Varicella Zoster Virus Reactivation Following COVID-19 Vaccination: A Systematic Review of Case Reports
Abstract
:1. Introduction
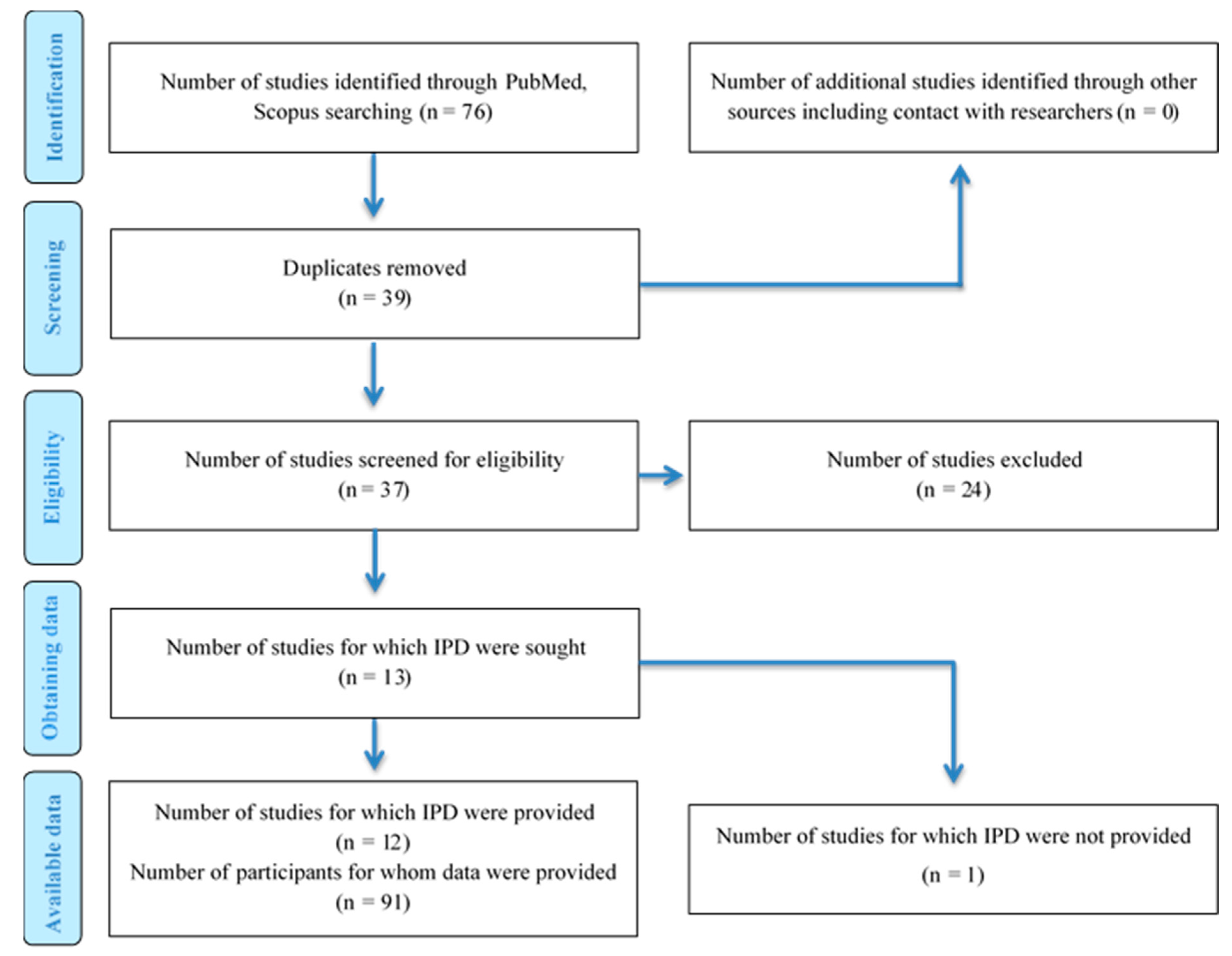
2. Methods
2.1. Literature Search
2.2. Eligibility Criteria
2.3. Data Extraction and Handling
2.4. Quality Assessment
3. Results
3.1. Study Characteristics
3.2. Quality of the Studies
4. Discussion
Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lu, H.; Stratton, C.W.; Tang, Y.W. Outbreak of pneumonia of unknown etiology in Wuhan, China: The mystery and the miracle. J. Med. Virol. 2020, 92, 401–402. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, X.; Yu, Y.; Xu, J.; Shu, H.; Liu, H.; Wu, Y.; Zhang, L.; Yu, Z.; Fang, M.; Yu, T. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: A single-centered, retrospective, observational study. Lancet Respir. Med. 2020, 8, 475–481. [Google Scholar] [CrossRef] [Green Version]
- Nicola, M.; Alsafi, Z.; Sohrabi, C.; Kerwan, A.; Al-Jabir, A.; Iosifidis, C.; Agha, M.; Agha, R. The socio-economic implications of the coronavirus pandemic (COVID-19): A review. Int. J. Surg. 2020, 78, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Loubet, P.; Wittkop, L.; Tartour, E.; Parfait, B.; Barrou, B.; Blay, J.-Y.; Hourmant, M.; Lachâtre, M.; Laplaud, D.-A.; Laville, M. A French cohort for assessing COVID-19 vaccine responses in specific populations. Nat. Med. 2021, 27, 1319–1321. [Google Scholar] [CrossRef]
- Johnson, J. Johnson & Johnson COVID-19 Vaccine Authorized by US FDA For Emergency Use-First Single-Shot Vaccine in Fight Against Global Pandemic; Johnson & Johnson: New Brunswick, NJ, USA, 2021. [Google Scholar]
- Mulligan, M.J.; Lyke, K.E.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Neuzil, K.; Raabe, V.; Bailey, R.; Swanson, K.A. Phase I/II study of COVID-19 RNA vaccine BNT162b1 in adults. Nature 2020, 586, 589–593. [Google Scholar] [CrossRef]
- Barrett, J.R.; Belij-Rammerstorfer, S.; Dold, C.; Ewer, K.J.; Folegatti, P.M.; Gilbride, C.; Halkerston, R.; Hill, J.; Jenkin, D.; Stockdale, L. Phase 1/2 trial of SARS-CoV-2 vaccine ChAdOx1 nCoV-19 with a booster dose induces multifunctional antibody responses. Nat. Med. 2021, 27, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Anderson, E.J.; Rouphael, N.G.; Widge, A.T.; Jackson, L.A.; Roberts, P.C.; Makhene, M.; Chappell, J.D.; Denison, M.R.; Stevens, L.J.; Pruijssers, A.J. Safety and immunogenicity of SARS-CoV-2 mRNA-1273 vaccine in older adults. N. Engl. J. Med. 2020, 383, 2427–2438. [Google Scholar] [CrossRef]
