Development of Diagnostic Tests Provides Technical Support for the Control of African Swine Fever
Abstract
1. Introduction
2. Epidemic Strains and Epidemic History of ASFV
3. Transmission
4. Control-Eradication Strategies
4.1. Africa
- Specific implementation options for the prevention and control of epidemics should be provided in light of the epidemic situation in specific countries and sectors.
- A complete monitoring system should be established. Disease monitoring and surveillance are key elements in the management of trans-boundary animal diseases (TADS), including ASF. This includes collecting, sorting, updating, and analyzing existing information related to epidemiology, which provide a knowledge base to understand the latest status of ASF in the region.
- The capabilities of diagnostic laboratories can be improved through the introduction of new diagnostic technologies and laboratory personnel training. Establish a network of diagnostic laboratories to predict the origin and evolution of disease outbreaks.
- Subregional management should be implemented. Domestic pigs in the ASF area must be placed in the enclosure and restricted to prevent contact with wild boars. In ASF-free zones, strict biosecurity measures will be carried out to protect domestic pig populations from diseases.
4.2. Europe
4.3. South America
5. Diagnosis of ASF
5.1. Clinical Features and Pathological Changes
5.2. Etiological Diagnosis
5.2.1. Virus Isolation and the Hemadsorption (HAD) Test
5.2.2. Polymerase Chain Reaction (PCR) Assay
5.2.3. Real-Time PCR Assay
5.2.4. Isothermal Amplification Technologies: RPA, LAMP and CPA
Recombinase Polymerase Amplification (RPA)
Loop-Mediated Isothermal Amplification (LAMP)
Cross-Priming Amplification (CPA)
5.3. Serological Diagnosis
5.3.1. Antigen-Based Detection
Fluorescent Antibody Test (FAT)
Direct ELISA
5.3.2. Antibody-Based Detection
Indirect ELISA
Indirect Fluorescent Antibody (IFA) Test, Indirect Immunoperoxidase Test (IPT), and Dot Immunobinding Assay (DIA)
Immunoblotting Test (IBT)
6. Discussion and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ASF | African swine fever |
| ASFV | African swine fever virus |
| ORFs | Open reading frames |
| NCLDVs | Nucleocytoplasmic large DNA viruses |
| FAO | Food and Agriculture Organization of Animal Health |
| TADS | Trans-boundary animal diseases |
| OIE | World Organization for Animal Health |
| MGF | Multigene families |
| HAD | Hemadsorption |
| PCR | Polymerase Chain Reaction |
| RPA | Recombinase polymerase amplification |
| LAMP | Loop-mediated isothermal amplification |
| CPA | Cross-priming amplification |
| FAT | Fluorescent antibody test |
| ELISA | Enzyme-linked immunosorbent assay |
| LFA | Lateral flow assay |
| IEOP | Immunoelectroosmophoresis |
References
- Costard, S.; Wieland, B.; De Glanville, W.; Jori, F.; Rowlands, R.; Vosloo, W.; Roger, F.; Pfeiffer, D.U.; Dixon, L.K. African swine fever: How can global spread be prevented? Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 2683–2696. [Google Scholar] [CrossRef]
- Gaudreault, N.N.; Madden, D.W.; Wilson, W.C.; Trujillo, J.D.; Richt, J.A. African Swine Fever Virus: An Emerging DNA Arbovirus. Front. Vet. Sci. 2020, 7, 215. [Google Scholar] [CrossRef]
