UV-Polymerized Vinylimidazolium Ionic Liquids for Permselective Membranes
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
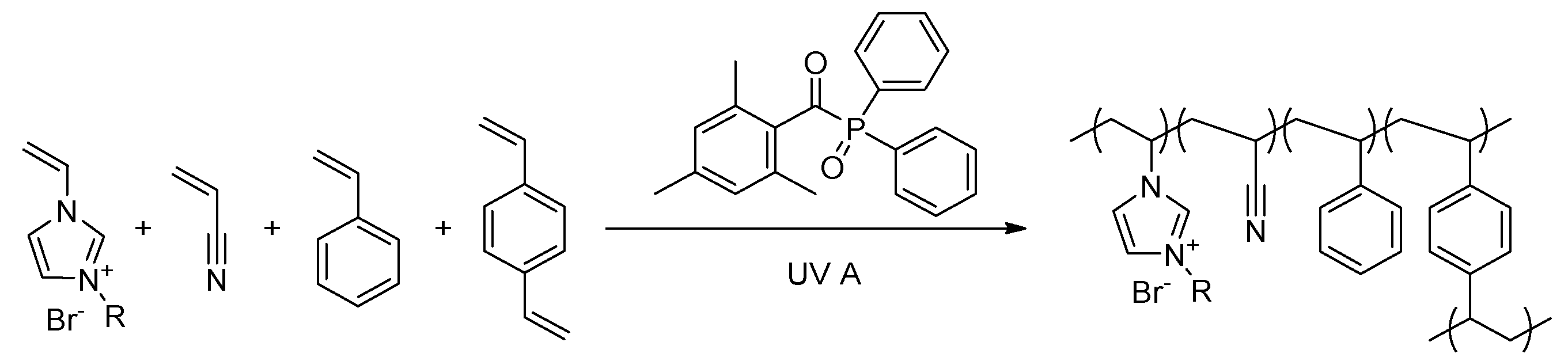
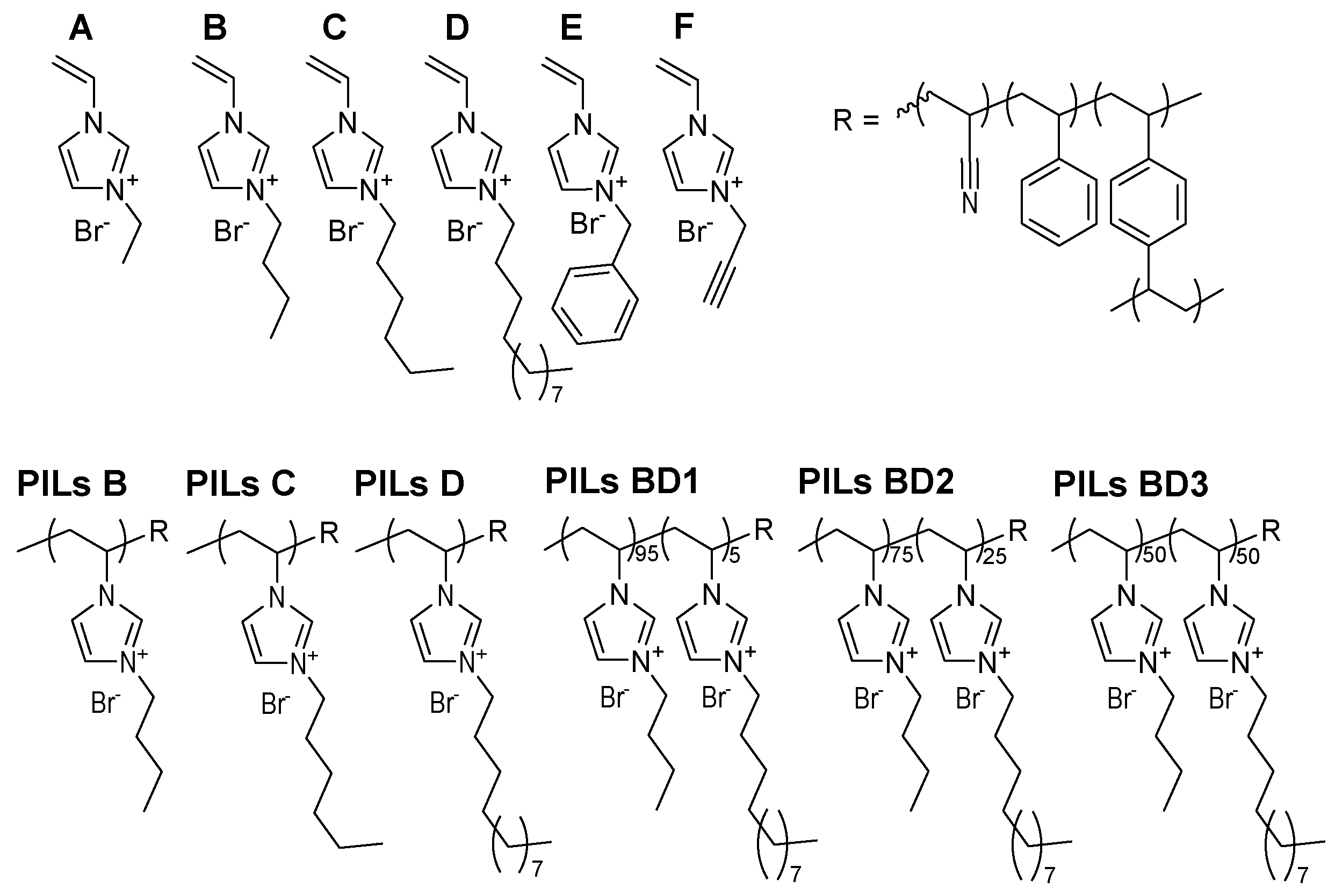
2.2. Synthesis of Ionic Liquids (ILs)
2.3. Formation of the PIL Layer
2.4. Analysis
2.5. Experimental Setup
2.6. AFM Measurements
3. Results and Discussion
3.1. Formation of Polymeric Layers
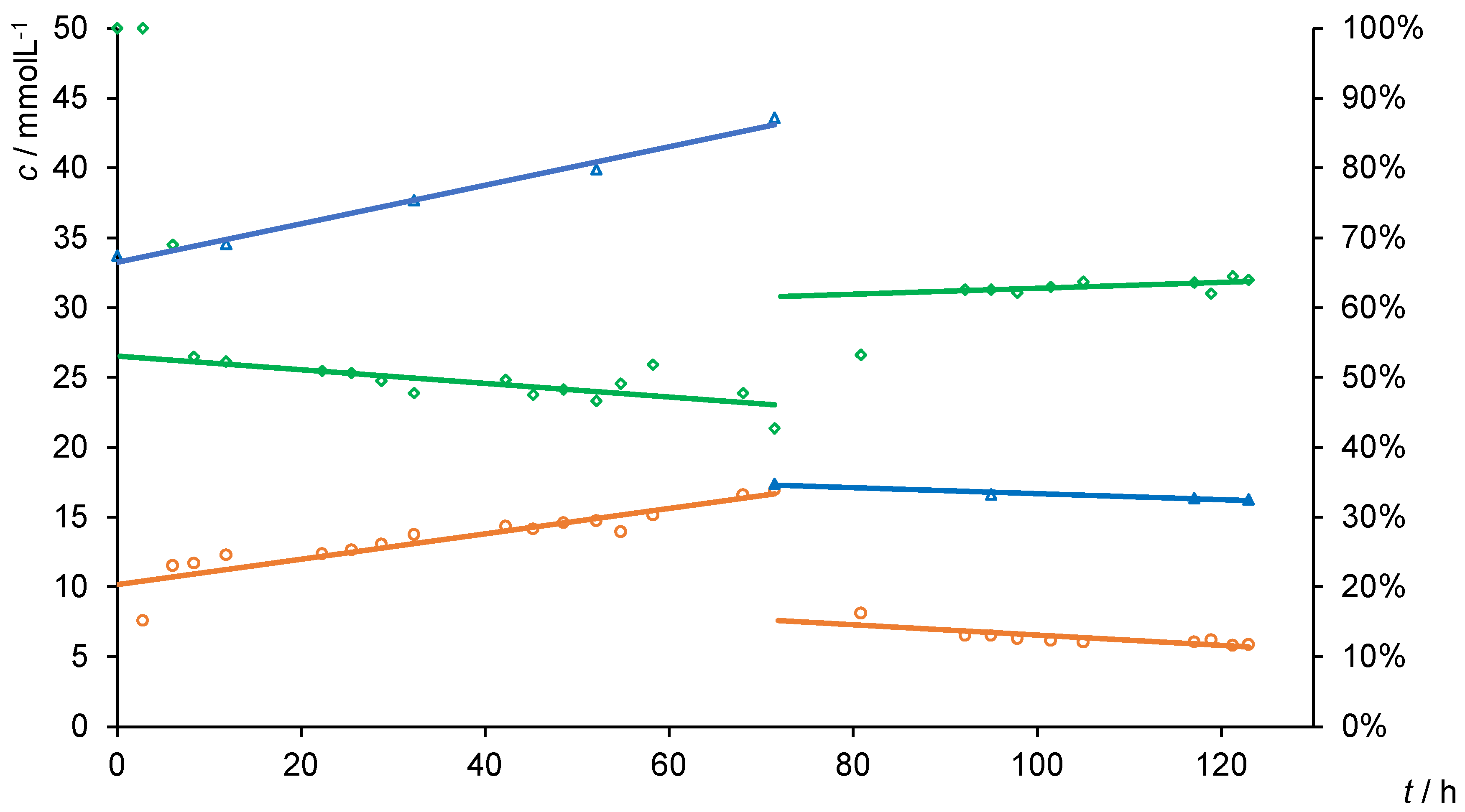
3.2. Flux Measurements
3.3. Separation Experiments
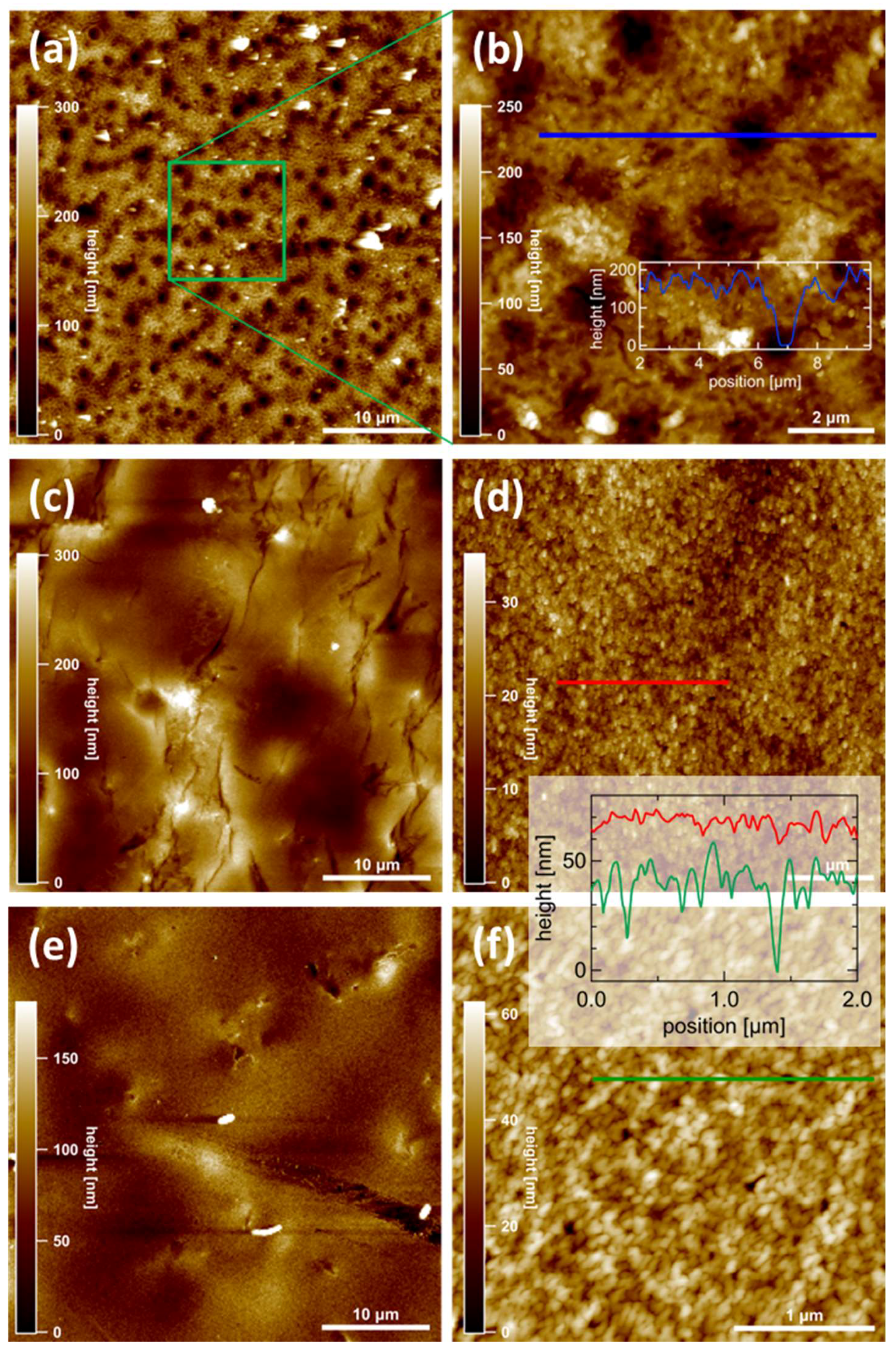
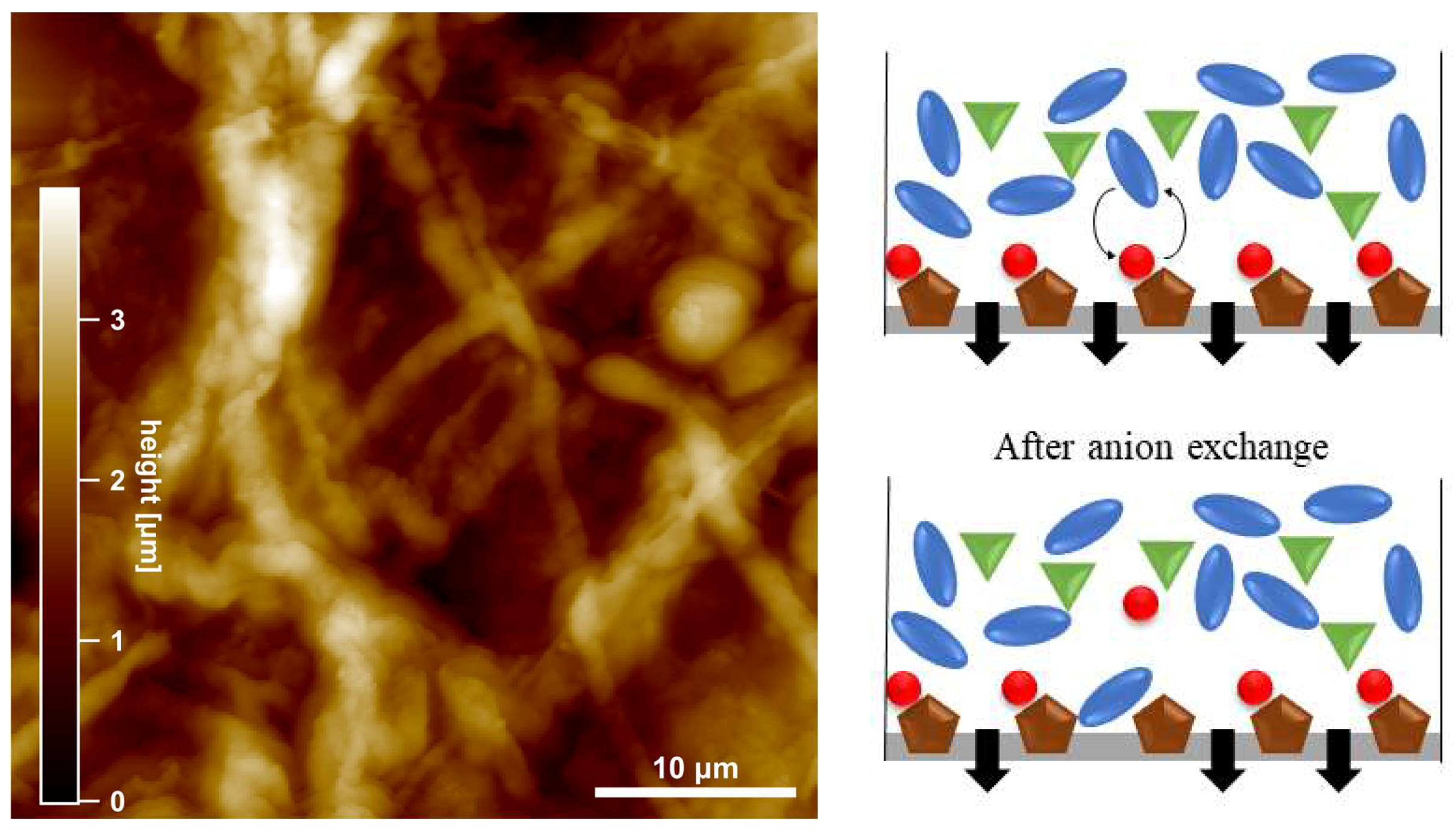
3.4. AFM Measurements
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Walden, P. Molecular weights and electrical conductivity of several fused salts. Bull. Acad. Imper. Sci. (St. Petersburg) 1914, 8, 405–422. [Google Scholar]
- Lu, J.; Yan, F.; Texter, J. Advanced applications of ionic liquids in polymer science. Prog. Polym. Sci. 2009, 34, 431–448. [Google Scholar] [CrossRef]
- Ohno, H. Electrochemical Aspects of Ionic Liquids, 2nd ed.; Ohno, H., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2011; ISBN 9781118003350. [Google Scholar]
