Surgical Management and Reconstruction of Diaphragm, Pericardium and Chest Wall in Mesothelioma Surgery: A Review
Abstract
:1. Introduction
2. Surgical Procedures for MPM Resection
3. Diaphragm
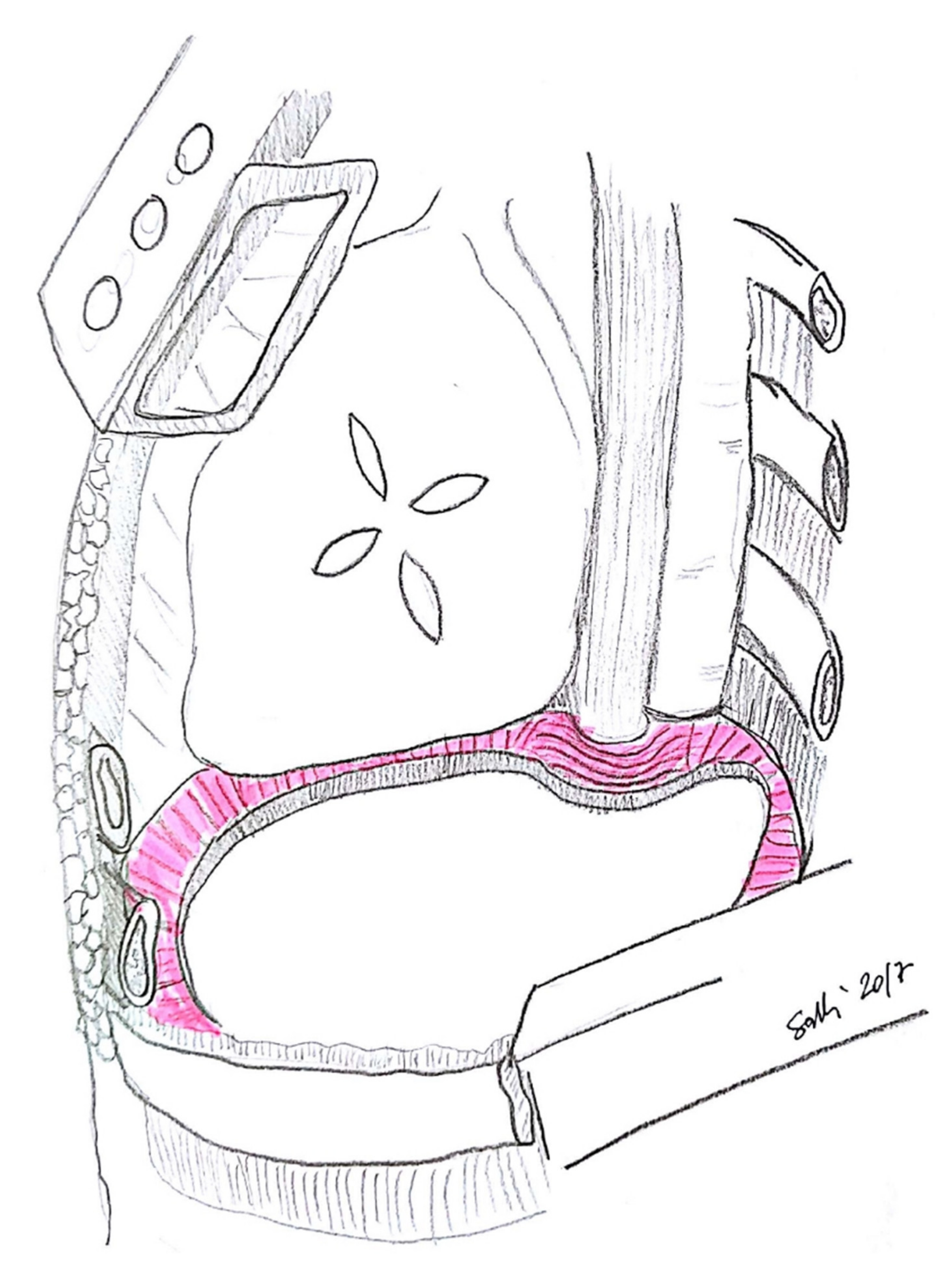
3.1. Diaphragm Management
3.2. Types of Prosthesis
3.3. Technical Aspects
4. Pericardium
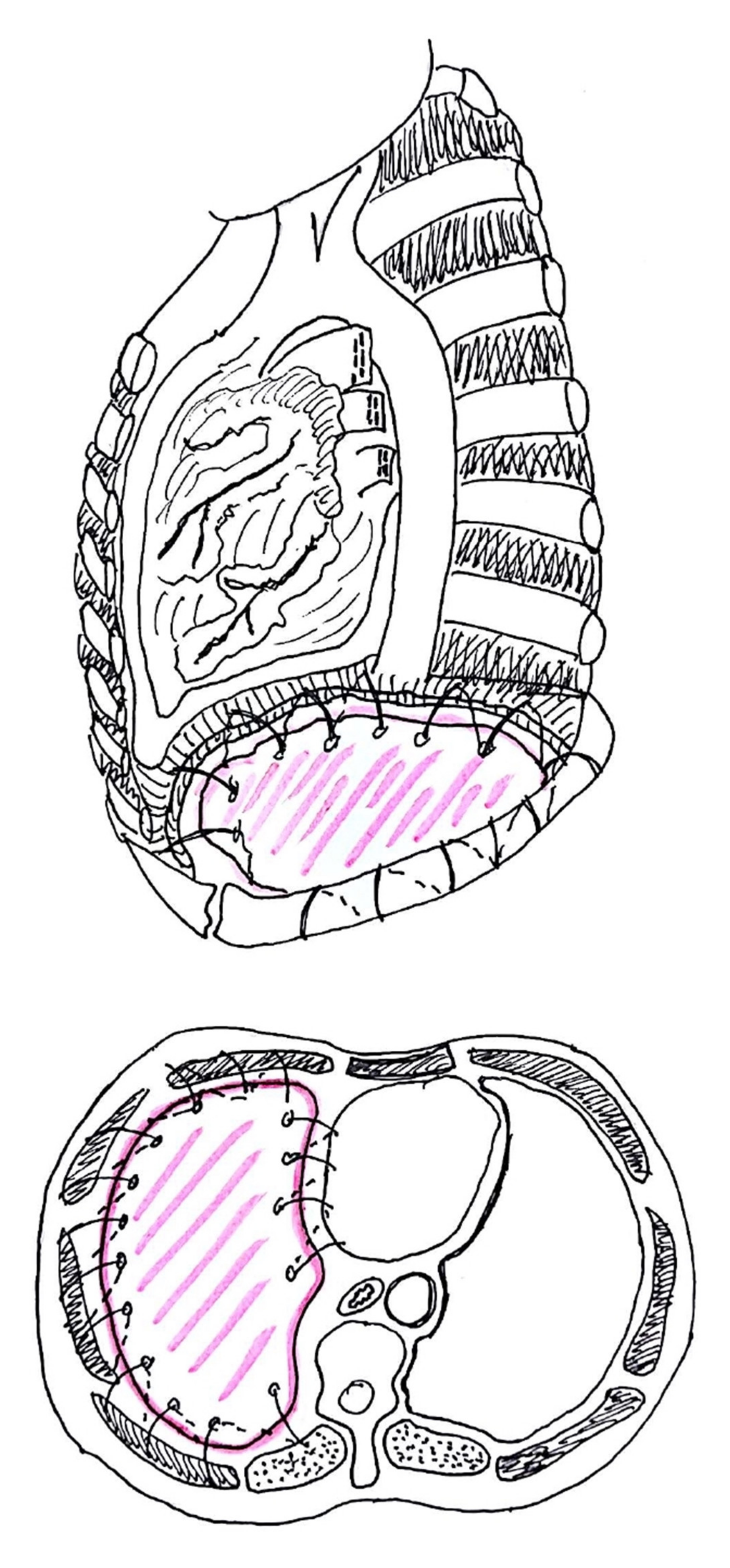
4.1. Pericardium Management
4.2. Types of Prosthesis
4.3. Technical Aspects
5. Chest Wall
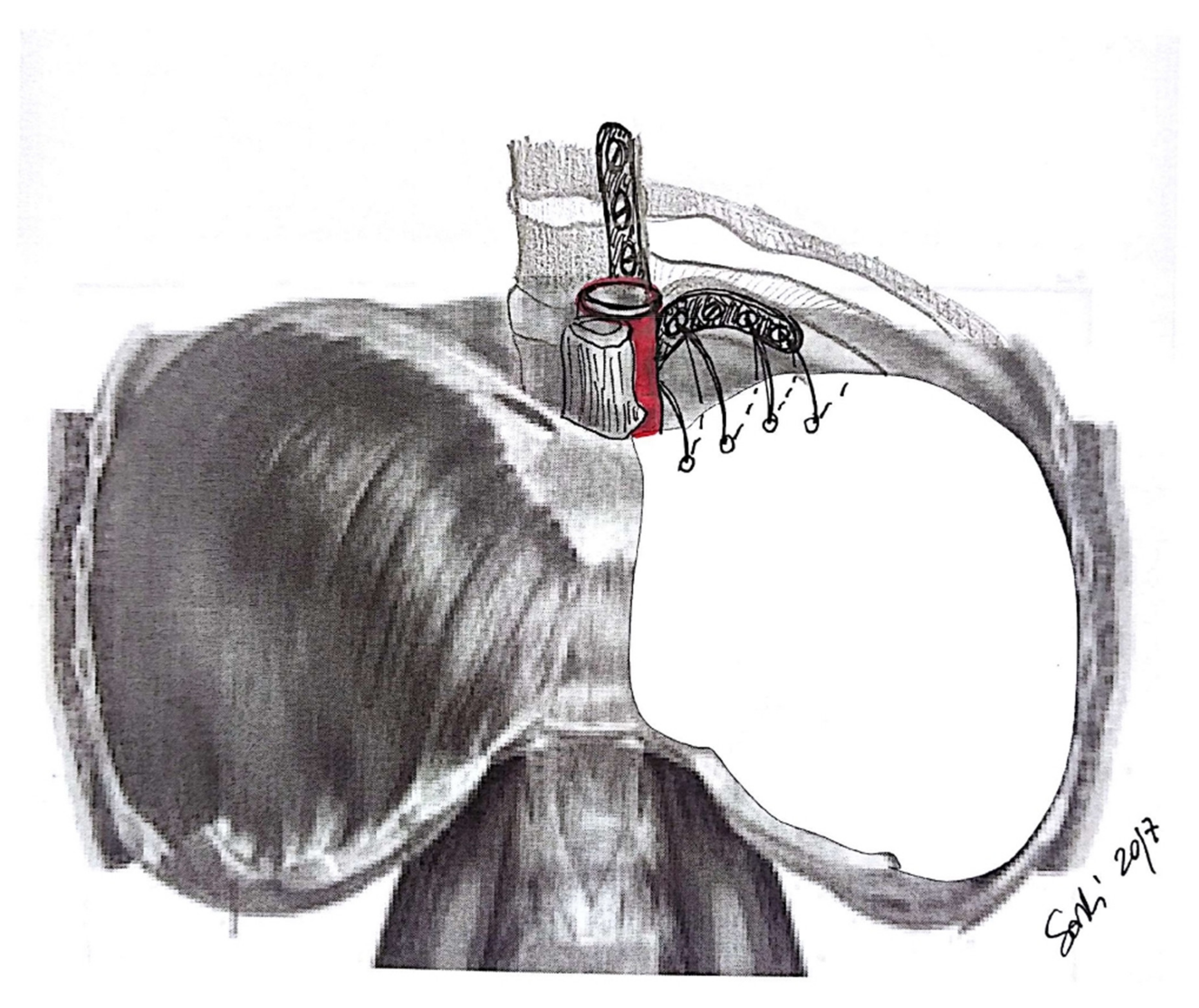
5.1. Chest Wall Management
5.2. Types of Prosthesis
5.3. Technical Aspects
6. Role of Minimally Invasive Surgery
7. Conclusions
Authors Contribution
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bertoglio, P.; Waller, D.A. The role of thoracic surgery in the management of mesothelioma: An expert opinion on the limited evidence. Expert Rev. Respir. Med. 2016, 10, 663–672. [Google Scholar] [CrossRef]
- Vogelzang, N.J.; Rusthoven, J.J.; Symanowski, J.; Denham, C.; Kaukel, E.; Ruffie, P.; Gotzemeier, U.; Boyer, M.; Emri, S.; Paoletti, P.; et al. Phase III study of pemetrexed in combination with cisplatin versus cisplatin alone in patients with malignant pleural mesothelioma. J. Clin. Oncol. 2003, 21, 2636–2644. [Google Scholar] [CrossRef] [PubMed]
- Mutti, L.; Peikert, T.; Robinson, B.W.; Scherpereel, A.; Tsao, A.S.; de Perrot, M.; Woodard, G.A.; Jablons, D.M.; Wiens, J.; Hirsch, F.R.; et al. Scientific Advances and New Frontiers in Mesothelioma Therapeutics. J. Thorac. Oncol. 2018, 13, 1269–1283. [Google Scholar] [CrossRef] [Green Version]
- Bertoglio, P.; Aprile, V.; Ambrogi, M.C.; Mussi, A.; Lucchi, M. The role of intracavitary therapies in the treatment of malignant pleural mesothelioma. J. Thorac. Dis. 2018, 10 (Suppl. 2), S293–S297. [Google Scholar] [CrossRef] [Green Version]
