Progress and Prospects of Nanocellulose-Based Membranes for Desalination and Water Treatment
Abstract
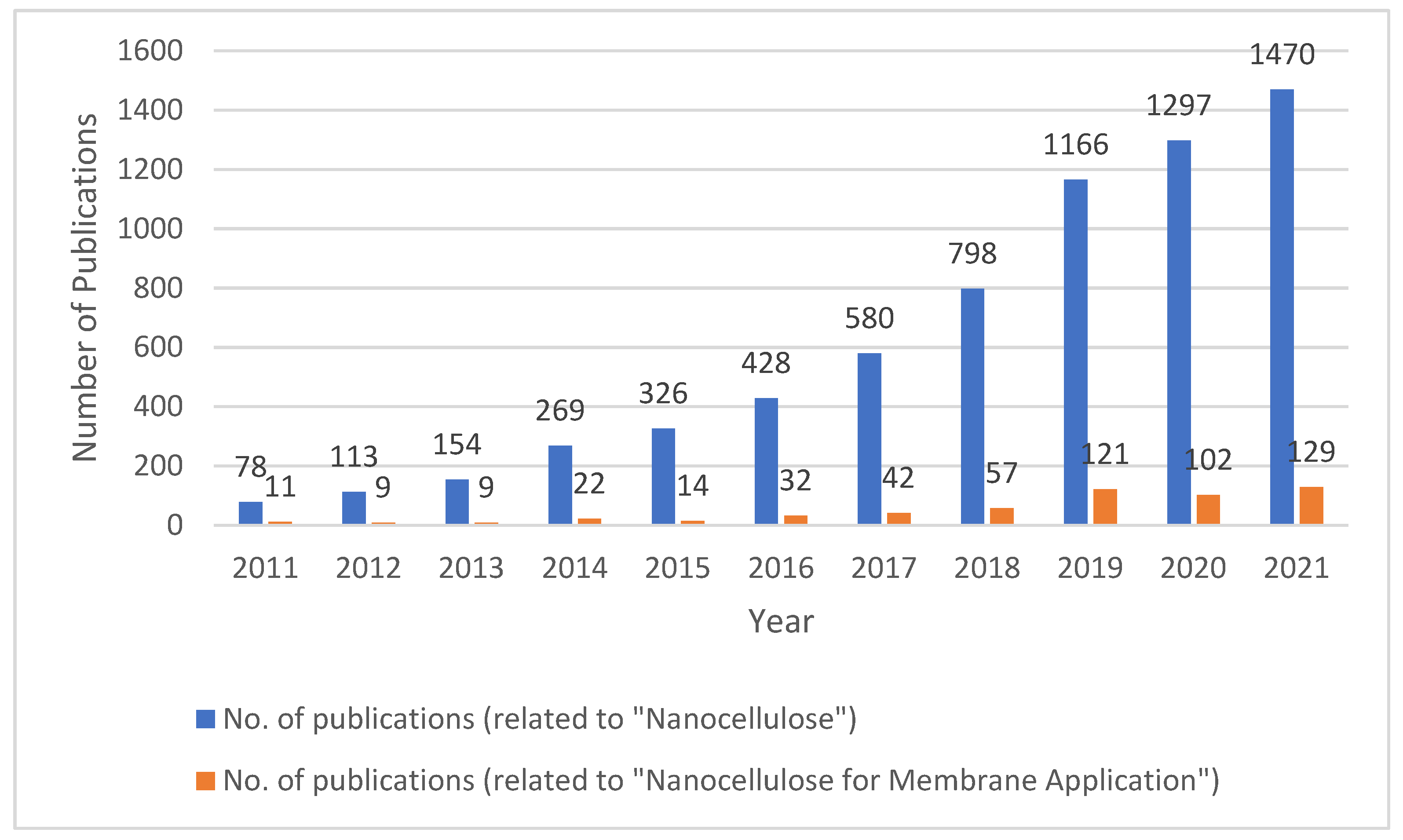
1. Introduction
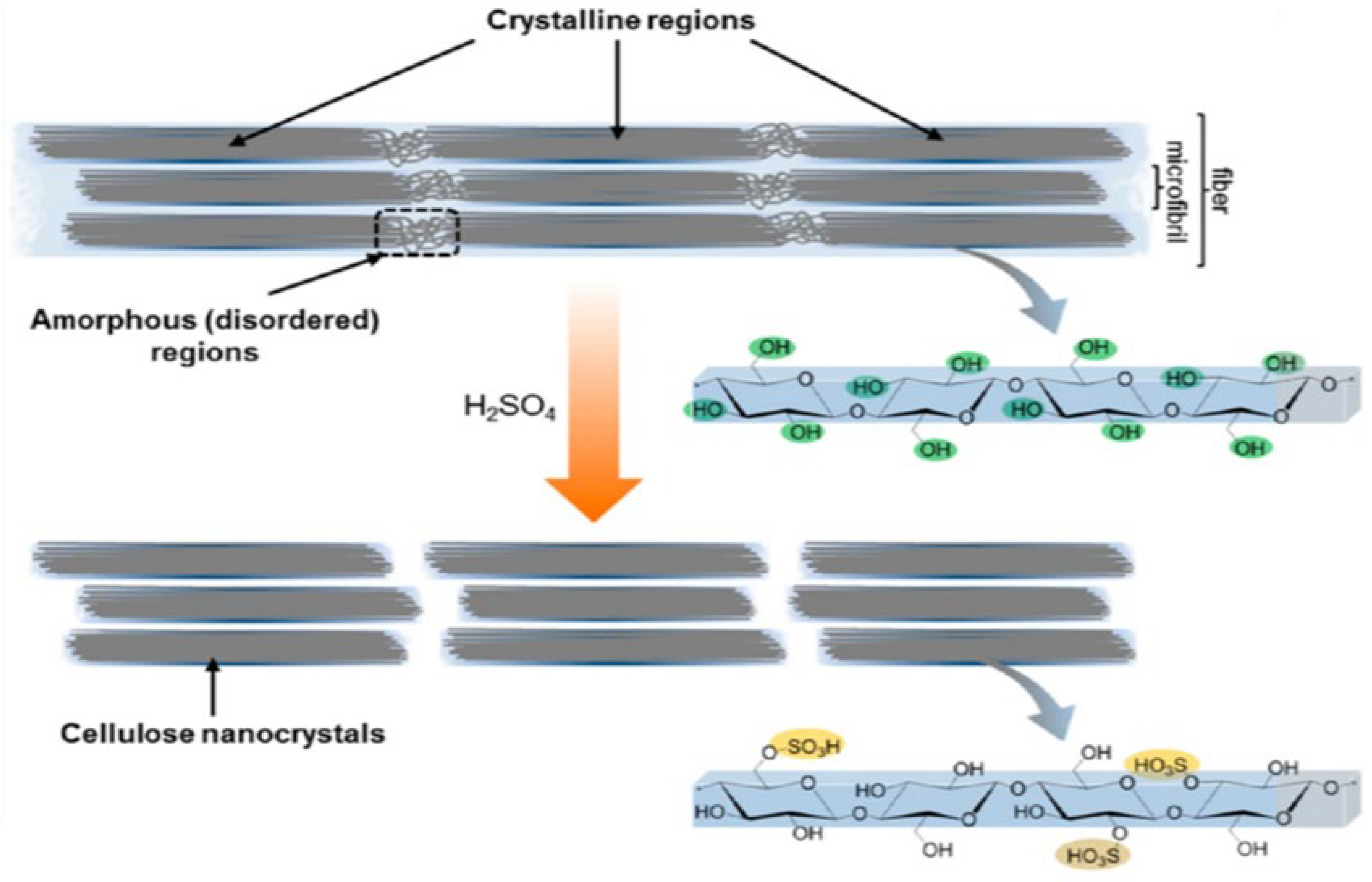
2. Structure and Properties of Nanocellulose
3. Sources for Obtaining Nanocellulose
4. Methods for Obtaining Nanocellulose
5. Application of Cellulose Nanocrystals in Desalination Application
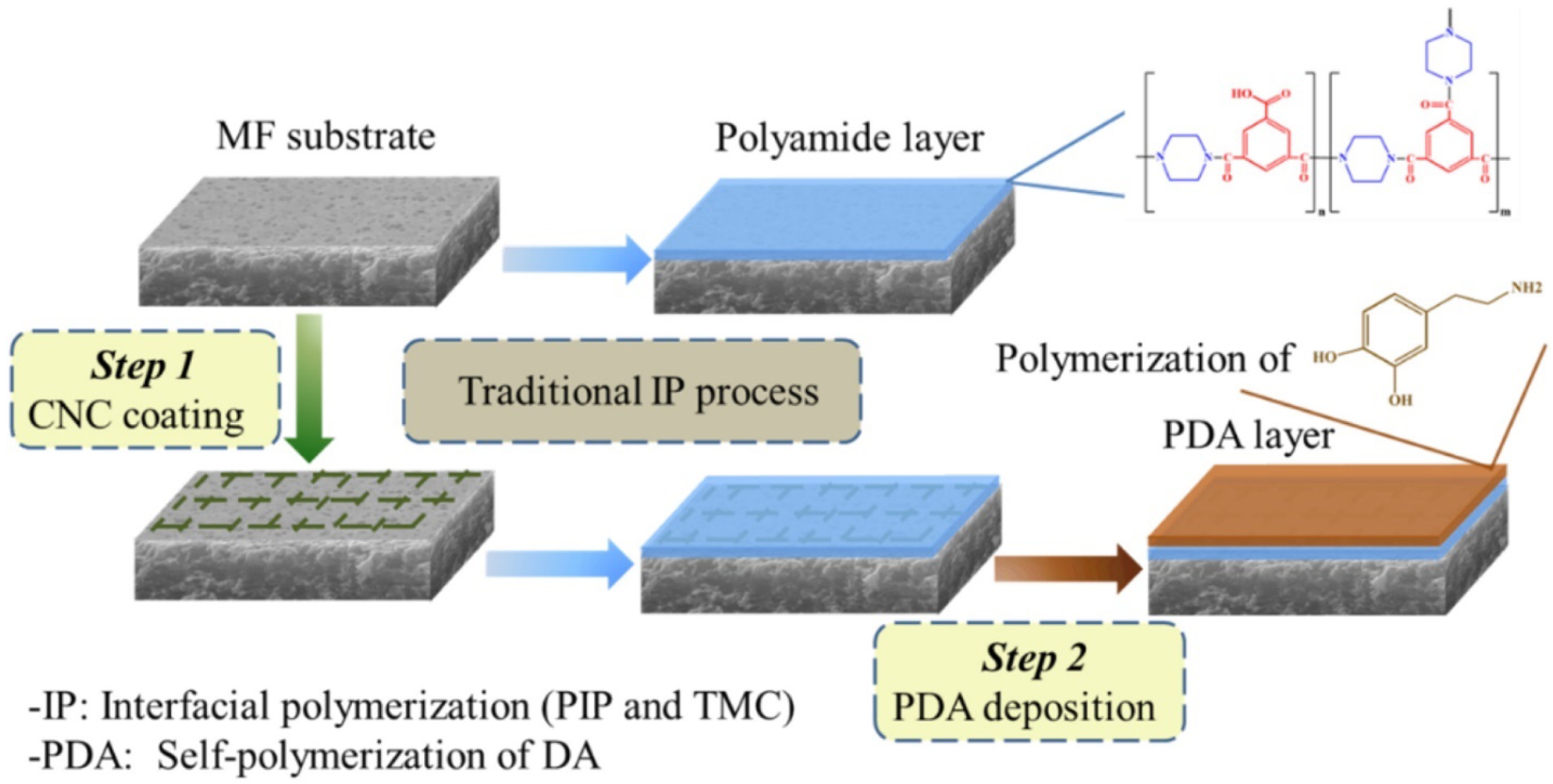
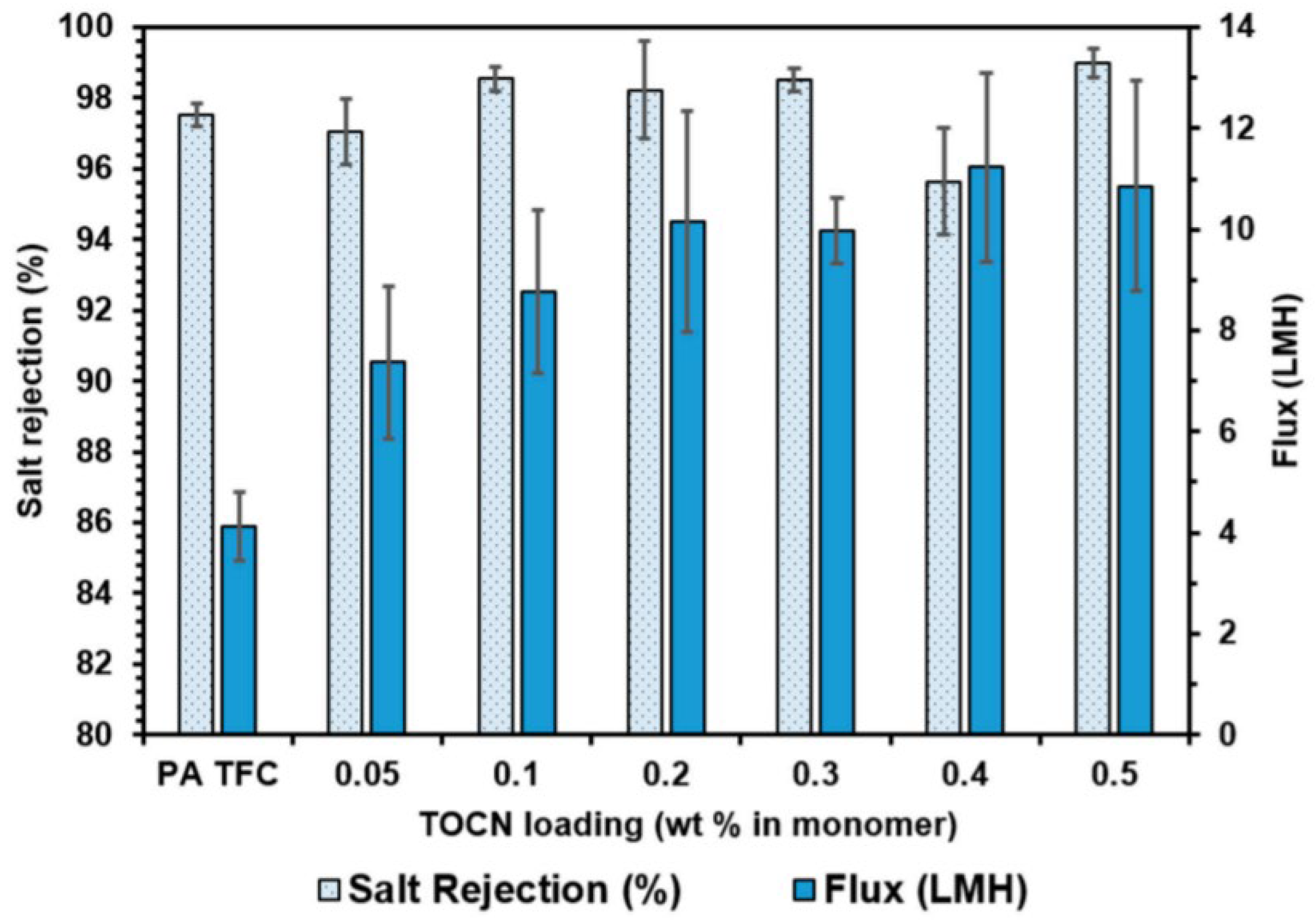
5.1. Cellulose Nanocrystals Application in Nanofiltration
5.2. Cellulose Nanocrystals Application in Reverse Osmosis
5.3. Cellulose Nanocrystals in Pervaporation
6. Cellulose Nanofibrils (CNF): Structure, Properties, and Desalination Application
6.1. Cellulose Nanofibers in Ultrafiltration Application
6.2. Cellulose Nanofibers in Nanofiltration
6.3. Cellulose Nanofibers in Reverse Osmosis
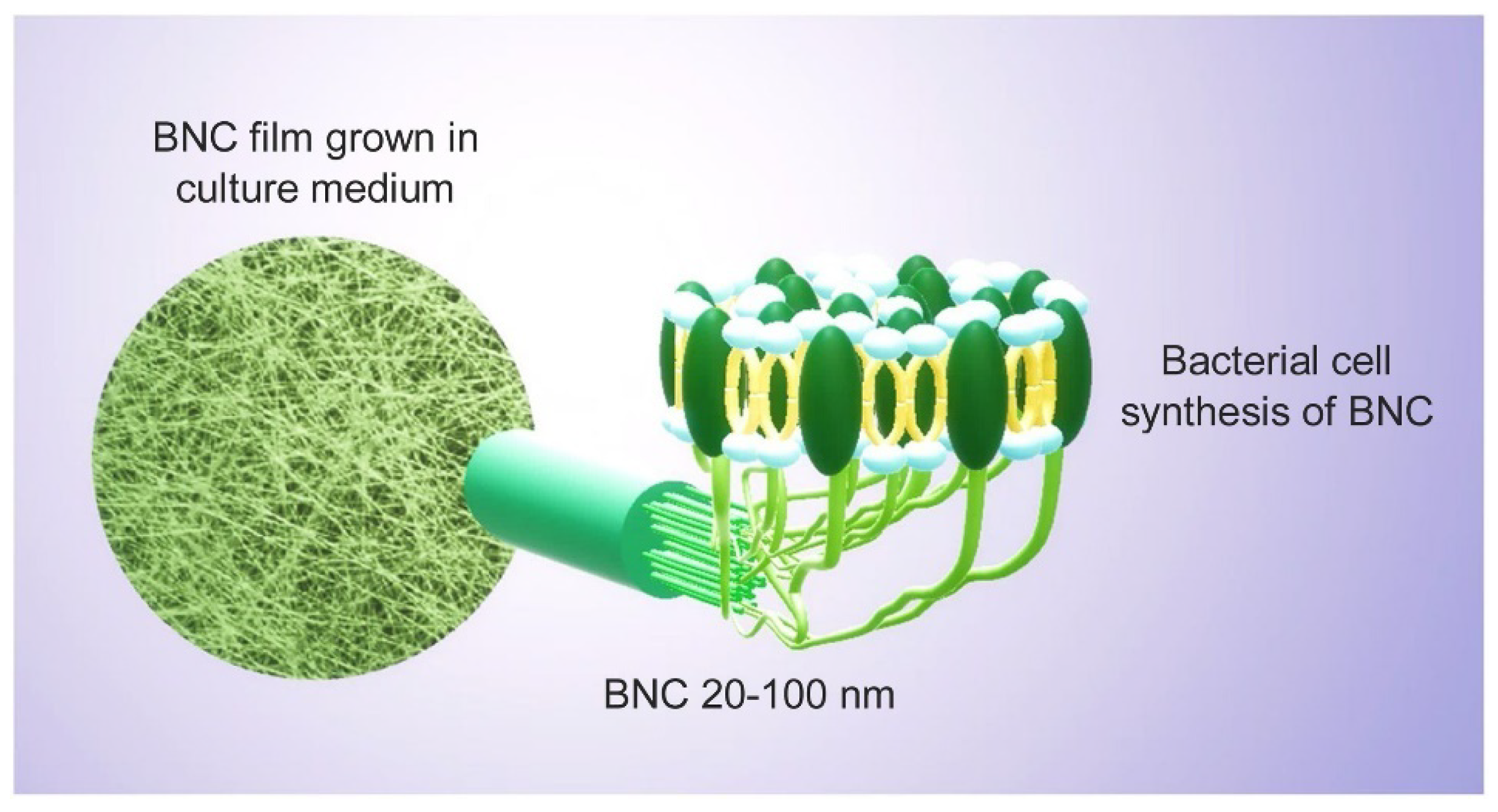
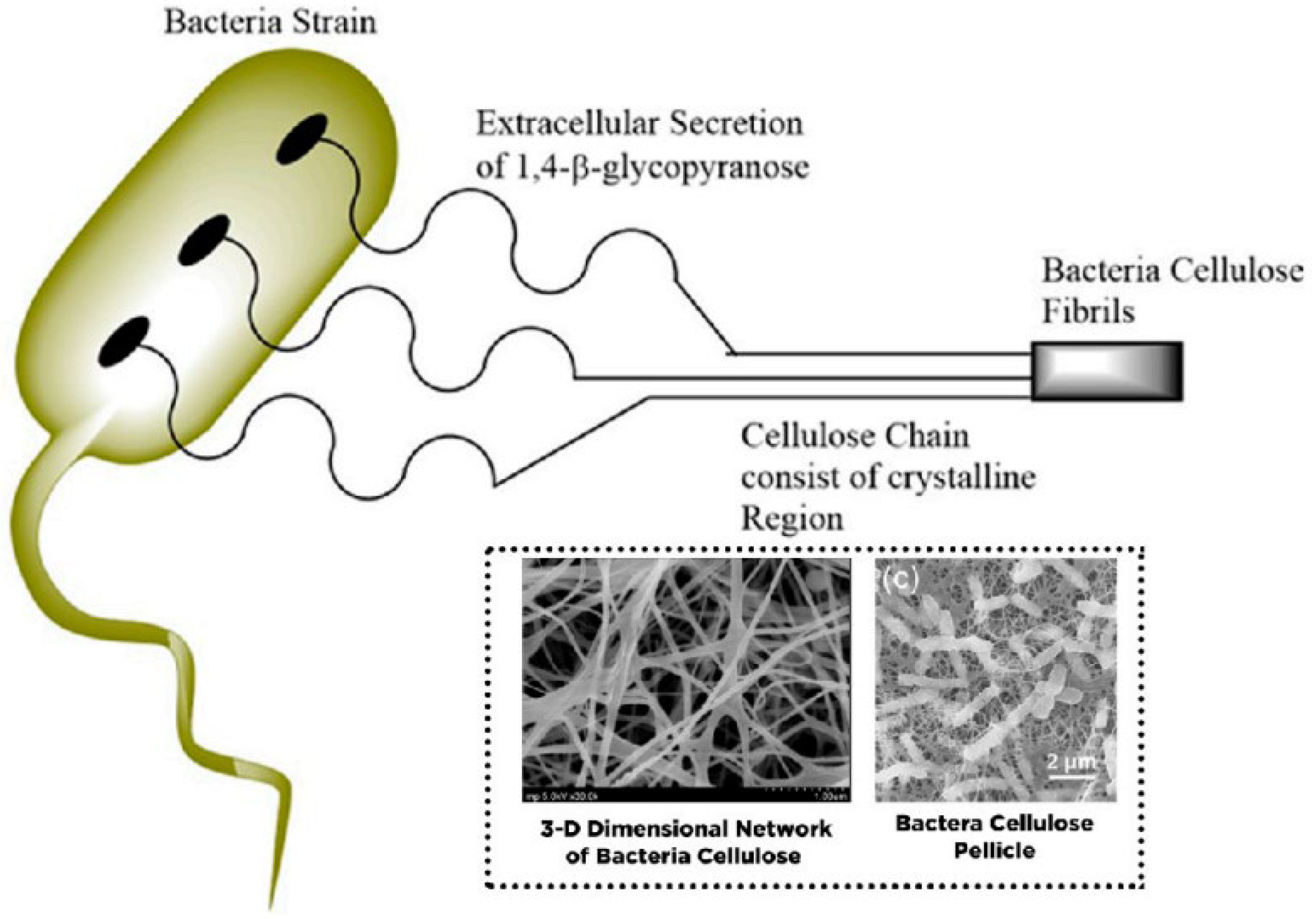
7. Bacterial Nano Cellulose (BNC): Structure, Property, Wastewater, and Desalination Application
7.1. Membrane Distillation
7.2. Ultrafiltration
7.3. Other Application
8. Future Perspectives
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Saleem, H.; Zaidi, S.J. Nanoparticles in Reverse Osmosis Membranes for Desalination: A State of the Art Review. Desalination 2020, 475, 114171. [Google Scholar] [CrossRef]
- Pendergast, M.M.; Hoek, E.M.V. A Review of Water Treatment Membrane Nanotechnologies. Energy Environ. Sci. 2011, 4, 1946–1971. [Google Scholar] [CrossRef]
- Kartal, B.; Koleva, M.; Arsov, R.; van der Star, W.; Jetten, M.S.; Strous, M. Adaptation of a freshwater anammox population to high salinity wastewater. J. Biotechnol. 2006, 126, 546–553. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Kuroda, M.; Nakatani, Y.; Soda, S.; Ike, M. Removal of selenite from artificial wastewater with high salinity by activated sludge in aerobic sequencing batch reactors. J. Biosci. Bioeng. 2019, 127, 618–624. [Google Scholar] [CrossRef]
- Liang, Y.; Zhu, H.; Bañuelos, G.; Yan, B.; Zhou, Q.; Yu, X.; Cheng, X. Constructed wetlands for saline wastewater treatment: A review. Ecol. Eng. 2017, 98, 275–285. [Google Scholar] [CrossRef]
- Pernetti, M.; Di Palma, L. Experimental Evaluation of Inhibition Effects of Saline Wastewater on Activated Sludge. Environ. Technol. 2005, 26, 695–704. [Google Scholar] [CrossRef]
- Vineis, P.; Chan, Q.; Khan, A. Climate change impacts on water salinity and health. J. Epidemiol. Glob. Health 2011, 1, 5–10. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhuang, X.; Ahmad, S.; Sung, S.; Ni, S.-Q. Biotreatment of high-salinity wastewater: Current methods and future directions. World J. Microbiol. Biotechnol. 2020, 36, 1–11. [Google Scholar] [CrossRef]
- Ai, L.; Su, J.; Wang, M.; Jiang, J. Bamboo-Structured Nitrogen-Doped Carbon Nanotube Coencapsulating Cobalt and Molybdenum Carbide Nanoparticles: An Efficient Bifunctional Electrocatalyst for Overall Water Splitting. ACS Sustain. Chem. Eng. 2018, 6, 9912–9920. [Google Scholar] [CrossRef]
- Xie, B.; Shan, C.; Xu, Z.; Li, X.; Zhang, X.; Chen, J.; Pan, B. One-step removal of Cr(VI) at alkaline pH by UV/sulfite process: Reduction to Cr(III) and in situ Cr(III) precipitation. Chem. Eng. J. 2017, 308, 791–797. [Google Scholar] [CrossRef]
- Bhadra, M.; Mitra, S. Advances in Nanostructured Membranes for Water Desalination. In Nanotechnology Applications for Clean Water: Solutions for Improving Water Quality, 2nd ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2014. [Google Scholar] [CrossRef]
- Charcosset, C. A review of membrane processes and renewable energies for desalination. Desalination 2009, 245, 214–231. [Google Scholar] [CrossRef]
- Othman, N.H.; Alias, N.H.; Fuzil, N.S.; Marpani, F.; Shahruddin, M.Z.; Chew, C.M.; Ng, K.M.D.; Lau, W.J.; Ismail, A.F. A Review on the Use of Membrane Technology Systems in Developing Countries. Membranes 2021, 12, 30. [Google Scholar] [CrossRef] [PubMed]
- Aliyu, U.M.; Rathilal, S.; Isa, Y.M. Membrane desalination technologies in water treatment: A review. Water Pr. Technol. 2018, 13, 738–752. [Google Scholar] [CrossRef]
- Choi, S.Y. A Review About Membrane-Based Water Desalination. Acton Sch. 2020, 1–8. Available online: https://www.actonscholars.org/how-can-differential-privacy-helps-deep-learning/ (accessed on 29 March 2022).
- El-Zanati, E.; El-Khatib, K.M. Integrated Membrane-Based Desalination System. Desalination 2007, 205, 15–25. [Google Scholar] [CrossRef]
- Saleem, H.; Zaidi, S.J. Innovative Nanostructured Membranes for Reverse Osmosis Water Desalination. In University of the Future: Re-Imagining Research and Higher Education; Qatar University Press: Doha, Qatar, 2020. [Google Scholar]
- Saleem, H.; Trabzon, L.; Kilic, A.; Zaidi, S.J. Recent advances in nanofibrous membranes: Production and applications in water treatment and desalination. Desalination 2020, 478, 114178. [Google Scholar] [CrossRef]
- Doornbusch, G.; Bel, M.; Tedesco, M.; Post, J.; Borneman, Z.; Nijmeijer, K. Effect of membrane area and membrane properties in multistage electrodialysis on seawater desalination performance. J. Membr. Sci. 2020, 611, 118303. [Google Scholar] [CrossRef]
- Sagle, A.; Freeman, B. Fundamentals of Membranes for Water Treatment. Futur. Desalin. Texas 2004, 2, 137. [Google Scholar]
- Schon, G. Auger and direct electron spectra in X-ray photoelectron studies of zinc, zinc oxide, gallium and gallium oxide. J. Electron Spectrosc. Relat. Phenom. 1973, 2, 75–86. [Google Scholar] [CrossRef]
