Monoclonal Antibodies Targeting Surface-Exposed and Secreted Proteins from Staphylococci
Abstract
1. Introduction
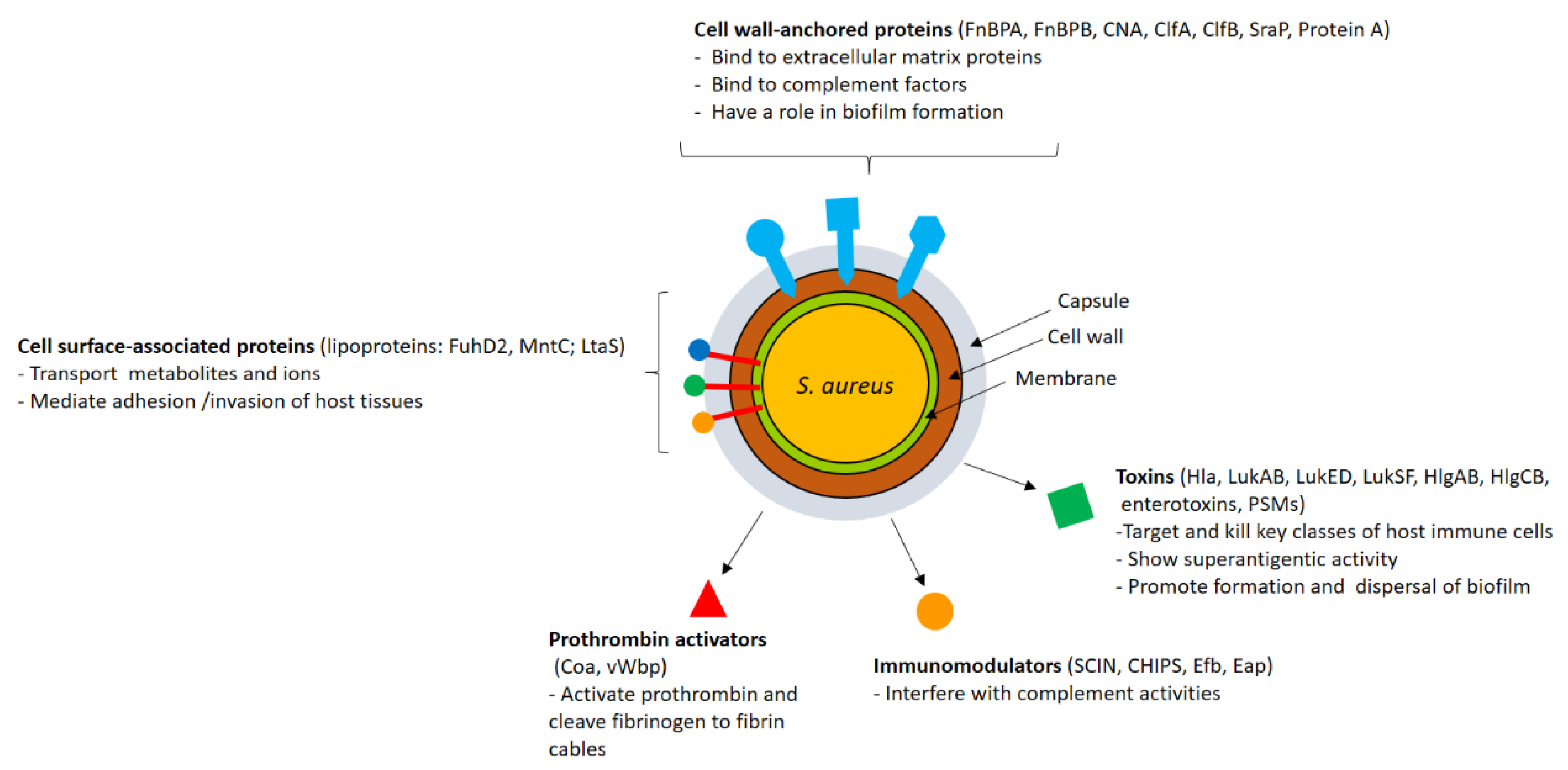
2. Staphylococcus Virulence Factors
2.1. CWA Proteins
2.2. Lipoproteins and Other Surface-Associated Proteins
2.3. Secreted Proteins
2.3.1. Factors Interfering with the Host Immune System
2.3.2. Toxins
2.4. Quorum Sensing Regulatory System
2.5. Biofilm Formation
3. Antibodies
3.1. General Properties of Antibodies
3.2. Monoclonal Antibodies and Their Derivatives
3.3. Active and Passive Immunization
4. mAbs Targeting Virulence Factors and Their Possible Therapeutic Use to Combat Staphylococcal Infections
4.1. mAbs against Surface and Surface-Associated Proteins
4.2. mAbs against Secreted Proteins/Peptides
4.3. mAbs That Block the Agr QS System
4.4. mAbs That Inhibit Biofilm Development
5. Antibody-Conjugated Derivatives and Their Use to Combat Staphylococcal Infections
6. Discussion and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Otto, M. Staphylococcus epidermidis—The ‘accidental’ pathogen. Nat. Rev. Microbiol. 2009, 7, 555–567. [Google Scholar] [CrossRef]
- Foster, T.J. Surface Proteins of Staphylococcus epidermidis. Front. Microbiol. 2020, 11, 1829. [Google Scholar] [CrossRef]
- Arciola, C.R.; Campoccia, D.; Montanaro, L. Implant infections: Adhesion, biofilm formation and immune evasion. Nat. Rev. Microbiol. 2018, 16, 397–409. [Google Scholar] [CrossRef]
- Lowy, F.D. Staphylococcus aureusInfections. N. Engl. J. Med. 1998, 339, 520–532. [Google Scholar] [CrossRef]
- Foster, T.J. The MSCRAMM Family of Cell-Wall-Anchored Surface Proteins of Gram-Positive Cocci. Trends Microbiol. 2019, 27, 927–941. [Google Scholar] [CrossRef] [PubMed]
- Foster, T.J. Surface Proteins of Staphylococcus aureus. Microbiol. Spectr. 2019, 7. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, M.-T.; Matsuo, M.; Niemann, S.; Herrmann, M.; Götz, F. Lipoproteins in Gram-Positive Bacteria: Abundance, Function, Fitness. Front. Microbiol. 2020, 11, 582582. [Google Scholar] [CrossRef]
- De Jong, N.W.M.; Van Kessel, K.P.M.; Van Strijp, J.A.G. Immune Evasion by Staphylococcus aureus. Microbiol. Spectr. 2019, 7. [Google Scholar] [CrossRef]
- Speziale, P.; Pietrocola, G.; Foster, T.J.; Geoghegan, J.A. Protein-based biofilm matrices in Staphylococci. Front. Cell. Infect. Microbiol. 2014, 4, 171. [Google Scholar] [CrossRef] [PubMed]
- Cheung, G.Y.C.; Bae, J.S.; Otto, M. Pathogenicity and virulence of Staphylococcus aureus. Virulence 2021, 12, 547–569. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.T.; Nguyen, T.H.; Otto, M. The staphylococcal exopolysaccharide PI–Biosynthesis and role in biofilm formation, colonization, and infection. Comput. Struct. Biotechnol. J. 2020, 18, 3324–3334. [Google Scholar] [CrossRef]
- Speziale, P.; Pietrocola, G. The Multivalent Role of Fibronectin-Binding Proteins A and B (FnBPA and FnBPB) of Staphylococcus aureus in Host Infections. Front. Microbiol. 2020, 11, 11. [Google Scholar] [CrossRef]
- Ganesh, V.K.; Rivera, J.J.; Smeds, E.; Ko, Y.-P.; Bowden, M.G.; Wann, E.R.; Gurusiddappa, S.; Fitzgerald, J.R.; Höök, M. A Structural Model of the Staphylococcus aureus ClfA–Fibrinogen Interaction Opens New Avenues for the Design of Anti-Staphylococcal Therapeutics. PLoS Pathog. 2008, 4, e1000226. [Google Scholar] [CrossRef] [PubMed]
- Pietrocola, G.; Nobile, G.; Gianotti, V.; Zapotoczna, M.; Foster, T.J.; Geoghegan, J.A.; Speziale, P. Molecular Interactions of Human Plasminogen with Fibronectin-binding Protein B (FnBPB), a Fibrinogen/Fibronectin-binding Protein from Staphylococcus aureus. J. Biol. Chem. 2016, 291, 18148–18162. [Google Scholar] [CrossRef]
- Pietrocola, G.; Nobile, G.; Alfeo, M.J.; Foster, T.J.; Geoghegan, J.A.; De Filippis, V.; Speziale, P. Fibronectin-binding protein B (FnBPB) from Staphylococcus aureus protects against the antimicrobial activity of histones. J. Biol. Chem. 2019, 294, 3588–3602. [Google Scholar] [CrossRef] [PubMed]
- Towell, A.M.; Feuillie, C.; Vitry, P.; Da Costa, T.M.; Mathelié-Guinlet, M.; Kezic, S.; Fleury, O.M.; McAleer, M.A.; Dufrêne, Y.F.; Irvine, A.D.; et al. Staphylococcus aureusbinds to the N-terminal region of corneodesmosin to adhere to the stratum corneum in atopic dermatitis. Proc. Natl. Acad. Sci. USA 2021, 118, e2014444118. [Google Scholar] [CrossRef]
- Speziale, P.; Arciola, C.R.; Pietrocola, G. Fibronectin and Its Role in Human Infective Diseases. Cells 2019, 8, 1516. [Google Scholar] [CrossRef] [PubMed]
- Walsh, E.J.; Miajlovic, H.; Gorkun, O.V.; Foster, T.J. Identification of the Staphylococcus aureus MSCRAMM clumping factor B (ClfB) binding site in the αC-domain of human fibrinogen. Microbiology 2008, 154, 550–558. [Google Scholar] [CrossRef] [PubMed]
