Modeling of Nanofiltration Process Using DSPM-DE Model for Purification of Amine Solution
Abstract
1. Introduction
2. Materials and Methods
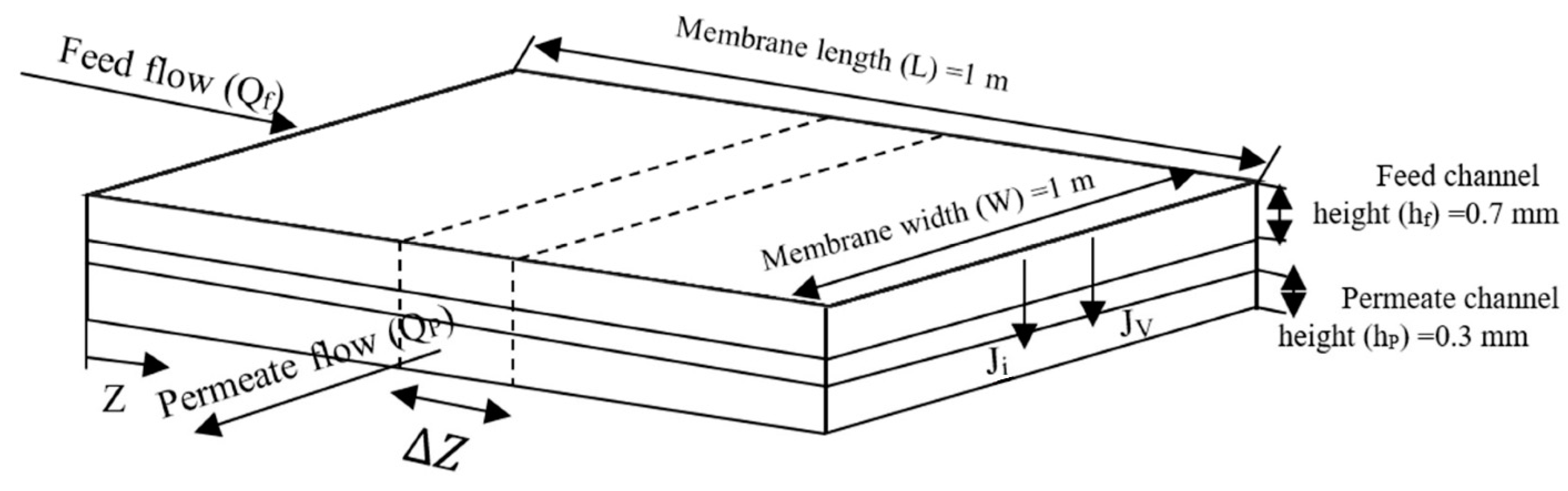
2.1. Experimental Set-Up
2.2. Process Modeling
- the gradient of the concentration is neglected in the width and length direction along feed side;
- flow rate profile along the module is obtained by total mass balance equation;
- mass transfer by diffusion in the axial direction is neglected due to the high flow rate of solvent.
3. Results and Discussion
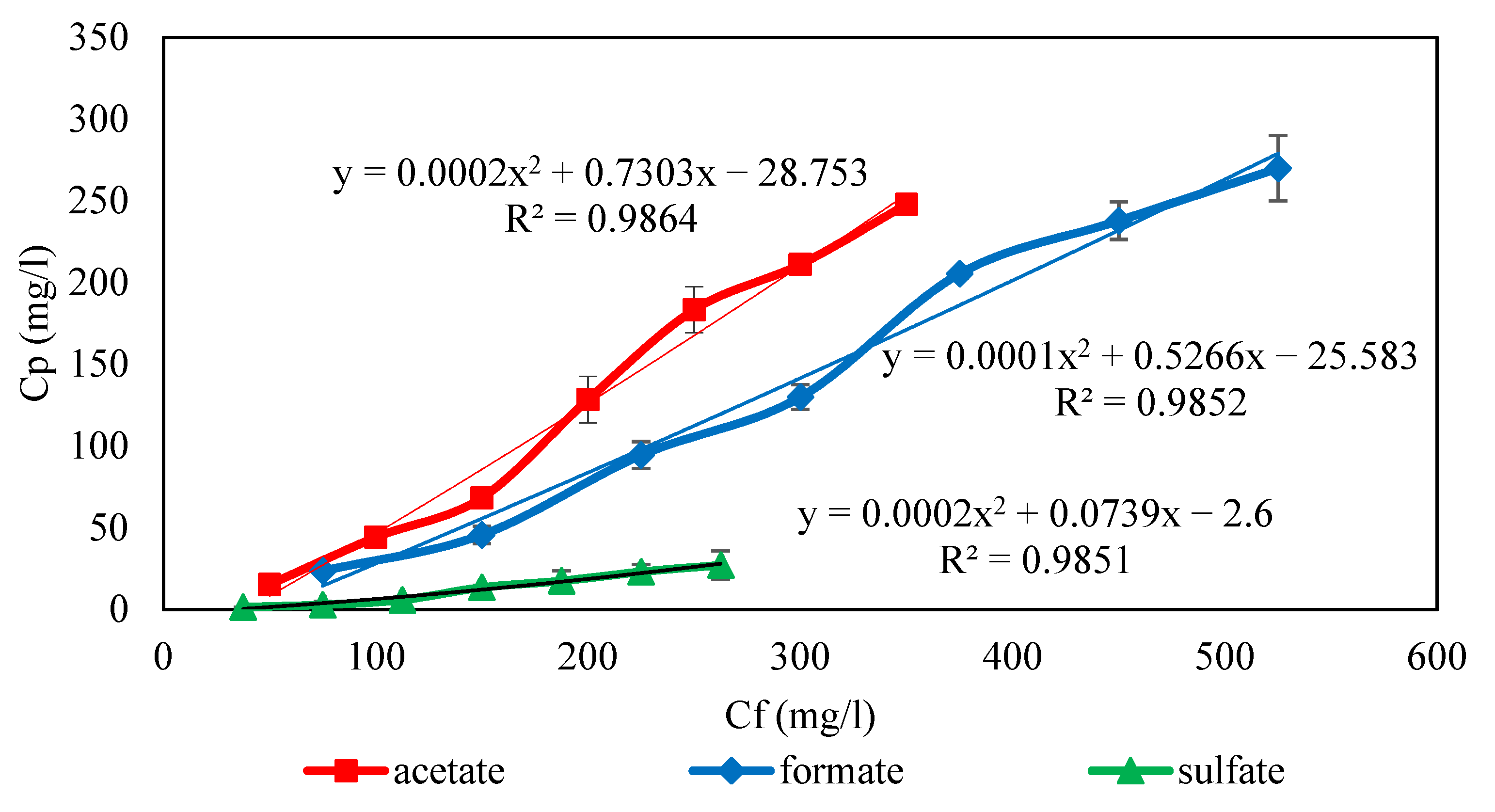
3.1. Correlation for Ci,P = f(Ci,f)
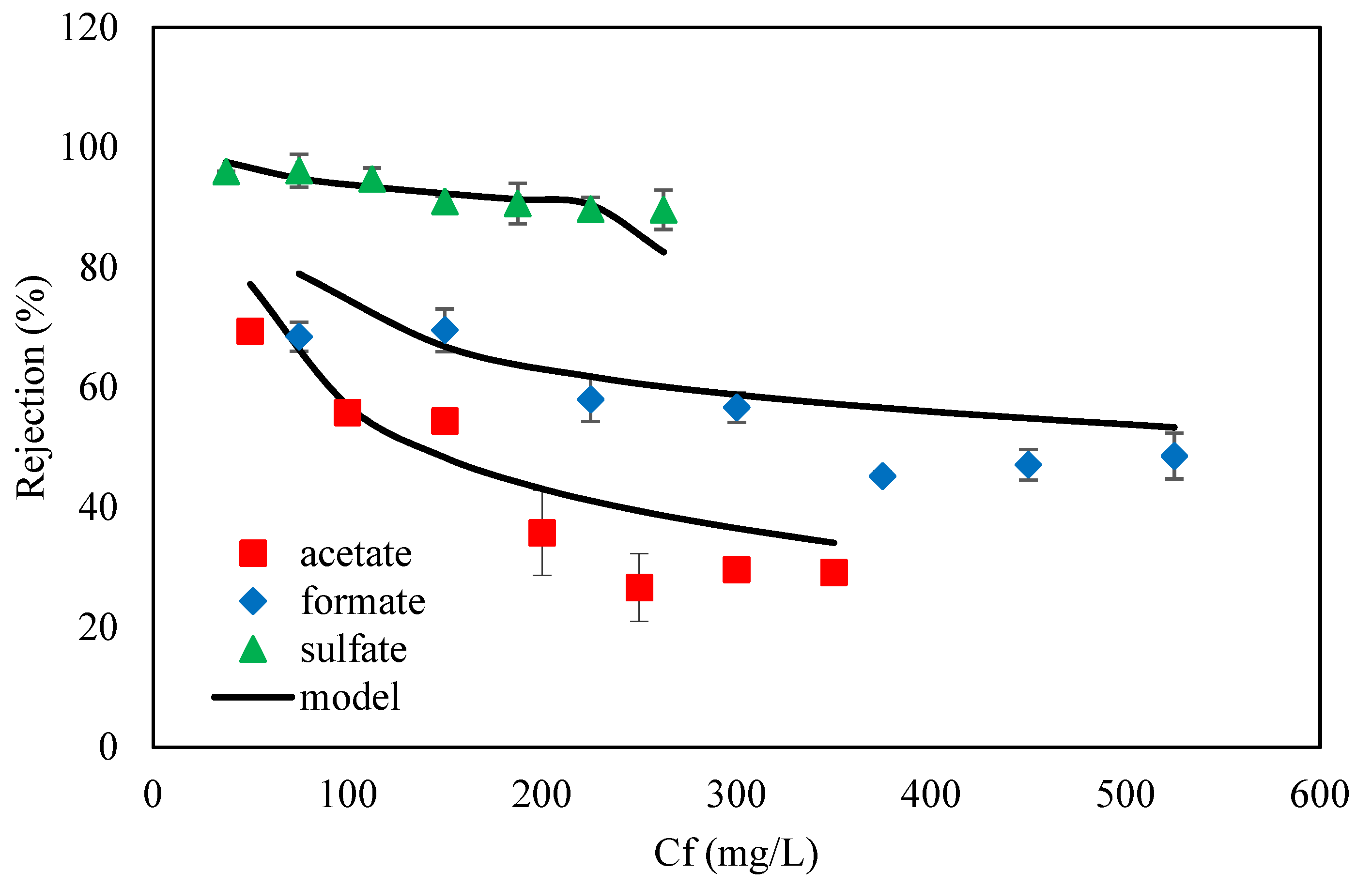
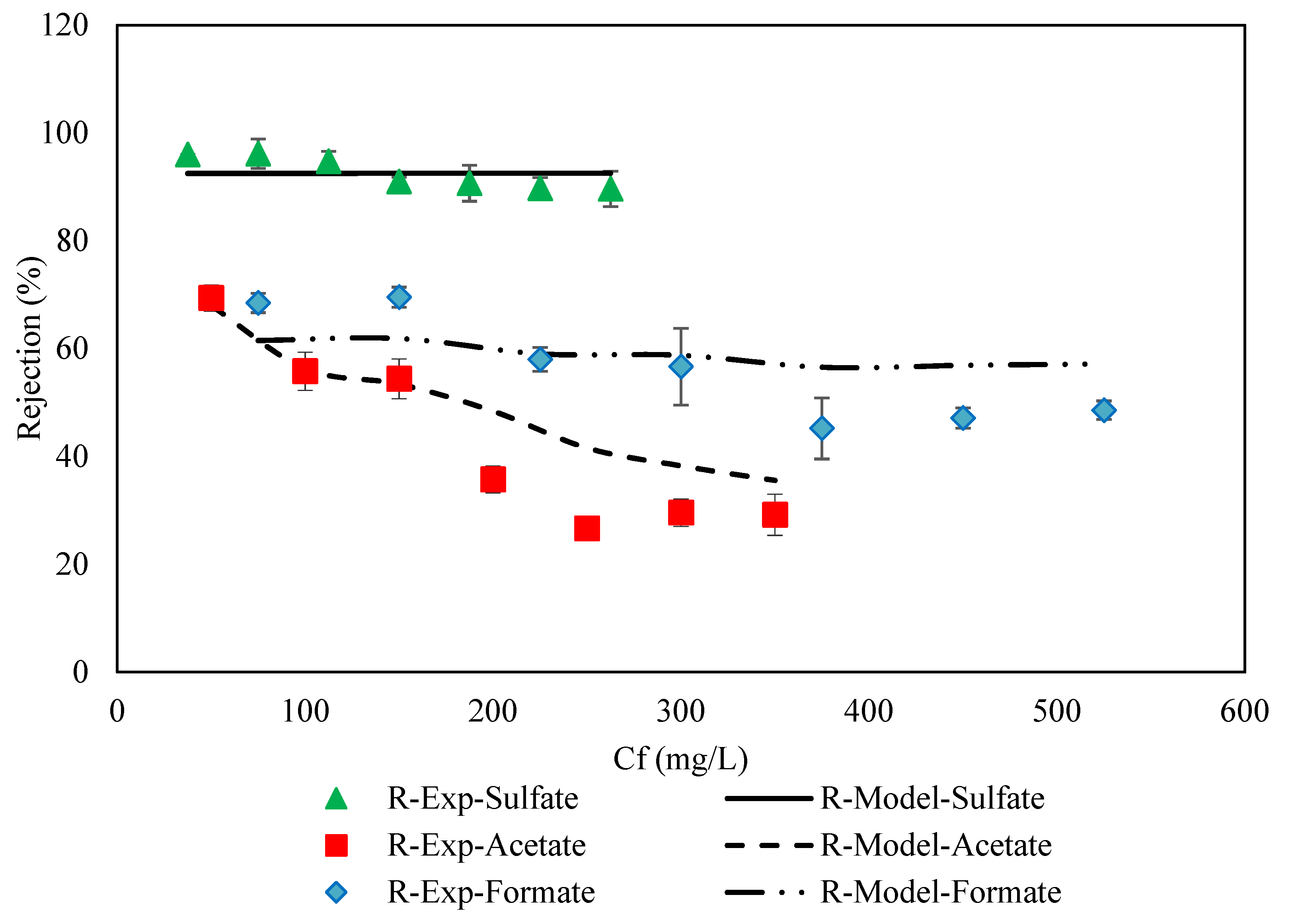
3.2. DSPM-DE Validation on Experimental Data
3.3. Ion Diffusion Coefficients Calculation
3.4. Analysis and Model Description
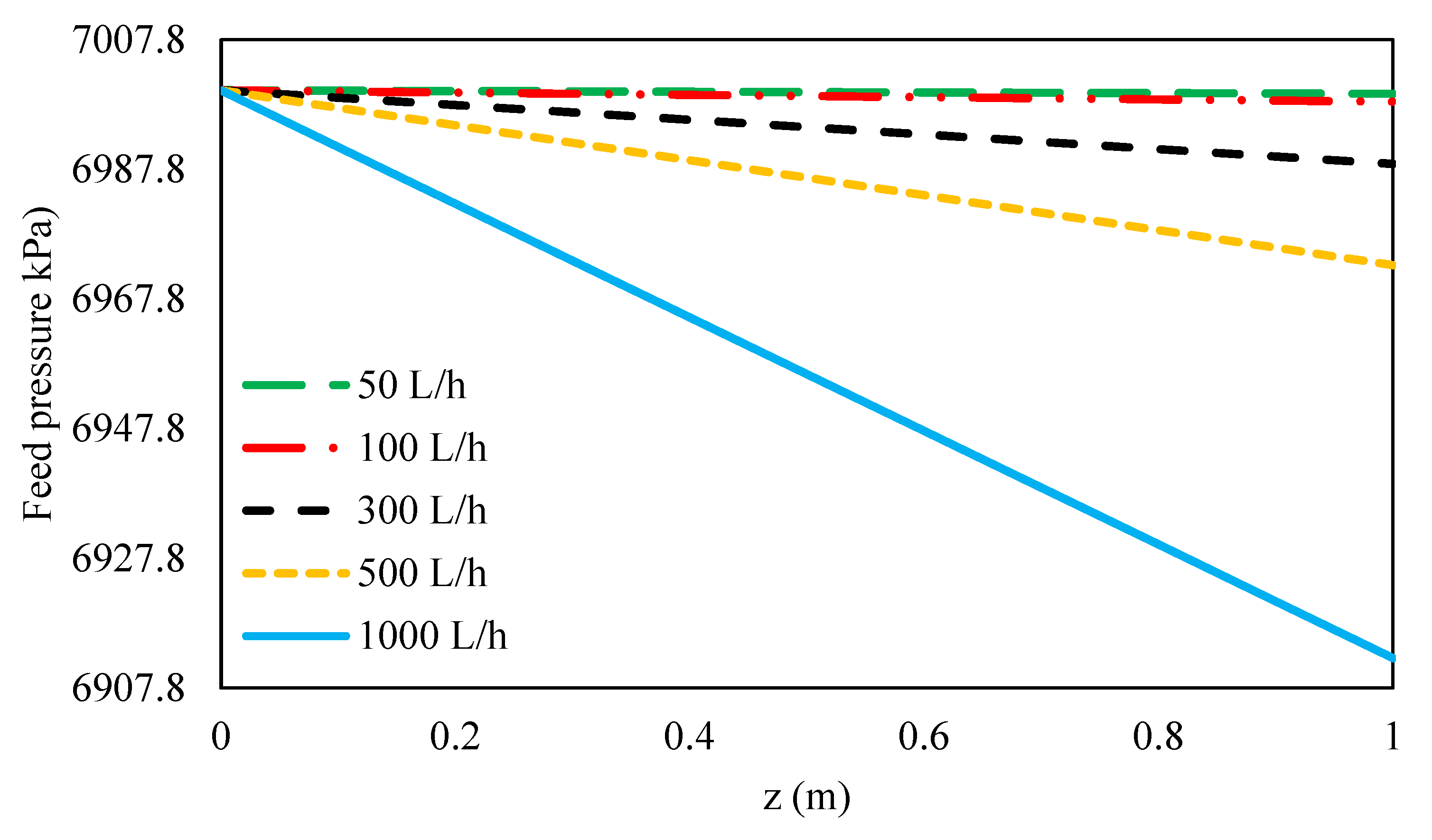
3.4.1. Effect of Feed Flow Rate on Feed Pressure Variation along the Feed Flow Direction
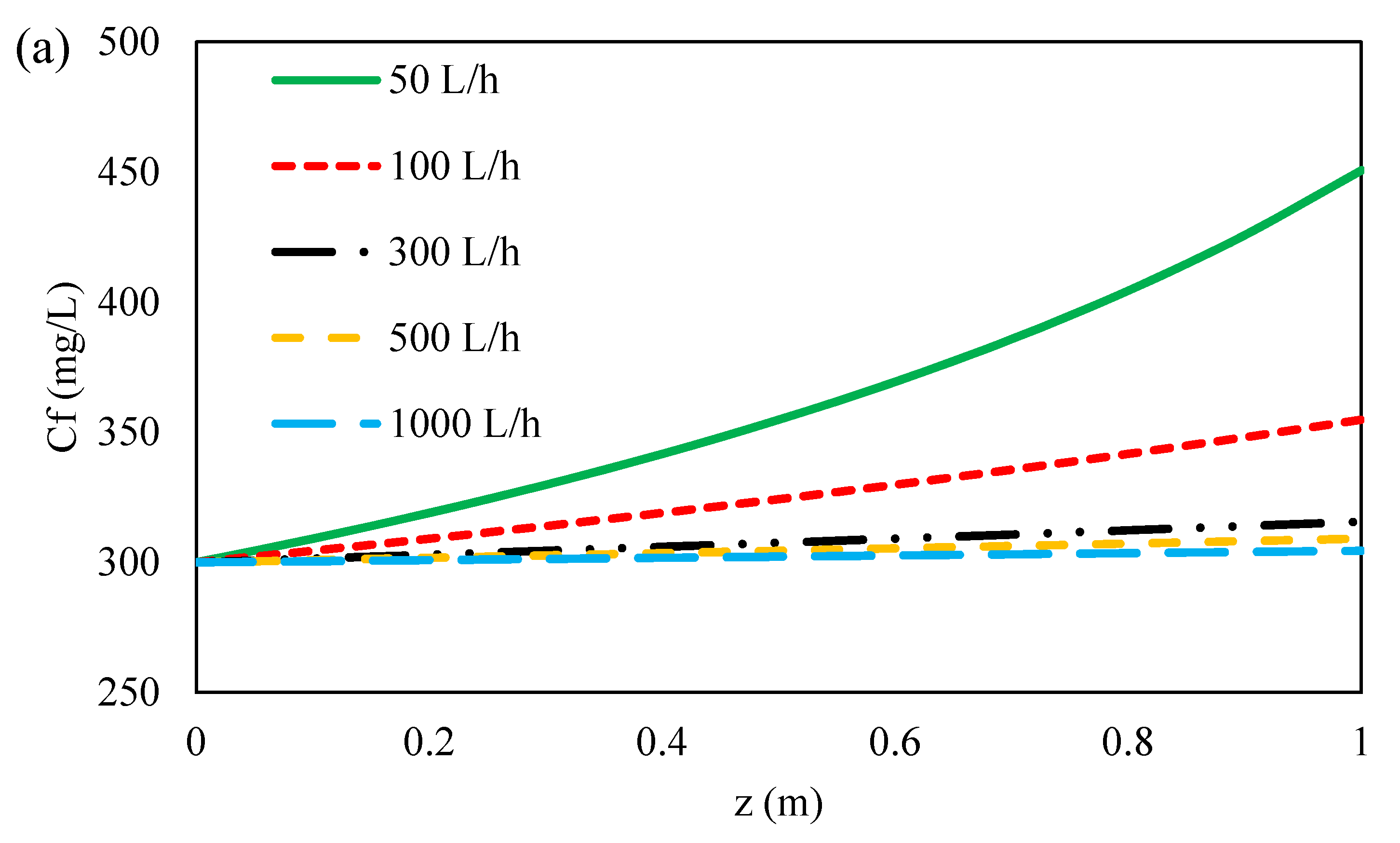
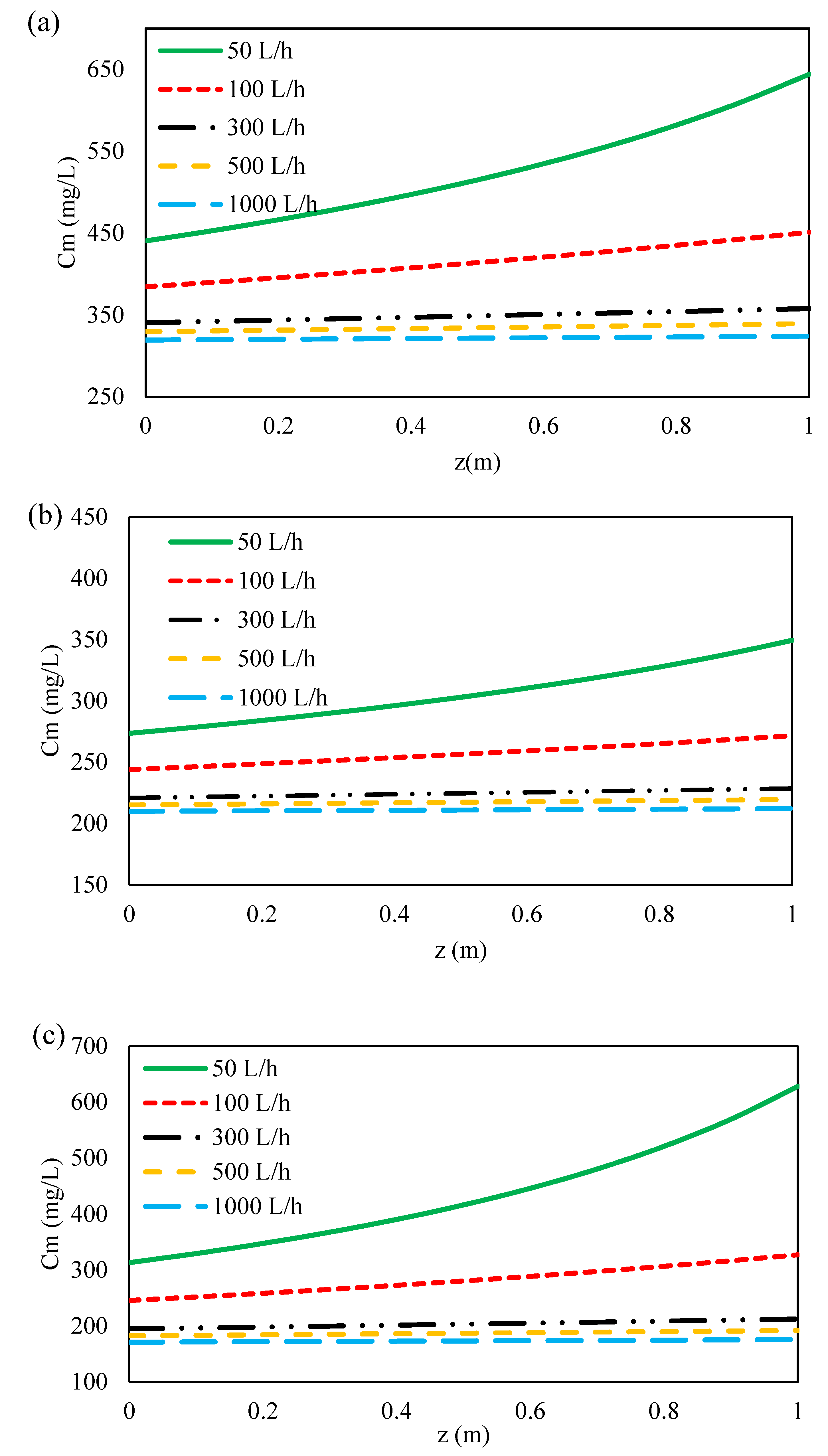
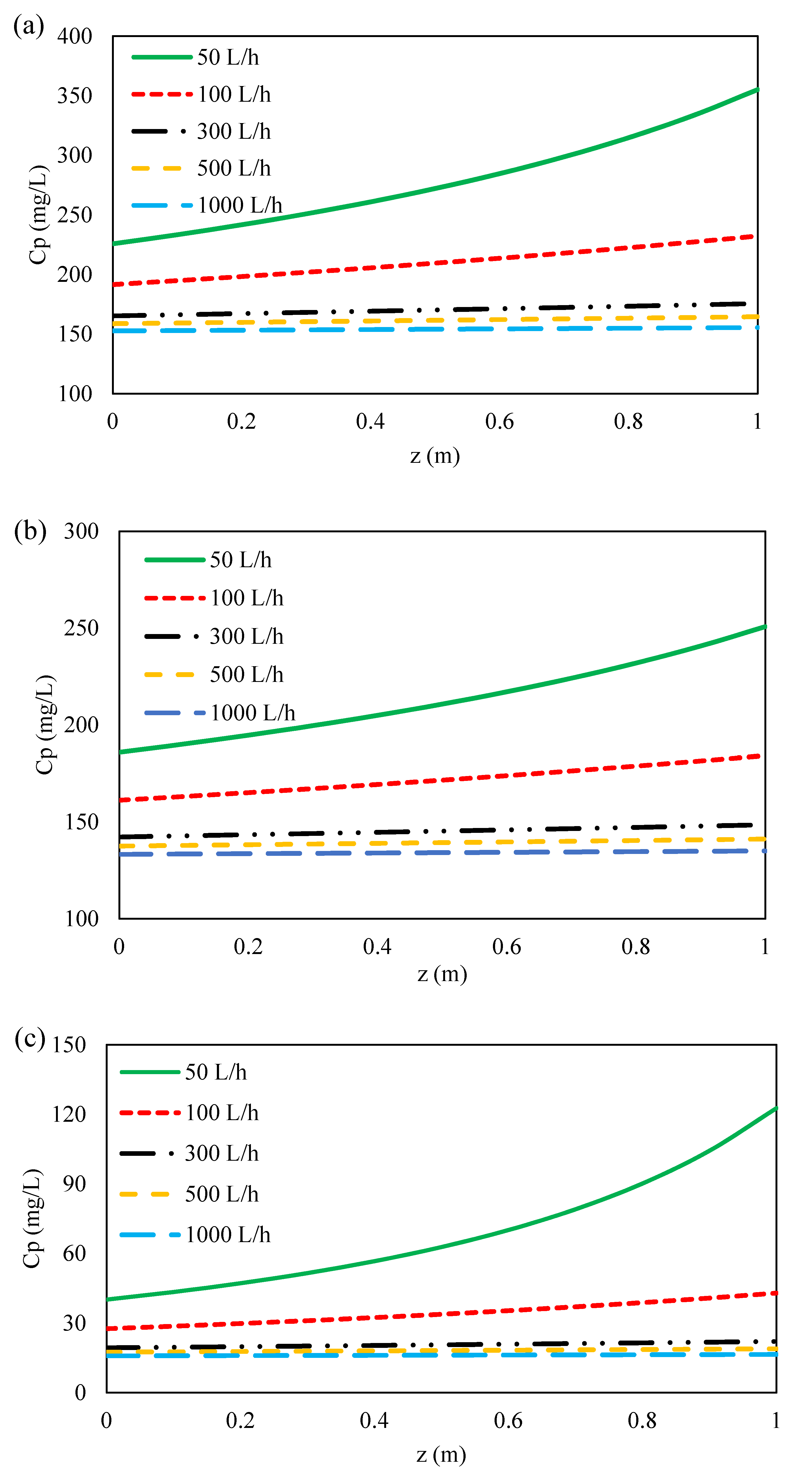
3.4.2. Effect of Feed Flow Rate on Ions Concentration along Flow Direction
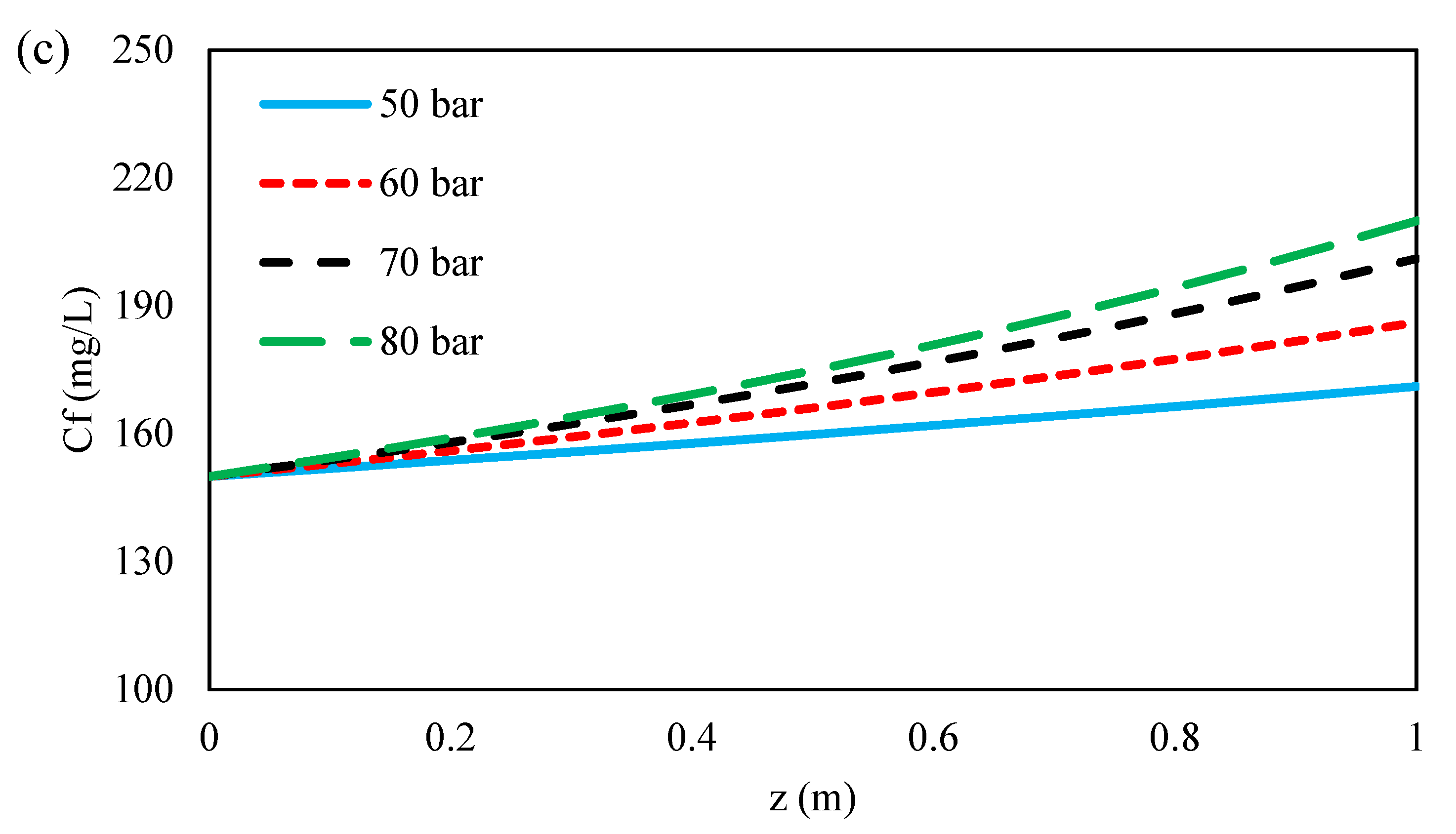
3.4.3. Effect of Inlet Feed Pressure on the Ions Concentration along Flow Direction
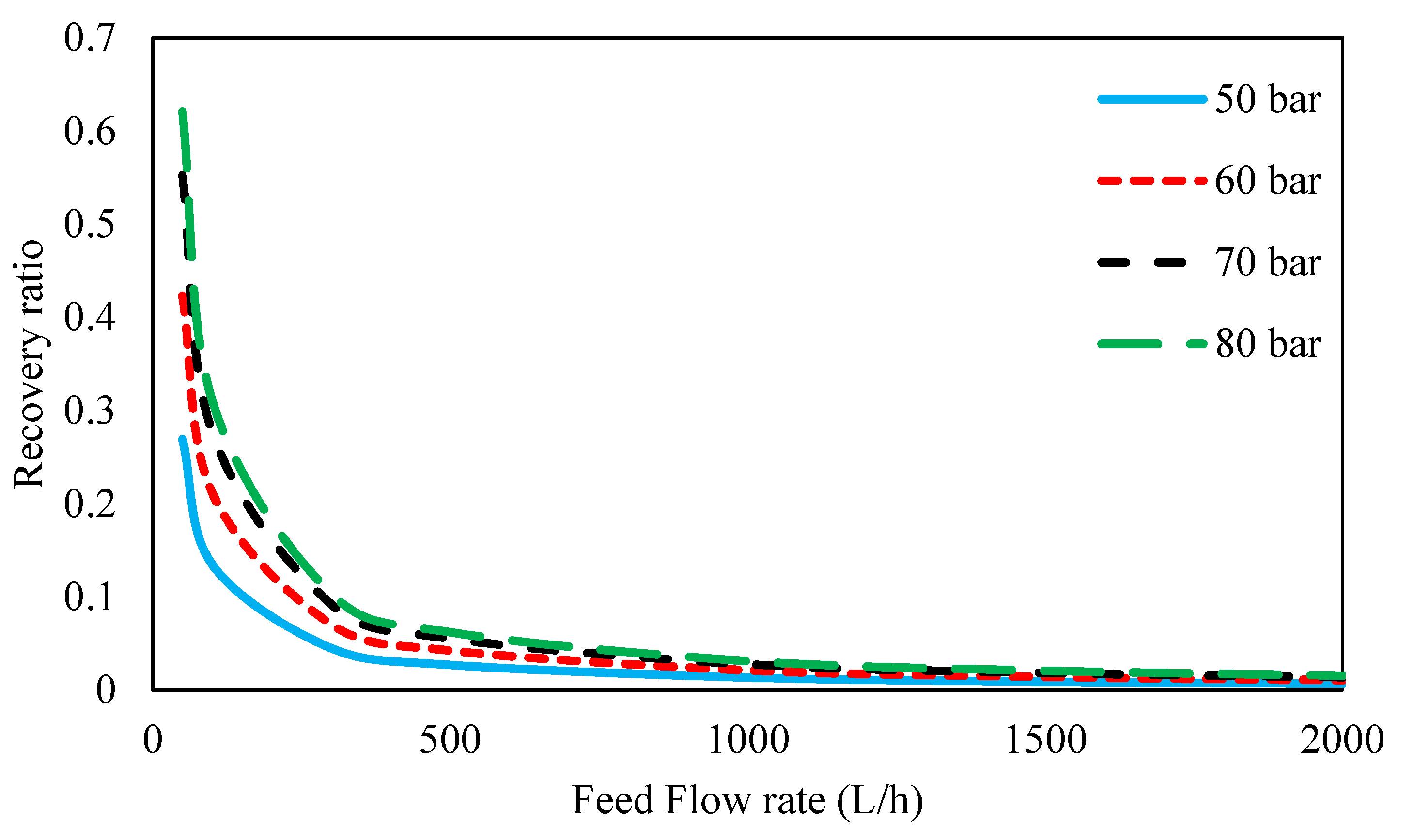
3.4.4. Effect of Inlet Feed Pressure and Feed Flow Rate on Rejection and Recovery Ratios
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A

References
- Al-Lagtah, N.M.; Al-Habsi, S.; Onaizi, S.A. Optimization and performance improvement of Lekhwair natural gas sweetening plant using Aspen HYSYS. J. Nat. Gas. Sci. Eng. 2015, 26, 367–381. [Google Scholar] [CrossRef]
- Gutierrez, J.P.; Benitez, L.A.; Ruiz, E.L.A.; Erdmann, E. A sensitivity analysis and a comparison of two simulators performance for the process of natural gas sweetening. J. Nat. Gas. Sci. Eng. 2016, 31, 800–807. [Google Scholar] [CrossRef]
- Jaafari, L.; Jaffary, B.; Idem, R. Screening study for selecting new activators for activating MDEA for natural gas sweetening. Sep. Purif. Technol. 2018, 199, 320–330. [Google Scholar] [CrossRef]
- Lu, H.; Kanehashi, S.; Scholes, C.; Kentish, S. The impact of ethylene glycol and hydrogen sulphide on the performance of cellulose triacetate membranes in natural gas sweetening. J. Membr. Sci. 2017, 539, 432–440. [Google Scholar] [CrossRef]
- Qiu, K.; Shang, J.; Ozturk, M.C.; Li, T.; Chen, S.; Zhang, L.; Gu, X. Studies of methyldiethanolamine process simulation and parameters optimization for high-sulfur gas sweetening. J. Nat. Gas. Sci. Eng. 2014, 21, 379–385. [Google Scholar] [CrossRef]
