Sustainable Fabrication of Organic Solvent Nanofiltration Membranes
Abstract
1. Introduction
2. Sustainable Membrane Materials
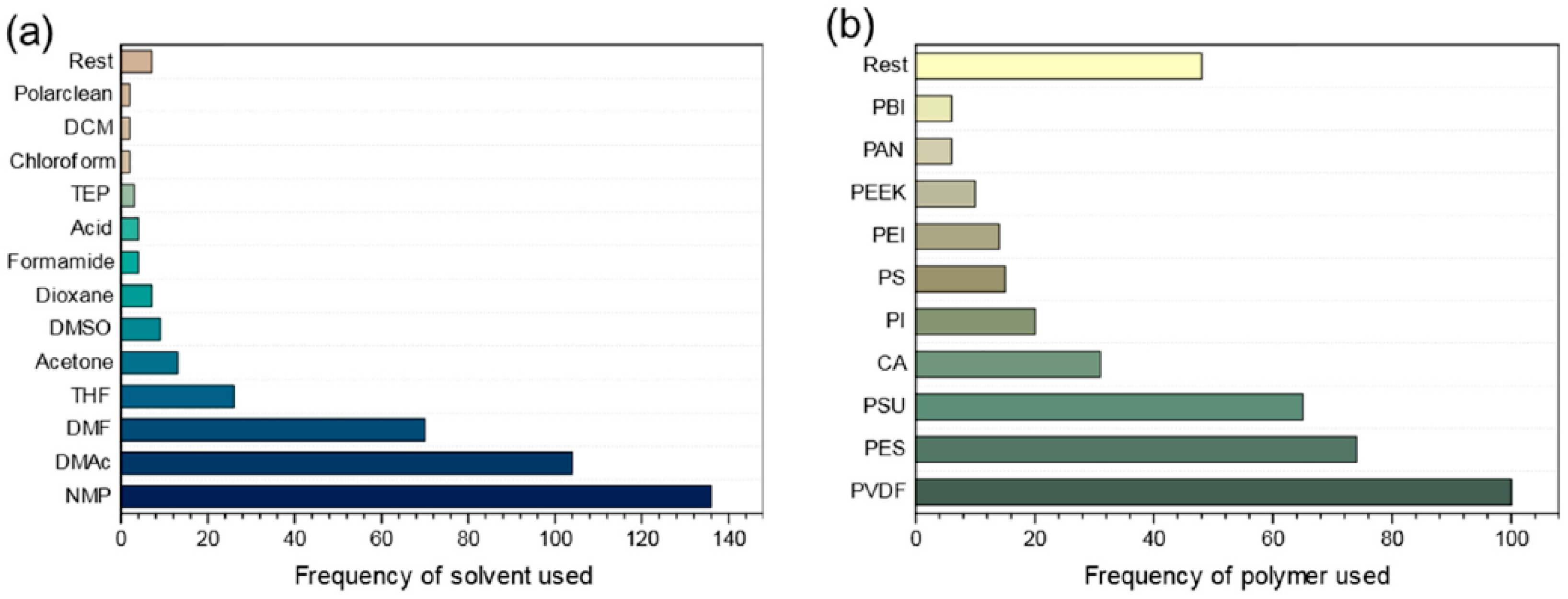
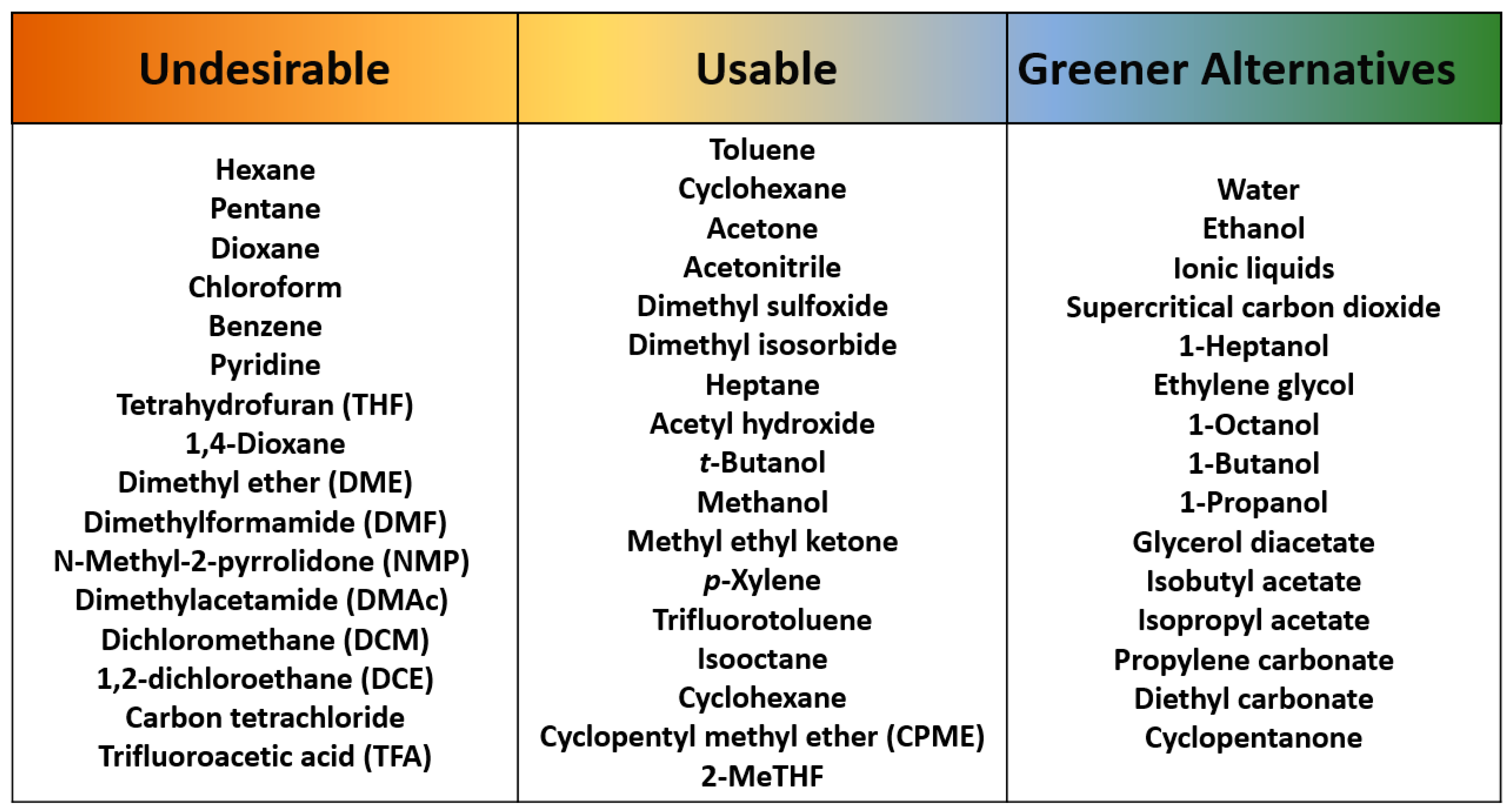
3. Sustainability for Employed Solvents
3.1. Water
3.2. γ-Valerolactone (GVL)
3.3. Methyl Lactate and Ethyl Lactate
3.4. Cyrene
3.5. Polarclean
3.6. Triethyl-Phosphate (TEP)
3.7. Ionic Liquid (IL) and Deep Eutectic Solvents (DES)
3.8. Other Green Solvents (Non-OSN Membranes)
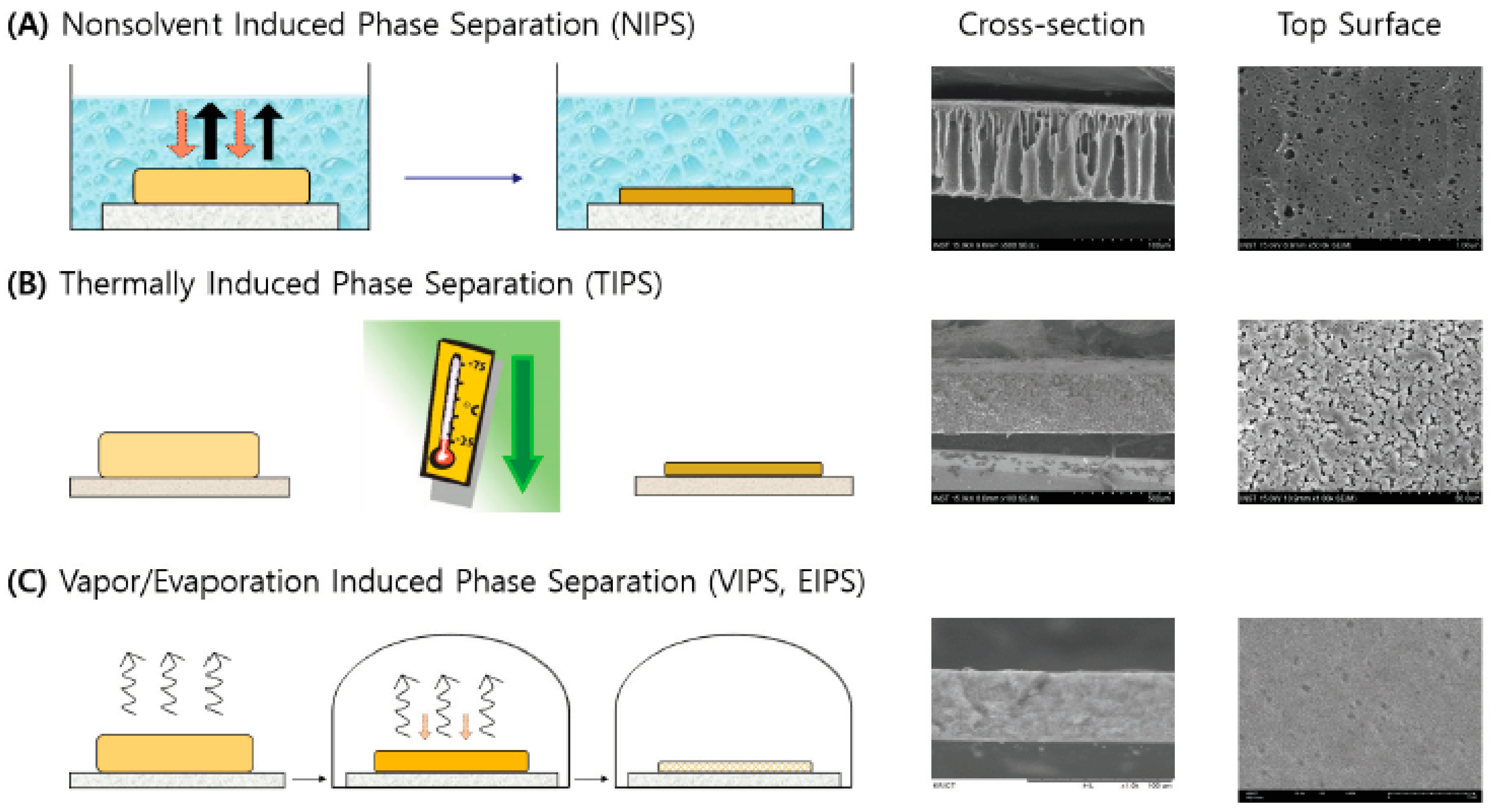
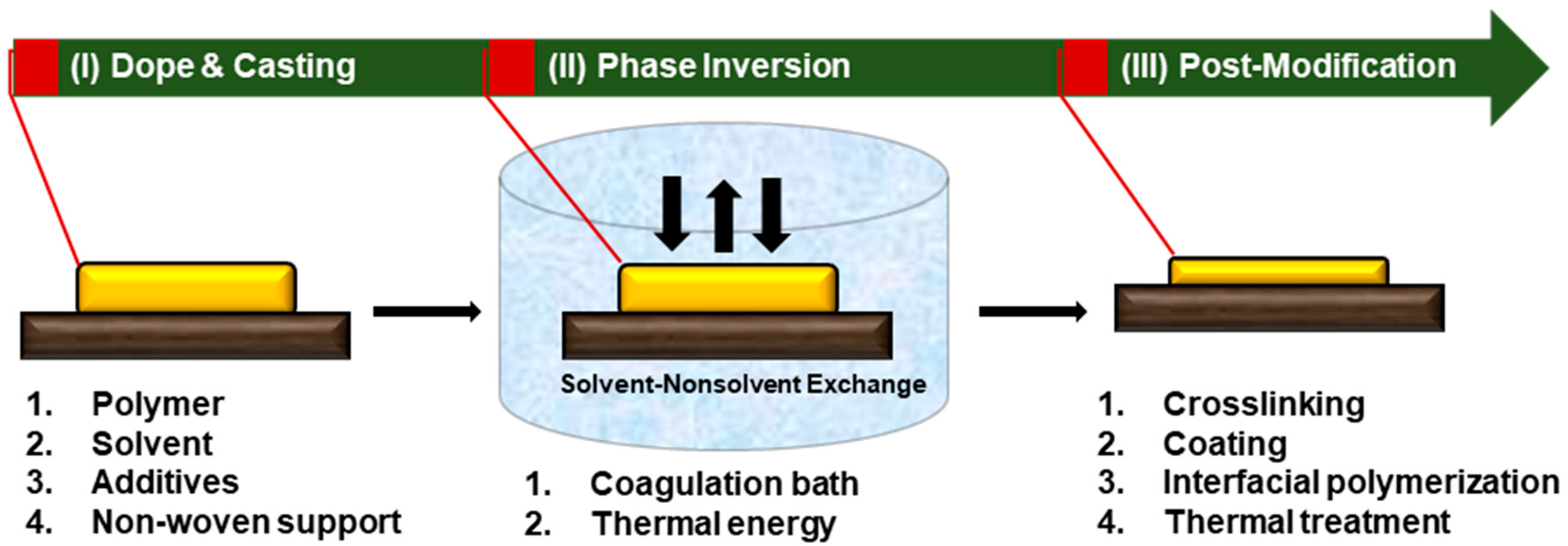
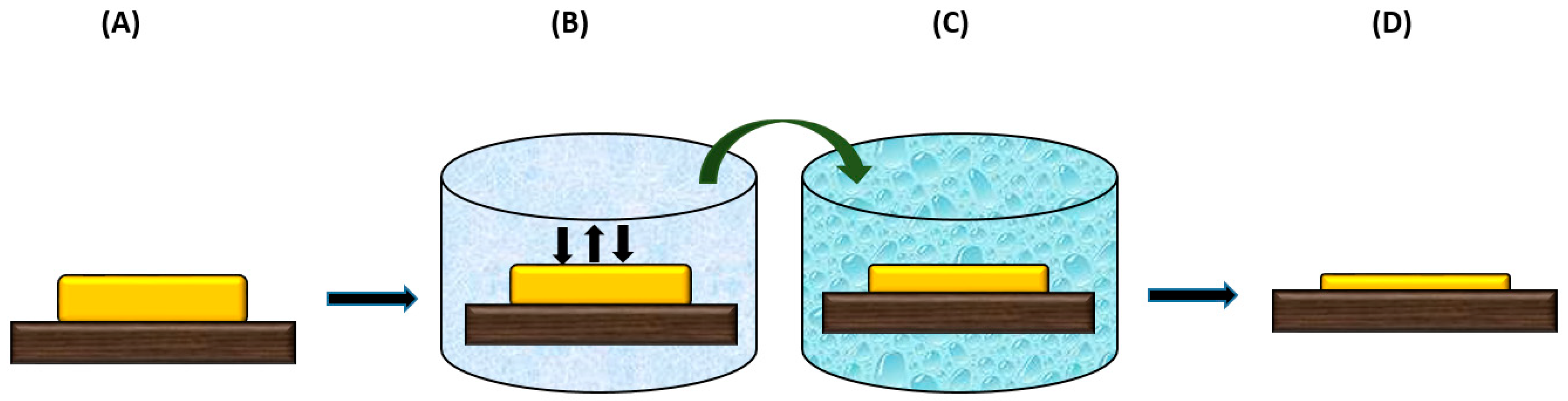
4. Sustainability for Membrane Fabrication Procedure
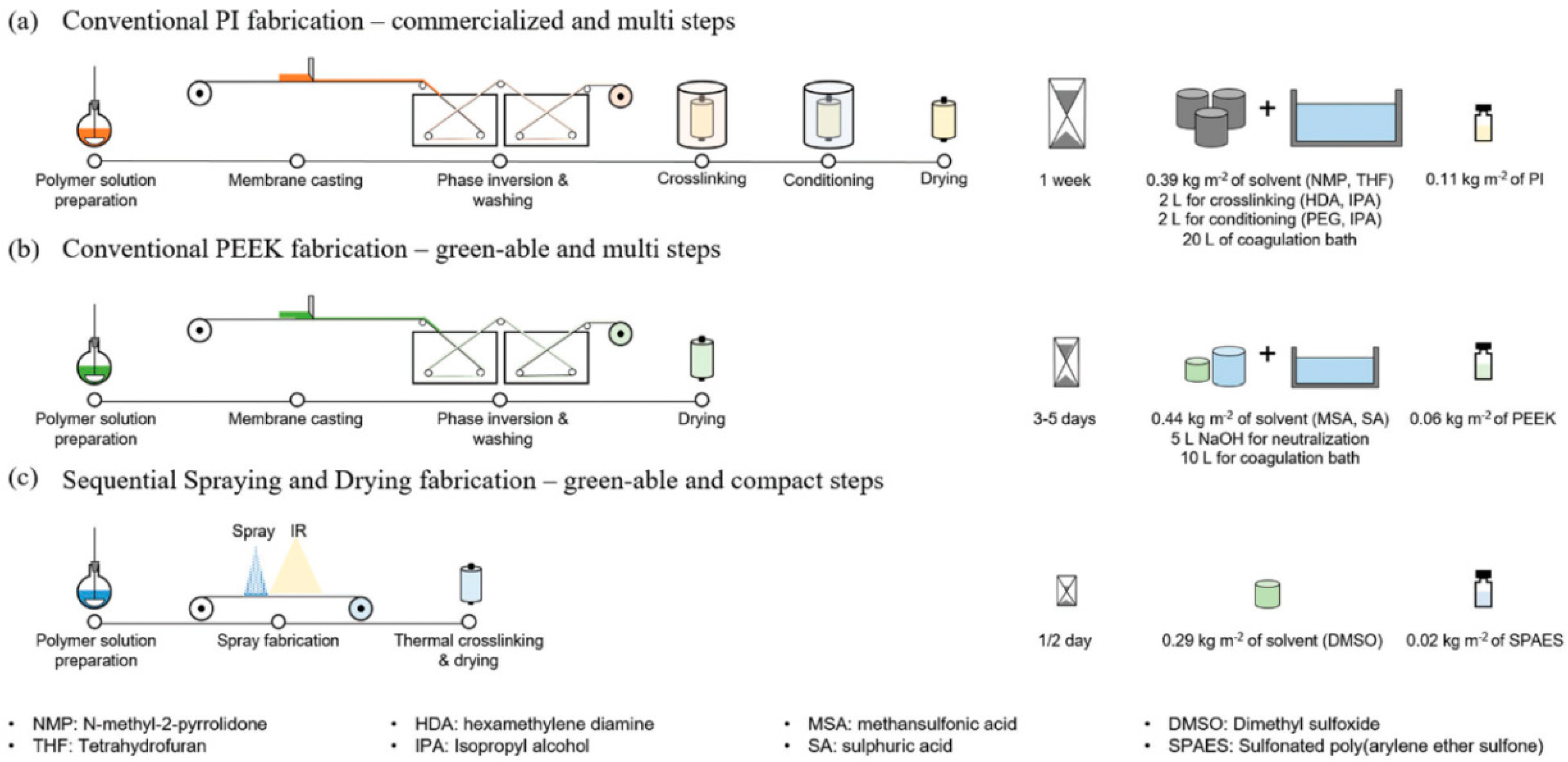
4.1. Minimizing the Number of Fabrication Steps and Materials
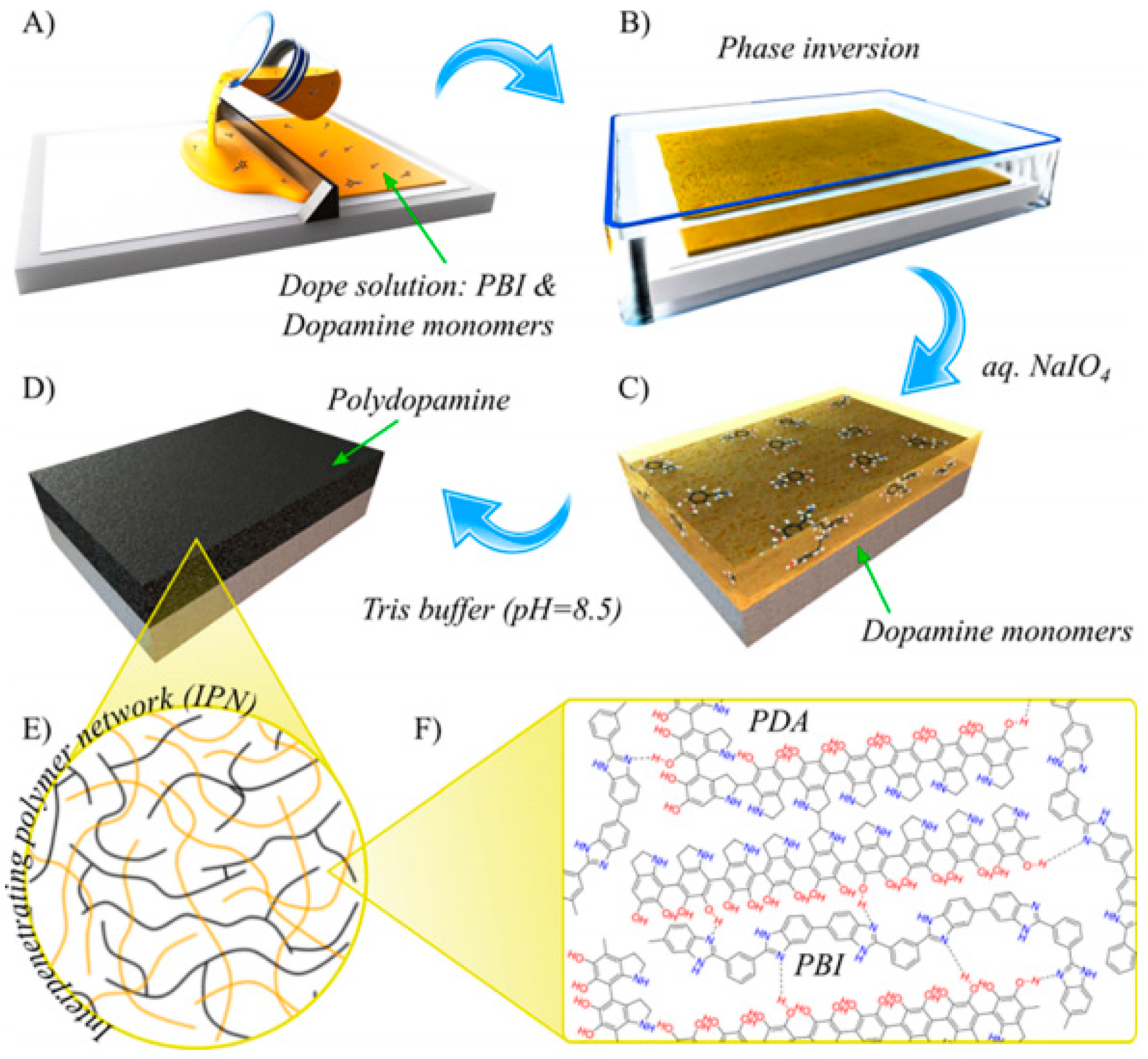
4.2. Sustainable Post-Modification
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| Abbreviation | Full Name |
| (EMIM)(Ac) | 1-ethyl-3-methylimidazolium acetate |
| (EMIM)(Cl) | 1,3-dimethylimidazolium chloride |
| (EMIM)(DEP) | 1-ethyl-3-ethylimidazolium diethyl phosphate |
| (EMIM)OAc | 1-ethyl-3-methylimidazolium acetate |
| AGET | Activator generated by electron transfer |
| ATBC | Acetyl tributyl citrate |
| ATRP | Atom transfer radical polymerization |
| CTA | Cellulose triacetate |
| DCMD | Direct contact membrane distillation |
| DES | Deep eutectic solvents |
| DFT | Discrete Fourier transform |
| DGE | Diglycidyl ether |
| DMC | Dimethyl carbonate |
| DMF | Dimethylformamide |
| DMSO | Dimethyl sulfoxide |
| DMSO2 | Dimethyl sulfone |
| EIPS | Evaporation induced phase separation |
| EU | Europe |
| GVL | γ-Valerolactone |
| HPC | Hydroxypropyl cellulose |
| IL | Ionic liquid |
| IP | Interfacial polymerization |
| LCST | Lower critical solution temperature |
| LDPE | Low-density polyethylene |
| MF | Microfiltration |
| MOF | Metal-organic frameworks |
| MPD | m-phenylenediamine |
| MW | Molecular weight |
| NaAlg | Sodium alginate |
| NF | Nanofiltration |
| NIPS | Non-solvent induced phase separation |
| NMP | N-methylpyrrolidone |
| N-TIPS | NIPS-TIPS |
| OSN | Organic Solvent Nanofiltration |
| PA | Polyamide |
| PAE | Poly(arylene ether sulfone) |
| PAES | Poly(arylene ether sulfone) |
| PAN | Polyacrylonitrile |
| PBI | Polybenzimidazole |
| PDA | Polydopamine |
| PE | Polyethylene |
| PEEK | Poly(ether ether ketone) |
| PEG | Poly(ethylene glycol) |
| PEI | Poly(ethylene imine) |
| PES | Polyethersulfone |
| PES-TA | Tertiary amine groups |
| PI | Polyimide |
| PLA | Polylactide |
| PP | Polypropylene |
| PS | Polystyrene |
| PSU | Polysulfone |
| PVDF-HFP | Poly(vinylidene fluoride-hexafluoropropylene) |
| PVP | Polyvinylpyrrolidone |
| RIPS | Reaction induced phase separation |
| SDS | Sodium dodecyl sulfate |
| SEM | Scanning electron microscopy |
| ST | Sodium tartrate |
| TA | Tannic acid |
| TEP | Triethyl-phosphate |
