Altered Cardiovascular Reactivity to and Recovery from Cold Face Test-Induced Parasympathetic Stimulation in Essential Hypertension
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Participants
2.2. Assessment of Essential Hypertension
2.2.1. Home BP Assessment
2.2.2. Study BP Assessment
2.3. Procedure
2.4. Cold Face Test (CFT)
2.5. Hemodynamic Measures
2.6. Psychological Assessment
2.6.1. Psychopathology
2.6.2. Chronic Stress
2.7. Statistical Analyses
3. Results
3.1. Participants’ Characteristics
3.2. CFT-Induced Reactivity
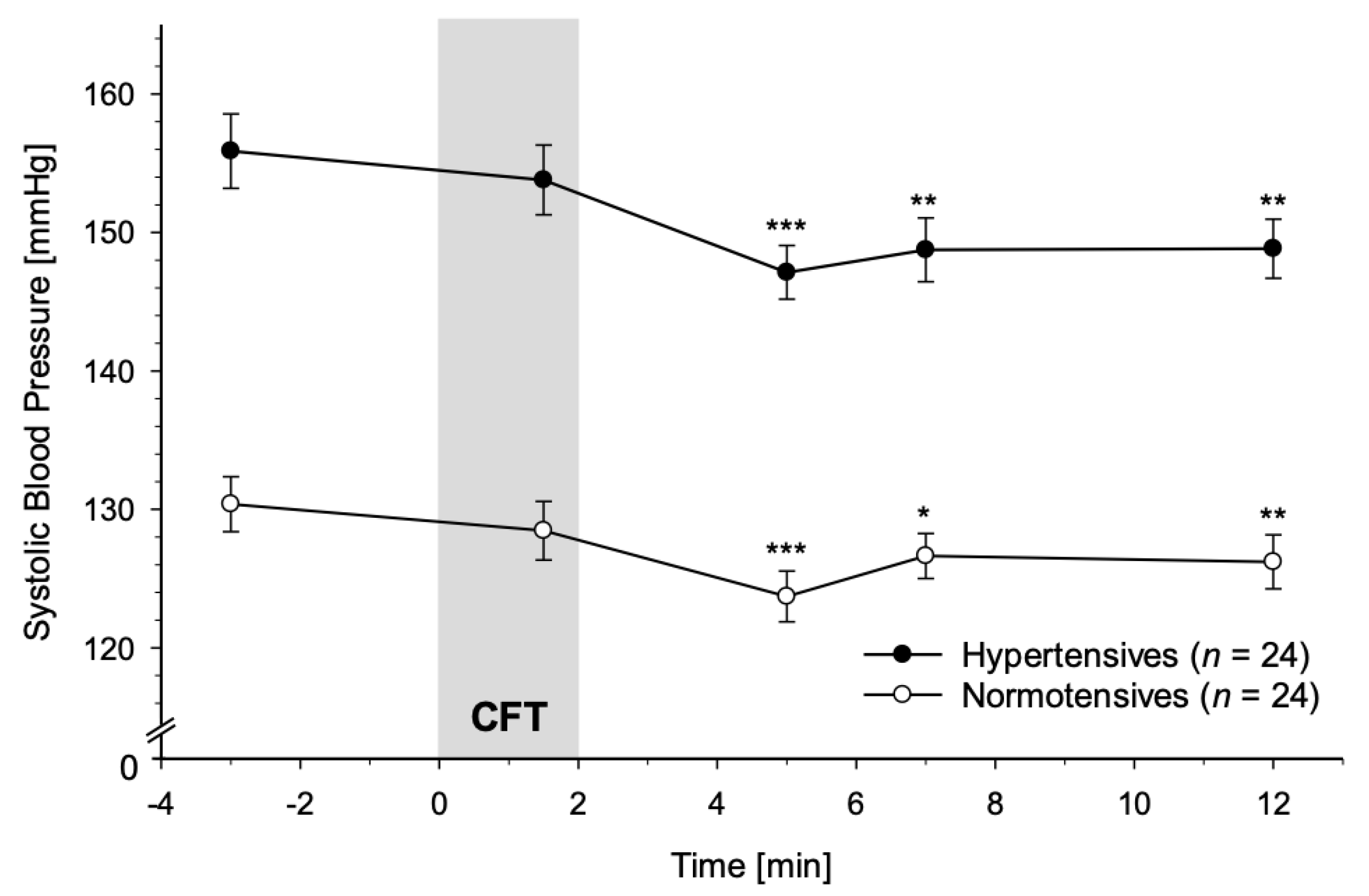
3.2.1. Systolic Blood Pressure
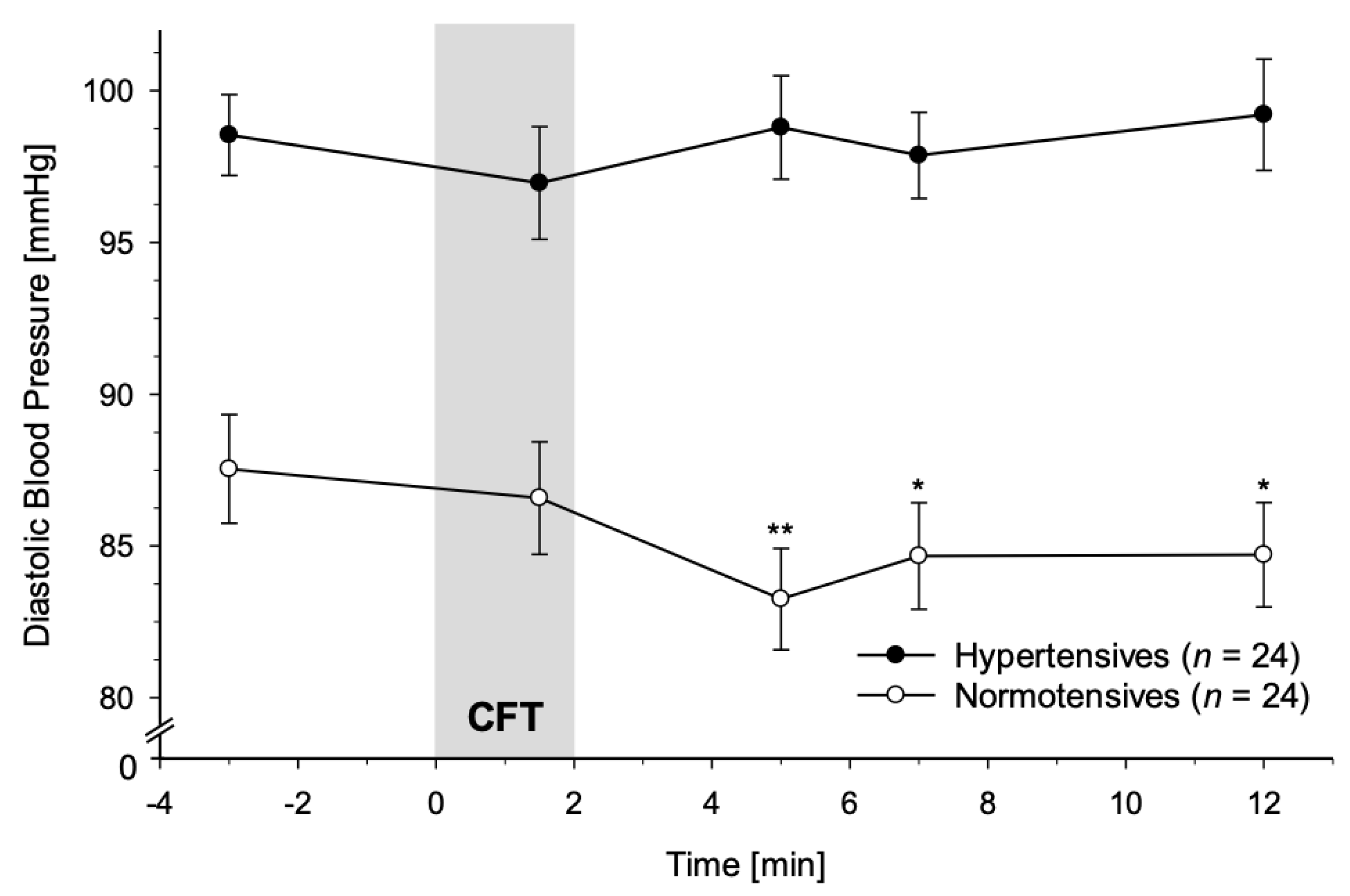
3.2.2. Diastolic Blood Pressure
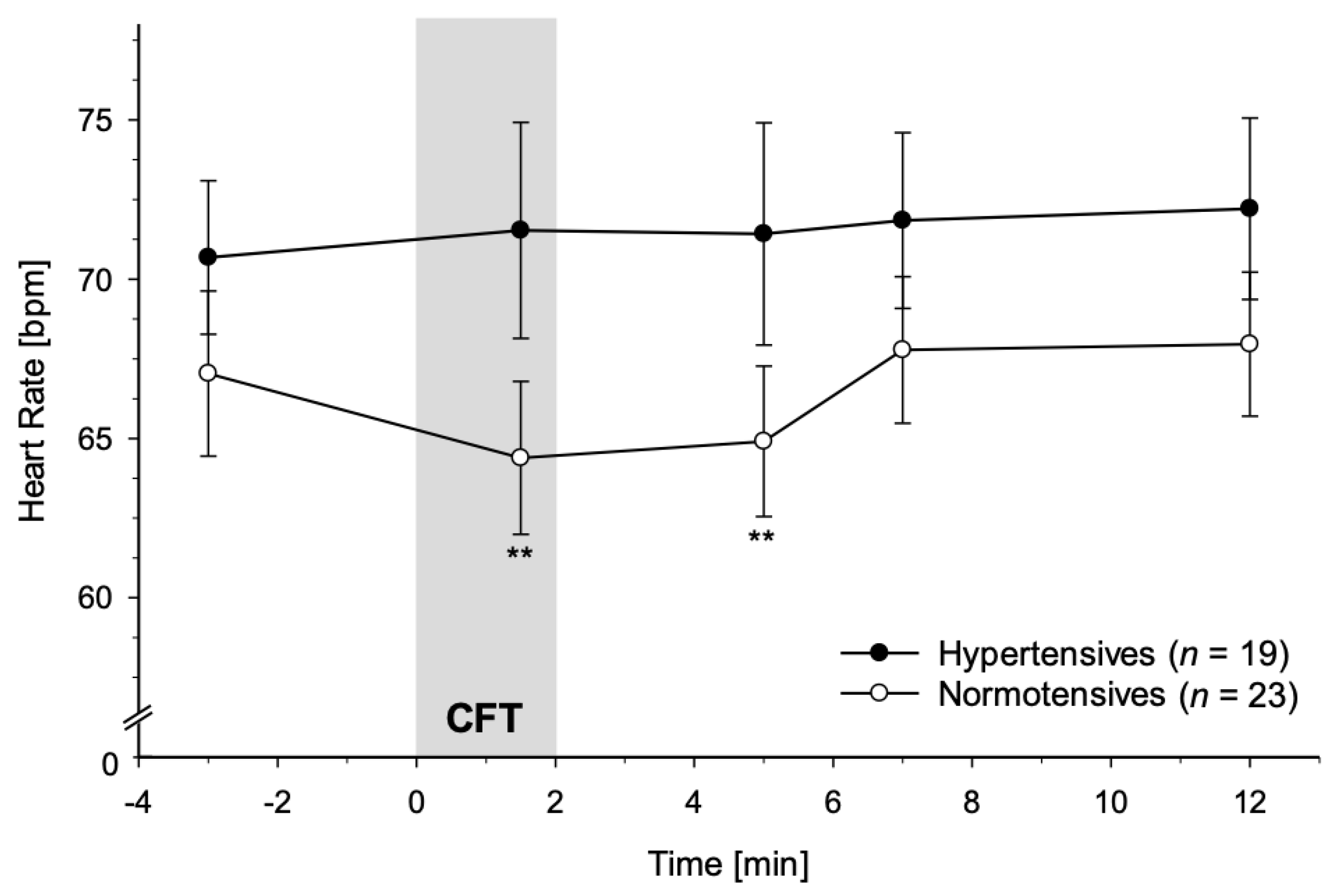
3.2.3. Heart Rate
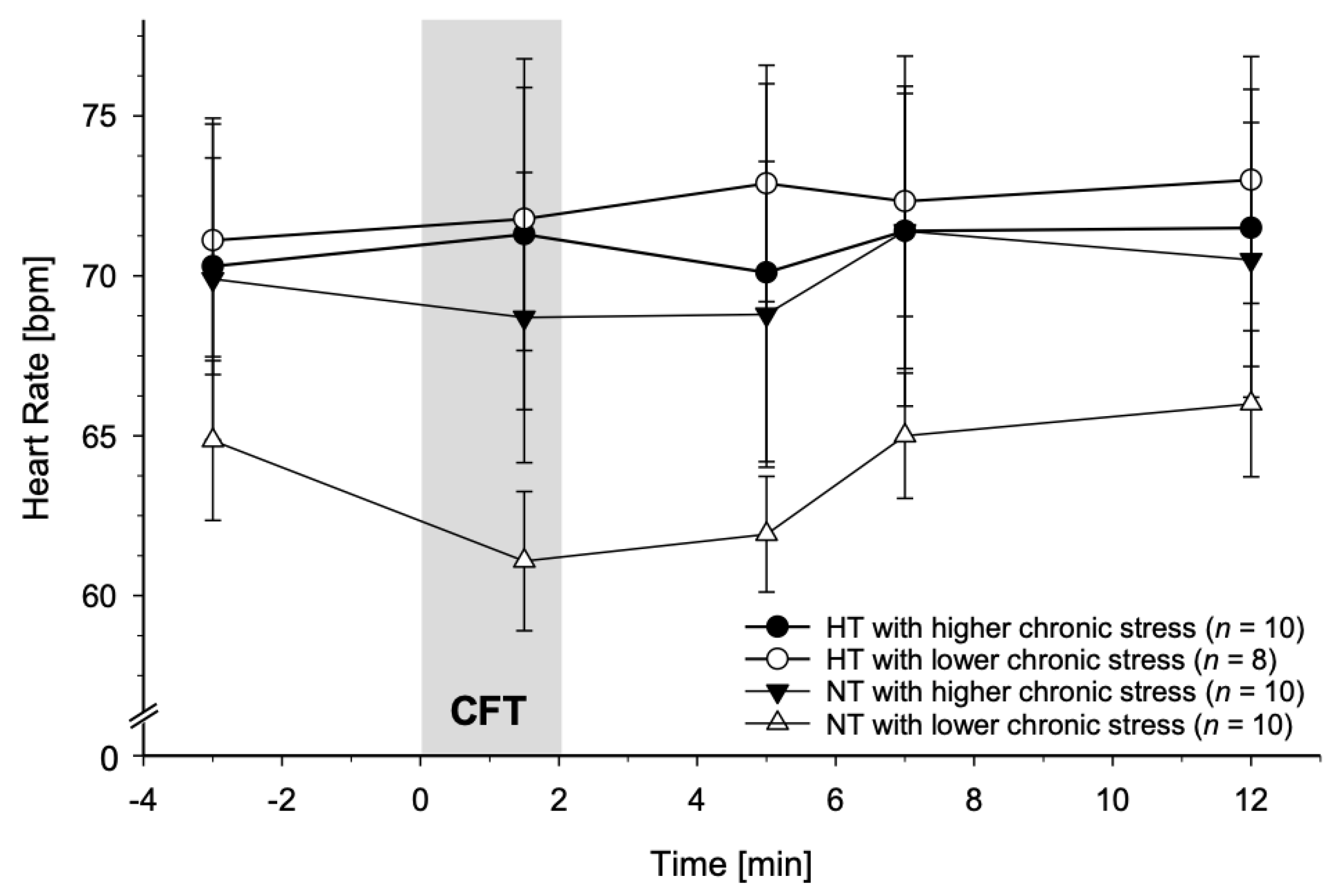
3.3. Associations between Chronic Stress and CFT-Induced Reactivity
3.3.1. Systolic and Diastolic Blood Pressure
3.3.2. Heart Rate
4. Discussion
4.1. Immediate Reactivity to CFT
4.2. Recovery from CFT
4.3. Moderation by Chronic Stress
4.3.1. SBP and DBP in NT
4.3.2. SBP and DBP Reactivity Divergence in HT vs. NT
4.3.3. Decrease in HR in NT and absence of HR Decreases in HT
4.4. Absence of HR Decreases in Chronically Stressed NT
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kaplan, N.; Mendis, S.; Poulter, N.; Whitworth, J. 2003 World Health Organization (WHO)/International Society of Hypertension (ISH) statement on management of hypertension. J. Hypertens. 2003, 21, 1983–1992. [Google Scholar] [CrossRef]
- Carretero, O.A.; Oparil, S. Essential Hypertension. Circulation 2000, 101, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Mancia, G.; Grassi, G. The Autonomic Nervous System and Hypertension. Circ. Res. 2014, 114, 1804–1814. [Google Scholar] [CrossRef]
- Goit, R.K.; Ansari, A.H. Reduced parasympathetic tone in newly diagnosed essential hypertension. Indian Heart J. 2016, 68, 153–157. [Google Scholar] [CrossRef] [PubMed]
