Composite Anion Exchange Membranes Containing a Long-Side Chain Ionomer and Exfoliated Lamellar Double Hydroxides
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. LDH Synthesis
2.3. LDH Exfoliation
2.4. Synthesis of PPO−Precursor
2.5. Synthesis of PPO-LC
2.6. Preparation of Composite Membranes
3. Characterization Techniques
3.1. 1H-NMR Spectroscopy
3.2. X-Ray Powder Diffraction
3.3. Ion Exchange Capacity
3.4. Thermogravimetric Analysis (TGA)
3.5. Water Uptake
3.6. Membrane Density and Volume Swelling
3.7. Stress–Strain Tests
3.8. Ionic Conductivity
3.9. Accelerated Alkaline Degradation Tests
4. Results and Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Merle, G.; Wessling, M.; Nijmeijer, K. Anion exchange membranes for alkaline fuel cells: A review. J. Membr. Sci. 2011, 377, 1–35. [Google Scholar] [CrossRef]
- Varcoe, J.R.; Atanassov, P.; Dekel, D.R.; Herring, A.M.; Hickner, M.A.; Kohl, P.A.; Kucernak, A.R.; Mustain, W.E.; Nijmeijer, K.; Scott, K.; et al. Anion-exchange membranes in electrochemical energy systems. Energy Environ. Sci. 2014, 7, 3135–3191. [Google Scholar] [CrossRef]
- Arges, C.G.; Zhang, L. Anion Exchange Membranes’ Evolution toward High Hydroxide Ion Conductivity and Alkaline Resiliency. ACS Appl. Energy Mater. 2018, 1, 2991–3012. [Google Scholar] [CrossRef]
- Nallayagari, A.R.; Sgreccia, E.; Pasquini, L.; Vacandio, F.; Kaciulis, S.; Di Vona, M.L.; Knauth, P. Catalytic electrodes for the oxygen reduction reaction based on co-doped (B-N, Si-N, S-N) carbon quantum dots and anion exchange ionomer. Electrochim. Acta 2022, 427, 140861. [Google Scholar] [CrossRef]
- Nallayagari, A.R.; Sgreccia, E.; Pasquini, L.; Sette, M.; Knauth, P.; Di Vona, M.L. Impact of Anion Exchange Ionomers on the Electrocatalytic Performance for the Oxygen Reduction Reaction of B-N Co-doped Carbon Quantum Dots on Activated Carbon. ACS Appl. Mater. Interfaces 2022, 14, 46537–46547. [Google Scholar] [CrossRef] [PubMed]
- Syahputra, S.; Sgreccia, E.; Nallayagari, A.R.; Vacandio, F.; Kaciulis, S.; Di Vona, M.L.; Knauth, P. Influence of Nitrogen Position on the Electrocatalytic Performance of B,N-Codoped Carbon Quantum Dots for the Oxygen Reduction Reaction. J. Electrochem. Soc. 2024, 171, 8. [Google Scholar] [CrossRef]
