Mixed Matrix Membranes for O2/N2 Separation: The Influence of Temperature
Abstract
:1. Introduction
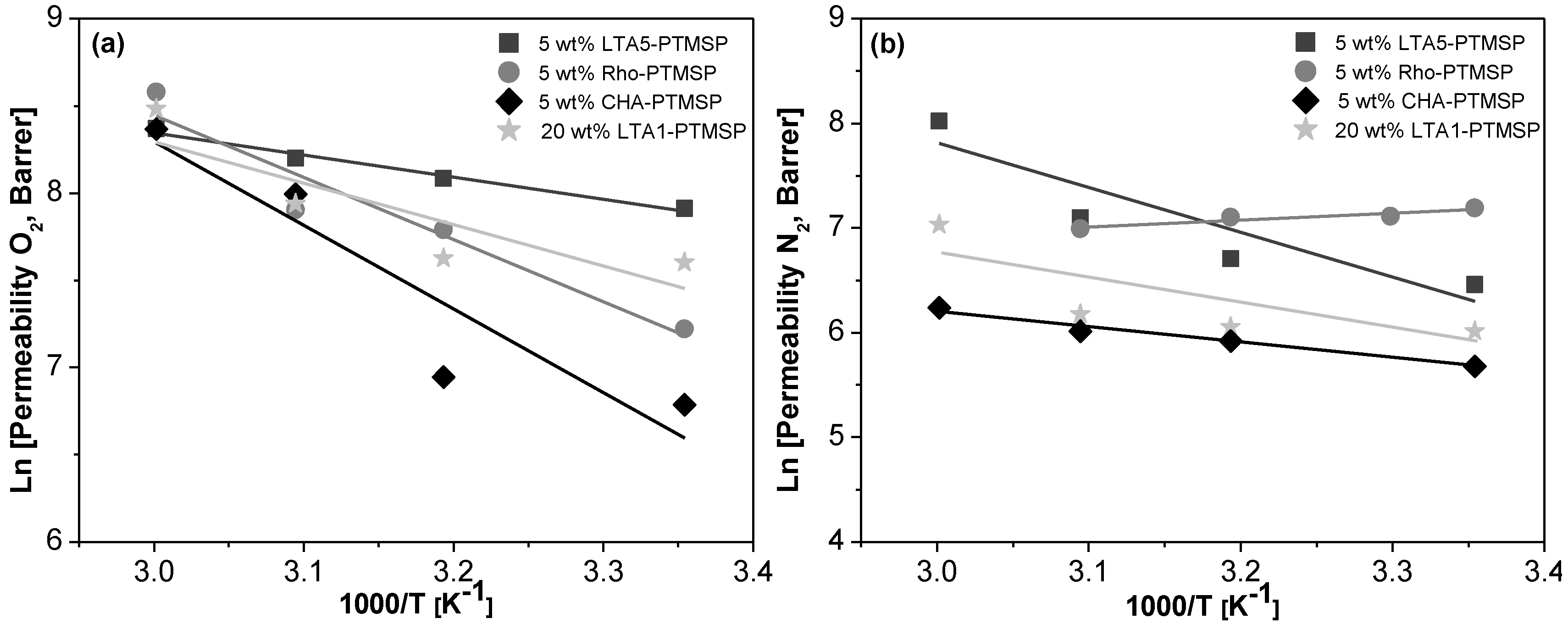
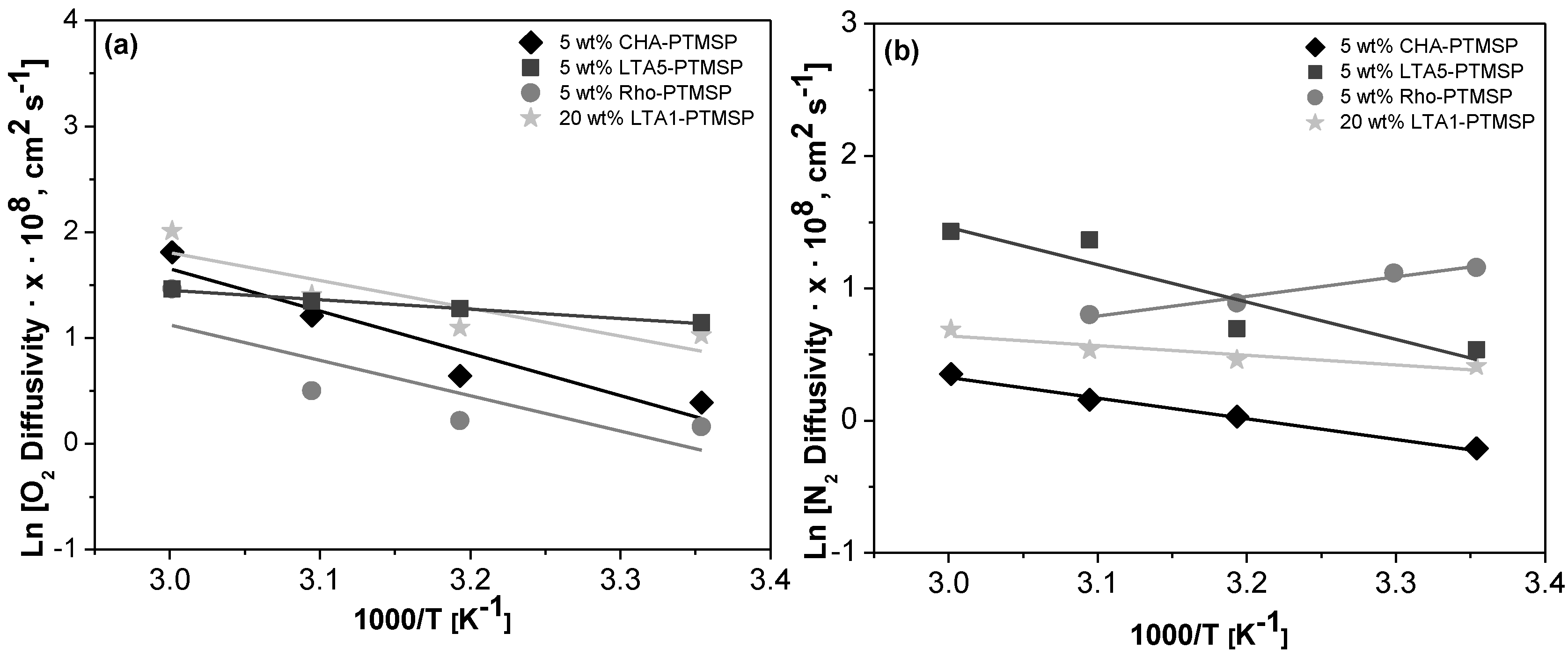
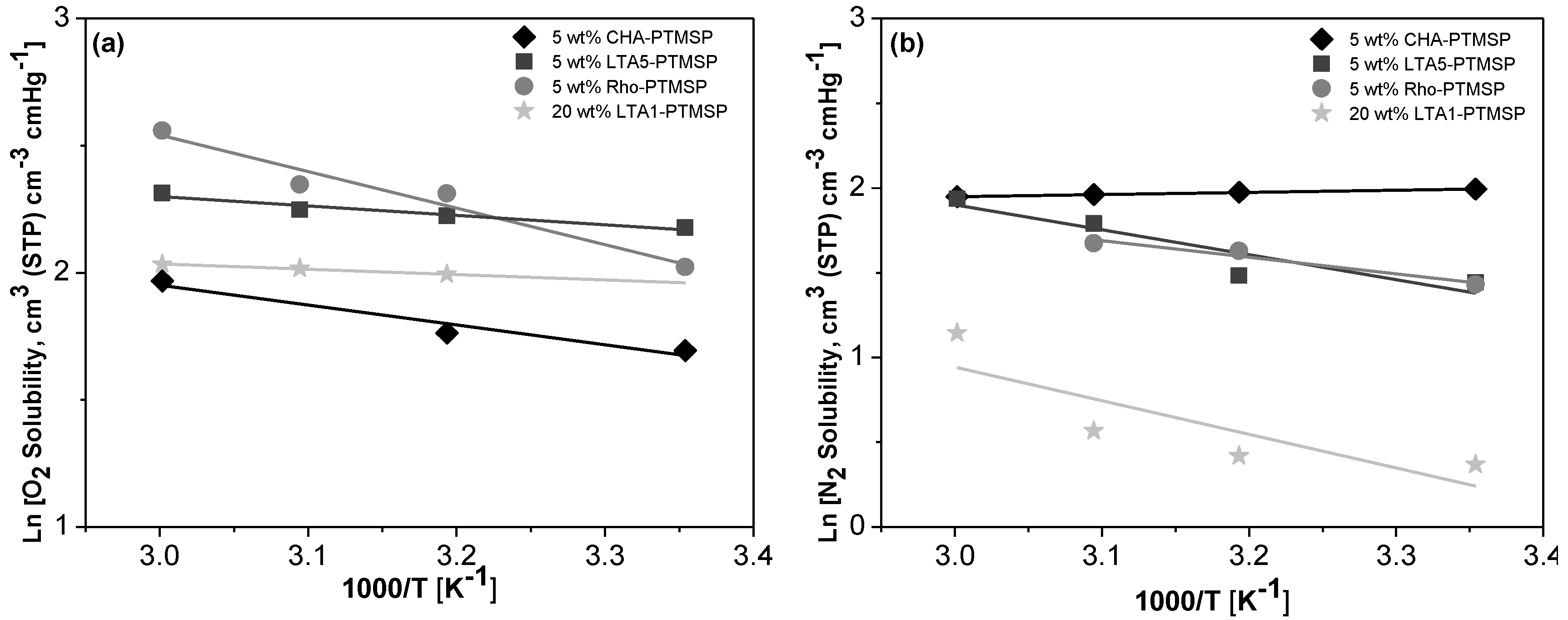
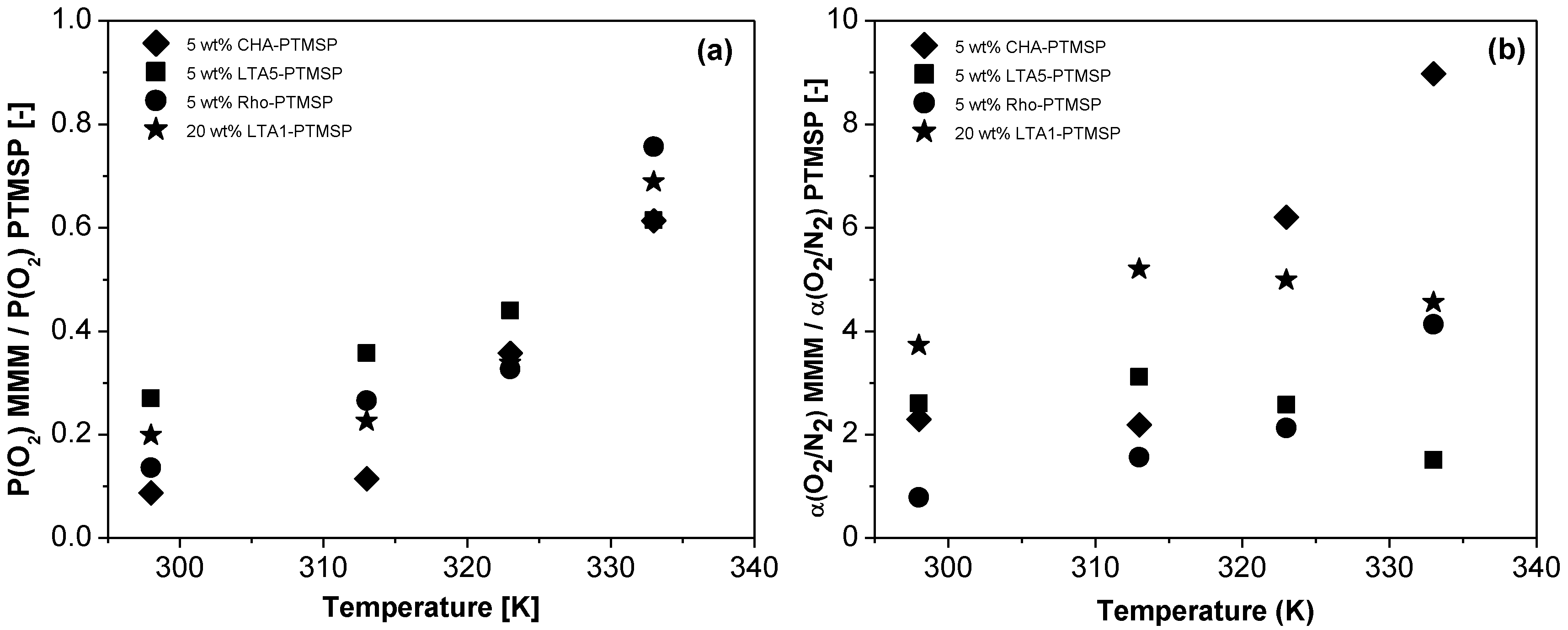
2. Results
3. Materials and Methods
3.1. Synthesis of MMM
3.2. Characterization
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Burdyny, T.; Struchtrup, H. Hybrid membrane/cryogenic separation of oxygen from air for use in the oxy-fuel process. Energy 2010, 35, 1884–1897. [Google Scholar] [CrossRef]
- Belaissaoui, B.; Moullec, Y.L.; Hagi, H.; Favre, E. Energy efficiency of oxygen enriched air production technologies: Cryogeny vs. membranes. Sep. Purif. Technol. 2014, 125, 142–150. [Google Scholar] [CrossRef]
- Baker, R.W. Membrane Technology and Applications, 2nd ed.; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 2004; p. 545. [Google Scholar]
- Koros, W.J.; Mahajan, R. Pushing the limits on possibilities for large scale gas separation: Which strategies? J. Membr. Sci. 2000, 175, 181–196. [Google Scholar] [CrossRef]
- Bredesen, R.; Jordal, K.; Bolland, O. High-temperature membranes in power generation with CO2 capture. Chem. Eng. Proc. 2004, 43, 1129–1158. [Google Scholar] [CrossRef]
- Carta, M.; Malpass-Evans, R.; Croad, M.; Rogan, Y.; Jansen, J.C.; Bernardo, P.; Bazzarelli, F.; McKeown, N.B. An efficient polymer molecular sieve for membrane gas separations. Science 2013, 339, 303–309. [Google Scholar] [CrossRef] [PubMed]
- Robeson, L.M. The upper bound revisited. J. Membr. Sci. 2008, 320, 390–400. [Google Scholar] [CrossRef]
- Smith, A.R.; Klosek, J. A review of air separation technologies and their integration with energy conversion processes. Fuel Proc. Technol. 2001, 70, 115–134. [Google Scholar] [CrossRef]
