Can Vaccination Trigger Autoimmune Disorders? A Meta-Analysis
Abstract
:1. Introduction
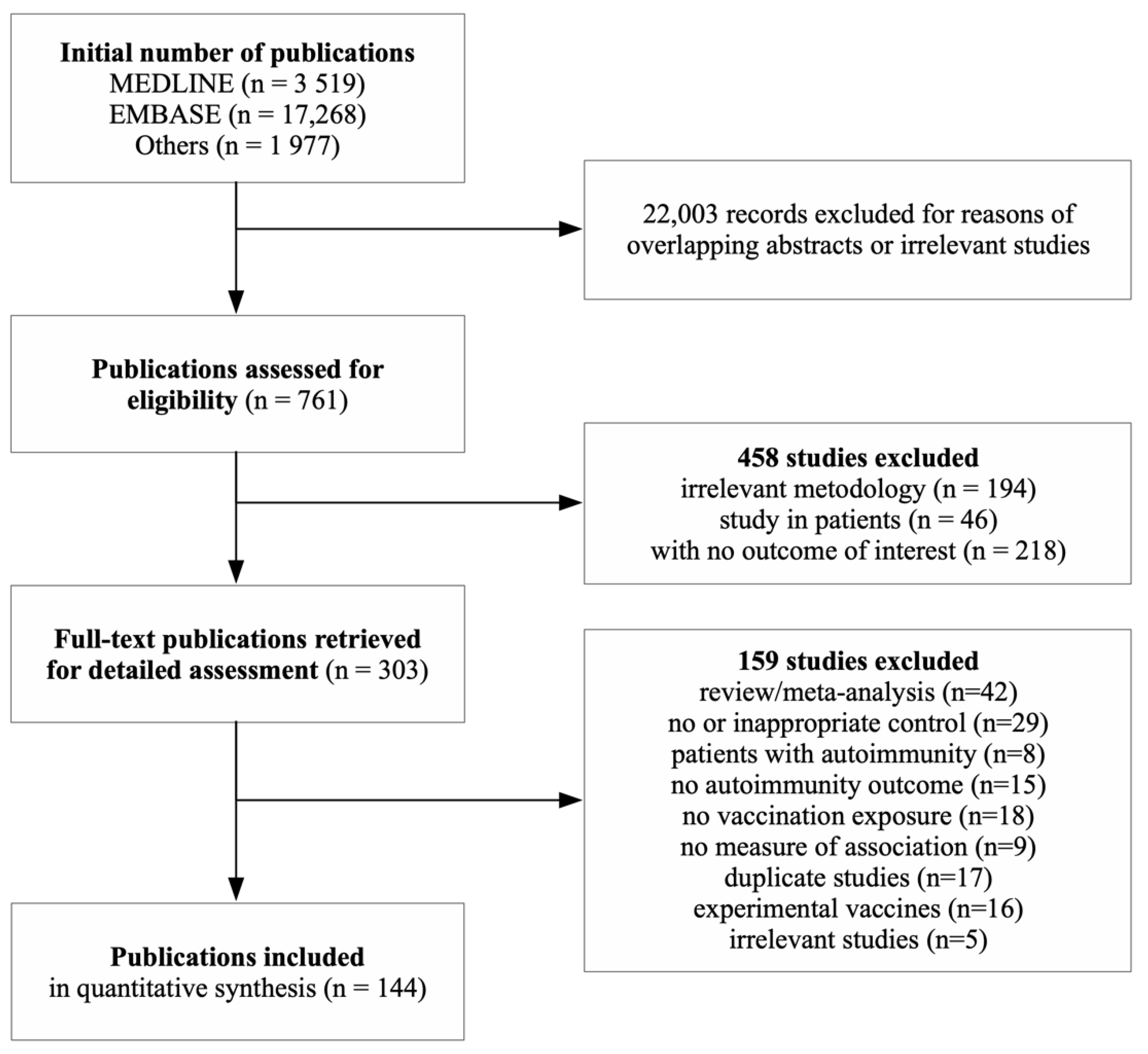
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Alter, M.; Speer, J. Clinical evaluation of possible etiologic factors in multiple sclerosis. Neurology 1968, 18, 109–116. [Google Scholar] [CrossRef]
- Stratton, K.R.; Howe, C.J.; Johnston, R.B., Jr. Adverse events associated with childhood vaccines other than pertussis and rubella. Summary of a report from the Institute of Medicine. JAMA 1994, 271, 602–605. [Google Scholar] [CrossRef]
- Jacobson, D.L.; Gange, S.J.; Rose, N.R.; Graham, N.M. Epidemiology and estimated population burden of selected autoimmune diseases in the United States. Clin. Immunol. Immunopathol. 1997, 84, 223–243. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Onkamo, P.; Väänänen, S.; Karvonen, M.; Tuomilehto, J. Worldwide increase in incidence of Type I diabetes—the analysis of the data on published incidence trends. Diabetologia 1999, 42, 1395–1403. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lerner, A.; Jeremias, P.; Matthias, T. The world incidence and prevalence of autoimmune diseases is increasing. Int. J. Celiac. Dis. 2015, 3, 151–155. [Google Scholar] [CrossRef] [Green Version]
- Wraith, D.C.; Goldman, M.; Lambert, P.H. Vaccination and autoimmune disease, what is the evidence? Lancet 2003, 362, 1659–1666. [Google Scholar] [CrossRef]
- Albert, L.J.; Inman, R.D. Molecular mimicry and autoimmunity. N. Engl. J. Med. 1999, 341, 2068–2074. [Google Scholar] [CrossRef]
- Langridge, W.H. Edible vaccines. Sci. Am. 2000, 283, 66–71. [Google Scholar] [CrossRef]
- Todd, J.A.; Wicker, L.S. Genetic protection from the inflammatory disease type 1 diabetes in humans and animal models. Immunity 2001, 15, 387–395. [Google Scholar] [CrossRef] [Green Version]
- Regner, M.; Lambert, P.H. Autoimmunity through infection or immunization? Nat. Immunol. 2001, 2, 185–188. [Google Scholar] [CrossRef] [PubMed]
- Duke, R.C. Self recognition by T cells. I. Bystander killing of target cells bearing syngeneic MHC antigens. J. Exp. Med. 1989, 170, 59–71. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses, the PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [Green Version]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B. Meta-analysis of observational studies in epidemiology, a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA 2000, 283, 2008–2012. [Google Scholar] [CrossRef] [PubMed]