- Dixon, L.K.; Chapman, D.A.; Netherton, C.L.; Upton, C. African swine fever virus replication and genomics. Virus Res. 2013, 173, 3–14. [Google Scholar] [CrossRef]
- Blasco, R.; Agüero, M.; Almendral, J.; Viñuela, E. Variable and constant regions in African swine fever virus DNA. Virology 1989, 168, 330–338. [Google Scholar] [CrossRef]
- Chapman, D.A.G.; Tcherepanov, V.; Upton, C.; Dixon, L.K. Comparison of the genome sequences of non-pathogenic and pathogenic African swine fever virus isolates. J. Gen. Virol. 2008, 89, 397–408. [Google Scholar] [CrossRef] [PubMed]
- Iyer, L.M.; Aravind, L.; Koonin, E.V. Common Origin of Four Diverse Families of Large Eukaryotic DNA Viruses. J. Virol. 2001, 75, 11720–11734. [Google Scholar] [CrossRef]
- Iyer, L.M.; Balaji, S.; Koonin, E.V.; Aravind, L. Evolutionary genomics of nucleo-cytoplasmic large DNA viruses. Virus Res. 2006, 117, 156–184. [Google Scholar] [CrossRef]
- Alonso, C.; Borca, M.; Dixon, L.; Revilla, Y.; Rodriguez, F.; Escribano, J.M. ICTV Report Consortium ICTV Virus Taxonomy Profile: Asfarviridae. J. Gen. Virol. 2018, 99, 613–614. [Google Scholar] [CrossRef]
- Salas, M.L.; Andrés, G. African swine fever virus morphogenesis. Virus Res. 2013, 173, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Andrés, G.; Simón-Mateo, C.; Viñuela, E. Assembly of African swine fever virus: Role of polyprotein pp220. J. Virol. 1997, 71, 2331–2341. [Google Scholar] [CrossRef] [PubMed]
- Andrés, G.; Alejo, A.; Salas, J.; Salas, M.L. African Swine Fever Virus Polyproteins pp220 and pp62 Assemble into the Core Shell. J. Virol. 2002, 76, 12473–12482. [Google Scholar] [CrossRef]
- Andrés, G.; Alejo, A.; Simón-Mateo, C.; Salas, M.L. African Swine Fever Virus Protease, a New Viral Member of the SUMO-1-specific Protease Family. J. Biol. Chem. 2001, 276, 780–787. [Google Scholar] [CrossRef] [PubMed]
- Alejo, A.; Matamoros, T.; Guerra, M.; Andrés, G. A Proteomic Atlas of the African Swine Fever Virus Particle. J. Virol. 2018, 92. [Google Scholar] [CrossRef] [PubMed]
- García-Escudero, R.; Andrés, G.; Almazán, F.; Viñuela, E. Inducible Gene Expression from African Swine Fever Virus Recombinants: Analysis of the Major Capsid Protein p72. J. Virol. 1998, 72, 3185–3195. [Google Scholar] [CrossRef] [PubMed]
- Andrés, G.; García-Escudero, R.; Viñuela, E.; Salas, M.L.; Rodríguez, J.M. African Swine Fever Virus Structural Protein pE120R Is Essential for Virus Transport from Assembly Sites to Plasma Membrane but Not for Infectivity. J. Virol. 2001, 75, 6758–6768. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Luo, Y.; Wang, Y.; Li, S.; Zhao, Z.; Bi, Y.; Sun, J.; Peng, R.; Song, H.; Zhu, D.; et al. Cryo-EM Structure of the African Swine Fever Virus. Cell Host Microbe 2019, 26, 836–843.e3. [Google Scholar] [CrossRef]
- Wang, N.; Zhao, D.; Wang, J.; Zhang, Y.; Wang, M.; Gao, Y.; Li, F.; Wang, J.; Bu, Z.; Rao, Z.; et al. Architecture of African swine fever virus and implications for viral assembly. Science 2019, 366, 640–644. [Google Scholar] [CrossRef]
- Jia, N.; Ou, Y.; Pejsak, Z.; Zhang, Y.; Zhang, J. Roles of African swine fever virus structural proteins in viral infection. J. Vet. Res. 2017, 61, 135–143. [Google Scholar] [CrossRef]