- Santaniello, A.; Golemme, G. Interfacial control in perfluoropolymer mixed matrix membranes for natural gas sweetening. J. Ind. Eng. Chem. 2018, 60, 169–176. [Google Scholar] [CrossRef]
- Alnaimat, F.; Alhseinat, E.; Banat, F. Electromagnetic separation of heat stable salt from gas sweetening amine. Int. J. Greenh. Gas. Control. 2018, 68, 256–268. [Google Scholar] [CrossRef]
- Fouad, W.A.; Berrouk, A.S. Using mixed tertiary amines for gas sweetening energy requirement reduction. J. Nat. Gas. Sci. Eng. 2013, 11, 12–17. [Google Scholar] [CrossRef]
- Najibi, H.; Maleki, N. Equilibrium solubility of carbon dioxide in N-methyldiethanolamine+piperazine aqueous solution: Experimental measurement and prediction. Fluid Phase Equilibria 2013, 354, 298–303. [Google Scholar] [CrossRef]
- Jassim, M.S. Sensitivity analyses and optimization of a gas sweetening plant for hydrogen sulfide and carbon dioxide capture using methyldiethanolamine solutions. J. Nat. Gas. Sci. Eng. 2016, 36, 175–183. [Google Scholar] [CrossRef]
- Keewan, M.; Banat, F.; Alhseinat, E.; Zain, J.; Pal, P. Effect of operating parameters and corrosion inhibitors on foaming behavior of aqueous methyldiethanolamine solutions. J. Pet. Sci. Eng. 2018, 165, 358–364. [Google Scholar] [CrossRef]
- Pal, P.; AbuKashabeh, A.; Al-Asheh, S.; Banat, F. Role of aqueous methyldiethanolamine (MDEA) as solvent in natural gas sweetening unit and process contaminants with probable reaction pathway. J. Nat. Gas. Sci. Eng. 2015, 24, 124–131. [Google Scholar] [CrossRef]
- Meng, H.; Zhang, S.; Li, C.; Li, L. Removal of heat stable salts from aqueous solutions of N-methyldiethanolamine using a specially designed three-compartment configuration electrodialyzer. J. Membr. Sci. 2008, 322, 436–440. [Google Scholar] [CrossRef]
- Pal, P.; Banat, F.; Alshoaibi, A. Adsorptive removal of heat stable salt anions from industrial lean amine solvent using anion exchange resins from gas sweetening unit. J. Nat. Gas. Sci. Eng. 2013, 15, 14–21. [Google Scholar] [CrossRef]
- Pal, P.; Abukashabeh, A.; Al-Asheh, S.; Banat, F. Accumulation of heat stable salts and degraded products during thermal degradation of aqueous methyldiethanolamine (MDEA) using microwave digester and high pressure reactor. J. Nat. Gas. Sci. Eng. 2014, 21, 1043–1047. [Google Scholar] [CrossRef]
- Cho, J.-H.; Jeon, S.-B.; Yang, K.-S.; Seo, J.-B.; Cho, S.-W.; Oh, K.-J. Regeneration of heat stable salts-loaded anion exchange resin by a novel zirconium pentahydroxide [Zr(OH) 5 − ] displacement technique in CO2 absorption process. Sep. Purif. Technol. 2015, 156, 465–471. [Google Scholar] [CrossRef]