- Claus, J.; Sommer, F.O.; Kragl, U. Ionic liquids in biotechnology and beyond. Solid State Ionics 2018, 314, 119–128. [Google Scholar] [CrossRef]
- Yuan, J.; Antonietti, M. Poly(ionic liquid)s: Polymers expanding classical property profiles. Polymer 2011, 52, 1469–1482. [Google Scholar] [CrossRef]
- Armand, M.; Endres, F.; MacFarlane, D.R.; Ohno, H.; Scrosati, B. Ionic-liquid materials for the electrochemical challenges of the future. Nat. Mater. 2009, 8, 621–629. [Google Scholar] [PubMed]
- Bandomir, J.; Schulz, A.; Taguchi, S.; Schmitt, L.; Ohno, H.; Sternberg, K.; Schmitz, K.-P.; Kragl, U. Synthesis and Characterization of Polymerized Ionic Liquids: Mechanical and Thermal Properties of a Novel Type of Hydrogels. Macromol. Chem. Phys. 2014, 215, 716–724. [Google Scholar] [CrossRef]
- Batra, D.; Seifert, S.; Firestone, M.A. The effect of cation structure on the mesophase architecture of self-assembled and polymerized imidazolium-based ionic liquids. Macromol. Chem. Phys. 2007, 208, 1416–1427. [Google Scholar] [CrossRef]