- Langewitz, W.; Rüddel, H.; Schächinger, H. Reduced parasympathetic cardiac control in patients with hypertension at rest and under mental stress. Am. Heart J. 1994, 127, 122–128. [Google Scholar] [CrossRef]
- Henquet, J.W.; van Baak, M.; Schols, M.; Rahn, K.H. Studies on the autonomic nervous system in borderline hypertension. Eur. J. Clin. Pharmacol. 1982, 22, 285–288. [Google Scholar] [CrossRef]
- Julius, S.; Pascual, A.V.; London, R. Role of Parasympathetic Inhibition in the Hyperkinetic Type of Borderline Hypertension. Circulation 1971, 44, 413–418. [Google Scholar] [CrossRef]
- Korner, P.; Shaw, J.; Uther, J.; West, M.; McRitchie, R.; Richards, J. Autonomic and Non-Autonomic Circulatory Components in Essential Hypertension in Man. Circulation 1973, 48, 107–117. [Google Scholar] [CrossRef]
- Rahn, K.; van Baak, M.; van Hooff, M.; Schols, M. Studies on salivary flow in borderline hypertension. J. Hypertens. Suppl. 1983, 1, 77–78. [Google Scholar]
- Best, S.A.; Bivens, T.B.; Palmer, M.D.; Boyd, K.N.; Galbreath, M.M.; Okada, Y.; Carrick-Ranson, G.; Fujimoto, N.; Shibata, S.; Hastings, J.L.; et al. Heart rate recovery after maximal exercise is blunted in hypertensive seniors. J. Appl. Physiol. 2014, 117, 1302–1307. [Google Scholar] [CrossRef]
- Kaushik, R.M.; Kaushik, R.; Mahajan, S.K.; Rajesh, V. Effects of mental relaxation and slow breathing in essential hypertension. Complement. Ther. Med. 2006, 14, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Joseph, C.N.; Porta, C.; Casucci, G.; Casiraghi, N.; Maffeis, M.; Rossi, M.; Bernardi, L. Slow Breathing Improves Arterial Baroreflex Sensitivity and Decreases Blood Pressure in Essential Hypertension. Hypertension 2005, 46, 714–718. [Google Scholar] [CrossRef]
- Benjelloun, H.; Aboudrar, S.; Jroundi, I.; Benjelloun-Bennani, H.; Coghlan, L.; Benomar, M. Sympathetic response in primary hypertension. Ann. Cardiol. Angeiol. 2009, 58, 139–143. [Google Scholar] [CrossRef]
