Immunization and Immunotherapy Approaches against Pseudomonas aeruginosa and Burkholderia cepacia Complex Infections
Abstract
1. Pseudomonas aeruginosa Infections and Their Current Management
2. Burkholderia cepacia Complex Infections and Their Current Management
3. Immunization Strategies against P. aeruginosa and Bcc Infections
4. Immunotherapy Strategies against P. aeruginosa and B. cepacia Complex Infections
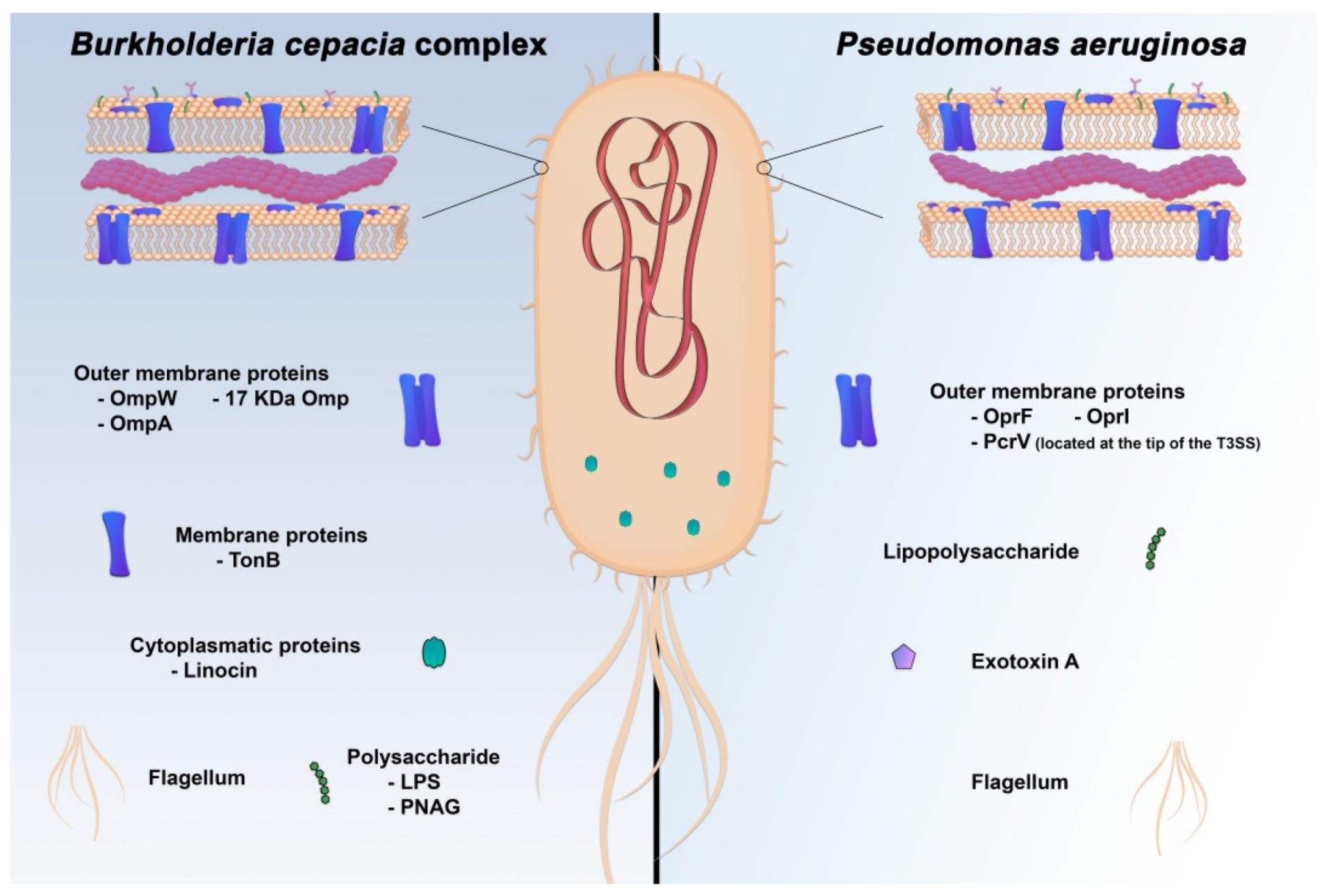
5. Novel Targets for Immunization and Immunotherapy Strategies against P. aeruginosa and Bcc Infections
6. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Gellatly, S.L.; Hancock, R.E.W. Pseudomonas aeruginosa: New insights into pathogenesis and host defenses. Pathog. Dis. 2013, 67, 159–173. [Google Scholar] [CrossRef]
- Malhotra, S.; Hayes, D.; Wozniak, D.J. Cystic Fibrosis and Pseudomonas aeruginosa: The Host-Microbe Interface. Clin. Microbiol. Rev. 2019, 32, e00138-18. [Google Scholar] [CrossRef]
- El Zowalaty, A.; A Al Thani, A.; Webster, T.J.; Schweizer, H.P.; Nasrallah, G.; Marei, H.; Ashour, H. Pseudomonas aeruginosa: Arsenal of resistance mechanisms, decades of changing resistance profiles, and future antimicrobial therapies. Futur. Microbiol. 2015, 10, 1683–1706. [Google Scholar] [CrossRef]
- European Centre for Disease Prevention and Control. Antimicrobial Resistance in the EU/EEA (EARS-Net)—Annual Epidemiological Report 2019; ECDC: Stockholm, Sweden, 2020.
