Polysaccharide-Based Membranes in Food Packaging Applications
Abstract
:1. Introduction
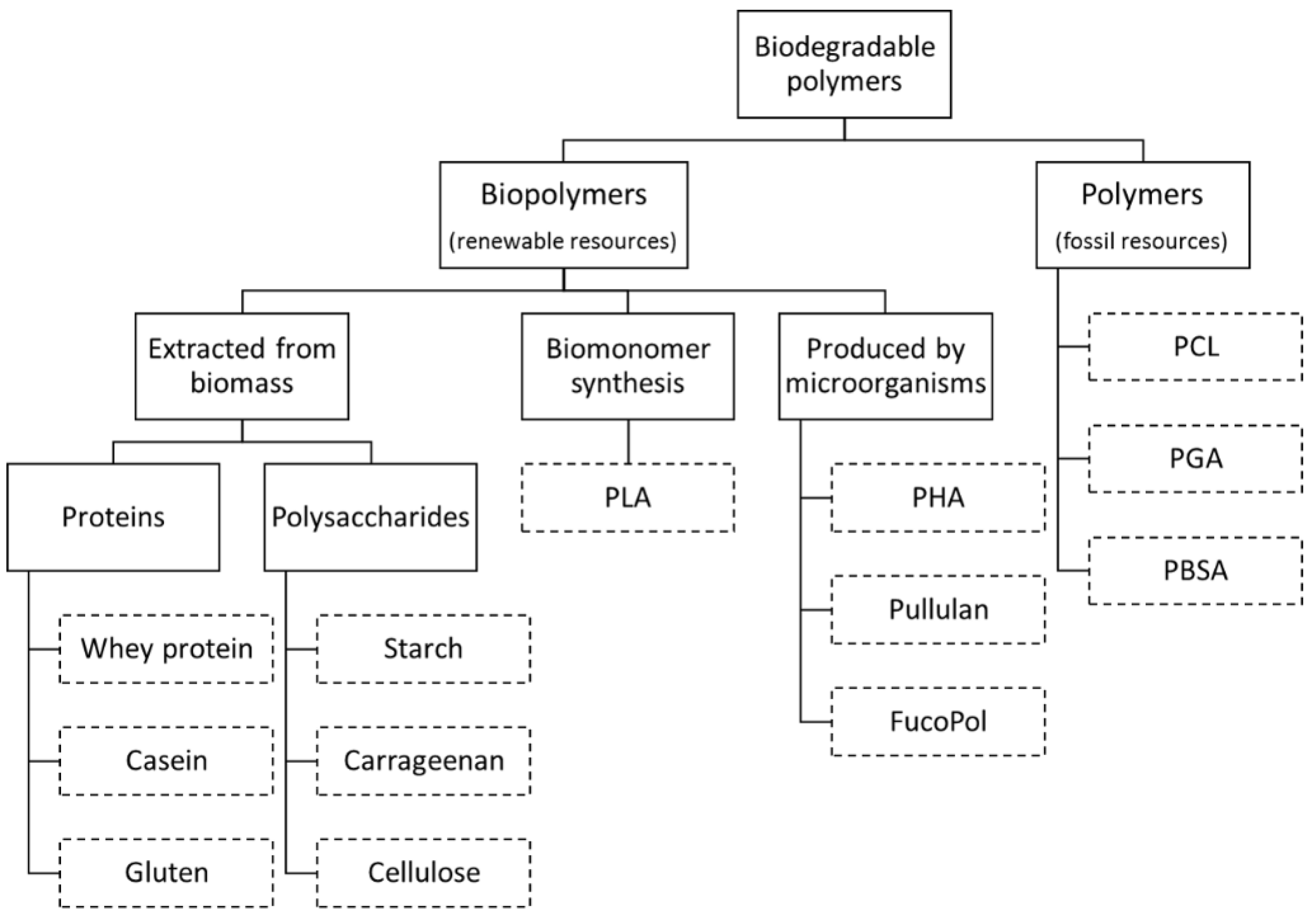
2. Biodegradable Polymers
- Polymers directly extracted/removed from biomass such as polysaccharides (e.g., starch, cellulose, and galactomannans) and proteins (e.g., casein and gluten).
- Polymers produced by chemical synthesis from renewable bio-derived monomers, such as polylactic acid (PLA), a thermoplastic aliphatic polyester derived from lactic acid monomers. The monomer itself is produced via fermentation of carbohydrate feedstocks.