- Ambrogi, M.C.; Bertoglio, P.; Aprile, V.; Chella, A.; Korasidis, S.; Fontanini, G.; Fanucchi, O.; Lucchi, M.; Mussi, A. Diaphragm and lung-preserving surgery with hyperthermic chemotherapy for malignant pleural mesothelioma: A 10-year experience. J. Thorac. Cardiovasc. Surg. 2018, 155, 1857–1866. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bertoglio, P.; Ambrogi, M.C.; Chella, A.; Aprile, V.; Dini, P.; Korasidis, S.; Fanucchi, O.; Mussi, A. Is less also better? A single-institution experience on treatment of early stage Malignant Pleural Mesothelioma. Eur. J. Surg. Oncol. 2017, 43, 1365–1371. [Google Scholar] [CrossRef] [PubMed]
- Casiraghi, M.; Maisonneuve, P.; Brambilla, D.; Solli, P.; Galetta, D.; Petrella, F.; Piperno, G.; De Marinis, F.; Spaggiari, L. Induction chemotherapy, extrapleural pneumonectomy and adjuvant radiotherapy for malignant pleural mesothelioma. Eur. J. Cardiothorac. Surg. 2017, 52, 975–981. [Google Scholar] [CrossRef] [Green Version]
- Sugarbaker, D.J. Macroscopic complete resection: The goal of primary surgery in multimodality therapy for pleural mesothelioma. J. Thorac. Oncol. 2006, 1, 175–176. [Google Scholar] [CrossRef]
- Rice, D.; Rusch, V.; Pass, H.; Asamura, H.; Nakano, T.; Edwards, J.; Giroux, D.; Hasegawa, S.; Kernstine, K.H.; Waller, D.; et al. Recommendations for uniform definitions of surgical techniques for malignant pleural mesothelioma: A consensus report of the international association for the study of lung cancer international staging committee and the international mesothelioma interest group. J. Thorac. Oncol. 2011, 6, 1304–1312. [Google Scholar] [PubMed] [Green Version]
- Bellini, A.; Dell’Amore, A.; Terzi, S.; Zambello, G.; Zuin, A.; Pasello, G.; Calabrese, F.; Schiavon, M.; Rea, F. Relapse Patterns and Tailored Treatment Strategies for Malignant Pleural Mesothelioma Recurrence after Multimodality Therapy. J. Clin. Med. 2021, 10, 1134. [Google Scholar] [CrossRef]