- Horcajada, J.P.; Montero, M.; Oliver, A.; Sorlí, L.; Luque, S.; Gómez-Zorrilla, S.; Benito, N.; Grau, S. Epidemiology and Treatment of Multidrug-Resistant and Extensively Drug-Resistant Pseudomonas aeruginosa Infections. Clin. Microbiol. Rev. 2019, 32, e00031-19. [Google Scholar] [CrossRef]
- Tacconelli, E.; Carrara, E.; Savoldi, A.; Harbarth, S.; Mendelson, M.; Monnet, D.L.; Pulcini, C.; Kahlmeter, G.; Kluytmans, J.; Carmeli, Y.; et al. Discovery, research, and development of new antibiotics: The WHO priority list of antibiotic-resistant bacteria and tuberculosis. Lancet Infect. Dis. 2018, 18, 318–327. [Google Scholar] [CrossRef]
- Langton Hewer, S.C.; Smyth, A.R. Antibiotic strategies for eradicating Pseudomonas aeruginosa in people with cystic fibrosis. Cochrane Database Syst. Rev. 2017, 4, CD004197. [Google Scholar] [CrossRef] [PubMed]
- Bassetti, M.; Vena, A.; Croxatto, A.; Righi, E.; Guery, B. How to manage Pseudomonas aeruginosa infections. Drugs Context 2018, 7, 1–18. [Google Scholar] [CrossRef]
- Ragupathi, N.K.D.; Veeraraghavan, B. Accurate identification and epidemiological characterization of Burkholderia cepacia complex: An update. Ann. Clin. Microbiol. Antimicrob. 2019, 18, 1–10. [Google Scholar] [CrossRef]
- Leitão, J.H.; Sousa, S.A.; Ferreira, A.S.; Ramos, C.G.; Silva, I.N.; Moreira, L. Pathogenicity, virulence factors, and strategies to fight against Burkholderia cepacia complex pathogens and related species. Appl. Microbiol. Biotechnol. 2010, 87, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Depoorter, E.; Bull, M.J.; Peeters, C.; Coenye, T.; Vandamme, P.; Mahenthiralingam, E. Burkholderia: An update on taxonomy and biotechnological potential as antibiotic producers. Appl. Microbiol. Biotechnol. 2016, 100, 5215–5229. [Google Scholar] [CrossRef] [PubMed]
- LiPuma, J.J. The Changing Microbial Epidemiology in Cystic Fibrosis. Clin. Microbiol. Rev. 2010, 23, 299–323. [Google Scholar] [CrossRef]
- Sfeir, M.M. Burkholderia cepacia complex infections: More complex than the bacterium name suggest. J. Infect. 2018, 77, 166–170. [Google Scholar] [CrossRef] [PubMed]
- Gilchrist, F.J.; Webb, A.K.; Bright-Thomas, R.J.; Jones, A. Successful treatment of cepacia syndrome with a combination of intravenous cyclosporin, antibiotics and oral corticosteroids. J. Cyst. Fibros. 2012, 11, 458–460. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.M.; Dodd, M.E.; Govan, J.R.W.; Barcus, V.; Doherty, C.J.; Morris, J.; Webb, A.K. Burkholderia cenocepacia and Burkholderia multivorans: Influence on survival in cystic fibrosis. Thorax 2004, 59, 948–951. [Google Scholar] [CrossRef] [PubMed]
- Chu, K.K.; Davidson, D.J.; Halsey, T.K.; Chung, J.W.; Speert, D.P. Differential Persistence among Genomovars of the Burkholderia cepacia Complex in a Murine Model of Pulmonary Infection. Infect. Immun. 2002, 70, 2715–2720. [Google Scholar] [CrossRef] [PubMed]
- Festini, F.; Buzzetti, R.; Bassi, C.; Braggion, C.; Salvatore, D.; Taccetti, G.; Mastella, G. Isolation measures for prevention of infection with respiratory pathogens in cystic fibrosis: A systematic review. J. Hosp. Infect. 2006, 64, 1–6. [Google Scholar] [CrossRef]
- Regan, K.H.; Bhatt, J. Eradication therapy for Burkholderia cepacia complex in people with cystic fibrosis. Cochrane Database Syst. Rev. 2019, 4, CD009876. [Google Scholar] [CrossRef]
- Saiman, L.; Siegel, J.D.; Lipuma, J.J.; Brown, R.F.; Bryson, E.A.; Chambers, M.J.; Downer, V.S.; Fliege, J.; Hazle, L.A.; Jain, M.; et al. Infection Prevention and Control Guideline for Cystic Fibrosis: 2013 Update. Infect. Control. Hosp. Epidemiology 2014, 35, s1–s67. [Google Scholar] [CrossRef]
- Sousa, S.A.; Ramos, C.G.; Leitão, J.H. Burkholderia cepacia complex: Emerging multihost pathogens equipped with a wide range of virulence factors and determinants. Int. J. Microbiol. 2011, 2011, 607575. [Google Scholar] [CrossRef]
- Callaghan, M.; McClean, S. Bacterial host interactions in cystic fibrosis. Curr. Opin. Microbiol. 2012, 15, 71–77. [Google Scholar] [CrossRef]
- Scoffone, V.C.; Chiarelli, L.R.; Trespidi, G.; Mentasti, M.; Riccardi, G.; Buroni, S. Burkholderia cenocepacia Infections in Cystic Fibrosis Patients: Drug Resistance and Therapeutic Approaches. Front. Microbiol. 2017, 8, 1592. [Google Scholar] [CrossRef] [PubMed]
- Leitão, J.H.; Sousa, S.A.; Cunha, M.V.; Salgado, M.J.; Melo-Cristino, J.; Barreto, M.C.; Sá-Correia, I. Variation of the antimicrobial susceptibility profiles of Burkholderia cepacia complex clonal isolates obtained from chronically infected cystic fibrosis patients: A five-year survey in the major Portuguese treatment center. Eur. J. Clin. Microbiol. Infect. Dis. 2008, 27, 1101–1111. [Google Scholar] [CrossRef] [PubMed]
- Tavares, M.; Kozak, M.; Balola, A.; Sá-Correia, I. Burkholderia cepacia Complex Bacteria: A Feared Contamination Risk in Water-Based Pharmaceutical Products. Clin. Microbiol. Rev. 2020, 33, e00139-19. [Google Scholar] [CrossRef]
