The Role of Methemoglobin and Carboxyhemoglobin in COVID-19: A Review
Abstract
1. Introduction
2. Methods
3. Results
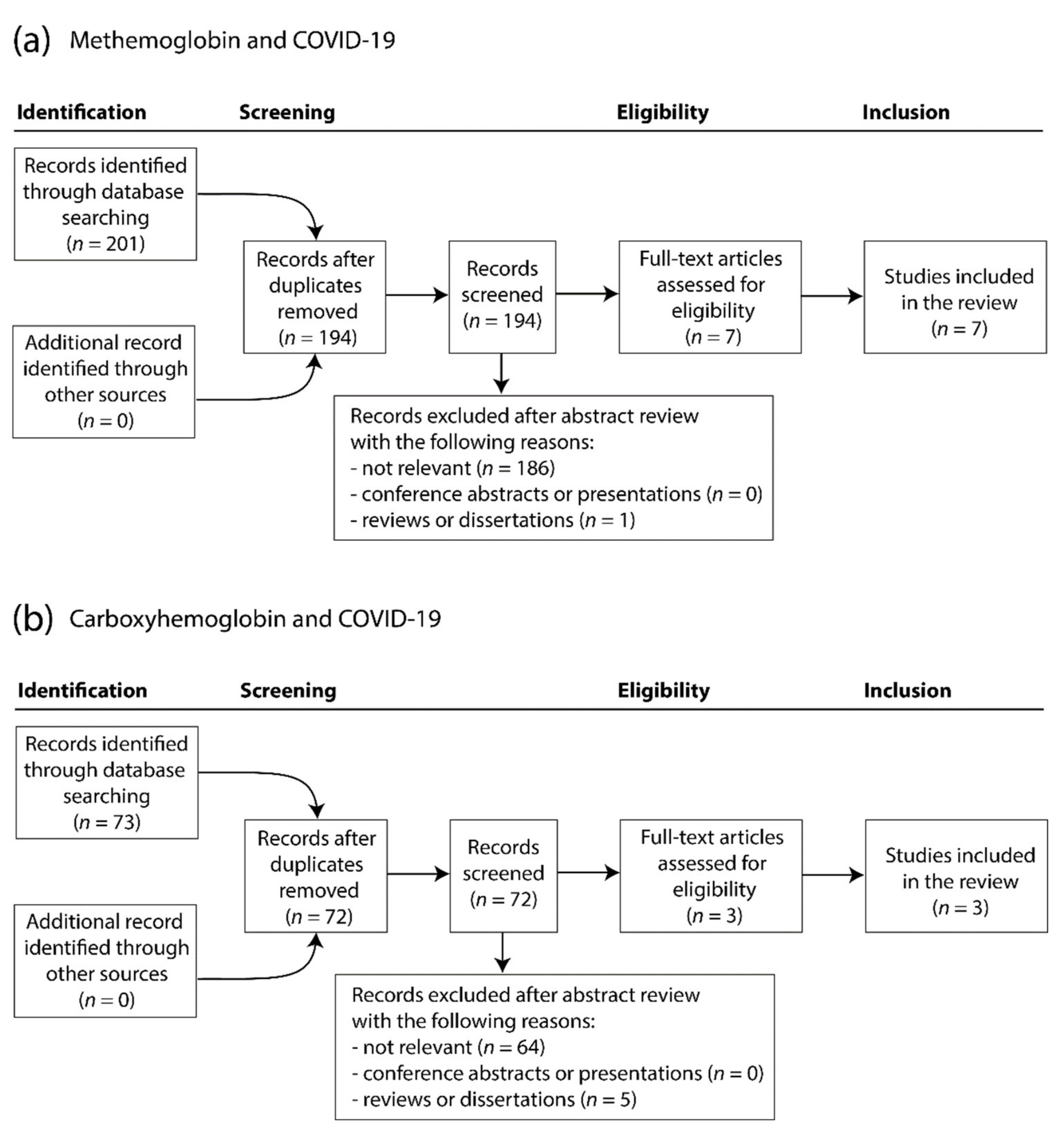
3.1. Literature Search Results
3.2. Characteristics of the Included Studies
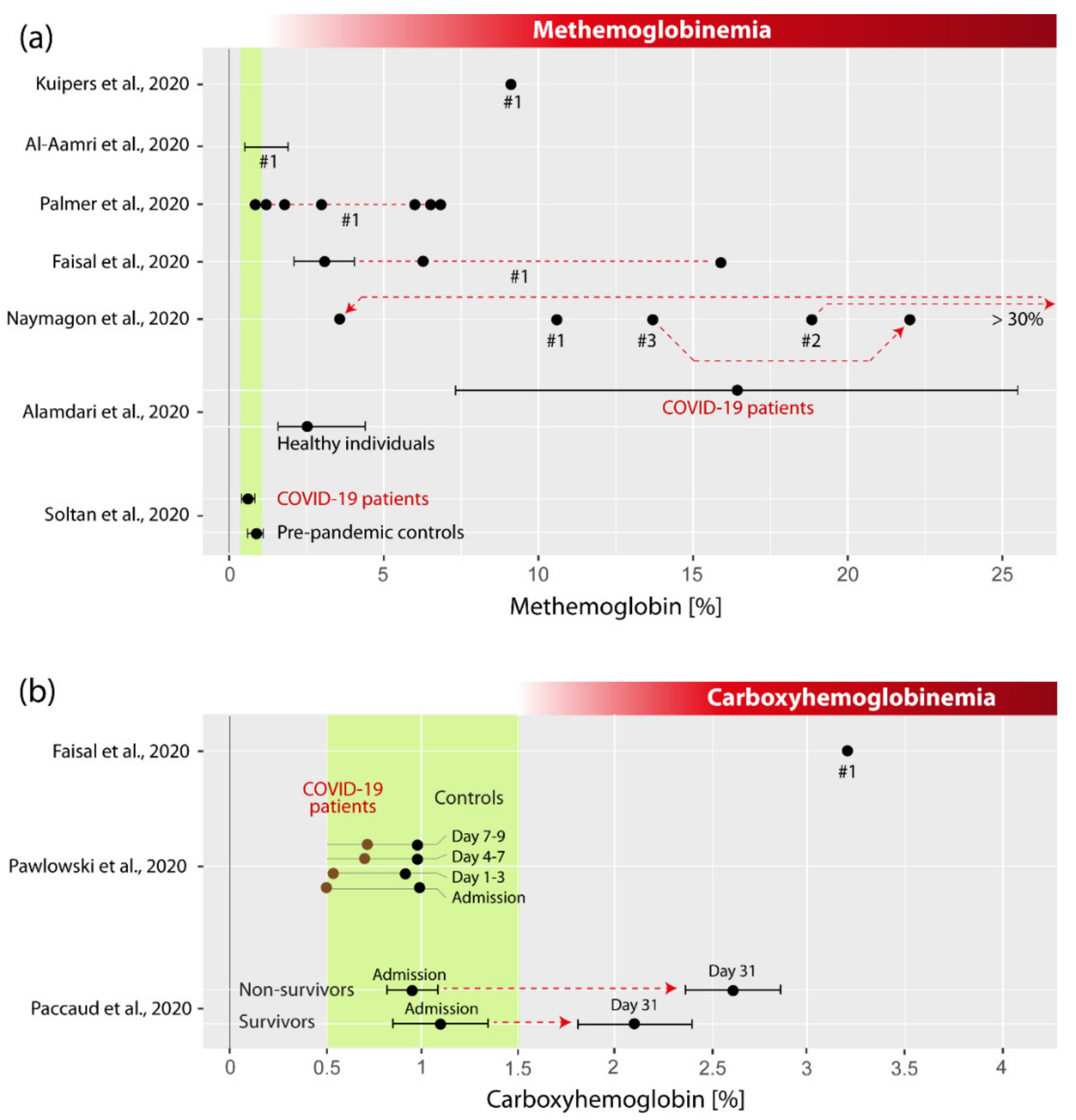
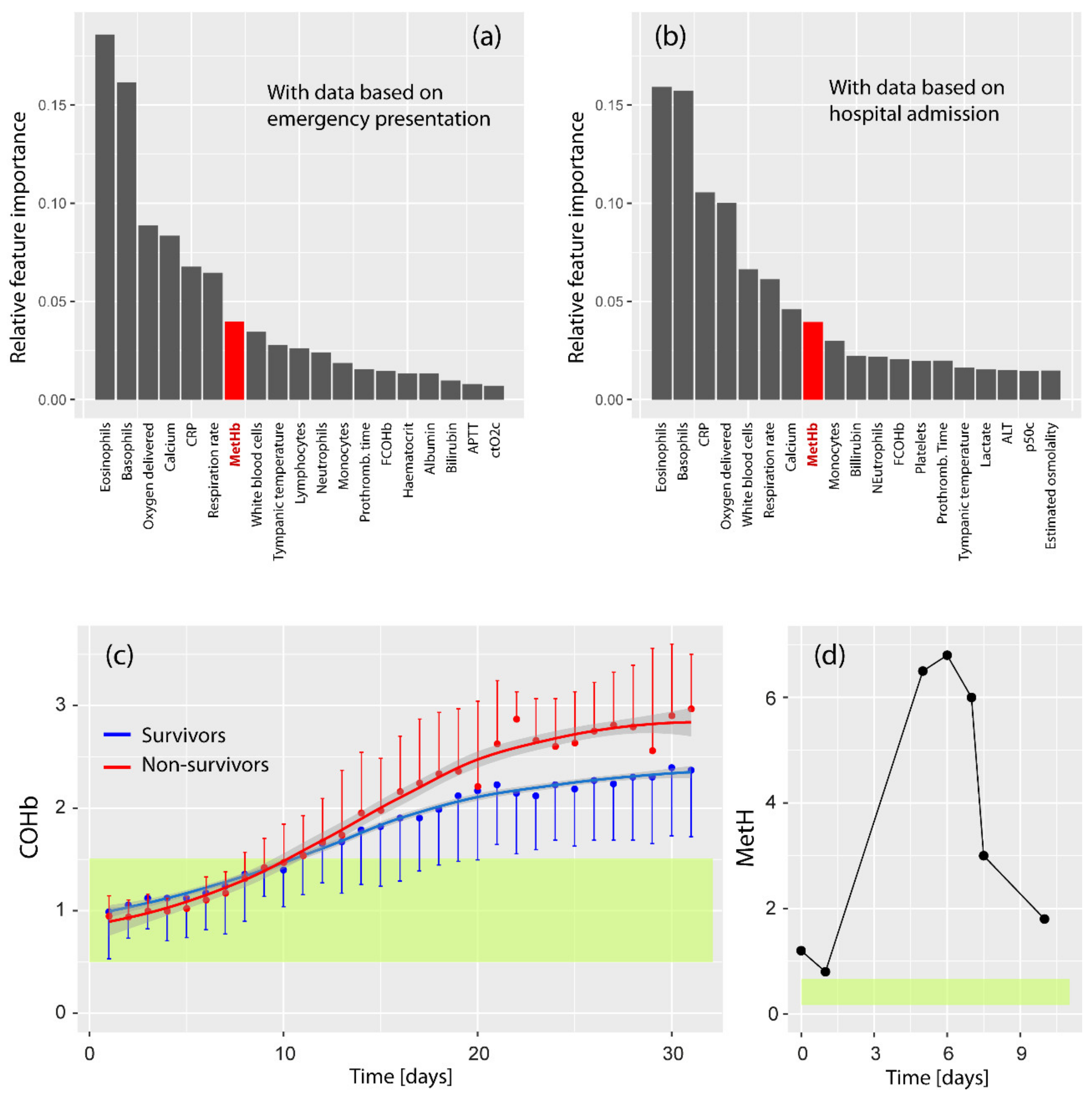
3.3. Summary of the Studies: MetHb and COVID-19
3.4. Summary of the Studies: COHb and COVID-19
4. Discussions
4.1. Is MetHb Increased in COVID-19 Patients?
4.2. Is COHb Increased in COVID-19 Patients?
4.3. Possible Reasons for Methemoglobinemia in COVID-19 Patients
4.4. Possible Reasons for Carboxyhemoglobinemia in COVID-19 Patients
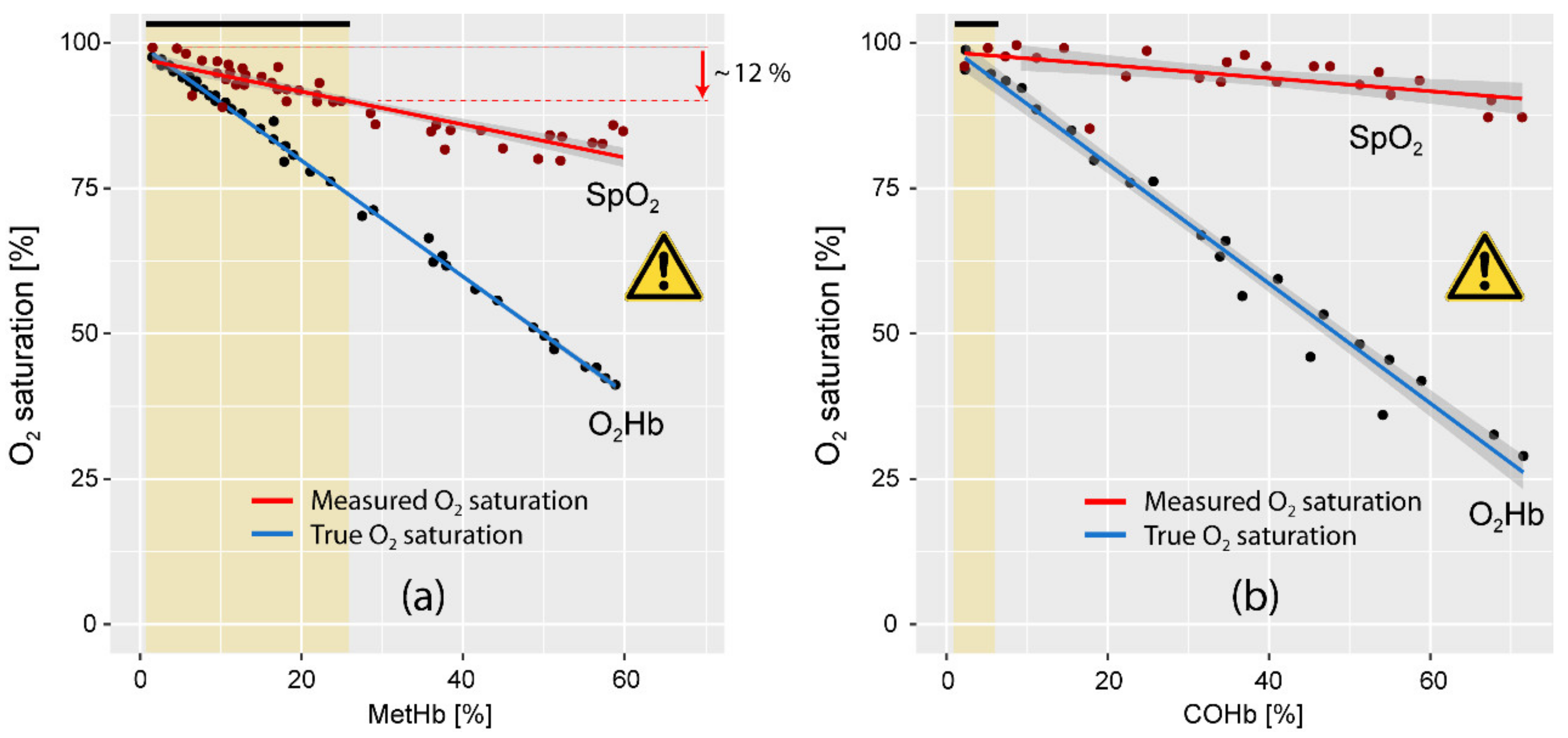
4.5. Methemoglobinemia and Carboxyhemoglobinemia in COVID-19 Patients: Consequences for Patient Monitoring and Treatment
5. Summary and Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wu, F.; Zhao, S.; Yu, B.; Chen, Y.-M.; Wang, W.; Song, Z.-G.; Hu, Y.; Tao, Z.-W.; Tian, J.-H.; Pei, Y.-Y.; et al. A new coronavirus associated with human respiratory disease in China. Nature 2020, 579, 265–269. [Google Scholar] [CrossRef]
- Zhou, P.; Yang, X.-L.; Wang, X.-G.; Hu, B.; Zhang, L.; Zhang, W.; Si, H.-R.; Zhu, Y.; Li, B.; Huang, C.-L.; et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 2020, 579, 270–273. [Google Scholar] [CrossRef]
- Lechien, J.R.; Chiesa-Estomba, C.M.; Place, S.; Van Laethem, Y.; Cabaraux, P.; Mat, Q.; Huet, K.; Plzak, J.; Horoi, M.; Hans, S.; et al. Clinical and epidemiological characteristics of 1420 European patients with mild-to-moderate coronavirus disease 2019. J. Intern. Med. 2020, 288, 335–344. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Guo, Y.; Mao, R.; Zhang, J. Proportion of asymptomatic coronavirus disease 2019: A systematic review and meta-analysis. J. Med. Virol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Yuki, K.; Fujiogi, M.; Koutsogiannaki, S. COVID-19 pathophysiology: A review. Clin. Immunol. 2020, 215, 108427. [Google Scholar] [CrossRef] [PubMed]
- Meyerowitz-Katz, G.; Merone, L. A systematic review and meta-analysis of published research data on COVID-19 infection-fatality rates. Int. J. Infect. Dis. 2020. [Google Scholar] [CrossRef]
- O’Driscoll, M.; Dos Santos, G.R.; Wang, L.; Cummings, D.A.T.; Azman, A.S.; Paireau, J.; Fontanet, A.; Cauchemez, S.; Salje, H. Age-specific mortality and immunity patterns of SARS-CoV-2. Nature 2020. [Google Scholar] [CrossRef]
- Krafte-Jacobs, B.; Brilli, R.; Szabo, C.; Denenberg, A.; Moore, L.; Salzman, A.L. Circulating methemoglobin and nitrite/nitrate concentrations as indicators of nitric oxide overproduction in critically ill children with septic shock. Crit. Care Med. 1997, 25, 1588–1593. [Google Scholar] [CrossRef]
- Wolak, E.; Byerly, F.L.; Mason, T.; Cairns, B.A. Methemoglobinemia in Critically Ill Burned Patients. Am. J. Crit. Care 2005, 14, 104–108. [Google Scholar] [CrossRef]