- Li, Y.; Guan, H.-M.; Chung, T.-S.; Kulprathipanja, S. Effects of novel silane modification of zeolite surface on polymer chain rigidification and partial pore blockage in polyethersulfone (PES)-zeolite a mixed matrix membranes. J. Membr. Sci. 2006, 275, 17–28. [Google Scholar] [CrossRef]
- Mahajan, R.; Koros, W.J. Factors controlling successful formation of mixed-matrix gas separation materials. Ind. Eng. Chem. Res. 2000, 39, 2692–2696. [Google Scholar] [CrossRef]
- Mahajan, R.; Burns, R.; Schaeffer, M.; Koros, W.J. Challenges in forming successful mixed matrix membranes with rigid polymeric materials. J. Appl. Polym. Sci. 2002, 86, 881–890. [Google Scholar] [CrossRef]
- Zimmerman, C.M.; Koros, W.J. Polypyrrolones for membrane gas separations. II. Activation energies and heats of sorption. J. Polym. Sci. B 1999, 37, 1251–1265. [Google Scholar] [CrossRef]
- Zimmerman, C.M.; Koros, W.J. Polypyrrolones for membrane gas separations. I. Structural comparison of gas transport and sorption properties. J. Polym. Sci. B 1999, 37, 1235–1249. [Google Scholar] [CrossRef]
- Bernardo, P.; Drioli, E.; Golemme, G. Membrane gas separation: A review/state of the art. Ind. Eng. Chem. Res. 2009, 48, 4638–4663. [Google Scholar] [CrossRef]
- Casado-Coterillo, C.; Soto, J.; Jimaré, M.T.; Valencia, S.; Corma, A.; Téllez, C.; Coronas, J. Preparation and characterization of ITQ-29/polysulfone mixed-matrix membranes for gas separation: Effect of zeolite composition and crystal size. Chem. Eng. Sci. 2012, 73, 116–122. [Google Scholar] [CrossRef]
- Chung, T.-S.; Jiang, L.Y.; Li, Y.; Kulprathipanja, S. Mixed matrix membranes (MMMs) comprising organic polymers with dispersed inorganic fillers for gas separation. Prog. Polym. Sci. 2007, 32, 483–507. [Google Scholar] [CrossRef]
