In-Situ Combination of Bipolar Membrane Electrodialysis with Monovalent Selective Anion-Exchange Membrane for the Valorization of Mixed Salts into Relatively High-Purity Monoprotic and Diprotic Acids
Abstract
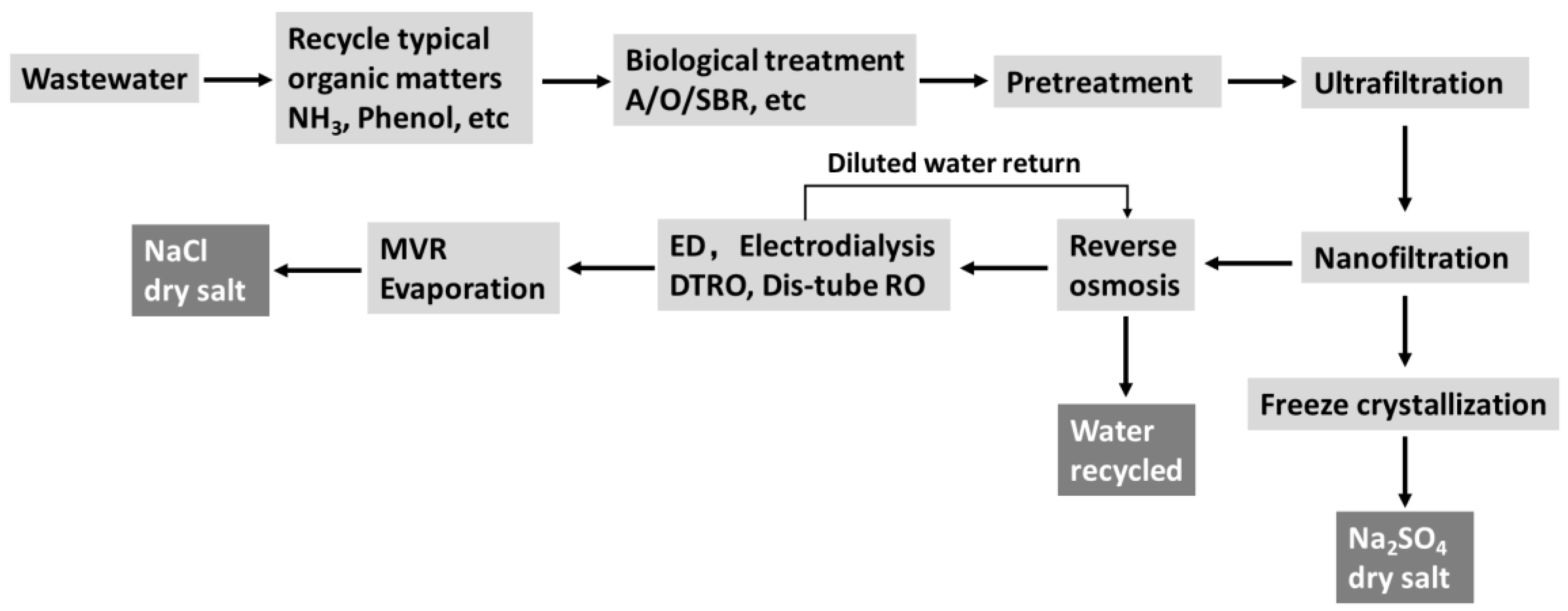
1. Introduction
2. Experimental Section
2.1. Material
2.2. Setup
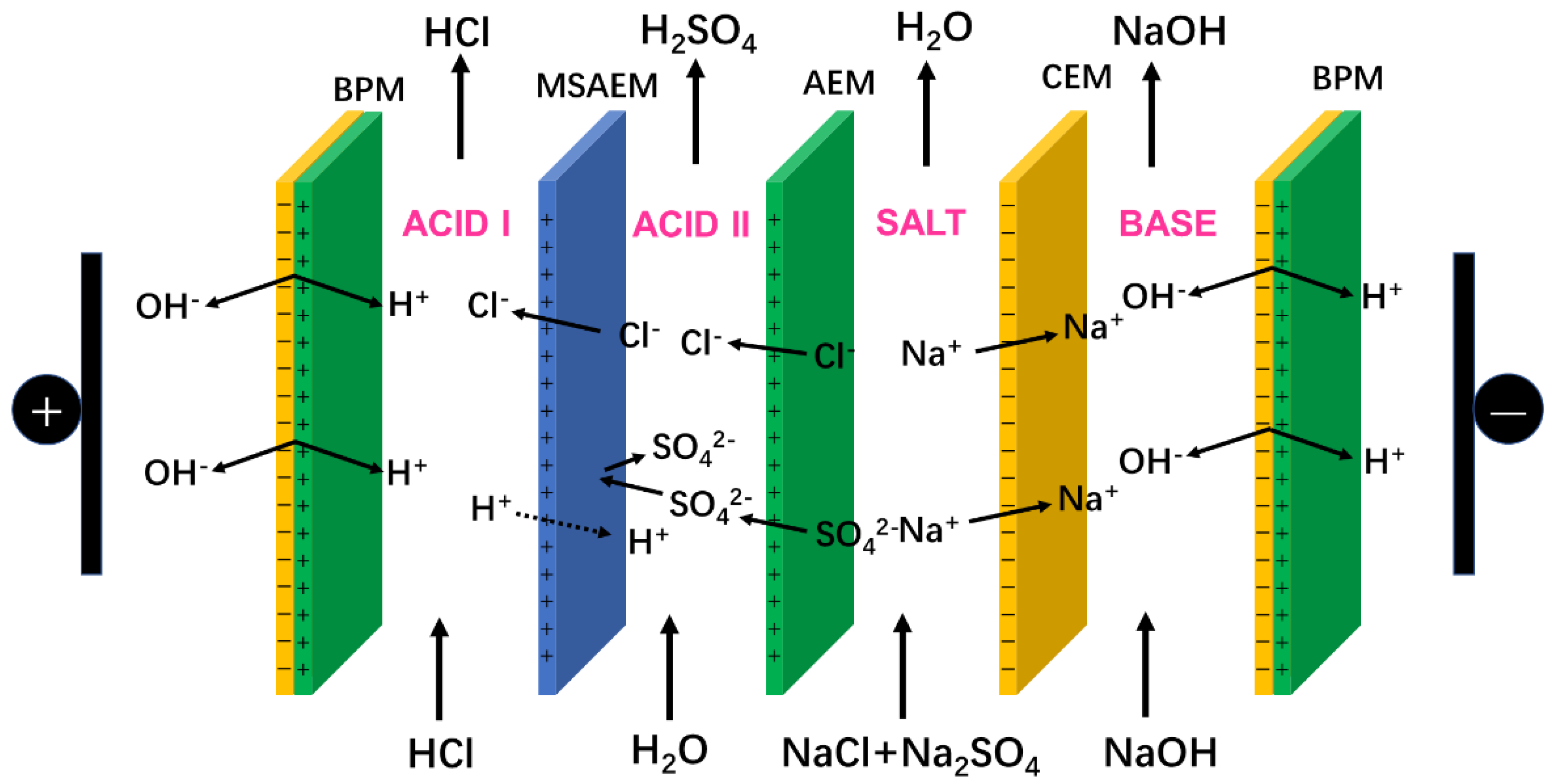
2.3. Operating Principle of the SBMED
2.4. Analyses and Calculations
3. Results and Discussion
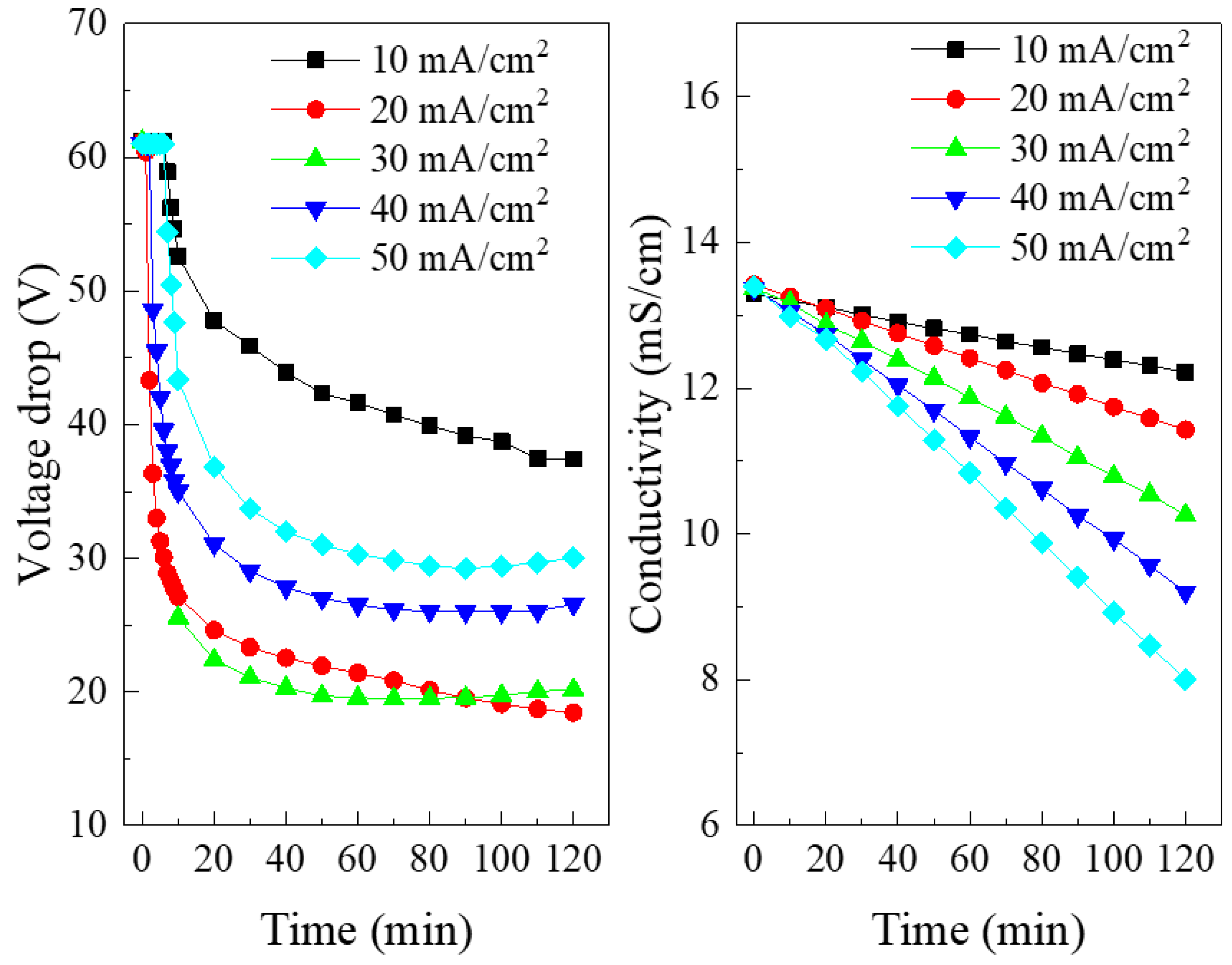
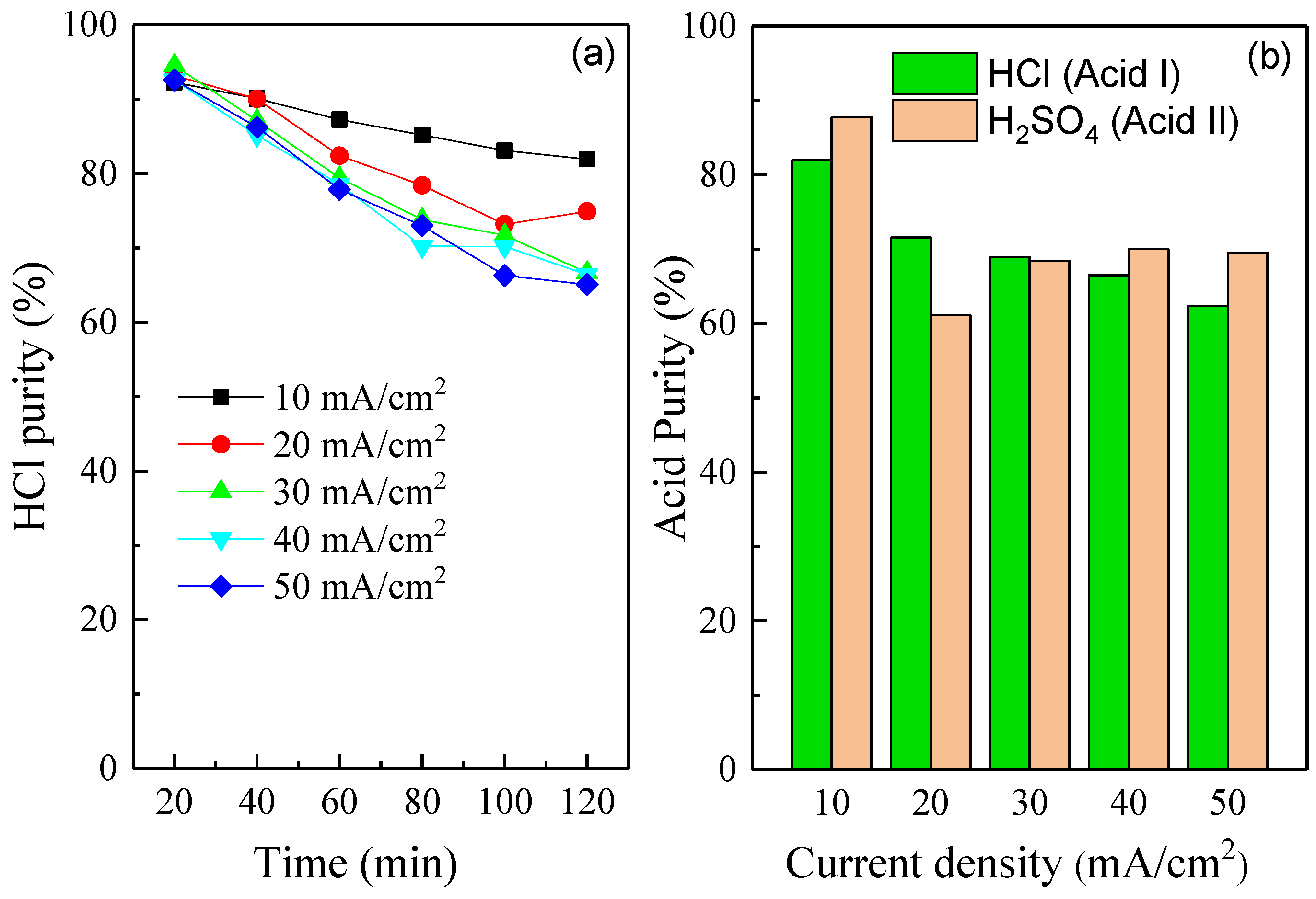
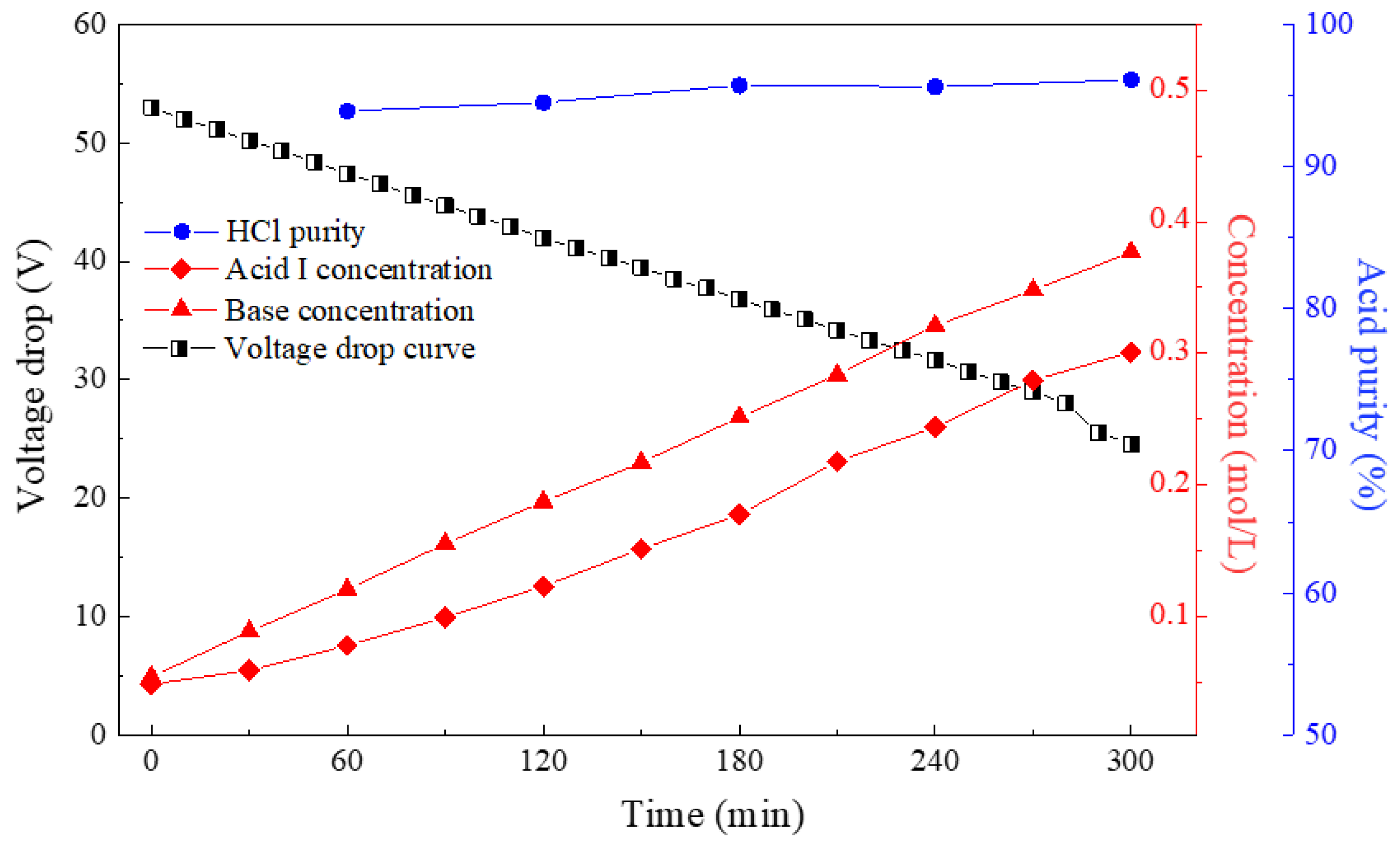
3.1. Effect of Current Density
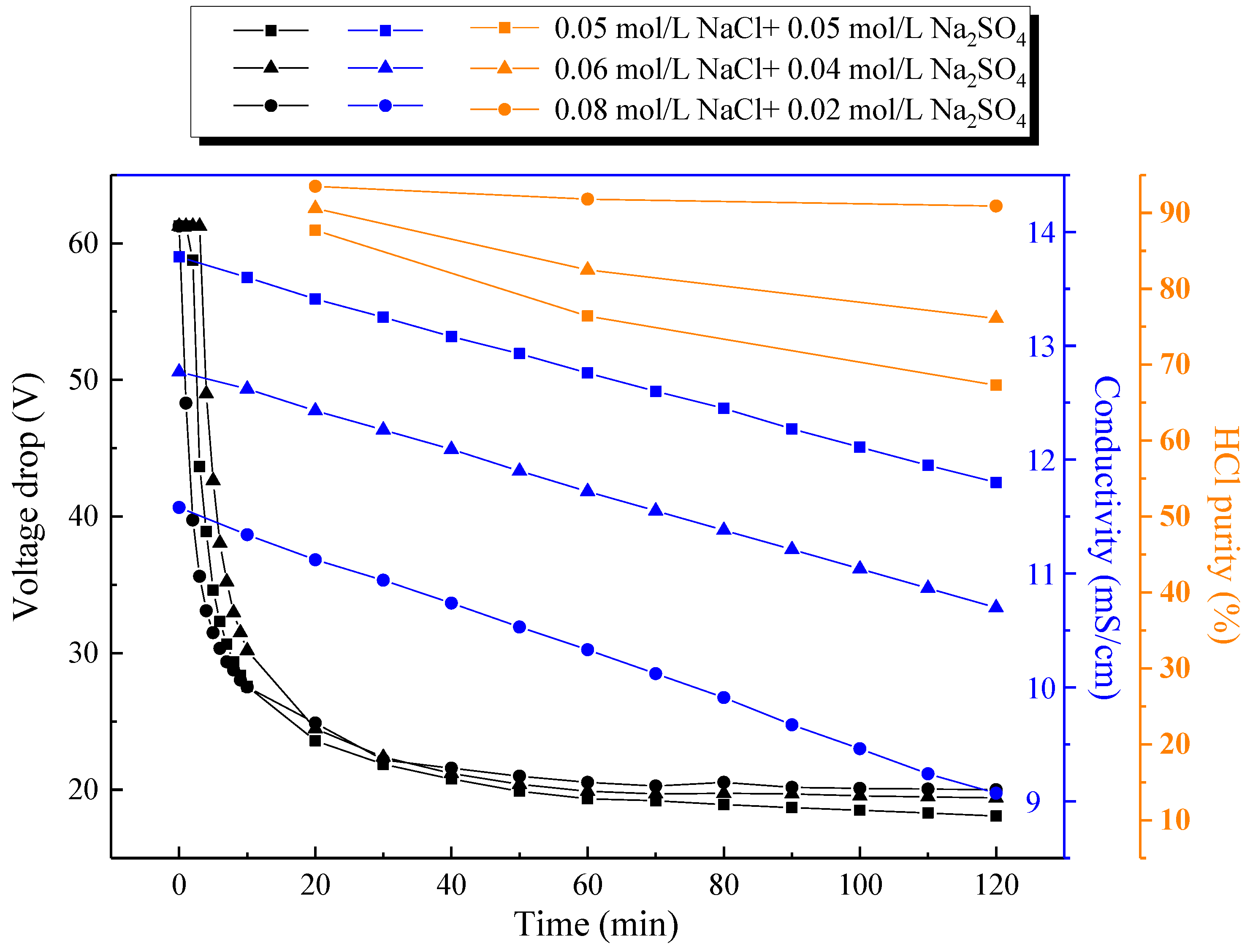
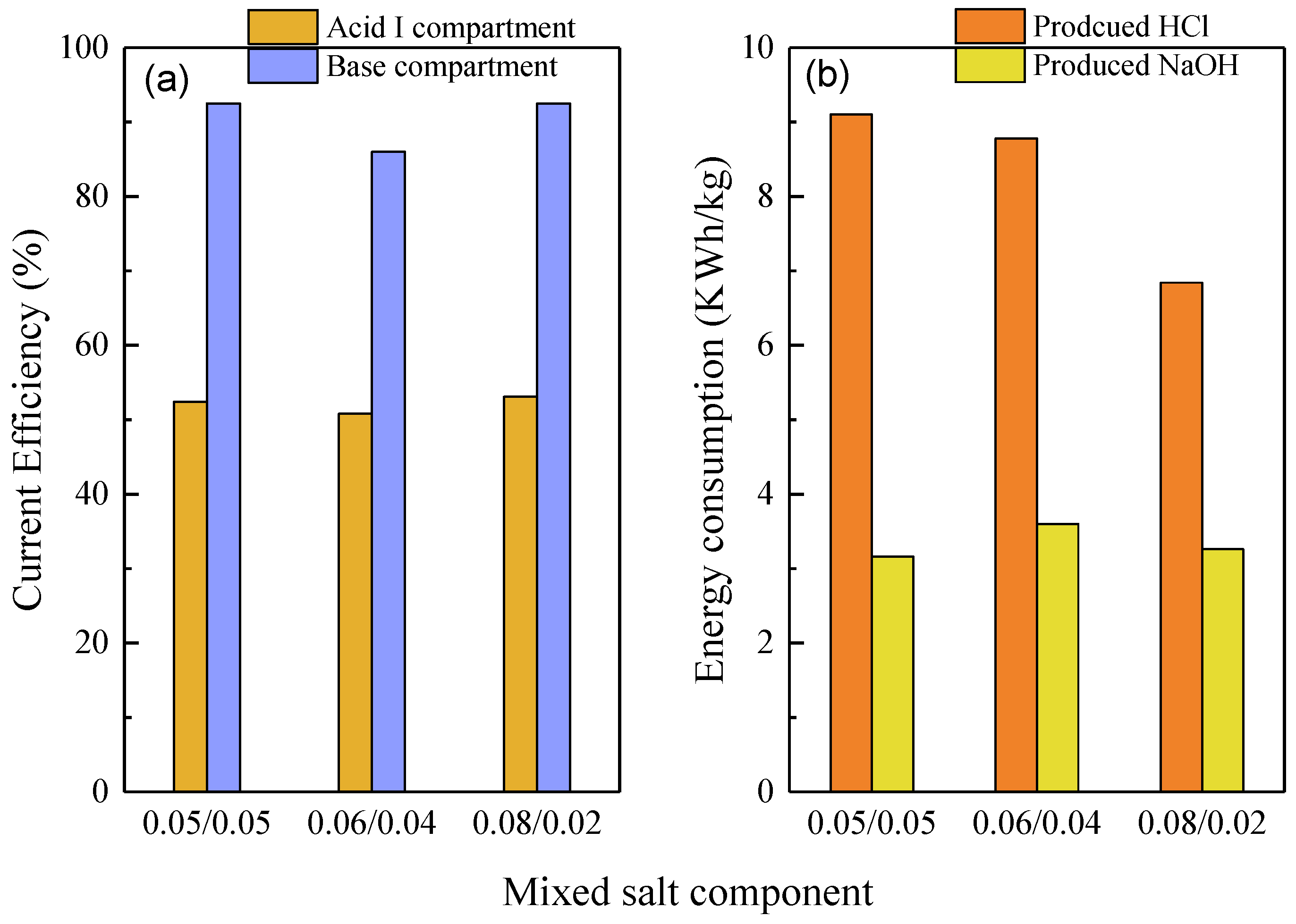
3.2. Effect of Monovalent and Divalent Salt Component
3.3. Simulated Seawater
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lefebvre, O.; Moletta, R. Treatment of organic pollution in industrial saline wastewater: A literature review. Water Res. 2006, 40, 3671–3682. [Google Scholar] [CrossRef]
- Tan, X.; Acquah, I.; Liu, H.; Li, W.; Tan, S. A critical review on saline wastewater treatment by membrane bioreactor (MBR) from a microbial perspective. Chemosphere 2019, 220, 1150–1162. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhuang, X.; Ahmad, S.; Sung, S.; Ni, S. Biotreatment of high-salinity wastewater: Current methods and future directions. World J. Microb. Biot. 2020, 36, 37. [Google Scholar] [CrossRef]
- Li, J.; Hu, S. History and future of the coal and coal chemical industry in China. Resour. Conserv. Recy. 2017, 124, 13–24. [Google Scholar] [CrossRef]