- Ohashi, K.; Yukioka, H.; Hayashi, M.; Asada, A. Elevated methemoglobin in patients with sepsis. Acta Anaesthesiol. Scand. 1998, 42, 713–716. [Google Scholar] [CrossRef]
- Melley, D.D.; Finney, S.J.; Elia, A.; Lagan, A.L.; Quinlan, G.J.; Evans, T.W. Arterial carboxyhemoglobin level and outcome in critically ill patients. Crit. Care Med. 2007, 35, 1882–1887. [Google Scholar] [CrossRef] [PubMed]
- Fazekas, A.S.; Wewalka, M.; Zauner, C.; Funk, G.-C. Carboxyhemoglobin levels in medical intensive care patients: A retrospective, observational study. Crit. Care 2012, 16, R6. [Google Scholar] [CrossRef] [PubMed]
- Umbreit, J. Methemoglobin—It’s not just blue: A concise review. Am. J. Hematol. 2007, 82, 134–144. [Google Scholar] [CrossRef] [PubMed]
- Hampson, N.B. Carboxyhemoglobin: A primer for clinicians. Undersea Hyperb. Med. 2018, 45, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Borland, C.; Harmes, K.; Cracknell, N.; Mack, D.; Higenbottam, T. Methemoglobin Levels in Smokers and Non-Smokers. Arch. Environ. Health Int. J. 1985, 40, 330–333. [Google Scholar] [CrossRef] [PubMed]
- Wilbur, S.; Williams, M.; Williams, R.; Scinicariello, F.; Klotzbach, J.M.; Diamond, G.L.; Citra, M. Toxicological Profile for Carbon Monoxide; Agency for Toxic Substances and Disease Registry (US): Atlanta, GA, USA, 2012.
- Kuipers, M.T.; Zwieten, R.; Heijmans, J.; Rutten, C.E.; Heer, K.; Kater, A.P.; Nur, E. Glucose-6-phosphate dehydrogenase deficiency-associated hemolysis and methemoglobinemia in a COVID-19 patient treated with chloroquine. Am. J. Hematol. 2020, 95. [Google Scholar] [CrossRef]
- Naymagon, L.; Berwick, S.; Kessler, A.; Lancman, G.; Gidwani, U.; Troy, K. The emergence of methemoglobinemia amidst the COVID-19 pandemic. Am. J. Hematol. 2020, 95. [Google Scholar] [CrossRef]
- Soltan, A.A.S.; Kouchaki, S.; Zhu, T.; Kiyasseh, D.; Taylor, T.; Hussain, Z.B.; Peto, T.; Brent, A.J.; Eyre, D.W.; Clifton, D. Artificial intelligence driven assessment of routinely collected healthcare data is an effective screening test for COVID-19 in patients presenting to hospital. MedRxiv 2020. [Google Scholar] [CrossRef]
- Faisal, H.; Bloom, A.; Gaber, A.O. Unexplained methemoglobinemia in coronavirus disease 2019: A case report. AA Pract. 2020, 14. [Google Scholar] [CrossRef]
- Alamdari, D.H.; Moghaddam, A.B.; Amini, S.; Keramati, M.R.; Zarmehri, A.M.; Alamdari, A.H.; Damsaz, M.; Banpour, H.; Yarahmadi, A.; Koliakos, G. Application of methylene blue -vitamin C –N-acetyl cysteine for treatment of critically ill COVID-19 patients, report of a phase-I clinical trial. Eur. J. Pharmacol. 2020, 885. [Google Scholar] [CrossRef]
