Towards Biohydrogen Separation Using Poly(Ionic Liquid)/Ionic Liquid Composite Membranes
Abstract
1. Introduction
2. Experimental Section
2.1. Materials
2.2. Preparation of PIL–IL Membranes
2.3. Gas Permeation Experiments
3. Results and Discussion
3.1. CO2 and H2 Permeation Properties
3.1.1. Gas Permeability (P)
3.1.2. Gas Diffusivity (D)
3.1.3. Gas Solubility (S)
3.2. CO2/H2 Separation Performance
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Δpd | Variation of downstream pressure |
| Δpi | Pressure driving force |
| A | Effective membrane surface area |
| bioH2 | Biohydrogen |
| CO2 | Carbon dioxide |
| D | Diffusivity |
| H2 | Hydrogen |
| H2O | Water |
| H2S | Hydrogen sulfide |
| ILs | Ionic liquids |
| Ji | Steady-state gas flux |
| ℓ | Membrane thickness |
| N2 | Nitrogen |
| O2 | Oxygen |
| P | Permeability |
| PILs | Poly(ionic liquid)s |
| R | Ideal gas law constant |
| S | Solubility |
| t | Time |
| T | Temperature |
| Vp | Permeate volume |
| αi/j | Permselectivity |
| θ | Time-lag parameter |
| Cations | |
| [C2mim]+ | 1-ethyl-3-methylimidazolium |
| [C4mpyr]+ | 1-butyl-3-methylpyrrolidinium |
| Anions | |
| [NTf2]− | Bis(tri-fluoromethylsulfonyl)imide |
| [C(CN)3]− | Tricyanomethanide |
References
- Mazloomi, K.; Gomes, C. Hydrogen as an energy carrier: Prospects and challenges. Renew. Sustain. Energy Rev. 2012, 16, 3024–3033. [Google Scholar] [CrossRef]
- Das, D.; Veziroǧlu, T.N. Hydrogen production by biological processes: A survey of literature. Int. J. Hydrog. Energy 2001, 26, 13–28. [Google Scholar] [CrossRef]
- Kalamaras, C.M.; Efstathiou, A.M. Hydrogen Production Technologies: Current State and Future Developments. Conf. Pap. Energy 2013, 2013, 9. [Google Scholar] [CrossRef]
- Das, D.; Veziroglu, T.N. Advances in biological hydrogen production processes. Int. J. Hydrog. Energy 2008, 33, 6046–6057. [Google Scholar] [CrossRef]
- Manish, S.; Banerjee, R. Comparison of biohydrogen production processes. Int. J. Hydrog. Energy 2008, 33, 279–286. [Google Scholar] [CrossRef]
- Singh, L.; Wahid, Z.A. Methods for enhancing bio-hydrogen production from biological process: A review. J. Ind. Eng. Chem. 2015, 21, 70–80. [Google Scholar] [CrossRef]
- Alves, H.J.; Bley Junior, C.; Niklevicz, R.R.; Frigo, E.P.; Frigo, M.S.; Coimbra-Araújo, C.H. Overview of hydrogen production technologies from biogas and the applications in fuel cells. Int. J. Hydrog. Energy 2013, 38, 5215–5225. [Google Scholar] [CrossRef]