- Khurana, R.K.; Watabiki, S.; Hebel, J.; Toro, R.; Nelson, E. Cold face test in the assessment of trigeminal-brainstem-vagal function in humans. Ann. Neurol. 1980, 7, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Heath, M.E.; Downey, J.A. The cold face test (diving reflex) in clinical autonomic assessment: Methodological considerations and repeatability of responses. Clin. Sci. 1990, 78, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Khurana, R.K.; Wu, R. The cold face test: A non-baroreflex mediated test of cardiac vagal function. Clin. Auton. Res. 2006, 16, 202–207. [Google Scholar] [CrossRef] [PubMed]
- Reyners, A.K.L.; Tio, R.A.; Vlutters, F.G.; van der Woude, G.F.; Reitsma, W.D.; Smit, A.J. Re-evaluation of the cold face test in humans. Eur. J. Appl. Physiol. 2000, 82, 487–492. [Google Scholar] [CrossRef]
- Hines, E.A., Jr.; Brown, G.E. The cold pressor test for measuring the reactibility of the blood pressure: Data concerning 571 normal and hypertensive subjects. Am. Heart J. 1936, 11, 1–9. [Google Scholar] [CrossRef]
- Velasco, M.; Gómez, J.; Blanco, M.; Rodriguez, I. The cold pressor test: Pharmacological and therapeutic aspects. Am. J. Ther. 1997, 4, 34–38. [Google Scholar] [CrossRef]
- Hurwitz, B.E.; Furedy, J.J. The human dive reflex: An experimental, topographical and physiological analysis. Physiol. Behav. 1986, 36, 287–294. [Google Scholar] [CrossRef]
- Murakami, M.; Suzuki, H.; Naitoh, M.; Nakamoto, H.; Ichihara, A.; Matsumoto, A.; Takeshita, E.; Kanno, Y.; Saruta, T. Evidence for Abnormalities in Parasympathetic Nerve-Mediated Reflexes in Borderline Hypertension. Hypertens. Res. 1993, 16, 185–190. [Google Scholar] [CrossRef][Green Version]
- Miziara, A.N.; Molina, R.J.; Ferreira, B.D.C.; Barbosa, C.J.D.G.; da Silva, V.J.D.; Prata, A.; Correia, D. Cardiac autonomic modulation in hypertensive patients with Chagas’ disease. Acta Trop. 2006, 97, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Ushakov, A.V.; Ivanchenko, V.S.; Gagarina, A.A. Psychological Stress in Pathogenesis of Essential Hypertension. Curr. Hypertens. Rev. 2016, 12, 203–214. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S. Stress, Adaptation, and Disease: Allostasis and Allostatic Load. Ann. N. Y. Acad. Sci. 1998, 840, 33–44. [Google Scholar] [CrossRef]