- Palmer, K.; Dick, J.; French, W.; Floro, L.; Ford, M. Methemoglobinemia in patient with G6PD deficiency and SARS-CoV-2 Infection. Emerg. Infect. Dis. 2020, 26, 2279–2281. [Google Scholar] [CrossRef] [PubMed]
- Al-Aamri, M.A.; Al-Khars, F.T.; Alkhwaitem, S.J.; AlHassan, A.K.; Al Aithan, A.M.; Alkhalifa, F.H.; Al-Abdi, S.Y. A Saudi G6PD deficient girl died with pediatric multisystem inflammatory syndrome-COVID-19. MedRxiv 2020. [Google Scholar] [CrossRef]
- Paccaud, P.; Castanares-Zapatero, D.; Gerard, L.; Montiel, V.; Wittebole, X.; Collienne, C.; Laterre, P.-F.; Hantson, P. Arterial carboxyhemoglobin levels in Covid-19 critically Ill patients. Res. Sq. 2020. [Google Scholar] [CrossRef]
- Pawlowski, C.; Wagner, T.; Puranik, A.; Murugadoss, K.; Loscalzo, L.; Venkatakrishnan, A.J.; Pruthi, R.K.; Houghton, D.E.; O’Horo, J.C.; Morice, W.G.; et al. Inference from longitudinal laboratory tests characterizes temporal evolution of COVID-19-associated coagulopathy (CAC). eLife 2020, 9. [Google Scholar] [CrossRef]
- Alanazi, M.Q. Drugs may be Induced Methemoglobinemia. J. Hematol. Thromboembolic Dis. 2017, 6. [Google Scholar] [CrossRef]
- Bodansky, O. Methemoglobinemia and methemoglobin-producing compounds. Pharmacol. Rev. 1951, 3, 144–196. [Google Scholar]
- Cefalu, J.N.; Joshi, T.V.; Spalitta, M.J.; Kadi, C.J.; Diaz, J.H.; Eskander, J.P.; Cornett, E.M.; Kaye, A.D. Methemoglobinemia in the operating room and intensive care unit: Early recognition, pathophysiology, and management. Adv. Ther. 2020, 37, 1714–1723. [Google Scholar] [CrossRef]
- Rizvi, I.; Zaman, S.; Zaidi, N.; Asif, S.M.; Abdali, N. Acute life-threatening methaemoglobinaemia following ingestion of chloroquine. Case Rep. 2012, 2012. [Google Scholar] [CrossRef]
- Cohen, R.J.; Sachs, J.R.; Wicker, D.J.; Conrad, M.E. Methemoglobinemia provoked by malarial chemoprophylaxis in Vietnam. N. Engl. J. Med. 1968, 279, 1127–1131. [Google Scholar] [CrossRef]
- Sharma, N.; Varma, S. Unusual life-threatening adverse drug effects with chloroquine in a young girl. J. Postgrad Med. 2003, 49, 187. [Google Scholar]
- Singh, A.K.; Singh, A.; Shaikh, A.; Singh, R.; Misra, A. Chloroquine and hydroxychloroquine in the treatment of COVID-19 with or without diabetes: A systematic search and a narrative review with a special reference to India and other developing countries. Diabetes Metab. Syndr. Clin. Res. Rev. 2020, 14, 241–246. [Google Scholar] [CrossRef] [PubMed]
- Gbinigie, K.; Frie, K. Should chloroquine and hydroxychloroquine be used to treat COVID-19? A rapid review. Bjgp Open 2020, 4. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.; Das, S.; Jain, A.; Misra, D.P.; Negi, V.S. A systematic review of the prophylactic role of chloroquine and hydroxychloroquine in coronavirus disease-19 (COVID-19). Int. J. Rheum. Dis. 2020, 23, 613–619. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, A.V.; Roman, Y.M.; Pasupuleti, V.; Barboza, J.J.; White, C.M. Hydroxychloroquine or Chloroquine for treatment or prophylaxis of COVID-19. Ann. Intern. Med. 2020, 173, 287–296. [Google Scholar] [CrossRef]