- Verma, N.; Verma, A. Amine system problems arising from heat stable salts and solutions to improve system performance. Fuel Process. Technol. 2009, 90, 483–489. [Google Scholar] [CrossRef]
- Chen, F.; Chi, Y.; Zhang, M.; Liu, Z.; Fei, X.; Yang, K.; Fu, C. Removal of heat stable salts from N-methyldiethanolamine wastewater by anion exchange resin coupled three-compartment electrodialysis. Sep. Purif. Technol. 2020, 242, 116777. [Google Scholar] [CrossRef]
- Gao, J.; Wang, S.; Wang, J.; Cao, L.; Tang, S.; Xia, Y. Effect of SO2 on the amine-based CO2 capture solvent and improvement using ion exchange resins. Int. J. Greenh. Gas. Control. 2015, 37, 38–45. [Google Scholar] [CrossRef]
- Bazhenov, S.; Rieder, A.; Schallert, B.; Vasilevsky, V.; Unterberger, S.; Grushevenko, E.; Volkov, V.; Volkov, A. Reclaiming of degraded MEA solutions by electrodialysis: Results of ED pilot campaign at post-combustion CO2 capture pilot plant. Int. J. Greenh. Gas. Control. 2015, 42, 593–601. [Google Scholar] [CrossRef]
- Dumée, L.; Scholes, C.; Stevens, G.W.; Kentish, S. Purification of aqueous amine solvents used in post combustion CO2 capture: A review. Int. J. Greenh. Gas. Control. 2012, 10, 443–455. [Google Scholar] [CrossRef]
- Edathil, A.A.; Pal, P.; Banat, F. Alginate clay hybrid composite adsorbents for the reclamation of industrial lean methyldiethanolamine solutions. Appl. Clay Sci. 2018, 156, 213–223. [Google Scholar] [CrossRef]
- Roy, Y.; Warsinger, D.M.; Lienhard, J.H. Effect of temperature on ion transport in nanofiltration membranes: Diffusion, convection and electromigration. Desalination 2017, 420, 241–257. [Google Scholar] [CrossRef]
- Ryzhkov, I.I.; Minakov, A.V. Theoretical study of electrolyte transport in nanofiltration membranes with constant surface potential/charge density. J. Membr. Sci. 2016, 520, 515–528. [Google Scholar] [CrossRef]
- Zhao, C.; Tang, C.Y.; Li, P.; Adrian, P.; Hu, G. Perfluorooctane sulfonate removal by nanofiltration membrane—The effect and interaction of magnesium ion/humic acid. J. Membr. Sci. 2016, 503, 31–41. [Google Scholar] [CrossRef]
- Wei, X.; Shi, Y.; Fei, Y.; Chen, J.; Lv, B.; Chen, Y.; Zheng, H.; Shen, J.; Zhu, L. Removal of trace phthalate esters from water by thin-film composite nanofiltration hollow fiber membranes. Chem. Eng. J. 2016, 292, 382–388. [Google Scholar] [CrossRef]
- Diaz, P.A.B.; Kronemberger, F.D.A.; Habert, A.C. Effect of feed conditions and added solutes on the performance of membrane nanofiltration of succinic acid solutions. Braz. J. Chem. Eng. 2020, 37, 283–295. [Google Scholar] [CrossRef]
