The Impact of COVID-19 and the Pandemic on Tinnitus: A Systematic Review
Abstract
:1. Introduction
2. Materials and Methods
2.1. Protocol and Registration
2.2. Eligibility Criteria
2.3. Information Sources
2.4. Search Strategy
2.5. Data Management and Study Selection
2.6. Data Collection Process and Data Items
2.7. Risk of Bias Assessment
2.8. Strategy for Data Synthesis
2.9. Subgroup Analysis
3. Results
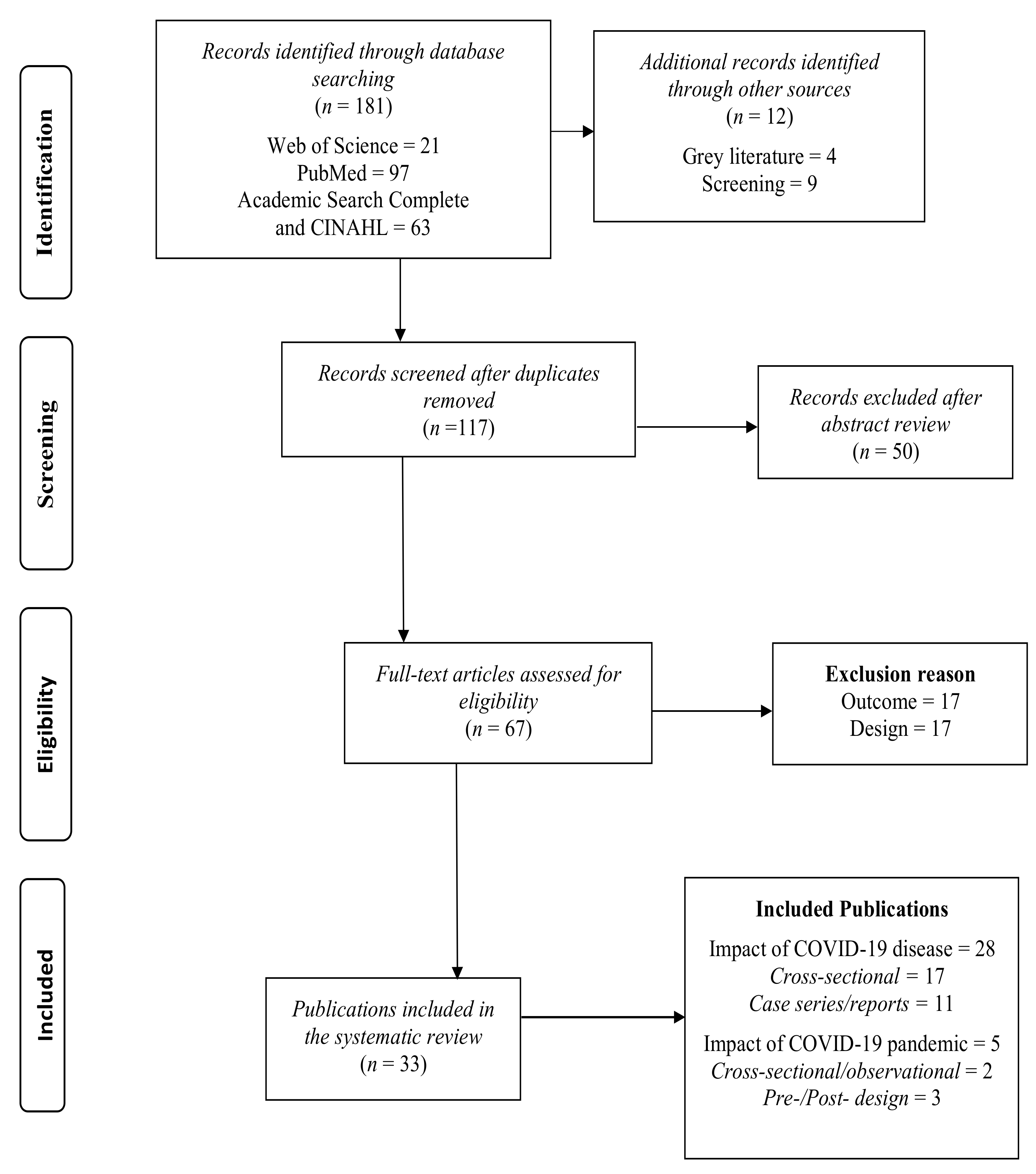
3.1. Study Selection
3.2. Study Characteristics
3.3. Risk of Bias in the Individual Studies
3.4. Case Reports/Case Series Disease Impact Studies
3.4.1. Study Characteristics
3.4.2. Pre-Existing Health Conditions
3.4.3. Tinnitus Characteristics
3.4.4. Tinnitus Initiation
3.4.5. Tinnitus Persistence or Recovery
3.4.6. Hearing Loss
3.4.7. Vestibular Impairment
3.4.8. COVID-19 Testing
3.4.9. Treatment of COVID-19
3.4.10. Quality Analysis of Case Reports
3.5. Cross-Sectional Studies Investigating Disease Impact
3.5.1. Study Characteristics
3.5.2. Study Designs and Outcomes
3.5.3. Pre-Existing Health Conditions
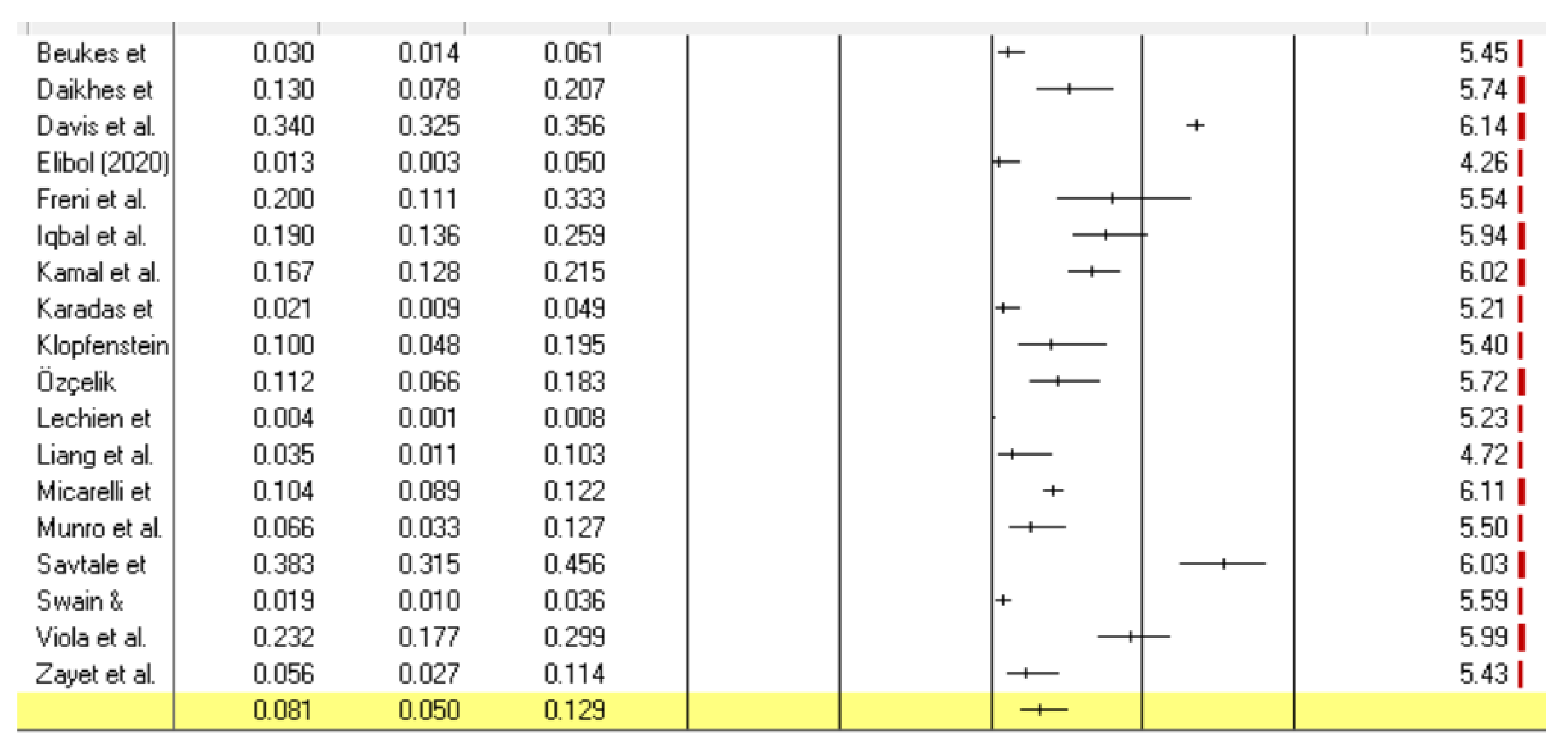
3.5.4. Tinnitus Overview
3.5.5. Tinnitus Characteristic
3.5.6. Hearing Loss
3.5.7. Vestibular Deficits