- Hashem, A.M.; Alghamdi, B.S.; Algaissi, A.A.; Alshehri, F.S.; Bukhari, A.; Alfaleh, M.A.; Memish, Z.A. Therapeutic use of chloroquine and hydroxychloroquine in COVID-19 and other viral infections: A narrative review. Travel Med. Infect. Dis. 2020, 35. [Google Scholar] [CrossRef]
- Pathak, D.S.K.; Salunke, D.A.A.; Thivari, D.P.; Pandey, A.; Nandy, D.K.; Harish, V.K.; Ratna, D.; Pandey, D.S.; Chawla, D.J.; Mujawar, D.J.; et al. No benefit of hydroxychloroquine in COVID-19: Results of systematic review and meta-analysis of randomized controlled trials”. Diabetes Metab. Syndr. Clin. Res. Rev. 2020, 14, 1673–1680. [Google Scholar] [CrossRef]
- Singh, A.K.; Singh, A.; Singh, R.; Misra, A. “Hydroxychloroquine in patients with COVID-19: A systematic review and meta-analysis”. Diabetes Metab. Syndr. Clin. Res. Rev. 2020, 14, 589–596. [Google Scholar] [CrossRef]
- Kashour, Z.; Riaz, M.; Garbati, M.A.; AlDosary, O.; Tlayjeh, H.; Gerberi, D.; Murad, M.H.; Sohail, M.R.; Kashour, T.; Tleyjeh, I.M. Efficacy of chloroquine or hydroxychloroquine in COVID-19 patients: A systematic review and meta-analysis. J. Antimicrob. Chemother. 2020. [Google Scholar] [CrossRef]
- Brewer, G.J. Rediscovery of the susceptibility of G6PD deficient persons to methemoglobinemia from oxidant drugs, and to hemolysis from methylene blue. Am. J. Hematol. 2007, 82, 87–88. [Google Scholar] [CrossRef]
- Zuchelkowski, B.E.; Wang, L.; Gingras, S.; Xu, Q.; Yang, M.; Triulzi, D.; Page, G.P.; Gordeuk, V.R.; Kim-Shapiro, D.B.; Lee, J.S.; et al. Brief report: Hydroxychloroquine does not induce hemolytic anemia or organ damage in a “humanized” G6PD A- mouse model. PLoS ONE 2020, 15, e0240266. [Google Scholar] [CrossRef]
- Hundsdoerfer, P.; Vetter, B.; Kulozik, A.E. Chronic Haemolytic Anaemia and Glucose-6-Phosphate Dehydrogenase deficiency. Acta Haematol. 2002, 108, 102–105. [Google Scholar] [CrossRef] [PubMed]
- Ramirez de Oleo, I.E.; Mejia Saldarriaga, M.; Johnson, B.K. Association of hydroxychloroquine use and Hemolytic Anemia in patients with low levels of Glucose-6-Phosphate Dehydrogenase. J. Clin. Rheumatol. Pract. Rep. Rheum. Musculoskelet. Dis. 2020. [Google Scholar] [CrossRef]
- Beauverd, Y.; Adam, Y.; Assouline, B.; Samii, K. COVID-19 infection and treatment with hydroxychloroquine cause severe haemolysis crisis in a patient with glucose-6-phosphate dehydrogenase deficiency. Eur. J. Haematol. 2020, 105, 357–359. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, J. Hemolytic anemia in a Glucose-6-Phosphate Dehydrogenase-deficient patient receiving hydroxychloroquine for COVID-19: A case report. Perm. J. 2020, 24. [Google Scholar] [CrossRef]