- McEwen, B.S. Protection and Damage from Acute and Chronic Stress: Allostasis and Allostatic Overload and Relevance to the Pathophysiology of Psychiatric Disorders. Ann. N. Y. Acad. Sci. 2004, 1032, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.-Y.; Li, N.; Li, W.A.; Khan, H. Association between psychosocial stress and hypertension: A systematic review and meta-analysis. Neurol. Res. 2017, 39, 573–580. [Google Scholar] [CrossRef] [PubMed]
- Gump, B.B.; Matthews, K.A. Do Background Stressors Influence Reactivity to and Recovery From Acute Stressors? J. Appl. Soc. Psychol. 1999, 29, 469–494. [Google Scholar] [CrossRef]
- de Looff, P.C.; Cornet, L.J.M.; Embregts, P.J.C.M.; Nijman, H.L.I.; Didden, H.C.M. Associations of sympathetic and parasympathetic activity in job stress and burnout: A systematic review. PLoS ONE 2018, 13, e0205741. [Google Scholar] [CrossRef]
- Koenig, J.; Thayer, J.F. Sex differences in healthy human heart rate variability: A meta-analysis. Neurosci. Biobehav. Rev. 2016, 64, 288–310. [Google Scholar] [CrossRef]
- Williams, B.; Mancia, G.; Spiering, W.; Agabiti Rosei, E.; Azizi, M.; Burnier, M.; Clement, D.L.; Coca, A.; De Simone, G.; Dominiczak, A. ESC/ESH Guidelines for the management of arterial hypertension: The Task Force for the management of arterial hypertension of the European Society of Cardiology (ESC) and the European Society of Hypertension (ESH). Eur. Heart J. 2018, 39, 3021–3104. [Google Scholar] [CrossRef] [PubMed]
- Chalmers, J.; MacMahon, S.; Mancia, G.; Whitworth, J.; Beilin, L.; Hansson, L.; Neal, B.; Rodgers, A.; Ni Mhurchu, C.; Clark, T. 1999 World Health Organization-International Society of Hypertension Guidelines for the management of hypertension. Guidelines sub-committee of the World Health Organization. Clin. Exp. Hypertens. 1999, 21, 1009–1060. [Google Scholar] [CrossRef] [PubMed]