| TFN | Thin-film nanocomposite |
| THF | Tetrahydrofuran |
| TIPS | Thermally induced phase separation |
| TMC | Trimesoyl chloride |
| UF | Ultrafiltration |
| VIPS | Vapor-induced phase separation |
References
- Marchetti, P.; Solomon, M.F.J.; Szekely, G.; Livingston, A.G. Molecular Separation with Organic Solvent Nanofiltration: A Critical Review. Chem. Rev. 2014, 114, 10735–10806. [Google Scholar] [CrossRef]
- Yang, W.-J.; Shao, D.-D.; Zhou, Z.; Xia, Q.-C.; Chen, J.; Cao, X.-L.; Zheng, T.; Sun, S.-P. Carbon quantum dots (CQDs) nanofiltration membranes towards efficient biogas slurry valorization. Chem. Eng. J. 2020, 385, 123993. [Google Scholar] [CrossRef]
- Szekely, G.; Jimenez-Solomon, M.F.; Marchetti, P.; Kim, J.F.; Livingston, A.G. Sustainability assessment of organic solvent nanofiltration: From fabrication to application. Green Chem. 2014, 16, 4440–4473. [Google Scholar] [CrossRef]
- Kim, J.F.; Gaffney, P.R.J.; Valtcheva, I.B.; Williams, G.; Buswell, A.M.; Anson, M.S.; Livingston, A.G. Organic Solvent Nanofiltration (OSN): A New Technology Platform for Liquid-Phase Oligonucleotide Synthesis (LPOS). Org. Process. Res. Dev. 2016, 20, 1439–1452. [Google Scholar] [CrossRef]
- Figoli, A.; Marino, T.; Simone, S.; Di Nicolò, E.; Li, X.-M.; He, T.; Tornaghi, S.; Drioli, E. Towards non-toxic solvents for membrane preparation: A review. Green Chem. 2014, 16, 4034–4059. [Google Scholar] [CrossRef]
- Zhu, J.; Zhang, Q.; Zheng, J.; Hou, S.; Mao, H.; Zhang, S. Green fabrication of a positively charged nanofiltration membrane by grafting poly(ethylene imine) onto a poly (arylene ether sulfone) membrane containing tertiary amine groups. J. Membr. Sci. 2016, 517, 39–46. [Google Scholar] [CrossRef]
- Muntha, S.T.; Kausar, A.; Siddiq, M. Advances in Polymeric Nanofiltration Membrane: A Review. Polym. Technol. Eng. 2016, 56, 841–856. [Google Scholar] [CrossRef]
- Gohil, J.M.; Choudhury, R.R. Chapter 2—Introduction to Nanostructured and Nano-enhanced Polymeric Membranes: Preparation, Function, and Application for Water Purification. In Nanoscale Materials in Water Purification; Thomas, S., Pasquini, D., Leu, S.-Y., Gopakumar, D.A., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 25–57. [Google Scholar]
- Zhu, Y.; Wang, Z.; Zhang, C.; Wang, J.; Wang, S. A Novel Membrane Prepared from Sodium Alginate Cross-linked with Sodium Tartrate for CO2 Capture. Chin. J. Chem. Eng. 2013, 21, 1098–1105. [Google Scholar] [CrossRef]
- Jung, J.T.; Kim, J.F.; Wang, H.H.; Di Nicolo, E.; Drioli, E.; Lee, Y.M. Understanding the non-solvent induced phase separation (NIPS) effect during the fabrication of microporous PVDF membranes via thermally induced phase separation (TIPS). J. Membr. Sci. 2016, 514, 250–263. [Google Scholar] [CrossRef]
- Kim, J.F.; Jung, J.T.; Wang, H.H.; Lee, S.Y.; Moore, T.; Sanguineti, A.; Drioli, E.; Lee, Y.M. Microporous PVDF membranes via thermally induced phase separation (TIPS) and stretching methods. J. Membr. Sci. 2016, 509, 94–104. [Google Scholar] [CrossRef]