- Chaney, S.; Basirat, A.; McDermott, R.; Keenan, N.; Moloney, E. COVID-19 and hydroxychloroquine side-effects: Glucose 6-phosphate dehydrogenase deficiency (G6PD) and acute haemolytic anaemia. QJM Int. J. Med. 2020. [Google Scholar] [CrossRef]
- Mastroianni, F.; Colombie, V.; Claes, G.; Gilles, A.; Vandergheynst, F.; Place, S. Hydroxychloroquine in a G6PD-Deficient patient with COVID-19 complicated by haemolytic anaemia: Culprit or innocent bystander? Eur. J. Case Rep. Intern. Med. 2020, 7, 001875. [Google Scholar] [CrossRef]
- Afra, T.P.; Vasudevan Nampoothiri, R.; Razmi T., M. Doubtful precipitation of hemolysis by hydroxychloroquine in glucose-6-phosphate dehydrogenase-deficient patient with COVID-19 infection. Eur. J. Haematol. 2020, 105, 512–513. [Google Scholar] [CrossRef]
- Rosen, P.J. Failure of Methylene Blue treatment in toxic methemoglobinemia. Ann. Intern. Med. 1971, 75. [Google Scholar] [CrossRef]
- Sikka, P.; Kapoor, S.; Saxena, K.K.; Bindra, V.K.; Jain, V. Blue cures blue but be cautious. J. Pharm. Bioallied Sci. 2011, 3, 543. [Google Scholar] [CrossRef]
- Hare, G.M.T.; Tsui, A.K.Y.; Crawford, J.H.; Patel, R.P. Is methemoglobin an inert bystander, biomarker or a mediator of oxidative stress—The example of anemia? Redox Biol. 2013, 1, 65–69. [Google Scholar] [CrossRef]
- Jurado, R.L. Iron, infections, and anemia of inflammation. Clin. Infect. Dis. 1997, 25, 888–895. [Google Scholar] [CrossRef] [PubMed]
- Ganz, T.; Longo, D.L. Anemia of Inflammation. New Engl. J. Med. 2019, 381, 1148–1157. [Google Scholar] [CrossRef] [PubMed]
- Bellmann-Weiler, R.; Lanser, L.; Barket, R.; Rangger, L.; Schapfl, A.; Schaber, M.; Fritsche, G.; Wöll, E.; Weiss, G. Prevalence and predictive value of anemia and dysregulated iron homeostasis in patients with COVID-19 infection. J. Clin. Med. 2020, 9, 2429. [Google Scholar] [CrossRef] [PubMed]
- Lazarian, G.; Quinquenel, A.; Bellal, M.; Siavellis, J.; Jacquy, C.; Re, D.; Merabet, F.; Mekinian, A.; Braun, T.; Damaj, G.; et al. Autoimmune haemolytic anaemia associated with COVID-19 infection. Br. J. Haematol. 2020, 190, 29–31. [Google Scholar] [CrossRef]
- Taneri, P.E.; Gómez-Ochoa, S.A.; Llanaj, E.; Raguindin, P.F.; Rojas, L.Z.; Roa-Díaz, Z.M.; Salvador, D.; Groothof, D.; Minder, B.; Kopp-Heim, D.; et al. Anemia and iron metabolism in COVID-19: A systematic review and meta-analysis. Eur. J. Epidemiol. 2020, 35, 763–773. [Google Scholar] [CrossRef]
- Hare, G.M.T.; Mu, A.; Romaschin, A.; Tsui, A.K.Y.; Shehata, N.; Beattie, W.S.; Mazer, C.D. Plasma methemoglobin as a potential biomarker of anemic stress in humans. Can. J. Anesth. J. Can. D’anesthésie 2012, 59, 348–356. [Google Scholar] [CrossRef]
- Nagababu, E.; Gulyani, S.; Earley, C.J.; Cutler, R.G.; Mattson, M.P.; Rifkind, J.M. Iron-deficiency anaemia enhances red blood cell oxidative stress. Free Radic. Res. 2009, 42, 824–829. [Google Scholar] [CrossRef]