3.5.8. COVID-19 Testing
3.5.9. Treatment of COVID-19
3.5.10. Quality Analysis of Cross-Sectional Observational Studies
3.6. Pandemic Impact Studies: Comparing Tinnitus before and during the Pandemic
3.6.1. Study Characteristics
3.6.2. Tinnitus Characteristics
3.6.3. Tinnitus Treatments
3.6.4. Quality Analysis of Pandemic Impact Study Comparing Tinnitus before and during the Pandemic
3.7. Cross-Sectional Studies Investigating the Effects of the Pandemic on Pre-existing Tinnitus
3.7.1. Study Characteristics
3.7.2. Outcome Measures
3.7.3. Individual Study Descriptions
3.7.4. Quality Analysis of Cross-Sectional Studies Investigating Effects of the Pandemic on Pre-Existing Tinnitus
4. Discussion
4.1. The Effect of Contracting COVID-19 on Tinnitus
4.2. Characteristics of Other Auditory Vestibular Conditions
4.3. The Impact of the Pandemic on Tinnitus Experiences
4.4. Limitations of the Evidence and Review Process
4.5. Implications for Practice, Policy, and Future Research
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organization. Coronavirus Disease (COVID-19) Pandemic. Available online: https://www.who.int/csr/don/05-january-2020-pneumonia-ofunkown-cause-china/en/ (accessed on 26 March 2021).