- Xie, K.; Li, W.; Zhao, W. Coal chemical industry and its sustainable development in China. Energy 2010, 35, 4349–4355. [Google Scholar] [CrossRef]
- Koppol, A.P.R.; Bagajewicz, M.J.; Dericks, B.J.; Savelski, M.J. On zero water discharge solutions in the process industry. Adv. Environ. Res. 2004, 8, 151–171. [Google Scholar] [CrossRef]
- Tong, T.; Elimelech, M. The global rise of zero liquid discharge for wastewater management: Drivers, technologies, and future directions. Environ. Sci. Technol. 2016, 50, 6846–6855. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Lv, W.; Xu, H. Modern Coal Mining Chemical Crystallization Technologies and Policies Analysis; China Petrochemical Press: Beijing, China, 2018; ISBN 9787511448330. [Google Scholar]
- Huang, C.; Xu, T. Electrodialysis with bipolar membranes for sustainable development. Environ. Sci. Technol. 2006, 40, 5233–5243. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, N.; Huang, C.; Xu, T. Production of monoprotic, diprotic, and triprotic organic acids by using electrodialysis with bipolar membranes: Effect of cell configurations. J. Membr. Sci. 2011, 385–386, 226–233. [Google Scholar] [CrossRef]
- Kumar, A.; Phillips, K.R.; Cai, J.; Schroder, U.; Lienhard, J.H. Integrated valorization of desalination brine through NaOH recovery: Opportunities and challenges. Angew. Chem. 2019, 131, 6570–6579. [Google Scholar] [CrossRef]