- Bauer, B.; Strathmann, H.; Effenberger, F. Anion-exchange membranes with improved alkaline stability. Desalination 1990, 79, 125–144. [Google Scholar] [CrossRef]
- Marino, M.G.; Kreuer, K.D. Alkaline Stability of Quaternary Ammonium Cations for Alkaline Fuel Cell Membranes and Ionic Liquids. ChemSusChem 2015, 8, 513–523. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Li, C.; Wang, J.; Li, L.; Wei, Z. A general strategy to enhance the alkaline stability of anion exchange membranes. J. Mater. Chem. A 2017, 5, 6318–6327. [Google Scholar] [CrossRef]
- Becerra-Arciniegas, R.A.; Narducci, R.; Ercolani, G.; Antonaroli, S.; Sgreccia, E.; Pasquini, L.; Knauth, P.; Di Vona, M.L. Alkaline stability of model anion exchange membranes based on poly(phenylene oxide) (PPO) with grafted quaternary ammonium groups: Influence of the functionalization route. Polymer 2019, 185, 121931. [Google Scholar] [CrossRef]
- Cheng, J.; He, G.; Zhang, F. A mini-review on anion exchange membranes for fuel cell applications: Stability issue and addressing strategies. Int. J. Hydrogen Energy 2015, 40, 7348–7360. [Google Scholar] [CrossRef]
- Pandey, T.P.; Sarode, H.N.; Yang, Y.; Yang, Y.; Vezzù, K.; Noto, V.D.; Seifert, S.; Knauss, D.M.; Liberatore, M.W.; Herring, A.M. A Highly Hydroxide Conductive, Chemically Stable Anion Exchange Membrane, Poly(2,6 dimethyl 1,4 phenylene oxide)-b-Poly(vinyl benzyl trimethyl ammonium), for Electrochemical Applications. J. Electrochem. Soc. 2016, 163, H513. [Google Scholar] [CrossRef]
- Narducci, R.; Becerra-Arciniegas, R.A.; Pasquini, L.; Ercolani, G.; Knauth, P.; Di Vona, M.L. Anion-Conducting Polymer Electrolyte without Ether Linkages and with Ionic Groups Grafted on Long Side Chains: Poly(Alkylene Biphenyl Butyltrimethyl Ammonium) (ABBA). Membranes 2022, 12, 337. [Google Scholar] [CrossRef] [PubMed]
- You, W.; Noonan, K.J.T.; Coates, G.W. Alkaline-stable anion exchange membranes: A review of synthetic approaches. Prog. Polym. Sci. 2020, 100, 101177. [Google Scholar] [CrossRef]
- Lin, C.; Gao, Y.; Li, N.; Zhang, M.; Luo, J.; Deng, Y.; Ling, L.; Zhang, Y.; Cheng, F.; Zhang, S. Quaternized Tröger’s base polymer with crown ether unit for alkaline stable anion exchange membranes. Electrochim. Acta 2020, 354, 136693. [Google Scholar] [CrossRef]