- Wells, G.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 11 December 2019).
- Guyatt, G.; Oxman, A.D.; Akl, E.A.; Kunz, R.; Vist, G.; Brozek, J.; Norris, S.; Falck-Ytter, Y.; Glasziou, P.; DeBeer, H.; et al. GRADE guidelines, 1. Introduction-GRADE evidence profiles and summary of findings tables. J. Clin. Epidemiol. 2011, 64, 383–394. [Google Scholar] [CrossRef]
- Deeks, J.J.; Higgins, J.P.; Altman, D.G. Analysing data and undertaking meta-analyses. In Cochrane Handbook for Systematic Reviews of Interventions, 2nd ed.; Thomas, J., Chandler, J., Cumpston, M., Li, T., Page, M.J., Welch, V.A., Eds.; John Wiley & Sons: Chichester, UK, 2009; pp. 243–296. [Google Scholar]
- Higgins, J.P.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [Green Version]
- Xie, S.; Xu, H.; Shan, X.; Liu, B.; Wang, K.; Cai, Z. Clinicopathological and prognostic significance of survivin expression in patients with oral squamous cell carcinoma, evidence from a meta-analysis. PLoS ONE 2015, 10, e0116517. [Google Scholar] [CrossRef] [PubMed]
- Chaimani, A.; Mavridis, D.; Salanti, G. A hands-on practical tutorial on performing meta-analysis with Stata. Evid. Based Ment. Health 2014, 17, 111–116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hedges, L.V.; Pigott, T.D. The power of statistical tests in meta-analysis. Psychol. Methods 2001, 6, 203–217. [Google Scholar] [CrossRef] [PubMed]
- Jackson, D.; Turner, R. Power analysis for random-effects meta-analysis. Res. Synth. Methods 2017, 8, 290–302. [Google Scholar] [CrossRef] [Green Version]
- Valentine, J.C.; Pigott, T.D.; Rothstein, H.R. How Many Studies Do You Need?, A Primer on Statistical Power for Meta-Analysis. J. Educ. Behav. Stat. 2010, 35, 215–247. [Google Scholar] [CrossRef]
- Genovese, C.; LA Fauci, V.; Squeri, A.; Trimarchi, G.; Squeri, R. HPV vaccine and autoimmune diseases, systematic review and meta-analysis of the literature. J. Prev. Med. Hyg. 2018, 59, E194–E199. [Google Scholar]