- Treasure, T.; Lang-Lazdunski, L.; Waller, D.; Bliss, J.M.; Tan, C.; Entwisle, J.; Snee, M.; O’brien, M.; Thomas, G.; Peto, J.; et al. Extra-pleural pneumonectomy versus no extra-pleural pneumonectomy for patients with malignant pleural mesothelioma: Clinical outcomes of the Mesothelioma and Radical Surgery (MARS) randomised feasibility study. Lancet Oncol. 2011, 12, 763–772. [Google Scholar] [CrossRef]
- Weder, W.; Stahel, R.A.; Baas, P.; Dafni, U.; de Perrot, M.; McCaughan, B.C.; Nakano, T.; Pass, H.I.; Robinson, B.W.; van Zandwijk, N.; et al. The MARS feasibility trial: Conclusions not supported by data. Lancet Oncol. 2011, 12, 1093–1094. [Google Scholar] [CrossRef]
- Sharkey, A.J.; Bilancia, R.; Tenconi, S.; Nakas, A.; Waller, D.A. The management of the diaphragm during radical surgery for malignant pleural mesothelioma. Eur. J. Cardiothorac. Surg. 2016, 50, 311–316. [Google Scholar] [CrossRef] [Green Version]
- Talon, I.; Schneider, A.; Ball, V.; Hemmerlé, J. Functionalization of PTFE Materials Using a Combination of Polydopamine and Platelet-Rich Fibrin. J. Surg. Res. 2020, 251, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Solli, P.; Brandolini, J.; Pardolesi, A.; Nardini, M.; Lacava, N.; Parri, S.F.; Kawamukai, K.; Bonfanti, B.; Bertolaccini, L. Diaphragmatic and pericardial reconstruction after surgery for malignant pleural mesothelioma. J. Thorac. Dis. 2018, 10 (Suppl. 2), S298–S303. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rolli, L.; Leuzzi, G.; Girotti, P.; Duranti, L.; Pastorino, U. Permeable Nonabsorbable Mesh for Total Diaphragmatic Replacement in Extended Pneumonectomy. Ann. Thorac. Surg. 2017, 104, e105–e107. [Google Scholar] [CrossRef]