- Franke, G.H. BSI: Brief Sympton Inventory von L.R. Derogatis (Kurzform der SCL-90-R)—Deutsche Version; Beltz: Göttingen, Germany, 2000. [Google Scholar]
- Schulz, P.; Schlotz, W.; Becker, P. Das Trierer Inventar zum Chronischen Stress (TICS); Hogrefe: Göttingen, Germany, 2004. [Google Scholar]
- Faul, F.; Erdfelder, E.; Lang, A.-G.; Buchner, A. G* Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 2007, 39, 175–191. [Google Scholar] [CrossRef] [PubMed]
- De Meersman, R.E.; Stein, P.K. Vagal modulation and aging. Biol. Psychol. 2007, 74, 165–173. [Google Scholar] [CrossRef]
- Koenig, J.; Jarczok, M.; Warth, M.; Ellis, R.; Bach, C.; Hillecke, T.; Thayer, J.F. Body mass index is related to autonomic nervous system activity as measured by heart rate variability—A replication using short term measurements. J. Nutr. Health Aging 2014, 18, 300–302. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioural Sciences.; Erlbaum: Hillsdale, NJ, USA, 1988. [Google Scholar]
- La Marca, R.; Waldvogel, P.; Thörn, H.; Tripod, M.; Wirtz, P.H.; Pruessner, J.C.; Ehlert, U. Association between Cold Face Test-induced vagal inhibition and cortisol response to acute stress. Psychophysiology 2011, 48, 420–429. [Google Scholar] [CrossRef] [PubMed]
- Signore, P.E.; Jones, D.R. Effect of pharmacological blockade on cardiovascular responses to voluntary and forced diving in muskrats. J. Exp. Biol. 1995, 198, 2307–2315. [Google Scholar] [CrossRef]
- Ulrich-Lai, Y.M.; Herman, J.P. Neural regulation of endocrine and autonomic stress responses. Nat. Rev. Neurosci. 2009, 10, 397–409. [Google Scholar] [CrossRef]
- Weber, C.S.; Thayer, J.F.; Rudat, M.; Wirtz, P.H.; Zimmermann-Viehoff, F.; Thomas, A.; Perschel, F.H.; Arck, P.C.; Deter, H.C. Low vagal tone is associated with impaired post stress recovery of cardiovascular, endocrine, and immune markers. Eur. J. Appl. Physiol. 2010, 109, 201–211. [Google Scholar] [CrossRef]