- Malakhov, A.O.; Knyazeva, E.E.; Novitsky, E.G. Gas transport properties of lia type zeolite-filled poly(trimethylsilylpropyne) membranes Petrol. Chem. 2015, 55, 708–715. [Google Scholar]
- Alentiev, A.; Yampolskii, Y. Correlation of gas permeability and diffusivity with selectivity: Orientations of the clouds of data points and the effects of temperature. Ind. Eng. Chem. Res. 2013, 52, 8864–8874. [Google Scholar] [CrossRef]
- Costello, L.M.; Koros, W.J. Temperature dependence of gas sorption and transport properties in polymers: Measurement and applications. Ind. Eng. Chem. Res. 1992, 31, 2708–2714. [Google Scholar] [CrossRef]
- Fernández-Barquín, A.; Casado-Coterillo, C.; Palomino, M.; Valencia, S.; Irabien, A. LTA/poly(1-trimethylsilyl-1-propyne) mixed matrix membranes for high-temperature CO2/N2 separation. Chem. Eng. Technol. 2015, 38, 658–666. [Google Scholar] [CrossRef]
- Fernández-Barquín, A.; Casado-Coterillo, C.; Palomino, M.; Valencia, S.; Irabien, A. Permselectivity improvement in membranes for CO2/N2 separation. Sep. Purif. Technol. 2016, 157, 102–111. [Google Scholar] [CrossRef]
- Merkel, T.C.; Bondar, V.I.; Nagai, K.; Freeman, B.D.; Pinnau, I. Gas sorption, diffusion, and permeation in poly(dimethylsiloxane). J. Polym. Sci. B 2000, 38, 415–434. [Google Scholar] [CrossRef]
- Qiu, J.; Zheng, J.-M.; Peinemann, K.-V. Gas transport properties in a novel poly(trimethylsilylpropyne) composite membrane with nanosized organic filler trimethylsilylglucose. Macromolecules 2006, 39, 4093–4100. [Google Scholar] [CrossRef]