- Merkel, T.C.; Zhou, M.; Baker, R.W. Carbon dioxide capture with membranes at an IGCC power plant. J. Memb. Sci. 2012, 389, 441–450. [Google Scholar] [CrossRef]
- Dunikov, D.; Borzenko, V.; Blinov, D.; Kazakov, A.; Lin, C.Y.; Wu, S.Y.; Chu, C.Y. Biohydrogen purification using metal hydride technologies. Int. J. Hydrog. Energy 2016, 41, 21787–21794. [Google Scholar] [CrossRef]
- Sanders, D.F.; Smith, Z.P.; Guo, R.; Robeson, L.M.; McGrath, J.E.; Paul, D.R.; Freeman, B.D. Energy-efficient polymeric gas separation membranes for a sustainable future: A review. Polymer 2013, 54, 4729–4761. [Google Scholar] [CrossRef]
- Bakonyi, P.; Nemestóthy, N.; Ramirez, J.; Ruiz-Filippi, G.; Bélafi-Bakó, K. Escherichia coli (XL1-BLUE) for continuous fermentation of bioH2 and its separation by polyimide membrane. Int. J. Hydrog. Energy 2012, 37, 5623–5630. [Google Scholar] [CrossRef]
- Bakonyi, P.; Kumar, G.; Nemestóthy, N.; Lin, C.Y.; Bélafi-Bakó, K. Biohydrogen purification using a commercial polyimide membrane module: Studying the effects of some process variables. Int. J. Hydrog. Energy 2013, 38, 15092–15099. [Google Scholar] [CrossRef]
- Bakonyi, P.; Nemestóthy, N.; Bélafi-Bakó, K. Biohydrogen purification by membranes: An overview on the operational conditions affecting the performance of non-porous, polymeric and ionic liquid based gas separation membranes. Int. J. Hydrog. Energy 2013, 38, 9673–9687. [Google Scholar] [CrossRef]
- Basu, S.; Khan, A.L.; Cano-Odena, A.; Liu, C.; Vankelecom, I.F.J. Membrane-based technologies for biogas separations. Chem. Soc. Rev. 2010, 39, 750–768. [Google Scholar] [CrossRef] [PubMed]
- Mohamad, I.N.; Rohani, R.; Mastar, M.S.; Nor, M.T.M.; Md. Jahim, J. Permeation properties of polymeric membranes for biohydrogen purification. Int. J. Hydrog. Energy 2016, 41, 4474–4488. [Google Scholar] [CrossRef]
- Pientka, Z.; Peter, J.; Zitka, J.; Bakonyi, P. Application of Polymeric Membranes in Biohydrogen Purification and Storage. Curr. Biochem. Eng. 2014, 1, 99–105. [Google Scholar] [CrossRef]
- Kanehashi, S.; Kishida, M.; Kidesaki, T.; Shindo, R.; Sato, S.; Miyakoshi, T.; Nagai, K. CO2 separation properties of a glassy aromatic polyimide composite membranes containing high-content 1-butyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide ionic liquid. J. Memb. Sci. 2013, 430, 211–222. [Google Scholar] [CrossRef]
