Solid-State Membrane Sensors Based on Man-Tailored Biomimetic Receptors for Selective Recognition of Isoproturon and Diuron Herbicides
Abstract
:1. Introduction
2. Experimental
2.1. Equipment
2.2. Reagents and Materials
2.3. Synthesis of Host-Tailored Polymers
2.4. Binding Experiments
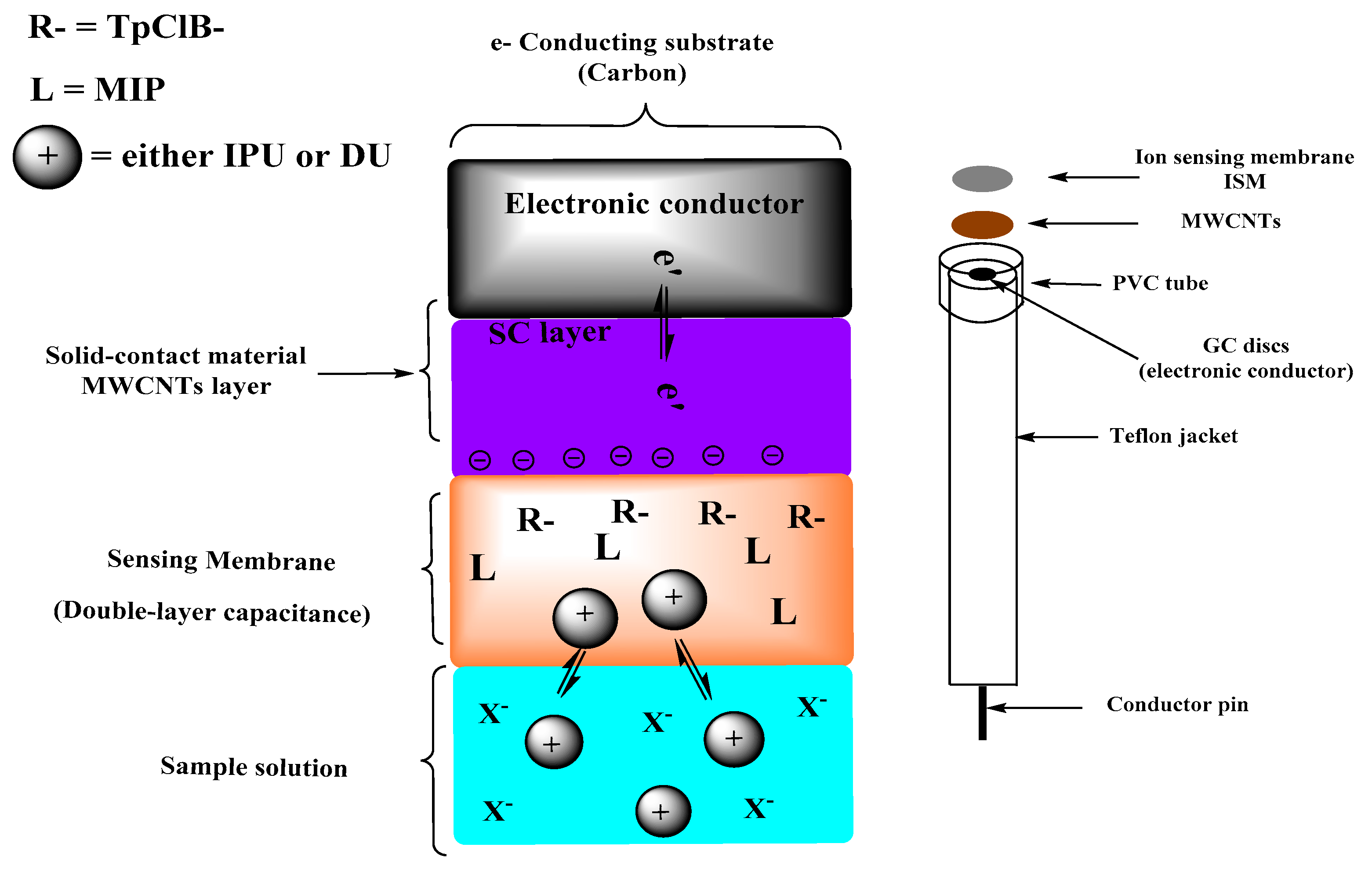
2.5. Sensor Preparation
2.6. Analytical Applications
3. Results and Discussion
3.1. Equilibrium Adsorption
3.2. Surface Morphology
3.3. ISE Performance Characteristics
3.4. Sensors’ Selectivity
3.5. Impedance and Chronopotentiometric Measurements
3.6. Analytical Application: Monitoring of Phenylureas in Water Samples
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Barbash, J.E.; Resek, E.A. Pesticides in Ground Water. Distribution, Trends and Governing Factors; Ann Arbor Press: Chelsea, MI, USA, 1996. [Google Scholar]
- Ragsdale, N.N.; Menzer, R.E. Carcinogenicity and Pesticides: Principles, Issues and Relationships; American Chemical Society: Washington, DC, USA, 1989. [Google Scholar]
- Lewis, R.J. Sax’s Dangerous Properties of Industrial Materials, 9th ed.; Van Nostrand Reinhold: New York, NY, USA, 1992. [Google Scholar]
- Krieger, R. Hayes’ Handbook of Pesticide Toxicology, 3rd ed.; Academic Press Inc.: Cambridge, MA, USA, 2010. [Google Scholar]
- Vanraes, P.; Wardenier, N.; Surmont, P.; Lynen, F.; Nikiforov, A.; Van Hulle, S.W.H.; Leys, C.; Bogaerts, A. Analysis of Short-Lived Reactive Species in Plasma−Air−WaterSystems: The Dos and the Do Nots. J. Hazard. Mater. 2018, 354, 180–190. [Google Scholar] [CrossRef] [PubMed]
- Starling, M.C.V.; Amorim, C.C.; Leão, M.M.D. Occurrence, control and fate of contaminants of emerging concern in environmental compartments in Brazil. J. Hazard. Mater. 2019, 372, 17–36. [Google Scholar] [CrossRef] [PubMed]
- Siisse, H.; Müller, H. Pesticide analysis by micellar electrokinetic capillary chromatography. J. Chromatogr. A 1996, 730, 337–343. [Google Scholar] [CrossRef]
- Sherma, J. Current Status of Pesticide Residue Analysis. J. AOAC Int. 1997, 80, 283–288. [Google Scholar] [CrossRef] [Green Version]
- Goger, B.; Kunert, O.; Seger, C.; Rinelli, R.; Winstersteiger, R. Quantification of phenylurea pesticides by HPLC/ECD and photolysis. Electroanalysis 2001, 3, 1335. [Google Scholar] [CrossRef]
- Farran, A.; Ruiz, S. Application of solid-phase extraction and micellar electrokinetic capillary chromatography to the study of hydrolytic and photolytic degradation of phenoxy acid and phenylurea herbicides. J. Chromatogr. A 2004, 1024, 267–274. [Google Scholar] [CrossRef]