- Sugarbaker, D.J.; Jaklitsch, M.T.; Bueno, R.; Richards, W.; Lukanich, J.; Mentzer, S.J.; Colson, Y.; Linden, P.; Chang, M.; Zellos, L.S.; et al. Prevention, early detection, and management of complications after 328 consecutive extrapleural pneumonectomies. J. Thorac. Cardiovasc. Surg. 2004, 128, 138–146. [Google Scholar] [CrossRef] [Green Version]
- Al-Nouri, O.; Hartman, B.; Freedman, R.; Thomas, C.; Esposito, T. Diaphragmatic rupture: Is management with biological mesh feasible? Int. J. Surg. Case Rep. 2012, 3, 349–353. [Google Scholar] [CrossRef] [Green Version]
- Bedini, A.V.; Andreani, S.M.; Muscolino, G. Latissimus dorsi reverse flap to substitute the diaphragm after extrapleural pneumonectomy. Ann. Thorac. Surg. 2000, 69, 986–988. [Google Scholar] [CrossRef]
- Schiavon, M.; De Filippis, A.; Marulli, G.; Rea, F. A new technique of diaphragmatic patch fixation in extrapleural pneumonectomy. Eur. J. Cardiothorac. Surg. 2010, 38, 798–800. [Google Scholar] [CrossRef]
- Chen, R.F.; Lai, C.P. Constrictive pericarditis associated with Marlex mesh. Two case reports. Tex. Heart Inst. J. 2001, 28, 63–64. [Google Scholar] [PubMed]
- Urschel, J.D.; Takita, H. Pericardial closure after intrapericardial pneumonectomy. Ann. Thorac. Surg. 1999, 67, 295–296. [Google Scholar]
- Bertoglio, P.; Fanucchi, O.; Ricciardi, S.; Chella, A.; Lucchi, M.; Mussi, A. Chest wall resection for mesothelioma recurrence after surgery. Asian Cardiovasc. Thorac. Ann. 2016, 24, 893–895. [Google Scholar] [CrossRef] [PubMed]
- Elsayed, H.H.; Hassaballa, A.S.; Ahmed, T.A.; Sharkawy, H.Y. Recurrence of mesothelioma after a macroscopic complete resection procedure: Is a second radical surgery justified? Interact. Cardiovasc. Thorac. Surg. 2021, 32, 761–763. [Google Scholar] [CrossRef] [PubMed]