- Kaur, S.P.; Gupta, V. COVID-19 Vaccine: A comprehensive status report. Virus Res. 2020, 288, 198114. [Google Scholar] [CrossRef]
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Diemert, D.; Spector, S.A.; Rouphael, N.; Creech, C.B. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N. Engl. J. Med. 2021, 384, 403–416. [Google Scholar] [CrossRef]
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Marc, G.P.; Moreira, E.D.; Zerbini, C. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef]
- Pormohammad, A.; Zarei, M.; Ghorbani, S.; Mohammadi, M.; Razizadeh, M.H.; Turner, D.L.; Turner, R.J. Efficacy and Safety of COVID-19 Vaccines: A Systematic Review and Meta-Analysis of Randomized Clinical Trials. Vaccines 2021, 9, 467. [Google Scholar] [CrossRef] [PubMed]
- Recalcati, S. Cutaneous manifestations in COVID-19: A first perspective. J. Eur. Acad. Derm. Venereol. 2020, 34, e212–e213. [Google Scholar] [CrossRef] [PubMed]
- Bostan, E.; Yalici-Armagan, B. Herpes zoster following inactivated COVID-19 vaccine: A coexistence or coincidence? J. Cosmet. Derm. 2021, 20, 1566–1567. [Google Scholar] [CrossRef]
- Gnann, J.W., Jr.; Whitley, R.J. Clinical practice. Herpes zoster. N. Engl. J. Med. 2002, 347, 340–346. [Google Scholar] [CrossRef] [PubMed]
- Marra, F.; Parhar, K.; Huang, B.; Vadlamudi, N. Risk factors for herpes zoster infection: A meta-analysis. Open Forum Infect Dis. 2020, 7, ofaa005. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, P. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kelly, W.N.; Arellano, F.M.; Barnes, J.; Bergman, U.; Edwards, I.R.; Fernandez, A.M.; Freedman, S.B.; Goldsmith, D.I.; Huang, K.; Jones, J.K. Guidelines for submitting adverse event reports for publication. Pharm. Drug Saf. 2007, 16, 581–587. [Google Scholar]
- Aksu, S.B.; Öztürk, G.Z. A rare case of shingles after COVID-19 vaccine: Is it a possible adverse effect? Clin. Exp. Vaccine Res. 2021, 10, 198–201. [Google Scholar] [CrossRef]
- Alpalhão, M.; Filipe, P. Herpes Zoster following SARS-CoV-2 vaccination–a series of 4 cases. J. Eur. Acad. Dermatol. Venereol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Arora, P.; Sardana, K.; Mathachan, S.R.; Malhotra, P. Herpes zoster after inactivated COVID-19 vaccine: A cutaneous adverse effect of the vaccine. J. Cosmet. Derm. 2021. [Google Scholar] [CrossRef]
- Català, A.; Muñoz-Santos, C.; Galván-Casas, C.; Roncero Riesco, M.; Revilla Nebreda, D.; Solá-Truyols, A.; Giavedoni, P.; Llamas-Velasco, M.; González-Cruz, C.; Cubiró, X. Cutaneous reactions after SARS-COV-2 vaccination: A cross-sectional Spanish nationwide study of 405 cases. Br. J. Derm. 2021. [Google Scholar] [CrossRef] [PubMed]