- Jiang, H.Y.; Shi, Y.D.; Zhang, X.; Pan, L.Y.; Xie, Y.R.; Jiang, C.M.; Deng, M.; Ruan, B. Human papillomavirus vaccination and the risk of autoimmune disorders, A systematic review and meta-analysis. Vaccine 2019, 37, 3031–3039. [Google Scholar] [CrossRef]
- Elwood, J.M.; Ameratunga, R. Autoimmune diseases after hepatitis B immunization in adults, Literature review and meta-analysis, with reference to ’autoimmune/autoinflammatory syndrome induced by adjuvants’ (ASIA). Vaccine 2018, 36, 5796–5802. [Google Scholar] [CrossRef]
- Morgan, E.; Halliday, S.R.; Campbell, G.R.; Cardwell, C.R.; Patterson, C.C. Vaccinations and childhood type 1 diabetes mellitus, a meta-analysis of observational studies. Diabetologia 2016, 59, 237–243. [Google Scholar] [CrossRef] [Green Version]
- Martínez-Sernández, V.; Figueiras, A. Central nervous system demyelinating diseases and recombinant hepatitis B vaccination, a critical systematic review of scientific production. J. Neurol. 2013, 260, 1951–1959. [Google Scholar] [CrossRef] [PubMed]
- Mouchet, J.; Salvo, F.; Raschi, E.; Poluzzi, E.; Antonazzo, I.C.; De Ponti, F.; Bégaud, B. Human papillomavirus vaccine and demyelinating diseases-A systematic review and meta-analysis. Pharmacol. Res. 2018, 132, 108–118. [Google Scholar] [CrossRef]
- Sestili, C.; Grazina, I.; La Torre, G. HBV vaccine and risk of developing multiple sclerosis, a systematic review and meta-analysis. Hum. Vaccines Immunother. 2018, 17, 2273–2278. [Google Scholar] [CrossRef]
- Meggiolaro, A.; Migliara, G.; La Torre, G. Association between Human Papilloma Virus (HPV) vaccination and risk of Multiple Sclerosis, A systematic review. Hum. Vaccines Immunother. 2018, 14, 1266–1274. [Google Scholar] [CrossRef]
- Mouchet, J.; Salvo, F.; Raschi, E.; Poluzzi, E.; Antonazzo, I.C.; De Ponti, F.; Bégaud, B. Hepatitis B vaccination and the putative risk of central demyelinating diseases—A systematic review and meta-analysis. Vaccine 2018, 36, 1548–1555. [Google Scholar] [CrossRef] [PubMed]
- Mailand, M.T.; Frederiksen, J.L. Vaccines and multiple sclerosis, a systematic review. J. Neurol. 2017, 264, 1035–1050. [Google Scholar] [CrossRef] [PubMed]
- Farez, M.F.; Correale, J. Immunizations and risk of multiple sclerosis, systematic review and meta-analysis. J. Neurol. 2011, 258, 1197–1206. [Google Scholar] [CrossRef] [PubMed]