3. Polysaccharides in Food Packaging
3.1. Polysaccharides Obtained from Animals
Chitin and Chitosan
3.2. Polysaccharides Obtained from Plants
3.2.1. Starch
3.2.2. Galactomannans
3.2.3. Cellulose
3.3. Polysaccharides Obtained from Algae
3.3.1. Carrageenan
3.3.2. Alginate
3.4. Polysaccharides Obtained from Microorganisms
3.4.1. Pullulan
3.4.2. Gellan Gum
3.4.3. Xanthan Gum
3.4.4. FucoPol
4. Conclusions and Future Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Coles, R.; McDowell, D.; Kirwan, M.J. Food Packaging Technology; Taylor & Francis: Oxford, United Kingdom, 2003. [Google Scholar]
- 94/62/ec on Packaging and Packaging Waste; European Parliament and Council Directive: European Union, 1994; l0062-en-05.04.2005-001.001.
- Trinetta, V. Definition and function of food packaging. In Reference Module in Food Science; Elsevier: Philadelphia, PA, EUA, 2016. [Google Scholar]
- Plastics Europe. Plastic Packaging: Born to Protect; Plastics Europe—Association of plastics manufacturers: Brussels, Belgium, 2012. [Google Scholar]
- Plastics Europe. Plastics—The facts 2014/2015; Plastics Europe—Association of plastics manufacturers: Brussels, Belgium, 2015. [Google Scholar]
- Plastics Europe. Packaging. Available online: http://www.plasticseurope.org/use-of-plastics/packaging.aspx (accessed on 2 December 2015).
- Plastic Packaging. Plastic packaging innovation. Safety. Sustainability. Available online: http://www.plasticpackagingfacts.org/plastic-packaging (accessed on 2 December 2015).
- American Chemistry Concil. Lifecycle of a plastic product. Available online: http://plastics.americanchemistry.com/Life-Cycle#uses (accessed on 2 December 2015).
- Plackett, D. Introductory overview. In Biopolymers: New Materials for Sustainable Films and Coatings; Wiley: Chichester, UK, 2011; pp. 1–14. [Google Scholar]
- Food and Agriculture Organization of the United Nations. Global food losses and food waste—Extent, causes and prevention; FAO: Rome, Italy, 2011. [Google Scholar]
- GreenFacts. Report highlights—Trends of plastic waste in the EU. Available online: http://www.greenfacts.org/highlights/2011/02/trends-of-plastic-waste-in-the-eu-environment-options-for-regulatory-initiatives/ (accessed on 3 December 2015).
- Tumwesigye, K.S.; Oliveira, J.C.; Sousa-Gallagher, M.J. New sustainable approach to reduce cassava borne environmental waste and develop biodegradable materials for food packaging applications. Food Packag. Shelf Life 2016, 7, 8–19. [Google Scholar] [CrossRef]
- ASTM D5488-94de1. Standard Terminology of Environmental Labeling of Packaging Materials and Packages (Withdrawn 2002); ASTM International: West Conshohocken, PA, USA, 1994; Available online: www.astm.org. [CrossRef]
- Galgano, F. Biodegradable packaging and edible coating for fresh-cut fruits and vegetables. Ital. J. Food Sci. 2015, 27. [Google Scholar] [CrossRef]
- Mensitieri, G.; di Maio, E.; Buonocore, G.G.; Nedi, I.; Oliviero, M.; Sansone, L.; Iannace, S. Processing and shelf life issues of selected food packaging materials and structures from renewable resources. Trends Food Sci. Technol. 2011, 22, 72–80. [Google Scholar] [CrossRef]
- Siracusa, V.; Rocculi, P.; Romani, S.; Rosa, M.D. Biodegradable polymers for food packaging: A review. Trends Food Sci. Technol. 2008, 19, 634–643. [Google Scholar] [CrossRef]
- Cruz-Romero, M.; Kerry, J.P. Crop-based biodegradable packaging and its environmental implications. CAB Rev. Perspect. Agric. Vet. Sci. Nutr. Nat. Resour. 2008, 3, 1–25. [Google Scholar] [CrossRef]
- Coelhoso, I.; Freitas, F.; Alves, V.; Reis, M.M. Biodegradable organic matter. In Encyclopedia of Membranes; Drioli, E., Giorno, L., Eds.; Springer Berlin Heidelberg: Heidelberg, Germany, 2015; pp. 1–2. [Google Scholar]
- Thakur, V.K.; Thakur, M.K. Handbook of sustainable polymers: Processing and applications; Pan Stanford Publishing: Singapore, 2016. [Google Scholar]