- Gordan, R.; Gwathmey, J.K.; Xie, L.H. Autonomic and endocrine control of cardiovascular function. World J. Cardiol. 2015, 7, 204–214. [Google Scholar] [CrossRef] [PubMed]
- Coote, J.H. Myths and realities of the cardiac vagus. J. Physiol. 2013, 591, 4073–4085. [Google Scholar] [CrossRef] [PubMed]
- Dhein, S.; van Koppen, C.J.; Brodde, O.-E. Muscarinic receptors in the Mammalian Heart. Pharmacol. Res. 2001, 44, 161–182. [Google Scholar] [CrossRef]
- Vanhoutte, P.M.; Shepherd, J.T. Muscarinic and β-adrenergic prejunctional modulation of adrenergic neurotransmission in the blood vessel wall. Gen. Pharmacol. 1983, 14, 35–37. [Google Scholar] [CrossRef]
- Dampney, R.; Coleman, M.; Fontes, M.; Hirooka, Y.; Horiuchi, J.; Li, Y.-W.; Polson, J.; Potts, P.; Tagawa, T. Central Mechanisms Underlying Short- And Long-Term Regulation Of The Cardiovascular System. Clin. Exp. Pharmacol. Physiol. 2002, 29, 261–268. [Google Scholar] [CrossRef]
- Belz, G.G. Elastic properties and Windkessel function of the human aorta. Cardiovasc. Drugs Ther. 1995, 9, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Safar, M.E.; Girerd, X.; Laurent, S. Structural changes of large conduit arteries in hypertension. J. Hypertens. 1996, 14, 545–555. [Google Scholar] [CrossRef] [PubMed]
- Cavalcante, J.L.; Lima, J.A.C.; Redheuil, A.; Al-Mallah, M.H. Aortic Stiffness: Current Understanding and Future Directions. J. Am. Coll. Cardiol. 2011, 57, 1511–1522. [Google Scholar] [CrossRef] [PubMed]
- Mayet, J.; Hughes, A. Cardiac and vascular pathophysiology in hypertension. Heart 2003, 89, 1104–1109. [Google Scholar] [CrossRef]
- Lind, L. Endothelium-dependent Vasodilation in Hypertension: A Review. Blood Press 2000, 9, 4–15. [Google Scholar] [CrossRef] [PubMed]
- Pal, G. Blood pressure and its regulation. In Textbook of Medical Physiology; Ahuja Publications: New Delhi, India, 2010; pp. 209–216. [Google Scholar]