- Dang, H.-S.; Jannasch, P. Exploring Different Cationic Alkyl Side Chain Designs for Enhanced Alkaline Stability and Hydroxide Ion Conductivity of Anion-Exchange Membranes. Macromolecules 2015, 48, 5742–5751. [Google Scholar] [CrossRef]
- Zhu, L.; Yu, X.; Hickner, M.A. Exploring backbone-cation alkyl spacers for multi-cation side chain anion exchange membranes. J. Power Sources 2018, 375, 433–441. [Google Scholar] [CrossRef]
- Long, H.; Kim, K.; Pivovar, B.S. Hydroxide Degradation Pathways for Substituted Trimethylammonium Cations: A DFT Study. J. Phys. Chem. C 2012, 116, 9419–9426. [Google Scholar] [CrossRef]
- Lee, W.-H.; Kim, Y.S.; Bae, C. Robust Hydroxide Ion Conducting Poly(biphenyl alkylene)s for Alkaline Fuel Cell Membranes. ACS Macro Lett. 2015, 4, 814–818. [Google Scholar] [CrossRef]
- Gokulapriyan, R.; Arunkumar, I.; Lee, H.-K.; Yoo, D.J. Anion Exchange Membrane Based on Cross-Linked Poly(2,6-dimethyl-1,4-phenylene oxide)/Poly(dimethylaminophenyl bisdiphenol) for Fuel Cell Applications. ACS Appl. Energy Mater. 2023, 6, 12549–12559. [Google Scholar] [CrossRef]
- Gokulapriyan, R.; Karthikeyan, S.C.; Yoo, D.J. Synthesis and characterization of ionic liquid incorporated quaternized poly(2,6-dimethyl-1,4-phenylene oxide) and NiO/IrO2 electrocatalyst for anion exchange membrane fuel cell applications. J. Mater. Chem. A 2024, 12, 23880–23896. [Google Scholar] [CrossRef]
- Di Vona, M.L.; Casciola, M.; Donnadio, A.; Nocchetti, M.; Pasquini, L.; Narducci, R.; Knauth, P. Anionic conducting composite membranes based on aromatic polymer and layered double hydroxides. Int. J. Hydrogen Energy 2017, 42, 3197–3205. [Google Scholar] [CrossRef]
- Pasquini, L.; Becerra-Arciniegas, R.-A.; Narducci, R.; Sgreccia, E.; Gressel, V.; Di Vona, M.-L.; Knauth, P. Properties and Alkaline Stability of Composite Anion Conducting Ionomers Based on Poly(phenylene oxide) Grafted with DABCO and Mg/Al Lamellar Double Hydroxide. ChemElectroChem 2020, 7, 2917–2924. [Google Scholar] [CrossRef]