- Hu, K.; Dickson, J.M. Nanofiltration membrane performance on fluoride removal from water. J. Membr. Sci. 2006, 279, 529–538. [Google Scholar] [CrossRef]
- Ku, Y.; Chen, S.-W.; Wang, W.-Y. Effect of solution composition on the removal of copper ions by nanofiltration. Sep. Purif. Technol. 2005, 43, 135–142. [Google Scholar] [CrossRef]
- Pérez, L.; Escudero, I.; Arcos-Martínez, M.J.; Benito, J.M. Application of the solution-diffusion-film model for the transfer of electrolytes and uncharged compounds in a nanofiltration membrane. J. Ind. Eng. Chem. 2017, 47, 368–374. [Google Scholar] [CrossRef]
- Tahaikt, M.; El Habbani, R.; Haddou, A.A.; Achary, I.; Amor, Z.; Taky, M.; Alami, A.; Boughriba, A.; Hafsi, M.; Elmidaoui, A. Fluoride removal from groundwater by nanofiltration. Desalination 2007, 212, 46–53. [Google Scholar] [CrossRef]
- Shang, W.; Sun, F.; Jia, W.; Guo, J.; Yin, S.; Wong, P.W.; An, A.K. High-performance nanofiltration membrane structured with enhanced stripe nano-morphology. J. Membr. Sci. 2020, 600, 117852. [Google Scholar] [CrossRef]
- Bolong, N.; Ismail, A.; Salim, M.R.; Rana, D.; Matsuura, T.; Tabe-Mohammadi, A. Negatively charged polyethersulfone hollow fiber nanofiltration membrane for the removal of bisphenol A from wastewater. Sep. Purif. Technol. 2010, 73, 92–99. [Google Scholar] [CrossRef]
- Epsztein, R.; Cheng, W.; Shaulsky, E.; Dizge, N.; Elimelech, M. Elucidating the mechanisms underlying the difference between chloride and nitrate rejection in nanofiltration. J. Membr. Sci. 2018, 548, 694–701. [Google Scholar] [CrossRef]
- Yaroshchuk, A. Non-steric mechanisms of nanofiltration: Superposition of Donnan and dielectric exclusion. Sep. Purif. Technol. 2001, 22–23, 143–158. [Google Scholar] [CrossRef]
- Zhang, R.; Shi, W.; Yu, S.; Wang, W.; Zhang, Z.; Zhang, B.; Li, L.; Bao, X. Influence of salts, anion polyacrylamide and crude oil on nanofiltration membrane fouling during desalination process of polymer flooding produced water. Desalination 2015, 373, 27–37. [Google Scholar] [CrossRef]
- Santafé-Moros, A.; Gozálvez-Zafrilla, J.; Lora-García, J. Performance of commercial nanofiltration membranes in the removal of nitrate ions. Desalination 2005, 185, 281–287. [Google Scholar] [CrossRef]
- Saliha, B.; Patrick, F.; Anthony, S. Investigating nanofiltration of multi-ionic solutions using the steric, electric and dielectric exclusion model. Chem. Eng. Sci. 2009, 64, 3789–3798. [Google Scholar] [CrossRef]
- Wang, Y.; Li, W.; Yan, H.; Xu, T. Removal of heat stable salts (HSS) from spent alkanolamine wastewater using electrodialysis. J. Ind. Eng. Chem. 2018, 57, 356–362. [Google Scholar] [CrossRef]