- Freitas, F.; Alves, V.D.; Reis, M.A.; Crespo, J.G.; Coelhoso, I.M. Microbial polysaccharide-based membranes: Current and future applications. J. Appl. Polym. Sci. 2014, 131. [Google Scholar] [CrossRef]
- Rhim, J.-W.; Park, H.-M.; Ha, C.-S. Bio-nanocomposites for food packaging applications. Prog. Polym. Sci. 2013, 38, 1629–1652. [Google Scholar] [CrossRef]
- Babu, R.; O’Connor, K.; Seeram, R. Current progress on bio-based polymers and their future trends. Prog. Biomater. 2013, 2, 1–16. [Google Scholar] [CrossRef]
- Van den Broek, L.A.M.; Knoop, R.J.I.; Kappen, F.H.J.; Boeriu, C.G. Chitosan films and blends for packaging material. Carbohydr. Polym. 2015, 116, 237–242. [Google Scholar] [CrossRef] [PubMed]
- Rinaudo, M. Chitin and chitosan: Properties and applications. Prog. Polym. Sci. 2006, 31, 603–632. [Google Scholar] [CrossRef]
- Pereda, M.; Aranguren, M.I.; Marcovich, N.E. Water vapor absorption and permeability of films based on chitosan and sodium caseinate. J. Appl. Polym. Sci. 2009, 111, 2777–2784. [Google Scholar] [CrossRef]
- Elsabee, M.Z.; Abdou, E.S. Chitosan based edible films and coatings: A review. Mater. Sci. Eng. C 2013, 33, 1819–1841. [Google Scholar] [CrossRef] [PubMed]
- Fajardo, P.; Martins, J.T.; Fuciños, C.; Pastrana, L.; Teixeira, J.A.; Vicente, A.A. Evaluation of a chitosan-based edible film as carrier of natamycin to improve the storability of saloio cheese. J. Food Eng. 2010, 101, 349–356. [Google Scholar] [CrossRef]
- Epure, V.; Griffon, M.; Pollet, E.; Avérous, L. Structure and properties of glycerol-plasticized chitosan obtained by mechanical kneading. Carbohydr. Polym. 2011, 83, 947–952. [Google Scholar] [CrossRef]
- Luo, Y.; Wang, Q. Recent development of chitosan-based polyelectrolyte complexes with natural polysaccharides for drug delivery. Int. J. Biol. Macromol. 2014, 64, 353–367. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.X.; Kim, K.M.; Hanna, M.A.; Nag, D. Chitosan–starch composite film: Preparation and characterization. Ind. Crops Prod. 2005, 21, 185–192. [Google Scholar] [CrossRef]
- Jindal, M.; Kumar, V.; Rana, V.; Tiwary, A.K. An insight into the properties of aegle marmelos pectin-chitosan cross-linked films. Int. J. Biol. Macromol. 2013, 52, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Rivero, S.; García, M.A.; Pinotti, A. Composite and bi-layer films based on gelatin and chitosan. J. Food Eng. 2009, 90, 531–539. [Google Scholar] [CrossRef]
- Kurek, M.; Galus, S.; Debeaufort, F. Surface, mechanical and barrier properties of bio-based composite films based on chitosan and whey protein. Food Packag. Shelf Life 2014, 1, 56–67. [Google Scholar] [CrossRef]
- Galus, S.; Kadzińska, J. Food applications of emulsion-based edible films and coatings. Trends Food Sci. Technol. 2015, 45, 273–283. [Google Scholar] [CrossRef]
- Hromiš, N.M.; Lazić, V.L.; Markov, S.L.; Vaštag, Ž.G.; Popović, S.Z.; Šuput, D.Z.; Džinić, N.R.; Velićanski, A.S.; Popović, L.M. Optimization of chitosan biofilm properties by addition of caraway essential oil and beeswax. J. Food Eng. 2015, 158, 86–93. [Google Scholar] [CrossRef]
- Vargas, M.; Perdones, Á.; Chiralt, A.; Cháfer, M.; González-Martínez, C. Effect of homogenization conditions on physicochemical properties of chitosan-based film-forming dispersions and films. Food Hydrocoll. 2011, 25, 1158–1164. [Google Scholar] [CrossRef]
- Sanuja, S.; Agalya, A.; Umapathy, M.J. Synthesis and characterization of zinc oxide–neem oil–chitosan bionanocomposite for food packaging application. Int. J. Biol. Macromol. 2015, 74, 76–84. [Google Scholar] [CrossRef] [PubMed]
- Ojagh, S.M.; Rezaei, M.; Razavi, S.H.; Hosseini, S.M.H. Development and evaluation of a novel biodegradable film made from chitosan and cinnamon essential oil with low affinity toward water. Food Chem. 2010, 122, 161–166. [Google Scholar] [CrossRef]
- Primex. Products—Chitoclear®. Available online: http://www.primex.is/products-services/chitoclear/ (accessed on 3 February 2015).