- Mishra, G.; Dash, B.; Pandey, S. Layered double hydroxides: A brief review from fundamentals to application as evolving biomaterials. Appl. Clay Sci. 2018, 153, 172–186. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, F.; Zhang, R.; Evans, D.G.; Duan, X. Preparation of Layered Double-Hydroxide Nanomaterials with a Uniform Crystallite Size Using a New Method Involving Separate Nucleation and Aging Steps. Chem. Mater. 2002, 14, 4286–4291. [Google Scholar] [CrossRef]
- Bukhtiyarova, M.V. A review on effect of synthesis conditions on the formation of layered double hydroxides. J. Solid State Chem. 2019, 269, 494–506. [Google Scholar] [CrossRef]
- Lopez, T.; Bosch, P.; Ramos, E.; Gomez, R.; Novaro, O.; Acosta, D.; Figueras, F. Synthesis and Characterization of Sol−Gel Hydrotalcites. Structure and Texture. Langmuir 1996, 12, 189–192. [Google Scholar] [CrossRef]
- Tadanaga, K.; Miyata, A.; Ando, D.; Yamaguchi, N.; Tatsumisago, M. Preparation of Co–Al and Ni–Al layered double hydroxide thin films by a sol–gel process with hot water treatment. J. Sol-Gel Sci. Technol. 2012, 62, 111–116. [Google Scholar] [CrossRef]
- Xu, Z.P.; Lu, G.Q. Hydrothermal Synthesis of Layered Double Hydroxides (LDHs) from Mixed MgO and Al2O3: LDH Formation Mechanism. Chem. Mater. 2005, 17, 1055–1062. [Google Scholar] [CrossRef]
- Lu, Y.; He, Y.; Li, J.; Wang, N.; An, Q.-F. One-step exfoliation and etching of layered double hydroxide nanosheets to construct nanofiltration membranes with high water permeance. Sep. Purif. Technol. 2025, 354, 129180. [Google Scholar] [CrossRef]
- Bao, W.; Tang, Y.; Yu, J.; Yan, W.; Wang, C.; Li, Y.; Wang, Z.; Yang, J.; Zhang, L.; Yu, F. Si-doped ZnAl-LDH nanosheets by layer-engineering for efficient photoelectrocatalytic water splitting. Appl. Catal. B Environ. Energy 2024, 346, 123706. [Google Scholar] [CrossRef]
- Phan, M.V.; Tran, T.K.T.; Pham, Q.N.; Do, M.H.; Nguyen, T.H.N.; Nguyen, M.T.; Phan, T.T.; To, T.X.H. Controllable synthesis of layered double hydroxide nanosheets to build organic inhibitor-loaded nanocontainers for enhanced corrosion protection of carbon steel. Nanoscale Adv. 2024, 6, 606–619. [Google Scholar] [CrossRef] [PubMed]
- Fan, Z.-W.; Zhang, J.; Zuo, B.-Y.; Tian, X.-B.; Wang, Z.; Ju, X.-J.; Xie, R.; Wang, W.; Pan, D.-W.; Liu, Z.; et al. Biomimetic Guttation Feature in 2D Hydrotalcite Membranes for Self-Sustaining Water Purification. Adv. Funct. Mater. 2024, 34, 2316247. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, H.; Lu, S. Preparation and application of layered double hydroxide nanosheets. RSC Adv. 2021, 11, 24254–24281. [Google Scholar] [CrossRef]