- Perkins, S.; Walsh, E.J.; Deivanayagam, C.C.S.; Narayana, S.V.L.; Foster, T.J.; Höök, M. Structural Organization of the Fibrinogen-binding Region of the Clumping Factor B MSCRAMM of Staphylococcus aureus. J. Biol. Chem. 2001, 276, 44721–44728. [Google Scholar] [CrossRef] [PubMed]
- Ganesh, V.K.; Barbu, E.M.; Deivanayagam, C.C.S.; Le, B.; Anderson, A.S.; Matsuka, Y.V.; Lin, S.L.; Foster, T.J.; Narayana, S.V.L.; Höök, M. Structural and Biochemical Characterization of Staphylococcus aureus Clumping Factor B/Ligand Interactions. J. Biol. Chem. 2011, 286, 25963–25972. [Google Scholar] [CrossRef]
- Walsh, E.J.; O’Brien, L.M.; Liang, X.; Hook, M.; Foster, T.J. Clumping Factor B, a Fibrinogen-binding MSCRAMM (Microbial Surface Components Recognizing Adhesive Matrix Molecules) Adhesin of Staphylococcus aureus, Also Binds to the Tail Region of Type I Cytokeratin 10. J. Biol. Chem. 2004, 279, 50691–50699. [Google Scholar] [CrossRef]
- Mulcahy, M.E.; Geoghegan, J.A.; Monk, I.R.; O’Keeffe, K.M.; Walsh, E.J.; Foster, T.J.; McLoughlin, R.M. Nasal Colonisation by Staphylococcus aureus Depends upon Clumping Factor B Binding to the Squamous Epithelial Cell Envelope Protein Loricrin. PLoS Pathog. 2012, 8, e1003092. [Google Scholar] [CrossRef] [PubMed]
- Speziale, P.; Höök, M.; Switalski, L.M.; Wadström, T. Fibronectin binding to a Streptococcus pyogenes strain. J. Bacteriol. 1984, 157, 420–427. [Google Scholar] [CrossRef]
- Switalski, L.M.; Patti, J.M.; Butcher, W.; Gristina, A.G.; Speziale, P.; Höök, M. A collagen receptor on Staphylococcus aureus strains isolated from patients with septic arthritis mediates adhesion to cartilage. Mol. Microbiol. 1993, 7, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Valotteau, C.; Prystopiuk, V.; Pietrocola, G.; Rindi, S.; Peterle, D.; De Filippis, V.; Foster, T.J.; Speziale, P.; Dufrene, Y.F. Single-Cell and Single-Molecule Analysis Unravels the Multifunctionality of theStaphylococcus aureusCollagen-Binding Protein Cna. ACS Nano 2017, 11, 2160–2170. [Google Scholar] [CrossRef]
- Thomas, M.G.; Peacock, S.; Daenke, S.; Berendt, A.R.; Young, B.; Johnson, S.; Minoo, B.; Shugarts, D.; Allen, M.; Ramey, R.R.; et al. Adhesion of Staphylococcus aureus to Collagen Is Not a Major Virulence Determinant for Septic Arthritis, Osteomyelitis, or Endocarditis. J. Infect. Dis. 1999, 179, 291–293. [Google Scholar] [CrossRef] [PubMed]
- Elasri, M.; Thomas, J.; Skinner, R.; Blevins, J.; Beenken, K.; Nelson, C.; Smelter, M. Staphylococcus aureus collagen adhesin contributes to the pathogenesis of osteomyelitis. Bone 2002, 30, 275–280. [Google Scholar] [CrossRef]
- Rhem, M.N.; Lech, E.M.; Patti, J.M.; McDevitt, D.; Hook, M.; Jones, D.B.; Wilhelmus, K.R. The Collagen-Binding Adhesin Is a Virulence Factor in Staphylococcus aureus Keratitis. Infect. Immun. 2000, 68, 3776–3779. [Google Scholar] [CrossRef]
- Patti, J.M.; Bremell, T.; Krajewska-Pietrasik, D.; Abdelnour, A.; Tarkowski, A.; Rydén, C.; Höök, M. The Staphylococcus aureus collagen adhesin is a virulence determinant in experimental septic arthritis. Infect. Immun. 1994, 62, 152–161. [Google Scholar] [CrossRef]
- Kang, M.; Ko, Y.-P.; Liang, X.; Ross, C.L.; Liu, Q.; Murray, B.E.; Höök, M. Collagen-binding Microbial Surface Components Recognizing Adhesive Matrix Molecule (MSCRAMM) of Gram-positive Bacteria Inhibit Complement Activation via the Classical Pathway. J. Biol. Chem. 2013, 288, 20520–20531. [Google Scholar] [CrossRef]
- Falugi, F.; Kim, H.K.; Missiakas, D.M.; Schneewind, O. Role of Protein A in the Evasion of Host Adaptive Immune Responses by Staphylococcus aureus. mBio 2013, 4, e00575. [Google Scholar] [CrossRef] [PubMed]
- O’Seaghdha, M.; Van Schooten, C.J.; Kerrigan, S.W.; Emsley, J.; Silverman, G.J.; Cox, D.; Lenting, P.J.; Foster, T.J. Staphylococcus aureusprotein A binding to von Willebrand factor A1 domain is mediated by conserved IgG binding regions. FEBS J. 2006, 273, 4831–4841. [Google Scholar] [CrossRef]
- Gómez, M.I.; O’Seaghdha, M.; Magargee, M.; Foster, T.J.; Prince, A.S. Staphylococcus aureus Protein A Activates TNFR1 Signaling through Conserved IgG Binding Domains. J. Biol. Chem. 2006, 281, 20190–20196. [Google Scholar] [CrossRef]
- Viela, F.; Prystopiuk, V.; Leprince, A.; Mahillon, J.; Speziale, P.; Pietrocola, G.; Dufrêne, Y.F. Binding ofStaphylococcus aureusProtein A to von Willebrand Factor Is Regulated by Mechanical Force. mBio 2019, 10, e00555. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.-H.; Jiang, Y.-L.; Zhang, J.; Wang, L.; Bai, X.-H.; Zhang, S.-J.; Ren, Y.-M.; Li, N.; Zhang, Y.-H.; Zhang, Z.; et al. Structural Insights into SraP-Mediated Staphylococcus aureus Adhesion to Host Cells. PLoS Pathog. 2014, 10, e1004169. [Google Scholar] [CrossRef] [PubMed]
- Kukita, K.; Kawada-Matsuo, M.; Oho, T.; Nagatomo, M.; Oogai, Y.; Hashimoto, M.; Suda, Y.; Tanaka, T.; Komatsuzawa, H. Staphylococcus aureus SasA Is Responsible for Binding to the Salivary Agglutinin gp340, Derived from Human Saliva. Infect. Immun. 2013, 81, 1870–1879. [Google Scholar] [CrossRef] [PubMed]