- Song, Y.J.; Kim, J.H.; Kim, Y.S.; Kim, S.D.; Cho, Y.H.; Park, H.S.; Nam, S.E.; Park, Y.I.; Son, E.H.; Kim, J.F. Controlling the Morphology of Polyvinylidene-co-hexafluoropropylene (PVDF-co-HFP) Membranes Via Phase Inversion Method. Membr. J. 2018, 28, 187–195. [Google Scholar] [CrossRef]
- Kim, J. Recent Progress on Improving the Sustainability of Membrane Fabrication. J. Membr. Sci. Res. 2019, 6, 241–250. [Google Scholar]
- Wang, H.H.; Jung, J.T.; Kim, J.F.; Kim, S.; Drioli, E.; Lee, Y.M. A novel green solvent alternative for polymeric membrane preparation via nonsolvent-induced phase separation (NIPS). J. Membr. Sci. 2019, 574, 44–54. [Google Scholar] [CrossRef]
- Dong, X.; Jeong, T.J.; Kline, E.; Banks, L.; Grulke, E.; Harris, T.; Escobar, I.C. Eco-friendly solvents and their mixture for the fabrication of polysulfone ultrafiltration membranes: An investigation of doctor blade and slot die casting methods. J. Membr. Sci. 2020, 614, 118510. [Google Scholar] [CrossRef]
- Galiano, F.; Briceño, K.; Marino, T.; Molino, A.; Christensen, K.V.; Figoli, A. Advances in biopolymer-based membrane preparation and applications. J. Membr. Sci. 2018, 564, 562–586. [Google Scholar] [CrossRef]
- Sheldon, R.A. Green and sustainable manufacture of chemicals from biomass: State of the art. Green Chem. 2014, 16, 950–963. [Google Scholar] [CrossRef]
- Aburabie, J.H.; Puspasari, T.; Peinemann, K.-V. Alginate-based membranes: Paving the way for green organic solvent nanofiltration. J. Membr. Sci. 2020, 596, 117615. [Google Scholar] [CrossRef]
- Aburabie, J.; Villalobos, L.F.; Peinemann, K.-V. Composite Membrane Formation by Combination of Reaction-Induced and Nonsolvent-Induced Phase Separation. Macromol. Mater. Eng. 2017, 302, 1700131. [Google Scholar] [CrossRef]
- Lackner, M.B. Kirk-Othmer Encyclopedia of Chemical Technology; Wiley & Sons: New York, NY, USA, 2015; pp. 1–41. [Google Scholar]
- Le Phuong, H.A.; Ayob, N.A.I.; Blanford, C.F.; Rawi, N.F.M.; Szekely, G. Nonwoven Membrane Supports from Renewable Resources: Bamboo Fiber Reinforced Poly(Lactic Acid) Composites. ACS Sustain. Chem. Eng. 2019, 7, 11885–11893. [Google Scholar] [CrossRef]
- Fan, L.; Ma, Y.; Su, Y.; Zhang, R.; Liu, Y.; Zhang, Q.; Jiang, Z. Green coating by coordination of tannic acid and iron ions for antioxidant nanofiltration membranes. RSC Adv. 2015, 5, 107777–107784. [Google Scholar] [CrossRef]
- Rasool, M.A.; Vankelecom, I.F. Use of γ-valerolactone and glycerol derivatives as bio-based renewable solvents for membrane preparation. Green Chem. 2019, 21, 1054–1064. [Google Scholar] [CrossRef]
- Rasool, M.A.; Van Goethem, C.; Vankelecom, I.F. Green preparation process using methyl lactate for cellulose-acetate-based nanofiltration membranes. Sep. Purif. Technol. 2020, 232, 115903. [Google Scholar] [CrossRef]