- Chicharro, M.; Bermejo, E.; Sanchez, A.; Zapardiel, A.; Fernandez-Gutierrez, A.; Arraez, D. Multiresidue analysis of phenylurea herbicides in environmental waters by capillary electrophoresis using electrochemical detection. Anal. Bioanal. Chem. 2005, 382, 519. [Google Scholar] [CrossRef]
- Su, M.; Jia, L.; Wu, X.; Sun, H. Residue investigation of some phenylureas and tebuthiuron herbicides in vegetables by ultra-performance liquid chromatography coupled with integrated selective accelerated solvent extraction-clean up in situ. J. Sci. Food Agric. 2018, 98, 4845–4853. [Google Scholar] [CrossRef]
- Gupta, V.; Sethi, B.; Sharma, R.; Agarwal, S.; Bharti, A. Mercury selective potentiometric sensor based on low rim functionalized thiacalix [4]-arene as a cationic receptor. J. Mol. Liq. 2013, 177, 114–118. [Google Scholar] [CrossRef]
- Duarte, E.H.; Casarin, J.; Sartori, E.R.; Tarley, C.R.T. Highly improved simultaneous herbicides determination in water samples by differential pulse voltammetry using boron-doped diamond electrode and solid phase extraction on cross-linked poly(vinylimidazole). Sens. Actuators B Chem. 2018, 255, 166–175. [Google Scholar] [CrossRef]
- Noyrod, P.; Chailapakul, O.; Wonsawat, W.; Chuanuawatanakul, S. Sensitive simultaneous voltammetric determination of the herbicides diuron and isoproturon at a platinum/chitosan bio-based sensing platform. J. Electroanal. Chem. 2018, 719, 54–59. [Google Scholar] [CrossRef]
- Mani, V.; Devasenathipathy, R.; Chen, S.-M.; Wu, T.-Y.; Kohilarani, K. High-performance electrochemical amperometric sensors for the sensitive determination of phenyl urea herbicides diuron and fenuron. Ionics 2015, 21, 2675–2683. [Google Scholar] [CrossRef]
- Morawski, F.D.M.; Winiarski, J.P.; De Campos, C.E.M.; Parize, A.L.; Jost, C.L. Sensitive simultaneous voltammetric determination of the herbicides diuron and isoproturon at a platinum/chitosan bio-based sensing platform. Ecotoxicol. Environ. Saf. 2020, 206, 111181. [Google Scholar] [CrossRef] [PubMed]
- Sadriu, I.; Bouden, S.; Nicolle, J.; Podvorica, F.; Bertagna, V.; Berho, C.; Amalric, L.; Vautrin-Ul, C. Molecularly imprinted polymer modified glassy carbon electrodes for the electrochemical analysis of isoproturon in water. Talanta 2020, 207, 120222. [Google Scholar] [CrossRef] [PubMed]
- Dimeski, G.; Badrick, T.; John, A.S. Ion Selective Electrodes (ISEs) and interferences—A review. Clin. Chim. Acta 2010, 411, 309–317. [Google Scholar] [CrossRef]
- Ashmawy, N.H.; Almehizia, A.A.; Youssef, T.A.; El-Galil, E.A.A.; Al-Omar, M.A.; Kamel, A.H. Novel Carbon/PEDOT/PSS-Based screen-printed biosensors for acetylcholine neurotransmitter and acetylcholinesterase detection in human serum. Molecules 2019, 24, 1539. [Google Scholar] [CrossRef] [Green Version]