- Torres, V.J.; Pishchany, G.; Humayun, M.; Schneewind, O.; Skaar, E.P. Staphylococcus aureus IsdB Is a Hemoglobin Receptor Required for Heme Iron Utilization. J. Bacteriol. 2006, 188, 8421–8429. [Google Scholar] [CrossRef]
- Pietrocola, G.; Pellegrini, A.; Alfeo, M.J.; Marchese, L.; Foster, T.J.; Speziale, P. The iron-regulated surface determinant B (IsdB) protein from Staphylococcus aureus acts as a receptor for the host protein vitronectin. J. Biol. Chem. 2020, 295, 10008–10022. [Google Scholar] [CrossRef]
- Ponnuraj, K.; Bowden, M.; Davis, S.; Gurusiddappa, S.; Moore, D.; Choe, D.; Xu, Y.; Hook, M.; Narayana, S.V. A “dock, lock, and latch” Structural Model for a Staphylococcal Adhesin Binding to Fibrinogen. Cell 2003, 115, 217–228. [Google Scholar] [CrossRef]
- Rohde, H.; Burdelski, C.; Bartscht, K.; Hussain, M.; Buck, F.; Horstkotte, M.A.; Knobloch, J.K.-M.; Heilmann, C.; Herrmann, M.; Mack, D. Induction ofStaphylococcus epidermidisbiofilm formation via proteolytic processing of the accumulation-associated protein by staphylococcal and host proteases. Mol. Microbiol. 2005, 55, 1883–1895. [Google Scholar] [CrossRef]
- Paharik, A.E.; Kotasinska, M.; Both, A.; Hoang, T.-M.N.; Büttner, H.; Roy, P.; Fey, P.D.; Horswill, A.R.; Rohde, H. The metalloprotease SepA governs processing of accumulation-associated protein and shapes intercellular adhesive surface properties inStaphylococcus epidermidis. Mol. Microbiol. 2017, 103, 860–874. [Google Scholar] [CrossRef]
- Brusselmans, K.; Timmermans, L.; Van de Sande, T.; Van Veldhoven, P.P.; Guan, G.; Shechter, I.; Claessens, F.; Verhoeven, G.; Swinnen, J.V. Squalene Synthase, a Determinant of Raft-associated Cholesterol and Modulator of Cancer Cell Proliferation. J. Biol. Chem. 2007, 282, 18777–18785. [Google Scholar] [CrossRef] [PubMed]
- Shahrooei, M.; Hira, V.; Stijlemans, B.; Merckx, R.; Hermans, P.W.M.; Van Eldere, J. Inhibition of Staphylococcus epidermidis Biofilm Formation by Rabbit Polyclonal Antibodies against the SesC Protein. Infect. Immun. 2009, 77, 3670–3678. [Google Scholar] [CrossRef] [PubMed]
- Khodaparast, L.; Khodaparast, L.; Van Mellaert, L.; Shahrooei, M.; Van Ranst, M.; Van Eldere, J. sesC as a genetic marker for easy identification of Staphylococcus epidermidis from other isolates. Infect. Genet. Evol. 2016, 43, 222–224. [Google Scholar] [CrossRef] [PubMed]
- Bartual, S.G.; Alcorlo, M.; Martínez-Caballero, S.; Molina, R.; Hermoso, J.A. Three-dimensional structures of Lipoproteins from Streptococcus pneumoniae and Staphylococcus aureus. Int. J. Med. Microbiol. 2018, 308, 692–704. [Google Scholar] [CrossRef]
- Mariotti, P.; Malito, E.; Biancucci, M.; Surdo, P.L.; Mishra, R.P.N.; Nardi-Dei, V.; Savino, S.; Nissum, M.; Spraggon, G.; Grandi, G.; et al. Structural and functional characterization of the Staphylococcus aureus virulence factor and vaccine candidate FhuD2. Biochem. J. 2013, 449, 683–693. [Google Scholar] [CrossRef]
- Horsburgh, M.J.; Wharton, S.J.; Cox, A.G.; Ingham, E.; Peacock, S.; Foster, S.J. MntR modulates expression of the PerR regulon and superoxide resistance in Staphylococcus aureus through control of manganese uptake. Mol. Microbiol. 2002, 44, 1269–1286. [Google Scholar] [CrossRef]
- Tribelli, P.M.; Luqman, A.; Nguyen, M.; Madlung, J.; Fan, S.; Macek, B.; Sass, P.; Bitschar, K.; Schittek, B.; Kretschmer, D.; et al. Staphylococcus aureus Lpl protein triggers human host cell invasion via activation of Hsp90 receptor. Cell. Microbiol. 2020, 22, e13111. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liu, F.-J.; Guan, Z.-C.; Dong, F.-T.; Cheng, J.-H.; Gao, Y.-P.; Li, D.; Yan, J.; Liu, C.-H.; Han, D.-P.; et al. The extracellular domain of Staphylococcus aureus LtaS binds insulin and induces insulin resistance during infection. Nat. Microbiol. 2018, 3, 622–631. [Google Scholar] [CrossRef]
- Heilmann, C.; Thumm, G.; Chhatwal, G.S.; Hartleib, J.; Uekötter, A.; Peters, G. Identification and characterization of a novel autolysin (Aae) with adhesive properties from Staphylococcus epidermidis. Microbiol. 2003, 149, 2769–2778. [Google Scholar] [CrossRef]
- Bowden, M.G.; Visai, L.; Longshaw, C.M.; Holland, K.T.; Speziale, P.; Höök, M. Is the GehD Lipase from Staphylococcus epidermidis a Collagen Binding Adhesin? J. Biol. Chem. 2002, 277, 43017–43023. [Google Scholar] [CrossRef]
- Christner, M.; Franke, G.C.; Schommer, N.N.; Wendt, U.; Wegert, K.; Pehle, P.; Kroll, G.; Schulze, C.; Buck, F.; Mack, D.; et al. The giant extracellular matrix-binding protein ofStaphylococcus epidermidismediates biofilm accumulation and attachment to fibronectin. Mol. Microbiol. 2010, 75, 187–207. [Google Scholar] [CrossRef] [PubMed]