- Yu, J.; Wang, Q.; O’Hare, D.; Sun, L. Preparation of two dimensional layered double hydroxide nanosheets and their applications. Chem. Soc. Rev. 2017, 46, 5950–5974. [Google Scholar] [CrossRef] [PubMed]
- Hou, W.; Kang, L.; Sun, R.; Liu, Z.-H. Exfoliation of layered double hydroxides by an electrostatic repulsion in aqueous solution. Colloids Surf. A Physicochem. Eng. Asp. 2008, 312, 92–98. [Google Scholar] [CrossRef]
- Wang, J.; Huang, L.; Gao, Y.; Yang, R.; Zhang, Z.; Guo, Z.; Wang, Q. A simple and reliable method for determining the delamination degree of nitrate and glycine intercalated LDHs in formamide. Chem. Commun. 2014, 50, 10130–10132. [Google Scholar] [CrossRef]
- Wang, S.; Qin, S.; Yang, G.; Liu, Y.; Yang, T.; Wang, Z.; Li, X.; Liu, D.; Lei, W. Phast Solid-Phase Exfoliation of Layered Double Hydroxides with Tunable Functionalization. ACS Appl. Mater. Interfaces 2024, 16, 69725–69732. [Google Scholar] [CrossRef] [PubMed]
- Mao, N.; Zhou, C.H.; Tong, D.S.; Yu, W.H.; Lin, C.C. Exfoliation of layered double hydroxide solids into functional nanosheets. Appl. Clay Sci. 2017, 144, 60–78. [Google Scholar] [CrossRef]
- Ma, R.; Liu, Z.; Li, L.; Iyi, N.; Sasaki, T. Exfoliating layered double hydroxides in formamide: A method to obtain positively charged nanosheets. J. Mater. Chem. 2006, 16, 3809–3813. [Google Scholar] [CrossRef]
- Chen, C.; Tao, L.; Du, S.; Chen, W.; Wang, Y.; Zou, Y.; Wang, S. Advanced Exfoliation Strategies for Layered Double Hydroxides and Applications in Energy Conversion and Storage. Adv. Funct. Mater. 2020, 30, 1909832. [Google Scholar] [CrossRef]
- Yuan, Y.; Tong, C.; Lü, C. Mussel-inspired functionalized LDH as covalent crosslinkers for constructing micro-crosslinking fluorenyl-containing polysulfone-based composite anion exchange membranes with enhanced properties. Appl. Clay Sci. 2020, 199, 105878. [Google Scholar] [CrossRef]
- Vinodh, R.; Atchudan, R.; Kim, H.-J.; Yi, M. Recent Advancements in Polysulfone Based Membranes for Fuel Cell (PEMFCs, DMFCs and AMFCs) Applications: A Critical Review. Polymers 2022, 14, 300. [Google Scholar] [CrossRef]