- Friess, K.; Jansen, J.C.; Bazzarelli, F.; Izák, P.; Jarmarová, V.; Kačírková, M.; Schauer, J.; Clarizia, G.; Bernardo, P. High ionic liquid content polymeric gel membranes: Correlation of membrane structure with gas and vapour transport properties. J. Memb. Sci. 2012, 415–416, 801–809. [Google Scholar] [CrossRef]
- Yuan, J.; Mecerreyes, D.; Antonietti, M. Poly(ionic liquid)s: An update. Prog. Polym. Sci. 2013, 38, 1009–1036. [Google Scholar] [CrossRef]
- Jeffrey Horne, W.; Andrews, M.A.; Shannon, M.S.; Terrill, K.L.; Moon, J.D.; Hayward, S.S.; Bara, J.E. Effect of branched and cycloalkyl functionalities on CO2 separation performance of poly(IL) membranes. Sep. Purif. Technol. 2015, 155, 89–95. [Google Scholar] [CrossRef]
- Bara, J.E.; Gabriel, C.J.; Hatakeyama, E.S.; Carlisle, T.K.; Lessmann, S.; Noble, R.D.; Gin, D.L. Improving CO2 selectivity in polymerized room-temperature ionic liquid gas separation membranes through incorporation of polar substituents. J. Memb. Sci. 2008, 321, 3–7. [Google Scholar] [CrossRef]
- Bara, J.E.; Hatakeyama, E.S.; Gin, D.L.; Noble, R.D. Improving CO2 permeability in polymerized room-temperature ionic liquid gas separation membranes through the formation of a solid composite with a room-temperature ionic liquid. Polym. Adv. Technol. 2008, 19, 1415–1420. [Google Scholar] [CrossRef]
- Bara, J.E.; Lessmann, S.; Gabriel, C.J.; Hatakeyama, E.S.; Noble, R.D.; Gin, D.L. Synthesis and Performance of Polymerizable Room-Temperature Ionic Liquids as Gas Separation Membranes. Ind. Eng. Chem. Res. 2007, 46, 5397–5404. [Google Scholar] [CrossRef]
- Hu, X.; Tang, J.; Blasig, A.; Shen, Y.; Radosz, M. CO2 permeability, diffusivity and solubility in polyethylene glycol-grafted polyionic membranes and their CO2 selectivity relative to methane and nitrogen. J. Memb. Sci. 2006, 281, 130–138. [Google Scholar] [CrossRef]
- Hao, L.; Li, P.; Yang, T.; Chung, T.-S. Room temperature ionic liquid/ZIF-8 mixed-matrix membranes for natural gas sweetening and post-combustion CO2 capture. J. Memb. Sci. 2013, 436, 221–231. [Google Scholar] [CrossRef]