- Büttner, H.; Perbandt, M.; Kohler, T.; Kikhney, A.; Wolters, M.; Christner, M.; Heise, M.; Wilde, J.; Weißelberg, S.; Both, A.; et al. A Giant Extracellular Matrix Binding Protein of Staphylococcus epidermidis Binds Surface-Immobilized Fibronectin via a Novel Mechanism. mBio 2020, 11, e01612-20. [Google Scholar] [CrossRef]
- Rooijakkers, S.H.M.; Ruyken, M.; Van Roon, J.; Van Kessel, K.P.M.; Van Strijp, J.A.G.; Van Wamel, W.J.B. Early expression of SCIN and CHIPS drives instant immune evasion by Staphylococcus aureus. Cell. Microbiol. 2006, 8, 1282–1293. [Google Scholar] [CrossRef]
- De Haas, C.J.; Veldkamp, K.E.; Peschel, A.; Weerkamp, F.; Van Wamel, W.J.; Heezius, E.C.; Poppelier, M.J.; Van Kessel, K.P.; Van Strijp, J.A. Chemotaxis Inhibitory Protein of Staphylococcus aureus, a Bacterial Antiinflammatory Agent. J. Exp. Med. 2004, 199, 687–695. [Google Scholar] [CrossRef] [PubMed]
- Postma, B.; Kleibeuker, W.; Poppelier, M.J.J.G.; Boonstra, M.; Van Kessel, K.P.M.; Van Strijp, J.A.G.; De Haas, C.J.C. Residues 10–18 within the C5a Receptor N Terminus Compose a Binding Domain for Chemotaxis Inhibitory Protein of Staphylococcus aureus. J. Biol. Chem. 2005, 280, 2020–2027. [Google Scholar] [CrossRef] [PubMed]
- Jongerius, I.; Köhl, J.; Pandey, M.K.; Ruyken, M.; Van Kessel, K.P.; Van Strijp, J.A.; Rooijakkers, S.H. Staphylococcal complement evasion by various convertase-blocking molecules. J. Exp. Med. 2007, 204, 2461–2471. [Google Scholar] [CrossRef]
- Ko, Y.-P.; Liang, X.; Smith, C.W.; Degen, J.L.; Höök, M. Binding of Efb from Staphylococcus aureus to Fibrinogen Blocks Neutrophil Adherence*. J. Biol. Chem. 2011, 286, 9865–9874. [Google Scholar] [CrossRef]
- Woehl, J.L.; Stapels, D.A.C.; Garcia, B.L.; Ramyar, K.X.; Keightley, A.; Ruyken, M.; Syriga, M.; Sfyroera, G.; Weber, A.B.; Zolkiewski, M.; et al. The Extracellular Adherence Protein fromStaphylococcus aureusInhibits the Classical and Lectin Pathways of Complement by Blocking Formation of the C3 Proconvertase. J. Immunol. 2014, 193, 6161–6171. [Google Scholar] [CrossRef]
- Stapels, D.A.; Ramyar, K.X.; Bischoff, M.; Köckritz-Blickwede, M.; Milder, F.J.; Ruyken, M.; Eisenbeis, J.; McWhorter, W.J.; Herrmann, M.; Kessel, K.P.; et al. Staphylococcus aureus secretes a unique class of neutrophil serine protease inhibitors. Proc. Natl. Acad. Sci. USA 2014, 111, 13187–13192. [Google Scholar] [CrossRef]
- Panizzi, P.; Friedrich, R.; Fuentes-Prior, P.; Kroh, H.K.; Briggs, J.; Tans, G.; Bode, W.; Bock, P.E. Novel Fluorescent Prothrombin Analogs as Probes of Staphylocoagulase-Prothrombin Interactions. J. Biol. Chem. 2006, 281, 1169–1178. [Google Scholar] [CrossRef] [PubMed]
- Friedrich, R.; Panizzi, P.; Fuentes-Prior, P.; Richter, K.; Verhamme, I.; Anderson, P.J.; Kawabata, S.-I.; Huber, R.; Bode, W.; Bock, P.E. Staphylocoagulase is a prototype for the mechanism of cofactor-induced zymogen activation. Nat. Cell Biol. 2003, 425, 535–539. [Google Scholar] [CrossRef] [PubMed]
- Bjerketorp, J.; Nilsson, M.; Ljungh, Å.; Flock, J.-I.; Jacobsson, K.; Frykberg, L. A novel von Willebrand factor binding protein expressed by Staphylococcus aureus a The GenBank accession number for the sequence reported in this paper is AY032850. Microbiology 2002, 148, 2037–2044. [Google Scholar] [CrossRef] [PubMed]
- Hendrix, H.; Lindhout, T.; Mertens, K.; Engels, W.; Hemker, H.C. Activation of human prothrombin by stoichiometric levels of staphylocoagulase. J. Biol. Chem. 1983, 258, 3637–3644. [Google Scholar] [CrossRef]
- Kroh, H.K.; Panizzi, P.; Bock, P.E. Von Willebrand factor-binding protein is a hysteretic conformational activator of prothrombin. Proc. Natl. Acad. Sci. USA 2009, 106, 7786–7791. [Google Scholar] [CrossRef] [PubMed]
- Thomer, L.; Schneewind, O.; Missiakas, D. Multiple Ligands of von Willebrand Factor-binding Protein (vWbp) Promote Staphylococcus aureus Clot Formation in Human Plasma. J. Biol. Chem. 2013, 288, 28283–28292. [Google Scholar] [CrossRef]
- Thomas, S.; Liu, W.; Arora, S.; Ganesh, V.; Ko, Y.-P.; Höök, M. The Complex Fibrinogen Interactions of the Staphylococcus aureus Coagulases. Front. Cell. Infect. Microbiol. 2019, 9, 106. [Google Scholar] [CrossRef]
- Bjerketorp, J.; Jacobsson, K.; Frykberg, L. The von Willebrand factor-binding protein (vWbp) of Staphylococcus aureus is a coagulase. FEMS Microbiol. Lett. 2004, 234, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Tam, K.; Torres, V.J. Staphylococcus aureus Secreted Toxins and Extracellular Enzymes. Microbiol. Spectr. 2019, 7, 10. [Google Scholar] [CrossRef]
- Caiazza, N.C.; O’Toole, G.A. Alpha-Toxin Is Required for Biofilm Formation by Staphylococcus aureus. J. Bacteriol. 2003, 185, 3214–3217. [Google Scholar] [CrossRef]
- Anderson, M.J.; Schaaf, E.; Breshears, L.M.; Wallis, H.W.; Johnson, J.R.; Tkaczyk, C.; Sellman, B.R.; Sun, J.; Peterson, M.L. Alpha-Toxin Contributes to Biofilm Formation among Staphylococcus aureus Wound Isolates. Toxins 2018, 10, 157. [Google Scholar] [CrossRef]