- Fisher, J.T.; Vincent, S.G.; Gomeza, J.; Yamada, M.; Wess, J. Loss of vagally mediated bradycardia and bronchoconstriction in mice lacking M2 or M3 muscarinic acetylcholine receptors. FASEB J. 2004, 18, 711–713. [Google Scholar] [CrossRef]
- LaCroix, C.; Freeling, J.; Giles, A.; Wess, J.; Li, Y.-F. Deficiency of M2 muscarinic acetylcholine receptors increases susceptibility of ventricular function to chronic adrenergic stress. Am. J. Physiol. Heart Circ. Physiol. 2008, 294, H810–H820. [Google Scholar] [CrossRef]
- Toda, N.; Nakanishi-Toda, M. How mental stress affects endothelial function. Pflugers Arch. 2011, 462, 779–794. [Google Scholar] [CrossRef]
- Petkovich, B.W.; Vega, J.; Thomas, S. Vagal modulation of hypertension. Curr. Hypertens. Rep. 2015, 17, 26. [Google Scholar] [CrossRef]
- Chapleau, M.W.; Rotella, D.L.; Reho, J.J.; Rahmouni, K.; Stauss, H.M. Chronic vagal nerve stimulation prevents high-salt diet-induced endothelial dysfunction and aortic stiffening in stroke-prone spontaneously hypertensive rats. Am. J. Physiol. Heart Circ. Physiol. 2016, 311, 276–285. [Google Scholar] [CrossRef]
- Grewal, S.; Gupta, V. Effect of obesity on autonomic nervous system. Int. J. Curr. Biol. Med. Sci. 2011, 1, 15–18. [Google Scholar]
- Thayer, J.F.; Yamamoto, S.S.; Brosschot, J.F. The relationship of autonomic imbalance, heart rate variability and cardiovascular disease risk factors. Int. J. Cardiol. 2010, 141, 122–131. [Google Scholar] [CrossRef] [PubMed]
- Pal, G.K.; Chandrasekaran, A.; Hariharan, A.P.; Dutta, T.K.; Pal, P.; Nanda, N.; Venugopal, L. Body mass index contributes to sympathovagal imbalance in prehypertensives. BMC Cardiovasc. Disord. 2012, 12, 54. [Google Scholar] [CrossRef] [PubMed]
- Lopes, R.; Neves, K.; Carneiro, F.; Tostes, R. Testosterone and Vascular Function in Aging. Front. Physiol. 2012, 3. [Google Scholar] [CrossRef] [PubMed]
| Normotensives (n = 24) Mean ± SEM (range) | Hypertensives (n = 24) Mean ± SEM (range) | p | |
|---|---|---|---|
| Age (years) | 52.71 ± 2.08 (29–64) | 54.13 ± 1.25 (38–64) | 0.56 |