- Hopkins, C.; Surda, P.; Whitehead, E.; Kumar, B.N. Early recovery following new onset anosmia during the COVID-19 pandemic—An observational cohort study. J. Otolaryngol. Head Neck Surg. 2020, 49, 1–6. [Google Scholar] [CrossRef]
- Guan, W.J.; Ni, Z.Y.; Hu, Y.; Liang, W.H.; Ou, C.Q.; He, J.X.; Liu, L.; Shan, H.; Lei, C.L.; Hui, D.S.C.; et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef] [PubMed]
- National Institute for Health and Care Excellence (NICE). COVID-19 Rapid Guideline: Managing the Long-Term Effects of COVID-19. Available online: https://www.nice.org.uk/guidance/ng188/resources/covid19-rapid-guideline-managing-thelongterm-effects-of-covid19-pdf-66142028400325 (accessed on 28 April 2021).
- Cohen, B.E.; Durstenfeld, A.; Roehm, P.C. Viral Causes of Hearing Loss: A Review for Hearing Health Professionals. Trends Hear. 2014, 18. [Google Scholar] [CrossRef]
- Young, Y.-H. Contemporary review of the causes and differential diagnosis of sudden sensorineural hearing loss. Int. J. Audiol. 2019, 59, 243–253. [Google Scholar] [CrossRef] [PubMed]
- Almufarrij, I.; Uus, K.; Munro, K.J. Does coronavirus affect the audio-vestibular system? A rapid systematic review. Int. J. Audiol. 2020, 59, 487–491. [Google Scholar] [CrossRef] [PubMed]
- Saniasiaya, J. Hearing Loss in SARS-CoV-2: What Do We Know? Ear. Nose Throat J. 2021, 100, 152S–154S. [Google Scholar] [CrossRef] [PubMed]
- Maharaj, S.; Alvarez, M.B.; Mungul, S.; Hari, K. Otologic dysfunction in patients with COVID-19: A systematic review. Laryngoscope 2020, 5, 1192–1196. [Google Scholar] [CrossRef]
- Almufarrij, I.; Munro, K.J. One year on: An updated systematic review of SARS-CoV-2, COVID-19 and audio-vestibular symptoms. Int. J. Audiol. 2021, 1–11. [Google Scholar] [CrossRef]
- Jafari, Z.; Kolb, B.E.; Mohajerani, M.H. Hearing Loss, Tinnitus, and Dizziness in COVID-19: A Systematic Review and Meta-Analysis. Can. J. Neurol. Sci. J. Can. Des Sci. Neurol. 2021, 1–33. [Google Scholar] [CrossRef]
- Lewnard, J.A.; Lo, N.C. Scientific and ethical basis for social-distancing interventions against COVID-19. Lancet Infect. Dis. 2020, 20, 631–633. [Google Scholar] [CrossRef] [Green Version]
- Cénat, J.M.; Blais-Rochette, C.; Kokou-Kpolou, C.K.; Noorishad, P.-G.; Mukunzi, J.N.; McIntee, S.-E.; Dalexis, R.D.; Goulet, M.-A.; Labelle, P.R. Prevalence of symptoms of depression, anxiety, insomnia, posttraumatic stress disorder, and psychological distress among populations affected by the COVID-19 pandemic: A systematic review and meta-analysis. Psychiatry Res. 2021, 295, 113599. [Google Scholar] [CrossRef]
- Kocevska, D.; Blanken, T.F.; Van Someren, E.J.; Rösler, L. Sleep quality during the COVID-19 pandemic: Not one size fits all. Sleep Med. 2020, 76, 86–88. [Google Scholar] [CrossRef] [PubMed]
- Vindegaard, N.; Benros, M.E. COVID-19 pandemic and mental health consequences: Systematic review of the current evidence. Brain Behav. Immun. 2020, 89, 531–542. [Google Scholar] [CrossRef] [PubMed]
- Xiong, J.; Lipsitz, O.; Nasri, F.; Lui, L.M.W.; Gill, H.; Phan, L.; Chen-Li, D.; Iacobucci, M.; Ho, R.; Majeed, A.; et al. Impact of COVID-19 pandemic on mental health in the general population: A systematic review. J. Affect. Disord. 2020, 277, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Mazurek, B.; Boecking, B.; Brueggemann, P. Association between Stress and Tinnitus—New Aspects. Otol. Neurotol. 2019, 40, e467–e473. [Google Scholar] [CrossRef]
- Beukes, E.W.; Baguley, D.M.; Jacquemin, L.; Lourenco, M.P.C.G.; Allen, P.M.; Onozuka, J.; Stockdale, D.; Kaldo, V.; Andersson, G.; Manchaiah, V. Changes in Tinnitus Experiences During the COVID-19 Pandemic. Front. Public Health 2020, 8, 592878. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. BMJ 2009, 339, 332–336. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johnson, N.; Phillips, M. Rayyan for systematic reviews. J. Electron. Resour. Librariansh. 2018, 30, 46–48. [Google Scholar] [CrossRef] [Green Version]
- Chirakkal, P.; Al Hail, A.N.; Zada, N.; Vijayakumar, D.S. COVID-19 and Tinnitus. Ear Nose Throat J. 2021, 100, 160S–162S. [Google Scholar] [CrossRef]