- Dixon, L.; Islam, M.; Nash, R.; Reis, A. African swine fever virus evasion of host defences. Virus Res. 2019, 266, 25–33. [Google Scholar] [CrossRef]
- Afonso, C.L.; Piccone, M.E.; Zaffuto, K.M.; Neilan, J.; Kutish, G.F.; Lu, Z.; Balinsky, C.A.; Gibb, T.R.; Bean, T.J.; Zsak, L.; et al. African Swine Fever Virus Multigene Family 360 and 530 Genes Affect Host Interferon Response. J. Virol. 2004, 78, 1858–1864. [Google Scholar] [CrossRef]
- Zsak, L.; Caler, E.; Lu, Z.; Kutish, G.F.; Neilan, J.G.; Rock, D.L. A Nonessential African Swine Fever Virus Gene UK Is a Significant Virulence Determinant in Domestic Swine. J. Virol. 1998, 72, 1028–1035. [Google Scholar] [CrossRef]
- De Oliveira, V.L.; Almeida, S.C.P.; Soares, H.R.; Crespo, A.; Marshall-Clarke, S.; Parkhouse, R.M.E. A novel TLR3 inhibitor encoded by African swine fever virus (ASFV). Arch. Virol. 2011, 156, 597–609. [Google Scholar] [CrossRef]
- Neilan, J.; Lu, Z.; Kutish, G.; Zsák, L.; Burrage, T.; Borca, M.; Carrillo, C.; Rock, D. A BIR Motif Containing Gene of African Swine Fever Virus, 4CL, is Nonessential for Growthin Vitroand Viral Virulence. Virology 1997, 230, 252–264. [Google Scholar] [CrossRef]
- Malogolovkin, A.; Burmakina, G.; Titov, I.; Sereda, A.; Gogin, A.; Baryshnikova, E.; Kolbasov, D. Comparative Analysis of African Swine Fever Virus Genotypes and Serogroups. Emerg. Infect. Dis. 2015, 21, 312–315. [Google Scholar] [CrossRef]
- Boshoff, C.I.; Bastos, A.D.; Gerber, L.; Vosloo, W. Genetic characterisation of African swine fever viruses from outbreaks in southern Africa (1973–1999). Vet. Microbiol. 2007, 121, 45–55. [Google Scholar] [CrossRef]
- Aslanyan, L.; Avagyan, H.; Karalyan, Z. Whole-genome-based phylogeny of African swine fever virus. Vet. World 2020, 13, 2118–2125. [Google Scholar] [CrossRef]
- Gallardo, C.; Anchuelo, R.; Pelayo, V.; Poudevigne, F.; Leon, T.; Nzoussi, J.; Bishop, R.; Pérez, C.; Soler, A.; Nieto, R.; et al. African Swine Fever Virus p72 Genotype IX in Domestic Pigs, Congo, 2009. Emerg. Infect. Dis. 2011, 17, 1556–1558. [Google Scholar] [CrossRef]
- Bisimwa, P.N.; Ongus, J.R.; Tiambo, C.K.; Machuka, E.M.; Bisimwa, E.B.; Steinaa, L.; Pelle, R. First detection of African swine fever (ASF) virus genotype X and serogroup 7 in symptomatic pigs in the Democratic Republic of Congo. Virol. J. 2020, 17, 1–11. [Google Scholar] [CrossRef]
- Lubisi, B.A.; Bastos, A.D.S.; Dwarka, R.M.; Vosloo, W. Molecular epidemiology of African swine fever in East Africa. Arch. Virol. 2005, 150, 2439–2452. [Google Scholar] [CrossRef]
- Lubisi, B.; Baratang, A.; Bastos, A.D.S.; Dwarka, R.M.; Vosloo, W. Intra-genotypic resolution of African swine fever viruses from an East African domestic pig cycle: A combined p72-CVR approach. Virus Genes 2007, 35, 729–735. [Google Scholar] [CrossRef]
- Gallardo, C.; Mwaengo, D.M.; Macharia, J.M.; Arias, M.; Taracha, E.A.; Soler, A.; Okoth, E.; Martín, E.; Kasiti, J.; Bishop, R.P. Enhanced discrimination of African swine fever virus isolates through nucleotide sequencing of the p54, p72, and pB602L (CVR) genes. Virus Genes 2008, 38, 85–95. [Google Scholar] [CrossRef]
- Rowlands, R.J.; Michaud, V.; Heath, L.; Hutchings, G.; Oura, C.; Vosloo, W.; Dwarka, R.; Onashvili, T.; Albina, E.; Dixon, L. African Swine Fever Virus Isolate, Georgia. Emerg. Infect. Dis. 2008, 14, 1870–1874. [Google Scholar] [CrossRef]