- Scherr, T.D.; Hanke, M.L.; Huang, O.; James, D.B.A.; Horswill, A.R.; Bayles, K.W.; Fey, P.D.; Torres, V.J.; Kielian, T. Staphylococcus aureus Biofilms Induce Macrophage Dysfunction Through Leukocidin AB and Alpha-Toxin. mBio 2015, 6, e01025. [Google Scholar] [CrossRef]
- Estellés, A.; Woischnig, A.-K.; Liu, K.; Stephenson, R.; Lomongsod, E.; Nguyen, D.; Zhang, J.; Heidecker, M.; Yang, Y.; Simon, R.J.; et al. A High-Affinity Native Human Antibody Disrupts Biofilm from Staphylococcus aureus Bacteria and Potentiates Antibiotic Efficacy in a Mouse Implant Infection Model. Antimicrob. Agents Chemother. 2016, 60, 2292–2301. [Google Scholar] [CrossRef]
- Xiong, Y.Q.; Estellés, A.; Li, L.; Abdelhady, W.; Gonzales, R.; Bayer, A.S.; Tenorio, E.; Leighton, A.; Ryser, S.; Kauvar, L.M. A Human Biofilm-Disrupting Monoclonal Antibody Potentiates Antibiotic Efficacy in Rodent Models of both Staphylococcus aureus and Acinetobacter baumannii Infections. Antimicrob. Agents Chemother. 2017, 61, e00904. [Google Scholar] [CrossRef] [PubMed]
- Goodman, S.D.; Obergfell, K.P.; Jurcisek, J.A.; Novotny, L.A.; Downey, J.S.; Ayala, E.A.; Tjokro, N.O.; Li, B.; Justice, S.S.; Bakaletz, L.O. Biofilms can be dispersed by focusing the immune system on a common family of bacterial nucleoid-associated proteins. Mucosal Immunol. 2011, 4, 625–637. [Google Scholar] [CrossRef]
- Kuby, J. Immunoglobulins: Structure and Functions. In Immunology, 2nd ed.; Schroeder, H.W., Ed.; Freeman and Company: New York, NY, USA, 1994; pp. 109–134. [Google Scholar]
- Janeway, C.A.; Travers, P.; Walport, M.; Shlomchik, M.J. The generation of lymphocyte antigen receptors. In Immuno-Biology, 5th ed.; Garland Science: New York, NY, USA, 2001; pp. 123–154. [Google Scholar]
- Moldenhauer, G. Selecting strategies I: Monoclonal antibodies. In Handbook of Therapeutic Antibodies; Dübel, S., Ed.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2010; pp. 19–44. [Google Scholar]
- Hall, A.E.; Domanski, P.J.; Patel, P.R.; Vernachio, J.H.; Syribeys, P.J.; Gorovits, E.L.; Johnson, M.A.; Ross, J.M.; Hutchins, J.T.; Patti, J.M. Characterization of a Protective Monoclonal AntibodyRecognizing Staphylococcus aureus MSCRAMM ProteinClumping FactorA. Infect. Immun. 2003, 71, 6864–6870. [Google Scholar] [CrossRef]
- Patti, J.M. A humanized monoclonal antibody targeting Staphylococcus aureus. Vaccine 2004, 22, S39–S43. [Google Scholar] [CrossRef] [PubMed]
- Weems, J.J.; Steinberg, J.P.; Filler, S.; Baddley, J.W.; Corey, G.R.; Sampathkumar, P.; Winston, L.; John, J.F.; Kubin, C.J.; Talwani, R.; et al. Phase II, Randomized, Double-Blind, Multicenter Study Comparing the Safety and Pharmacokinetics of Tefibazumab to Placebo for Treatment of Staphylococcus aureus Bacteremia. Antimicrob. Agents Chemother. 2006, 50, 2751–2755. [Google Scholar] [CrossRef]
- Schaffer, A.C.; Solinga, R.M.; Cocchiaro, J.; Portoles, M.; Kiser, K.B.; Risley, A.; Randall, S.M.; Valtulina, V.; Speziale, P.; Walsh, E.; et al. Immunization with Staphylococcus aureus Clumping Factor B, a Major Determinant in Nasal Carriage, Reduces Nasal Colonization in a Murine Model. Infect. Immun. 2006, 74, 2145–2153. [Google Scholar] [CrossRef]
- Visai, L.; Xu, Y.; Casolini, F.; Rindi, S.; Höök, M.; Speziale, P. Monoclonal Antibodies to CNA, a Collagen-binding Microbial Surface Component Recognizing Adhesive Matrix Molecules, DetachStaphylococcus aureus from a Collagen Substrate. J. Biol. Chem. 2000, 275, 39837–39845. [Google Scholar] [CrossRef] [PubMed]
- Herman-Bausier, P.; Valotteau, C.; Pietrocola, G.; Rindi, S.; Alsteens, D.; Foster, T.J.; Speziale, P.; Dufrene, Y.F. Mechanical Strength and Inhibition of the Staphylococcus aureus Collagen-Binding Protein Cna. mBio 2016, 7, e01529. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.K.; Emolo, C.; DeDent, A.C.; Falugi, F.; Missiakas, D.M.; Schneewind, O. Protein A-Specific Monoclonal Antibodies and Prevention of Staphylococcus aureus Disease in Mice. Infect. Immun. 2012, 80, 3460–3470. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.K.; Cheng, A.G.; Kim, H.-Y.; Missiakas, D.M.; Schneewind, O. Nontoxigenic protein A vaccine for methicillin-resistant Staphylococcus aureus infections in mice. J. Exp. Med. 2010, 207, 1863–1870. [Google Scholar] [CrossRef]
- Chen, X.; Sun, Y.; Missiakas, D.; Schneewind, O. Staphylococcus aureus Decolonization of Mice With Monoclonal Antibody Neutralizing Protein A. J. Infect. Dis. 2019, 219, 884–888. [Google Scholar] [CrossRef]
- Thammavongsa, V.; Rauch, S.; Kim, H.K.; Missiakas, D.M.; Schneewind, O. Protein A-neutralizing monoclonal antibody protects neonatal mice against Staphylococcus aureus. Vaccine 2015, 33, 523–526. [Google Scholar] [CrossRef] [PubMed]