- Kumar, A.; Phillips, K.R.; Thiel, G.P.; Schröer, U.; Lienhard, J.H. Direct electrosynthesis of sodium hydroxide and hydrochloric acid from brine streams. Nat. Catal. 2019, 2, 106–113. [Google Scholar] [CrossRef]
- Wei, Y.; Li, C.; Wang, Y.; Zhang, X.; Li, Q.; Xu, T. Regenerating sodium hydroxide from the spent caustic by bipolar membrane electrodialysis (BMED). Sep. Purif. Technol. 2012, 86, 49–54. [Google Scholar] [CrossRef]
- Zhang, X.; Lu, W.; Ren, H.; Cong, W. Sulfuric acid and ammonia generation by bipolar membranes electrodialysis: Transport rate model for ion and water through anion exchange membrane. Chem. Biochem. Eng. 2008, 22, 1–8. [Google Scholar]
- Paleologou, M.; Thibault, A.; Wong, P.Y.; Thompson, R.; Berry, R. Enhancement of the current efficiency for sodium hydroxide production from sodium sulphate in a two-compartment bipolar membrane electrodialysis system. Sep. Purif. Technol. 1997, 11, 159–171. [Google Scholar] [CrossRef]
- Yang, Y.; Gao, X.; Fan, A.; Fu, L.; Gao, C. An innovative beneficial reuse of seawater concentrate using bipolar membrane electrodialysis. J. Membr. Sci. 2014, 449, 119–126. [Google Scholar] [CrossRef]
- Zhang, Y.; Paepen, S.; Pinoy, L.; Meesschaert, B.; Van der Bruggen, B. Selectrodialysis: Fractionation of divalent ions from monovalent ions in a novel electrodialysis stack. Sep. Purif. Technol. 2012, 88, 191–201. [Google Scholar] [CrossRef]
- Zhang, Y.; Desmidt, E.; Looveren, A.V.; Pinoy, L.; Meesschaert, B.; Van der Bruggen, B. Phosphate separation and recovery from wastewater by novel Electrodialysis. Environ. Sci. Technol. 2013, 47, 5888–5895. [Google Scholar] [CrossRef]