- Xian, F.; Jia, L.; Sugahara, Y.; Xue, H.; Yamauchi, Y.; Sasaki, T.; Ma, R. Constructing Fast Transmembrane Pathways in a Layered Double Hydroxide Nanosheets/Nanoparticles Composite Film for an Inorganic Anion-Exchange Membrane. ACS Appl. Mater. Interfaces 2022, 14, 51212–51221. [Google Scholar] [CrossRef] [PubMed]
- Sun, P.; Ma, R.; Sasaki, T. Recent progress on exploring exceptionally high and anisotropic H+/OH− ion conduction in two-dimensional materials. Chem. Sci. 2018, 9, 33–43. [Google Scholar] [CrossRef]
- Wang, Y.; Ying, Z.; Gao, Y.; Shi, L. Layered Double Hydroxide Nanosheets: Synthesis Strategies and Applications in the Field of Energy Conversion. Chem. A Eur. J. 2024, 30, e202303025. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.; Long, C.; Li, Y.; Wang, D.; Zhu, H. High-performance layered double hydroxide/poly(2,6-dimethyl-1,4-phenylene oxide) membrane with porous sandwich structure for anion exchange membrane fuel cell applications. J. Membr. Sci. 2018, 552, 51–60. [Google Scholar] [CrossRef]
- Ni, J.; Wang, J.; Zhao, S.; Zhong, F.; Qu, T.; Hu, F.; Liu, H.; Gong, C.; Wen, S. LDH nanosheets anchored on bacterial cellulose-based composite anion exchange membranes for significantly enhanced strength and ionic conductivity. Appl. Clay Sci. 2022, 217, 106391. [Google Scholar] [CrossRef]
- He, X.; Cao, L.; He, G.; Zhao, A.; Mao, X.; Huang, T.; Li, Y.; Wu, H.; Sun, J.; Jiang, Z. A highly conductive and robust anion conductor obtained via synergistic manipulation in intra- and inter-laminate of layered double hydroxide nanosheets. J. Mater. Chem. A 2018, 6, 10277–10285. [Google Scholar] [CrossRef]
- Liu, Z.; Ma, R.; Osada, M.; Iyi, N.; Ebina, Y.; Takada, K.; Sasaki, T. Synthesis, Anion Exchange, and Delamination of Co−Al Layered Double Hydroxide: Assembly of the Exfoliated Nanosheet/Polyanion Composite Films and Magneto-Optical Studies. J. Am. Chem. Soc. 2006, 128, 4872–4880. [Google Scholar] [CrossRef]
- Becerra-Arciniegas, R.A.; Narducci, R.; Ercolani, G.; Sgreccia, E.; Pasquini, L.; Di Vona, M.L.; Knauth, P. Model Long Side-Chain PPO-Based Anion Exchange Ionomers: Properties and Alkaline Stability. J. Phys. Chem. C 2020, 124, 1309–1316. [Google Scholar] [CrossRef]