- Cui, C.; Yao, Q.; Zhang, D.; Zhao, Y.; Zhang, K.; Nisenbaum, E.; Cao, P.; Zhao, K.; Huang, X.; Leng, D.; et al. Approaching Otolaryngology Patients During the COVID-19 Pandemic. Otolaryngol. Neck Surg. 2020, 163, 121–131. [Google Scholar] [CrossRef]
- Degen, C.; Lenarz, T.; Willenborg, K. Acute Profound Sensorineural Hearing Loss after COVID-19 Pneumonia. Mayo Clin. Proc. 2020, 95, 1801–1803. [Google Scholar] [CrossRef]
- Fidan, V. New type of corona virus induced acute otitis media in adult. Am. J. Otolaryngol. 2020, 41, 102487. [Google Scholar] [CrossRef] [PubMed]
- Karimi-Galougahi, M.; Naeini, A.S.; Raad, N.; Mikaniki, N.; Ghorbani, J. Vertigo and hearing loss during the COVID-19 pandemic–is there an association? Acta Otorhinolaryngol. Ital. 2020, 40, 463–465. [Google Scholar] [CrossRef]
- Koumpa, F.S.; Forde, C.T.; Manjaly, J.G. Sudden irreversible hearing loss post COVID-19. BMJ Case Rep. 2020, 13, e238419. [Google Scholar] [CrossRef] [PubMed]
- Lamounier, P.; Franco, V.; Ramos, H.; Gobbo, D.A.; Teixeira, R.P.; Dos Reis, P.C.; Bahmad, F., Jr.; Costa, C.C. A 67-Year-Old Woman with Sudden Hearing Loss Associated with SARS-CoV-2 Infection. Am. J. Case Rep. 2020, 21, e927519-1–e927519-6. [Google Scholar] [CrossRef]
- Lang, B.; Hintze, J.; Conlon, B. Coronavirus disease 2019 and sudden sensorineural hearing loss. J. Laryngol. Otol. 2020, 134, 1026–1028. [Google Scholar] [CrossRef] [PubMed]
- Maharaj, S.; Hari, K. Congenital Inner Ear Abnormalities and COVID-19-Related Ear Infections. Ear Nose Throat J. 2020. [Google Scholar] [CrossRef] [PubMed]
- Rhman, S.A.; Wahid, A.A. COVID -19 and sudden sensorineural hearing loss, a case report. Otolaryngol. Case Rep. 2020, 16, 100198. [Google Scholar] [CrossRef]
- Sun, R.; Liu, H.; Wang, X. Mediastinal Emphysema, Giant Bulla, and Pneumothorax Developed during the Course of COVID-19 Pneumonia. Korean J. Radiol. 2020, 21, 541–544. [Google Scholar] [CrossRef] [Green Version]
- Daikhes, N.; Карнеева, О.В.; Machalov, A.; Kuznetcov, A.; Sapozhnikov, Y.; Балакина, A.B.; Khulugurova, L.; Карпoв, V.L. Audiological profile of patients with SARS-Co-V-2 PCR-positive cases. Vestnik Otorinolaringol. 2020, 85, 6–11. [Google Scholar] [CrossRef]
- Davis, H.E.; Assaf, G.S.; McCorkell, L.; Wei, H.; Low, R.J.; Re’em, Y.; Redfield, S.; Austin, J.P.; Akrami, A. Characterizing long COVID in an international cohort: 7 months of symptoms and their impact. medRxiv 2020. [Google Scholar] [CrossRef]
- Elibol, E. Otolaryngological symptoms in COVID-19. Eur. Arch. Oto-Rhino-Laryngol. 2021, 278, 1233–1236. [Google Scholar] [CrossRef] [PubMed]
- Freni, F.; Meduri, A.; Gazia, F.; Nicastro, V.; Galletti, C.; Aragona, P.; Galletti, B.; Galletti, F. Symptomatology in head and neck district in coronavirus disease (COVID-19): A possible neuroinvasive action of SARS-CoV-2. Am. J. Otolaryngol. 2020, 41, 102612. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, A.; Iqbal, K.; Ali, S.A.; Azim, D.; Farid, E.; Baig, M.D.; Bin Arif, T.; Raza, M. The COVID-19 Sequelae: A Cross-Sectional Evaluation of Post-recovery Symptoms and the Need for Rehabilitation of COVID-19 Survivors. Cureus 2021, 13. [Google Scholar] [CrossRef]
- Kamal, M.; Omirah, M.A.; Hussein, A.; Saeed, H. Assessment and characterisation of post-COVID-19 manifestations. Int. J. Clin. Pract. 2021, 75. [Google Scholar] [CrossRef] [PubMed]
- Karadaş, Ö.; Öztürk, B.; Sonkaya, A.R. A prospective clinical study of detailed neurological manifestations in patients with COVID-19. Neurol. Sci. 2020, 41, 1991–1995. [Google Scholar] [CrossRef]
- Klopfenstein, T.; Zahra, H.; Kadiane-Oussou, N.J.; Lepiller, Q.; Royer, P.-Y.; Toko, L.; Gendrin, V.; Zayet, S. New loss of smell and taste: Uncommon symptoms in COVID-19 patients on Nord Franche-Comte cluster, France. Int. J. Infect. Dis. 2020, 100, 117–122. [Google Scholar] [CrossRef]
- Özçelik Korkmaz, M.; Eğilmez, O.K.; Özçelik, M.A.; Güven, M. Otolaryngological manifestations of hospitalised patients with confirmed COVID-19 infection. Eur. Arch. Oto-Rhino-Laryngol. 2021, 278, 1675–1685. [Google Scholar] [CrossRef]