- Varshney, A.K.; Kuzmicheva, G.A.; Lin, J.; Sunley, K.M.; Bowling, R.A., Jr.; Kwan, T.-Y.; Mays, H.R.; Rambhadran, A.; Zhang, Y.; Martin, R.L.; et al. A natural human monoclonal antibody targeting Staphylococcus Protein A protects against Staphylococcus aureus bacteremia. PLoS ONE 2018, 13, e0190537. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.-T.; Yue, Y.; Zheng, F.; Liang, X.-D.; Cao, Q.-X.; Wang, Y.-W.; Zhu, J. Monoclonal antibody against l-lectin module of SraP blocks adhesion and protects mice against Staphylococcus aureus challenge. J. Microbiol. Immunol. Infect. 2019, 1684, 30115. [Google Scholar] [CrossRef] [PubMed]
- Thomer, L.; Emolo, C.; Thammavongsa, V.; Kim, H.K.; McAdow, M.E.; Yu, W.; Kieffer, M.; Schneewind, O.; Missiakas, D. Antibodies against a secreted product of Staphylococcus aureus trigger phagocytic killing. J. Exp. Med. 2016, 213, 293–301. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Jagasia, R.; Kaufmann, G.F.; Mathison, J.C.; Ruiz, D.I.; Moss, J.A.; Meijler, M.M.; Ulevitch, R.J.; Janda, K.D. Infection Control by Antibody Disruption of Bacterial Quorum Sensing Signaling. Chem. Biol. 2007, 14, 1119–1127. [Google Scholar] [CrossRef] [PubMed]
- Kirchdoerfer, R.N.; Garner, A.L.; Flack, C.E.; Mee, J.M.; Horswill, A.R.; Janda, K.D.; Kaufmann, G.F.; Wilson, I.A. Structural Basis for Ligand Recognition and Discrimination of a Quorum-quenching Antibody. J. Biol. Chem. 2011, 286, 17351–17358. [Google Scholar] [CrossRef]
- Oganesyan, V.; Peng, L.; Damschroder, M.M.; Cheng, L.; Sadowska, A.; Tkaczyk, C.; Sellman, B.R.; Wu, H.; Dall’Acqua, W.F. Mechanisms of Neutralization of a Human Anti-α-toxin Antibody. J. Biol. Chem. 2014, 289, 29874–29880. [Google Scholar] [CrossRef]
- Hua, L.; Hilliard, J.J.; Shi, Y.; Tkaczyk, C.; Cheng, L.I.; Yu, X.; Datta, V.; Ren, S.; Feng, H.; Zinsou, R.; et al. Assessment of an Anti-Alpha-Toxin Monoclonal Antibody for Prevention and Treatment of Staphylococcus aureus-Induced Pneumonia. Antimicrob. Agents Chemother. 2013, 58, 1108–1117. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.-Q.; Robbie, G.J.; Wu, Y.; Esser, M.T.; Jensen, K.; Schwartz, H.I.; Bellamy, T.; Hernandez-Illas, M.; Jafri, H.S. Safety, Tolerability, and Pharmacokinetics of MEDI4893, an Investigational, Extended-Half-Life, Anti-Staphylococcus aureus Alpha-Toxin Human Monoclonal Antibody, in Healthy Adults. Antimicrob. Agents Chemother. 2017, 61, 01020-16. [Google Scholar] [CrossRef] [PubMed]
- Ruzin, A.; Wu, Y.; Yu, L.; Yu, X.-Q.; Tabor, D.E.; Mok, H.; Tkaczyk, C.; Jensen, K.; Bellamy, T.; Roskos, L.; et al. Charac-terisation of anti-alpha toxin antibody levels and colonisation status after administration of an investigational human monoclonal antibody, MEDI4893, against Staphylococcus aureus alpha toxin. Clin. Transl. Immunol. 2018, 7, e1009. [Google Scholar] [CrossRef]
- Aguilar, J.L.; Varshney, A.K.; Pechuan, X.; Dutta, K.; Nosanchuk, J.D.; Fries, B.C. Monoclonal antibodies protect from Staphylococcal Enterotoxin K (SEK) induced toxic shock and sepsis by USA300Staphylococcus aureus. Virulence 2016, 8, 741–750. [Google Scholar] [CrossRef]
- Hall, A.E.; Patel, P.R.; Domanski, P.J.; Prater, B.D.; Gorovits, E.L.; Syribeys, P.J.; Vernachio, J.H.; Patti, J.M.; Hutchins, J.T. A Panel of Monoclonal Antibodies Recognizing theStaphylococcus epidermidisFibrinogen-BindingMSCRAMM SdrG. Hybridoma 2007, 26, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.; Kowalski, R.; Zorman, J.; Wang, X.-M.; Towne, V.; Zhao, Q.; Secore, S.; Finnefrock, A.C.; Ebert, T.; Pancari, G.; et al. Selection and Characterization of Murine Monoclonal Antibodies to Staphylococcus aureus Iron-Regulated Surface Determinant B with Functional Activity In Vitro and In Vivo. Clin. Vaccine Immunol. 2009, 16, 1095–1104. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Shi, M.; Tong, X.; Kim, H.K.; Wang, L.-X.; Schneewind, O.; Missiakas, D. Glycosylation-dependent opsonophagocytic activity of staphylococcal protein A antibodies. Proc. Natl. Acad. Sci. USA 2020, 117, 22992–23000. [Google Scholar] [CrossRef]
- Huynh, T.; Stecher, M.; McKinnon, J.; Jung, N.; Rupp, M.E. Safety and Tolerability of 514G3, a True Human Anti-Protein A Monoclonal Antibody for the Treatment of S. aureus Bacteremia. Open Forum Infect. Dis. 2016, 3, 1354. [Google Scholar] [CrossRef]
- Yang, Y.; Qian, M.; Yi, S.; Liu, S.; Li, B.; Yu, R.; Guo, Q.; Zhang, X.; Yu, C.; Li, J.; et al. Monoclonal Antibody Targeting Staphylococcus aureus Surface Protein A (SasA) Protect Against Staphylococcus aureus Sepsis and Peritonitis in Mice. PLoS ONE 2016, 11, e0149460. [Google Scholar] [CrossRef]
- Anderson, A.S.; Scully, I.L.; Timofeyeva, Y.; Murphy, E.; McNeil, L.K.; Mininni, T.; Nuñez, L.; Carriere, M.; Singer, C.; Dilts, D.A.; et al. Staphylococcus aureus Manganese Transport Protein C Is a Highly Conserved Cell Surface Protein That Elicits Protective Immunity Against S. aureus and Staphylococcus epidermidis. J. Infect. Dis. 2012, 205, 1688–1696. [Google Scholar] [CrossRef] [PubMed]