- Bandini, S.; Vezzani, D. Nanofiltration modeling: The role of dielectric exclusion in membrane characterization. Chem. Eng. Sci. 2003, 58, 3303–3326. [Google Scholar] [CrossRef]
- Ren, T.; Han, L.; Liu, R.; Ma, C.; Chen, X.; Zhao, S.; Zhang, Y. Influence of inorganic salt on retention of ibuprofen by nanofiltration. Sep. Purif. Technol. 2017, 189, 382–388. [Google Scholar] [CrossRef]
- Fridman-Bishop, N.; Tankus, K.A.; Freger, V. Permeation mechanism and interplay between ions in nanofiltration. J. Membr. Sci. 2018, 548, 449–458. [Google Scholar] [CrossRef]
- Nicolini, J.V.; Borges, C.P.; Ferraz, H.C. Selective rejection of ions and correlation with surface properties of nanofiltration membranes. Sep. Purif. Technol. 2016, 171, 238–247. [Google Scholar] [CrossRef]
- Park, M.; Snyder, S.A. Chapter 6-Attenuation of contaminants of emerging concerns by nanofiltration membrane: Rejection mechanism and application in water reuse. In Contaminants of Emerging Concern in Water and Wastewater; Hernán-dez-Maldonado, A.J., Blaney, L., Eds.; Butterworth-Heinemann: Oxford, UK, 2020; pp. 177–206. [Google Scholar] [CrossRef]
- Lim, J.; Scholes, C.A.; Dumee, L.; Kentish, S.E. Nanofiltration for the concentration of heat stable salts prior to MEA reclamation. Int. J. Greenh. Gas. Control. 2014, 30, 34–41. [Google Scholar] [CrossRef]
- Ghorbani, A.; Bayati, B.; Poerio, T.; Argurio, P.; Kikhavani, T.; Namdari, M.; Ferreira, L.M. Application of NF Polymeric Membranes for Removal of Multicomponent Heat-Stable Salts (HSS) Ions from Methyl Diethanolamine (MDEA) Solutions. Molecules 2020, 25, 4911. [Google Scholar] [CrossRef]
- Ghorbani, A.; Bayati, B.; Kikhavani, T. Modelling Ion Transport in an Amine Solution through a Nanofiltration Membrane. Braz. J. Chem. Eng. 2019, 36, 1667–1677. [Google Scholar] [CrossRef]
- Roy, Y.; Sharqawy, M.H.; Lienhard, J.H. Modeling of flat-sheet and spiral-wound nanofiltration configurations and its application in seawater nanofiltration. J. Membr. Sci. 2015, 493, 360–372. [Google Scholar] [CrossRef]
- Léniz-Pizarro, F.; Liu, C.; Colburn, A.; Escobar, I.C.; Bhattacharyya, D. Positively charged nanofiltration membrane synthesis, transport models, and lanthanides separation. J. Membr. Sci. 2021, 620, 118973. [Google Scholar] [CrossRef]
- Xu, R.; Zhou, M.; Wang, H.; Wang, X.; Wen, X. Influences of temperature on the retention of PPCPs by nanofiltration membranes: Experiments and modeling assessment. J. Membr. Sci. 2020, 599, 117817. [Google Scholar] [CrossRef]