- Horsley, A.; Jones, A.M.; Lord, R. Antibiotic treatment for Burkholderia cepacia complex in people with cystic fibrosis experiencing a pulmonary exacerbation. Cochrane Database Syst. Rev. 2016, 2016, CD009529. [Google Scholar] [CrossRef] [PubMed]
- Gautam, V.; Shafiq, N.; Singh, M.; Ray, P.; Singhal, L.; Jaiswal, N.P.; Prasad, A.; Singh, S.; Agarwal, A. Clinical and in vitro evidence for the antimicrobial therapy in Burkholderia cepacia complex infections. Expert Rev. Anti. Infect. Ther. 2015, 13, 629–663. [Google Scholar] [CrossRef] [PubMed]
- Priebe, G.P.; Goldberg, J.B. Vaccines for Pseudomonas aeruginosa: A long and winding road. Expert Rev. Vaccines 2014, 13, 507–519. [Google Scholar] [CrossRef]
- Sainz-Mejías, M.; Jurado-Martín, I.; McClean, S. Understanding Pseudomonas aeruginosa–Host Interactions: The Ongoing Quest for an Efficacious Vaccine. Cells 2020, 9, 2617. [Google Scholar] [CrossRef]
- Hatano, K.; Boisot, S.; DesJardins, D.; Wright, D.C.; Brisker, J.; Pier, G.B. Immunogenic and antigenic properties of a heptavalent high-molecular-weight O-polysaccharide vaccine derived from Pseudomonas aeruginosa. Infect. Immun. 1994, 62, 3608–3616. [Google Scholar] [CrossRef]
- Hancock, E.R.; Mutharia, L.M.; Chan, L.; Darveau, R.P.; Speert, D.P.; Pier, G.B. Pseudomonas aeruginosa isolates from patients with cystic fibrosis: A class of serum-sensitive, nontypable strains deficient in lipopolysaccharide O side chains. Infect. Immun. 1983, 42, 170–177. [Google Scholar] [CrossRef]
- Govan, J.R.; Deretic, V. Microbial pathogenesis in cystic fibrosis: Mucoid Pseudomonas aeruginosa and Burkholderia cepacia. Microbiol. Rev. 1996, 60, 539–574. [Google Scholar] [CrossRef]
- Campodónico, V.L.; Llosa, N.J.; Grout, M.; Döring, G.; Maira-Litrán, T.; Pier, G.B. Evaluation of Flagella and Flagellin of Pseudomonas aeruginosa as Vaccines. Infect. Immun. 2010, 78, 746–755. [Google Scholar] [CrossRef] [PubMed]
- Doring, G.; Meisner, C.; Stern, M.; Flagella Vaccine Trial Study Group. A double-blind randomized placebo-controlled phase III study of a Pseudomonas aeruginosa flagella vaccine in cystic fibrosis patients. Proc. Natl. Acad. Sci. USA 2007, 104, 11020–11025. [Google Scholar] [CrossRef]
- Mutharia, L.M.; Nicas, T.I.; Hancock, R.E.W. Outer Membrane Proteins of Pseudomonas aeruginosa Serotype Strains. J. Infect. Dis. 1982, 146, 770–779. [Google Scholar] [CrossRef] [PubMed]
- Cassin, E.K.; Tseng, B.S. Pushing beyond the Envelope: The Potential Roles of OprF in Pseudomonas aeruginosa Biofilm Formation and Pathogenicity. J. Bacteriol. 2019, 201. [Google Scholar] [CrossRef] [PubMed]
- Loots, K.; Revets, H.; Goddeeris, B.M. Attachment of the outer membrane lipoprotein (OprI) of Pseudomonas aeruginosa to the mucosal surfaces of the respiratory and digestive tract of chickens. Vaccine 2008, 26, 546–551. [Google Scholar] [CrossRef]
- Westritschnig, K.; Hochreiter, R.; Wallner, G.; Firbas, C.; Schwameis, M.; Jilma, B. A randomized, placebo-controlled phase i study assessing the safety and immunogenicity of a Pseudomonas aeruginosa hybrid outer membrane protein OprF/I vaccine (IC43) in healthy volunteers. Hum. Vaccines Immunother. 2014, 10, 170–183. [Google Scholar] [CrossRef]
- Adlbrecht, C.; Wurm, R.; Depuydt, P.; Spapen, H.; Lorente, J.A.; Staudinger, T.; Creteur, J.; Zauner, C.; Meier-Hellmann, A.; Eller, P.; et al. Efficacy, immunogenicity, and safety of IC43 recombinant Pseudomonas aeruginosa vaccine in mechanically ventilated intensive care patients—A randomized clinical trial. Crit. Care 2020, 24, 74. [Google Scholar] [CrossRef] [PubMed]
- Johansen, H.K.; Gøtzsche, P.C. Vaccines for preventing infection with Pseudomonas aeruginosa in cystic fibrosis. Cochrane Database Syst. Rev. 2015, 2015, CD001399. [Google Scholar] [CrossRef]
- Naim, H.Y. Applications and challenges of multivalent recombinantvaccines. In Human Vaccines and Immunotherapeutics. Landes Biosci. 2013, 9, 457–461. [Google Scholar] [CrossRef]
- Merakou, C.; Schaefers, M.; Priebe, G.P. Progress toward the elusive Pseudomonas aeruginosa vaccine. Surg. Infect. 2018, 19, 757–768. [Google Scholar] [CrossRef]
- Kamei, A.; Coutinho-Sledge, Y.S.; Goldberg, J.B.; Priebe, G.P.; Pier, G.B. Mucosal vaccination with a multivalent, live-attenuated vaccine induces multifactorial immunity against Pseudomonas aeruginosa acute lung infection. Infect. Immun. 2011, 79, 1289–1299. [Google Scholar] [CrossRef]
- Cripps, A.W.; Peek, K.; Dunkley, M.; Vento, K.; Marjason, J.K.; McIntyre, M.E.; Sizer, P.; Croft, D.; Sedlak-Weinstein, L. Safety and immunogenicity of an oral inactivated whole-cell Pseudomonas aeruginosa vaccine administered to healthy human subjects. Infect. Immun. 2006, 74, 968–974. [Google Scholar] [CrossRef]
- Baker, S.M.; McLachlan, J.B.; Morici, L.A. Immunological considerations in the development of Pseudomonas aeruginosa vaccines. Hum. Vaccines Immunother. 2019, 16, 412–418. [Google Scholar] [CrossRef]