- Lechien, J.R.; Chiesa-Estomba, C.M.; Place, S.; Laethem, Y.V.; Cabaraux, P.; Mat, Q.; Huet, K.; Plzak, J.; Horoi, M.; Hans, S.; et al. Clinical and epidemiological characteristics of 1420 European patients with mild-to-moderate coronavirus disease 2019. J. Intern. Med. 2020, 288, 335–344. [Google Scholar] [CrossRef]
- Liang, Y.; Xu, J.; Chu, M.; Mai, J.; Lai, N.; Tang, W.; Yang, T.; Zhang, S.; Guan, C.; Zhong, F.; et al. Neurosensory dysfunction: A diagnostic marker of early COVID-19. Int. J. Infect. Dis. 2020, 98, 347–352. [Google Scholar] [CrossRef]
- Micarelli, A.; Granito, I.; Carlino, P.; Micarelli, B.; Alessandrini, M. Self-perceived general and ear-nose-throat symptoms related to the COVID-19 outbreak: A survey study during quarantine in Italy. J. Int. Med. Res. 2020, 48. [Google Scholar] [CrossRef]
- Munro, K.J.; Uus, K.; Almufarrij, I.; Chaudhuri, N.; Yioe, V. Persistent self-reported changes in hearing and tinnitus in post-hospitalisation COVID-19 cases. Int. J. Audiol. 2020, 59, 889–890. [Google Scholar] [CrossRef]
- Savtale, S.; Hippargekar, P.; Bhise, S.; Kothule, S. Prevalence of Otorhinolaryngological Symptoms in Covid 19 Patients. Indian J. Otolaryngol. Head Neck Surg. 2021, 1–7. [Google Scholar] [CrossRef]
- Swain, S.K.; Pani, S.R. Incidence of Hearing Loss in COVID-19 Patients: A COVID Hospital-based Study in the Eastern Part of India. Int. J. Curr. Res. Rev. 2021, 13, 103–107. [Google Scholar] [CrossRef]
- Viola, P.; Ralli, M.; Pisani, D.; Malanga, D.; Sculco, D.; Messina, L.; Laria, C.; Aragona, T.; Leopardi, G.; Ursini, F.; et al. Tinnitus and equilibrium disorders in COVID-19 patients: Preliminary results. Eur. Arch. Oto-Rhino-Laryngol. 2020, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Zayet, S.; Kadiane-Oussou, N.J.; Lepiller, Q.; Zahra, H.; Royer, P.-Y.; Toko, L.; Gendrin, V.; Klopfenstein, T. Clinical features of COVID-19 and influenza: A comparative study on Nord Franche-Comte cluster. Microbes Infect. 2020, 22, 481–488. [Google Scholar] [CrossRef] [PubMed]
- Naylor, G.; Burke, L.A.; Holman, J.A. Covid-19 Lockdown Affects Hearing Disability and Handicap in Diverse Ways: A Rapid Online Survey Study. Ear Hear. 2020, 41, 1442–1449. [Google Scholar] [CrossRef]
- Anzivino, R.; Sciancalepore, P.I.; Petrone, P.; D’Elia, A.; Petrone, D.; Quaranta, N. Tinnitus revival during COVID-19 lockdown: How to deal with it? Eur. Arch. Oto-Rhino-Laryngol. 2021, 278, 295–296. [Google Scholar] [CrossRef]
- Schlee, W.; Hølleland, S.; Bulla, J.; Simoes, J.; Neff, P.; Schoisswohl, S.; Woelflick, S.; Schecklmann, M.; Schiller, A.; Staudinger, S.; et al. The Effect of Environmental Stressors on Tinnitus: A Prospective Longitudinal Study on the Impact of the COVID-19 Pandemic. J. Clin. Med. 2020, 9, 2756. [Google Scholar] [CrossRef]
- Xia, L.; He, G.; Feng, Y.; Yu, X.; Zhao, X.; Chen, Z.; Yin, S.; Wang, J.; Fan, J.; Dong, C. Impact of anxiety associated with COVID-19 on tinnitus. medRxiv 2020. [Google Scholar] [CrossRef]
- National Institutes of Health (NIH). Study quality assessment tools. National Heart, Lung, and Blood Institute. Available online: https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools (accessed on 2 February 2021).
- Campbell, M.; McKenzie, J.E.; Sowden, A.; Katikireddi, S.V.; Brennan, S.E.; Ellis, S.; Hartmann-Boyce, J.; Ryan, R.; Shepperd, S.; Thomas, J.; et al. Synthesis without meta-analysis (SWiM) in systematic reviews: Reporting guideline. BMJ 2020, 368, l6890. [Google Scholar] [CrossRef] [Green Version]
- Popay, J.; Roberts, H.; Sowden, A.; Petticrew, M.; Arai, L.; Rodgers, M.; Britten, N.; Roen, K.; Duffy, S. Guidance on the Conduct of Narrative Synthesis in Systematic Reviews; A product from the ESRC methods programme Version 1; Institute for Health Research: London, UK, 2006. [Google Scholar]
- Borenstein, M.; Hedges, L.; Higgins, J.; Rothstein, H. Comprehensive Meta-Analysis Version 3.0; Biostat: Englewood, NJ, USA, 2013. [Google Scholar]
- Borenstein, M.; Hedges, L.; Rothstein, H. Meta-Analysis: Fixed Effect vs. Random Effects. Meta-analysis.com. 2007. Available online: https://www.meta-analysis.com/downloads/M-af_e_v_r_e_sv.pdf (accessed on 2 February 2021).