- Zhao, D.; Liu, R.; Zhang, X.; Li, F.; Wang, J.; Zhang, J.; Liu, X.; Wang, L.; Zhang, J.; Wu, X.; et al. Replication and virulence in pigs of the first African swine fever virus isolated in China. Emerg. Microbes Infect. 2019, 8, 438–447. [Google Scholar] [CrossRef]
- Gallardo, C.; Fernández-Pinero, J.; Pelayo, V.; Gazaev, I.; Markowska-Daniel, I.; Pridotkas, G.; Nieto, R.; Fernández-Pacheco, P.; Bokhan, S.; Nevolko, O.; et al. Genetic Variation among African Swine Fever Genotype II Viruses, Eastern and Central Europe. Emerg. Infect. Dis. 2014, 20, 1544–1547. [Google Scholar] [CrossRef] [PubMed]
- Eustace, M.R. On a Form of Swine Fever Occurring in British East Africa (Kenya Colony). J. Comp. Pathol. Ther. 1921, 34, 159–191. [Google Scholar] [CrossRef]
- Danzetta, M.L.; Marenzoni, M.L.; Iannetti, S.; Tizzani, P.; Calistri, P.; Feliziani, F. African Swine Fever: Lessons to Learn From Past Eradication Experiences. A Systematic Review. Front. Vet. Sci. 2020, 7, 296. [Google Scholar] [CrossRef] [PubMed]
- Penrith, M.-L.; Vosloo, W.; Jori, F.J.; Bastos, A.D. African swine fever virus eradication in Africa. Virus Res. 2013, 173, 228–246. [Google Scholar] [CrossRef]
- Sánchez-Vizcaíno, J.M.; Mur, L.; Martínez-López, B. African Swine Fever: An Epidemiological Update. Transbound. Emerg. Dis. 2012, 59, 27–35. [Google Scholar] [CrossRef]
- Sánchez-Vizcaíno, J.M.; Mur, L.; Martínez-López, B. African swine fever (ASF): Five years around Europe. Vet. Microbiol. 2013, 165, 45–50. [Google Scholar] [CrossRef]
- Jurado, C.; Mur, L.; Rolesu, S.; Laddomada, A.; Fernández-Carrión, E.; Sánchez-Vizcaíno, J.M. Why is African swine fever still present in Sardinia? Transbound. Emerg. Dis. 2017, 65, 557–566. [Google Scholar] [CrossRef]
- Laddomada, A.; Rolesu, S.; Loi, F.; Cappai, S.; Oggiano, A.; Madrau, M.P.; Sanna, M.L.; Pilo, G.; Bandino, E.; Brundu, D.; et al. Surveillance and control of African Swine Fever in free-ranging pigs in Sardinia. Transbound. Emerg. Dis. 2019, 66, 1114–1119. [Google Scholar] [CrossRef] [PubMed]
- Franzoni, G.; Giudici, S.D.; Loi, F.; Sanna, D.; Floris, M.; Fiori, M.; Sanna, M.L.; Madrau, P.; Scarpa, F.; Zinellu, S.; et al. African Swine Fever Circulation among Free-Ranging Pigs in Sardinia: Data from the Eradication Program. Vaccines 2020, 8, 549. [Google Scholar] [CrossRef] [PubMed]
- Loi, F.; Cappai, S.; Laddomada, A.; Feliziani, F.; Oggiano, A.; Franzoni, G.; Rolesu, S.; Guberti, V. Mathematical Approach to Estimating the Main Epidemiological Parameters of African Swine Fever in Wild Boar. Vaccines 2020, 8, 521. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Vizcaíno, J.; Mur, L.; Gomez-Villamandos, J.; Carrasco, L. An Update on the Epidemiology and Pathology of African Swine Fever. J. Comp. Pathol. 2015, 152, 9–21. [Google Scholar] [CrossRef]
- Costard, S.; Mur, L.; Lubroth, J.; Sanchez-Vizcaino, J.; Pfeiffer, D. Epidemiology of African swine fever virus. Virus Res. 2013, 173, 191–197. [Google Scholar] [CrossRef]
- OIE. Reports on ASF: OIE-World Organisation for Animal Health. Available online: https://www.oie.int/en/animal-health-in-the-world/information-on-aquatic-and-terrestrial-animal-diseases/african-swine-fever/reports-on-asf/ (accessed on 2 January 2021).