- Xie, M.; Nghiem, L.D.; Price, W.E.; Elimelech, M. Relating rejection of trace organic contaminants to membrane properties in forward osmosis: Measurements, modelling and implications. Water Res. 2014, 49, 265–274. [Google Scholar] [CrossRef]
- Amiralian, N.; Mustapic, M.; Hossain, M.S.A.; Wang, C.; Konarova, M.; Tang, J.; Na, J.; Khan, A.; Rowan, A. Magnetic Nanocellulose: A Potential Material for Removal of Dye from Water. J. Hazard. Mater. 2020, 394, 122571. [Google Scholar] [CrossRef] [PubMed]
- Johnston, J.; Lou, J.; Tilton, N. Application of Projection Methods to Simulating Mass Transport in Reverse Osmosis Systems. Comput. Fluids 2022, 232, 105189. [Google Scholar] [CrossRef]
- Khan, B.; Haider, S.; Khurram, R.; Wang, Z.; Wang, X. Preparation of an Ultrafiltration (UF) Membrane with Narrow and Uniform Pore Size Distribution via Etching of SiO2 Nano-Particles in a Membrane Matrix. Membranes 2020, 10, 150. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Bian, L.; Bi, Q.; Li, Q.; Wang, X. Evaluation of the Pore Size Distribution of a Forward Osmosis Membrane in Three Different Ways. J. Memb. Sci. 2014, 454, 390–397. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, N.; Shi, J.; Xia, Y.; Zhu, B.; Shao, R.; Min, C.; Xu, Z.; Deng, H. Extra-Thin Composite Nanofiltration Membranes Tuned by γ-Cyclodextrins Containing Amphipathic Cavities for Efficient Separation of Magnesium/Lithium Ions. Sep. Purif. Technol. 2022, 286, 120419. [Google Scholar] [CrossRef]
- Li, T.; Wang, Y.; Wang, X.; Cheng, C.; Zhang, K.; Yang, J.; Han, G.; Wang, Z.; Wang, X.; Wang, L. Desalination Characteristics of Cellulose Acetate FO Membrane Incorporated with ZIF-8 Nanoparticles. Membranes 2022, 12, 122. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Thampi, S.P.; Suggala, S.V.; Bhattacharya, P.K. Pervaporation from a Dense Membrane: Roles of Permeant-Membrane Interactions, Kelvin Effect, and Membrane Swelling. Langmuir 2004, 20, 4708–4714. [Google Scholar] [CrossRef] [PubMed]
- Reza Shirzad Kebria, M.; Rahimpour, A. Membrane Distillation: Basics, Advances, and Applications. Adv. Membr. Technol. 2020, 1–21. [Google Scholar] [CrossRef]
- Rajaeian, B.; Rahimpour, A.; Tade, M.O.; Liu, S. Fabrication and Characterization of Polyamide Thin Film Nanocomposite (TFN) Nanofiltration Membrane Impregnated with TiO2 Nanoparticles. Desalination 2013, 313, 176–188. [Google Scholar] [CrossRef]
- Lau, W.J.; Ismail, A.F.; Misdan, N.; Kassim, M.A. A Recent Progress in Thin Film Composite Membrane: A Review. Desalination 2012, 287, 190–199. [Google Scholar] [CrossRef]
- Zarrabi, H.; Yekavalangi, M.E.; Vatanpour, V.; Shockravi, A.; Safarpour, M. Improvement in Desalination Performance of Thin Film Nanocomposite Nanofiltration Membrane Using Amine-Functionalized Multiwalled Carbon Nanotube. Desalination 2016, 394, 83–90. [Google Scholar] [CrossRef]
- Kebria, M.R.S.; Jahanshahi, M.; Rahimpour, A. SiO2 Modified Polyethyleneimine-Based Nanofiltration Membranes for Dye Removal from Aqueous and Organic Solutions. Desalination 2015, 367, 255–264. [Google Scholar] [CrossRef]
- Liu, S.; Zeng, T.H.; Hofmann, M.; Burcombe, E.; Wei, J.; Jiang, R.; Kong, J.; Chen, Y. Antibacterial Activity of Graphite, Graphite Oxide, Graphene Oxide, and Reduced Graphene Oxide: Membrane and Oxidative Stress. ACS Nano 2011, 5, 6971–6980. [Google Scholar] [CrossRef] [PubMed]
- Saleem, H.; Zaidi, S.J. Developments in the Application of Nanomaterials for Water Treatment and Their Impact on the Environment. Nanomaterials 2020, 10, 1764. [Google Scholar] [CrossRef] [PubMed]
- Saleem, H.; Zaidi, S.J.; Ismail, A.F.; Goh, P.S. Advances of Nanomaterials for Air Pollution Remediation and Their Impacts on the Environment. Chemosphere 2022, 287, 132083. [Google Scholar] [CrossRef] [PubMed]
- Saleem, H.; Zaidi, S.J. Sustainable Use of Nanomaterials in Textiles and Their Environmental Impact. Materials 2020, 13, 5134. [Google Scholar] [CrossRef] [PubMed]
- Saleem, H.; Zaidi, S.J. Recent Developments in the Application of Nanomaterials in Agroecosystems. Nanomaterials 2020, 10, 2411. [Google Scholar] [CrossRef]
- Tan, H.F.; Ooi, B.S.; Leo, C.P. Future Perspectives of Nanocellulose-Based Membrane for Water Treatment. J. Water Process Eng. 2020, 37, 101502. [Google Scholar] [CrossRef]
- George, J.; Sabapathi, S.N. Cellulose Nanocrystals: Synthesis, Functional Properties, and Applications. Nanotechnol. Sci. Appl. 2015, 8, 45–54. [Google Scholar] [CrossRef]
- Abol-Fotouh, D.; Hassan, M.A.; Shokry, H.; Roig, A.; Azab, M.S.; Kashyout, A.E.H.B. Bacterial Nanocellulose from Agro-Industrial Wastes: Low-Cost and Enhanced Production by Komagataeibacter Saccharivorans MD1. Sci. Rep. 2020, 10, 1–14. [Google Scholar] [CrossRef]
- Muhamad, I.I.; Pa’e, N.; Yusof, A.H.M. Bacterial Nanocellulose and Its Application in Wastewater Treatment. Sustain. Nanocellulose Nanohydrogels Nat. Sources 2020, 299–314. [Google Scholar] [CrossRef]
- Carter, N.; Grant, I.; Dewey, M.; Bourque, M.; Neivandt, D.J. Production and Characterization of Cellulose Nanofiber Slurries and Sheets for Biomedical Applications. Front. Nanotechnol. 2021, 3, 1–11. [Google Scholar] [CrossRef]
- Van Goethem, C.; Verbeke, R.; Pfanmöller, M.; Koschine, T.; Dickmann, M.; Timpel-Lindner, T.; Egger, W.; Bals, S.; Vankelecom, I.F.J. The Role of MOFs in Thin-Film Nanocomposite (TFN) Membranes. J. Memb. Sci. 2018, 563, 938–948. [Google Scholar] [CrossRef]