- Burt, B.M.; Ali, S.O.; DaSilva, M.C.; Yeap, B.Y.; Richards, W.G.; Baldini, E.H.; Sugarbaker, D.J. Clinical indications and results after chest wall resection for recurrent mesothelioma. J. Thorac. Cardiovasc. Surg. 2013, 146, 1373–1379. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Politi, L.; Borzellino, G. Second surgery for recurrence of malignant pleural mesothelioma after extrapleural pneumonectomy. Ann. Thorac. Surg. 2010, 89, 207–210. [Google Scholar] [CrossRef]
- Khullar, O.V.; Fernandez, F.G. Prosthetic Reconstruction of the Chest Wall. Thorac. Surg. Clin. 2017, 27, 201–208. [Google Scholar] [CrossRef]
- Weyant, M.J.; Bains, M.S.; Venkatraman, E.; Downey, R.J.; Park, B.J.; Flores, R.M.; Rizk, N.; Rusch, V.W. Results of chest wall resection and reconstruction with and without rigid prosthesis. Ann. Thorac. Surg. 2006, 81, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Berthet, J.P.; Caro, A.G.; Solovei, L.; Gilbert, M.; Bommart, S.; Gaudard, P.; Canaud, L.; Alric, P.; Marty-Ané, C.H. Titanium Implant Failure After Chest Wall Osteosynthesis. Ann. Thorac. Surg. 2015, 99, 1945–1952. [Google Scholar] [CrossRef]
- Sanna, S.; Brandolini, J.; Pardolesi, A.; Argnani, D.; Mengozzi, M.; Dell’Amore, A.; Solli, P. Materials and techniques in chest wall reconstruction: A review. J. Vis. Surg. 2017, 3, 95. [Google Scholar] [CrossRef] [Green Version]
- Rintoul, R.C.; Ritchie, A.J.; Edwards, J.G.; Waller, D.A.; Coonar, A.S.; Bennett, M.; Lovato, E.; Hughes, V.; Fox-Rushby, J.A.; Sharples, L.D.; et al. Efficacy and cost of video-assisted thoracoscopic partial pleurectomy versus talc pleurodesis in patients with malignant pleural mesothelioma (MesoVATS): An open-label, randomised, controlled trial. Lancet 2014, 384, 1118–1127. [Google Scholar] [CrossRef] [Green Version]
- Demmy, T.L. Video-assisted thoracoscopic extrapleural pneumonectomy for malignant pleural mesothelioma. Ann. Cardiothorac. Surg. 2012, 1, 533. [Google Scholar] [PubMed]