- Chiu, H.-H.; Wei, K.-C.; Chen, A.; Wang, W.-H. Herpes zoster following COVID-19 vaccine: A report of three cases. QJM Int. J. Med. 2021, hcab208. [Google Scholar] [CrossRef]
- Eid, E.; Abdullah, L.; Kurban, M.; Abbas, O. Herpes zoster emergence following mRNA COVID-19 vaccine. J. Med. Virol. 2021, 93, 5231–5232. [Google Scholar] [CrossRef]
- Furer, V.; Zisman, D.; Kibari, A.; Rimar, D.; Paran, Y.; Elkayam, O. Herpes zoster following BNT162b2 mRNA Covid-19 vaccination in patients with autoimmune inflammatory rheumatic diseases: A case series. Rheumatology 2021. [Google Scholar] [CrossRef]
- Lee, C.; Cotter, D.; Basa, J.; Greenberg, H.L. 20 Post-COVID-19 vaccine-related shingles cases seen at the Las Vegas Dermatology clinic and sent to us via social media. J. Cosmet. Derm. 2021, 20, 1960–1964. [Google Scholar] [CrossRef]
- Psichogiou, M.; Samarkos, M.; Mikos, N.; Hatzakis, A. Reactivation of Varicella Zoster Virus after Vaccination for SARS-CoV-2. Vaccines 2021, 9, 572. [Google Scholar] [CrossRef]
- Rodríguez-Jiménez, P.; Chicharro, P.; Cabrera, L.-M.; Seguí, M.; Morales-Caballero, Á.; Llamas-Velasco, M.; Sánchez-Pérez, J. Varicella-Zoster virus reactivation after SARS-CoV-2 BNT162b2 mRNA vaccination: Report of 5 cases. Jaad Case Rep. 2021, 12, 58–59. [Google Scholar] [CrossRef]
- Tessas, I.; Kluger, N. Ipsilateral Herpes Zoster after the first dose of BNT162b2 mRNA COVID-19 vaccine. J. Eur. Acad. Derm. Venereol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Eshleman, E.; Shahzad, A.; Cohrs, R.J. Varicella zoster virus latency. Future Virol. 2011, 6, 341–355. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Depledge, D.P.; Sadaoka, T.; Ouwendijk, W.J. Molecular aspects of varicella-zoster virus latency. Viruses 2018, 10, 349. [Google Scholar] [CrossRef] [Green Version]
- Crooke, S.N.; Ovsyannikova, I.G.; Poland, G.A.; Kennedy, R.B. Immunosenescence and human vaccine immune responses. Immun. Ageing 2019, 16, 25. [Google Scholar] [CrossRef] [Green Version]
- Zak-Prelich, M.; Borkowski, J.; Alexander, F.; Norval, M. The role of solar ultraviolet irradiation in zoster. Epidemiol. Infect. 2002, 129, 593–597. [Google Scholar] [CrossRef] [PubMed]
- Medicines & Healthcare Products Regulatory Agency. Coronavirus Vaccine Weekly Summary of Yellow Card Reporting. gov.uk. 2021. Available online: https://www.gov.uk/government/publications/coronavirus-covid-19-vaccine-adverse-reactions/coronavirus-vaccine-summary-of-yellow-card-reporting (accessed on 24 July 2021).