- Xu, P.; Capito, M.; Cath, T.Y. Selective removal of arsenic and monovalent ions from brackish water reverse osmosis concentrate. J. Harzard. Mater. 2013, 260, 885–891. [Google Scholar] [CrossRef]
- Cohena, B.; Lazarovitcha, N.; Gilronb, J. Upgrading groundwater for irrigation using monovalent selective Electrodialysis. Desalination 2018, 431, 126–139. [Google Scholar] [CrossRef]
- Zhang, W.; Miao, M.; Pan, J.; Sotto, A.; Shen, J.; Gao, C.; Van der Bruggen, B. Separation of divalent ions from seawater concentrate to enhance the purity of coarse salt by electrodialysis with monovalent-selective membranes. Desalination 2017, 411, 28–37. [Google Scholar] [CrossRef]
- Reig, M.; Valderrama, C.; Gibert, O.; Cortina, J.L. Selectrodialysis and bipolar membrane electrodialysis combination for industrial process brines treatment: Monovalent-divalent ions separation and acid and base production. Desalination 2016, 399, 88–95. [Google Scholar] [CrossRef]
- Qiu, Y.; Yao, L.; Tang, C.; Zhao, Y.; Zhu, J.; Shen, J. Integration of selectrodialysis and selectrodialysis with bipolar membrane to salt lake treatment for the production of lithium hydroxide. Desalination 2019, 465, 1–12. [Google Scholar] [CrossRef]
- Chen, B.; Jiang, C.; Wang, Y.; Fu, R.; Liu, Z.; Xu, T. Selectrodialysis with bipolar membrane for the reclamation of concentrated brine from RO plant. Desalination 2018, 442, 8–15. [Google Scholar] [CrossRef]