- Li, X.-G. High-resolution thermogravimetry of poly(2,6-dimethyl-1,4-phenylene oxide). J. Appl. Polym. Sci. 1999, 71, 1887–1892. [Google Scholar] [CrossRef]
- Ehlers, G.F.L.; Fisch, K.R.; Powell, W.R.J.J.o.P.S.P.A. Thermal degradation of polymers with phenylene units in the chain. III. Polyarylates. J. Polym. Sci. Part A-1 Polym. Chem. 1969, 7, 2969–2981. [Google Scholar] [CrossRef]
- Kameda, T.; Fubasami, Y.; Uchiyama, N.; Yoshioka, T. Elimination behavior of nitrogen oxides from a NO3−-intercalated Mg–Al layered double hydroxide during thermal decomposition. Thermochim. Acta 2010, 499, 106–110. [Google Scholar] [CrossRef]
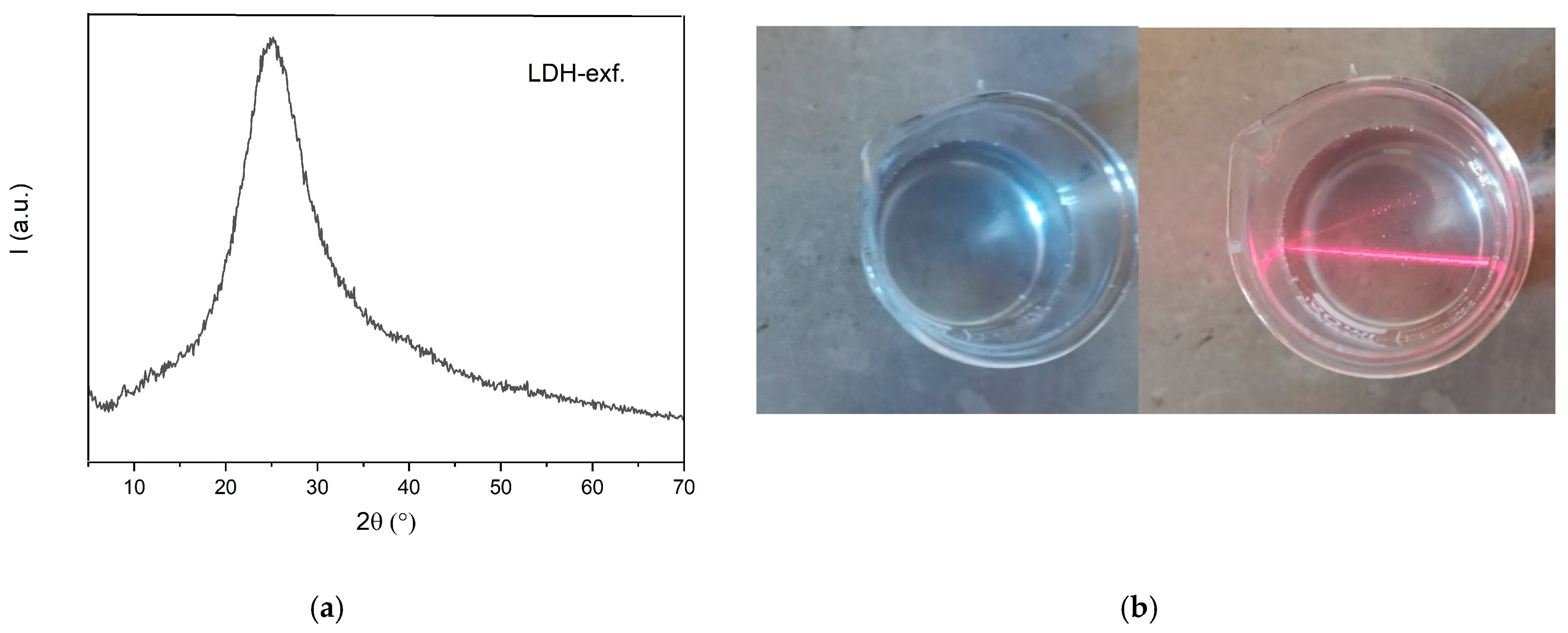

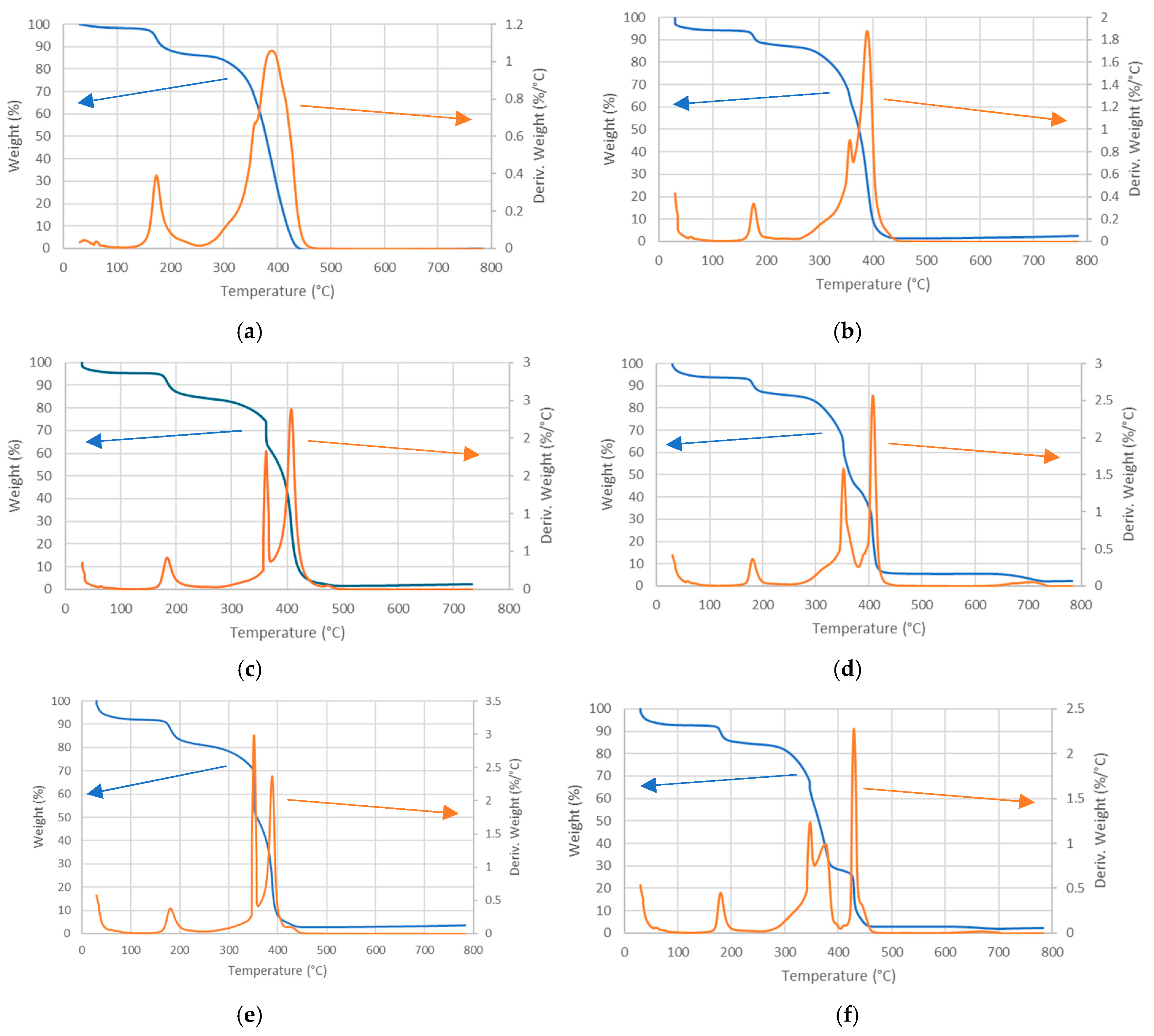
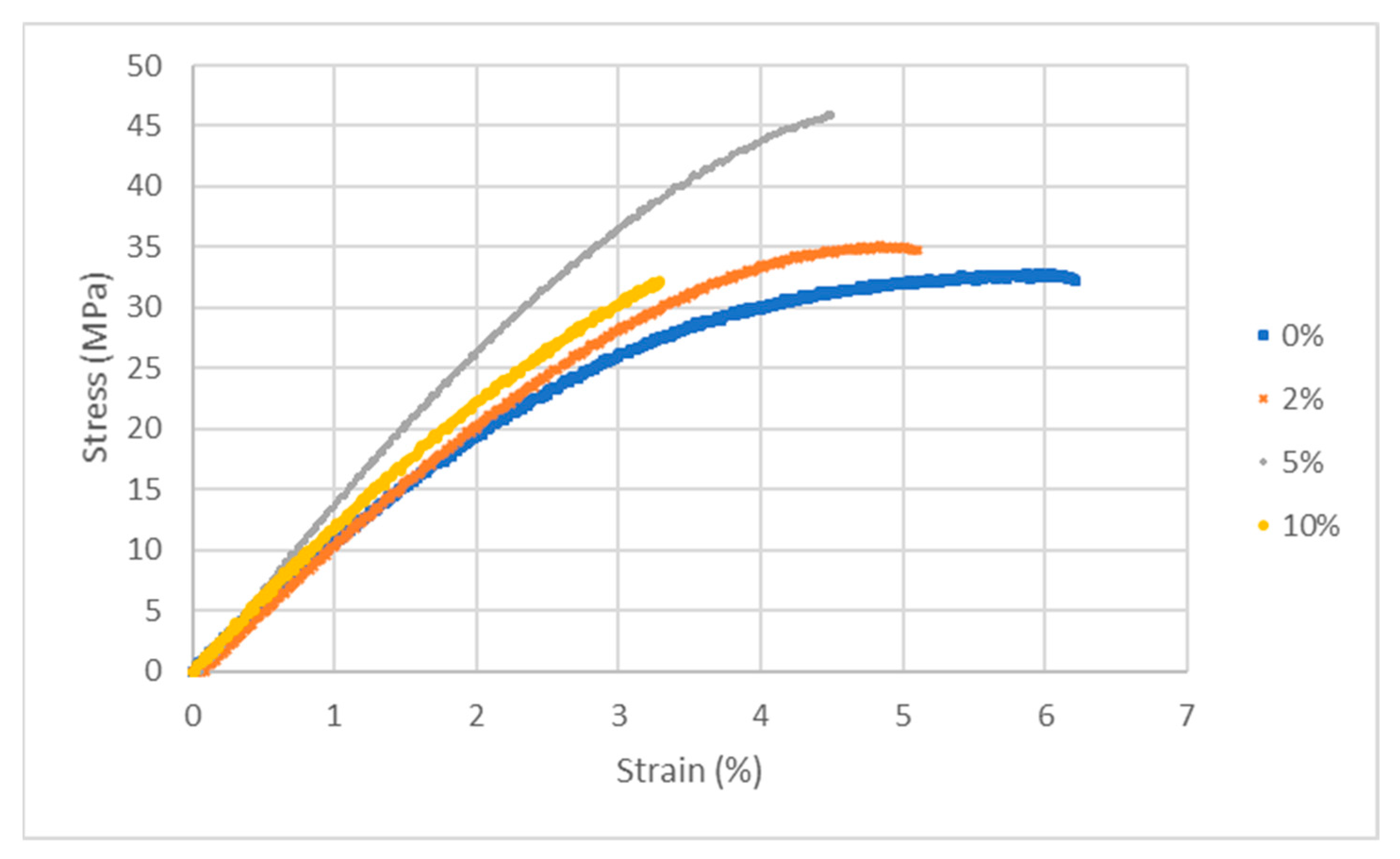
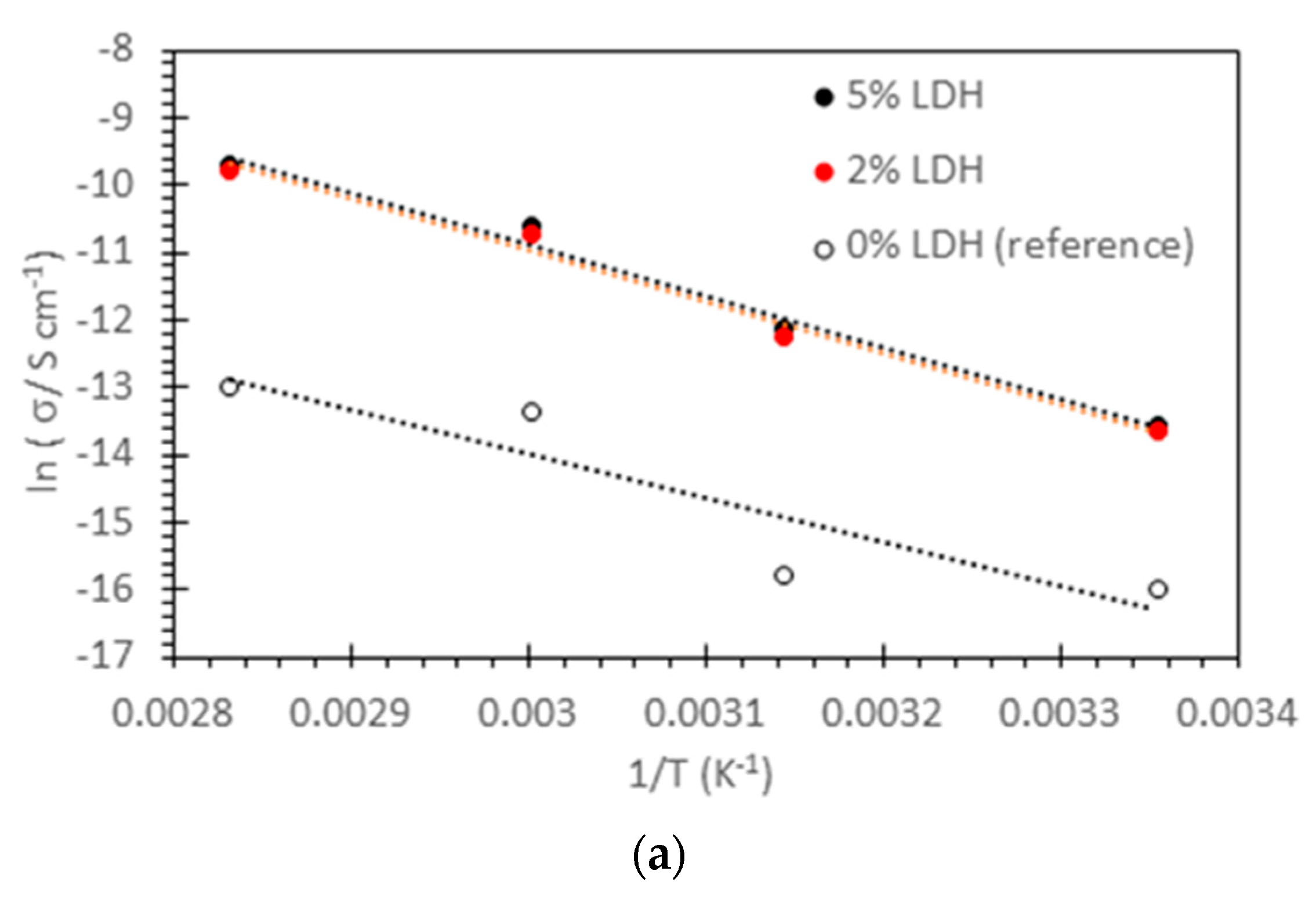

| (a) PPO-LC-LDH (Cl−) | ||||
|---|---|---|---|---|
| % LDH | WU (%) | SV (%) | Ddry (g/cm3) | |
| 0 | 32.0 | - | - | |
| 2 | 30.3 | 39.6 | 1.07 | |
| 5 | 21.3 | 21.3 | 1.24 | |
| 10 | 14.6 | 15.9 | 1.27 | |
| (b) PPO-LC-LDH (OH−) | ||||
| % LDH | WU (%) | Sv (%) | Ddry (g/cm3) | IEC (meq/g) |
| 0 | 46 | - | 0.90 | - |
| 2 | 41.2 | 46.0 | 1.09 | 1.27 |
| 5 | 39.8 | 42.0 | 1.23 | 1.24 |
| 10 | 34.3 | 29.0 | 1.35 | 1.20 |
| (c) PPO-LC-LDH (OH−) After Accelerated Degradation Test (2 M KOH, 80 °C, 3 Days) | ||||
| % LDH | WU (%) | Sv (%) | Ddry (g/cm3) | |
| 0 | 22.0 | 14.6 | - | |
| 2 | 25.8 | 24.1 | 1.09 | |
| 5 | 39.8 | 36.6 | 1.22 | |
| 10 | 33.7 | 25.7 | 1.24 | |
| % LDH | Mass Loss Around 180 °C (%) | Variation (%) |
|---|---|---|
| 0 | 12.2 | 36 |
| After Degradation | 7.8 | |
| 2 | 12.0 | 28 |
| After Degradation | 8.7 | |
| 5 | 11.2 | 19 |
| After Degradation | 9.1 |
| % LDH | Young’s Modulus (MPa) | Yield Stress (MPa) | Ultimate Stress (MPa) | Elongation @ Break (%) |
|---|---|---|---|---|
| 0 | 1020 ± 20 | 20 ± 6 | 32 ± 2 | 6 ± 1 |
| 2 | 1040 ± 25 | 15 ± 1 | 32 ± 4 | 4 ± 1 |
| 5 | 1380 ± 2 | 42 ± 1 | 44 ± 3 | 4 ± 1 |
| 10 | 1150 ± 7 | 7 ± 1 | 31 ± 2 | 3 ± 1 |
| PPO-LC (mS/cm) | PPO-LC 2%LDH (mS/cm) | PPO-LC 5%LDH (mS/cm) | PPO-LC 10%LDH (mS/cm) | ||||
|---|---|---|---|---|---|---|---|
| Temperature | As Cast | After Degradation | As Cast | After Degradation | As Cast | After Degradation | As Cast |