- Großeheilmann, J.; Bandomir, J.; Kragl, U. Preparation of Poly(ionic liquid)s-Supported Recyclable Organocatalysts for the Asymmetric Nitroaldol (Henry) Reaction. Chem. Eur. J. 2015, 21, 18957–18960. [Google Scholar] [CrossRef]
- Liu, Z.; Wang, Y.; Ren, Y.; Jin, G.; Zhang, C.; Chen, W.; Yan, F. Poly(ionic liquid) hydrogel-based anti-freezing ionic skin for a soft robotic gripper. Mater. Horizons 2020, 7, 919–927. [Google Scholar] [CrossRef]
- Shaplov, A.S.; Ponkratov, D.O.; Vygodskii, Y.S. Poly(ionic liquid)s: Synthesis, properties, and application. Polym. Sci. Ser. B 2016, 58, 73–142. [Google Scholar] [CrossRef]
- Souza, I.D.; Hantao, L.W.; Queiroz, M.E.C. Polymeric ionic liquid open tubular capillary column for on-line in-tube SPME coupled with UHPLC-MS/MS to determine endocannabinoids in plasma samples. Anal. Chim. Acta 2019, 1045, 108–116. [Google Scholar] [CrossRef]
- Ohno, H.; Yoshizawa, M.; Ogihara, W. Development of new class of ion conductive polymers based on ionic liquids. Electrochim. Acta 2004, 50, 255–261. [Google Scholar] [CrossRef]