- Lorrain, Y.; Pourcelly, G.; Gavach, C. Influence of cations on the proton leakage through anion-exchange membranes. J. Membr. Sci. 1996, 110, 181–190. [Google Scholar] [CrossRef]
- Lorrain, Y.; Pourcelly, G.; Gavach, C. Transport mechanism of sulfuric acid through an anion exchange membrane. Desalination 1997, 109, 231–239. [Google Scholar] [CrossRef]
- Beck, A.; Ernst, M. Kinetic modeling and selectivity of anion exchange in Donnan dialysis. J. Membr. Sci. 2015, 479, 132–140. [Google Scholar] [CrossRef]
- McDonald, M.B.; Freund, M.S.; Hammond, P.T. Catalytic, conductive bipolar membrane interfaces through layer-by-layer deposition for the design of membrane-integrated artificial photosynthesis systems. ChemSusChem 2017, 10, 4599–4609. [Google Scholar] [CrossRef]
- John, J.K. Monopolar and Bipolar Ion Exchange Membranes Mass Transport Limitations; Ipskamp: Enschede, The Netherlands, 1997. [Google Scholar]
- Onsager, L. Deviations from Ohm’s law in weak electrolytes. J. Chem. Phys. 1934, 2, 599–615. [Google Scholar] [CrossRef]
- Simons, R. Electric field effects on proton transfer between ionizable groups and water in ion exchange membranes. Electrochem. Acta 1984, 29, 151–158. [Google Scholar] [CrossRef]
- Koter, S.; Warszawski, A. A new model for characterization of bipolar membrane electrodialysis of brine. Desalination 2006, 198, 111–123. [Google Scholar] [CrossRef]
- Luo, T.; Abdu, S.; Wessling, M. Selectivity of ion exchange membranes: A review. J. Membr. Sci. 2018, 555, 429–454. [Google Scholar] [CrossRef]
