection In Vitro. Nutrients 2021, 13, 328. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Meng, X.; Zhang, F.; Xiang, Y.; Wang, J. The in vitro antiviral activity of lactoferrin against common human coronaviruses and SARS-CoV-2 is mediated by targeting the heparan sulfate co-receptor. Emerg. Microbes Infect. 2021, 10, 317–330. [Google Scholar] [CrossRef]
- de Sá Almeida, J.S.; de Oliveira Marre, A.T.; Teixeira, F.L.; Boente, R.F.; Domingues, R.; de Paula, G.R.; Lobo, L.A. Lactoferrin and lactoferricin B reduce adhesion and biofilm formation in the intestinal symbionts Bacteroides fragilis and Bacteroides thetaiotaomicron. Anaerobe 2020, 64, 102232. [Google Scholar] [CrossRef]
- Silva, J.O.; Martins Reis, A.C.; Quesada-Gómez, C.; Pinheiro, A.Q.; Freire, R.S.; Oriá, R.B.; de Carvalho, C.B. In vitro effect of antibiotics on biofilm formation by Bacteroides fragilis group strains isolated from intestinal microbiota of dogs and their antimicrobial susceptibility. Anaerobe 2014, 28, 24–28. [Google Scholar] [CrossRef]
- Chatzidaki-Livanis, M.; Coyne, M.J.; Roelofs, K.G.; Gentyala, R.R.; Caldwell, J.M.; Comstock, L.E. Gut Symbiont Bacteroides fragilis Secretes a Eukaryotic-Like Ubiquitin Protein That Mediates Intraspecies Antagonism. mBio 2017, 8, e01902-17. [Google Scholar] [CrossRef] [Green Version]
- Husson, M.O.; Ley, D.; Portal, C.; Gottrand, M.; Hueso, T.; Desseyn, J.L.; Gottrand, F. Modulation of host defence against bacterial and viral infections by omega-3 polyunsaturated fatty acids. J. Infect. 2016, 73, 523–535. [Google Scholar] [CrossRef]
- Hathaway, D.; Pandav, K.; Patel, M.; Riva-Moscoso, A.; Singh, B.M.; Patel, A.; Min, Z.C.; Singh-Makkar, S.; Sana, M.K.; Sanchez-Dopazo, R.; et al. Omega 3 Fatty Acids and COVID-19: A Comprehensive Review. Infect. Chemother. 2020, 52, 478–495. [Google Scholar] [CrossRef]
- Schwarz, B.; Sharma, L.; Roberts, L.; Peng, X.; Bermejo, S.; Leighton, I.; Casanovas-Massana, A.; Minasyan, M.; Farhadian, S.; Ko, A.I.; et al. Cutting Edge: Severe SARS-CoV-2 Infection in Humans Is Defined by a Shift in the Serum Lipidome, Resulting in Dysregulation of Eicosanoid Immune Mediators. J. Immunol. 2021, 206, 329–334. [Google Scholar] [CrossRef]
- Hammock, B.D.; Wang, W.; Gilligan, M.M.; Panigrahy, D. Eicosanoids: The Overlooked Storm in Coronavirus Disease 2019 (COVID-19)? Am. J. Pathol. 2020, 190, 1782–1788. [Google Scholar] [CrossRef]
- Vivar-Sierra, A.; Araiza-Macías, M.J.; Hernández-Contreras, J.P.; Vergara-Castañeda, A.; Ramírez-Vélez, G.; Pinto-Almazán, R.; Salazar, J.R.; Loza-Mejía, M.A. In Silico Study of Polyunsaturated Fatty Acids as Potential SARS-CoV-2 Spike Protein Closed Conformation Stabilizers: Epidemiological and Computational Approaches. Molecules 2021, 26, 711. [Google Scholar] [CrossRef]
- Yu, H.N.; Zhu, J.; Pan, W.S.; Shen, S.R.; Shan, W.G.; Das, U.N. Effects of fish oil with a high content of n-3 polyunsaturated fatty acids on mouse gut microbiota. Arch. Med. Res. 2014, 45, 195–202. [Google Scholar] [CrossRef]