- Milescu, R.A.; McElroy, C.R.; Farmer, T.J.; Williams, P.M.; Walters, M.J.; Clark, J.H. Fabrication of PES/PVP Water Filtration Membranes Using Cyrene®, a Safer Bio-Based Polar Aprotic Solvent. Adv. Polym. Technol. 2019, 2019, 1–15. [Google Scholar] [CrossRef]
- Jung, J.T.; Wang, H.H.; Kim, J.F.; Lee, J.; Kim, J.S.; Drioli, E.; Lee, Y.M. Tailoring nonsolvent-thermally induced phase separation (N-TIPS) effect using triple spinneret to fabricate high performance PVDF hollow fiber membranes. J. Membr. Sci. 2018, 559, 117–126. [Google Scholar] [CrossRef]
- Fadhil, S.; Marino, T.; Makki, H.F.; Alsalhy, Q.F.; Blefari, S.; Macedonio, F.; Di Nicolò, E.; Giorno, L.; Drioli, E.; Figoli, A. Novel PVDF-HFP flat sheet membranes prepared by triethyl phosphate (TEP) solvent for direct contact membrane distillation. Chem. Eng. Process. Process. Intensif. 2016, 102, 16–26. [Google Scholar] [CrossRef]
- Falca, G.; Musteata, V.-E.; Behzad, A.R.; Chisca, S.; Nunes, S.P. Cellulose hollow fibers for organic resistant nanofiltration. J. Membr. Sci. 2019, 586, 151–161. [Google Scholar] [CrossRef]
- Sukma, F.; Çulfaz-Emecen, P. Cellulose membranes for organic solvent nanofiltration. J. Membr. Sci. 2018, 545, 329–336. [Google Scholar] [CrossRef]
- Hanafia, A.; Faur, C.; Deratani, A.; Guenoun, P.; Garate, H.; Quemener, D.; Pochat-Bohatier, C.; Bouyer, D. Fabrication of novel porous membrane from biobased water-soluble polymer (hydroxypropylcellulose). J. Membr. Sci. 2017, 526, 212–220. [Google Scholar] [CrossRef]
- Liang, H.-Q.; Wu, Q.-Y.; Wan, L.-S.; Huang, X.-J.; Xu, Z.-K. Polar polymer membranes via thermally induced phase separation using a universal crystallizable diluent. J. Membr. Sci. 2013, 446, 482–491. [Google Scholar] [CrossRef]
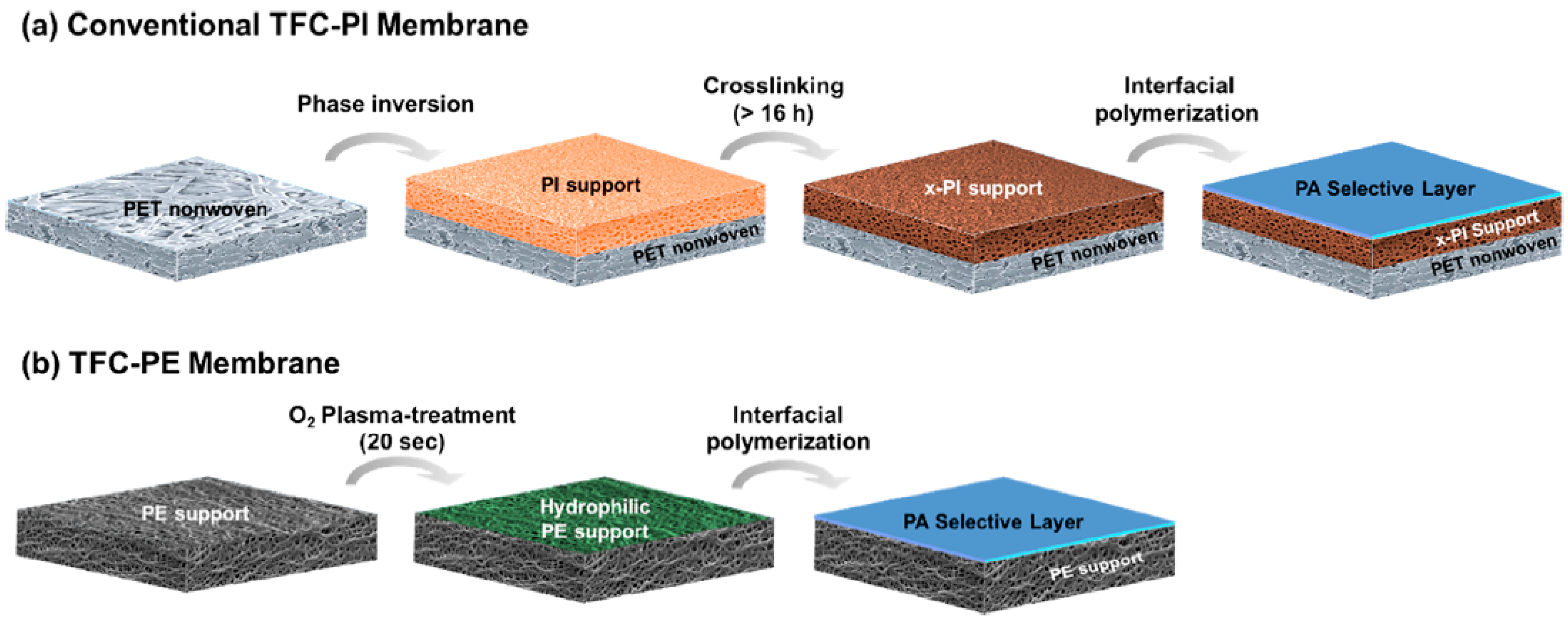
- Park, S.H.; Kim, Y.J.; Kwon, S.J.; Shin, M.G.; Nam, S.E.; Cho, Y.H.; Park, Y.I.; Kim, J.F.; Lee, J.H. Polyethylene Battery Separator as a Porous Support for Thin Film Composite Organic Solvent Nanofiltration Membranes. ACS Appl. Mater. Interfaces 2018, 10, 44050–44058. [Google Scholar] [CrossRef]
- Kim, J.H.; Cook, M.; Park, S.H.; Moon, S.J.; Kim, J.F.; Livingston, A.G.; Lee, Y.M. A compact and scalable fabrication method for robust thin film composite membranes. Green Chem. 2018, 20, 1887–1898. [Google Scholar] [CrossRef]
- Zhao, D.; Kim, J.F.; Ignacz, G.; Pogany, P.; Lee, Y.M.; Szekely, G. Bio-Inspired Robust Membranes Nanoengineered from Interpenetrating Polymer Networks of Polybenzimidazole/Polydopamine. ACS Nano 2019, 13, 125–133. [Google Scholar] [CrossRef]
- Fei, F.; Le Phuong, H.A.; Blanford, C.F.; Szekely, G. Tailoring the Performance of Organic Solvent Nanofiltration Membranes with Biophenol Coatings. ACS Appl. Polym. Mater. 2019, 1, 452–460. [Google Scholar] [CrossRef]