- Hernán, M.A.; Alonso, A.; Hernández-Díaz, S. Tetanus vaccination and risk of multiple sclerosis, a systematic review. Neurology 2006, 67, 212–215. [Google Scholar] [CrossRef] [PubMed]
- Martín Arias, L.H.; Sanz, R.; Sáinz, M.; Treceño, C.; Carvajal, A. Guillain-Barré syndrome and influenza vaccines, A meta-analysis. Vaccine 2015, 33, 3773–3778. [Google Scholar] [CrossRef] [PubMed]
- Petráš, M.; Lesná, I.K.; Dáňová, J.; Čelko, A.M. Is an Increased Risk of Developing Guillain-Barré Syndrome Associated with Seasonal Influenza Vaccination? A Systematic Review and Meta-Analysis. Vaccines 2020, 8, 150. [Google Scholar] [CrossRef] [Green Version]
- Pineton de Chambrun, G.; Dauchet, L.; Gower-Rousseau, C.; Cortot, A.; Colombel, J.F.; Peyrin-Biroulet, L. Vaccination and Risk for Developing Inflammatory Bowel Disease, A Meta-Analysis of Case-Control and Cohort Studies. Clin. Gastroenterol. Hepatol. 2015, 13, 1405–1415. [Google Scholar] [CrossRef] [PubMed]
- Mellone, N.G.; Silva, M.T.; Paglia, M.D.G.; Lopes, L.C.; Barberato-Filho, S.; Del Fiol, F.S.; Bergamaschi, C.C. Kawasaki Disease and the Use of the Rotavirus Vaccine in Children, A Systematic Review and Meta-Analysis. Front. Pharmacol. 2019, 10, 1075. [Google Scholar] [CrossRef]
- Mantadakis, E.; Farmaki, E.; Buchanan, G.R. Thrombocytopenic purpura after measles-mumps-rubella vaccination, a systematic review of the literature and guidance for management. J. Pediatr. 2010, 156, 623–628. [Google Scholar] [CrossRef]
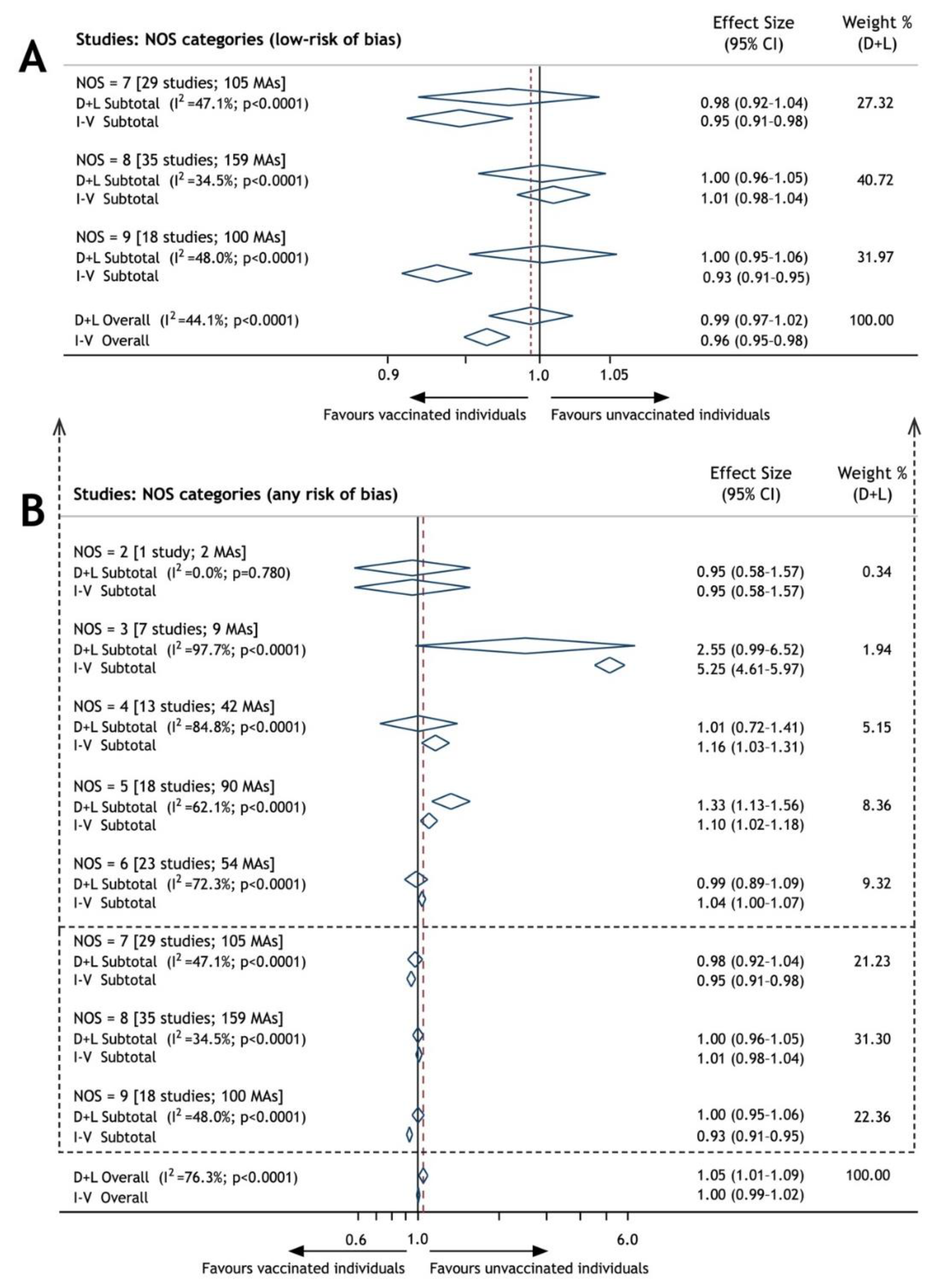
| Studies’ NOS 1-Scaled Groups | Pooled ES 2 (95% CI 3) * | No Serious Publication Bias (Pooled ES; 95% CI) ** | 95% Prediction Interval | Number of MAs 4 | No Serious Studies’ Inconsistency (I2) 5 | No Serious Impression (SE) 6 | Power of Test (%) | Robustness |
|---|---|---|---|---|---|---|---|---|
| 2 | 0.95 (0.58–1.57) | Yes (0.95; 0.58–1.57) | 0.02–39.1 | 2 | Yes (0.0%) | No (0.25) | - | NA 8 |
| 3 | 2.55 (0.99–6.52) | No (5.25; 4.61–5.97) | 0.03–194 | 9 | No (97.7%) | No (0.48) | 43.3 | NA |
| 4 | 1.01 (0.72–1.41) | No (1.16; 1.03–1.31) | 0.10–10.0 | 43 | No (84.8%) | No (0.17) | 8.5 | NA |
| 5 | 1.33 (1.13–1.56) | Yes (1.10; 1.02–1.18) | 0.43–4.14 | 90 | No (62.1%) | Yes (0.08) | 83.0 | NA |