- Hassan, S.S.M.; Elnemma, E.M.; Mohamed, A.H.K. Novel Biomedical Sensors for Flow Injection Potentiometric Determination of Creatinine in Human Serum. Electroanalysis 2005, 17, 2246–2253. [Google Scholar] [CrossRef]
- Kamel, A.H.; Amr, A.E.; Galal, H.R.; Al-Omar, M.A.; Almehizia, A.A. Screen-Printed Sensor Based on Potentiometric Transduction for Free Bilirubin Detection as a Biomarker for Hyperbilirubinemia Diagnosis. Chemosensors 2020, 8, 86. [Google Scholar] [CrossRef]
- Abd-Rabboh, H.S.M.; Kamel, A.H.; Amr, A.E. Article All-Solid-State Calcium Sensors Modified with Polypyrrol (PPY) and Graphene Oxide (GO) as Solid-Contact Ion-to-Electron Transducers. Chemosensors 2020, 8, 93. [Google Scholar] [CrossRef]
- Eldin, A.G.; Amr, A.E.-G.E.; Kamel, A.H.; Hassan, S.S.M. Screen-printed Microsensors Using Polyoctyl-thiophene (POT) Conducting Polymer as Solid Transducer for Ultratrace Determination of Azides. Molecules 2019, 24, 1392. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van De Velde, L.; D’Angremont, E.; Olthuis, W. Solid contact potassium selective electrodes for biomedical applications—A review. Talanta 2016, 160, 56–65. [Google Scholar] [CrossRef]
- Hauser, P.C.; Chiang, D.W.; Wright, G.A. A potassium-ion selective electrode with valinomycin based poly(vinyl chloride) membrane and a poly(vinyl ferrocene) solid contact. Anal. Chim. Acta 1995, 302, 241–248. [Google Scholar] [CrossRef]
- Guziński, M.; Lisak, G.; Sokalski, T.; Bobacka, J.; Ivaska, A.; Bocheńska, M.; Lewenstam, A. Solid contact Ion-selective electrodes with highly selective thioamide derivatives of p-tetr-butylcalix[4]arene for the determination of lead(II) in environmental samples. Anal. Chem. 2013, 85, 1555–1561. [Google Scholar] [CrossRef] [PubMed]
- Lindfors, T. Light sensitivity and potential stability of electrically conducting polymers commonly used in solid contact ion-selective electrodes. J. Solid State Electrochem. 2008, 13, 77–89. [Google Scholar] [CrossRef]
- Bobacka, J.; Ivaska, A.; Lewenstam, A. Potentiometric Ion Sensors. Chem. Rev. 2008, 108, 329–351. [Google Scholar] [CrossRef] [PubMed]
- Mirata, F.; Resmini, M. Molecularly Imprinted Polymers for Catalysis and Synthesis, Molecularly Imprinted Polymers in Biotechnology; Springer: Berlin/Heidelberg, Germany, 2015; pp. 107–129. [Google Scholar]
- Lok, C.; Son, R. Application of molecularly imprinted polymers in food sample analysis—A perspective. Int. Food Res. J. 2009, 16, 127–140. [Google Scholar]