- Tang, J.; Radosz, M.; Shen, Y. Poly(ionic liquid)s as Optically Transparent Microwave-Absorbing Materials. Macromolecules 2008, 41, 493–496. [Google Scholar] [CrossRef]
- Ohno, H. Design of Ion Conductive Polymers Based on Ionic Liquids. Macromol. Symp. 2007, 249–250, 551–556. [Google Scholar] [CrossRef]
- Green, M.D.; Salas-de la Cruz, D.; Ye, Y.; Layman, J.M.; Elabd, Y.A.; Winey, K.I.; Long, T.E. Alkyl-Substituted N-Vinylimidazolium Polymerized Ionic Liquids: Thermal Properties and Ionic Conductivities. Macromol. Chem. Phys. 2011, 212, 2522–2528. [Google Scholar] [CrossRef]
- Yoshio, M.; Kagata, T.; Hoshino, K.; Mukai, T.; Ohno, H.; Kato, T. One-Dimensional Ion-Conductive Polymer Films: Alignment and Fixation of Ionic Channels Formed by Self-Organization of Polymerizable Columnar Liquid Crystals. J. Am. Chem. Soc. 2006, 128, 5570–5577. [Google Scholar] [CrossRef] [PubMed]
- Eftekhari, A.; Saito, T. Synthesis and properties of polymerized ionic liquids. Eur. Polym. J. 2017, 90, 245–272. [Google Scholar] [CrossRef]