- Walter, R.; Hartmann, K.; Fleisch, F.; Reinhart, W.H.; Kuhn, M. Reactivation of herpesvirus infections after vaccinations? Lancet 1999, 353, 810. [Google Scholar] [CrossRef]
- Bo, X.; Fan, C.-Y.; Wang, A.-L.; Zou, Y.-L.; Yu, Y.-H.; Cong, H.; Xia, W.-G.; Zhang, J.-X.; Qing, M. Suppressed T cell-mediated immunity in patients with COVID-19: A clinical retrospective study in Wuhan, China. J. Infect. 2020, 81, e51–e60. [Google Scholar]
- Diez-Domingo, J.; Parikh, R.; Bhavsar, A.B.; Cisneros, E.; McCormick, N.; Lecrenier, N. Can COVID-19 Increase the Risk of Herpes Zoster? A Narrative Review. Dermatol. Ther. 2021, 11, 1119–1126. [Google Scholar] [CrossRef]
- Pardi, N.; Hogan, M.J.; Porter, F.W.; Weissman, D. mRNA vaccines—A new era in vaccinology. Nat. Rev. Drug Discov. 2018, 17, 261–279. [Google Scholar] [CrossRef] [Green Version]
- Zhang, C.; Maruggi, G.; Shan, H.; Li, J. Advances in mRNA vaccines for infectious diseases. Front. Immunol. 2019, 10, 594. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- West, J.A.; Gregory, S.M.; Damania, B. Toll-like receptor sensing of human herpesvirus infection. Front. Cell. Infect. Microbiol. 2012, 2, 122. [Google Scholar] [CrossRef] [Green Version]
| Author, Year | Country | Study Design | N of Patients (Males/Females) | Mean Age | Comorbidities (N of Cases) | Vaccine Type (N of Patients) | Vaccine Doses before Symptoms (N of Patients) | Days of HZ Onset after Vaccination (Mean) | Treatment (Duration) |
|---|---|---|---|---|---|---|---|---|---|
| Aksu, 2021 [19] | Turkey | Case study | 1 (1/0) | 68 |
| N/A | 2nd dose (1) | 5 | Valaciclovir (7 days) |
| Alpalhão, 2021 [20] | Portugal | Case series | 4 (1/3) | 69 |
| Pfizer-BNT162b2 (2) AstraZeneca- ChAdOx1-nCoV19 (2) | 1st dose (2) 2nd dose (2) | 4 | Valacyclovir (N/A) |
| Arora, 2021 [21] | India | Case study | 1 (1/0) | 60 |
| AstraZeneca- ChAdOx1-nCoV19 (1) | 1st dose (1) | 4 | Valacyclovir, Fusidic acid (7 days) |
| Bostan, 2021 [14] | Turkey | Case study | 1 (1/0) | 78 |
| N/A | N/A | 5 | Valacyclovir (7 days) |
| Català, 2021 [22] | Spain | Cross sectional | 41 (16/25) | 61 |
| Pfizer-BNT162b2 (28) Moderna-mRNA-1273 (6) AstraZeneca- ChAdOx1-nCoV19 (7) | 1st dose (26) 2nd dose (15) | 6.9 | N/A |
| Chiu, 2021 [23] | Taiwan | Case series | 3 (3/0) | 53 | N/A | Moderna-mRNA-1273 (1) AstraZeneca- ChAdOx1-nCoV19 (2) | 1st dose (3) | 3.6 | Acyclovir (7 days) |