- Bowen, W.; Welfoot, J.S. Modelling the performance of membrane nanofiltration—Critical assessment and model development. Chem. Eng. Sci. 2002, 57, 1121–1137. [Google Scholar] [CrossRef]
- Wang, R.; Lin, S. Pore model for nanofiltration: History, theoretical framework, key predictions, limitations, and prospects. J. Membr. Sci. 2021, 620, 118809. [Google Scholar] [CrossRef]
- Chakrabortty, S.; Roy, M.; Pal, P. Removal of fluoride from contaminated groundwater by cross flow nanofiltration: Transport modeling and economic evaluation. Desalination 2013, 313, 115–124. [Google Scholar] [CrossRef]
- Geraldes, V.; Alves, A.M.B. Computer program for simulation of mass transport in nanofiltration membranes. J. Membr. Sci. 2008, 321, 172–182. [Google Scholar] [CrossRef]
- Zerafat, M.; Shariati-Niassar, M.; Hashemi, S.; Sabbaghi, S.; Ismail, A.; Matsuura, T. Mathematical modeling of nanofiltration for concentrated electrolyte solutions. Desalination 2013, 320, 17–23. [Google Scholar] [CrossRef]
- Kumar, R.; Chakrabortty, S.; Pal, P. Membrane-integrated physico-chemical treatment of coke-oven wastewater: Transport modelling and economic evaluation. Environ. Sci. Pollut. Res. 2014, 22, 6010–6023. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, S.; Rezaei, M.; Bag-Mohammadi, M.; Altzibar, H.; Olazar, M. Smart models to predict the minimum spouting velocity of conical spouted beds with non-porous draft tube. Chem. Eng. Res. Des. 2018, 138, 331–340. [Google Scholar] [CrossRef]
- Hosseini, S.; Rezaei, M.; Bag-Mohammadi, M.; Zendehboudi, A.; Ahmadi, G.; Bag-Mohammdi, M. Prediction of frost layer over flat plates under natural and forced convection conditions using intelligent and least-square fitting approaches. Appl. Therm. Eng. 2019, 148, 33–42. [Google Scholar] [CrossRef]
- Karimi, M.; Vaferi, B.; Hosseini, S.H.; Rasteh, M. Designing an Efficient Artificial Intelligent Approach for Estimation of Hydrodynamic Characteristics of Tapered Fluidized Bed from Its Design and Operating Parameters. Ind. Eng. Chem. Res. 2018, 57, 259–267. [Google Scholar] [CrossRef]
- Zendehboudi, A.; Saidur, R.; Mahbubul, I.; Hosseini, S. Data-driven methods for estimating the effective thermal conductivity of nanofluids: A comprehensive review. Int. J. Heat Mass Transf. 2019, 131, 1211–1231. [Google Scholar] [CrossRef]
- Oatley, D.L.; Llenas, L.; Pérez, R.; Williams, P.M.; Martínez-Lladó, X.; Rovira, M. Review of the dielectric properties of nanofiltration membranes and verification of the single oriented layer approximation. Adv. Colloid Interface Sci. 2012, 173, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Pérez-González, A.; Ibáñez, R.; Gómez, P.; Urtiaga, A.; Ortiz, I.; Irabien, J. Nanofiltration separation of polyvalent and monovalent anions in desalination brines. J. Membr. Sci. 2015, 473, 16–27. [Google Scholar] [CrossRef]