- Noriega, B.S.; Sanchez-Gonzalez, M.A.; Salyakina, D.; Coffman, J. Understanding the Impact of Omega-3 Rich Diet on the Gut Microbiota. Case. Rep. Med. 2016, 2016, 3089303. [Google Scholar] [CrossRef] [Green Version]
- Davis, D.J.; Hecht, P.M.; Jasarevic, E.; Beversdorf, D.Q.; Will, M.J.; Fritsche, K.; Gillespie, C.H. Sex-specific effects of docosahexaenoic acid (DHA) on the microbiome and behavior of socially-isolated mice. Brain Behav. Immun. 2017, 59, 38–48. [Google Scholar] [CrossRef]
- Ghosh, S.; DeCoffe, D.; Brown, K.; Rajendiran, E.; Estaki, M.; Dai, C.; Yip, A.; Gibson, D.L. Fish oil attenuates omega-6 polyunsaturated fatty acid-induced dysbiosis and infectious colitis but impairs LPS dephosphorylation activity causing sepsis. PLoS ONE 2013, 8, e55468. [Google Scholar] [CrossRef]
- Watson, H.; Mitra, S.; Croden, F.C.; Taylor, M.; Wood, H.M.; Perry, S.L.; Spencer, J.A.; Quirke, P.; Toogood, G.J.; Lawton, C.L.; et al. A randomised trial of the effect of omega-3 polyunsaturated fatty acid supplements on the human intestinal microbiota. Gut 2018, 67, 1974–1983. [Google Scholar] [CrossRef]
- Zhu, L.; Sha, L.; Li, K.; Wang, Z.; Wang, T.; Li, Y.; Liu, P.; Dong, X.; Dong, Y.; Zhang, X.; et al. Dietary flaxseed oil rich in omega-3 suppresses severity of type 2 diabetes mellitus via anti-inflammation and modulating gut microbiota in rats. Lipids Health Dis. 2020, 19, 20. [Google Scholar] [CrossRef] [Green Version]
- Zeng, H.L.; Zhang, B.; Wang, X.; Yang, Q.; Cheng, L. Urinary trace elements in association with disease severity and outcome in patients with COVID-19. Environ. Res. 2021, 194, 110670. [Google Scholar] [CrossRef]
- Read, S.A.; Obeid, S.; Ahlenstiel, C.; Ahlenstiel, G. The Role of Zinc in Antiviral Immunity. Adv. Nutr. 2019, 10, 696–710. [Google Scholar] [CrossRef] [Green Version]
- Costagliola, G.; Spada, E.; Comberiati, P.; Peroni, D.G. Could nutritional supplements act as therapeutic adjuvants in COVID-19? Ital. J. Pediatr. 2021, 47, 32. [Google Scholar] [CrossRef]
- Reed, S.; Neuman, H.; Moscovich, S.; Glahn, R.P.; Koren, O.; Tako, E. Chronic Zinc Deficiency Alters Chick Gut Microbiota Composition and Function. Nutrients 2015, 7, 5497. [Google Scholar] [CrossRef]
- Shao, Y.; Lei, Z.; Yuan, J.; Yang, Y.; Guo, Y.; Zhang, B. Effect of zinc on growth performance, gut morphometry, and cecal microbial community in broilers challenged with Salmonella enterica serovar typhimurium. J. Microbiol. 2014, 52, 1002–1011. [Google Scholar] [CrossRef]
- Feng, D.; Zhang, M.; Tian, S.; Wang, J.; Zhu, W. Chitosan-chelated zinc modulates cecal microbiota and attenuates inflammatory response in weaned rats challenged with Escherichia coli. J. Microbiol. 2020, 58, 780–792. [Google Scholar] [CrossRef]
- Zackular, J.P.; Moore, J.L.; Jordan, A.T.; Juttukonda, L.J.; Noto, M.J.; Nicholson, M.R.; Crews, J.D.; Semler, M.W.; Zhang, Y.; Ware, L.B.; et al. Dietary zinc alters the microbiota and decreases resistance to Clostridium difficile infection. Nat. Med. 2016, 22, 1330–1334. [Google Scholar] [CrossRef] [PubMed]
- Rayman, M.P. Selenium and human health. Lancet 2012, 379, 1256–1268. [Google Scholar] [CrossRef]