- Plowright, W.; Parker, J.; Peirce, M.A. African Swine Fever Virus in Ticks (Ornithodoros moubata, Murray) collected from Animal Burrows in Tanzania. Nature 1969, 221, 1071–1073. [Google Scholar] [CrossRef]
- Haresnape, J.M.; Wilkinson, P.J. A study of African swine fever virus infected ticks (Ornithodoros moubata) collected from three villages in the ASF enzootic area of Malawi following an outbreak of the disease in domestic pigs. Epidemiol. Infect. 1989, 102, 507–522. [Google Scholar] [CrossRef]
- Jori, F.; Vial, L.; Penrith, M.; Pérez-Sánchez, R.; Etter, E.; Albina, E.; Michaud, V.; Roger, F. Review of the sylvatic cycle of African swine fever in sub-Saharan Africa and the Indian ocean. Virus Res. 2013, 173, 212–227. [Google Scholar] [CrossRef]
- Mur, L.; Boadella, M.; Martinez-Lopez, B.; Gallardo, C.; Gortazar, C.; Sánchez-Vizcaíno, J.M. Monitoring of African Swine Fever in the Wild Boar Population of the Most Recent Endemic Area of Spain. Transbound. Emerg. Dis. 2012, 59, 526–531. [Google Scholar] [CrossRef] [PubMed]
- Dixon, L.K.; Stahl, K.; Jori, F.; Vial, L.; Pfeiffer, D.U. African Swine Fever Epidemiology and Control. Annu. Rev. Anim. Biosci. 2020, 8, 221–246. [Google Scholar] [CrossRef]
- Guinat, C.; Reis, A.L.; Netherton, C.L.; Goatley, L.; Pfeiffer, D.U.; Dixon, L. Dynamics of African swine fever virus shedding and excretion in domestic pigs infected by intramuscular inoculation and contact transmission. Vet. Res. 2014, 45, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, C.; Blome, S.; Malogolovkin, A.; Parilov, S.; Kolbasov, D.; Teifke, J.P.; Beer, M. Characterization of African Swine Fever Virus Caucasus Isolate in European Wild Boars. Emerg. Infect. Dis. 2011, 17, 2342–2345. [Google Scholar] [CrossRef]
- McVicar, J.W. Quantitative aspects of the transmission of African swine fever. Am. J. Vet. Res. 1984, 45, 1535–1541. [Google Scholar] [PubMed]
- Olesen, A.S.; Lohse, L.; Boklund, A.; Halasa, T.; Gallardo, C.; Pejsak, Z.; Belsham, G.J.; Rasmussen, T.B.; Bøtner, A. Transmission of African swine fever virus from infected pigs by direct contact and aerosol routes. Vet. Microbiol. 2017, 211, 92–102. [Google Scholar] [CrossRef] [PubMed]
- FAO. AU-IBAR or ILRI. Regional Strategy to Control African Swine Fever in Africa. Available online: http://www.fao.org/3/a-i6053e.pdf (accessed on 2 January 2021).
- Official Journal of the European Communities. Laying Down Specific Provisions for the Control of African Swine Fever and Amending Directive 92/119/EEC as Regards Teschen Disease and African Swine Fever. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32002L0060&from=EN (accessed on 2 January 2021).