- Mavukkandy, M.O.; McBride, S.A.; Warsinger, D.M.; Dizge, N.; Hasan, S.W.; Arafat, H.A. Thin Film Deposition Techniques for Polymeric Membranes—A Review. J. Memb. Sci. 2020, 610, 118258. [Google Scholar] [CrossRef]
- Moon, R.J.; Martini, A.; Nairn, J.; Simonsen, J.; Youngblood, J. Cellulose Nanomaterials Review Structure, Properties and Nanocomposites. Chem. Soc. Rev. 2011, 40, 3941–3994. [Google Scholar] [CrossRef] [PubMed]
- Ambika Gopakumar, D. Nanocellulose Based Functional Constructs for Clean Water and Microwave Suppression. Ph.D. Thesis, Université de Bretagne Sud, Lorient, France, 20 November 2017. [Google Scholar]
- Espíndola, S.P.; Pronk, M.; Zlopasa, J.; Picken, S.J.; van Loosdrecht, M.C.M. Nanocellulose Recovery from Domestic Wastewater. J. Clean. Prod. 2021, 280, 124507. [Google Scholar] [CrossRef]
- Jamshaid, A.; Hamid, A.; Muhammad, N.; Naseer, A.; Ghauri, M.; Iqbal, J.; Rafiq, S.; Shah, N.S. Cellulose-Based Materials for the Removal of Heavy Metals from Wastewater—An Overview. ChemBioEng Rev. 2017, 4, 240–256. [Google Scholar] [CrossRef]
- Reshmy, R.; Philip, E.; Paul, S.A.; Madhavan, A.; Sindhu, R.; Binod, P.; Pandey, A.; Sirohi, R. Nanocellulose-Based Products for Sustainable Applications-Recent Trends and Possibilities. Rev. Environ. Sci. Biotechnol. 2020, 19, 779–806. [Google Scholar] [CrossRef]
- Shen, J.; Xu, X.; Mi, O. Adsorption of Pb (II) from Aqueous Solutions Using Nanocrystalline Cellulose/Sodium Alginate/K-Carrageenan Composite Hydrogel Beads. J. Polym. Environ. 2022, 30, 1995–2006. [Google Scholar] [CrossRef]
- Zhu, X.; Tong, J.; Lan, H.; Pan, D. Fabrication of Polyethyleneimine-Functionalized Magnetic Cellulose Nanocrystals for the Adsorption of Diclofenac Sodium from Aqueous Solutions. Polymers 2022, 14, 720. [Google Scholar] [CrossRef]
- Peng, B.; Yao, Z.; Wang, X.; Crombeen, M.; Sweeney, D.G.; Tam, K.C. Cellulose-Based Materials in Wastewater Treatment of Petroleum Industry. Green Energy Environ. 2020, 5, 37–49. [Google Scholar] [CrossRef]
- Zhao, X.Q.; Wahid, F.; Cui, J.X.; Wang, Y.Y.; Zhong, C. Cellulose-Based Special Wetting Materials for Oil/Water Separation: A Review. Int. J. Biol. Macromol. 2021, 185, 890–906. [Google Scholar] [CrossRef] [PubMed]
- Akter, M.; Bhattacharjee, M.; Dhar, A.K.; Rahman, F.B.A.; Haque, S.; Ur Rashid, T.U.; Kabir, S.M.F. Cellulose-Based Hydrogels for Wastewater Treatment: A Concise Review. Gels 2021, 7, 30. [Google Scholar] [CrossRef] [PubMed]
- Klemm, D.; Heublein, B.; Fink, H.P.; Bohn, A. Cellulose: Fascinating Biopolymer and Sustainable Raw Material. Angew. Chem. Int. Ed. 2005, 44, 3358–3393. [Google Scholar] [CrossRef] [PubMed]
- Sadare, O.O.; Yoro, K.O.; Moothi, K.; Daramola, M.O. Lignocellulosic Biomass-Derived Nanocellulose Crystals as Fillers in Membranes for Water and Wastewater Treatment: A Review. Membranes 2022, 12, 320. [Google Scholar] [CrossRef]
- Chen, X.; Yuan, F.; Zhang, H.; Huang, Y.; Yang, J.; Sun, D. Recent Approaches and Future Prospects of Bacterial Cellulose-Based Electroconductive Materials. J. Mater. Sci. 2016, 51, 5573–5588. [Google Scholar] [CrossRef]
- Wang, Z.; Ma, H.; Chu, B.; Hsiao, B.S. Fabrication of Cellulose Nanofiber-Based Ultrafiltration Membranes by Spray Coating Approach. J. Appl. Polym. Sci. 2017, 134, 1–6. [Google Scholar] [CrossRef]
- Carpenter, A.W.; De Lannoy, C.F.; Wiesner, M.R. Cellulose Nanomaterials in Water Treatment Technologies. Environ. Sci. Technol. 2015, 49, 5277–5287. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, H.; Shen, Z. Nanocellulose Based Filtration Membrane in Industrial Waste Water Treatment: A Review. Materials 2021, 14, 5398. [Google Scholar] [CrossRef]
- Dhali, K.; Ghasemlou, M.; Daver, F.; Cass, P.; Adhikari, B. A Review of Nanocellulose as a New Material towards Environmental Sustainability. Sci. Total Environ. 2021, 775, 145871. [Google Scholar] [CrossRef]
- Lin, N.; Dufresne, A. Nanocellulose in Biomedicine: Current Status and Future Prospect. Eur. Polym. J. 2014, 59, 302–325. [Google Scholar] [CrossRef]
- Gao, Z.; Peng, J.; Zhong, T.; Sun, J.; Wang, X.; Yue, C. Biocompatible Elastomer of Waterborne Polyurethane Based on Castor Oil and Polyethylene Glycol with Cellulose Nanocrystals. Carbohydr. Polym. 2012, 87, 2068–2075. [Google Scholar] [CrossRef]
- Thomas, B.; Raj, M.C.; Athira, B.K.; Rubiyah, H.M.; Joy, J.; Moores, A.; Drisko, G.L.; Sanchez, C. Nanocellulose, a Versatile Green Platform: From Biosources to Materials and Their Applications. Chem. Rev. 2018, 118, 11575–11625. [Google Scholar] [CrossRef] [PubMed]
- Mahfoudhi, N.; Boufi, S. Nanocellulose as a Novel Nanostructured Adsorbent for Environmental Remediation: A Review. Cellulose 2017, 24, 1171–1197. [Google Scholar] [CrossRef]
- Reshmy, R.; Thomas, D.; Philip, E.; Paul, S.A.; Madhavan, A.; Sindhu, R.; Binod, P.; Pugazhendhi, A.; Sirohi, R.; Tarafdar, A.; et al. Potential of Nanocellulose for Wastewater Treatment. Chemosphere 2021, 281, 130738. [Google Scholar] [CrossRef]
- Yuan, B.; Li, L.; Murugadoss, V.; Vupputuri, S.; Wang, J.; Alikhani, N.; Guo, Z. Nanocellulose-Based Composite Materials for Wastewater Treatment and Waste-Oil Remediation. ES Food Agrofor. 2020, 1, 41–52. [Google Scholar] [CrossRef]
- Mbakop, S.; Nthunya, L.N.; Onyango, M.S. Recent Advances in the Synthesis of Nanocellulose Functionalized-Hybrid Membranes and Application in Water Quality Improvement. Processes 2021, 9, 611. [Google Scholar] [CrossRef]
- Mazlan, N.M.; Marchetti, P.; Maples, H.A.; Gu, B.; Karan, S.; Bismarck, A.; Livingston, A.G. Organic Fouling Behaviour of Structurally and Chemically Different Forward Osmosis Membranes—A Study of Cellulose Triacetate and Thin Film Composite Membranes. J. Memb. Sci. 2016, 520, 247–261. [Google Scholar] [CrossRef]