- Hudiono, Y.C.; Carlisle, T.K.; LaFrate, A.L.; Gin, D.L.; Noble, R.D. Novel mixed matrix membranes based on polymerizable room-temperature ionic liquids and SAPO-34 particles to improve CO2 separation. J. Memb. Sci. 2011, 370, 141–148. [Google Scholar] [CrossRef]
- Hudiono, Y.C.; Carlisle, T.K.; Bara, J.E.; Zhang, Y.; Gin, D.L.; Noble, R.D. A three-component mixed-matrix membrane with enhanced CO2 separation properties based on zeolites and ionic liquid materials. J. Memb. Sci. 2010, 350, 117–123. [Google Scholar] [CrossRef]
- Tomé, L.C.; Marrucho, I.M. Ionic liquid-based materials: A platform to design engineered CO2 separation membranes. Chem. Soc. Rev. 2016, 45, 2785–2824. [Google Scholar] [CrossRef] [PubMed]
- Sadeghpour, M.; Yusoff, R.; Aroua Mohamed, K. Polymeric ionic liquids (PILs) for CO2 capture. Rev. Chem. Eng. 2017, 33, 183–220. [Google Scholar] [CrossRef]
- Qian, W.; Texter, J.; Yan, F. Frontiers in poly(ionic liquid)s: Syntheses and applications. Chem. Soc. Rev. 2017, 46, 1124–1159. [Google Scholar] [CrossRef]
- Ajjan, F.N.; Ambrogi, M.; Tiruye, G.A.; Cordella, D.; Fernandes, A.M.; Grygiel, K.; Isik, M.; Patil, N.; Porcarelli, L.; Rocasalbas, G.; et al. Innovative polyelectrolytes/poly(ionic liquid)s for energy and the environment. Polym. Int. 2017, 66, 1119–1128. [Google Scholar] [CrossRef]
- Zulfiqar, S.; Sarwar, M.I.; Mecerreyes, D. Polymeric ionic liquids for CO2 capture and separation: Potential, progress and challenges. Polym. Chem. 2015, 6, 6435–6451. [Google Scholar] [CrossRef]
- Carlisle, T.K.; Nicodemus, G.D.; Gin, D.L.; Noble, R.D. CO2/light gas separation performance of cross-linked poly(vinylimidazolium) gel membranes as a function of ionic liquid loading and cross-linker content. J. Memb. Sci. 2012, 397–398, 24–37. [Google Scholar] [CrossRef]
- Shaligram, S.V.; Wadgaonkar, P.P.; Kharul, U.K. Polybenzimidazole-based polymeric ionic liquids (PILs): Effects of ‘substitution asymmetry’ on CO2 permeation properties. J. Memb. Sci. 2015, 493, 403–413. [Google Scholar] [CrossRef]
- Carlisle, T.K.; Wiesenauer, E.F.; Nicodemus, G.D.; Gin, D.L.; Noble, R.D. Ideal CO2/Light Gas Separation Performance of Poly(vinylimidazolium) Membranes and Poly(vinylimidazolium)-Ionic Liquid Composite Films. Ind. Eng. Chem. Res. 2012, 52, 1023–1032. [Google Scholar] [CrossRef]
- Li, P.; Paul, D.R.; Chung, T.-S. High performance membranes based on ionic liquid polymers for CO2 separation from the flue gas. Green Chem. 2012, 14, 1052–1063. [Google Scholar] [CrossRef]