- Boinas, F.S.; Wilson, A.J.; Hutchings, G.H.; Martins, C.; Dixon, L.J. The Persistence of African Swine Fever Virus in Field-Infected Ornithodoros erraticus during the ASF Endemic Period in Portugal. PLoS ONE 2011, 6, e20383. [Google Scholar] [CrossRef]
- Morelle, K.; Bubnicki, J.; Churski, M.; Gryz, J.; Podgórski, T.; Kuijper, D.P.J. Disease-Induced Mortality Outweighs Hunting in Causing Wild Boar Population Crash After African Swine Fever Outbreak. Front. Vet. Sci. 2020, 7, 378. [Google Scholar] [CrossRef]
- Cwynar, P.; Stojkov, J.; Wlazlak, K. African Swine Fever Status in Europe. Viruses 2019, 11, 310. [Google Scholar] [CrossRef]
- Schulz, K.; Staubach, C.; Blome, S.; Nurmoja, I.; Viltrop, A.; Conraths, F.J.; Kristian, M.; Sauter-Louis, C. How to Demonstrate Freedom from African Swine Fever in Wild Boar—Estonia as an Example. Vaccines 2020, 8, 336. [Google Scholar] [CrossRef] [PubMed]
- Alexander, F.C.M. Experiences with African Swine Fever in Haiti. Ann. N. Y. Acad. Sci. 1992, 653, 251–256. [Google Scholar] [CrossRef]
- Lyra, L.T.D.P.; Saraiva, V.; Lage, G.H.; Samarcos, M. Eradication of African swine fever from Brazil. Rev. Sci. Tech. 1986, 5, 771–787. [Google Scholar] [CrossRef]
- Moura, J.; McManus, C.; Bernal, F.; De Melo, C. An analysis of the 1978 African swine fever outbreak in Brazil and its eradication. Rev. Sci. Tech. 2010, 29, 549–563. [Google Scholar] [CrossRef]
- Blome, S.; Gabriel, C.; Beer, M. Pathogenesis of African swine fever in domestic pigs and European wild boar. Virus Res. 2013, 173, 122–130. [Google Scholar] [CrossRef]
- Wilkinson, P.; Wardley, R.; Williams, S. African swine fever virus (Malta/78) in pigs. J. Comp. Pathol. 1981, 91, 277–284. [Google Scholar] [CrossRef]
- Beltrán-Alcrudo, D.; Arias, M.; Gallardo, C.; Kramer, S.; Penrith, M.L. African Swine Fever: Detection and Diagnosis—A Manual for Veterinarians. FAO Animal Production and Health Manual. No. 19; FAO: Rome, Italy, 2017; Available online: http://www.fao.org/3/i7228e/i7228e.pdf (accessed on 2 January 2021).
- Gallardo, M.C.; Reoyo, A.D.L.T.; Fernández-Pinero, J.; Iglesias, I.; Muñoz, M.J.; Arias, M.L. African swine fever: A global view of the current challenge. Porc. Health Manag. 2015, 1, 21. [Google Scholar] [CrossRef] [PubMed]
- Nga, B.T.T.; Dao, B.T.A.; Thi, L.N.; Osaki, M.; Kawashima, K.; Song, D.; Salguero, F.J.; Le, V.P. Clinical and Pathological Study of the First Outbreak Cases of African Swine Fever in Vietnam, 2019. Front. Vet. Sci. 2020, 7, 392. [Google Scholar] [CrossRef] [PubMed]
- Villeda, C.J.; Williams, S.M.; Wilkinson, P.J.; Viñuela, E. Consumption coagulopathy associated with shock in acute African swine fever. Arch. Virol. 1993, 133, 467–475. [Google Scholar] [CrossRef]
- Villeda, C.J.; Williams, S.M.; Wilkinson, P.J.; Viñuela, E. Haemostatic abnormalities in African swine fever/A comparison of two virus strains of different virulence (Dominican Republic’78 and Malta’78). Arch. Virol. 1993, 130, 71–83. [Google Scholar] [CrossRef]
- Gómez-Villamandos, J.; Bautista, M.; Carrasco, L.; De Lara, F.C.-M.; Hervás, J.; Wilkinson, P.; Sierra, M. Thrombocytopenia associated with apoptotic megakaryocytes in a viral haemorrhagic syndrome induced by a moderately virulent strain of African swine fever virus. J. Comp. Pathol. 1998, 118, 1–13. [Google Scholar] [CrossRef]
- Sánchez-Cordón, P.J.; Montoya, M.; Reis, A.L.; Dixon, L.K. African swine fever: A re-emerging viral disease threatening the global pig industry. Vet. J. 2018, 233, 41–48. [Google Scholar] [CrossRef]