- Nakamura, K.; Kitagawa, T.; Nara, S.; Wakamatsu, T.; Ishiba, Y.; Kanehashi, S.; Sasto, S.; Nagai, K. Permeability of dry gases and those dissolved in water through hydrophobic high free-volume silicon- or fluorine-containing nonporous glassy polymer membranes. Ind. Eng. Chem. Res. 2013, 52, 1133–1140. [Google Scholar] [CrossRef]
- Shao, L.; Samseth, J.; Hägg, M.-B. Crosslinking and stabilization of nanoparticle filled poly(1-trimethylsilyl-1-propyne) nanocomposite membranes for gas separations. J. Appl. Polym. Sci. 2009, 113, 3078–3088. [Google Scholar] [CrossRef]
- Merkel, T.C.; Freeman, B.D.; Spontak, R.J.; He, Z.; Pinnau, I.; Meakin, P.; Hill, A.J. Ultrapermeable, reversible-selective nanocomposite membranes. Science 2002, 296, 519–522. [Google Scholar] [CrossRef] [PubMed]
- Merkel, T.C.; Freeman, B.D.; Spontak, R.J.; He, Z.; Pinnau, I.; Meakin, P.; Hill, A.J. Sorption, transport and structural evidence for enhanced free volume in poly(4-methyl-2-pentyne)/fumed silica nanocomposite membranes. Chem. Mater. 2003, 15, 109–123. [Google Scholar] [CrossRef]
- Yave, W.; Peinemann, K.-V.; Shishatsky, S.; Khotmiskiy, V.; Chirkova, M.; Matson, S.; Litvinova, E.; Lecerf, N. Synthesis, characterization and membrane properties of poly(1-trimethylgermyl-1-propyne) and its nanocomposite with TiO2. Macromolecules 2007, 40, 8991–8998. [Google Scholar] [CrossRef]
- Woo, M.; Choi, J.; Tsapatsis, M. Poly(1-trimethylsilyl-1-propyne)/MFI composite membranes for butane separations. Microporous Mesoporous Mater. 2008, 110, 330–338. [Google Scholar] [CrossRef]
- Rowe, B.W.; Robeson, L.M.; Freeman, B.D.; Paul, D.R. Influence of temperature on the upper bound: Theoretical considerations and comparison with experimental results. J. Membr. Sci. 2010, 360, 58–69. [Google Scholar] [CrossRef]
- Clarizia, G.; Algieri, C.; Drioli, E. Filler-polymer combination: A route to modify gas transport properties of a polymeric membrane. Polymer 2004, 45, 5671–5681. [Google Scholar] [CrossRef]