- Yang, Z.; Peng, H.; Wang, W.; Liu, T. Crystallization Behavior of Poly(ε-Caprolactone)/Layered Double Hydroxide Nanocomposites. J. Appl. Polym. Sci. 2010, 116, 2658–2667. [Google Scholar] [CrossRef]
- Cheng, Q.; Ye, D.; Chang, C.; Zhang, L. Facile Fabrication of Superhydrophilic Membranes Consisted of Fibrous Tunicate Cellulose Nanocrystals for Highly Efficient Oil/Water Separation. J. Memb. Sci. 2017, 525, 1–8. [Google Scholar] [CrossRef]
- Nechyporchuk, O.; Belgacem, M.N.; Bras, J. Production of Cellulose Nanofibrils: A Review of Recent Advances. Ind. Crops Prod. 2016, 93, 2–25. [Google Scholar] [CrossRef]
- Park, C.W.; Han, S.Y.; Namgung, H.W.; Seo, P.N.; Lee, S.Y.; Lee, S.H. Preparation and Characterization of Cellulose Nanofibrils with Varying Chemical Compositions. BioResources 2017, 12, 5031–5044. [Google Scholar] [CrossRef]
- Fan, M.; Dai, D.; Yang, A. High Strength Natural Fiber Composite: Defibrillation and Its Mechanisms of Nano Cellulose Hemp Fibers. Int. J. Polym. Mater. Polym. Biomater. 2011, 60, 1026–1040. [Google Scholar] [CrossRef]
- Newman, R.H.; Hemmingson, J.A. Carbon-13 NMR Distinction between Categories of Molecular Order and Disorder in Cellulose. Cellulose 1995, 2, 95–110. [Google Scholar] [CrossRef]
- Domingues, R.M.A.; Gomes, M.E.; Reis, R.L. The Potential of Cellulose Nanocrystals in Tissue Engineering Strategies. Biomacromolecules 2014, 15, 2327–2346. [Google Scholar] [CrossRef] [PubMed]
- Habibi, Y.; Lucia, L.A.; Rojas, O.J. Cellulose nanocrystals: Chemistry, self-assembly, and applications. Chem. Rev. 2010, 110, 3479–3500. [Google Scholar] [CrossRef] [PubMed]
- Hardian, R.; Cywar, R.M.; Chen, E.Y.-X.; Szekely, G. Sustainable Nanofiltration Membranes Based on Biosourced Fully Recyclable Polyesters and Green Solvents. J. Membr. Sci. Lett. 2022, 2, 100016. [Google Scholar] [CrossRef]
- Waheed, A.; Baig, U.; Ansari, M.A. Fabrication of CuO Nanoparticles Immobilized Nanofiltration Composite Membrane for Dye/Salt Fractionation: Performance and Antibiofouling. J. Environ. Chem. Eng. 2022, 10, 106960. [Google Scholar] [CrossRef]
- Liu, S.; Low, Z.X.; Hegab, H.M.; Xie, Z.; Ou, R.; Yang, G.; Simon, G.P.; Zhang, X.; Zhang, L.; Wang, H. Enhancement of Desalination Performance of Thin-Film Nanocomposite Membrane by Cellulose Nanofibers. J. Memb. Sci. 2019, 592, 117363. [Google Scholar] [CrossRef]
- Wang, D.; Yuan, H.; Chen, Y.; Ni, Y.; Huang, L.; Mondal, A.K.; Lin, S.; Huang, F.; Zhang, H. A Cellulose-Based Nanofiltration Membrane with a Stable Three-Layer Structure for the Treatment of Drinking Water. Cellulose 2020, 27, 8237–8253. [Google Scholar] [CrossRef]
- Li, S.; Wang, D.; Xiao, H.; Zhang, H.; Cao, S.; Chen, L.; Ni, Y.; Huang, L. Ultra-Low Pressure Cellulose-Based Nanofiltration Membrane Fabricated on Layer-by-Layer Assembly for Efficient Sodium Chloride Removal. Carbohydr. Polym. 2021, 255, 117352. [Google Scholar] [CrossRef]
- Ewulonu, C.M.; Liu, X.; Wu, M.; Yong, H. Lignin-Containing Cellulose Nanomaterials: A Promising New Nanomaterial for Numerous Applications. J. Bioresour. Bioprod. 2019, 4, 3–10. [Google Scholar] [CrossRef]
- Liu, X.; Huang, K.; Lin, X.; Li, H.; Tao, T.; Wu, Q.; Zheng, Q.; Huang, L.; Ni, Y.; Chen, L.; et al. Transparent and Conductive Cellulose Film by Controllably Growing Aluminum Doped Zinc Oxide on Regenerated Cellulose Film. Cellulose 2020, 27, 4847–4855. [Google Scholar] [CrossRef]
- Fei, G.; Wang, Y.; Wang, H.; Ma, Y.; Guo, Q.; Huang, W.; Yang, D.; Shao, Y.; Ni, Y. Fabrication of Bacterial Cellulose/Polyaniline Nanocomposite Paper with Excellent Conductivity, Strength, and Flexibility. ACS Sustain. Chem. Eng. 2019, 7, 8215–8225. [Google Scholar] [CrossRef]
- Wang, J.J.; Yang, H.C.; Wu, M.B.; Zhang, X.; Xu, Z.K. Nanofiltration Membranes with Cellulose Nanocrystals as an Interlayer for Unprecedented Performance. J. Mater. Chem. A 2017, 5, 16289–16295. [Google Scholar] [CrossRef]
- Bai, L.; Liu, Y.; Ding, A.; Ren, N.; Li, G.; Liang, H. Fabrication and Characterization of Thin-Film Composite (TFC) Nanofiltration Membranes Incorporated with Cellulose Nanocrystals (CNCs) for Enhanced Desalination Performance and Dye Removal. Chem. Eng. J. 2019, 358, 1519–1528. [Google Scholar] [CrossRef]
- Xu, C.; Chen, W.; Gao, H.; Xie, X.; Chen, Y. Cellulose Nanocrystal/Silver (CNC/Ag) Thin-Film Nanocomposite Nanofiltration Membranes with Multifunctional Properties. Environ. Sci. Nano 2020, 7, 803–816. [Google Scholar] [CrossRef]
- Bai, L.; Liu, Y.; Bossa, N.; Ding, A.; Ren, N.; Li, G.; Liang, H.; Wiesner, M.R. Incorporation of Cellulose Nanocrystals (CNCs) into the Polyamide Layer of Thin-Film Composite (TFC) Nanofiltration Membranes for Enhanced Separation Performance and Antifouling Properties. Environ. Sci. Technol. 2018, 52, 11178–11187. [Google Scholar] [CrossRef]
- Bai, L.; Ding, J.; Wang, H.; Ren, N.; Li, G.; Liang, H. High-Performance Nanofiltration Membranes with a Sandwiched Layer and a Surface Layer for Desalination and Environmental Pollutant Removal. Sci. Total Environ. 2020, 743, 140766. [Google Scholar] [CrossRef]
- Yang, L.; Liu, X.; Zhang, X.; Chen, T.; Ye, Z.; Rahaman, M.S. High Performance Nanocomposite Nanofiltration Membranes with Polydopamine-Modified Cellulose Nanocrystals for Efficient Dye/Salt Separation. Desalination 2022, 521, 115385. [Google Scholar] [CrossRef]