- McCormack, A.; Edmondson-Jones, M.; Somerset, S.; Hall, D. A systematic review of the reporting of tinnitus prevalence and severity. Hear. Res. 2016, 337, 70–79. [Google Scholar] [CrossRef]
- Mustafa, M. Audiological profile of asymptomatic Covid-19 PCR-positive cases. Am. J. Otolaryngol. 2020, 41, 102483. [Google Scholar] [CrossRef]
- Harenberg, J.; Jonas, J.B.; Trecca, E.M.C. A Liaison between Sudden Sensorineural Hearing Loss and SARS-CoV-2 Infection. Thromb. Haemost. 2020, 120, 1237–1239. [Google Scholar] [CrossRef] [PubMed]
- Cure, E.; Cure, M.C. Comment on “Hearing loss and COVID-19: A note”. Am. J. Otolaryngol. 2020, 41, 102513. [Google Scholar] [CrossRef]
- Beukes, E.W.; Onozuka, J.; Brazell, T.P.; Manchaiah, V. Coping with Tinnitus during the COVID-19 Pandemic. Am. J. Audiol. 2021, 1–9. [Google Scholar] [CrossRef]
- Beukes, E.W.; Lourenco, M.P.C.G.; Biot, L.; Andersson, G.; Kaldo, V.; Manchaiah, V.; Jacquemin, L. Suggestions for shaping tinnitus service provision in Western Europe: Lessons from the COVID-19 pandemic. Int. J. Clin. Pract. 2021, e14196. [Google Scholar] [CrossRef]
- Manchaiah, V.; Eikelboom, R.H.; Bennett, B.J.; Swanepoel, D.W. International survey of audiologists during the COVID-19 pandemic: Effects on the workplace. Int. J. Audiol. 2021, 1–8. [Google Scholar] [CrossRef]
- Beukes, E.W.; Baguley, D.M.; Allen, P.M.; Manchaiah, V.; Andersson, G. Audiologist-Guided Internet-Based Cognitive Behavior Therapy for Adults with Tinnitus in the United Kingdom: A Randomized Controlled Trial. Ear Hear. 2018, 39, 423–433. [Google Scholar] [CrossRef] [Green Version]
- Beukes, E.W.; Andersson, G.; Allen, P.M.; Manchaiah, V.; Baguley, D.M. Effectiveness of Guided Internet-Based Cognitive Behavioral Therapy vs Face-to-Face Clinical Care for Treatment of Tinnitus. JAMA Otolaryngol. Neck Surg. 2018, 144, 1126–1133. [Google Scholar] [CrossRef] [Green Version]
- Beukes, E.W.; Manchaiah, V.; Allen, P.M.; Baguley, D.M.; Andersson, G. Internet-Based Interventions for Adults with Hearing Loss, Tinnitus, and Vestibular Disorders: A Systematic Review and Meta-Analysis. Trends Hear. 2019, 23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beukes, E.W.; Andersson, G.; Fagelson, M.A.; Manchaiah, V. Dismantling internet-based cognitive behavioral therapy for tinnitus. The contribution of applied relaxation: A randomized controlled trial. Internet Interv. 2021, 25, 100402. [Google Scholar] [CrossRef] [PubMed]
- Trecca, E.M.; Gelardi, M.; Cassano, M. COVID-19 and hearing difficulties. Am. J. Otolaryngol. 2020, 41, 102496. [Google Scholar] [CrossRef] [PubMed]
- Ariapooran, S.; Khezeli, M. Correction to: Symptoms of anxiety disorders in Iranian adolescents with hearing loss during the COVID-19 pandemic. BMC Psychiatry 2021, 21, 1–5. [Google Scholar] [CrossRef]
- Ciorba, A.; Skarżyński, P.H.; Pelucchi, S.; Hatzopoulos, S. Ototoxicity prevention during the SARS-CoV-2 (COVID-19) emergency. J. Glob. Antimicrob. Resist. 2020, 23, 263–264. [Google Scholar] [CrossRef] [PubMed]
- Tserga, E.; Nandwani, T.; Edvall, N.K.; Bulla, J.; Patel, P.; Canlon, B.; Cederroth, C.R.; Baguley, D.M. The genetic vulnerability to cisplatin ototoxicity: A systematic review. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef] [Green Version]
| Inclusion | Exclusion | |
|---|---|---|
| Population | Individuals of any age experiencing tinnitus during the COVID-19 pandemic or due to contracting SARS-CoV-2 (COVID-19). | Individuals without tinnitus |
| Condition | Tinnitus, which is the perception of sound in the ears or head in the absence of any external sound. | No tinnitus |
| Exposure | Confirmed, probable, or suspected exposure to COVID-19 or the pandemic. | Not exposed to COVID-19 or the pandemic. |
| Comparator | Not applicable | Not applicable |
| Outcomes | Self-reported experiences of tinnitus | No tinnitus reports |
| Study designs | Any study designs, including commentaries and case studies | Systematic reviews, secondary studies discussing other studies |
| Timings | At least one time point | No exclusions regarding the length of follow up assessments |
| Language | All languages | None |