- Au, W.Y.; Ngai, C.-W.; Chan, W.-M.; Leung, R.Y.Y.; Chan, S.-C. Hemolysis and methemoglobinemia due to hepatitis E virus infection in patient with G6PD deficiency. Ann. Hematol. 2011, 90, 1237–1238. [Google Scholar] [CrossRef]
- Avner, J.R. Severe methemoglobinemia in a 3-week-old infant with a urinary tract infection. Crit. Care Med. 1992, 20. [Google Scholar] [CrossRef]
- Saxena, N.; Puri, S.K.; Sahni, S.K.; Dutta, G.P.; Pandey, V.C. Studies on methemoglobin and methemoglobin reductase in monkey erythrocytes during Plasmodium knowlesi infection. Clin. Chem. Enzymol. Commun. 1989, 2, 55–59. [Google Scholar]
- Srivastava, S.; Alhomida, A.S.; Siddiqi, N.J. Studies on erythrocytic methemoglobin reductase systems in Plasmodium yoelii nigeriensis infected mice. In Vivo 2000, 14, 547–550. [Google Scholar] [PubMed]
- Srivastava, S.; Alhomida, A.S.; Siddiqi, N.J.; Pandey, V.C. Changes in rodent-erythrocyte methemoglobin reductase system produced by two malaria parasites, viz. Plasmodium yoelii nigeriensis and Plasmodium berghei. Comp. Biochem. Physiol. Part. B Biochem. Mol. Biol. 2001, 129, 725–731. [Google Scholar] [CrossRef]
- Tyan, M.L. Age-related increase in erythrocyte oxidant sensitivity. Mech. Ageing Dev. 1982, 20, 25–32. [Google Scholar] [CrossRef]
- Liu, X.; Spolarics, Z. Methemoglobin is a potent activator of endothelial cells by stimulating IL-6 and IL-8 production and E-selectin membrane expression. Am. J. Physiol. Cell Physiol. 2003, 285, C1036–C1046. [Google Scholar] [CrossRef] [PubMed]
- Mumby, S.; Ramakrishnan, L.; Evans, T.W.; Griffiths, M.J.D.; Quinlan, G.J. Methemoglobin-induced signaling and chemokine responses in human alveolar epithelial cells. Am. J. Physiol. Lung Cell. Mol. Physiol. 2014, 306, L88–L100. [Google Scholar] [CrossRef]
- Vaninov, N. In the eye of the COVID-19 cytokine storm. Nat. Rev. Immunol. 2020, 20, 277. [Google Scholar] [CrossRef]
- Ragab, D.; Salah Eldin, H.; Taeimah, M.; Khattab, R.; Salem, R. The COVID-19 cytokine storm; what we know so far. Front. Immunol. 2020, 11. [Google Scholar] [CrossRef]
- Jose, R.J.; Manuel, A. COVID-19 cytokine storm: The interplay between inflammation and coagulation. Lancet Respir. Med. 2020, 8, e46–e47. [Google Scholar] [CrossRef]
- Mangalmurti, N.; Hunter, C.A. Cytokine storms: Understanding COVID-19. Immunity 2020, 53, 19–25. [Google Scholar] [CrossRef]
- Tamm, M.; Bihl, M.; Eickelberg, O.; Stulz, P.; Perruchoud, A.P.; Roth, M. Hypoxia-induced Interleukin-6 and Interleukin-8 production is mediated by platelet-activating factor and platelet-derived growth factor in primary human lung cells. Am. J. Respir. Cell Mol. Biol. 1998, 19, 653–661. [Google Scholar] [CrossRef]
- Eltzschig, H.K.; Schwartz, R.S.; Carmeliet, P. Hypoxia and inflammation. N. Engl. J. Med. 2011, 364, 656–665. [Google Scholar] [CrossRef] [PubMed]