- Huang, Z.; Rose, A.H.; Hoffmann, P.R. The role of selenium in inflammation and immunity: From molecular mechanisms to therapeutic opportunities. Antioxid. Redox Signal. 2012, 16, 705–743. [Google Scholar] [CrossRef] [Green Version]
- Avery, J.C.; Hoffmann, P.R. Selenium, Selenoproteins, and Immunity. Nutrients 2018, 10, 1203. [Google Scholar] [CrossRef] [Green Version]
- Gangadoo, S.; Dinev, I.; Chapman, J.; Hughes, R.J.; Van, T.T.H.; Moore, R.J.; Stanley, D. Selenium nanoparticles in poultry feed modify gut microbiota and increase abundance of Faecalibacterium prausnitzii. Appl. Microbiol. Biotechnol. 2018, 102, 1455–1466. [Google Scholar] [CrossRef]
- Wei, J.Y.; Wang, J.; Liu, W.; Zhang, K.Z.; Sun, P. Short communication: Effects of different selenium supplements on rumen fermentation and apparent nutrient and selenium digestibility of mid-lactation dairy cows. J. Dairy Sci. 2019, 102, 3131–3135. [Google Scholar] [CrossRef] [Green Version]
- DiNicolantonio, J.J.; O’Keefe, J.H. Magnesium and Vitamin D Deficiency as a Potential Cause of Immune Dysfunction, Cytokine Storm and Disseminated Intravascular Coagulation in covid-19 patients. Mol. Med. 2021, 118, 68–73. [Google Scholar]
- Serefko, A.; Szopa, A.; Poleszak, E. Magnesium and depression. Magnes Res. 2016, 29, 112–119. [Google Scholar] [CrossRef]
- Veronese, N.; Zanforlini, B.M.; Manzato, E.; Sergi, G. Magnesium and healthy aging. Magnes Res 2015, 28, 112–115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malpuech-Brugère, C.; Nowacki, W.; Daveau, M.; Gueux, E.; Linard, C.; Rock, E.; Lebreton, J.; Mazur, A.; Rayssiguier, Y. Inflammatory response following acute magnesium deficiency in the rat. Biochim. Biophys. Acta 2000, 1501, 91–98. [Google Scholar] [CrossRef] [Green Version]
- Scanlan, B.J.; Tuft, B.; Elfrey, J.E.; Smith, A.; Zhao, A.; Morimoto, M.; Chmielinska, J.J.; Tejero-Taldo, M.I.; Mak Iu, T.; Weglicki, W.B.; et al. Intestinal inflammation caused by magnesium deficiency alters basal and oxidative stress-induced intestinal function. Mol. Cell. Biochem. 2007, 306, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Pyndt Jørgensen, B.; Winther, G.; Kihl, P.; Nielsen, D.S.; Wegener, G.; Hansen, A.K.; Sørensen, D.B. Dietary magnesium deficiency affects gut microbiota and anxiety-like behaviour in C57BL/6N mice. Acta Neuropsychiatr. 2015, 27, 307–311. [Google Scholar] [CrossRef] [PubMed]
- Pachikian, B.D.; Neyrinck, A.M.; Deldicque, L.; De Backer, F.C.; Catry, E.; Dewulf, E.M.; Sohet, F.M.; Bindels, L.B.; Everard, A.; Francaux, M.; et al. Changes in intestinal bifidobacteria levels are associated with the inflammatory response in magnesium-deficient mice. J. Nutr. 2010, 140, 509–514. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- García-Legorreta, A.; Soriano-Pérez, L.A.; Flores-Buendía, A.M.; Medina-Campos, O.N.; Noriega, L.G.; Granados-Portillo, O.; Nambo-Venegas, R.; Tovar, A.R.; Mendoza-Vargas, A.; Barrera-Oviedo, D.; et al. Effect of Dietary Magnesium Content on Intestinal Microbiota of Rats. Nutrients 2020, 12, 2889. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).