- Vikman, M.; Vartiainen, J.; Tsitko, I.; Korhonen, P. Biodegradability and Compostability of Nanofibrillar Cellulose- Based Products. J. Polym. Environ. 2015, 23, 206–215. [Google Scholar] [CrossRef]
- Wenten, I.G. Khoiruddin Reverse Osmosis Applications: Prospect and Challenges. Desalination 2016, 391, 112–125. [Google Scholar] [CrossRef]
- Shalaby, S.M.; Sharshir, S.W.; Kabeel, A.E.; Kandeal, A.W.; Abosheiasha, H.F.; Abdelgaied, M.; Hamed, M.H.; Yang, N. Reverse Osmosis Desalination Systems Powered by Solar Energy: Preheating Techniques and Brine Disposal Challenges – A Detailed Review. Energy Convers. Manag. 2022, 251, 114971. [Google Scholar] [CrossRef]
- Honarparvar, S.; Zhang, X.; Chen, T.; Alborzi, A.; Afroz, K.; Reible, D. Frontiers of Membrane Desalination Processes for Brackish Water Treatment: A Review. Membranes 2021, 11, 246. [Google Scholar] [CrossRef]
- Aladwani, S.H.; Al-Obaidi, M.A.; Mujtaba, I.M. Performance of Reverse Osmosis Based Desalination Process Using Spiral Wound Membrane: Sensitivity Study of Operating Parameters under Variable Seawater Conditions. Clean. Eng. Technol. 2021, 5, 100284. [Google Scholar] [CrossRef]
- Liu, Y.; Bai, L.; Zhu, X.; Xu, D.; Li, G.; Liang, H.; Wiesner, M.R. The Role of Carboxylated Cellulose Nanocrystals Placement in the Performance of Thin-Film Composite (TFC) Membrane. J. Memb. Sci. 2021, 617, 118581. [Google Scholar] [CrossRef]
- Asempour, F.; Emadzadeh, D.; Matsuura, T.; Kruczek, B. Synthesis and Characterization of Novel Cellulose Nanocrystals-Based Thin Film Nanocomposite Membranes for Reverse Osmosis Applications. Desalination 2018, 439, 179–187. [Google Scholar] [CrossRef]
- Singh, R. Hybrid Membrane System Design and Operation. Hybrid Membr. Syst. Water Purif. 2005, 197–242. [Google Scholar] [CrossRef]
- Kang, G.; Cao, Y. Development of Antifouling Reverse Osmosis Membranes for Water Treatment: A Review. Water Res. 2012, 46, 584–600. [Google Scholar] [CrossRef]
- Park, C.H.; Jeon, S.; Park, S.H.; Shin, M.G.; Park, M.S.; Lee, S.Y.; Lee, J.H. Cellulose Nanocrystal-Assembled Reverse Osmosis Membranes with High Rejection Performance and Excellent Antifouling. J. Mater. Chem. A 2019, 7, 3992–4001. [Google Scholar] [CrossRef]
- Misdan, N.; Ismail, A.F.; Hilal, N. Recent Advances in the Development of (Bio)Fouling Resistant Thin Film Composite Membranes for Desalination. Desalination 2016, 380, 105–111. [Google Scholar] [CrossRef]
- Lu, X.; Arias Chavez, L.H.; Romero-Vargas Castrillón, S.; Ma, J.; Elimelech, M. Influence of Active Layer and Support Layer Surface Structures on Organic Fouling Propensity of Thin-Film Composite Forward Osmosis Membranes. Environ. Sci. Technol. 2015, 49, 1436–1444. [Google Scholar] [CrossRef]
- Ly, Q.V.; Hu, Y.; Li, J.; Cho, J.; Hur, J. Characteristics and Influencing Factors of Organic Fouling in Forward Osmosis Operation for Wastewater Applications: A Comprehensive Review. Environ. Int. 2019, 129, 164–184. [Google Scholar] [CrossRef] [PubMed]
- Akhator, P.; Obanor, A.; Ugege, A. Nigerian Wood Waste: A Potential Resource for Economic Development. J. Appl. Sci. Environ. Manag. 2017, 21, 246. [Google Scholar] [CrossRef]
- Adeniyi, A.; Gonzalez-Ortiz, D.; Pochat-Bohatier, C.; Oyewo, O.; Sithole, B.; Onyango, M. Incorporation of Cellulose Nanocrystals (CNC) Derived from Sawdust into Polyamide Thin-Film Composite Membranes for Enhanced Water Recovery. Alex. Eng. J. 2020, 59, 4201–4210. [Google Scholar] [CrossRef]
- Smith, E.D.; Hendren, K.D.; Haag, J.V.; Foster, E.J.; Martin, S.M. Functionalized Cellulose Nanocrystal Nanocomposite Membranes with Controlled Interfacial Transport for Improved Reverse Osmosis Performance. Nanomaterials 2019, 9, 125. [Google Scholar] [CrossRef]
- Hayakawa, D.; Nishiyama, Y.; Mazeau, K.; Ueda, K. Evaluation of Hydrogen Bond Networks in Cellulose Iβ and II Crystals Using Density Functional Theory and Car–Parrinello Molecular Dynamics. Carbohydr. Res. 2017, 449, 103–113. [Google Scholar] [CrossRef]
- Joy, J.; George, N.; Chirayil, C.J.; Wilson, R. Nanocellulose/Polymer Nanocomposite Membranes for Pervaporation Application; INC: New York, NY, USA, 2020; ISBN 9780128167854. [Google Scholar]
- Golubeva, O.Y.; Yudin, V.E.; Didenko, A.L.; Svetlichnyi, V.M.; Gusarov, V.V. Nanocomposites Based on Polyimide Thermoplastics and Magnesium Silicate Nanoparticles with Montmorillonite Structure. Russ. J. Appl. Chem. 2007, 80, 106–109. [Google Scholar] [CrossRef]
- Prihatiningtyas, I.; Volodin, A.; Van Der Bruggen, B. 110th Anniversary: Cellulose Nanocrystals as Organic Nanofillers for Cellulose Triacetate Membranes Used for Desalination by Pervaporation. Ind. Eng. Chem. Res. 2019, 58, 14340–14349. [Google Scholar] [CrossRef]
- Gofman, I.V.; Svetlichnyi, V.M.; Yudin, V.E.; Dobrodumov, A.V.; Didenko, A.L.; Abalov, I.V.; Korytkova, E.N.; Egorov, A.I.; Gusarov, V.V. Modification of Films of Heat-Resistant Polyimides by Adding Hydrosilicate and Carbon Nanoparticles of Various Geometries. Russ. J. Gen. Chem. 2007, 77, 1158–1163. [Google Scholar] [CrossRef]
- Yudin, V.E.; Otaigbe, J.U.; Nazarenko, S.I.; Kim, W.D.; Korytkova, E.N. A Comparative Study on the Mechanical and Barrier Characteristics of Polyimide Nanocomposite Films Filled with Nanoparticles of Planar and Tubular Morphology. Mech. Compos. Mater. 2011, 47, 335–342. [Google Scholar] [CrossRef]
- Prihatiningtyas, I.; Li, Y.; Hartanto, Y.; Vananroye, A.; Coenen, N.; Van der Bruggen, B. Effect of Solvent on the Morphology and Performance of Cellulose Triacetate Membrane/Cellulose Nanocrystal Nanocomposite Pervaporation Desalination Membranes. Chem. Eng. J. 2020, 388, 124216. [Google Scholar] [CrossRef]