- Li, P.; Pramoda, K.P.; Chung, T.-S. CO2 Separation from Flue Gas Using Polyvinyl-(Room Temperature Ionic Liquid)–Room Temperature Ionic Liquid Composite Membranes. Ind. Eng. Chem. Res. 2011, 50, 9344–9353. [Google Scholar] [CrossRef]
- Tomé, L.C.; Mecerreyes, D.; Freire, C.S.R.; Rebelo, L.P.N.; Marrucho, I.M. Pyrrolidinium-based polymeric ionic liquid materials: New perspectives for CO2 separation membranes. J. Memb. Sci. 2013, 428, 260–266. [Google Scholar] [CrossRef]
- Tomé, L.C.; Isik, M.; Freire, C.S.R.; Mecerreyes, D.; Marrucho, I.M. Novel pyrrolidinium-based polymeric ionic liquids with cyano counter-anions: High performance membrane materials for post-combustion CO2 separation. J. Memb. Sci. 2015, 483, 155–165. [Google Scholar] [CrossRef]
- Tomé, L.C.; Gouveia, A.S.L.; Freire, C.S.R.; Mecerreyes, D.; Marrucho, I.M. Polymeric ionic liquid-based membranes: Influence of polycation variation on gas transport and CO2 selectivity properties. J. Memb. Sci. 2015, 486, 40–48. [Google Scholar] [CrossRef]
- Teodoro, R.M.; Tomé, L.C.; Mantione, D.; Mecerreyes, D.; Marrucho, I.M. Mixing poly(ionic liquid)s and ionic liquids with different cyano anions: Membrane forming ability and CO2/N2 separation properties. J. Memb. Sci. 2018, 552, 341–348. [Google Scholar]
- Tomé, L.C.; Florindo, C.; Freire, C.S.; Rebelo, L.P.; Marrucho, I.M. Playing with ionic liquid mixtures to design engineered CO2 separation membranes. Phys. Chem. Chem. Phys. 2014, 16, 17172–17182. [Google Scholar] [PubMed]
- Pont, A.-L.; Marcilla, R.; De Meatza, I.; Grande, H.; Mecerreyes, D. Pyrrolidinium-based polymeric ionic liquids as mechanically and electrochemically stable polymer electrolytes. J. Power Sources 2009, 188, 558–563. [Google Scholar] [CrossRef]
- Tomé, L.C.; Guerreiro, D.C.; Teodoro, R.M.; Alves, V.D.; Marrucho, I.M. Effect of polymer molecular weight on the physical properties and CO2/N2 separation of pyrrolidinium-based poly(ionic liquid) membranes. J. Memb. Sci. 2018, 549, 267–274. [Google Scholar] [CrossRef]
- Wijmans, J.G.; Baker, R.W. The solution-diffusion model: A review. J. Memb. Sci. 1995, 107, 1–21. [Google Scholar] [CrossRef]
- Matteucci, S.; Yampolskii, Y.; Freeman, B.D.; Pinnau, I. Transport of Gases and Vapors in Glassy and Rubbery Polymers. In Materials Science of Membranes for Gas and Vapor Separation; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2006; pp. 1–47. [Google Scholar]