- Kim, S.; Hoek, E.M. Modeling concentration polarization in reverse osmosis processes. Desalination 2005, 186, 111–128. [Google Scholar] [CrossRef]
- Schock, G.; Miquel, A. Mass transfer and pressure loss in spiral wound modules. Desalination 1987, 64, 339–352. [Google Scholar] [CrossRef]
- Chilyumova, E.; Thöming, J. Nanofiltration of bivalent nickel cations—Model parameter determination and process simulation. Desalination 2008, 224, 12–17. [Google Scholar] [CrossRef]
- Wang, J.; Luo, J.; Zhang, X.; Wan, Y. Concentration of ionic liquids by nanofiltration for recycling: Filtration behavior and modeling. Sep. Purif. Technol. 2016, 165, 18–26. [Google Scholar] [CrossRef]
- Nakari, O.; Pihlajamäki, A.; Mänttäri, M. Permeability of dilute ionic liquid solutions through a nanofiltration membrane—Effect of ionic liquid concentration, filtration pressure and temperature. Sep. Purif. Technol. 2016, 163, 267–274. [Google Scholar] [CrossRef]
- Meschke, K.; Hansen, N.; Hofmann, R.; Haseneder, R.; Repke, J.-U. Influence of process parameters on separation performance of strategic elements by polymeric nanofiltration membranes. Sep. Purif. Technol. 2020, 235, 116186. [Google Scholar] [CrossRef]
- Szymczyk, A.; Fievet, P. Ion transport through nanofiltration membranes: The steric, electric and dielectric exclusion model. Desalination 2006, 200, 122–124. [Google Scholar] [CrossRef]
- Escoda, A.; Déon, S.; Fievet, P. Assessment of dielectric contribution in the modeling of multi-ionic transport through nanofiltration membranes. J. Membr. Sci. 2011, 378, 214–223. [Google Scholar] [CrossRef]
- Worou, C.; Kang, J.; Shen, J.; Yan, P.; Wang, W.; Gong, Y.; Chen, Z. Runge–Kutta Numerical Method Followed by Richardson’s Extrapolation for Efficient Ion Rejection Reassessment of a Novel Defect-Free Synthesized Nanofiltration Membrane. Membranes 2021, 11, 130. [Google Scholar] [CrossRef]
- Geraldes, V. Flow management in nanofiltration spiral wound modules with ladder-type spacers. J. Membr. Sci. 2002, 203, 87–102. [Google Scholar] [CrossRef]
- Koo, C.H.; Mohammad, A.W.; Suja’, F. Effect of cross-flow velocity on membrane filtration performance in relation to membrane properties. DESALINATION Water Treat. 2014, 55, 678–692. [Google Scholar] [CrossRef]