- Paseta, L.; Navarro, M.; Coronas, J.; Téllez, C. Greener processes in the preparation of thin film nanocomposite membranes with diverse metal-organic frameworks for organic solvent nanofiltration. J. Ind. Eng. Chem. 2019, 77, 344–354. [Google Scholar] [CrossRef]
- Byrne, F.P.; Jin, S.; Paggiola, G.; Petchey, T.H.M.; Clark, J.H.; Farmer, T.J.; Hunt, A.J.; McElroy, C.R.; Sherwood, J. Tools and techniques for solvent selection: Green solvent selection guides. Sustain. Chem. Process. 2016, 4, 1. [Google Scholar] [CrossRef]
- Alder, C.M.; Hayler, J.D.; Henderson, R.K.; Redman, A.M.; Shukla, L.; Shuster, L.E.; Sneddon, H.F. Updating and further expanding GSK’s solvent sustainability guide. Green Chem. 2016, 18, 3879–3890. [Google Scholar] [CrossRef]
- Henderson, R.K.; Jiménez-González, C.; Constable, D.J.; Alston, S.R.; Inglis, G.G.; Fisher, G.; Sherwood, J.; Binks, S.P.; Curzons, A.D. Expanding GSK’s Solvent Selection Guide—Embedding sustainability into solvent selection starting at Medicinal Chemistry. Green Chem. 2011, 13, 854–862. [Google Scholar] [CrossRef]
- Keçili, R.; Yılmaz, E.; Ersöz, A.; Say, R. Chapter 12—Imprinted Materials: From Green Chemistry to Sustainable Engineering. In Sustainable Nanoscale Engineering; Szekely, G., Livingston, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 317–350. [Google Scholar]
- Kim, J.; Jung, J.; Wang, H.; Drioli, E.; Lee, Y.M. 1.15 Effect of Solvents on Membrane Fabrication via Thermally Induced Phase Separation (TIPS): Thermodynamic and Kinetic Perspectives. In Comprehensive Membrane Science and Engineering; Elsevier: Amsterdam, The Netherlands, 2017; pp. 386–417. [Google Scholar]
- Gu, Y.; Jérôme, F. Bio-based solvents: An emerging generation of fluids for the design of eco-efficient processes in catalysis and organic chemistry. Chem. Soc. Rev. 2013, 42, 9550–9570. [Google Scholar] [CrossRef]
- Shen, X.; Xia, D.; Xiang, Y.; Gao, J. γ-valerolactone (GVL) as a bio-based green solvent and ligand for iron-mediated AGET ATRP. e-Polymers 2019, 19, 323–329. [Google Scholar] [CrossRef]
- Häckl, K.; Kunz, W. Some aspects of green solvents. C. R. Chim. 2018, 21, 572–580. [Google Scholar] [CrossRef]
- Pereira, C.S.M.; Silva, V.M.T.M.; Rodrigues, A.E. Ethyl lactate as a solvent: Properties, applications and production processes—A review. Green Chem. 2011, 13, 2658–2671. [Google Scholar] [CrossRef]
- Zare, K. Characterization and synthesis of methyl lactate through esterification reaction under supercritical state of carbon dioxide. Res. J. Eng. 2018, 7, 1–4. [Google Scholar]
- Sherwood, J.; De Bruyn, M.; Constantinou, A.; Moity, L.; McElroy, C.R.; Farmer, T.J.; Duncan, T.; Raverty, W.; Hunt, A.J.; Clark, J.H. Dihydrolevoglucosenone (Cyrene) as a bio-based alternative for dipolar aprotic solvents. Chem. Commun. 2014, 50, 9650–9652. [Google Scholar] [CrossRef]
- Marino, T.; Galiano, F.; Molino, A.; Figoli, A. New frontiers in sustainable membrane preparation: Cyrene™ as green bioderived solvent. J. Membr. Sci. 2019, 580, 224–234. [Google Scholar] [CrossRef]