- Kamtsikakis, A.; Delepierre, G.; Weder, C. Cellulose Nanocrystals as a Tunable Nanomaterial for Pervaporation Membranes with Asymmetric Transport Properties. J. Memb. Sci. 2021, 635, 119473. [Google Scholar] [CrossRef]
- Prihatiningtyas, I.; Hartanto, Y.; Van der Bruggen, B. Ultra-High Flux Alkali-Treated Cellulose Triacetate/Cellulose Nanocrystal Nanocomposite Membrane for Pervaporation Desalination. Chem. Eng. Sci. 2021, 231, 116276. [Google Scholar] [CrossRef]
- Rahimi-Kashkouli, Y.; Rahbari-Sisakht, M.; Ghadami Jadval Ghadam, A. Thin Film Nanocomposite Nanofiltration Membrane Incorporated with Cellulose Nanocrystals with Superior Anti-Organic Fouling Affinity. Environ. Sci. Water Res. Technol. 2020, 6, 715–723. [Google Scholar] [CrossRef]
- Rezaei-DashtArzhandi, M.; Sarrafzadeh, M.H.; Goh, P.S.; Lau, W.J.; Ismail, A.F.; Wong, K.C.; Mohamed, M.A. Enhancing the Desalination Performance of Forward Osmosis Membrane through the Incorporation of Green Nanocrystalline Cellulose and Halloysite Dual Nanofillers. J. Chem. Technol. Biotechnol. 2020, 95, 2359–2370. [Google Scholar] [CrossRef]
- Rafieian, F.; Jonoobi, M.; Yu, Q. A Novel Nanocomposite Membrane Containing Modified Cellulose Nanocrystals for Copper Ion Removal and Dye Adsorption from Water. Cellulose 2019, 26, 3359–3373. [Google Scholar] [CrossRef]
- Liang, L.; Zhang, S.; Goenaga, G.A.; Meng, X.; Zawodzinski, T.A.; Ragauskas, A.J. Chemically Cross-Linked Cellulose Nanocrystal Aerogels for Effective Removal of Cation Dye. Front. Chem. 2020, 8, 1–9. [Google Scholar] [CrossRef]
- Lalia, B.S.; Guillen, E.; Arafat, H.A.; Hashaikeh, R. Nanocrystalline Cellulose Reinforced PVDF-HFP Membranes for Membrane Distillation Application. Desalination 2014, 332, 134–141. [Google Scholar] [CrossRef]
- Lv, J.; Zhang, G.; Zhang, H.; Yang, F. Graphene Oxide-Cellulose Nanocrystal (GO-CNC) Composite Functionalized PVDF Membrane with Improved Antifouling Performance in MBR: Behavior and Mechanism. Chem. Eng. J. 2018, 352, 765–773. [Google Scholar] [CrossRef]
- Suman; Kardam, A.; Gera, M.; Jain, V.K. A Novel Reusable Nanocomposite for Complete Removal of Dyes, Heavy Metals and Microbial Load from Water Based on Nanocellulose and Silver Nano-Embedded Pebbles. Environ. Technol. 2015, 36, 706–714. [Google Scholar] [CrossRef]
- Kocaman, C.; Bukusoglu, E.; Culfaz-Emecen, P.Z. Controlling Ultrafiltration Membrane Rejection via Shear-Aligned Deposition of Cellulose Nanocrystals from Aqueous Suspensions. ACS Appl. Mater. Interfaces 2021, 13, 36548–36557. [Google Scholar] [CrossRef] [PubMed]
- Moeinzadeh, R.; Emadzadeh, D. Petroleum Effluent Treatment Using Ultrafiltration Nanocomposite Membrane Made of Polysulfone and Cellulose Nanocrystal. J. Water Wastewater 2020, 31, 138–146. [Google Scholar] [CrossRef]
- Nishino, T.; Takano, K.; Nakamae, K. Elastic Modulus of the Crystalline Regions of Cellulose Polymorphs. J. Polym. Sci. Part B Polym. Phys. 1995, 33, 1647–1651. [Google Scholar] [CrossRef]
- Tsuzuki, T.; Zhang, L.; Rana, R.; Liu, Q.; Wang, X. Production of Green Nanomaterials. In Proceedings of the 2010 International Conference on Nanoscience and Nanotechnology, Sydney, NSW, Australia, 22–26 February 2010; 2010; pp. 150–153. [Google Scholar] [CrossRef]
- Syverud, K.; Stenius, P. Strength and Barrier Properties of MFC Films. Cellulose 2009, 16, 75–85. [Google Scholar] [CrossRef]
- Jonoobi, M.; Harun, J.; Mathew, A.P.; Oksman, K. Mechanical Properties of Cellulose Nanofiber (CNF) Reinforced Polylactic Acid (PLA) Prepared by Twin Screw Extrusion. Compos. Sci. Technol. 2010, 70, 1742–1747. [Google Scholar] [CrossRef]
- Frone, A.N.; Panaitescu, D.M.; Chiulan, I.; Nicolae, C.A.; Vuluga, Z.; Vitelaru, C.; Damian, C.M. The Effect of Cellulose Nanofibers on the Crystallinity and Nanostructure of Poly(Lactic Acid) Composites. J. Mater. Sci. 2016, 51, 9771–9791. [Google Scholar] [CrossRef]
- Kramar, A.; Javier, F. Cellulose-Based Nanofibers Processing Techniques and Methods Based on Bottom-Up Approach—A Review. Polymers 2022, 14, 286. [Google Scholar] [CrossRef]
- Cruz-Silva, R.; Izu, K.; Maeda, J.; Saito, S.; Morelos-Gomez, A.; Aguilar, C.; Takizawa, Y.; Yamanaka, A.; Tejiima, S.; Fujisawa, K.; et al. Nanocomposite Desalination Membranes Made of Aromatic Polyamide with Cellulose Nanofibers: Synthesis, Performance, and Water Diffusion Study. Nanoscale 2020, 12, 19628–19637. [Google Scholar] [CrossRef]
- Zhang, Q.G.; Deng, C.; Soyekwo, F.; Liu, Q.L.; Zhu, A.M. Sub-10 Nm Wide Cellulose Nanofibers for Ultrathin Nanoporous Membranes with High Organic Permeation. Adv. Funct. Mater. 2016, 26, 792–800. [Google Scholar] [CrossRef]
- Hassan, M.L.; Fadel, S.M.; Abouzeid, R.E.; Abou Elseoud, W.S.; Hassan, E.A.; Berglund, L.; Oksman, K. Water Purification Ultrafiltration Membranes Using Nanofibers from Unbleached and Bleached Rice Straw. Sci. Rep. 2020, 10, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Soyekwo, F.; Zhang, Q.; Gao, R.; Qu, Y.; Lin, C.; Huang, X.; Zhu, A.; Liu, Q. Cellulose Nanofiber Intermediary to Fabricate Highly-Permeable Ultrathin Nanofiltration Membranes for Fast Water Purification. J. Memb. Sci. 2017, 524, 174–185. [Google Scholar] [CrossRef]
- Fang, X.; Wei, S.; Liu, S.; Li, R.; Zhang, Z.; Liu, Y.; Zhang, X. Metal-Coordinated Nanofiltration Membranes Constructed on Metal Ions Blended Support toward Enhanced Dye/Salt Separation and Antifouling Performances. Membranes 2022, 12, 340. [Google Scholar] [CrossRef]