- Norwegian Chitosan. Products. Available online: http://www.chitosan.no/?page_id=1266 (accessed on 4 February 2016).
- G.T.C. BioCorporation. Gtc bio corporation—Product show. Available online: http://gtcbiotex.en.ec21.com/product_list.html?type=all (accessed on 4 February 2016).
- Ortega-Toro, R.; Morey, I.; Talens, P.; Chiralt, A. Active bilayer films of thermoplastic starch and polycaprolactone obtained by compression molding. Carbohydr. Polym. 2015, 127, 282–290. [Google Scholar] [CrossRef] [PubMed]
- Jiménez, A.; Fabra, M.J.; Talens, P.; Chiralt, A. Effect of sodium caseinate on properties and ageing behaviour of corn starch based films. Food Hydrocoll. 2012, 29, 265–271. [Google Scholar] [CrossRef]
- Souza, A.C.; Benze, R.; Ferrão, E.S.; Ditchfield, C.; Coelho, A.C.V.; Tadini, C.C. Cassava starch biodegradable films: Influence of glycerol and clay nanoparticles content on tensile and barrier properties and glass transition temperature. LWT-Food Sci. Technol. 2012, 46, 110–117. [Google Scholar] [CrossRef]
- Müller, C.M.O.; Yamashita, F.; Laurindo, J.B. Evaluation of the effects of glycerol and sorbitol concentration and water activity on the water barrier properties of cassava starch films through a solubility approach. Carbohydr. Polym. 2008, 72, 82–87. [Google Scholar] [CrossRef]
- Isotton, F.S.; Bernardo, G.L.; Baldasso, C.; Rosa, L.M.; Zeni, M. The plasticizer effect on preparation and properties of etherified corn starchs films. Ind. Crops Prod. 2015, 76, 717–724. [Google Scholar] [CrossRef]
- Abdorreza, M.N.; Cheng, L.H.; Karim, A.A. Effects of plasticizers on thermal properties and heat sealability of sago starch films. Food Hydrocoll. 2011, 25, 56–60. [Google Scholar] [CrossRef]
- Averous, L.; Moro, L.; Dole, P.; Fringant, C. Properties of thermoplastic blends: Starch-polycaprolactone. Polymer 2000, 41, 4157–4167. [Google Scholar] [CrossRef]
- Gupta, A.P.; Kumar, V. New emerging trends in synthetic biodegradable polymers—Polylactide: A critique. Eur. Polym. J. 2007, 43, 4053–4074. [Google Scholar] [CrossRef]
- Avella, M.; de Vlieger, J.J.; Errico, M.E.; Fischer, S.; Vacca, P.; Volpe, M.G. Biodegradable starch/clay nanocomposite films for food packaging applications. Food Chem. 2005, 93, 467–474. [Google Scholar] [CrossRef]
- Novamont. Mater-bi®. Available online: http://www.novamont.com/eng/page.php?id_page=9&id_first=9 (accessed on 11 February 2016).
- Eco-Go. Eco go products. Available online: http://www.eco-go.net/products.php (accessed on 11 February 2016).
- Plantic. Plantic™. Available online: http://www.plantic.com.au/product/plantic-e (accessed on 11 February 2016).
- Cerqueira, M.A.; Bourbon, A.I.; Pinheiro, A.C.; Martins, J.T.; Souza, B.W.S.; Teixeira, J.A.; Vicente, A.A. Galactomannans use in the development of edible films/coatings for food applications. Trends Food Sci. Technol. 2011, 22, 662–671. [Google Scholar] [CrossRef]
- Pawar, H.A.; Lalitha, K.G. Isolation, purification and characterization of galactomannans as an excipient from senna tora seeds. Int. J. Biol.Macromol. 2014, 65, 167–175. [Google Scholar] [CrossRef] [PubMed]
- Prajapati, V.D.; Jani, G.K.; Moradiya, N.G.; Randeria, N.P.; Nagar, B.J.; Naikwadi, N.N.; Variya, B.C. Galactomannan: A versatile biodegradable seed polysaccharide. Int. J. Biol. Macromol. 2013, 60, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Mikkonen, K.S.; Rita, H.; Helén, H.; Talja, R.A.; Hyvönen, L.; Tenkanen, M. Effect of polysaccharide structure on mechanical and thermal properties of galactomannan-based films. Biomacromolecules 2007, 8, 3198–3205. [Google Scholar] [CrossRef] [PubMed]
- Conforti, F.D.; Totty, J.A. Effect of three lipid/hydrocolloid coatings on shelf life stability of golden delicious apples. Int. J. Food Sci. Technol. 2007, 42, 1101–1106. [Google Scholar] [CrossRef]
- Cerqueira, M.A.; Lima, Á.M.; Souza, B.W.S.; Teixeira, J.A.; Moreira, R.A.; Vicente, A.A. Functional polysaccharides as edible coatings for cheese. J. Agric. Food Chem. 2009, 57, 1456–1462. [Google Scholar] [CrossRef] [PubMed]
- Credou, J.; Berthelot, T. Cellulose: From biocompatible to bioactive material. J. Mater. Chem. B 2014, 2, 4767–4788. [Google Scholar] [CrossRef]
- Duan, J.; Reddy, K.O.; Ashok, B.; Cai, J.; Zhang, L.; Rajulu, A.V. Effects of spent tea leaf powder on the properties and functions of cellulose green composite films. J. Environ. Chem. Eng. 2016, 4, 440–448. [Google Scholar] [CrossRef]
- Innovia FILMS. Cellulose film attributes. Available online: http://www.innoviafilms.com/Our-Products/Packaging/Cellulose-Film-Attributes.aspx (accessed on 12 February 2016).