- Randová, A.; Bartovská, L.; Morávek, P.; Matějka, P.; Novotná, M.; Matějková, S.; Drioli, E.; Figoli, A.; Lanč, M.; Friess, K. A fundamental study of the physicochemical properties of Rhodiasolv®Polarclean: A promising alternative to common and hazardous solvents. J. Mol. Liq. 2016, 224, 1163–1171. [Google Scholar] [CrossRef]
- Marino, T.; Blasi, E.; Tornaghi, S.; Di Nicolò, E.; Figoli, A. Polyethersulfone membranes prepared with Rhodiasolv®Polarclean as water soluble green solvent. J. Membr. Sci. 2018, 549, 192–204. [Google Scholar] [CrossRef]
- Abdellah, M.; Pérez-Manriquez, L.; Puspasari, T.; Scholes, C.A.; Kentish, S.E.; Peinemann, K.-V. Effective Interfacially Polymerized Polyester Solvent Resistant Nanofiltration Membrane from Bioderived Materials. Adv. Sustain. Syst. 2018, 2, 1800043. [Google Scholar] [CrossRef]
- Marino, T.; Blefari, S.; Di Nicolò, E.; Figoli, A. A more sustainable membrane preparation using triethyl phosphate as solvent. Green Process. Synth. 2017, 6, 295–300. [Google Scholar] [CrossRef]
- Esmaeili, M.; Anugwom, I.; Mänttäri, M.; Kallioinen, M. Utilization of DES-Lignin as a Bio-Based Hydrophilicity Promoter in the Fabrication of Antioxidant Polyethersulfone Membranes. Membranes 2018, 8, 80. [Google Scholar] [CrossRef]
- Dsouza, S.A.; Pereira, M.M.; Polisetti, V.; Mondal, D.; Nataraj, S.K. Introducing deep eutectic solvents as flux boosting and surface cleaning agents for thin film composite polyamide membranes. Green Chem. 2020, 22, 2381–2387. [Google Scholar] [CrossRef]
- Ajari, H.; Chaouachi, B.; Galiano, F.; Marino, T.; Russo, F.; Figoli, A. A novel approach for dissolving crystalline LDPE using non-toxic solvents for membranes preparation. Int. J. Environ. Sci. Technol. 2018, 16, 5375–5386. [Google Scholar] [CrossRef]
- Karan, S.; Jiang, Z.; Livingston, A.G. Sub-10 nm polyamide nanofilms with ultrafast solvent transport for molecular separation. Science 2015, 348, 1347–1351. [Google Scholar] [CrossRef] [PubMed]
- Solomon, M.F.J.; Bhole, Y.; Livingston, A.G. High flux membranes for organic solvent nanofiltration (OSN)—Interfacial polymerization with solvent activation. J. Membr. Sci. 2012, 423–424, 371–382. [Google Scholar] [CrossRef]
- Solomon, M.F.J.; Bhole, Y.; Livingston, A.G. High flux hydrophobic membranes for organic solvent nanofiltration (OSN)—Interfacial polymerization, surface modification and solvent activation. J. Membr. Sci. 2013, 434, 193–203. [Google Scholar] [CrossRef]
| No. | Selective Membrane Material | Support Material | Solvent for Fabrication | Testing Solvent | Marker (MW) | Permeance (L/m2·h·bar) | Highest Rejection (%) | Ref. |
|---|---|---|---|---|---|---|---|---|
| 1. | NaAlg | PAN | Water | Methanol | B12 (1355 g·mol−1) | 1.27 ± 0.2 | 98 ± 2 | [18] |
| 2. | NaAlg | Crosslinked PAN | Water | DMF | B12 (1355 g·mol−1) | 0.21 ± 0.1 | 98 ± 1 | [18] |