- Abdalla, N.S.; Amr, A.E.-G.E.; El-Tantawy, A.S.M.; Al-Omar, M.A.; Kamel, A.H.; Khalifa, N.M. Tailor-Made Specific Recognition of Cyromazine Pesticide Integrated in a Potentiometric Strip Cell for Environmental and Food Analysis. Polymers 2019, 11, 1526. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, G.; Jin, M.; Du, P.-F.; Zhang, C.; Cui, X.; Zhang, Y.; She, Y.; Shao, H.; Jin, F.; Wang, S.; et al. A sensitive chemiluminescence enzyme immunoassay based on molecularly imprinted polymers solid-phase extraction of parathion. Anal. Biochem. 2017, 530, 87–93. [Google Scholar] [CrossRef]
- Azodi-Deilami, S.; Abdouss, M.; Kordestani, D. Synthesis and Characterization of the Magnetic Molecularly Imprinted Polymer Nanoparticles Using N, N-bis Methacryloyl Ethylenediamine as a New Cross-linking Agent for Controlled Release of Meloxicam. Appl. Biochem. Biotechnol. 2014, 172, 3271–3286. [Google Scholar] [CrossRef]
- Asadi, E.; Azodi-Deilami, S.; Abdouss, M.; Khaghani, S. Cyproterone Synthesis, Recognition and Controlled Release by Molecularly Imprinted Nanoparticle. Appl. Biochem. Biotechnol. 2012, 167, 2076–2087. [Google Scholar] [CrossRef]
- Haupt, K. Molecularly imprinted polymers in analytical chemistry. Analyst 2001, 126, 747–756. [Google Scholar] [CrossRef] [PubMed]
- Ye, L.; Haupt, K. Molecularly imprinted polymers as antibody and receptor mimics for assays, sensors and drug discovery. Anal. Bioanal. Chem. 2004, 378, 1887–1897. [Google Scholar] [CrossRef] [PubMed]
- Li, H.-H.; Wang, H.-H.; Li, W.-T.; Fang, X.-X.; Guo, X.-C.; Zhou, W.-H.; Cao, X.; Kou, D.-X.; Zhou, Z.-J.; Wu, S.-X. A novel electrochemical sensor for epinephrine based on three dimensional molecularly imprinted polymer arrays. Sens. Actuators B Chem. 2016, 222, 1127–1133. [Google Scholar] [CrossRef]
- Chen, H.; Zhang, W.; Yang, N. Chitosan-Based Surface Molecularly Imprinted Polymer Microspheres for Sustained Release of Sinomenine Hydrochloride in Aqueous Media. Appl. Biochem. Biotechnol. 2017, 185, 370–384. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Tao, H.; Li, J. Molecularly Imprinted Electrochemical Luminescence Sensor Based on Enzymatic Amplification for Ultratrace Isoproturon Determination. Electroanalysis 2012, 24, 1664–1670. [Google Scholar] [CrossRef]
- Kamel, A.H.; Jiang, X.; Li, P.; Liang, R. A paper-based potentiometric sensing platform based on molecularly imprinted nanobeads for determination of bisphenol A. Anal. Methods 2018, 10, 3890–3895. [Google Scholar] [CrossRef]
- El-Kosasy, A.; Kamel, A.H.; Hussin, L.; Ayad, M.F.; Fares, N. Mimicking new receptors based on molecular imprinting and their application to potentiometric assessment of 2,4-dichlorophenol as a food taint. Food Chem. 2018, 250, 188–196. [Google Scholar] [CrossRef]