- Zhao, Y.; Tang, K.N.; Ruan, H.M.; Xue, L.X.; Van der Bruggen, B.; Gao, C.J.; Shen, J.N. Sulfonated reduced graphene oxide modification layers to improve monovalent anions selectivity and controllable resistance of anion exchange membrane. J. Membr. Sci. 2017, 536, 167–175. [Google Scholar] [CrossRef]
- Pan, J.F.; Ding, J.C.; Tan, R.Q.; Chen, G.Y.; Zhao, Y.; Gao, C.J.; Van der Bruggen, B.; Shen, J.N. Preparation of a monovalent selective anion exchange membrane through constructing a covalently crosslinked interface by electro-deposition of polyethyleneimine. J. Membr. Sci. 2017, 539, 263–272. [Google Scholar] [CrossRef]
- Zhang, Y.; Van der Bruggen, B.; Pinoy, L.; Meesschaert, B. Separation of nutrient ions and organic compounds from salts in RO concentrates by standard and monovalent selective ion-exchange membranes used in electrodialysis. J. Membr. Sci. 2009, 332, 104–112. [Google Scholar] [CrossRef]
- Wang, W.; Liu, R.; Tan, M.; Suna, H.; Jason Niu, Q.; Xu, T.; Nikonenkoe, V.; Zhang, Y. Evaluation of the ideal selectivity and the performance of selectrodialysis by using TFC ion exchange membranes. J. Membr. Sci. 2019, 582, 236–245. [Google Scholar] [CrossRef]
- Zhang, Y.; Pinoy, L.; Meesschaert, B.; Van der Bruggen, B. Separation of small organic ions from salts by ion-exchange membrane in electrodialysis. AlChE J. 2011, 57, 2070–2078. [Google Scholar] [CrossRef]
- Van der Bruggen, B.; Koninckx, A.; Vandecasteele, C. Separation of monovalent and divalent ions from aqueous solution by electrodialysis and nanofiltration. Water Res. 2004, 38, 1347–1353. [Google Scholar] [CrossRef]