| 6 | 0.99 (0.89–1.09) | No (1.04; 1.01–1.07) | 0.59–1.66 | 54 | No (72.3%) | Yes (0.05) | 92.0 | NA |
| 7 | 0.98 (0.92–1.04) | Yes (0.95; 0.91–0.98) | 0.63–1.51 | 105 | Yes (47.1%) | Yes (0.03) | 99.3 | NA |
| 8 | 1.00 (0.96–1.05) | Yes (1.01; 0.98–1.04) | 0.74–1.36 | 159 | Yes (34.5%) | Yes (0.02) | >99.9 | NA |
| 9 | 1.00 (0.95–1.06) | Yes (0.93; 0.91–0.95) | 0.72–1.40 | 100 | Yes (48.0%) | Yes (0.03) | 99.6 | NA |
| Overall, any RoB 7 | 1.05 (1.01–1.09) | No (1.00; 0.99–1.02) | 0.55–1.99 | 562 | No (76.3%) | Yes (0.02) | 90.7 | NA |
| Overall, low RoB | 0.99 (0.97–1.02) | Yes (0.96; 0.95–0.98) | 0.73–1.36 | 364 | Yes (44.1%) | Yes (0.02) | >99.9 | Yes |
| Location | Autoimmune Disorders |
|---|---|
| Endocrine | Addison’s disease, Graves–Basedow’s disease, Hashimoto’s thyroiditis, Hyperthyroidism, Hypothyroidism, Pernicious anemia, Type 1 diabetes, Thyroiditis |
| Neurological | Acute disseminated encephalomyelitis, Acquired/Acute demyelinating syndrome, Bell’s palsy, Guillain–Barré syndrome, Myasthenia gravis, Multiple sclerosis, Optic neuritis, Narcolepsy |
| Gastrointestinal | Autoimmune hepatitis, Celiac disease, Crohn’s disease, Inflammatory bowel disease, Pancreatitis, Ulcerative colitis |
| Haematological | Autoimmune haemolytic anemia, Immune thrombocytopenia/Idiopathic thrombocytopenic purpura |
| Dermatological | Alopecia areata, Psoriasis, Vitiligo |
| Connective tissue | Ankylosing spondylitis, Sjögren’s syndrome, Inflammatory polyarthritis, Juvenile arthritis, Myositis, Psoriatic arthritis, Rheumatoid arthritis, Scleroderma (local and/or systemic), Systemic lupus |
| Vasculitis | Behcet’s disease, Erythema nodosum, Henoch–Schönlein purpura, Kawasaki syndrome, Polyarteritis nodosa, Vasculitis, Wegener’s granulomatosis |
| Others | Glomerulonephritis, Raynaud’s syndrome, Uveitis |
| Subgroups | Pooled ES 1 (95% CI 2) * | No Serious Publication Bias (Pooled ES; 95% CI) ** | 95% Prediction Interval | Number of MAs 3 | No Serious Studies’ Inconsistency (I2) 4 | No Serious Impression (SE) 5 | Power of Test (%) | Robustness (Pooled ES; 95% CI) † |
|---|---|---|---|---|---|---|---|---|
| Autoimmune disorders | ||||||||
| Endocrine | 0.96 (0.92–1.00) | Yes (0.97; 0.95–1.00) | 0.72–1.29 | 120 | Yes (45.6%) | Yes (0.02) | 90.0 | Yes (0.93; 0.89–0.98) |