- Rutherford, S.W.; Do, D.D. Review of time lag permeation technique as a method for characterisation of porous media and membranes. Adsorption 1997, 3, 283–312. [Google Scholar] [CrossRef]
- Wang, S.; Li, X.; Wu, H.; Tian, Z.; Xin, Q.; He, G.; Peng, D.; Chen, S.; Yin, Y.; Jiang, Z.; et al. Advances in high permeability polymer-based membrane materials for CO2 separations. Energy Environ. Sci. 2016, 9, 1863–1890. [Google Scholar] [CrossRef]
- Raeissi, S.; Peters, C.J. Understanding temperature dependency of hydrogen solubility in ionic liquids, including experimental data in [bmim][Tf2N]. AIChE J. 2012, 58, 3553–3559. [Google Scholar] [CrossRef]
- Kumełan, J.; Pérez-Salado Kamps, Á.; Tuma, D.; Maurer, G. Solubility of H2 in the Ionic Liquid [bmim][PF6]. J. Chem. Eng. Data 2006, 51, 11–14. [Google Scholar] [CrossRef]
- Finotello, A.; Bara, J.E.; Camper, D.; Noble, R.D. Room-Temperature Ionic Liquids: Temperature Dependence of Gas Solubility Selectivity. Ind. Eng. Chem. Res. 2008, 47, 3453–3459. [Google Scholar] [CrossRef]
- Kumełan, J.; Tuma, D.; Pérez-Salado Kamps, Á.; Maurer, G. Solubility of the Single Gases Carbon Dioxide and Hydrogen in the Ionic Liquid [bmpy][Tf2N]. J. Chem. Eng. Data 2010, 55, 165–172. [Google Scholar] [CrossRef]
- Raeissi, S.; Florusse, L.J.; Peters, C.J. Hydrogen Solubilities in the IUPAC Ionic Liquid 1-Hexyl-3-methylimidazolium Bis(Trifluoromethylsulfonyl)Imide. J. Chem. Eng. Data 2011, 56, 1105–1107. [Google Scholar] [CrossRef]
- Robeson, L.M.; Smith, Z.P.; Freeman, B.D.; Paul, D.R. Contributions of diffusion and solubility selectivity to the upper bound analysis for glassy gas separation membranes. J. Memb. Sci. 2014, 453, 71–83. [Google Scholar] [CrossRef]
- Rowe, B.W.; Robeson, L.M.; Freeman, B.D.; Paul, D.R. Influence of temperature on the upper bound: Theoretical considerations and comparison with experimental results. J. Memb. Sci. 2010, 360, 58–69. [Google Scholar] [CrossRef]
- Robeson, L.M. The upper bound revisited. J. Memb. Sci. 2008, 320, 390–400. [Google Scholar] [CrossRef]
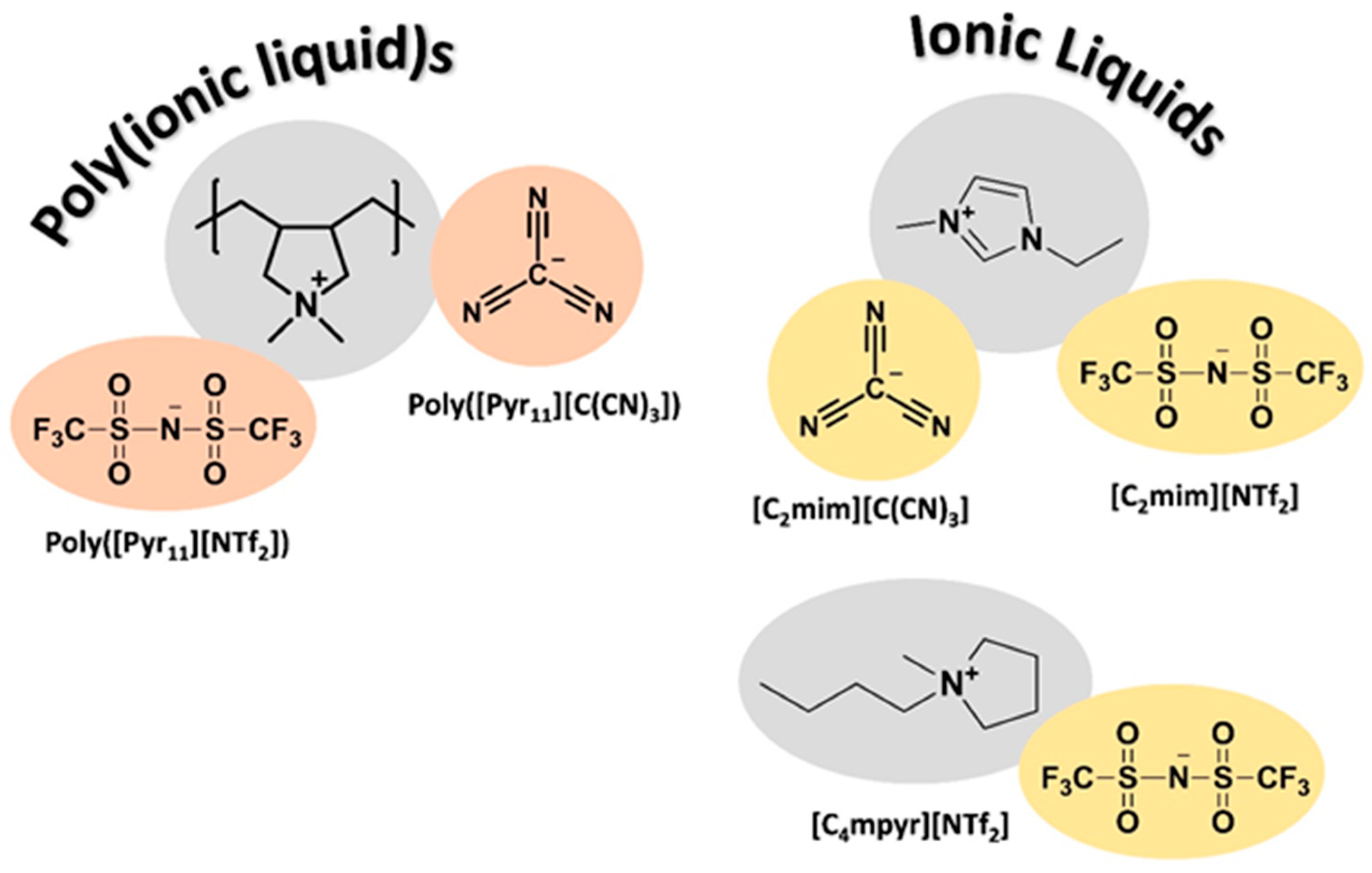
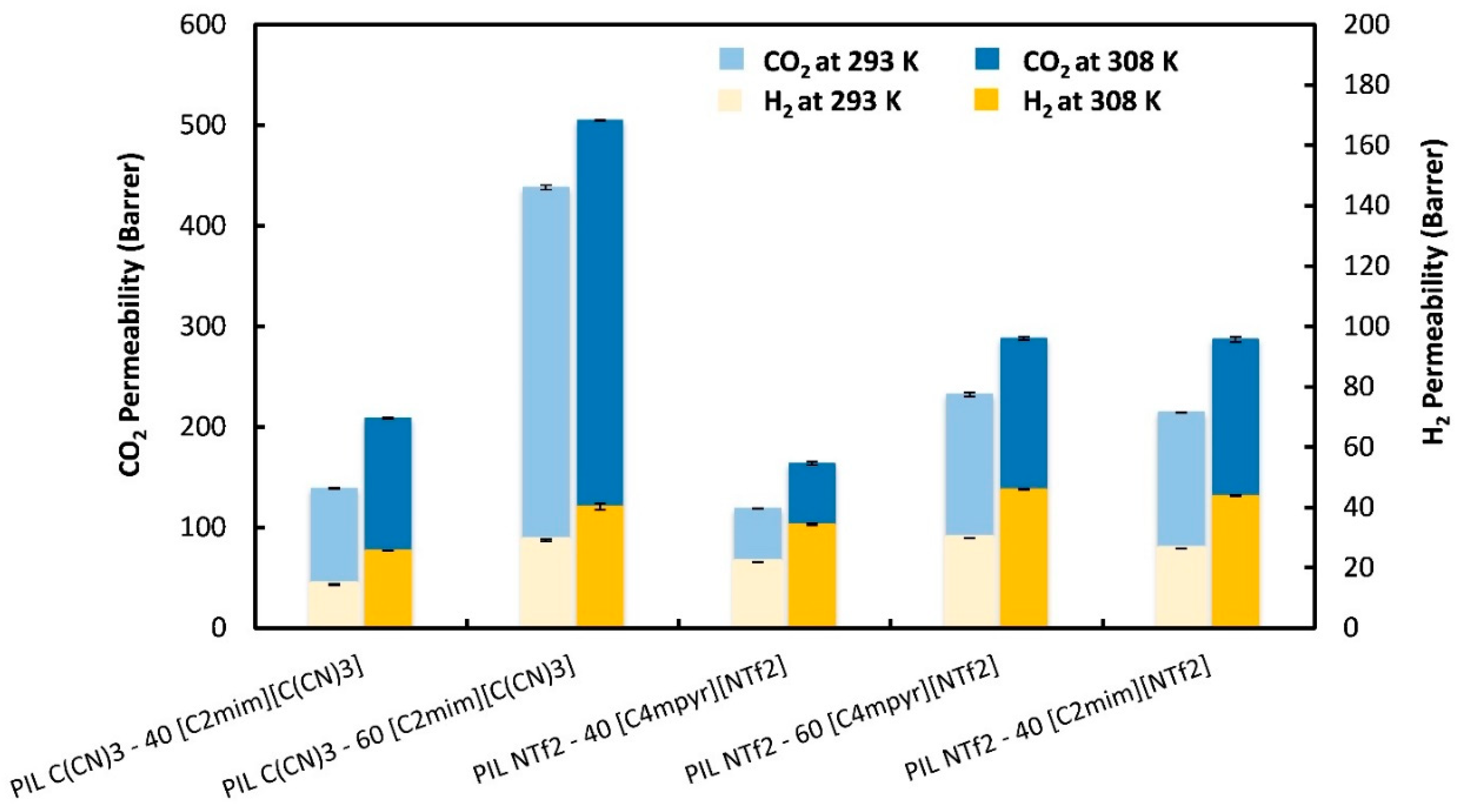
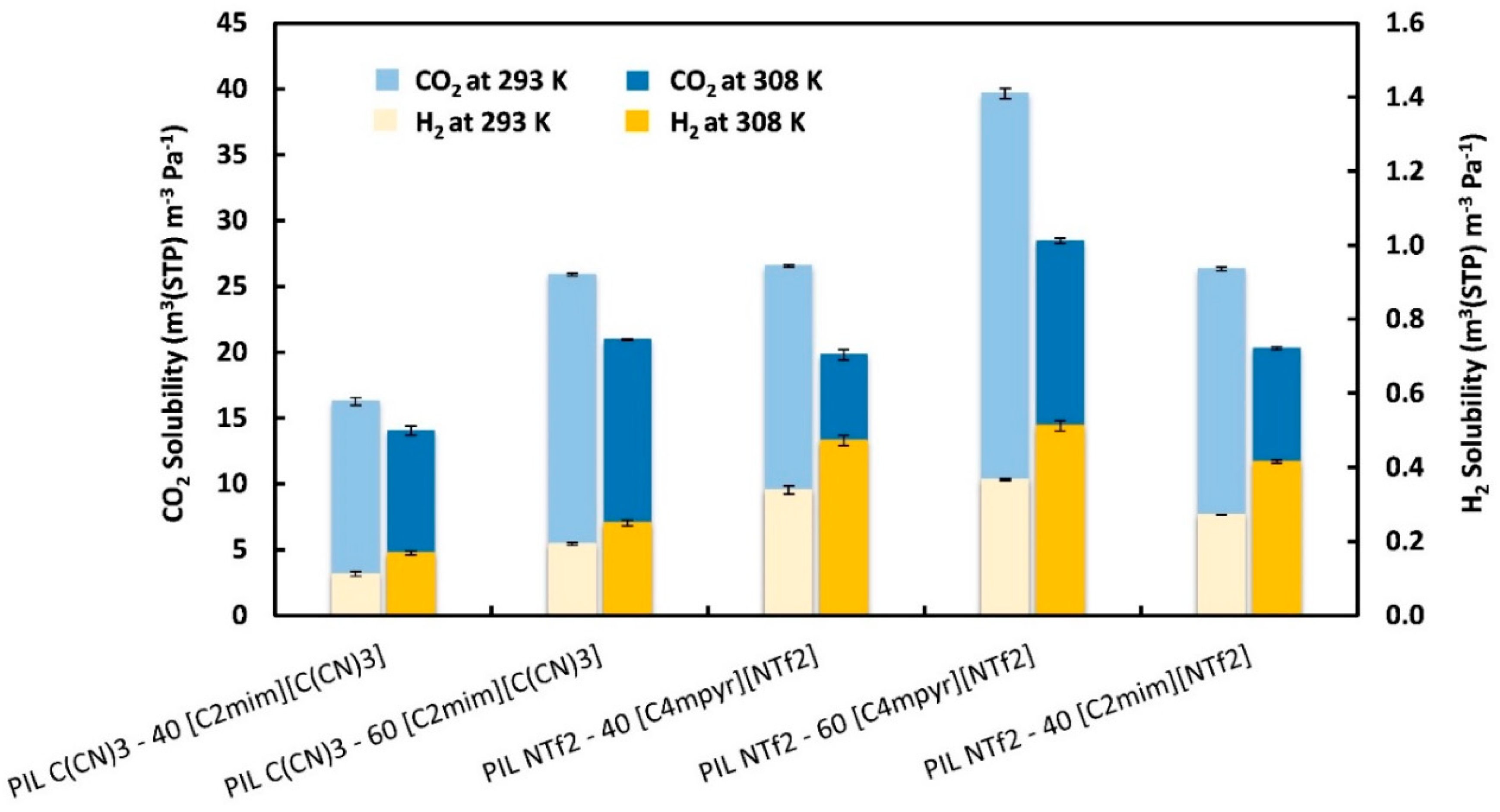
 ) from other reported PIL–IL membranes are also displayed for comparison [33].