| Eid, 2021 [24] | Lebanon | Case study | 1 (1/0) | 79 |
| N/A | N/A | 5 | N/A |
| Furer, 2021 [25] | Israel | Case series | 6 (0/6) | 49 |
| Pfizer-BNT162b2 (6) | 1st dose (1) 2nd dose (5) | 8 | Valacyclovir, Acyclovir (7 days) |
| Lee, 2021 [26] | USA | Case series | 20 (10/10) | 56 |
| Moderna-mRNA-1273 (14) Pfizer-BNT162b2 (6) | 1st dose (15) 2nd dose (5) | 6.9 | Valacyclovir (N/A) |
| Psichogiou, 2021 [27] | Greece | Case series | 7 (4/3) | 77 |
| Pfizer-BNT162b2 (7) | 1st dose (1) 2nd dose (6) | 9 | Valacyclovir (N/A) |
| Rodriguez-Jimenez, 2021 [28] | Spain | Case series | 5 (2/3) | 48 |
| Pfizer-BNT162b2 (5) | 1st dose (3) 2nd dose (2) | 5.4 | N/A |
| Tessas, 2021 [29] | Finland | Case study | 1 (1/0) | 44 |
| Pfizer-BNT162b2 (1) | 1st dose (1) | 7 | Valacyclovir (14 days) |
| Author, Year | Case Number | Dermatome/Anatomical Site |
|---|---|---|
| Aksu, 2021 | Case 1 | T3-T5 (Right chest) |
| Alpalhão, 2021 | Case 1 | 5th cranial nerve |
| Case 2 | 5th cranial nerve | |
| Case 3 | C8 | |
| Case 4 | 5th cranial nerve | |
| Arora, 2021 | Case 1 | L2–L3 (Right leg) |
| Bostan, 2021 | Case 1 | T3–T5 (Right chest) |
| Català, 2021 | Case 1–41 | N/A |
| Chiu, 2021 | Case 1 | T8 |
| Case 2 | T10 | |
| Case 3 | T11 | |
| Eid, 2021 | Case 1 | Right leg |
| Furer, 2021 | Case 1 | L5 |
| Case 2 | 5th cranial nerve | |
| Case 3 | L1–L2 | |
| Case 4 | T10 | |
| Case 5 | T4 | |
| Case 6 | T6 | |
| Lee, 2021 | Case 1 | Mid-abdomen, right flank, and right mid-back |
| Case 2 | Left axilla, left shoulder, left triceps | |
| Case 3 | Right chest | |
| Case 4 | Left back, left shoulder, left triceps | |
| Case 5 | Right neck, right collarbone, right lower jaw | |
| Case 6 | Left axilla, left upper chest | |
| Case 7 | N/A | |
| Case 8 | T1 | |
| Case 9 | Right back, right flank | |
| Case 10 | Mid chest, right arm | |
| Case 11 | Left flank | |
| Case 12 | Left axilla, left triceps, left scapula | |
| Case 13 | Right flank | |
| Case 14 | Right forehead | |
| Case 15 | Right flank, back | |
| Case 16 | Under right eye | |
| Case 17 | Left eyebrow | |
| Case 18 | Left back, left abdomen | |
| Case 19 | Right back, right arm | |
| Case 20 | Left arm, left upper back, left breast | |
| Psichogiou, 2021 | Case 1 | Lumbar region |
| Case 2 | Thoracic region (Right chest) | |
| Case 3 | 5th cranial nerve | |
| Case 4 | Thoracic region (Right chest) | |
| Case 5 | Thoracic region (Right chest) | |
| Case 6 | 5th cranial nerve | |
| Case 7 | Thoracic region (Right chest) | |
| Rodriguez, 2021 | Case 1 | C6 |