- Qian, W.; Texter, J.; Yan, F. Frontiers in poly(ionic liquid)s: Syntheses and applications. Chem. Soc. Rev. 2017, 46, 1124–1159. [Google Scholar] [CrossRef]
- Pourjavadi, A.; Hosseini, S.H.; Soleyman, R. Crosslinked poly(ionic liquid) as high loaded dual acidic organocatalyst. J. Mol. Catal. A Chem. 2012, 365, 55–59. [Google Scholar] [CrossRef]
- Ziółkowski, B.; Diamond, D. Thermoresponsive poly(ionic liquid) hydrogels. Chem. Commun. 2013, 49, 10308. [Google Scholar] [CrossRef]
- Maksym, P.; Tarnacka, M.; Bielas, R.; Hachuła, B.; Zajac, A.; Szpecht, A.; Smiglak, M.; Kaminski, K.; Paluch, M. Structure-property relationships of tailored imidazolium-and pyrrolidinium-based poly(ionic liquid)s. Solid-like vs. gel-like systems. Polymer 2020, 192, 122262. [Google Scholar] [CrossRef]
- Maksym, P.; Tarnacka, M.; Dzienia, A.; Erfurt, K.; Chrobok, A.; Zięba, A.; Wolnica, K.; Kaminski, K.; Paluch, M. A facile route to well-defined imidazolium-based poly(ionic liquid)s of enhanced conductivity via RAFT. Polym. Chem. 2017, 8, 5433–5443. [Google Scholar] [CrossRef]