) from other reported PIL–IL membranes are also displayed for comparison [33].
 ) from other reported PIL–IL membranes are also displayed for comparison [33].
) from other reported PIL–IL membranes are also displayed for comparison [33].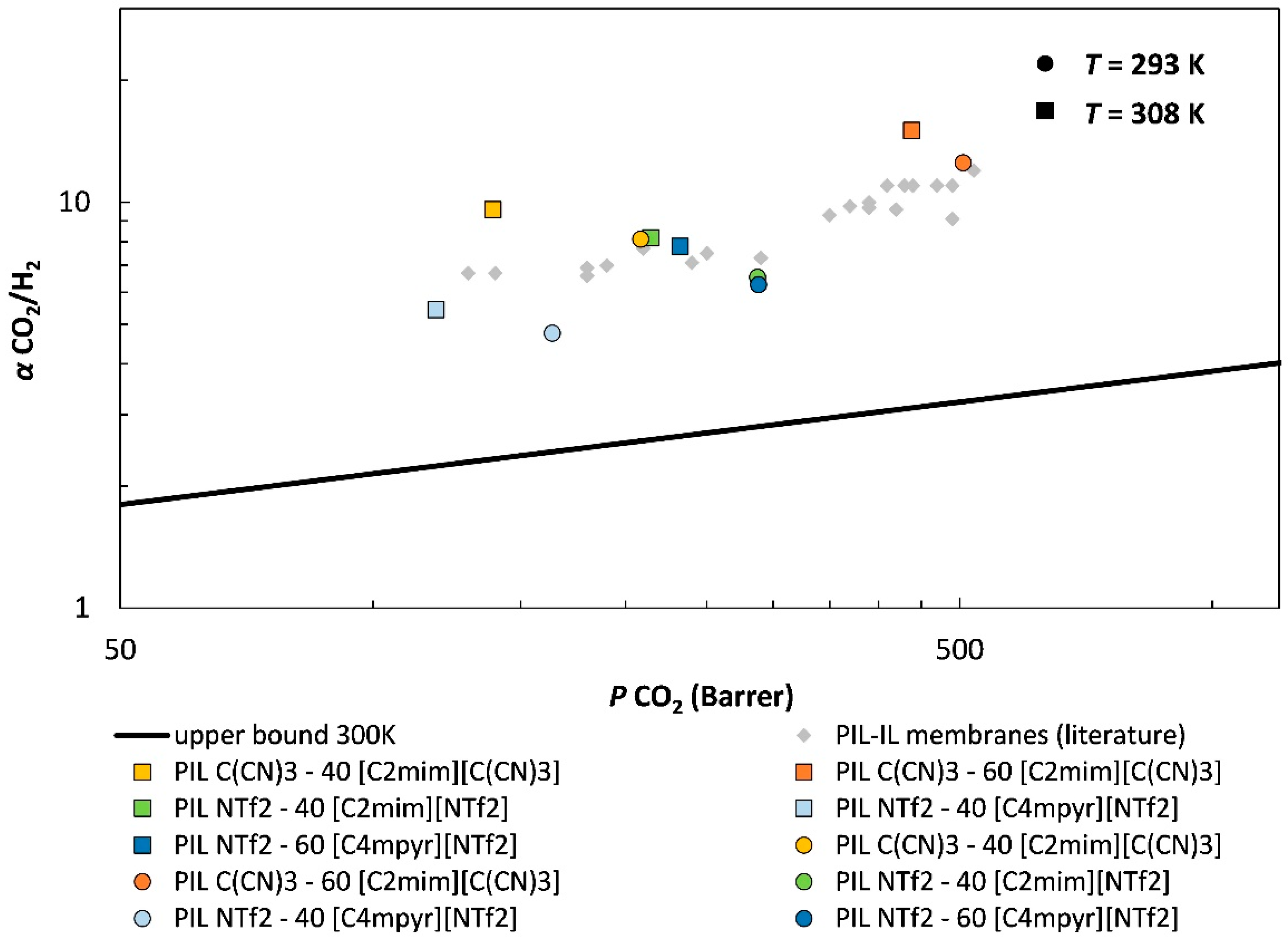
| PIL–IL Membrane | Polymer (PIL) | wt % of IL | Solvent | T (K) | Evaporation Time (Days) |
|---|---|---|---|---|---|
| PIL C(CN)3–40 [C2mim][C(CN)3] | Poly([Pyr11][C(CN)3]) | 40 | Acetonitrile | 298 | 3 |
| PIL C(CN)3–60 [C2mim][C(CN)3] | 60 | ||||
| PIL NTf2–40 [C4mpyr][NTf2] | Poly([Pyr11][NTf2]) | 40 | Acetone | 298 | 2 |
| PIL NTf2–60 [C4mpyr][NTf2] | 60 | ||||
| PIL NTf2–40 [C2mim][NTf2] | Poly([Pyr11][NTf2]) | 40 | Acetone | 298 | 2 |
| PIL–IL Membrane | Gas Diffusivity (×1012) (m2 s−1) (T = 293 K) | Gas Diffusivity (×1012) (m2 s−1) (T = 308 K) | ||
|---|---|---|---|---|
| D CO2 ± σ | D H2 ± σ | D CO2 ± σ | D H2 ± σ | |
| PIL C(CN)3–40 [C2mim][C(CN)3] | 64 ± 1.0 | 970 ± 36.2 | 112 ± 2.5 | 1146 ± 34.0 |
| PIL C(CN)3–60 [C2mim][C(CN)3] | 127 ± 1.1 | 1130 ± 5.70 | 181 ± 0.6 | 1211 ± 3.2 |
| PIL NTf2–40 [C4mpyr][NTf2] | 34 ± 0.1 | 484 ± 18.5 | 62 ± 1.8 | 546 ± 20.6 |
| PIL NTf2–60 [C4mpyr][NTf2] | 44 ± 0.7 | 610 ± 6.30 | 76 ± 0.5 | 673 ± 16.9 |
| PIL NTf2–40 [C2mim][NTf2] | 61 ± 0.4 | 722 ± 1.80 | 106 ± 1.5 | 792 ± 3.70 |
| PIL–IL Membrane | Gas Permeability (Barrer) (T = 293 K) | Gas Permeability (Barrer) (T = 308 K) | ||||
|---|---|---|---|---|---|---|
| P CO2 ± σ | P H2 ± σ | α CO2/H2 | P CO2 ± σ | P H2 ± σ | α CO2/H2 | |