| Case 2 | Dorsal 2-Dorsal 4 | |
| Case 3 | Dorsal 4 | |
| Case 4 | 5th cranial nerve | |
| Case 5 | Dorsal 5 | |
| Tessas, 2021 | Case 1 | C5-C6 (Left upper back, left arm) |
| Author, Year | Title | Demographics | Current Health Status | Medical History | Physical Examination | Patient Disposition | Drug Identification | Dosage | Administration Drug- Reaction Interface | Concomitant Therapies | Adverse Events | Discussion | Overall Rating |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Aksu, 2021 | ● | ● | ● | ● | ● | ● | ○ | ● | ● | ● | ○ | ● | 10 |
| Alpalhão, 2021 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | 12 |
| Arora, 2021 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | 12 |
| Bostan, 2021 | ● | ● | ● | ● | ● | ○ | ○ | ● | ○ | ● | ○ | ● | 8 |
| Català, 2021 | ● | ● | ○ | ● | ○ | ● | ● | ● | ○ | ○ | ● | ● | 8 |
| Chiu, 2021 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ○ | ○ | ● | 10 |
| Eid, 2021 | ● | ● | ● | ● | ● | ○ | ○ | ● | ○ | ● | ● | ● | 9 |
| Furer, 2021 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | 12 |
| Lee, 2021 | ● | ● | ● | ● | ● | ○ | ● | ● | ● | ○ | ● | ● | 10 |
| Psichogiou, 2021 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | 12 |
| Rodriguez-Jimenez, 2021 | ● | ● | ● | ● | ● | ○ | ● | ● | ● | ● | ○ | ● | 10 |
| Tessas, 2021 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | 12 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Katsikas Triantafyllidis, K.; Giannos, P.; Mian, I.T.; Kyrtsonis, G.; Kechagias, K.S. Varicella Zoster Virus Reactivation Following COVID-19 Vaccination: A Systematic Review of Case Reports. Vaccines 2021, 9, 1013. https://doi.org/10.3390/vaccines9091013
Katsikas Triantafyllidis K, Giannos P, Mian IT, Kyrtsonis G, Kechagias KS. Varicella Zoster Virus Reactivation Following COVID-19 Vaccination: A Systematic Review of Case Reports. Vaccines. 2021; 9(9):1013. https://doi.org/10.3390/vaccines9091013
Chicago/Turabian StyleKatsikas Triantafyllidis, Konstantinos, Panagiotis Giannos, Imran Tariq Mian, George Kyrtsonis, and Konstantinos S. Kechagias. 2021. "Varicella Zoster Virus Reactivation Following COVID-19 Vaccination: A Systematic Review of Case Reports" Vaccines 9, no. 9: 1013. https://doi.org/10.3390/vaccines9091013
APA StyleKatsikas Triantafyllidis, K., Giannos, P., Mian, I. T., Kyrtsonis, G., & Kechagias, K. S. (2021). Varicella Zoster Virus Reactivation Following COVID-19 Vaccination: A Systematic Review of Case Reports. Vaccines, 9(9), 1013. https://doi.org/10.3390/vaccines9091013