- Wang, X.-Y.; Hill, A.J.; Freeman, B.D.; Sanchez, I.C. Structural, sorption and transport characteristics of an ultrapermeable polymer. J. Membr. Sci. 2008, 314, 15–23. [Google Scholar] [CrossRef]
- Shantarovich, V.P.; Kevdina, I.B.; Yampolskiii, Y.P.; Alentiev, A.Y. Positron annihilation lifetime study of high and low free volume glassy polymers: Effects of free volume sizes on the permeability and permselectivity. Macromolecules 2000, 33, 7453–7466. [Google Scholar] [CrossRef]
- Rutherford, S.W.; Do, D.D. Review of time lag permeation technique as a method for characterisation of porous media and membranes. Adsorption 1997, 3, 283–312. [Google Scholar] [CrossRef]
- Bastani, D.; Esmaeili, N.; Asadollahi, M. Polymeric mixed matrix membranes containing zeolites as a filler for gas separation applications: A review. J. Ind. Eng. Chem. 2013, 19, 375–393. [Google Scholar] [CrossRef]
- Li, Y.; Chung, T.-S.; Cao, C.; Kulprathipanja, S. The effects of polymer chain rigidification, zeolite pore size and pore blockage on polyether sulfone (PES)-zeolite a mixed matrix membranes. J. Membr. Sci. 2005, 260, 45–55. [Google Scholar] [CrossRef]
- Zhou, J.-H.; Zhu, R.-X.; Zhou, J.-M.; Chen, M.-B. Molecular dynamics simulation of diffusion of gases in pure and silica-filled poly(1-trimethylsilyl-1-propyne) [PTMSP]. Polymer 2006, 47, 5206–5212. [Google Scholar] [CrossRef]
- Ahmad, J.; Hägg, M.-B. Development of matrimid/zeolite 4A mixed matrix membrane using low boiling point solvent. Sep. Purif. Technol. 2013, 115, 190–197. [Google Scholar] [CrossRef]
- Jia, W.; Murad, S. Separation of gas mixtures using a range of zeolite membranes: A molecular dynamics study. J. Phys. Chem. 2005, 122, 234708(1-11). [Google Scholar] [CrossRef] [PubMed]