- Nishimura, N.; Ohno, H. 15th anniversary of polymerised ionic liquids. Polymer 2014, 55, 3289–3297. [Google Scholar] [CrossRef]
- Claus, J.; Brietzke, A.; Lehnert, C.; Oschatz, S.; Grabow, N.; Kragl, U. Swelling characteristics and biocompatibility of ionic liquid based hydrogels for biomedical applications. PLoS ONE 2020, 15, e0231421. [Google Scholar] [CrossRef]
- Claus, J.; Jastram, A.; Piktel, E.; Bucki, R.; Janmey, P.; Kragl, U. Polyerized Ionic Liquids-Based Hydrogels With Intrinsic Antibacterial Activity—Modern Weapons Against Multidrug-Resistant Infections. J. Appl. Polym. Sci. 2020, 105, 135–149. [Google Scholar]
- Costa, S.P.F.; Azevedo, A.M.O.; Pinto, P.C.A.G.; Saraiva, M.L.M.F.S. Environmental Impact of Ionic Liquids: Recent Advances in (Eco)toxicology and (Bio)degradability. ChemSusChem 2017, 10, 2321–2347. [Google Scholar] [CrossRef] [PubMed]
- European Chemical Agency. Available online: https://echa.europa.eu/ (accessed on 23 October 2020).