| Neurological | 1.02 (0.95–1.10) | Yes (0.91; 0.89–0.94) | 0.65–1.60 | 95 | Yes (47.0%) | Yes (0.04) | 73.0 | No (1.10; 1.02–1.20) |
| Gastrointestinal | 1.03 (0.93–1.13) | No (1.06; 1.00–1.12) | 0.63–1.66 | 41 | No (50.9%) | Yes (0.05) | 40.3 | Yes (1.05; 0.96–1.14) |
| Hematologic | 1.12 (0.91–1.36) | Yes (1.09; 0.97–1.24) | 0.43–2.89 | 27 | No (52.4%) | No (0.10) | 5.2 | No (1.22; 1.02–1.47) |
| Dermatological | 0.98 (0.85–1.13) | Yes (0.98; 0.87–1.11) | 0.68–1.41 | 10 | Yes (7.9%) | Yes (0.07) | 17.5 | Yes (1.28; 0.86–1.90) |
| Connective tissue | 0.98 (0.93–1.04) | Yes (0.98; 0.93–1.04) | 0.87–1.10 | 47 | Yes (<0.1%) | Yes (0.03) | 5.0 | Yes (1.07; 0.97–1.19) |
| Vasculitis | 1.09 (0.91–1.29) | Yes (1.08; 0.94–1.25) | 0.60–1.96 | 20 | Yes (22.6%) | Yes (0.09) | 5.3 | Yes (1.12; 0.96–1.31) |
| Type of vaccine | ||||||||
| Killed | 1.00 (0.97–1.03) | Yes (1.00; 0.98–1.02) | 0.73–1.36 | 251 | Yes (38.0%) | Yes (0.02) | >99.9 | No (1.07; 1.01–1.12) |
| Live | 0.99 (0.92–1.07) | Yes (0.94; 0.90–0.98) | 0.60–1.62 | 72 | No (54.4%) | Yes (0.04) | 90.5 | Yes (1.00; 0.93–1.07) |
| Unspecific | 1.03 (0.93–1.15) | Yes (0.90; 0.87–0.93) | 0.73–1.47 | 19 | Yes (41.4%) | Yes (0.06) | 41.2 | Yes (1.09; 0.95–1.25) |
| Lipid-adjuvants | 0.94 (0.83–1.07) | Yes (0.95; 0.85–1.06) | 0.63–1.41 | 22 | Yes (14.5%) | Yes (0.07) | 68.3 | Yes (0.96; 0.86–1.06) |
| Study methodology | ||||||||
| Case–control | 0.96 (0.91–0.99) | Yes (0.91; 0.89–0.93) | 0.74–1.22 | 152 | Yes (21.6%) | Yes (0.02) | >99.9 | Yes (1.03; 0.98–1.09) |
| Cohort | 1.00 (0.95–1.04) | Yes (0.98; 0.95–1.00) | 0.67–1.47 | 176 | Yes (49.4%) | Yes (0.02) | >99.9 | Yes (1.05; 0.97–1.13) |
| Modified | 1.08 (0.99–1.16) | Yes (1.03; 0.99–1.07) | 0.82–1.41 | 36 | Yes (26.9%) | Yes (0.04) | 11.6 | No (1.11; 1.01–1.22) |
| Measures of association | ||||||||
| Crude | 0.97 (0.90–1.05) | Yes (0.97; 0.92–1.02) | 0.60–1.56 | 94 | Yes (35.7%) | Yes (0.04) | 96.4 | Yes (1.07; 0.98–1.18) |
| Adjusted | 1.00 (0.97–1.03) | Yes (0.96; 0.95–0.98) | 0.74–1.36 | 270 | Yes (46.7%) | Yes (0.02) | >99.9 | Yes (1.04; 0.99–1.08) |
| Age of study population | ||||||||
| <18 years | 1.02 (0.98–1.06) | Yes (1.02; 0.99–1.04) | 0.78–1.32 | 184 | Yes (27.3%) | Yes (0.02) | 99.9 | Yes (1.00; 0.95–1.04) |