- Suda, T.; Kitamura, Y.; Hasegawa, S.; Negi, K.; Hattori, Y. Video-assisted thoracoscopic extrapleural pneumonectomy for malignant pleural mesothelioma. J. Thorac. Cardiovasc. Surg. 2007, 134, 1088–1089. [Google Scholar] [CrossRef] [PubMed] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bertoglio, P.; Garelli, E.; Brandolini, J.; Kawamukai, K.; Antonacci, F.; Ricciardi, S.; Cipolli, A.; Bonfanti, B.; Forti Parri, S.N.; Daddi, N.; et al. Surgical Management and Reconstruction of Diaphragm, Pericardium and Chest Wall in Mesothelioma Surgery: A Review. J. Clin. Med. 2021, 10, 2330. https://doi.org/10.3390/jcm10112330
Bertoglio P, Garelli E, Brandolini J, Kawamukai K, Antonacci F, Ricciardi S, Cipolli A, Bonfanti B, Forti Parri SN, Daddi N, et al. Surgical Management and Reconstruction of Diaphragm, Pericardium and Chest Wall in Mesothelioma Surgery: A Review. Journal of Clinical Medicine. 2021; 10(11):2330. https://doi.org/10.3390/jcm10112330
Chicago/Turabian StyleBertoglio, Pietro, Elena Garelli, Jury Brandolini, Kenji Kawamukai, Filippo Antonacci, Sara Ricciardi, Alessandro Cipolli, Barbara Bonfanti, Sergio Nicola Forti Parri, Niccolò Daddi, and et al. 2021. "Surgical Management and Reconstruction of Diaphragm, Pericardium and Chest Wall in Mesothelioma Surgery: A Review" Journal of Clinical Medicine 10, no. 11: 2330. https://doi.org/10.3390/jcm10112330
APA StyleBertoglio, P., Garelli, E., Brandolini, J., Kawamukai, K., Antonacci, F., Ricciardi, S., Cipolli, A., Bonfanti, B., Forti Parri, S. N., Daddi, N., Dolci, G., & Solli, P. (2021). Surgical Management and Reconstruction of Diaphragm, Pericardium and Chest Wall in Mesothelioma Surgery: A Review. Journal of Clinical Medicine, 10(11), 2330. https://doi.org/10.3390/jcm10112330