- Kumar, R.; Pal, P. Membrane-integrated hybrid system for the effective treatment of ammoniacal wastewater of coke-making plant: A volume reduction approach. Environ. Technol. 2014, 35, 2018–2027. [Google Scholar] [CrossRef]





| NF-3 Membrane | ||||||
|---|---|---|---|---|---|---|
| MWCO (Da) | Pore Radius (nm) | Membrane Thickness (μm) | (∆x/Ak) (μm) | Pure Water Permeate (L m−2 h−1 bar−1) | Operation Limits | Rejection (%) |
| 250–300 | 0.55 | 0.9 | 0.51 | 8.86 | 50 °C, 83 bar, 3–10 pH | NaCl: 60% MgSO4: 98% |
| Parameters | Values | References |
|---|---|---|
| Feed flow rate (L h−1) | 0.693 | |
| Cross flow velocity (m s−1 1 × 106) | 7.67 | |
| Temperature maintained in units (K) | 308 | |
| Solute radius of HCO2− ion (rs nm) | 0.00738 | |
| Solute radius of C2H3O2− ion (rs nm) | 0.00832 | |
| Solute radius of SO42− ion (rs nm) | 0.0102 | |
| Solute radius of Na+ ion (rs nm) | 0.116 | [53] |
| Solute radius of H+ (rs nm) | 0.025 | [56] |
| Bulk diffusivity of HCO2− ion (Di,∞ × 109 m2/s) | 1.33 | |
| Bulk diffusivity of C2H3O2− ion (Di,∞ × 109 m2/s) | 1.18 | |
| Bulk diffusivity of SO42− ion (Di,∞ × 109 m2/s) | 0.96 | |
| Bulk diffusivity of Na+ ion (Di,∞ × 109 m2 s−1) | 1.9 | [53] |
| Bulk diffusivity of H+ ion (Di,∞ × 109 m2 s−1) | 9.3 | [56] |
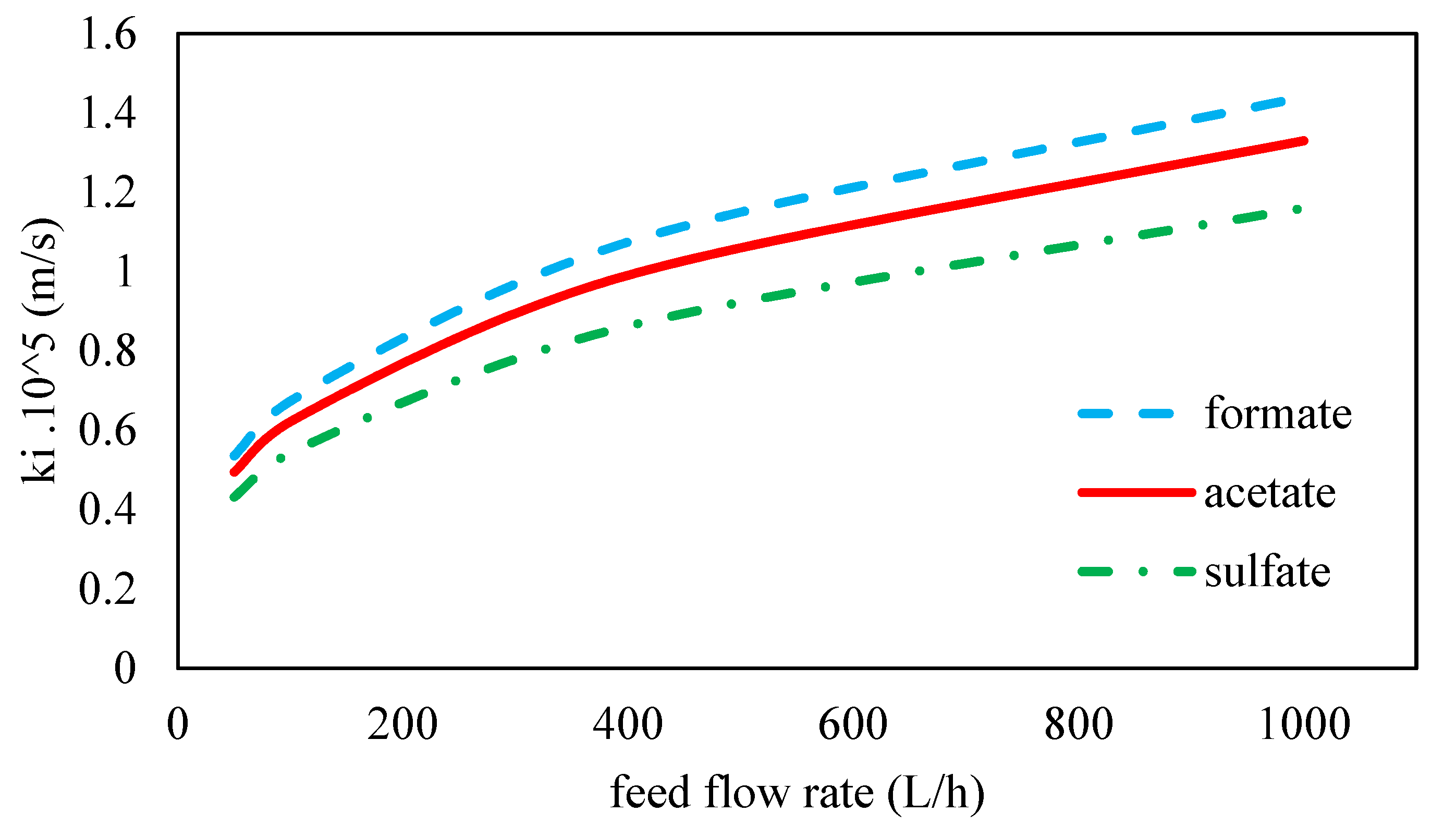
| Mass transfer coefficient of HCO2− ion (m s−1 × 105) | 1.44 | |
| Mass transfer coefficient of C2H3O2− ion (m s−1 × 105) | 1.33 | |
| Mass transfer coefficient of SO42− ion (m s−1 × 105) | 1.16 | |
| Boltzmann constant (k) (J K−1 × 10−25) | 1.38066 | [71] |
| Faraday constant | 96,500 | [71] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghorbani, A.; Bayati, B.; Drioli, E.; Macedonio, F.; Kikhavani, T.; Frappa, M. Modeling of Nanofiltration Process Using DSPM-DE Model for Purification of Amine Solution. Membranes 2021, 11, 230. https://doi.org/10.3390/membranes11040230
Ghorbani A, Bayati B, Drioli E, Macedonio F, Kikhavani T, Frappa M. Modeling of Nanofiltration Process Using DSPM-DE Model for Purification of Amine Solution. Membranes. 2021; 11(4):230. https://doi.org/10.3390/membranes11040230
Chicago/Turabian StyleGhorbani, Asma, Behrouz Bayati, Enrico Drioli, Francesca Macedonio, Tavan Kikhavani, and Mirko Frappa. 2021. "Modeling of Nanofiltration Process Using DSPM-DE Model for Purification of Amine Solution" Membranes 11, no. 4: 230. https://doi.org/10.3390/membranes11040230
APA StyleGhorbani, A., Bayati, B., Drioli, E., Macedonio, F., Kikhavani, T., & Frappa, M. (2021). Modeling of Nanofiltration Process Using DSPM-DE Model for Purification of Amine Solution. Membranes, 11(4), 230. https://doi.org/10.3390/membranes11040230