| 25 °C | 1.0 | 0.6 | 2.6 | 0.9 | 1.6 | 1.3 | 2.6 |
| 45 °C | 1.5 | 0.9 | 4.2 | 1.3 | 3.7 | 2.1 | 4.0 |
| 60 °C | 2.0 | 1.3 | 5.4 | 1.7 | 6 | 2.5 | 5.1 |
| 80 °C | 2.4 | 2 | 7.3 | 2.3 | 9.9 | 3.9 | 6.8 |
| 60 °C cool | 1.9 | 1.3 | 5.1 | 2 | 8.1 | 3.1 | 5.0 |
| 45 °C cool | 1.5 | 1.2 | 3.9 | 1.5 | 6.3 | 2.6 | 3.7 |
| 25 °C cool | 0.9 | 0.9 | 2.5 | 0.9 | 4 | 1.5 | 2.4 |
| Membrane | T/°C | RH/% | |||
|---|---|---|---|---|---|
| 30 | 50 | 70 | 95 | ||
| PPO-LC 0% LDH | 25 | 1.1 10−7 | 2.4 10−6 | 1.7 10−5 | 1.4 10−3 |
| 45 | 1.4 10−7 | 1.0 10−5 | 3.7 10−5 | 1.6 10−3 | |
| 60 | 1.6 10−6 | 1.9 10−5 | 5.7 10−4 | 9.8 10−4 | |
| 80 | 2.3 10−6 | 1.3 10−5 | 9.7 10−5 | 3.3 10−4 | |
| PPO-LC 2% LDH | 25 | 1.2 10−6 | 1.5 10−4 | 4.3 10−4 | 7.3 10−3 |
| 45 | 4.8 10−6 | 2.2 10−4 | 9.8 10−4 | 8.5 10−3 | |
| 60 | 2.2 10−5 | 3.8 10−4 | 8.6 10−4 | 1.2 10−2 | |
| 80 | 5.6 10−5 | 5.9 10−4 | 1.8 10−3 | 4.3 10−3 | |
| PPO-LC 5% LDH | 25 | 1.3 10−6 | 1.9 10−4 | 8.0 10−4 | 3.9 10−3 |
| 45 | 5.3 10−6 | 5.9 10−4 | 1.6 10−3 | 8.8 10−3 | |
| 60 | 2.5 10−5 | 5.1 10−4 | 1.4 10−3 | 1.3 10−2 | |
| 80 | 6.2 10−5 | 5.8 10−4 | 2.7 10−3 | 8.4 10−3 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Narducci, R.; Syahputra, S.; Di Vona, M.L.; Knauth, P.; Pasquini, L. Composite Anion Exchange Membranes Containing a Long-Side Chain Ionomer and Exfoliated Lamellar Double Hydroxides. Membranes 2024, 14, 275. https://doi.org/10.3390/membranes14120275
Narducci R, Syahputra S, Di Vona ML, Knauth P, Pasquini L. Composite Anion Exchange Membranes Containing a Long-Side Chain Ionomer and Exfoliated Lamellar Double Hydroxides. Membranes. 2024; 14(12):275. https://doi.org/10.3390/membranes14120275
Chicago/Turabian StyleNarducci, Riccardo, Suanto Syahputra, Maria Luisa Di Vona, Philippe Knauth, and Luca Pasquini. 2024. "Composite Anion Exchange Membranes Containing a Long-Side Chain Ionomer and Exfoliated Lamellar Double Hydroxides" Membranes 14, no. 12: 275. https://doi.org/10.3390/membranes14120275
APA StyleNarducci, R., Syahputra, S., Di Vona, M. L., Knauth, P., & Pasquini, L. (2024). Composite Anion Exchange Membranes Containing a Long-Side Chain Ionomer and Exfoliated Lamellar Double Hydroxides. Membranes, 14(12), 275. https://doi.org/10.3390/membranes14120275