- Pennington, J.E.; Reynolds, H.Y.; Wood, R.E.; Robinson, R.A.; Levine, A.S. Use of a Pseudomonas aeruginosa vaccine in patients with acute leukemia and cystic fibrosis. Am. J. Med. 1975, 58, 629–636. [Google Scholar] [CrossRef]
- Langford, D.T.; Hiller, J. Prospective, controlled study of a polyvalent pseudomonas vaccine in cystic fibrosis—Three year results. Arch. Dis. Child. 1984, 59, 1131–1134. [Google Scholar] [CrossRef]
- Zuercher, A.W.; Horn, M.P.; Que, J.U.; Ruedeberg, A.; Schoeni, M.H.; Schaad, U.B.; Marcus, P.; Lang, A.B. Antibody responses induced by long-term vaccination with an octovalent conjugate Pseudomonas aeruginosa vaccine in children with cystic fibrosis. FEMS Immunol. Med. Microbiol. 2006, 47, 302–308. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.K.; Kim, J.J.; Kim, J.H.; Woo, Y.M.; Kim, S.; Yoon, D.W.; Choi, C.S.; Kim, I.S.; Park, W.J.; Lee, N.G.; et al. Comparison of two immunization schedules for a Pseudomonas aeruginosa outer membrane proteins vaccine in burn patients. Vaccine 2000, 19, 1274–1283. [Google Scholar] [CrossRef]
- Rello, J.; Krenn, C.-G.; Locker, G.; Pilger, E.; Madl, C.; Balica, L.; Dugernier, T.; Laterre, P.-F.; Spapen, H.; Depuydt, P.; et al. A randomized placebo-controlled phase II study of a Pseudomonas vaccine in ventilated ICU patients. Crit. Care 2017, 21, 1–13. [Google Scholar] [CrossRef]
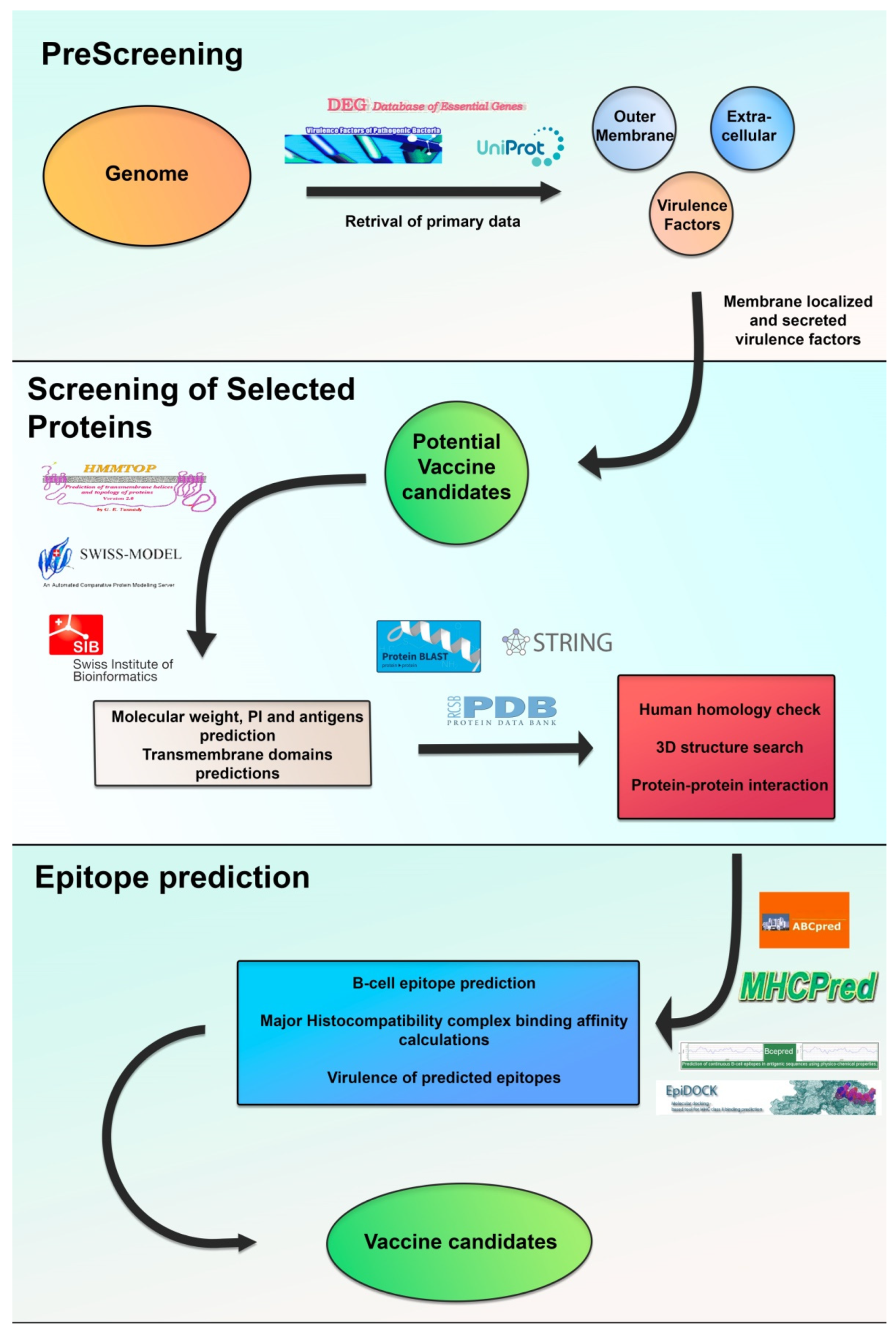
- Sousa, S.A.; Seixas, A.M.M.; Leitão, J.H. Postgenomic approaches and bioinformatics tools to advance the development of vaccines against bacteria of the Burkholderia cepacia complex. Vaccines 2018, 6, 34. [Google Scholar] [CrossRef] [PubMed]
- Pradenas, G.A.; Ross, B.N.; Torres, A.G. Burkholderia cepacia Complex Vaccines: Where Do We Go from here? Vaccines 2016, 4, 10. [Google Scholar] [CrossRef] [PubMed]
- Choh, L.-C.C.; Ong, G.-H.O.; Vellasamy, K.M.K.; Kalaiselvam, K.K.; Kang, W.-T.K.; Al-Maleki, A.R.A.; Mariappan, V.V.; Vadivelu, J.J. Burkholderia vaccines: Are we moving forward? Front. Cell. Infect. Microbiol. 2013, 3, 5. [Google Scholar] [CrossRef]
- Wang, G.; Zarodkiewicz, P.; Valvano, M.A. Current advances in Burkholderia vaccines development. Cells 2020, 9, 2671. [Google Scholar] [CrossRef]
- Pradenas, G.A.; Myers, J.N.; Torres, A.G. Characterization of the Burkholderia cenocepacia TonB Mutant as a potential live attenuated vaccine. Vaccines 2017, 5, 33. [Google Scholar] [CrossRef] [PubMed]
- Makidon, P.E.; Knowlton, J.; Groom, J.V.; Blanco, L.P.; Lipuma, J.J.; Bielinska, A.U.; Baker, J.R. Induction of immune response to the 17 kDa OMPA Burkholderia cenocepacia polypeptide and protection against pulmonary infection in mice after nasal vaccination with an OMP nanoemulsion-based vaccine. Med Microbiol. Immunol. 2009, 199, 81–92. [Google Scholar] [CrossRef]
- Sousa, S.A.; Morad, M.; Feliciano, J.R.; Pita, T.; Nady, S.; El-Hennamy, R.E.; Abdel-Rahman, M.; Cavaco, J.; Pereira, L.; Barreto, C.; et al. The Burkholderia cenocepacia OmpA-like protein BCAL2958: Identification, characterization, and detection of anti-BCAL2958 antibodies in serum from B. cepacia complex-infected Cystic Fibrosis patients. AMB Express 2016, 6, 41. [Google Scholar] [CrossRef]
- McClean, S.; Healy, M.E.; Collins, C.; Carberry, S.; O’Shaughnessy, L.; Dennehy, R.; Adams, Á.; Kennelly, H.; Corbett, J.M.; Carty, F.; et al. Linocin and OmpW are involved in attachment of the Cystic Fibrosis-associated pathogen Burkholderia cepacia complex to lung epithelial cells and protect mice against infection. Infect. Immun. 2016, 84, 1424–1437. [Google Scholar] [CrossRef] [PubMed]
- Bertot, G.M.; Restelli, M.A.; Galanternik, L.; Urey, R.C.A.; Valvano, M.A.; Grinstein, S. Nasal immunization with Burkholderia multivorans outer membrane proteins and the mucosal adjuvant adamantylamide dipeptide confers efficient protection against experimental lung infections with B. multivorans and B. cenocepacia. Infect. Immun. 2007, 75, 2740–2752. [Google Scholar] [CrossRef]
- Döring, G.; Pier, G. Vaccines and immunotherapy against Pseudomonas aeruginosa. Vaccine 2008, 26, 1011–1024. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Quintanilla, M.; Pulido, M.R.; Carretero-Ledesma, M.; McConnell, M.J. Vaccines for antibiotic-resistant bacteria: Possibility or pipe dream? Trends Pharmacol. Sci. 2016, 37, 143–152. [Google Scholar] [CrossRef]
- Lazar, H.; Horn, M.P.; Zuercher, A.W.; Imboden, M.A.; Durrer, P.; Seiberling, M.; Pokorny, R.; Hammer, C.; Lang, A.B. Pharmacokinetics and safety profile of the human anti—Pseudomonas aeruginosa serotype O11 immunoglobulin M monoclonal antibody KBPA-101 in healthy volunteers. Antimicrob. Agents Chemother. 2009, 53, 3442–3446. [Google Scholar] [CrossRef]
- Duvall, M.; Bradley, N.; Fiorini, R.N. A novel platform to produce human monoclonal antibodies: The next generation of therapeutic human monoclonal antibodies discovery. MAbs 2011, 3, 203–208. [Google Scholar] [CrossRef][Green Version]