- Prajapati, V.D.; Maheriya, P.M.; Jani, G.K.; Solanki, H.K. Carrageenan: A natural seaweed polysaccharide and its applications. Carbohydr. Polym. 2014, 105, 97–112. [Google Scholar] [CrossRef] [PubMed]
- Tavassoli-Kafrani, E.; Shekarchizadeh, H.; Masoudpour-Behabadi, M. Development of edible films and coatings from alginates and carrageenans. Carbohydr. Polym. 2016, 137, 360–374. [Google Scholar] [CrossRef] [PubMed]
- Necas, J.; Bartosikova, L. Carrageenan: A review. Vet. Med. 2013, 58, 187–205. [Google Scholar]
- Campos, C.A.; Gerschenson, L.N.; Flores, S.K. Development of edible films and coatings with antimicrobial activity. Food Bioprocess Technol. 2011, 4, 849–875. [Google Scholar] [CrossRef]
- McHugh, D.J. A Guide to the Seaweed Industry—FAO Fisheries Technical Paper 441; Food Agriculture Organization of the United Nations: Rome, Italy, 2003. [Google Scholar]
- Bico, S.L.S.; Raposo, M.F.J.; Morais, R.M.S.C.; Morais, A.M.M.B. Combined effects of chemical dip and/or carrageenan coating and/or controlled atmosphere on quality of fresh-cut banana. Food Control 2009, 20, 508–514. [Google Scholar] [CrossRef]
- Plotto, A.; Narciso, J.A.; Rattanapanone, N.; Baldwin, E.A. Surface treatments and coatings to maintain fresh-cut mango quality in storage. J. Sci. Food Agric. 2010, 90, 2333–2341. [Google Scholar] [CrossRef] [PubMed]
- Hambleton, A.; Fabra, M.-J.; Debeaufort, F.; Dury-Brun, C.; Voilley, A. Interface and aroma barrier properties of iota-carrageenan emulsion–based films used for encapsulation of active food compounds. J. Food Eng. 2009, 93, 80–88. [Google Scholar] [CrossRef]
- Fabra, M.J.; Chambin, O.; Voilley, A.; Gay, J.-P.; Debeaufort, F. Influence of temperature and nacl on the release in aqueous liquid media of aroma compounds encapsulated in edible films. J. Food Eng. 2012, 108, 30–36. [Google Scholar] [CrossRef]
- FMC. Carrageenan; Bubnis, W.A., Ed.; Corporation, F.: Philadelphia, PA, USA, 2000. [Google Scholar]
- Jet Net. NutrafilmTM. Available online: http://www.jetnetcorp.com/nutrafilm.html (accessed on 12 February 2016).
- Pawar, S.N.; Edgar, K.J. Alginate derivatization: A review of chemistry, properties and applications. Biomaterials 2012, 33, 3279–3305. [Google Scholar] [CrossRef] [PubMed]
- Vu, C.H.T.; Won, K. Novel water-resistant uv-activated oxygen indicator for intelligent food packaging. Food Chem. 2013, 140, 52–56. [Google Scholar] [CrossRef] [PubMed]
- Blanco-Pascual, N.; Montero, M.P.; Gómez-Guillén, M.C. Antioxidant film development from unrefined extracts of brown seaweeds laminaria digitata and ascophyllum nodosum. Food Hydrocoll. 2014, 37, 100–110. [Google Scholar] [CrossRef]
- CBI. Cbi Product Factsheet: Alginates in Europe; CBI Market Intelligence: Hague, The Netherlands, 2015; p. 13. [Google Scholar]
- Lu, Y.; Sun, Q.; She, X.; Xia, Y.; Liu, Y.; Li, J.; Yang, D. Fabrication and characterisation of α-chitin nanofibers and highly transparent chitin films by pulsed ultrasonication. Carbohydr. Polym. 2013, 98, 1497–1504. [Google Scholar] [CrossRef] [PubMed]
- Srinivasa, P.C.; Tharanathan, R.N. Chitin/chitosan—Safe, ecofriendly packaging materials with multiple potential uses. Food Rev. Int. 2007, 23, 53–72. [Google Scholar] [CrossRef]
- n-chitopack. N-chitopack project—European R&D project. Available online: http://www.n-chitopack.eu/ (accessed on 22 February 2016).