- Burnie, J.P.; Matthews, R.C.; Carter, T.; Beaulieu, E.; Donohoe, M.; Chapman, C.; Williamson, P.; Hodgetts, S.J. Identification of an Immunodominant ABC Transporter in Methicillin-Resistant Staphylococcus aureusInfections. Infect. Immun. 2000, 68, 3200–3209. [Google Scholar] [CrossRef]
- Otto, M. Novel targeted immunotherapy approaches for staphylococcal infection. Expert Opin. Biol. Ther. 2010, 10, 1049–1059. [Google Scholar] [CrossRef]
- Baker, M. Anti-infective antibodies: Finding the path forward. Nat. Biotechnol. 2006, 24, 1491–1493. [Google Scholar] [CrossRef]
- Georgoutsou-Spyridonos, M.; Ricklin, D.; Pratsinis, H.; Perivolioti, E.; Pirmettis, I.; Garcia, B.L.; Geisbrecht, B.V.; Foukas, P.G.; Lambris, J.D.; Mastellos, D.C.; et al. Attenuation ofStaphylococcus aureus–Induced Bacteremia by Human Mini-Antibodies Targeting the Complement Inhibitory Protein Efb. J. Immunol. 2015, 195, 3946–3958. [Google Scholar] [CrossRef]
- Haas, P.-J.; De Haas, C.J.C.; Kleibeuker, W.; Poppelier, M.J.J.G.; Van Kessel, K.P.M.; Kruijtzer, J.A.W.; Liskamp, R.M.J.; Van Strijp, J.A.G. N-Terminal Residues of the Chemotaxis Inhibitory Protein of Staphylococcus aureus Are Essential for Blocking Formylated Peptide Receptor but Not C5a Receptor. J. Immunol. 2004, 173, 5704–5711. [Google Scholar] [CrossRef]
- Hoekstra, H.; Pastrana, F.R.; Bonarius, H.P.J.; Van Kessel, K.P.M.; Elsinga, G.S.; Kooi, N.; Groen, H.; Van Dijl, J.M.; Buist, G. A human monoclonal antibody that specifically binds and inhibits the staphylococcal complement inhibitor protein SCIN. Virulence 2018, 9, 70–82. [Google Scholar] [CrossRef] [PubMed]
- Rouha, H.; Badarau, A.; Visram, Z.C.; Battles, M.B.; Prinz, B.; Magyarics, Z.; Nagy, G.; Mirkina, I.; Stulik, L.; Zerbs, M.; et al. Five birds, one stone: Neutralization of α-hemolysin and 4 bi-component leukocidins of Staphylococcus aureus with a single human monoclonal antibody. mAbs 2015, 7, 243–254. [Google Scholar] [CrossRef]
- Stulik, L.; Rouha, H.; Labrousse, D.; Visram, Z.C.; Badarau, A.; Maierhofer, B.; Groß, K.; Weber, S.; Kramarić, M.D.; Glojnarić, I.; et al. Preventing lung pathology and mortality in rabbit Staphylococcus aureus pneumonia models with cytotoxin-neutralizing monoclonal IgGs penetrating the epithelial lining fluid. Sci. Rep. 2019, 9, 5339. [Google Scholar] [CrossRef]
- Varshney, A.K.; Wang, X.; MacIntyre, J.; Zollner, R.S.; Kelleher, K.; Kovalenko, O.V.; Pechuan, X.; Byrne, F.R.; Fries, B.C. Humanized Staphylococcal Enterotoxin B (SEB)–Specific Monoclonal Antibodies Protect From SEB Intoxication and Staphylococcus aureus Infections Alone or as Adjunctive Therapy With Vancomycin. J. Infect. Dis. 2014, 210, 973–981. [Google Scholar] [CrossRef] [PubMed]
- Karau, M.J.; Tilahun, M.E.; Krogman, A.; Osborne, B.A.; Goldsby, R.A.; David, C.S.; Mandrekar, J.N.; Patel, R.; Rajagopalan, G. Passive therapy with humanized anti-staphylococcal enterotoxin B antibodies attenuates systemic inflammatory response and protects from lethal pneumonia caused by staphylococcal enterotoxin B-producingStaphylococcus aureus. Virulence 2017, 8, 1148–1159. [Google Scholar] [CrossRef] [PubMed]
- Rukkawattanakul, T.; Sookrung, N.; Seesuay, W.; Onlamoon, N.; Diraphat, P.; Chaicumpa, W.; Indrawattana, N. Human scFvs That Counteract Bioactivities of Staphylococcus aureus TSST-1. Toxins 2017, 9, 50. [Google Scholar] [CrossRef]
- Sun, D.; Accavitti, M.A.; Bryers, J.D. Inhibition of Biofilm Formation by Monoclonal Antibodies against Staphylococcus epidermidis RP62A Accumulation-Associated Protein. Clin. Diagn. Lab. Immunol. 2005, 12, 93–100. [Google Scholar] [CrossRef]
- Ortines, R.V.; Liu, H.; Cheng, L.I.; Cohen, T.S.; Lawlor, H.; Gami, A.; Wang, Y.; Dillen, C.A.; Archer, N.K.; Miller, R.J.; et al. Neutralizing Alpha-Toxin Accelerates Healing of Staphylococcus aureus -Infected Wounds in Nondiabetic and Diabetic Mice. Antimicrob. Agents Chemother. 2018, 62, 2288. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Cheng, L.I.; Helfer, D.R.; Ashbaugh, A.G.; Miller, R.J.; Tzomides, A.J.; Thompson, J.M.; Ortines, R.V.; Tsai, A.S.; Liu, H.; et al. Mouse model of hematogenous implant-relatedStaphylococcus aureusbiofilm infection reveals therapeutic targets. Proc. Natl. Acad. Sci. USA 2017, 114, E5094–E5102. [Google Scholar] [CrossRef] [PubMed]
- Lehar, S.M.; Pillow, T.; Xu, M.; Staben, L.; Kajihara, K.K.; Vandlen, R.; DePalatis, L.; Raab, H.; Hazenbos, W.L.; Morisaki, J.H.; et al. Novel antibody–antibiotic conjugate eliminates intracellular S. aureus. Nature 2015, 527, 323–328. [Google Scholar] [CrossRef]
- Mariathasan, S.; Tan, M.-W. Antibody–Antibiotic Conjugates: A Novel Therapeutic Platform against Bacterial Infections. Trends Mol. Med. 2017, 23, 135–149. [Google Scholar] [CrossRef] [PubMed]