- Cussler, E.L. Diffusion: Mass Transfer in Fluid Systems, 3rd ed.; Cambridge University Press: Cambridge, UK, 2007; p. 631. [Google Scholar]
- Bara, J.E.; Lessmann, S.; Gabriel, C.J.; Hatakeyama, E.S.; Noble, R.D.; Gin, D.L. Synthesis and performance of polymerizable room-temperature ionic liquids as gas separation membranes. Ind. Eng. Chem. Res. 2007, 46, 5397–5404. [Google Scholar] [CrossRef]
- Crank, J. The Mathematics of Diffusion, 2nd ed.; Clarendon Press: Oxford, UK, 1975; p. 411. [Google Scholar]
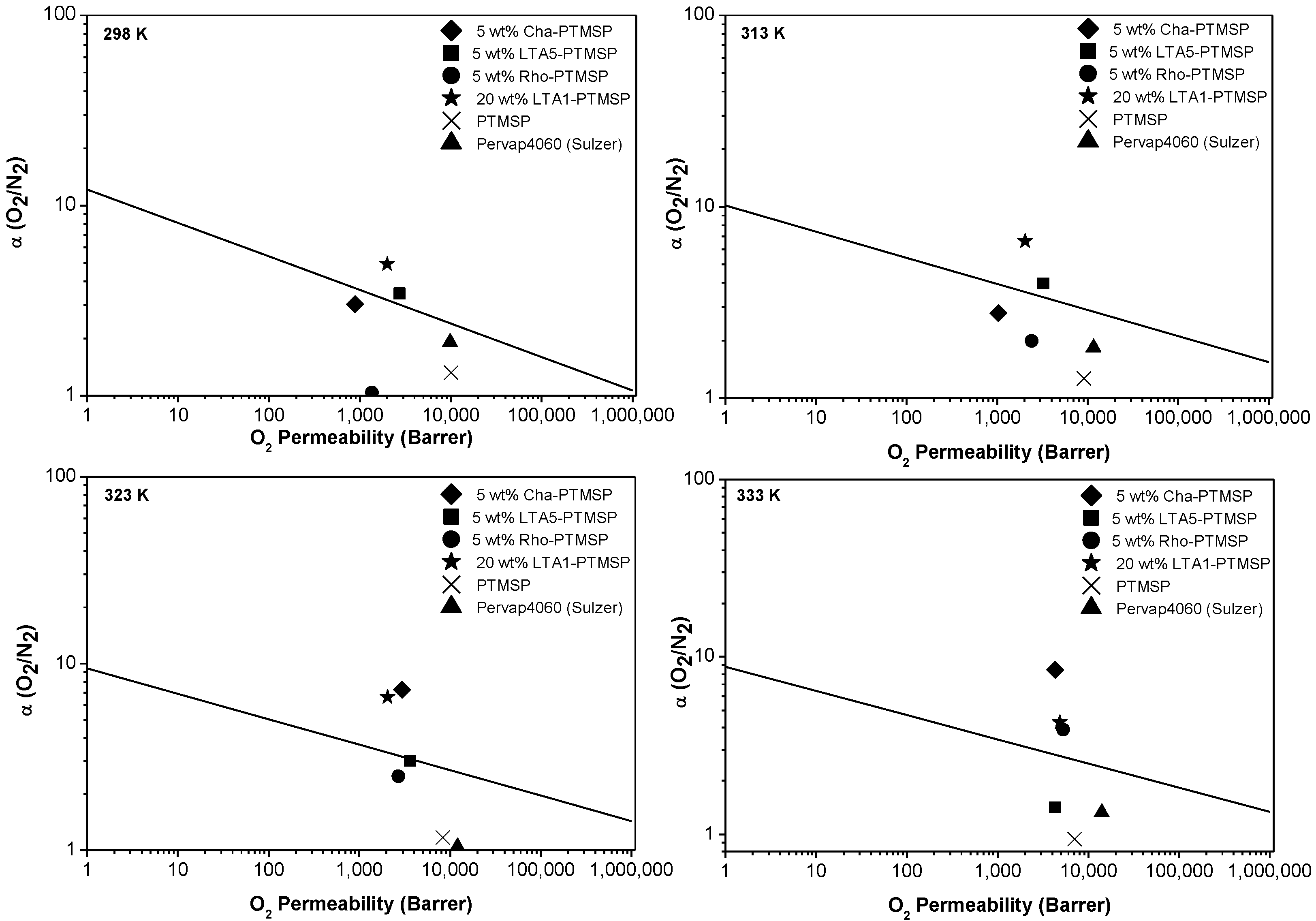
| Membrane | T = 298 K | T = 313 K | T = 323 K | T = 333 K | ||||
|---|---|---|---|---|---|---|---|---|
| P (O2) | αP (O2/N2) | P (O2) | αP (O2/N2) | P (O2) | αP (O2/N2) | P (O2) | αP (O2/N2) | |
| PTMSP | 10083 ± 936 | 1.32 ± 0.12 | 9072 ± 256 | 1.27 ± 0.04 | 8293 ± 851 | 1.27 ± 0.12 | 7024 ± 1050 | 0.94 ± 0.41 |
| 5 wt % CHA/PTMSP | 885 ± 395 | 3.03 ± 0.96 | 1038 ± 270 | 2.79 ± 0.51 | 2965 ± 1321 | 7.25 ± 2.28 | 4308 ± 1608 | 8.43 ± 2.23 |
| 5 wt % LTA5/PTMSP | 2727 ± 813 | 3.43 ± 0.72 | 3244 ± 193 | 3.97 ± 0.17 | 3642 ± 33 | 3.02 ± 0.02 | 4316 ± 1107 | 1.42 ± 2.23 |
| 5 wt % Rho/PTMSP | 1368 ± 573 | 1.03 ± 0.63 | 2414 ± 921 | 1.99 ± 1.58 | 2707 ± 601 | 2.49 ± 1.66 | 5312 ± 1910 | 3.88 ± 1.02 |
| 20 wt % LTA1/PTMSP | 2000 ± 198 | 4.91 ± 0.49 | 2051 ± 756 | 6.62 ± 2.44 | 2803 ± 1592 | 5.83 ± 3.31 | 4833 ± 2542 | 4.28 ± 2.25 |
| 5 wt % TMSP/PTMSP [23] | 2346 | 1.92 | 2203 | 1.88 | 2018 | 1.82 | – | – |
| LiA/PTMSP [17] | – | – | 10390 | 1.45 | – | – | – | – |
| 20 vol % zeolite A/Matrimid | 4.0 | 7.20 | – | – | – | – | – | – |
| 15 wt % zeolite NaA/PDMS [31] | 330 | 2.23 | 403 | 2.12 | – | – | 493 | 1.65 |