- Moncure, M.; Brathwaite, C.E.M.; Samaha, E.; Marburger, R.; Ross, S.E. Carboxyhemoglobin elevation in trauma victims. J. Trauma Inj. Infect. Crit. Care 1999, 46, 424–427. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, H. Increased blood carboxyhaemoglobin concentrations in inflammatory pulmonary diseases. Thorax 2002, 57, 779–783. [Google Scholar] [CrossRef] [PubMed]
- Hampson, N.B. Carboxyhemoglobin elevation due to hemolytic anemia. J. Emerg. Med. 2007, 33, 17–19. [Google Scholar] [CrossRef]
- Zegdi, R.; Caïd, R.; Van De Louw, A.; Perrin, D.; Burdin, M.; Boiteau, R.; Tenaillon, A. Exhaled carbon monoxide in mechanically ventilated critically ill patients: Influence of inspired oxygen fraction. Intensive Care Med. 2014, 26, 1228–1231. [Google Scholar] [CrossRef]
- Kakavas, S.; Papanikolaou, A.; Ballis, E.; Tatsis, N.; Goga, C.; Tatsis, G. Carboxyhemoglobin and methemoglobin levels as prognostic markers in acute pulmonary embolism. Am. J. Emerg. Med. 2015, 33, 563–568. [Google Scholar] [CrossRef]
- Paine, A.; Eiz-Vesper, B.; Blasczyk, R.; Immenschuh, S. Signaling to heme oxygenase-1 and its anti-inflammatory therapeutic potential. Biochem. Pharmacol. 2010, 80, 1895–1903. [Google Scholar] [CrossRef]
- Dorresteijn, M.; Pickkers, P. Carboxyhemoglobin levels during human inflammation. Crit. Care 2012, 16, 24. [Google Scholar] [CrossRef][Green Version]
- Schimmel, J.; George, N.; Schwarz, J.; Yousif, S.; Suner, S.; Hack, J.B. Carboxyhemoglobin levels induced by cigarette smoking outdoors in smokers. J. Med Toxicol. 2017, 14, 68–73. [Google Scholar] [CrossRef]
- Deller, A.; Stenz, R.; Forstner, K.; Schreiber, M.N.; Konrad, F.; Fosel, T. [Carbomonoxyhemoglobin and methemoglobin in patients with and without a smoking history during ambulatory anesthesia. Consequences for the use of pulse oximetry]. Anasthesiol. Intensivmed. Notf. Schmerzther. 1991, 26, 186–190. [Google Scholar] [CrossRef]
- Gavrilovska-Brzanov, A.; Shosholcheva, M.; Kuzmanovska, B.; Kartalov, A.; MojsovaMijovska, M.; JovanovskiSrceva, M.; Taleska, G.; Brzanov, N.; Simeonov, R.; Miceska, M. The influence of smoking on the variations in carboxyhemoglobin and methemoglobin during urologic surgery. Med Arch. 2017, 71. [Google Scholar] [CrossRef]
- Annabi, E.H.; Barker, S.J. Severe methemoglobinemia detected by pulse oximetry. Anesth. Analg. 2009, 108, 898–899. [Google Scholar] [CrossRef] [PubMed]
- Hampson, N.B. Noninvasive pulse CO-oximetry expedites evaluation and management of patients with carbon monoxide poisoning. Am. J. Emerg. Med. 2012, 30, 2021–2024. [Google Scholar] [CrossRef] [PubMed]
- Frasca, D.; Dahyot-Fizelier, C.; Catherine, K.; Levrat, Q.; Debaene, B.; Mimoz, O. Accuracy of a continuous noninvasive hemoglobin monitor in intensive care unit patients*. Crit. Care Med. 2011, 39, 2277–2282. [Google Scholar] [CrossRef]