- Jain, R.; Beckett, V.; Konstan, M.; Accurso, F.; Burns, J.; Mayer-Hamblett, N.; Milla, C.; VanDevanter, D.; Chmiel, J. KB001-A, a novel anti-inflammatory, found to be safe and well-tolerated in cystic fibrosis patients infected with Pseudomonas aeruginosa. J. Cyst. Fibros. 2018, 17, 484–491. [Google Scholar] [CrossRef]
- François, B.; Luyt, C.E.; Dugard, A.; Wolff, M.; Diehl, J.L.; Jaber, S.; Forel, J.M.; Garot, D.; Kipnis, E.; Mebazaa, A.; et al. Safety and pharmacokinetics of an anti-PcrV PEGylated monoclonal antibody fragment in mechanically ventilated patients colonized with Pseudomonas aeruginosa: A randomized, double-blind, placebo-controlled trial. Crit. Care Med. 2012, 40, 2320–2326. [Google Scholar] [CrossRef] [PubMed]
- Jain, M.; Ramirez, D.; Seshadri, R.; Cullina, J.F.; Powers, C.A.; Schulert, G.S.; Bar-Meir, M.; Sullivan, C.L.; McColley, S.A.; Hauser, A.R. Type III Secretion Phenotypes of Pseudomonas aeruginosa Strains Change during Infection of Individuals with Cystic Fibrosis. J. Clin. Microbiol. 2004, 42, 5229–5237. [Google Scholar] [CrossRef]
- Warrener, P.; Varkey, R.; Bonnell, J.C.; DiGiandomenico, A.; Camara, M.; Cook, K.; Peng, L.; Zha, J.; Chowdury, P.; Sellman, B.; et al. A novel anti-PcrV antibody providing enhanced protection against pseudomonas aeruginosa in multiple animal infection models. Antimicrob. Agents Chemother. 2014, 58, 4384–4391. [Google Scholar] [CrossRef] [PubMed]
- Coutinho, A.; Gronowicz, E.; Bullock, W.W.; Möller, G. Mechanism of thymus-independent immunocyte triggering: Mitogenic activation of B cells results in specific immune responses. J. Exp. Med. 1974, 139, 74–92. [Google Scholar] [CrossRef] [PubMed]
- Keyt, B.A.; Baliga, R.; Sinclair, A.M.; Carroll, S.F.; Peterson, M.S. Structure, function, and therapeutic use of IgM antibodies. Antibodies 2020, 9, 53. [Google Scholar] [CrossRef]
- Que, Y.-A.; Lazar, H.; Wolff, M.; François, B.; Laterre, P.-F.; Mercier, E.; Garbino, J.; Pagani, J.-L.; Revelly, J.-P.; Mus, E.; et al. Assessment of panobacumab as adjunctive immunotherapy for the treatment of nosocomial Pseudomonas aeruginosa pneumonia. Eur. J. Clin. Microbiol. Infect. Dis. 2014, 33, 1861–1867. [Google Scholar] [CrossRef]
- DiGiandomenico, A.; Keller, A.E.; Gao, C.; Rainey, G.J.; Warrener, P.; Camara, M.M.; Bonnell, J.; Fleming, R.; Bezabeh, B.; DiMasi, N.; et al. A multifunctional bispecific antibody protects against Pseudomonas aeruginosa. Sci. Transl. Med. 2014, 6, 262ra155. [Google Scholar] [CrossRef]
- Chastre, J.; François, B.; Bourgeois, M.; Komnos, A.; Ferrer, R.; Rahav, G.; De Schryver, N.; Lepape, A.; Koksal, I.; Luyt, C.-E.; et al. Efficacy, pharmacokinetics (PK), and safety profile of MEDI3902, an anti—Pseudomonas aeruginosa bispecific human monoclonal antibody in mechanically ventilated intensive care unit patients; results of the phase 2 EVADE study conducted by the public-private COMBACTE-MAGNET consortium in the Innovative Medicines Initiative (IMI) program. Open Forum Infect. Dis. 2020, 7, S377–S378. [Google Scholar] [CrossRef]
- Nilsson, E.; Larsson, A.; Olesen, H.V.; Wejåker, P.E.; Kollberg, H. Good effect of IgY against Pseudomonas aeruginosa infections in cystic fibrosis patients. Pediatr. Pulmonol. 2008, 43, 892–899. [Google Scholar] [CrossRef]
- Leiva, C.L.; Gallardo, M.J.; Casanova, N.; Terzolo, H.; Chacana, P. IgY-technology (egg yolk antibodies) in human medicine: A review of patents and clinical trials. Int. Immunopharmacol. 2020, 81, 106269. [Google Scholar] [CrossRef]
- Carlander, D.; Kollberg, H.; Wejåker, P.-E.; Larsson, A. Peroral immunotheraphy with yolk antibodies for the prevention and treatment of enteric infections. Immunol. Res. 2000, 21, 1–6. [Google Scholar] [CrossRef]
- Thomsen, K.; Christophersen, L.; Bjarnsholt, T.; Jensen, P.Ø.; Moser, C.; Høiby, N. Anti—Pseudomonas aeruginosa IgY antibodies augment bacterial clearance in a murine pneumonia model. J. Cyst. Fibros. 2016, 15, 171–178. [Google Scholar] [CrossRef] [PubMed]
- EudraCT Number 2011-000801-39—Clinical Trial Results—EU Clinical Trials Register. Available online: https://www.clinicaltrialsregister.eu/ctr-search/trial/2011-000801-39/results (accessed on 13 May 2021).
- Milla, C.E.; Chmiel, J.F.; Accurso, F.J.; Vandevanter, D.R.; Konstan, M.W.; Yarranton, G.; Geller, D.E. Anti-PcrV antibody in cystic fibrosis: A novel approach targeting Pseudomonas aeruginosa airway infection. Pediatr. Pulmonol. 2014, 49, 650–658. [Google Scholar] [CrossRef]
- Ali, S.; Yu, X.Q.; Robbie, G.J.; Wu, Y.; Shoemaker, K.; Yu, L.; DiGiandomenico, A.; Keller, A.E.; Anude, C.; Hernandez-Illas, M.; et al. Phase 1 study of MEDI3902, an investigational anti–Pseudomonas aeruginosa PcrV and Psl bispecific human monoclonal antibody, in healthy adults. Clin. Microbiol. Infect. 2019, 25, 629.e1–629.e6. [Google Scholar] [CrossRef]
- Aridis Pharmaceuticals | AR-105 (Aerucin®). Available online: https://www.aridispharma.com/ar-105/ (accessed on 12 June 2021).