| >18 years | 0.99 (0.94–1.05) | Yes (0.96; 0.93–1.00) | 0.65–1.50 | 91 | No (56.3%) | Yes (0.03) | 98.7 | Yes (1.06; 0.99–1.13) |
| General population | 0.95 (0.90–1.02) | Yes (0.90 (0.88–0.93) | 0.65–1.39 | 89 | Yes (42.3%) | Yes (0.03) | >99.9 | Yes (1.09; 0.99–1.21) |
| Study locality | ||||||||
| Asia | 1.09 (0.94–1.26) | Yes (1.09; 0.94–1.26) | 0.80–1.48 | 16 | Yes (<0.1%) | Yes (0.08) | 5.0 | Yes (1.28; 0.84–1.94) |
| Australia | 1.10 (0.86–1.41) | Yes (1.10; 0.86–1.41) | 0.63–1.95 | 6 | Yes (<0.1%) | No (0.13) | 5.0 | Yes (0.92; 0.77–1.09) |
| Europe | 1.01 (0.98–1.04) | Yes (0.97; 0.95–0.99) | 0.76–1.33 | 229 | Yes (41.2%) | Yes (0.02) | >99.9 | Yes (1.02; 0.98–1.06) |
| North America | 0.96 (0.90–1.02) | Yes (0.92; 0.89–0.96) | 0.58–1.57 | 113 | No (53.0%) | Yes (0.03) | 99.9 | Yes (1.07; 0.99–1.15) |
| Year of publication | ||||||||
| 1990 or older | 1.04 (0.83–1.31) | Yes (1.04; 0.83–1.31) | 0.62–1.75 | 7 | Yes (<0.1%) | No (0.12) | 5.0 | Yes (0.88; 0.75–1.03) |
| 1991–2000 | 0.98 (0.91–1.06) | Yes (0.98; 0.92–1.05) | 0.77–1.25 | 43 | Yes (9.6%) | Yes (0.04) | 87.5 | Yes (0.93; 0.86–1.01) |
| 2001–2010 | 1.00 (0.93–1.08) | Yes (0.99; 0.94–1.04) | 0.63–1.58 | 73 | Yes (44.5%) | Yes (0.04) | 82.5 | Yes (1.04; 0.97–1.11) |
| 2011 or newer | 1.00 (0.96–1.03) | Yes (0.96; 0.94–0.98) | 0.72–1.37 | 241 | Yes (48.5%) | Yes (0.02) | >99.9 | No (1.09; 1.03–1.15) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petráš, M.; Lesná, I.K.; Dáňová, J.; Čelko, A.M. Can Vaccination Trigger Autoimmune Disorders? A Meta-Analysis. Vaccines 2021, 9, 821. https://doi.org/10.3390/vaccines9080821
Petráš M, Lesná IK, Dáňová J, Čelko AM. Can Vaccination Trigger Autoimmune Disorders? A Meta-Analysis. Vaccines. 2021; 9(8):821. https://doi.org/10.3390/vaccines9080821
Chicago/Turabian StylePetráš, Marek, Ivana Králová Lesná, Jana Dáňová, and Alexander M. Čelko. 2021. "Can Vaccination Trigger Autoimmune Disorders? A Meta-Analysis" Vaccines 9, no. 8: 821. https://doi.org/10.3390/vaccines9080821
APA StylePetráš, M., Lesná, I. K., Dáňová, J., & Čelko, A. M. (2021). Can Vaccination Trigger Autoimmune Disorders? A Meta-Analysis. Vaccines, 9(8), 821. https://doi.org/10.3390/vaccines9080821