- Mohammed, S.; Hegab, H.M.; Ou, R.; Liu, S.; Ma, H.; Chen, X.; Sridhar, T.; Wang, H. Effect of Oxygen Plasma Treatment on the Nanofiltration Performance of Reduced Graphene Oxide/Cellulose Nanofiber Composite Membranes. Green Chem. Eng. 2021, 2, 122–131. [Google Scholar] [CrossRef]
- Ortiz-Medina, J.; Inukai, S.; Araki, T.; Morelos-Gomez, A.; Cruz-Silva, R.; Takeuchi, K.; Noguchi, T.; Kawaguchi, T.; Terrones, M.; Endo, M. Robust Water Desalination Membranes against Degradation Using High Loads of Carbon Nanotubes. Sci. Rep. 2018, 8, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Wang, Z.; Xu, J.; Wang, J.; Wang, S. Surface Modification of Commercial Aromatic Polyamide Reverse Osmosis Membranes by Crosslinking Treatments. Chin. J. Chem. Eng. 2013, 21, 473–484. [Google Scholar] [CrossRef]
- Hassan, M.; Hassan, E.; Fadel, S.M.; Abou-Zeid, R.E.; Berglund, L.; Oksman, K. Metallo-Terpyridine-Modified Cellulose Nanofiber Membranes for Papermaking Wastewater Purification. J. Inorg. Organomet. Polym. Mater. 2018, 28, 439–447. [Google Scholar] [CrossRef]
- Battirola, L.C.; Andrade, P.F.; Marson, G.V.; Hubinger, M.D.; Gonçalves, M.d.C. Cellulose Acetate/Cellulose Nanofiber Membranes for Whey and Fruit Juice Microfiltration. Cellulose 2017, 24, 5593–5604. [Google Scholar] [CrossRef]
- Asper, M.; Hanrieder, T.; Quellmalz, A.; Mihranyan, A. Removal of Xenotropic Murine Leukemia Virus by Nanocellulose Based Filter Paper. Biologicals 2015, 43, 452–456. [Google Scholar] [CrossRef]
- Ma, H.; Burger, C.; Hsiao, B.S.; Chu, B. Fabrication and Characterization of Cellulose Nanofiber Based Thin-Film Nanofibrous Composite Membranes. J. Memb. Sci. 2014, 454, 272–282. [Google Scholar] [CrossRef]
- Xiong, Y.; Wang, C.; Wang, H.; Jin, C.; Sun, Q.; Xu, X. Nano-Cellulose Hydrogel Coated Flexible Titanate-Bismuth Oxide Membrane for Trinity Synergistic Treatment of Super-Intricate Anion/Cation/Oily-Water. Chem. Eng. J. 2018, 337, 143–151. [Google Scholar] [CrossRef]
- Anokhina, T.S.; Bazhenov, S.D.; Borisov, I.L.; Vasilevsky, V.P.; Vinokurov, V.A.; Volkov, A.V. Nanocellulose as Modifier for Hollow Fiber Ultrafiltration PSF Membranes. Key Eng. Mater. 2019, 816, 238–243. [Google Scholar] [CrossRef]
- Torres, F.G.; Arroyo, J.J.; Troncoso, O.P. Bacterial Cellulose Nanocomposites: An All-Nano Type of Material. Mater. Sci. Eng. C 2019, 98, 1277–1293. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.; Li, J.; Bao, Z.; Hu, M.; Nian, R.; Feng, D.; An, D.; Li, X.; Xian, M.; Zhang, H. A Natural in Situ Fabrication Method of Functional Bacterial Cellulose Using a Microorganism. Nat. Commun. 2019, 10, 1–10. [Google Scholar] [CrossRef]
- Ullah, H.; Wahid, F.; Santos, H.A.; Khan, T. Advances in Biomedical and Pharmaceutical Applications of Functional Bacterial Cellulose-Based Nanocomposites. Carbohydr. Polym. 2016, 150, 330–352. [Google Scholar] [CrossRef]
- Junka, A.; Żywicka, A.; Chodaczek, G.; Dziadas, M.; Czajkowska, J.; Duda-Madej, A.; Bartoszewicz, M.; Mikołajewicz, K.; Krasowski, G.; Szymczyk, P.; et al. Potential of Biocellulose Carrier Impregnated with Essential Oils to Fight Against Biofilms Formed on Hydroxyapatite. Sci. Rep. 2019, 9, 1–13. [Google Scholar] [CrossRef]
- Roig-Sanchez, S.; Jungstedt, E.; Anton-Sales, I.; Malaspina, D.C.; Faraudo, J.; Berglund, L.A.; Laromaine, A.; Roig, A. Nanocellulose Films with Multiple Functional Nanoparticles in Confined Spatial Distribution. Nanoscale Horiz. 2019, 4, 634–641. [Google Scholar] [CrossRef]
- Li, D.; Tian, X.; Wang, Z.; Guan, Z.; Li, X.; Qiao, H.; Ke, H.; Luo, L.; Wei, Q. Multifunctional Adsorbent Based on Metal-Organic Framework Modified Bacterial Cellulose/Chitosan Composite Aerogel for High Efficient Removal of Heavy Metal Ion and Organic Pollutant. Chem. Eng. J. 2020, 383, 123127. [Google Scholar] [CrossRef]
- Lunardi, V.B.; Soetaredjo, F.E.; Putro, J.N.; Santoso, S.P.; Yuliana, M.; Sunarso, J.; Ju, Y.H.; Ismadji, S. Nanocelluloses: Sources, Pretreatment, Isolations, Modification, and Its Application as the Drug Carriers. Polymers 2021, 13, 2052. [Google Scholar] [CrossRef]
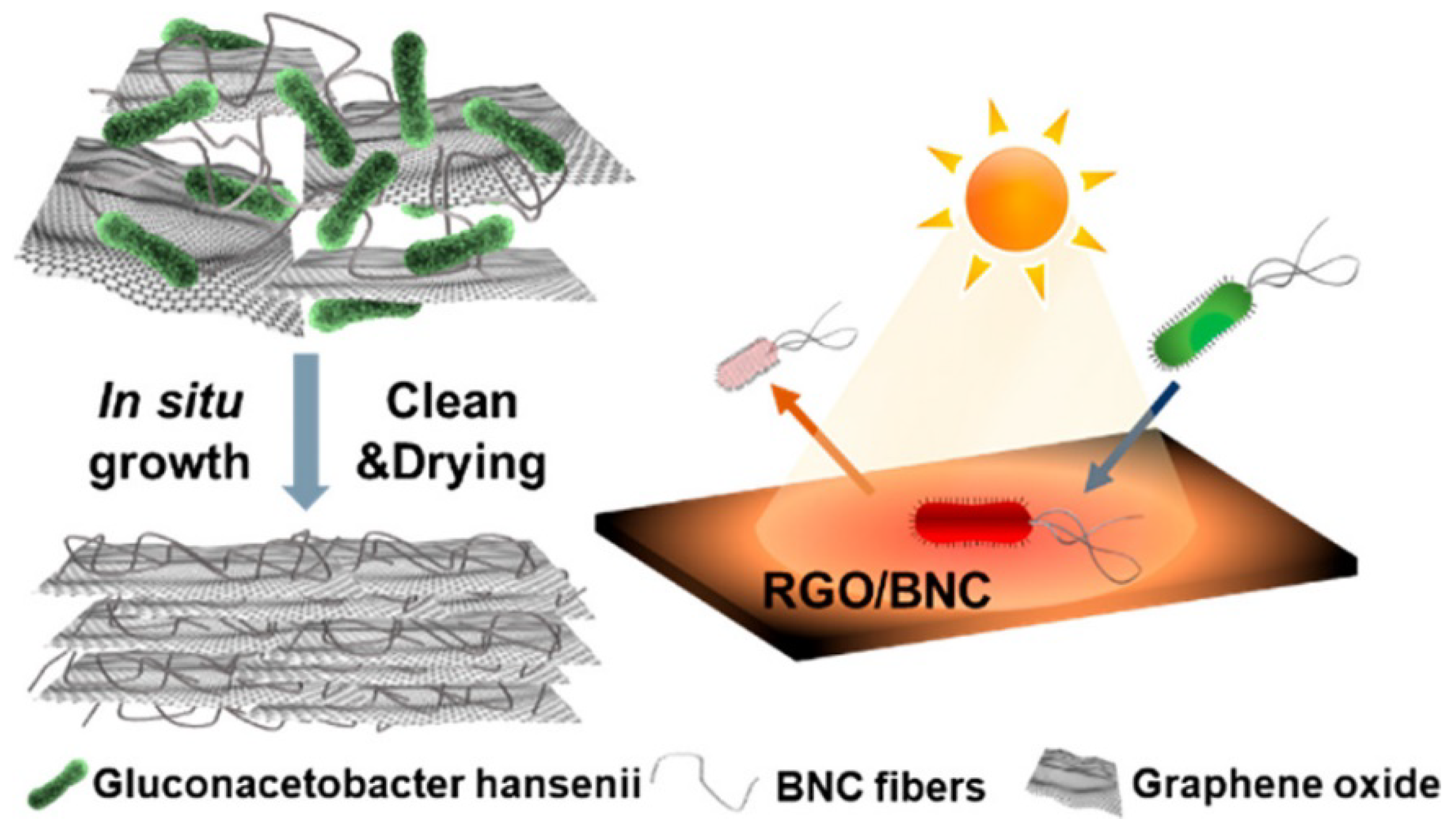
- Jiang, Q.; Ghim, D.; Cao, S.; Tadepalli, S.; Liu, K.K.; Kwon, H.; Luan, J.; Min, Y.; Jun, Y.S.; Singamaneni, S. Photothermally Active Reduced Graphene Oxide/Bacterial Nanocellulose Composites as Biofouling-Resistant Ultrafiltration Membranes. Environ. Sci. Technol. 2019, 53, 412–421. [Google Scholar] [CrossRef]
- Alkhudhiri, A.; Hilal, N. Membrane Distillation-Principles, Applications, Configurations, Design, and Implementation; Elsevier Inc.: Amsterdam, The Netherlands, 2018; ISBN 9780128167120. [Google Scholar]
- Souhaimi, M.K.; Matsuura, T. Membrane Distillation. Membr. Distill. 2011, 483–496. [Google Scholar] [CrossRef]
- Leitch, M.E.; Li, C.; Ikkala, O.; Mauter, M.S.; Lowry, G.V. Bacterial Nanocellulose Aerogel Membranes: Novel High-Porosity Materials for Membrane Distillation. Environ. Sci. Technol. Lett. 2016, 3, 85–91. [Google Scholar] [CrossRef]
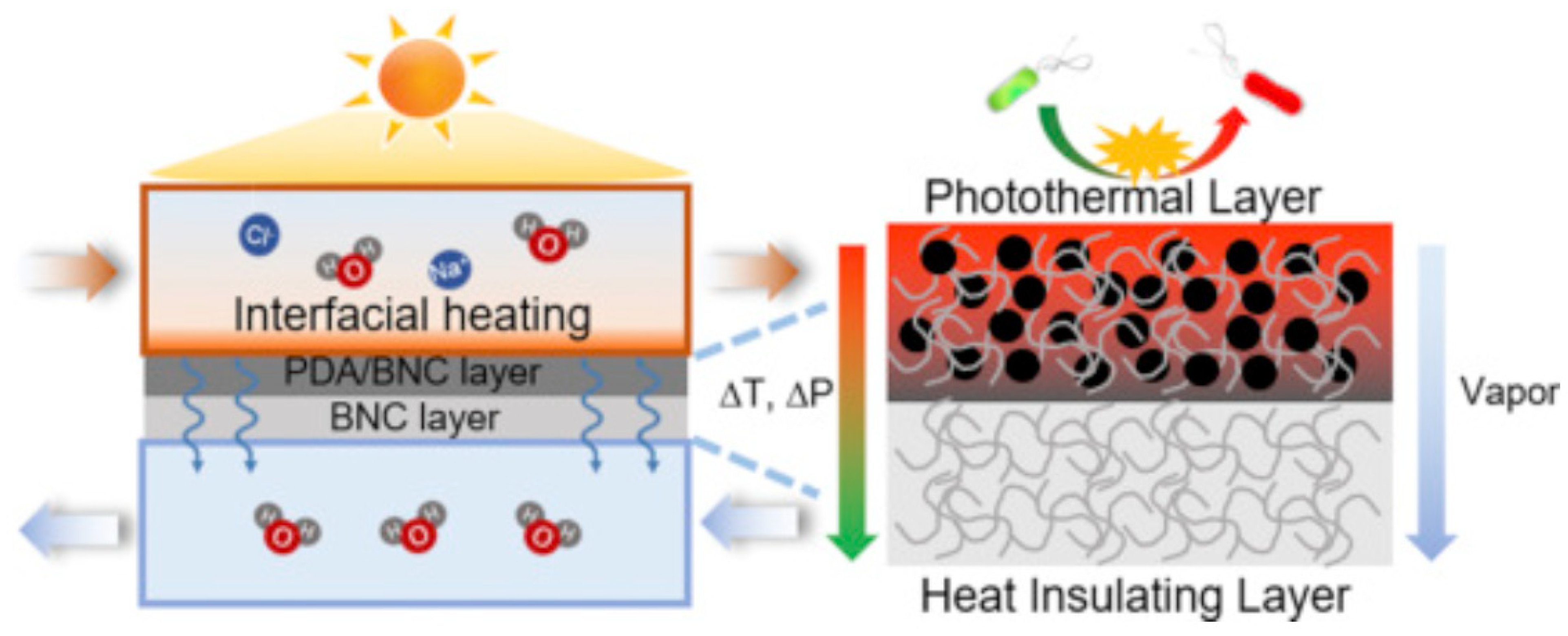
- Wu, X.; Cao, S.; Ghim, D.; Jiang, Q.; Singamaneni, S.; Jun, Y.S. A Thermally Engineered Polydopamine and Bacterial Nanocellulose Bilayer Membrane for Photothermal Membrane Distillation with Bactericidal Capability. Nano Energy 2021, 79, 105353. [Google Scholar] [CrossRef]