- Kerch, G. Chitosan films and coatings prevent losses of fresh fruit nutritional quality: A review. Trends Food Sci. Technol. 2015, 46, 159–166. [Google Scholar] [CrossRef]
- Cheng, K.C.; Demirci, A.; Catchmark, J.M. Pullulan: Biosynthesis, production, and applications. Appl. Microbiol. Biotechnol. 2011, 92, 29–44. [Google Scholar] [CrossRef] [PubMed]
- Prajapati, V.D.; Jani, G.K.; Khanda, S.M. Pullulan: An exopolysaccharide and its various applications. Carbohydr. Polym. 2013, 95, 540–549. [Google Scholar] [CrossRef] [PubMed]
- Kristo, E.; Biliaderis, C.G. Physical properties of starch nanocrystal-reinforced pullulan films. Carbohydr. Polym. 2007, 68, 146–158. [Google Scholar] [CrossRef]
- Gniewosz, M.; Synowiec, A. Antibacterial activity of pullulan films containing thymol. Flavour Fragr. J. 2011, 26, 389–395. [Google Scholar] [CrossRef]
- Shih, F.F.; Daigle, K.W.; Champagne, E.T. Effect of rice wax on water vapour permeability and sorption properties of edible pullulan films. Food Chem. 2011, 127, 118–121. [Google Scholar] [CrossRef]
- Singh, R.S.; Kaur, N.; Kennedy, J.F. Pullulan and pullulan derivatives as promising biomolecules for drug and gene targeting. Carbohydr. Polym. 2015, 123, 190–207. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Zhong, F.; Li, Y.; Shoemaker, C.F.; Xia, W. Preparation and characterization of pullulan–chitosan and pullulan–carboxymethyl chitosan blended films. Food Hydrocoll. 2013, 30, 82–91. [Google Scholar] [CrossRef]
- Trovatti, E.; Fernandes, S.C.M.; Rubatat, L.; Perez, D.D.S.; Freire, C.S.R.; Silvestre, A.J.D.; Neto, C.P. Pullulan–nanofibrillated cellulose composite films with improved thermal and mechanical properties. Compos. Sci. Technol. 2012, 72, 1556–1561. [Google Scholar] [CrossRef]
- Kanmani, P.; Lim, S.T. Development and characterization of novel probiotic-residing pullulan/starch edible films. Food Chem. 2013, 141, 1041–1049. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-Y.; Choi, Y.-G.; Byul Kim, S.R.; Lim, S.-T. Humidity stability of tapioca starch–pullulan composite films. Food Hydrocoll. 2014, 41, 140–145. [Google Scholar] [CrossRef]
- Tong, Q.; Xiao, Q.; Lim, L.-T. Preparation and properties of pullulan–alginate–carboxymethylcellulose blend films. Food Res. Int. 2008, 41, 1007–1014. [Google Scholar] [CrossRef]
- Zhang, C.; Gao, D.; Ma, Y.; Zhao, X. Effect of gelatin addition on properties of pullulan films. J Food Sci. 2013, 78, C805–C810. [Google Scholar] [CrossRef] [PubMed]
- Jinmei. Product name: Pullulan. Available online: http://www.jinmeibio.com/en/showproduct/?pone=38&id=44 (accessed on 15 February 2016).