- Sata, T. Studies on anion exchange membranes having permselectivity for specific anions in electrodialysis —Effect of hydrophilicity of anion exchange membranes on permselectivity of anions. J. Membr. Sci. 2000, 167, 1–31. [Google Scholar] [CrossRef]
- Zabolotsky, V.I.; Manzanares, J.A.; Nikonenko, V.V.; Lebedev, K.A.; Lovtsov, E.G. Space charge effect on competitive ion transport through ion-exchange membranes. Desalination 2002, 147, 387–392. [Google Scholar] [CrossRef]
- Zabolotsky, V.I.; Achoh, A.R.; Lebedev, K.A.; Melnikov, S.S. Permselectivity of bilayered ion-exchange membranes in ternary electrolyte. J. Membr. Sci. 2020, 608, 118–152. [Google Scholar] [CrossRef]
- Lide, D. Handbook of Chemistry and Physics, 74th ed.; CRC Press Inc.: Boca Raton, FL, USA, 1993. [Google Scholar]

| Coal Transformation Type | Developed Scale | Waste Salt Discharge Capacity (104 t) | Under Development Scale | Estimated Waste Salt Discharge Capacity (104 t) |
|---|---|---|---|---|
| Coal to oil (104 t/a) | 640 | 64 | 800 | 80 |
| Coal to syngas (108 Nm3/a) | 131 | 21.3 | 222 | 36 |
| Coal to olefin (104 t/a) | 887 | 88.7 | 330 | 33 |
| Coal to ethylene glycol (104 t/a) | 285 | 46.3 | 305 | 49.6 |
| Total | 220.3 | 198.7 |
| Membrane Type | Thickness (µm) | IEC (mmol/g) | Resistance (Ω·cm2) | Voltage Drop c (V) | Efficiency (%) |
|---|---|---|---|---|---|
| Neosepta BP-1E | 220 | - | - | 1.2 | >98 |
| Neosepta CMX | 170 | 1.5–1.8 | 3.0 b | - | - |
| Neosepta AMX | 140 | 1.4–1.7 | 2.4 b | - | - |