- Nzula, S.; Vandamme, P.; Govan, J.R.W. Infuence of taxonomic status on the in vitro antimicrobial susceptibility of the Burkholderia cepacia complex. J. Antimicrob. Chemother. 2002, 50, 265–269. [Google Scholar] [CrossRef]
- Peeters, E.; Nelis, H.J.; Coenye, T. In vitro activity of ceftazidime, ciprofloxacin, meropenem, minocycline, tobramycin and trimethoprim/sulfamethoxazole against planktonic and sessile Burkholderia cepacia complex bacteria. J. Antimicrob. Chemother. 2009, 64, 801–809. [Google Scholar] [CrossRef] [PubMed]
- Gibson, R.L.; Burns, J.L.; Ramsey, B.W. Pathophysiology and management of pulmonary infections in Cystic Fibrosis. Am. J. Respir. Crit. Care Med. 2003, 168, 918–951. [Google Scholar] [CrossRef]
- De Groot, R.; Smith, A.L. Antibiotic pharmacokinetics in Cystic Fibrosis: Differences and clinical significance. Clin. Pharmacokinet. 1987, 13, 228–253. [Google Scholar] [CrossRef]
- Prandota, J. Drug disposition in Cystic Fibrosis: Progress in understanding pathophysiology and pharmacokinetics. Pediatr. Infect. Dis. J. 1987, 6, 1111–1126. [Google Scholar] [CrossRef]
- Patel, N.; Conejero, L.; De Reynal, M.; Easton, A.; Bancroft, G.J.; Titball, R.W. Development of vaccines against Burkholderia pseudomallei. Front. Microbiol. 2011, 2, 198. [Google Scholar] [CrossRef]
- Skurnik, D.; Davis, M.R.; Benedetti, D.; Moravec, K.L.; Cywes-Bentley, C.; Roux, D.; Traficante, D.C.; Walsh, R.L.; Maira-Litrn, T.; Cassidy, S.K.; et al. Targeting pan-resistant bacteria with antibodies to a broadly conserved surface polysaccharide expressed during infection. J. Infect. Dis. 2012, 205, 1709–1718. [Google Scholar] [CrossRef] [PubMed]
- Pimenta, A.I.; Kilcoyne, M.; Bernardes, N.; Mil-Homens, D.; Joshi, L.; Fialho, A.M. Burkholderia cenocepacia BCAM2418 -induced antibody inhibits bacterial adhesion, confers protection to infection and enables identification of host glycans as adhesin targets. Cell. Microbiol. 2021, 1–16. [Google Scholar] [CrossRef]
- Santos, N.C.; Figueira-Coelho, J.; Martins-Silva, J.; Saldanha, C. Multidisciplinary utilization of dimethyl sulfoxide: Pharmacological, cellular, and molecular aspects. Biochem. Pharmacol. 2003, 65, 1035–1041. [Google Scholar] [CrossRef]
- Wu, W.; Huang, J.; Duan, B.; Traficante, D.C.; Hong, H.; Risech, M.; Lory, S.; Priebe, G.P. Th17-stimulating Protein Vaccines Confer Protection against Pseudomonas aeruginosa Pneumonia. Am. J. Respir. Crit. Care Med. 2012, 186, 420–427. [Google Scholar] [CrossRef] [PubMed]
- Motta, S.; Vecchietti, D.; Martorana, A.; Brunetti, P.; Bertoni, G.; Polissi, A.; Mauri, P.; Di Silvestre, D. The Landscape of Pseudomonas aeruginosa Membrane-Associated Proteins. Cells 2020, 9, 2421. [Google Scholar] [CrossRef] [PubMed]
- Gong, Q.; Ruan, M.; Niu, M.; Qin, C.; Hou, Y.; Guo, J. Immune efficacy of DNA vaccines based on oprL and oprF genes of Pseudomonas aeruginosa in chickens. Poult. Sci. 2018, 97, 4219–4227. [Google Scholar] [CrossRef]
- Yang, F.; Gu, J.; Zou, J.; Lei, L.; Jing, H.; Zhang, J.; Zeng, H.; Zou, Q.; Lv, F.; Zhang, J. PA0833 Is an OmpA C-Like Protein That Confers Protection Against Pseudomonas aeruginosa Infection. Front. Microbiol. 2018, 9, 1062. [Google Scholar] [CrossRef] [PubMed]
- Shinoy, M.; Dennehy, R.; Coleman, L.; Carberry, S.; Schaffer, K.; Callaghan, M.; Doyle, S.; McClean, S. Immunoproteomic analysis of proteins expressed by two related pathogens, Burkholderia multivorans and Burkholderia cenocepacia, during human infection. PLoS ONE 2013, 8, e80796. [Google Scholar] [CrossRef]
- Sanchez-Campillo, M.; Bini, L.; Comanducci, M.; Raggiaschi, R.; Marzocchi, B.; Pallini, V.; Ratti, G. Identification of immunoreactive proteins of Chlamydia trachomatis by Western blot analysis of a two-dimensional electrophoresis map with patient sera. Electrophoresis 1999, 20, 2269–2279. [Google Scholar] [CrossRef]
- Vidakovics, M.L.P.; Paba, J.; Lamberti, Y.; Ricart, C.A.; De Sousa, M.V.; Rodriguez, M.E. Profiling the Bordetella pertussis proteome during iron starvation. J. Proteome Res. 2007, 6, 2518–2528. [Google Scholar] [CrossRef]
- Olekhnovich, I.N.; Kadner, R.J. Contribution of the RpoA C-terminal domain to stimulation of the Salmonella enterica hilA promoter by HilC and HilD. J. Bacteriol. 2004, 186, 3249–3253. [Google Scholar] [CrossRef] [PubMed]
- Mariappan, V.; Vellasamy, K.M.; Thimma, J.S.; Hashim, O.H.; Vadivelu, J. Identification of immunogenic proteins from Burkholderia cepacia secretome using proteomic analysis. Vaccine 2010, 28, 1318–1324. [Google Scholar] [CrossRef]
- Nieves, W.; Heang, J.; Asakrah, S.; Zu Bentrup, K.H.; Roy, C.J.; Morici, L.A. Immunospecific responses to bacterial elongation factor Tu during Burkholderia infection and immunization. PLoS ONE 2010, 5, e14361. [Google Scholar] [CrossRef]
- Harding, S.V.; Sarkar-Tyson, M.; Smither, S.J.; Atkins, T.P.; Oyston, P.C.; Brown, K.A.; Liu, Y.; Wait, R.; Titball, R.W. The identification of surface proteins of Burkholderia pseudomallei. Vaccine 2007, 25, 2664–2672. [Google Scholar] [CrossRef] [PubMed]
- Upritchard, H.G.; Cordwell, S.J.; Lamont, I.L. Immunoproteomics to examine Cystic Fibrosis host interactions with extracellular Pseudomonas aeruginosa proteins. Infect. Immun. 2008, 76, 4624–4632. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Garduño, R.A.; Garduño, E.; Hoffman, P.S. Surface-associated Hsp60 Chaperonin of Legionella pneumophila mediates invasion in a HeLa cell model. Infect. Immun. 1998, 66, 4602–4610. [Google Scholar] [CrossRef]
- Khan, M.N.; Shukla, D.; Bansal, A.; Mustoori, S.; Ilavazhagan, G. Immunogenicity and protective efficacy of GroEL (hsp60) of Streptococcus pneumoniae against lethal infection in mice. FEMS Immunol. Med. Microbiol. 2009, 56, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Sinha, K.; Bhatnagar, R. GroEL provides protection against Bacillus anthracis infection in BALB/c mice. Mol. Immunol. 2010, 48, 264–271. [Google Scholar] [CrossRef]
- Sousa, S.A.; Soares-Castro, P.; Seixas, A.; Feliciano, J.R.; Balugas, B.; Barreto, C.; Pereira, M.L.; Santos, P.M.; Leitão, J.H. New insights into the immunoproteome of B. cenocepacia J2315 using serum samples from cystic fibrosis patients. New Biotechnol. 2020, 54, 62–70. [Google Scholar] [CrossRef]
- Bazzini, S.; Udine, C.; Sass, A.; Pasca, M.R.; Longo, F.; Emiliani, G.; Fondi, M.; Perrin, E.; Decorosi, F.; Viti, C.; et al. Deciphering the role of RND Efflux transporters in Burkholderia cenocepacia. PLoS ONE 2011, 6, e18902. [Google Scholar] [CrossRef] [PubMed]