- Morris, E.R.; Nishinari, K.; Rinaudo, M. Gelation of gellan—A review. Food Hydrocoll. 2012, 28, 373–411. [Google Scholar] [CrossRef]
- Prajapati, V.D.; Jani, G.K.; Zala, B.S.; Khutliwala, T.A. An insight into the emerging exopolysaccharide gellan gum as a novel polymer. Carbohydr. Polym. 2013, 93, 670–678. [Google Scholar] [CrossRef] [PubMed]
- Kirchmajer, D.M.; Steinhoff, B.; Warren, H.; Clark, R.; in het Panhuis, M. Enhanced gelation properties of purified gellan gum. Carbohydr. Res. 2014, 388, 125–129. [Google Scholar] [CrossRef] [PubMed]
- De Filpo, G.; Palermo, A.M.; Munno, R.; Molinaro, L.; Formoso, P.; Nicoletta, F.P. Gellan gum/titanium dioxide nanoparticle hybrid hydrogels for the cleaning and disinfection of parchment. Int. Biodeterior. Biodegrad. 2015, 103, 51–58. [Google Scholar] [CrossRef]
- CPKelco. Kelcogel® gellan gum book 5th edition. Available online: http://www.appliedbioscience.com/docs/Gellan_Book_5th_Edition.pdf (accessed on 22 February 2016).
- Palaniraj, A.; Jayaraman, V. Production, recovery and applications of xanthan gum by xanthomonas campestris. J. Food Eng. 2011, 106, 1–12. [Google Scholar] [CrossRef]
- Freitas, F.; Alves, V.D.; Reis, M.A.M. Advances in bacterial exopolysaccharides: From production to biotechnological applications. Trends Biotechnol. 2011, 29, 388–398. [Google Scholar] [CrossRef] [PubMed]
- Garcı́a-Ochoa, F.; Santos, V.E.; Casas, J.A.; Gómez, E. Xanthan gum: Production, recovery, and properties. Biotechnol. Adv. 2000, 18, 549–579. [Google Scholar] [CrossRef]
- Freitas, F.; Alves, V.D.; Coelhoso, I.; Reis, M.A.M. Production and food applications of microbial biopolymers. In Engineering Aspects of Food Biotechnology; Teixeira, J.A., Vicente, A.A., Eds.; CRC Press: Boca Raton, FL, USA, 2013. [Google Scholar]
- Faria, S.; de Oliveira Petkowicz, C.L.; de Morais, S.A.L.; Terrones, M.G.H.; de Resende, M.M.; de França, F.P.; Cardoso, V.L. Characterization of xanthan gum produced from sugar cane broth. Carbohydr. Polym. 2011, 86, 469–476. [Google Scholar] [CrossRef]
- Rosalam, S.; England, R. Review of xanthan gum production from unmodified starches by Xanthomonas comprestris sp. Enzyme Microb. Technol. 2006, 39, 197–207. [Google Scholar] [CrossRef]
- Quoc, L.P.T.; Hoa, D.P.; Ngoc, H.T.B.; Phi, T.T.Y. Effect of xanthan gum solution on the preservation of acerola. Cercet. Agron. Mold. 2015, 48, 89–97. [Google Scholar]
- Torres, C.A.V.; Marques, R.; Antunes, S.; Alves, V.D.; Sousa, I.; Ramos, A.M.; Oliveira, R.; Freitas, F.; Reis, M.A.M. Kinetics of production and characterization of the fucose-containing exopolysaccharide from enterobacter A47. J. Biotechnol. 2011, 156, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Alves, V.D.; Freitas, F.; Torres, C.A.V.; Cruz, M.; Marques, R.; Grandfils, C.; Gonçalves, M.P.; Oliveira, R.; Reis, M.A.M. Rheological and morphological characterization of the culture broth during exopolysaccharide production by Enterobacter sp. Carbohydr. Polym. 2010, 81, 758–764. [Google Scholar] [CrossRef]
- Torres, C.A.V.; Marques, R.; Ferreira, A.R.V.; Antunes, S.; Grandfils, C.; Freitas, F.; Reis, M.A.M. Impact of glycerol and nitrogen concentration on enterobacter A47 growth and exopolysaccharide production. Int. J. Biol. Macromol. 2014, 71, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Freitas, F.; Alves, V.D.; Torres, C.A.V.; Cruz, M.; Sousa, I.; Melo, M.J.; Ramos, A.M.; Reis, M.A.M. Fucose-containing exopolysaccharide produced by the newly isolated enterobacter strain A47 DSM 23139. Carbohydr. Polym. 2011, 83, 159–165. [Google Scholar] [CrossRef]
- Torres, C.A.V.; Ferreira, A.R.V.; Freitas, F.; Reis, M.A.M.; Coelhoso, I.; Sousa, I.; Alves, V.D. Rheological studies of the fucose-rich exopolysaccharide fucopol. Int. J. Biol. Macromol. 2015, 79, 611–617. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, A.R.V.; Torres, C.A.V.; Freitas, F.; Reis, M.A.M.; Alves, V.D.; Coelhoso, I.M. Biodegradable films produced from the bacterial polysaccharide fucopol. Int. J. Biol. Macromol. 2014, 71, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, A.R.V.; Torres, C.A.V.; Freitas, F.; Sevrin, C.; Grandfils, C.; Reis, M.A.M.; Alves, V.D.; Coelhoso, I.M. Development and characterization of bilayer films of FucoPol and chitosan. Carbohydr. Polym. 2016, in press. [Google Scholar] [CrossRef]