| 50 wt % zeolite NaA/PDMS * [31] | 221 | 2.23 | 284 | 2.12 | – | – | 330 | 1.82 |
| 20 wt % Zeolite A/PES [9] | – | – | 0.35 | 6.0 | – | – | – | – |
| 15 wt % Zeolite A/PVAc [10] | – | – | 0.45 | 7.45 | – | – | – | – |
| Membrane | T = 298 K | T = 313 K | T = 323 K | T = 333 K | ||||
|---|---|---|---|---|---|---|---|---|
| D (O2) | αD (O2/N2) | P(O2) | αD (O2/N2) | P (O2) | αD (O2/N2) | P (O2) | αD (O2/N2) | |
| PTMSP | 5.47 ± 1.72 | 0.11 ± 0.04 | 3.50 ± 0.35 | 0.18 ± 0.03 | 3.46 ± 1.54 | 0.26 ± 0.09 | 3.40 ± 0.53 | 0.31 ± 0.06 |
| 5 wt % CHA/PTMSP | 1.48 ± 0.28 | 1.83 ± 0.43 | 1.90 ± 0.07 | 1.84 ± 0.23 | 3.35 ± 0.95 | 2.86 ± 0.87 | 6.11 ± 1.87 | 4.30 ± 1.50 |
| 5 wt % LTA5/PTMSP | 3.14 ± 0.82 | 1.84 ± 0.67 | 3.58 ± 0.57 | 1.79 ± 0.39 | 3.85 ± 0.11 | 0.98 ± 0.41 | 4.33 ± 1.02 | 1.04 ± 0.48 |
| 5 wt % Rho/PTMSP | 1.17 ±0.13 | 0.37 ± 0.14 | 1.25 ± 0.14 | 0.51 ± 0.18 | 1.65 ± 0.22 | 0.74 ± 0.26 | 4.31 ± 1.15 | 1.02 ± 0.50 |
| 20 wt % LTA1/PTMSP | 2.78 ± 1.01 | 1.84 ± 1.03 | 3.00 ± 1.08 | 1.90 ± 1.04 | 4.08 ± 1.83 | 2.40 ± 1.40 | 7.45 ± 3.53 | 3.74 ± 2.12 |
| Membrane | T = 298 K | T = 313 K | T = 323 K | T = 333 K | ||||
|---|---|---|---|---|---|---|---|---|
| S(O2) | αS (O2/N2) | S(O2) | αS (O2/N2) | S(O2) | αS (O2/N2) | S(O2) | αS (O2/N2) | |
| PTMSP | 18.43 ± 1.21 | 11.59 ± 3.22 | 25.92 ± 0.52 | 7.18 ± 1.02 | 23.97 ± 1.77 | 4.49 ± 0.55 | 20.66 ± 6.23 | 3.05 ± 0.72 |
| 5 wt % CHA/PTMSP | 5.44 ± 1.14 | 0.76 ± 0.16 | 5.83 ± 1.12 | 0.79 ± 0.15 | 8.76 ± 0.43 | 1.25 ± 0.09 | 7.15 ± 0.46 | 1.00 ± 0.11 |
| 5 wt % LTA5/PTMSP | 8.82 ± 0.68 | 2.08 ± 0.33 | 9.24 ± 1.54 | 2.10 ± 1.25 | 9.48 ± 0.31 | 1.58 ± 0.18 | 10.11 ± 0.80 | 1.46 ±0.77 |
| 5 wt % Rho/PTMSP | 7.55 ± 0.58 | 1.87 ± 0.16 | 10.09 ± 0.54 | 1.89 ± 0.80 | 10.46 ± 4.89 | 2.05 ± 1.00 | 12.93 ± 0.44 | 3.83 ± 0.58 |