- Chan, Y.Y.; Chua, K.L. The Burkholderia pseudomallei BpeAB-OprB Efflux pump: Expression and impact on quorum sensing and virulence. J. Bacteriol. 2005, 187, 4707–4719. [Google Scholar] [CrossRef] [PubMed]
- Darwin, A.J. Stress relief during host infection: The phage shock protein response supports bacterial virulence in various ways. PLoS Pathog. 2013, 9, e1003388. [Google Scholar] [CrossRef]
- Southern, S.J.; Tavassoli, A.; Sarkar-Tyson, M.; Milne, T.; Male, A.; Oyston, P.C.F. Evaluating the role of phage-shock protein A in Burkholderia pseudomallei. Microbiology 2015, 161, 2192–2203. [Google Scholar] [CrossRef]
- Sonnenberg, M.G.; Belisle, J.T. Definition of Mycobacterium tuberculosis culture filtrate proteins by two-dimensional polyacrylamide gel electrophoresis, N-terminal amino acid sequencing, and electrospray mass spectrometry. Infect. Immun. 1997, 65, 4515–4524. [Google Scholar] [CrossRef]
- Coombes, B.K.; Valdez, Y.; Finlay, B. Evasive maneuvers by secreted bacterial proteins to avoid innate immune responses. Curr. Biol. 2004, 14, R856–R867. [Google Scholar] [CrossRef]
- Sousa, S.A.; Seixas, A.M.; Mandal, M.; Rodríguez-Ortega, M.J.; Leitão, J.H. Characterization of the Burkholderia cenocepacia J2315 surface-exposed immunoproteome. Vaccines 2020, 8, 509. [Google Scholar] [CrossRef]
- Sriramulu, D.D.; Lünsdorf, H.; Lam, J.S.; Römling, U. Microcolony formation: A novel biofilm model of Pseudomonas aeruginosa for the cystic fibrosis lung. J. Med Microbiol. 2005, 54, 667–676. [Google Scholar] [CrossRef] [PubMed]
- Worlitzsch, D.; Tarran, R.; Ulrich, M.; Schwab, U.; Cekici, A.; Meyer, K.C.; Birrer, P.; Bellon, G.; Berger, J.; Weiss, T.; et al. Effects of reduced mucus oxygen concentration in airway Pseudomonas infections of cystic fibrosis patients. J. Clin. Invest. 2002, 109, 317–325. [Google Scholar] [CrossRef] [PubMed]
| Immunogen | Clinical Trial | Immunization Protocol | Results | References | |
|---|---|---|---|---|---|
| Pros | Cons | ||||
| Pseudostat (Paraformaldehyde-killed P. aeruginosa) | (Phase 1): Safety and immunogenicity test of a vaccine administered to healthy human subjects | Healthy volunteers (18–50 years of age) Oral dose (150 mg), 2 doses (day 0 and 28). Serological follow-up to 56-days post-vaccination. | Immunogenic in humans. Pooled sera collected after immunization had higher capacity to promote opsonophagocytotic killing (OPK) of P. aeruginosa. | Some neurological, gastrointestinal, and respiratory disorders were detected | [43] |
| Pseudogen (Heptavalent O-antigen) | (Phase 2): Efficacy evaluation of the vaccine in patients with acute leukemia and CF. | Intramuscular (IM) administration of 6–12 µg/kg. | Efficacy in preventing fatal P. aeruginosa infections in cancer and burn patients. | No benefit for leukemia and CF patients. Limited use due to their toxicity in 92% of patients tested. | [45] |
| PEV-01 (Polyvalent LPS extracts) | (Phase 2): Prospective, controlled study of a polyvalent vaccine in CF. | Three doses, SC, 1 month apart and the dose 4 after 1 year. 0.25 mL for CF patients under 12 years of age and 0.5 mL for patients over 12. | No benefit for CF patients. | [46] | |
| Aerugen (Octavalent OPS-Toxin A conjugate) | (Phase 3): Analysis of the serological response after 10 years of repeated immunization of children with CF and efficacy on prevention of P. aeruginosa colonization. | Initial inoculations were given at 0, 2 and 12 months, and annual booster doses after the third year. | Increase of IgG levels to all vaccine components. Has a good safety profile for long-term use. The incidence of P. aeruginosa infection was lower compared with the non-vaccinated group. | Later unpublished results from a prospective trial of this vaccine in Europe did not show a delay in colonization and this vaccine was abandoned. | [47] |
| Bivalent FliC | (Phase 3): Immunization of CF patients not colonized with P. aeruginosa to evaluate its safety and efficacy. | Double-blind, placebo-controlled, multicenter trial. IM; 40 µg in CF patients (2–18 years of age), 4 doses, administrated each dose with 4 weeks apart and a booster dose after 1 year. Addition of adjuvant Al(OH)3, thiomersal. | Vaccine well tolerated. Active immunization of CF patients delayed the onset of chronic infection with P. aeruginosa, resulting in longer survival of these patients. High serum IgG titers to flagella vaccine subtypes. | [33] | |
| CFC-101 (OMP extracted from 4 P. aeruginosa strains) | (Phase 2): Analysis of 2 immunization schedules of the OMP vaccine in burn patients. | Double-blind, randomized and placebo-controled trial. Adult patients with burn injuries in greater than 10% total body surface. IM; 3 doses (0.5 or 1.0 mg) with 3- or 7-day intervals. | The vaccine was safe and highly immunogenic in burn patients, especially with 1 mg doses at 3-day intervals. | [48] | |
| IC43 (OprF/I) | NCT00778388 (Phase 1): Against P. aeruginosa in healthy volunteers | Placebo controlled, double-blind, multi-center, randomized trial. Four different doses (50–200 mcg) administered intramuscularly (IM) to healthy adults (18–65 years of age), with 2 doses given 7 days apart | No serious adverse effect. IC43 doses ≥50 mcg were sufficient to induce plateau of IgG antibody responses in healthy volunteers. At day 90, titers declined but remained higher than the placebo group for up to 6 months. | Higher doses, whether adjuvanted or not, were not more effective. | [37,38,49] |
| NCT00876252 (Phase 2): Immunogenicity of IC43 in ICU admitted patients requiring MV | Patients were randomized to receive 3 different vaccine doses (100 mcg or 200 mcg IC43 with adjuvant, or 100 mcg without adjuvant) or placebo IM at days 0 and 7. Evaluation for 90 days. | At day 14 all IC43 administered groups had higher anti-OprF/I titers. Lower mortality in patients immunized with IC43 compared with placebo. | No statistical difference in P. aeruginosa infection rates between patients vaccinated with IC43 and placebo. However, most P. aeruginosa infections occurred before 14 days. | ||
| NCT01563263 (Phase 2/3): Confirmatory study assessing efficacy, immunogenicity and safety of IC43 vaccine in intensive care unit (ICU) patients | Placebo controlled, double-blind, multi-center, randomized trial. ICU patients requiring MV for more than 48 h, age 18–80 years. Patients were randomized to receive an IM injection of 100 mcg of IC43 or placebo on days 0 and 7. | Vaccine was well tolerated in the large population of medically ill, MV patients. The vaccine achieved high immunogenicity. | However, no clinical benefit over placebo was provided in terms of overall mortality. | ||