| PIL C(CN)3–40 [C2mim][C(CN)3] | 139 ± 0.5 | 14.5 ± 0.2 | 9.6 ± 0.2 | 209 ± 0.9 | 25.7 ± 0.1 | 8.1 ± 0.1 |
| PIL C(CN)3–60 [C2mim][C(CN)3] | 438 ± 2.1 | 29.1 ± 0.4 | 15.1 ± 0.3 | 505 ± 0.3 | 40.3 ± 1.1 | 12.5 ± 0.3 |
| PIL NTf2–40 [C4mpyr][NTf2] | 119 ± 0.2 | 21.9 ± 0.1 | 5.4 ± 0.1 | 164 ± 1.6 | 34.4 ± 0.3 | 4.8 ± 0.1 |
| PIL NTf2–60 [C4mpyr][NTf2] | 232 ± 2.2 | 29.8 ± 0.1 | 7.8 ± 0.1 | 288 ± 1.6 | 46.0 ± 0.1 | 6.3 ± 0.1 |
| PIL NTf2–40 [C2mim][NTf2] | 214 ± 0.6 | 26.2 ± 0.1 | 8.2 ± 0.1 | 287 ± 2.4 | 43.8 ± 0.2 | 6.5 ± 0.1 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gouveia, A.S.L.; Ventaja, L.; Tomé, L.C.; Marrucho, I.M. Towards Biohydrogen Separation Using Poly(Ionic Liquid)/Ionic Liquid Composite Membranes. Membranes 2018, 8, 124. https://doi.org/10.3390/membranes8040124
Gouveia ASL, Ventaja L, Tomé LC, Marrucho IM. Towards Biohydrogen Separation Using Poly(Ionic Liquid)/Ionic Liquid Composite Membranes. Membranes. 2018; 8(4):124. https://doi.org/10.3390/membranes8040124
Chicago/Turabian StyleGouveia, Andreia S. L., Lucas Ventaja, Liliana C. Tomé, and Isabel M. Marrucho. 2018. "Towards Biohydrogen Separation Using Poly(Ionic Liquid)/Ionic Liquid Composite Membranes" Membranes 8, no. 4: 124. https://doi.org/10.3390/membranes8040124
APA StyleGouveia, A. S. L., Ventaja, L., Tomé, L. C., & Marrucho, I. M. (2018). Towards Biohydrogen Separation Using Poly(Ionic Liquid)/Ionic Liquid Composite Membranes. Membranes, 8(4), 124. https://doi.org/10.3390/membranes8040124