- Yuan, J.; Mecerreyes, D.; Antonietti, M. Poly(ionic liquid)s: An update. Prog. Polym. Sci. 2013, 38, 1009–1036. [Google Scholar] [CrossRef]
- Bengani, P.; Kou, Y.; Asatekin, A. Zwitterionic copolymer self-assembly for fouling resistant, high flux membranes with size-based small molecule selectivity. J. Memb. Sci. 2015, 493, 755–765. [Google Scholar] [CrossRef]
- Zheng, Z.; Xu, Q.; Guo, J.; Qin, J.; Mao, H.; Wang, B.; Yan, F. Structure–Antibacterial Activity Relationships of Imidazolium-Type Ionic Liquid Monomers, Poly(ionic liquids) and Poly(ionic liquid) Membranes: Effect of Alkyl Chain Length and Cations. ACS Appl. Mater. Interfaces 2016, 8, 12684–12692. [Google Scholar] [CrossRef]
- Sengupta, A.; Kumar Ethirajan, S.; Kamaz, M.; Jebur, M.; Wickramasinghe, R. Synthesis and characterization of antibacterial poly ionic liquid membranes with tunable performance. Sep. Purif. Technol. 2019, 212, 307–315. [Google Scholar] [CrossRef]
- Matandabuzo, M.; Ajibade, P.A. Vinyl pyridinium polymeric ionic liquid functionalized carbon nanotube composites as adsorbent for chromium(VI) in aqueous solution. J. Mol. Liq. 2019, 296, 111778. [Google Scholar] [CrossRef]
- Wieszczycka, K.; Filipowiak, K.; Wojciechowska, I.; Aksamitowski, P. Novel ionic liquid-modified polymers for highly effective adsorption of heavy metals ions. Sep. Purif. Technol. 2020, 236, 116313. [Google Scholar] [CrossRef]
- Si, Z.; Qiu, L.; Dong, H.; Gu, F.; Li, Y.; Yan, F. Effects of Substituents and Substitution Positions on Alkaline Stability of Imidazolium Cations and Their Corresponding Anion-Exchange Membranes. ACS Appl. Mater. Interfaces 2014, 6, 4346–4355. [Google Scholar] [CrossRef] [PubMed]
- Lin, B.; Dong, H.; Li, Y.; Si, Z.; Gu, F.; Yan, F. Alkaline Stable C2-Substituted Imidazolium-Based Anion-Exchange Membranes. Chem. Mater. 2013, 25, 1858–1867. [Google Scholar] [CrossRef]
- Gu, F.; Dong, H.; Li, Y.; Sun, Z.; Yan, F. Base Stable Pyrrolidinium Cations for Alkaline Anion Exchange Membrane Applications. Macromolecules 2014, 47, 6740–6747. [Google Scholar] [CrossRef]
- Gu, F.; Dong, H.; Li, Y.; Si, Z.; Yan, F. Highly Stable N3-Substituted Imidazolium-Based Alkaline Anion Exchange Membranes: Experimental Studies and Theoretical Calculations. Macromolecules 2014, 47, 208–216. [Google Scholar] [CrossRef]
- Xu, Q.; Zheng, Z.; Wang, B.; Mao, H.; Yan, F. Zinc Ion Coordinated Poly(Ionic Liquid) Antimicrobial Membranes for Wound Healing. ACS Appl. Mater. Interfaces 2017, 9, 15. [Google Scholar] [CrossRef]
- Zheng, Z.; Guo, J.; Mao, H.; Xu, Q.; Qin, J.; Yan, F. Metal-Containing Poly(ionic liquid) Membranes for Antibacterial Applications. ACS Biomater. Sci. Eng. 2017, 3, 922–928. [Google Scholar] [CrossRef]
- Marcilla, R.; Blazquez, J.A.; Rodriguez, J.; Pomposo, J.A.; Mecerreyes, D. Tuning the solubility of polymerized ionic liquids by simple anion-exchange reactions. J. Polym. Sci. Part A Polym. Chem. 2004, 42, 208–212. [Google Scholar] [CrossRef]
- Coupillaud, P.; Pinaud, J.; Guidolin, N.; Vignolle, J.; Fèvre, M.; Veaudecrenne, E.; Mecerreyes, D.; Taton, D. Poly(ionic liquid)s based on imidazolium hydrogen carbonate monomer units as recyclable polymer-supported N-heterocyclic carbenes: Use in organocatalysis. J. Polym. Sci. Part A Polym. Chem. 2013, 51, 4530–4540. [Google Scholar] [CrossRef]
- Ulbricht, M. Advanced functional polymer membranes. Polymer 2006, 47, 2217–2262. [Google Scholar] [CrossRef]
- Saleh, T.A.; Gupta, V.K. Nanomaterial and Polymer Membranes: Synthesis, Characterization, and Applications; Elsevier: Amsterdam, The Netherlands, 2016; ISBN 9780128047033. [Google Scholar]