| 3. | NaAlg | Cellulose | Water | Methanol | B12 (1355 g·mol−1) | 0.38 ± 0.1 | 95 ± 1 | [18] |
| 4. | NaAlg | Alumina support | Water | Methanol | B12 (1355 g·mol−1) | 1.6 ± 0.1 | 90± 2 | [18] |
| 5. | NaAlg | Alumina support | Water | DMF | B12 (1355 g·mol−1) | 0.25 ± 0.02 | 70 ± 1 | [18] |
| 6. | NaAlg | Alumina support | Water | DMSO | B12 (1355 g·mol−1) | 0.15 ± 0.05 | 76 ± 2 | [18] |
| 7. | NaAlg | Alumina support | Water | NMP | B12 (1355 g·mol−1) | 0.11 ± 0.03 | 80 ± 3 | [18] |
| 8. | PBI | Bamboo fiber/PLA | DMC, DMAc | Water | 1068 ± 32 | [21] | ||
| 9. | CTA | Polypropylene nonwoven | GVL | Water | Rose Bengal (1017 g·mol−1) | >90 | [23] | |
| 10 | CA | Polypropylene nonwoven | Methyl lactate | Water | Rose Bengal (1017 g·mol−1) | >90 | [23] | |
| 11. | CA | Polypropylene nonwoven | GVL | Water | Rose Bengal (1017 g·mol−1) | >90 | [23] | |
| 12. | CA | Polypropylene nonwoven | Methyl lactate 2-methyl THF | Water | Rose Bengal (1017 g·mol−1) | 2.4 | 99.5 | [24] |
| 13. | PES | Polyester nonwoven | Cyrene | Water | 2542.7 | [25] | ||
| 14. | PES | Polyester nonwoven | Cyrene | Water | 898.4 | [25] | ||
| 15. | PVDF | PolarClean | Water | 3000 | [10,24] | |||
| 16. | PVDF | PolarClean | Water | Polystyrene | 99.99 | [26] | ||
| 17. | PVDF-HFP | TEP | Distilled water | NaCl | 16.1 | 99.3 | [27] | |
| 18. | Cellulose | ((EMIM)(DEP)) | Ethanol | Congo Red (696 g·mol−1) | 19 ± 1 | >90 | [28] | |
| 19. | Cellulose | ((EMIM)(DEP)) | Water | Congo Red (696 g·mol−1) | 48 ± 3 | >99 | [28] | |
| 20. | Cellulose | (EMIM)OAc | Ethanol | Bromothymol Blue (624.4 g·mol−1) | 0.3 | 94 | [29] | |
| 21. | Cellulose | (EMIM)OAc/Acetone | Ethanol | Bromothymol Blue (624.4 g·mol−1) | 8.4 | 69.8 | [29] | |
| 22. | TA/Fe3+ | PES | Water | Water | Orange GII (452.4 g·mol−1) | 45.6 | 94.8 | [22] |
| 23. | TA/Fe3+ | PES | Water | Water | Orange GII (452.4 g·mol−1) | 34.3 | 95.5 | [22] |
| 24. | HPC | Water | Water | 3 ± 0.2 − 38 ± 5 | [30] | |||
| 25. | PVDF | ATBC | Water | 538 | [11] | |||
| 26. | PVDF | DMSO2 | Water | 1491 | [31] | |||
| 27. | PA | PE battery separator | Hexane, Water | Acetone | Styrene oligomer (~1000 g·mol−1) | ~20 | >99 | [32] |
| 28. | PAES | Porous substrate(PE separator, TR-NFM, and PET nonwoven) | DMSO | DMF | Styrene oligomer (1595 g·mol−1) | 0.37 ± 0.018 | >99 | [33] |
| 29. | PAES, PES-TA | PAES membrane with PES-TA group | PEGDGE 10% wt aqueous solutions, PEI% wt solution | Water | Methyl violet (407.979 g·mol−1) | 15.5 | 99.8 | [6] |
| 30. | PBI/PDA | Polypropylene | DMAc, Water | DMF | Polystyrene (610 g·mol−1) | 12 | ~100 | [34] |
| 31. | Bio-phenol coated PI | PI membrane | Water:EtOH | Acetone | Polystyrene (390–1550 g·mol−1) | 1–10 | 5~100 | [35] |
| 32. | PI | Crosslinked PI membrane | DMSO | Methanol | Sunset Yellow (452 g·mol−1) | 11 | 93 | [36] |
| No. | Solvent | Chemical Structure | Soluble Polymer | Sustainable Characteristics | Ref. |