| Immunogen | Clinical Trial | Immunization Protocol | Results | References | |
|---|---|---|---|---|---|
| Pros | Cons | ||||
| KB001 (anti-PcrV PEGylated mouse Mab) | Phase 1/2 (NCT00691587): Safety and pharmacokinetics (PK) of KB001 in mechanically ventilated (MV) ICU patients colonized with P. aeruginosa. | Patients (older than 18 years) will receive randomly either placebo, or single low-dose or single high-dose of KB001 intravenously (IV). | Safe and well-tolerated. | No anti-KB001 antibodies were detected. | [64,77] |
| Decrease in the incidence of P. aeruginosa associated pneumonia in patients on MV. | |||||
| Phase 1/2 (NCT00638365): Dose escalation study of KB001 in CF patients colonized with P. aeruginosa. | Patients (older than 12 years) will randomly receive either placebo, single-dose 3 mg/kg or single-dose 10 mg/kg of KB001 IV. | Safe and well-tolerated. | No significant differences between KB001 and the placebo | ||
| Reduced lung inflammation of KB001 vaccinated patients. | group in P. aeruginosa colonization of CF patients. | ||||
| KB001-A | Phase 2 (NCT01695343): Evaluation of the effect of KB001-A on time-to-need for antibiotic treatment of CF patients. | Patients (12–50 years of age) will randomly receive either placebo, KB001-A up to 5× IV at 10 mg/kg to a maximum dose of 800 mg per dose. | Safe and well-tolerated. | Reduced clinical efficacy, being not associated with an increased time to need for antibiotics. | [63] |
| (anti-PcrV PEGylated mouse MAb) | Modest FEV1 benefit and reduction in selected sputum inflammatory markers (IL-8). | ||||
| (One amino acid difference from KB001) | |||||
| V2L2MD | Preclinical | Good prophylactic protection in several mouse models of P. aeruginosa infection. | [66] | ||
| (anti-PcrV Human MAb) | |||||
| MEDI3902 (anti-PcrV and Psl bispecific human MAb) | Phase 1 (NCT02255760): Safety evaluation, PK, anti-drug antibody (ADA) responses, ex vivo anticytotoxicity and OPK of MEDI3902 in healthy adults | Single IV infusion in healthy adults aged 18–60 years. | The safety and tolerability profile of MEDI3902 was acceptable. | Infusion-related reaction (e.g., skin rash). | [71,78] |
| Dose-escalation study: subjects were randomized in a 3:1 ratio to receive 250, 750, 1500 or 3000 mg of MEDI3902 or placebo. | Anti–P. aeruginosa activity was demonstrated in sera of treated subjects. | ||||
| Subjects followed for 60 days afterwards. | |||||
| Phase 2 (NCT02696902): Evaluation of MEDI3902 efficacy and safety on the prevention of P. aeruginosa nosocomial pneumonia in MV patients | Participants will receive a single IV dose of placebo, MEDI3902 500 mg or MEDI3902 1500 mg. | Some clinical efficacy in ICU patients with lower baseline inflammation. | |||
| Panobacumab or KBPA-101 or AR-101 (IgM/κ isotype directed against the LPS O-polysaccharide moiety of P. aeruginosa serotype O11) | Phase 2: PK and safety profile of KBPA-101 in healthy volunteers | No adverse effects in healthy volunteers. | [61,69] | ||
| NCT00851435 (phase 2): Safety and PK in patients with hospital acquired pneumonia (HAP) caused by serotype O11 P. aeruginosa | HAP patients (older than 18 years of age) were treated by IV infusion of 1.2 mg/kg KBPA-101, 3 separate doses, every third day. | Improve clinical outcome in a shorter time. | |||
| Passive immunotherapy targeting LPS can be a complementary strategy for the treatment of nosocomial P. aeruginosa pneumonia. | |||||
| Aerucin or AR-105 (Human IgG1 MAb that targets P. aeruginosa alginate) | NCT02486770 (phase 1): Safety evaluation of Aerucin in healthy individuals. | IV administration up to 20 mg/kg monitored for 84 days in healthy individuals. | Safety up to doses of 20 mg/kg. | [79] | |
| NCT03027609 (phase 2): Efficacy, safety and PK evaluation of Aerucin in combination with standard antibiotic treatment in P. aeruginosa VAP patients. | Placebo controlled, double-blind, randomized trial. | No significant difference between Aerucin and placebo patient groups for treatment of P. aeruginosa VAP patients. | |||
| Single IV infusion of Aerucin 20 mg/kg. | |||||
| PseudIgY | NCT00633191 (phase 2): Study of anti-pseudomonas IgY in prevention of recurrence of P. aeruginosa infections in CF Patients. | Oral administration (gargle solution) of CF patients every night after toothbrushing. | After 12 years of prophylactic anti-Pseudomonas IgY treatment a reduction was observed in the level of infections with P. aeruginosa in the treated CF patients and no decrease in lung function. | [72] | |
| (anti-pseudomonas IgY gargle) | |||||
| PsAer-IgY (anti-pseudomonas IgY gargle) | NCT01455675 (phase 3): Evaluation of the clinical efficacy and safety of anti-Pseudomonas IgY in prevention of recurrence of P. aeruginosa infection in CF patients | Randomized, double-blind, | IgY antibodies were present in the oral cavity of treated patients for up to 24 h. | Clinical efficacy results were unclear. | [76] |
| placebo-controlled. | No adverse immune or allergic reaction. | ||||
| Oral administration of CF patients (older than 5 years of age), every day with 70 mL gargling solution (contains 50 mg IgY) or placebo. Treatment for 24 months. | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sousa, S.A.; Seixas, A.M.M.; Marques, J.M.M.; Leitão, J.H. Immunization and Immunotherapy Approaches against Pseudomonas aeruginosa and Burkholderia cepacia Complex Infections. Vaccines 2021, 9, 670. https://doi.org/10.3390/vaccines9060670
Sousa SA, Seixas AMM, Marques JMM, Leitão JH. Immunization and Immunotherapy Approaches against Pseudomonas aeruginosa and Burkholderia cepacia Complex Infections. Vaccines. 2021; 9(6):670. https://doi.org/10.3390/vaccines9060670
Chicago/Turabian StyleSousa, Sílvia A., António M. M. Seixas, Joana M. M. Marques, and Jorge H. Leitão. 2021. "Immunization and Immunotherapy Approaches against Pseudomonas aeruginosa and Burkholderia cepacia Complex Infections" Vaccines 9, no. 6: 670. https://doi.org/10.3390/vaccines9060670
APA StyleSousa, S. A., Seixas, A. M. M., Marques, J. M. M., & Leitão, J. H. (2021). Immunization and Immunotherapy Approaches against Pseudomonas aeruginosa and Burkholderia cepacia Complex Infections. Vaccines, 9(6), 670. https://doi.org/10.3390/vaccines9060670