- Xu, T.; Jiang, Q.; Ghim, D.; Liu, K.K.; Sun, H.; Derami, H.G.; Wang, Z.; Tadepalli, S.; Jun, Y.S.; Zhang, Q.; et al. Catalytically Active Bacterial Nanocellulose-Based Ultrafiltration Membrane. Small 2018, 14, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Gholami Derami, H.; Jiang, Q.; Ghim, D.; Cao, S.; Chandar, Y.J.; Morrissey, J.J.; Jun, Y.S.; Singamaneni, S. A Robust and Scalable Polydopamine/Bacterial Nanocellulose Hybrid Membrane for Efficient Wastewater Treatment. ACS Appl. Nano Mater. 2019, 2, 1092–1101. [Google Scholar] [CrossRef]
- Sijabat, E.K.; Nuruddin, A.; Aditiawati, P.; Purwasasmita, B.S. Synthesis and Characterization of Bacterial Nanocellulose from Banana Peel for Water Filtration Membrane Application. J. Phys. Conf. Ser. 2019, 1230. [Google Scholar] [CrossRef]
- Amin Abdel-Fatah, M.; Ahmed Al Bazedi, G. Water Treatment and Desalination. Desalin. Challenges Oppor. 2020, 1–15. [Google Scholar] [CrossRef]
- Mautner, A.; Bismarck, A. Bacterial Nanocellulose Papers with High Porosity for Optimized Permeance and Rejection of Nm-Sized Pollutants. Carbohydr. Polym. 2021, 251, 117130. [Google Scholar] [CrossRef]
- Zhang, X.; Fang, Y.; Chen, W. Preparation of Silver/Bacterial Cellulose Composite Membrane and Study on Its Antimicrobial Activity. Synth. React. Inorg. Met. Nano-Met. Chem. 2013, 43, 907–913. [Google Scholar] [CrossRef]
- Jiang, Q. Multifunctional Nanocomposites Based on Bacterial Cellulose. Ph.D. Thesis, Washington University, St. Louis, MO, USA, 15 December 2018. [Google Scholar]





| Process | Reverse Osmosis | Forward Osmosis | Nanofiltration | Ultrafiltration | Pervaporation | Membrane Distillation |
|---|---|---|---|---|---|---|
| Membrane Pore diameter | <0.5 nm | 0.50–0.60 nm | <2.0 nm | 0.001 –0.05 mm | 0.2 to 0.6 nm | 100 Å and 1 μm. |
| Required pressure (bar) | High pressure | Mild pressure | High pressure | Low pressure | Low pressure | - |
| Cellulose Material | Cellulose Raw Material | Method of Synthesis | Application | Target | Foulant | Removal Efficiency | Reference |
|---|---|---|---|---|---|---|---|
| CNC incorporated membrane | Filter paper | Acidic hydrolysis | Osmotically derived FO | NaCl | BSA | Salt rejection: 89.79% | [120] |
| PES/CNC/APTES membrane | - | - | adsorption and ion removal | Cu ion and Red-16 | - | 90% Cu ion 99% Red-16 | [121] |
| CNCs/PMVEMA/PEG aerogel | Wood pulp | - | adsorption | cation methylene blue dye | - | 116.2 mg g−1 | [122] |
| CNC membrane | Kapok fiber | adsorption | Methyl blue | 85% for 5 mg/L MB | |||
| PVDF-HFP/CNC membrane | cellulose KimWipes | Acid Hydrolysis | MD | NaCl | - | Salt rejection 99% and Water flux 10.2–11.5 Lh−1 m−2 | [123] |
| GO-CNC/PVDF membrane | Hawp04700-Merck Millipore cellulose membrane | Acid Hydrolysis | Membrane bioreactor | Municipal waste water | BSA | 2.8 times less fouling | [124] |
| CNC/AgNPs pebble | Cellulose powder | Acid hydrolysis | Adsorption | Pb and Cr ion | 99.48% Pb(II) and 98.30% Cr(III) | [125] | |
| CNC/UF | Cotton linter | Hydrolysis | UF | vitamin B12, Blue Dextran, and β-lactoglobulin | 2.6, 4.1, and 26% | [126] | |
| PSF/CNC | - | Hydrolysis | UF | Petroleum effluent | Oil particles | 94.4% | [127] |
| Cellulose Material | Cellulose Raw Material | Method of Synthesis | Application | Target | Foulant | Removal Efficiency | Reference |
|---|---|---|---|---|---|---|---|
| CNF based filter paper | Cladophora sp. algae | - | Size-exclusion filtration | xenotrpic murine leukemia virus | - | 5.25log xMuLV | [144] |
| CNF/TFN | Wood pulp | TEMPO/NaBr/NaClO method | UF | oil/water emulsion | sodium alginate | 96.3% | [145] |
| Titanate-bismuth oxide/CNF | Bamboo pulp | TEMPO oxidation | Adsorption | Cs+ and I− ion | Industrial oil | is <80% | [146] |
| Fe3O4/CNF magnetic membrane | Sugarcane pulp | Hydrolysis | Catalytic dye removal | RhB | - | 94.9% | [23] |
| UF/PSF/CNF | - | - | UF | Blue dextran | 99% | [147] |
| Cellulose Material | Cellulose Raw Material | Method of Synthesis | Application | Target | Foulant | Removal Efficiency | Reference |
|---|---|---|---|---|---|---|---|
| BNC/Ag | BNC | - | Antimicrobial activity | E. coli and S. aureus | - | Inhibitory rate 99.4% and 98.4% | [165] |
| BNC based membrane | Banana Peel | Fermentation with Gluconacetobacter xylinus bacteria | Filtration | - | - | - | [162] |
| Pd/GO/Bacterial NC | Pd, Graphene oxide and BNC | Fermentation | Filtration | Gold Nanoparticles | 99.7% | [164] | |
| BNC based nanopaper | Bacterial Cellulose | - | UF | MO | 99.3% over different concentration of MO | [160] | |
| rGO/BNC | Gluconacetobacter hansenii | In situ incorporation | UF | Au, E. coli | 97% | [166] | |
| PDA/BNC | Gluconacetobacter hansenii | In situ incorporation | NF | R6G, MO, MB | Avg. 89% | [161] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saud, A.; Saleem, H.; Zaidi, S.J. Progress and Prospects of Nanocellulose-Based Membranes for Desalination and Water Treatment. Membranes 2022, 12, 462. https://doi.org/10.3390/membranes12050462
Saud A, Saleem H, Zaidi SJ. Progress and Prospects of Nanocellulose-Based Membranes for Desalination and Water Treatment. Membranes. 2022; 12(5):462. https://doi.org/10.3390/membranes12050462
Chicago/Turabian StyleSaud, Asif, Haleema Saleem, and Syed Javaid Zaidi. 2022. "Progress and Prospects of Nanocellulose-Based Membranes for Desalination and Water Treatment" Membranes 12, no. 5: 462. https://doi.org/10.3390/membranes12050462
APA StyleSaud, A., Saleem, H., & Zaidi, S. J. (2022). Progress and Prospects of Nanocellulose-Based Membranes for Desalination and Water Treatment. Membranes, 12(5), 462. https://doi.org/10.3390/membranes12050462