|---|---|---|---|---|---|
| 1. | Water |  | HPC | An inorganic, transparent, tasteless, odorless and nearly colorless chemical substance with low risk of hazard, low cost and high availability | [30] |
| 2. | γ-Valerolactone (GVL) | 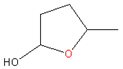 | PI, PES, CA, CTA, PSU | Colorless liquid, bio-based green solvent, low toxicity and miscible with water, stability under neutral media | [23] |
| 3. | Methyl lactate | 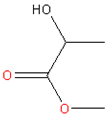 | CA, cellulose derivatives | Colorless and clear substance with a peculiar odor, high boiling point and slow volatility rate with biodegradability, water miscibility, noncorrosive, noncarcinogenic, and non-ozone-depleting features | [23,24] |
| 4. | Ethyl lactate |  | CA | Colorless, sweet smell and clear substance, low toxicity, and agreeable odor, water miscibility, noncorrosive, noncarcinogenic, and non-ozone-depleting features | [45] |
| 5. | Cyrene | 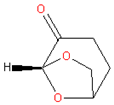 | PES, PVDF | Bio-based origin, clear colorless to light yellow liquid, mild, smoky ketone-like odor, with a comparatively high dynamic viscosity, comparable solvent properties to NMP without nitrogen or sulfur heteroatoms | [47,48] |
| 6. | Rhodiasolv® Polarclean | 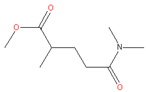 | PI, CA, PVDF | Clear, colorless to yellow liquid with slight odor, solubility in water, high solvency capability, eco-friendly sustainable solvent | [10,24,26] |
| 7. | Triethyl phosphate (TEP) | 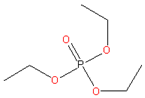 | CA, PVDF | Colorless, corrosive liquid, combustible, slowly dissolves in water and sinks in water, no components supposed of persistence, bio-accumulation, and toxicity or high persistence in the environment | [27] |
| 8. | Ionic Liquids (ILs) | 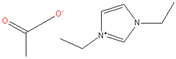 Ex) 1-ethyl-3-ethylimidazolium acetate | PAN, Cellulose, PES, CA | Low melting point, high thermal stability, low viscosity, low chemical reactivity, and negligible vapor pressure without flammability | [28,29] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen Thi, H.Y.; Nguyen, B.T.D.; Kim, J.F. Sustainable Fabrication of Organic Solvent Nanofiltration Membranes. Membranes 2021, 11, 19. https://doi.org/10.3390/membranes11010019
Nguyen Thi HY, Nguyen BTD, Kim JF. Sustainable Fabrication of Organic Solvent Nanofiltration Membranes. Membranes. 2021; 11(1):19. https://doi.org/10.3390/membranes11010019
Chicago/Turabian StyleNguyen Thi, Hai Yen, Bao Tran Duy Nguyen, and Jeong F. Kim. 2021. "Sustainable Fabrication of Organic Solvent Nanofiltration Membranes" Membranes 11, no. 1: 19. https://doi.org/10.3390/membranes11010019
APA StyleNguyen Thi, H. Y., Nguyen, B. T. D., & Kim, J. F. (2021). Sustainable Fabrication of Organic Solvent Nanofiltration Membranes. Membranes, 11(1), 19. https://doi.org/10.3390/membranes11010019