- Salguero, F.; Ruiz-Villamor, E.; Bautista, M.; Sánchez-Cordón, P.; Carrasco, L.; Gómez-Villamandos, J. Changes in macrophages in spleen and lymph nodes during acute African swine fever: Expression of cytokines. Vet. Immunol. Immunopathol. 2002, 90, 11–22. [Google Scholar] [CrossRef]
- Carrasco, L.; Núñez, A.; Salguero, F.; Segundo, F.D.S.; Sánchez-Cordón, P.; Gómez-Villamandos, J.; Sierra, M. African Swine Fever: Expression of Interleukin-1 alpha and Tumour Necrosis Factor-alpha by Pulmonary Intravascular Macrophages. J. Comp. Pathol. 2002, 126, 194–201. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Villamandos, J.; Bautista, M.; Sánchez-Cordón, P.; Carrasco, L. Pathology of African swine fever: The role of monocyte-macrophage. Virus Res. 2013, 173, 140–149. [Google Scholar] [CrossRef]
- Carrascosa, A.L.; Bustos, M.J.; De Leon, P. Methods for Growing and Titrating African Swine Fever Virus: Field and Laboratory Samples. Curr. Protoc. Cell Biol. 2011, 53. [Google Scholar] [CrossRef]
- Enjuanes, L.; Carrascosa, A.L.; Viñuela, E. Isolation and Properties of the DNA of African Swine Fever (ASF) Virus. J. Gen. Virol. 1976, 32, 479–492. [Google Scholar] [CrossRef] [PubMed]
- Parker, J.; Plowright, W. Plaque Formation by African Swine Fever Virus. Nat. Cell Biol. 1968, 219, 524–525. [Google Scholar] [CrossRef] [PubMed]
- Santurde, G.; Gonzalvo, F.R.; Carnero, M.E.; Tabarés, E. Genetic stability of African swine fever virus grown in monkey kidney cells. Brief report. Arch. Virol. 1988, 98, 117–122. [Google Scholar] [CrossRef]
- Hurtado, C.; Bustos, M.J.; Carrascosa, A.L. The use of COS-1 cells for studies of field and laboratory African swine fever virus samples. J. Virol. Methods 2010, 164, 131–134. [Google Scholar] [CrossRef]
- Sánchez, E.G.; Riera, E.; Nogal, M.; Gallardo, C.; Fernández, P.; Bello-Morales, R.; López-Guerrero, J.A.; Chitko-McKown, C.G.; Richt, J.A.; Revilla, Y. Phenotyping and susceptibility of established porcine cells lines to African Swine Fever Virus infection and viral production. Sci. Rep. 2017, 7, 10369. [Google Scholar] [CrossRef]
- Malmquist, W.A.; Hay, D. Hemadsorption and cytopathic effect produced by African Swine Fever virus in swine bone marrow and buffy coat cultures. Am. J. Vet. Res. 1960, 21, 104–108. [Google Scholar]
- Gallardo, C.; Soler, A.; Rodze, I.; Nieto, R.; Cano-Gómez, C.; Fernandez-Pinero, J.; Arias, M. Attenuated and non-haemadsorbing (non-HAD) genotypeIIAfrican swine fever virus (ASFV) isolated in Europe, Latvia 2017. Transbound. Emerg. Dis. 2019, 66, 1399–1404. [Google Scholar] [CrossRef] [PubMed]
- Leitão, A.; Cartaxeiro, C.; Coelho, R.; Cruz, B.; Parkhouse, R.M.E.; Portugal, F.C.; Vigário, J.D.; Martins, C.L.V. The non-haemadsorbing African swine fever virus isolate ASFV/NH/P68 provides a model for defining the protective anti-virus immune response. J. Gen. Virol. 2001, 82, 513–523. [Google Scholar] [CrossRef]
- Gonzague, M.; Roger, F.; Bastos, A.; Burger, C.; Randriamparany, T.; Smondack, S.; Cruciere, C. Isolation of a non-haemadsorbing, non-cytopathic strain of African swine fever virus in Madagascar. Epidemiol. Infect. 2001, 126, 453–459. [Google Scholar] [CrossRef][Green Version]
- OIE. Manual of Diagnostic Tests and Vaccines for Terrestrial Animals, 2012. Chapter 2.8.1. Available online: http://www.oie.int/ (accessed on 2 January 2021).