- Basile, A.; Nunes, S.P. Advanced Membrane Science and Technology for Sustainable Energy and Enviromental Applications; Woodhead Publishing: Philadelphia, PA, USA, 2011; ISBN 9781845699697. [Google Scholar]
- Khulbe, K.C.; Feng, C.Y.; Matsuura, T. Synthetic Polymeric Membranes; Springer Laboratory: Berlin/Heidelberg, Germany, 2008; ISBN 978-3-540-73993-7. [Google Scholar]
- Liao, Y.; Loh, C.H.; Tian, M.; Wang, R.; Fane, A.G. Progress in electrospun polymeric nanofibrous membranes for water treatment: Fabrication, modification and applications. Prog. Polym. Sci. 2018, 77, 69–94. [Google Scholar] [CrossRef]
- Guo, J.; Xu, Q.; Shi, R.; Zheng, Z.; Mao, H.; Yan, F. Polyanionic Antimicrobial Membranes: An Experimental and Theoretical Study. Langmuir 2017, 33, 4346–4355. [Google Scholar] [CrossRef]
- Kamaz, M.; Sengupta, A.; DePaz, S.S.; Chiao, Y.-H.; Ranil Wickramasinghe, S. Poly(ionic liquid) augmented membranes for π electron induced separation/fractionation of aromatics. J. Memb. Sci. 2019, 579, 102–110. [Google Scholar] [CrossRef]
- Cheng, W.; Liu, C.; Tong, T.; Epsztein, R.; Sun, M.; Verduzco, R.; Ma, J.; Elimelech, M. Selective removal of divalent cations by polyelectrolyte multilayer nanofiltration membrane: Role of polyelectrolyte charge, ion size, and ionic strength. J. Memb. Sci. 2018, 559, 98–106. [Google Scholar] [CrossRef]
- Marcus, Y. A simple empirical model describing the thermodynamics of hydration of ions of widely varying charges, sizes, and shapes. Biophys. Chem. 1994, 51, 111–127. [Google Scholar] [CrossRef]
- Nightingale, E.R. Phenomenological Theory of Ion Solvation. Effective Radii of Hydrated Ions. J. Phys. Chem. 1959, 63, 1381–1387. [Google Scholar] [CrossRef]
- Bürger, J.; Kunnathully, V.S.; Kool, D.; Lindner, J.K.N.; Brassat, K. Characterisation of the PS-PMMA interfaces in microphase separated block copolymer thin films by analytical (S)TEM. Nanomaterials 2020, 10, 141. [Google Scholar] [CrossRef] [PubMed]

| Membrane | LH2O/L m−2 h−1 bar−1 |
|---|---|
| Ultracell® 30 kDa | 193.4 ± 10.92 |
| PILs B | 74.09 ± 8.63 |
| Ultracell® 5 kDa | 12.24 ± 0.57 |
| PILs C | 5.160 ± 0.53 |
| DuraMem™ 200 | 1.880 ± 0.06 |
| PILs D | 0.027 ± 0.0021 |
| PILs BD1 | 0.291 ± 0.0002 |
| PILs BD2 | 0.259 ± 0.0041 |
| PILs BD3 | 0.051 ± 0.0025 a |
| Sugar | M/g∙mol−1 | Charge | R |
|---|---|---|---|
| K-gluconate | 234.25 | yes | 25% |
| Na-gluconate | 218.14 | yes | 53% |
| Ca-gluconate | 448.39 | yes | 75% |
| d-glucose | 180.16 | no | 3% |
| d-mannitol | 182.17 | no | 6% |
| Sucrose | 342.30 | no | 14% |
| Raffinose | 504.44 | no | 23% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sommer, F.O.; Appelt, J.-S.; Barke, I.; Speller, S.; Kragl, U. UV-Polymerized Vinylimidazolium Ionic Liquids for Permselective Membranes. Membranes 2020, 10, 308. https://doi.org/10.3390/membranes10110308
Sommer FO, Appelt J-S, Barke I, Speller S, Kragl U. UV-Polymerized Vinylimidazolium Ionic Liquids for Permselective Membranes. Membranes. 2020; 10(11):308. https://doi.org/10.3390/membranes10110308
Chicago/Turabian StyleSommer, Fridolin O., Jana-Sophie Appelt, Ingo Barke, Sylvia Speller, and Udo Kragl. 2020. "UV-Polymerized Vinylimidazolium Ionic Liquids for Permselective Membranes" Membranes 10, no. 11: 308. https://doi.org/10.3390/membranes10110308
APA StyleSommer, F. O., Appelt, J.-S., Barke, I., Speller, S., & Kragl, U. (2020). UV-Polymerized Vinylimidazolium Ionic Liquids for Permselective Membranes. Membranes, 10(11), 308. https://doi.org/10.3390/membranes10110308