| Neosepta ACS | 130 | 1.4–2.0 | 3.8 b | - | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yan, H.; Li, W.; Zhou, Y.; Irfan, M.; Wang, Y.; Jiang, C.; Xu, T. In-Situ Combination of Bipolar Membrane Electrodialysis with Monovalent Selective Anion-Exchange Membrane for the Valorization of Mixed Salts into Relatively High-Purity Monoprotic and Diprotic Acids. Membranes 2020, 10, 135. https://doi.org/10.3390/membranes10060135
Yan H, Li W, Zhou Y, Irfan M, Wang Y, Jiang C, Xu T. In-Situ Combination of Bipolar Membrane Electrodialysis with Monovalent Selective Anion-Exchange Membrane for the Valorization of Mixed Salts into Relatively High-Purity Monoprotic and Diprotic Acids. Membranes. 2020; 10(6):135. https://doi.org/10.3390/membranes10060135
Chicago/Turabian StyleYan, Haiyang, Wei Li, Yongming Zhou, Muhammad Irfan, Yaoming Wang, Chenxiao Jiang, and Tongwen Xu. 2020. "In-Situ Combination of Bipolar Membrane Electrodialysis with Monovalent Selective Anion-Exchange Membrane for the Valorization of Mixed Salts into Relatively High-Purity Monoprotic and Diprotic Acids" Membranes 10, no. 6: 135. https://doi.org/10.3390/membranes10060135
APA StyleYan, H., Li, W., Zhou, Y., Irfan, M., Wang, Y., Jiang, C., & Xu, T. (2020). In-Situ Combination of Bipolar Membrane Electrodialysis with Monovalent Selective Anion-Exchange Membrane for the Valorization of Mixed Salts into Relatively High-Purity Monoprotic and Diprotic Acids. Membranes, 10(6), 135. https://doi.org/10.3390/membranes10060135