- Kamel, A.H. Solid Contact Potentiometric Sensors Based on Host-Tailored Molecularly Imprinted Polymers for Creatine Assessment. Int. J. Electrochem. Sci. 2016, 11, 8938–8949. [Google Scholar] [CrossRef]
- Hassan, S.S.M.; Amr, A.E.-G.E.; El-Naby, H.A.; Al-Omar, M.A.; Kamel, A.H. Non-Equilibrium Potential Responses towards Neutral Orcinol Using All-Solid-State Potentiometric Sensors Integrated with Molecularly Imprinted Polymers. Polymers 2019, 11, 1232. [Google Scholar] [CrossRef] [Green Version]
- Abd-Rabboh, H.S.M.; Kamel, A.H. Mimicking a Receptor for Cyanide Ion Based on Ion Imprinting and Its Applications in Potential Transduction. Electroanalysis 2012, 24, 1409–1415. [Google Scholar] [CrossRef]
- Moreira, F.T.; Guerreiro, J.R.L.; Azevedo, V.L.; Kamel, A.H.; Sales, M.G.F. New biomimetic sensors for the determination of tetracycline in biological samples: Batch and flow mode operations. Anal. Methods 2010, 2, 2039. [Google Scholar] [CrossRef] [Green Version]
- Yamamura, H.L.; Enna, S.J.; Kuhar, M.J. Neurotransmitter Receptor Binding; Raven Press: New York, NY, USA, 1985. [Google Scholar]
- Taylor, J.K. Quality Assurance of Chemical Measurements; CRC Press: Boca Raton, FL, USA, 1987. [Google Scholar]
- Bakker, E.; Pretsch, E.; Bühlmann, P. Selectivity of potentiometric ion sensors. Anal. Chem. 2000, 72, 1127–1133. [Google Scholar] [CrossRef] [PubMed]
- Konopka, A.; Sokalski, T.; Michalska, A.; Lewenstam, A.; Maj-Żurawska, M. Factors Affecting the Potentiometric Response of All-Solid-State Solvent Polymeric Membrane Calcium-Selective Electrode for Low-Level Measurements. Anal. Chem. 2004, 76, 6410–6418. [Google Scholar] [CrossRef] [PubMed]
- Bobacka, J. Potential Stability of All-Solid-State Ion-Selective Electrodes Using Conducting Polymers as Ion-to-Electron Transducers. Anal. Chem. 1999, 71, 4932–4937. [Google Scholar] [CrossRef]








| Parameter | MIP(IPU)/MWCNTs-ISEs (Sensor 1) | MIP(DU)/MWCNTs-ISEs (Sensor 2) | NIP/IPU (Sensor 3) | NIP/DU (Sensor 4) |
|---|---|---|---|---|
| Slope, mV/decade | 53.1 ± 1.2 | 57.2 ± 1.2 | 39.8 ± 0.9 | 48.1 ± 0.6 |
| Correlation coefficient, r2 | 0.997 | 0.998 | 0.998 | 0.998 |
| Linear range, M | 2.2 × 10−6–1.0 × 10−3 | 3.2 × 10−6–1.0 × 10−3 | 3.2 × 10−5–1.0 × 10−3 | 3.4 × 10−5–1.0 × 10−3 |
| Detection limit, M | 8.3 × 10−7 | 1.4 × 10−6 | 1.3 × 10−5 | 1.2 × 10−5 |
| Working range, pH | 2.5–4.5 | 2.5–4.5 | 2.5–4.5 | 2.5–4.5 |
| Response time, s | <10 | <10 | <10 | <10 |
| Standard deviation (σv), mV | 1.1 | 0.7 | 1.2 | 0.9 |