- Barker, S.J.; Tremper, K.K.; Hyatt, J. Effects of methemoglobinemia on pulse oximetry and mixed venous oximetry. Anesthesiology 1989, 70, 112–117. [Google Scholar] [CrossRef]
- Barker, S.J.; Tremper, K.K. The effect of carbon monoxide inhalation on pulse oximetry and transcutaneous Po2. Anesthesiology 1987, 66, 677–679. [Google Scholar] [CrossRef]
- Dhont, S.; Derom, E.; Van Braeckel, E.; Depuydt, P.; Lambrecht, B.N. The pathophysiology of ‘happy’ hypoxemia in COVID-19. Respir. Res. 2020, 21. [Google Scholar] [CrossRef]
- Tobin, M.J.; Laghi, F.; Jubran, A. Why COVID-19 silent hypoxemia is baffling to physicians. Am. J. Respir. Crit. Care Med. 2020, 202, 356–360. [Google Scholar] [CrossRef]
- Mæhlen, J.O.; Seim, M.; Ottestad, W. Covid-19 med stille hypoksemi. Tidsskr. Den. Nor. Legeforening 2020. [Google Scholar] [CrossRef]
- Tobin, M.J.; Jubran, A.; Laghi, F. Misconceptions of pathophysiology of happy hypoxemia and implications for management of COVID-19. Respir. Res. 2020, 21. [Google Scholar] [CrossRef]
- Bek, M.; Laule, S.; Reichert-Jünger, C.; Holtkamp, R.; Wiesner, M.; Keyl, C. Methemoglobinemia in critically ill patients during extended hemodialysis and simultaneous disinfection of the hospital water supply. Crit. Care 2009, 13, R162. [Google Scholar] [CrossRef] [PubMed]
- Uchida, I.; Tashiro, C.; Koo, Y.H.; Mashimo, T.; Yoshiya, I. Carboxyhemoglobin and methemoglobin levels in banked blood. J. Clin. Anesth. 1990, 2, 86–90. [Google Scholar] [CrossRef]
- Boehm, R.E.; Arbo, B.D.; Leal, D.; Hansen, A.W.; Pulcinelli, R.R.; Thiesen, F.V.; Balsan, A.M.; Onsten, T.G.H.; Gomez, R. Smoking fewer than 20 cigarettes per day and remaining abstinent for more than 12 hours reduces carboxyhemoglobin levels in packed red blood cells for transfusion. PLoS ONE 2018, 13, e0204102. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scholkmann, F.; Restin, T.; Ferrari, M.; Quaresima, V. The Role of Methemoglobin and Carboxyhemoglobin in COVID-19: A Review. J. Clin. Med. 2021, 10, 50. https://doi.org/10.3390/jcm10010050
Scholkmann F, Restin T, Ferrari M, Quaresima V. The Role of Methemoglobin and Carboxyhemoglobin in COVID-19: A Review. Journal of Clinical Medicine. 2021; 10(1):50. https://doi.org/10.3390/jcm10010050
Chicago/Turabian StyleScholkmann, Felix, Tanja Restin, Marco Ferrari, and Valentina Quaresima. 2021. "The Role of Methemoglobin and Carboxyhemoglobin in COVID-19: A Review" Journal of Clinical Medicine 10, no. 1: 50. https://doi.org/10.3390/jcm10010050
APA StyleScholkmann, F., Restin, T., Ferrari, M., & Quaresima, V. (2021). The Role of Methemoglobin and Carboxyhemoglobin in COVID-19: A Review. Journal of Clinical Medicine, 10(1), 50. https://doi.org/10.3390/jcm10010050