| 20 wt % LTA1/PTMSP | 9.26 ± 5.71 | 6.40 ± 3.25 | 7.34 ± 1.06 | 4.16 ± 1.00 | 7.51 ± 1.87 | 4.95 ± 2.45 | 7.64 ± 2.42 | 2.43 ± 0.62 |
| Membrane | EP (kJ/mol) | ED (kJ/mol) | −ΔHS (kJ/mol) | |||
|---|---|---|---|---|---|---|
| O2 | N2 | O2 | N2 | O2 | N2 | |
| PTMSP | −8.19 ± 1.72 | −12.60 ± 4.10 | −11.02 ± 1.71 | −17.29 ± 8.34 | −2.34 ±1.89 | 5.36 ± 2.32 |
| 5 wt % CHA/PTMSP | 39.9 ± 13.44 | 12.13 ± 3.17 1 | 33.2 ± 13.16 | 12.85 ± 2.28 2 | 6.51 ± 3.28 | −1.02 ± 8.56 |
| 5 wt % LTA5/PTMSP | 10.58 ± 4.27 | 42.29 ± 13.05 1 | 7.37 ± 3.29 | 28.25 ± 1.65 2 | 3.07 ± 1.56 | 12.21 ± 1.21 |
| 5 wt % Rho/PTMSP | 29.65 ± 9.59 | −5.04 ± 0.69 1 | 27.82 ± 1.53 | −12.35 ± 8.15 2 | 11.95 ± 1.72 | 8.11 ± 1.76 |
| 20 wt % LTA1/PTMSP | 19.78 ± 7.87 | 19.96 ± 10.7 2 | 21.93 ± 5.24 | 8.54 ± 5.18 | 1.72 ± 1.00 | 16.50 ± 9.08 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernández-Barquín, A.; Casado-Coterillo, C.; Valencia, S.; Irabien, A. Mixed Matrix Membranes for O2/N2 Separation: The Influence of Temperature. Membranes 2016, 6, 28. https://doi.org/10.3390/membranes6020028
Fernández-Barquín A, Casado-Coterillo C, Valencia S, Irabien A. Mixed Matrix Membranes for O2/N2 Separation: The Influence of Temperature. Membranes. 2016; 6(2):28. https://doi.org/10.3390/membranes6020028
Chicago/Turabian StyleFernández-Barquín, Ana, Clara Casado-Coterillo, Susana Valencia, and Angel Irabien. 2016. "Mixed Matrix Membranes for O2/N2 Separation: The Influence of Temperature" Membranes 6, no. 2: 28. https://doi.org/10.3390/membranes6020028
APA StyleFernández-Barquín, A., Casado-Coterillo, C., Valencia, S., & Irabien, A. (2016). Mixed Matrix Membranes for O2/N2 Separation: The Influence of Temperature. Membranes, 6(2), 28. https://doi.org/10.3390/membranes6020028