| BMI (kg/m2) | 23.82 ± 0.41(20.73–29.04) | 26.13 ± 0.51 (21.29–30.76) | 0.001 ** |
| MAP (mmHg) | 92.80 ± 1.25 (81.67–103.67) | 112.94 ± 1.27 (97.83–123.83) | <0.001 *** |
| Resting SBP α (mmHg) | 122.98 ± 1.46 (105.5–134.0) | 149.40 ± 1.64 (133.5–163.5) | <0.001 *** |
| Resting DBP α (mmHg) | 77.71 ± 1.37 (62.0–89.0) | 94.71 ± 1.43 (74.0–107.0) | <0.001 *** |
| Resting HR α (min−1) | 69.52 ± 2.14 (53.0–91.5), n = 23 | 69.96 ± 2.23 (49.0–99.5) | 0.89 |
| GSI | 0.20 ± 0.04 (0.00–0.89) | 0.25 ± 0.04 (0.02–0.72), n = 23 | 0.40 |
| TICS-CSSS | 10.19 ± 1.67 (2–34), n = 21 | 11.35 ± 1.50 (0–29), n = 23 | 0.61 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Walther, L.-M.; von Känel, R.; Heimgartner, N.; Zuccarella-Hackl, C.; Ehlert, U.; Wirtz, P.H. Altered Cardiovascular Reactivity to and Recovery from Cold Face Test-Induced Parasympathetic Stimulation in Essential Hypertension. J. Clin. Med. 2021, 10, 2714. https://doi.org/10.3390/jcm10122714
Walther L-M, von Känel R, Heimgartner N, Zuccarella-Hackl C, Ehlert U, Wirtz PH. Altered Cardiovascular Reactivity to and Recovery from Cold Face Test-Induced Parasympathetic Stimulation in Essential Hypertension. Journal of Clinical Medicine. 2021; 10(12):2714. https://doi.org/10.3390/jcm10122714
Chicago/Turabian StyleWalther, Lisa-Marie, Roland von Känel, Nadja Heimgartner, Claudia Zuccarella-Hackl, Ulrike Ehlert, and Petra H. Wirtz. 2021. "Altered Cardiovascular Reactivity to and Recovery from Cold Face Test-Induced Parasympathetic Stimulation in Essential Hypertension" Journal of Clinical Medicine 10, no. 12: 2714. https://doi.org/10.3390/jcm10122714
APA StyleWalther, L.-M., von Känel, R., Heimgartner, N., Zuccarella-Hackl, C., Ehlert, U., & Wirtz, P. H. (2021). Altered Cardiovascular Reactivity to and Recovery from Cold Face Test-Induced Parasympathetic Stimulation in Essential Hypertension. Journal of Clinical Medicine, 10(12), 2714. https://doi.org/10.3390/jcm10122714