- Xiao, G.; Zhu, Y.; Wang, L.; You, Q.; Huo, P.; You, Y. Production and storage of edible film using gellan gum. Proced. Environ. Sci. 2011, 8, 756–763. [Google Scholar] [CrossRef]
| Polysaccharide | Composition | Membrane Properties | Main Food Applications | Refs |
|---|---|---|---|---|
| Chitin | N-acetylglucosamine | Biodegradable Antibacterial and fungistatic properties; Biocompatible and non-toxic Highly transparent | Coffee capsules Food bags Packaging films | [24,78,79,80] |
| Chitosan | d-glucosamine N-acetyl-d-glucosamine | Biodegradable Biocompatible and non-toxic Antifungal and antibacterial properties; Good mechanical properties Barrier to gases High water vapor permeability Brittle—need to use plasticizer | Edible membranes and coatings (Strawberries, cherries, mango, guava, among others) Packaging membranes for vegetables and fruit | [14,28,66,79,81] |
| Starch | Glucose | Biodegradable Transparent Odorless and tasteless Retrogradation High elongation and tensile strength | Flexible packaging:
Thermoformed trays and containers for packaging fresh food | [17,66] |
| Galactomannans | Mannose Galactose | Biodegradable EdibleSemi-permeable barrier to gases | Edible membranes and coatings
| [14,54] |
| Cellulose | Glucose | Biodegradable Good mechanical properties Transparent Highly sensitive to water Resistance to fats and oils Need to perform modification, use of plasticizer or polymer blend | Cellophane membranes | [17,66] |
| Carrageenan | Galactose | Biodegradable Fragile and ductile behavior Usually blended with other polymers | Coatings Fruits Meet Encapsulation of aroma compounds | [64,66] |
| Alginate | Mannuronic Glucuronic acid | Biodegradable High water vapor permeability Poor water resistance Strong and brittle membranes Cross-link with calcium | Coatings Prevent water loss in fresh cut fruit (apple, papaya, pear and melon) Inhibition of microbial growth (turkey products) Microwaveable food (increase warming efficiency) | [14,64,66] |
| Polysaccharide | Microorganism | Composition | Membrane Properties | Main Food Applications | Refs. |
|---|---|---|---|---|---|
| Pullulan | Aureobasidium pullulans | Maltotriose (three glucose) | Biodegradable Transparent Edible Oil and grease resistant Heat sealable High water solubility Barrier to oxygen | Coating material Wrapping material Blends with other polymers to improvement of mechanical properties Inner package
| [79,82,93] |
| Gellan gum | Sphingomonas elodea | Glucose Rhamnose Glucuronic acid | Biodegradable Edible Lipid barrier Excellent gas barrier Good tensile strength | Edible Coatings in breading and batters for chicken, fish, cheese, vegetables and potatoes. Encapsulation of flavor and bioactive ingredients | [20,95,114] |
| Xanthan gum | Xanthomonas campestris | Glucose Mannose Glucuronic acid AcetatePyruvate | Biodegradable Edible | Edible coating
| [79,106] |
| FucoPol | Enterobacter A47 | Fucose Galactose Glucose Glucuronic acid Acetate Succinate Pyruvate | Biodegradable Transparent High gas barrier Poor water resistance | Possible application as inner layer in multilayer packaging | [112,113] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferreira, A.R.V.; Alves, V.D.; Coelhoso, I.M. Polysaccharide-Based Membranes in Food Packaging Applications. Membranes 2016, 6, 22. https://doi.org/10.3390/membranes6020022
Ferreira ARV, Alves VD, Coelhoso IM. Polysaccharide-Based Membranes in Food Packaging Applications. Membranes. 2016; 6(2):22. https://doi.org/10.3390/membranes6020022
Chicago/Turabian StyleFerreira, Ana R. V., Vítor D. Alves, and Isabel M. Coelhoso. 2016. "Polysaccharide-Based Membranes in Food Packaging Applications" Membranes 6, no. 2: 22. https://doi.org/10.3390/membranes6020022
APA StyleFerreira, A. R. V., Alves, V. D., & Coelhoso, I. M. (2016). Polysaccharide-Based Membranes in Food Packaging Applications. Membranes, 6(2), 22. https://doi.org/10.3390/membranes6020022