- Bispo, M.; Anaya-Sanchez, A.; Suhani, S.; Raineri, E.J.M.; López-Álvarez, M.; Heuker, M.; Szymański, W.; Pastrana, F.R.; Buist, G.; Horswill, A.R.; et al. Fighting Staphylococcus aureus infections with light and photoimmunoconjugates. JCI Insight 2020, 5, 139512. [Google Scholar] [CrossRef]
- Millenbaugh, N.J.; Baskin, J.B.; DeSilva, M.N.; Elliott, W.R.; Glickman, R.D. Photothermal killing of Staphylococcus aureus using antibody-targeted gold nanoparticles. Int. J. Nanomed. 2015, 10, 1953–1960. [Google Scholar] [CrossRef]
- Vor, L.; Dijk, B.; Kessel, K.P.M.; Kavanaugh, J.S.; Haas, C.J.C.; Aerts, P.C.; Viveen, M.C.; Boel, E.C.H.; Fluit, A.D.C.; Kwiecinski, J.M.; et al. Human monoclonal antibodies against Staphylococcus aureus surface antigens recognize in vitro biofilm and in vivo implant infections. Biorxiv Microbiol. 2021. [Google Scholar] [CrossRef]
- Swerlick, R.A.; Cunningham, M.W.; Hall, N.K. Monoclonal Antibodies Cross-Reactive with Group A Streptococci and Normal and Psoriatic Human Skin. J. Investig. Dermatol. 1986, 87, 367–371. [Google Scholar] [CrossRef] [PubMed]
- Descotes, J. Immunotoxicity of monoclonal antibodies. mAbs 2009, 1, 104–111. [Google Scholar] [CrossRef] [PubMed]

| Antigen (UniProtKB Accession Code) | Antibody | Ig Class | Recognized Domain/Epitope | In Vitro Activities of the Antibody | In Vivo Efficiency of the Antibody | Refs |
|---|---|---|---|---|---|---|
| ClfA (Q5HHM8) | Tefibazumab, a humanized mAb | IgG1 | N3 subdomain of A region | Blocks fibrinogen binding to ClfA. | Protects against infective endocarditis. | [79,80,81] |
| ClfB (O86476) | Mu/3D6 | IgG1k | N2-N3 subregion | Inhibits bacterial binding to cytokeratin 10. | Reduces nasal colonization by bacteria. | [82] |
| CNA (Q53654) | Mu/mAbs | IgG1k | CNA151-318 | Inhibits collagen binding to bacteria. | ̶ | [83,84] |
| SpA (A0A0H3K686) | Mu/Hum/3F6 Mu/514G3 | 3F6: IgG2a 514G3: IgG3 | 3F6: binds to each of the five immunoglobulin binding domains.514G3: binds to epitope by CDR. | 3F6: neutralizes domains of SpA associated with IgG/IgM binding. 514G3: induces phagocytosis. | 3F6: protects animals against bloodstream infection. 514G3: rescues mice from bacteria-mediated bacteremia. | [85,86,87,88,89] |
| SraP (Q5HCP3) | Mu/mAb | Unknown | Lectin domain | Inhibits bacterial adherence to epithelial cells. | ̶ | [90] |
| LtaS (Q5HHV4) | Hum/YG2 | Unknown | Extracellular domain of LtaS (eLtaS) | Inhibits the interaction between eLtsA and insulin. | Restores impaired glucose tolerance in mice. | [49] |
| Coa (P07767) | Mu/Hum/3B3 | IgG1 | C-terminal R domain | Promotes phagocytosis of fibrin-coated bacteria. | Protects mice against lethal bloodstream infection. | [91] |
| Autoinducing peptide-4 (AIP-4) | Mu/mAb AP4-Z4H11 | Unknown | YSTCYFIM | Inhibits QS in vitro through sequestration of AIP-4. | Suppresses bacterial pathogenicity in an abscess formation mouse model. | [92,93] |
| DNABII (Q5HFV0) | Hum/TRL 1068 | IgG1 | GRNPQTGKEID | Disrupts biofilm formation. | Blocks biofilm formation in a murine tissue cage infection model. | [73] |
| Hla (P09616) | Hum/MEDI4893 | IgG1k | Recognizes a conformational epitope in the “rim” domain of Hla. | Blocks the binding to ADAM10. | Affords protection to mice in a model of acute pneumonia. | [94,95,96,97] |
| SEK (A0A0H2WWN7) | Mu/mAb 4G3 Mu/mAb 5G2 | 4G3: IgG2b 5G2: IgG1 | 4G3 and 5G2 recognize distinct epitopes on SEK. | 4G3 and 5G2 inhibit SEK-induced proliferation of human immune cells. | Combination of 4G3 + 5G2 enhances survival of mice infected with bacteria. | [98] |
| Agent | Target Antigen | Primary Clinical Indication | Status |
|---|---|---|---|
| Aurexis (tefibazumab) | ClfA | Treatment of bacteremia | Not in active development |
| Aurograb | GrfA | Treatment of deep-seated infection | Ineffective in a phase II trial |
| MEDI4893 | Hla | Prevention of pneumonia | Phase II ongoing |
| 514G3 | SpA | Treatment of bacteremia | Phase I/II ongoing |
| ASN100 | Hla, HlgAB, HlgCB, LukED and LukSF | Treatment of pneumonia | Clinical trial failed |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Speziale, P.; Pietrocola, G. Monoclonal Antibodies Targeting Surface-Exposed and Secreted Proteins from Staphylococci. Vaccines 2021, 9, 459. https://doi.org/10.3390/vaccines9050459
Speziale P, Pietrocola G. Monoclonal Antibodies Targeting Surface-Exposed and Secreted Proteins from Staphylococci. Vaccines. 2021; 9(5):459. https://doi.org/10.3390/vaccines9050459
Chicago/Turabian StyleSpeziale, Pietro, and Giampiero Pietrocola. 2021. "Monoclonal Antibodies Targeting Surface-Exposed and Secreted Proteins from Staphylococci" Vaccines 9, no. 5: 459. https://doi.org/10.3390/vaccines9050459
APA StyleSpeziale, P., & Pietrocola, G. (2021). Monoclonal Antibodies Targeting Surface-Exposed and Secreted Proteins from Staphylococci. Vaccines, 9(5), 459. https://doi.org/10.3390/vaccines9050459