| Accuracy, % | 99.1 | 98.8 | 97.6 | 96.8 |
| Precision (CVw), % | 0.6 | 0.8 | 0.6 | 0.5 |
| Between-day variability (CVb), % | 1.1 | 0.9 | 0.8 | 1.2 |
| Interfering Ion, B | MIP (DU)/MWCNTs-ISEs | MIP(IPU)/MWCNTs-ISEs | NIP/DU | NIP/IPU |
|---|---|---|---|---|
| Isoproturon (IPU) | 0 | −3.1 ± 0.6 | 0 | −3.3 ± 0.2 |
| Diuron (DU) | −3.3 ± 0.6 | 0 | −3.0 ± 0.1 | 0 |
| Linuron (LU) | −2.9 ± 0.4 | −0.7 ± 0.03 | −1.7 ± 0.3 | −1.4 ± 0.1 |
| Fenuron (FU) | −2.8 ± 0.2 | −2.2 ± 0.6 | −1.8 ± 0.2 | −1.7 ± 0.2 |
| Methiuron (MU) | −2.7 ± 0.7 | −2.9 ± 0.5 | −2.0 ± 0.4 | −2.8 ± 0.4 |
| Phenylurea | −3.4 ± 0.6 | −3.5 ± 0.3 | −2.4 ± 0.2 | −2.6 ± 0.4 |
| Phenylalanine | −4.7 ± 0.8 | −4.6 ± 0.9 | −3.1 ± 0.3 | −3.8 ± 0.2 |
| Urea | −5.1 ± 0.2 | −5.2 ± 0.1 | −4.3 ± 0.5 | −4.8 ± 0.4 |
| NH4+ | −5.6 ± 0.3 | −5.5 ± 0.4 | −4.6 ± 0.4 | −5.2 ± 0.2 |
| K+ | −5.9 ± 0.2 | −5.8 ± 0.3 | −4.8 ± 0.1 | −4.6 ± 0.5 |
| Na+ | −6.1 ± 0.1 | −5.7 ± 0.9 | −5.3 ± 0.3 | −5.6 ± 0.2 |
| Ca2+ | −6.4 ± 0.2 | −6.6 ± 0.1 | −5.4 ± 0.6 | −5.5 ± 0.1 |
| Sample | * Concentration of Phenylurea Herbicide, µg/mL | ||||
|---|---|---|---|---|---|
| Amount Added | (IPU)/MWCNTs-ISEs | MIP (DU)/MWCNTs-ISEs | |||
| Found, IPU | Recovery, % | Found, DU | Recovery, % | ||
| Sample 1 | 0.5 | 0.53 ± 0.2 | 106.1 | 0.52 ± 0.3 | 104.0 |
| Sample2 | 2.5 | 2.42 ± 0.4 | 96.8 | 2.53 ± 0.2 | 101.2 |
| Sample3 | 5.0 | 4.87 ± 0.3 | 97.4 | 4.93 ± 0.4 | 98.6 |
| Sample 4 | 10.0 | 9.7 ± 0.4 | 97.0 | 9.6 ± 0.5 | 96.0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kamel, A.H.; Amr, A.E.-G.E.; Al-Omar, M.A.; Almehizia, A.A. Solid-State Membrane Sensors Based on Man-Tailored Biomimetic Receptors for Selective Recognition of Isoproturon and Diuron Herbicides. Membranes 2020, 10, 279. https://doi.org/10.3390/membranes10100279
Kamel AH, Amr AE-GE, Al-Omar MA, Almehizia AA. Solid-State Membrane Sensors Based on Man-Tailored Biomimetic Receptors for Selective Recognition of Isoproturon and Diuron Herbicides. Membranes. 2020; 10(10):279. https://doi.org/10.3390/membranes10100279
Chicago/Turabian StyleKamel, Ayman H., Abd El-Galil E. Amr, Mohamed A. Al-Omar, and Abdulrahman A. Almehizia. 2020. "Solid-State Membrane Sensors Based on Man-Tailored Biomimetic Receptors for Selective Recognition of Isoproturon and Diuron Herbicides" Membranes 10, no. 10: 279. https://doi.org/10.3390/membranes10100279
APA StyleKamel, A. H., Amr, A. E.-G. E., Al-Omar, M. A., & Almehizia, A. A. (2020). Solid-State Membrane Sensors Based on Man-Tailored Biomimetic Receptors for Selective Recognition of Isoproturon and Diuron Herbicides. Membranes, 10(10), 279. https://doi.org/10.3390/membranes10100279