| Study | Country Study Period | Quality Assessment | Participant Characteristics | Other Audio-Vestibular Manifestations | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Publication Date | Participants (n = 35) | Gender | Age in Years (Mean, Median, Range) | Tinnitus | Hearing Loss | Vestibular Impairments | Taste Disorders | Smell Disorders | |||
| Chirakkal et al. [21] | State of Qatar Not stated | Fair | 04/12/2020 | n = 1 | M = 0% F = 100% | 35 | n = 1 (Ringing, matched at 4 kHz at 10 dBHL) | X | |||
| Cui et al. [22] | China 14/01/2020–20/30/2020 | Fair | 01/07/2020 | n = 20 | M = 55% F = 45% | NA, 63 32–72 | n = 1 | X | X | ||
| Degen et al. [23] | Germany Not stated | Fair | 01/08/2020 | n = 1 | M = 100% F = 0% | 60 | n = 1 (bilateral, described as loud white noise) | X | |||
| Fidan [24] | Turkey Not stated | Fair | 01/05/2020 | n = 1 | M = 0% F = 100% | 35 | n = 1 | X | |||
| Karimi-Galougahi et al. [25] | Iran Not stated | Fair | 10/06/2020 | n = 6 | M = 33% F = 67% | 32 | n = 4 | X | X | ||
| Koumpa et al. [26] | United Kingdom Not stated | Good | 13/10/2020 | n = 1 | M =100% F = 0% | 45 | n = 1 (left sided) | X | |||
| Lamounier et al. [27] | Brazil 12/03/2020–23/05/2020 | Good | 03/11/2020 | n = 1 | M = 0% F = 100% | 67 | n = 1 (disabling) | X | |||
| Lang et al. [28] | Ireland 19/04/2020–09/06/2020 | Fair | 01/10/2020 | n = 1 | M = 0% F = 100% | 30 | n = 1 (right-sided) | X | |||
| Maharaj & Hari [29] | Malaysia Not stated | Good | 23/10/2020 | n = 1 | M = 100% F = 0% | 44 | n = 1 “Right-sided non-pulsatile” | X | |||
| Abdel-Rhman and Abdel- Wahid [30] | Egypt 15/04/2020–05/2020 | Fair | 08/07/2020 | n = 1 | M = 100% F = 0% | 52 | n = 1 Gradually worsening | X | |||
| Sun et al. [31] | China 16/01/20202–24/02/2020 | Poor | 01/05/2020 | n = 1 | M = 100% F = 0% | 38 | n = 1 | X | |||
| Study | Country Study Period | Quality Assessment | Participant Characteristics | Other Audio-Vestibular Manifestations | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Publicat-ion Date | Participants (n = 8913) | Gender | Age in Years (Mean, Median, and Range) | Tinnitus | Hearing Loss | Vestibular Impairments | Taste Disorders | Smell Disorders | |||
| Daikhes et al. [32] | Russia 4/2020–6/2020 | Poor | 20/07/2020 | n = 108 (including n = 30 as a control) | Not provided | NA, 20–50, NA | n = 14 (17%) | X | |||
| Davis et al. [33] | International (39 countries) 9/6/2020–11/25/2020 | Fair | 26/12/2020 | n = 3762 | M = 19.1% F = 78.9% Other = 2% | NA, NA,30–60 | n = 1280 (34%) | X | X | X | X |
| Elibol [34] | Turkey 3/25/2020–4/25/2020 | Fair | 01/09/2020 | n = 155 | M = 41.3% F = 58.7% | 36.3 (8.1), NA, 18–72 | n = 2 (1.3%) | X | X | X | |
| Freni et al. [35] | Italy Not stated | Fair | 18/06/2020 | n = 50 (including n = 20 as a control) | M = 60% F = 40% | 37.7 (17.9), NA, 18–65 | n = 10 (20%) | X | X | X | |
| Iqbal et al. [36] | Pakistan 9/2020–12/2020 | Good | 02/02/2021 | n = 158 | M = 44.9% F = 55.1% | 32.1 (12.42), NA, 19–80 | n = 30 (19%) | X | X | ||
| Kamal et al. [37] | Egypt Not stated | Fair | 29/09/2020 | n = 287 | M = 35.9% F = 64.1% | 32.3 (8.5), NA, 20–60 | n = 48 (17%) | ||||
| Karadaş et al. [38] | Turkey | Fair | 25/06/2020 | n = 239 | M = 55.6% F= 44.4% | 46.46 (15.41), 19–88 | n = 5 (2.1%) | X | X | X | X |
| Klopfenstein et al. [39] | France 3/1/2020–3/14/2020 | Fair | 04/08/2020 | n = 70 | M = 33% F = 67% | 47 (16), NA, NA | n = 7 (10%) | X | X | X | |
| Özçelik Korkmaz et al. [40] | Turkey 4/2020–5/2020 | Fair | 03/10/2020 | n = 116 | M = 50% F = 50% | 57.4 (14.32), NA, 19–83 | n = 13 (11%) | X | X | X | X |
| Lechien et al. [41] | Europe 3/22/2020–4/10/2020 | Fair | 30/04/2020 | n = 1420 | M = 32.3% F = 67.7% | 39.17 (12.09), 37, NA | n = 5 (0.3%) | X | X | ||
| Liang et al. [42] | China 3/16/2020–4/12/2020 | Fair | 24/06/2020 | n = 86 | M = 51.2% F = 48.8% | NA, 25.5, 6–57 | n = 3 (3.5%) | X | X | ||
| Micarelli et al. [43] | Italy 3/23/2020–3/30/2020 | Fair | 20-Oct | n = 1380 | M = 39.3% F = 60.6% | NA, 23–72, NA | n = 144 (10%) | X | X | X | |
| Munro et al. [44] | UK Not stated | Fair | 31/07/2020 | n = 121 | M = 87.5% F = 12.5% (of those with a change in tinnitus/hearing) | NA, 64, 44–82 | n = 8 (7%) | X | |||
| Savtale et al. [45] | India 10/1/2020–10/15/2020 | Fair | 08/02/2021 | n= 180 | M= 33.4% F= 66.6% | 37.8 (12.5), NA, 18–65 | n = 120 (67%) | X | X | X | |
| Swain andPani [46] | India 3/2020–8/2020 | Fair | 03/02/2021 | n = 472 | M = 64.3% F = 35.7% | 28.2, NA, 16–52 | n = 9 (2%) | X | X | X | |
| Viola et al. [47] | Italy 5/5/2020–6/10/2020 | Fair | 23/10/2020 | n = 185 | M = 53.5% F = 46.5% | 52.15 (13), 53, 19–81 | n = 43 (23%) | X | |||
| Zayet et al. [48] | UK (France) 2/26/2020–3/14/2020 | Fair | 16/06/2020 | n = 124, COVID group: n = 70; Influenza group: n = 54 | Overall group: M = 31% F = 69% COVID only group: M = 41.4% F = 58.6% | 59(13), NA, 19–98 | n = 7 (6%) out of the COVID only group | X | X | X | |
| Study | Country Study Period | Quality Assessment | Participant Characteristics | Other Audio-Vestibular Manifestations | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Publication Date | Participants (n = 3558) | Gender | Age in Years (Mean, Median, and Range) | Tinnitus | Hearing Loss | Vestibular Impairments | Taste/Smell Disorders | |||
| Cross-sectional studies (n = 3232) | ||||||||||
| Beukes et al. [18] | International 4/29/2020–6/21/2020 | Fair | 05/11/2020 | n = 3103 | M = 50%F = 50% | 58 (14), NA, 18–100 | n = 3996 had pre-existing tinnitus, n = 7 post-COVID tinnitus (0.2%) | X | ||
| Naylor et al. [49] | Scotland 5/29/2020–6/15/2020 | Fair | 01/11/2020 | n = 129 | M = 51.9% F = 48.1% | 64.4, NA, 27–76 | n = 70 | X | ||
| Pre-/post-design (n = 326) | ||||||||||
| Anzivino et al. [50] | Italy ~5/1 to 5/15/2020 | Poor | 22/06/2020 | n = 16 | Not provided | Not provided | n = 16 | |||
| Schlee et al. [51] | Germany 3/28/2018–8/20/2018 and 4/14/2020–4/29/2020 | Fair | 26/08/2020 | n = 122 | M = 65.6% F = 34.4% | 54.0 (10.9), NA, NA | n = 122 | X | X | |
| Xia et al. [52] | China 3/1/2019–4/14/2019 and 3/1/2020–4/14/2020 | Fair | 05/02/2021 | n = 188, n = 89 prior the pandemic, n = 99 during pandemic | 2020:M = 43.4% F = 56.6% 2019: M = 48.3% F= 51.7% | 2020 = 50.8 (15.1), NA, NA, 2019 = 52.6 (14.7), NA, NA | n = 188 | X | ||
| Study Design Considerations | Data Collection and Reporting Suggestions |
|---|---|
| Including control groups with and without the presence of the disease or symptoms being investigated | Reporting basic demographic information such as age, gender, and additional health and mental health difficulties. |
| Utilizing standardized self-reported outcome measures to track the changes in severity of presenting symptoms | Reporting how COVID-19 was tested and managed, and how severe the symptom presentation was |
| Studying wider populations not only form one clinic or region | Reporting possible pre-existing associated factors such as local or systemic infections; vascular or autoimmune disorders; and stress, anxiety, and depression. |
| Undertaking audiometric assessments and comparing these with baseline audiograms or OAE results where available | Describing the tinnitus presentation such as its onset, frequency, descriptions, location, duration, and if it changes or resolves |
| Studies including longitudinal follow-up periods to identify the trajectory of the symptoms to indicate whether the tinnitus resolves or remain and if the severity changes | Investigating psychosocial factors that may contribute such as stress, anxiety, and depression |
| Providing management options to those presenting with audio-vestibular symptoms | Reporting tinnitus or auditory treatments offered and their effects |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Beukes, E.; Ulep, A.J.; Eubank, T.; Manchaiah, V. The Impact of COVID-19 and the Pandemic on Tinnitus: A Systematic Review. J. Clin. Med. 2021, 10, 2763. https://doi.org/10.3390/jcm10132763
Beukes E, Ulep AJ, Eubank T, Manchaiah V. The Impact of COVID-19 and the Pandemic on Tinnitus: A Systematic Review. Journal of Clinical Medicine. 2021; 10(13):2763. https://doi.org/10.3390/jcm10132763
Chicago/Turabian StyleBeukes, Eldre, Alyssa Jade Ulep, Taylor Eubank, and Vinaya Manchaiah. 2021. "The Impact of COVID-19 and the Pandemic on Tinnitus: A Systematic Review" Journal of Clinical Medicine 10, no. 13: 2763. https://doi.org/10.3390/jcm10132763
APA StyleBeukes, E., Ulep, A. J., Eubank, T., & Manchaiah, V. (2021). The Impact of COVID-19 and the Pandemic on Tinnitus: A Systematic Review. Journal of Clinical Medicine, 10(13), 2763. https://doi.org/10.3390/jcm10132763