- Aguero, M.; Fernandez, J.; Romero, L.; Mascaraque, C.S.; Arias, M.; Sanchez-Vizcaino, J.M. Highly Sensitive PCR Assay for Routine Diagnosis of African Swine Fever Virus in Clinical Samples. J. Clin. Microbiol. 2003, 41, 4431–4434. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Atim, S.A.; Shao, L.; Ayebazibwe, C.; Sun, Y.; Liu, Y.; Ji, S.; Meng, X.-Y.; Li, S.; Li, Y.; et al. Development of an updated PCR assay for detection of African swine fever virus. Arch. Virol. 2017, 162, 191–199. [Google Scholar] [CrossRef]
- Basto, A.P.; Portugal, R.S.; Nix, R.J.; Cartaxeiro, C.; Boinas, F.; Dixon, L.K.; Leitao, A.; Martins, C. Development of a nested PCR and its internal control for the detection of African swine fever virus (ASFV) in Ornithodoros erraticus. Arch. Virol. 2005, 151, 819–826. [Google Scholar] [CrossRef]
- Hu, L.; Lin, X.; Yang, Z.; Yao, X.; Li, G.; Peng, S.; Wang, Y. A multiplex PCR for simultaneous detection of classical swine fever virus, African swine fever virus, highly pathogenic porcine reproductive and respiratory syndrome virus, porcine reproductive and respiratory syndrome virus and pseudorabies in swines. Pol. J. Vet. Sci. 2015, 18, 715–723. [Google Scholar] [CrossRef]
- Erickson, A.; Fisher, M.; Furukawa-Stoffer, T.; Ambagala, A.; Hodko, D.; Pasick, J.; King, D.P.; Nfon, C.; Polo, R.O.; Lung, O. A multiplex reverse transcription PCR and automated electronic microarray assay for detection and differentiation of seven viruses affecting swine. Transbound. Emerg. Dis. 2017, 65, e272–e283. [Google Scholar] [CrossRef] [PubMed]
- Xiao, L.; Wang, Y.; Kang, R.; Wu, X.; Lin, H.; Ye, Y.; Yu, J.; Ye, J.; Xie, J.; Cao, Y.; et al. Development and application of a novel Bio–Plex suspension array system for high–throughput multiplexed nucleic acid detection of seven respiratory and reproductive pathogens in swine. J. Virol. Methods 2018, 261, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Belák, S.; Thorén, P. Molecular diagnosis of animal diseases: Some experiences over the past decade. Expert Rev. Mol. Diagn. 2001, 1, 434–443. [Google Scholar] [CrossRef] [PubMed]
- King, D.P.; Reid, S.M.; Hutchings, G.H.; Grierson, S.S.; Wilkinson, P.J.; Dixon, L.K.; Bastos, A.D.; Drew, T.W. Development of a TaqMan® PCR assay with internal amplification control for the detection of African swine fever virus. J. Virol. Methods 2003, 107, 53–61. [Google Scholar] [CrossRef]
- Tignon, M.; Gallardo, C.; Iscaro, C.; Hutet, E.; Van der Stede, Y.; Kolbasov, D.; De Mia, G.M.; Le Potier, M.-F.; Bishop, R.P.; Arias, M.; et al. Development and inter-laboratory validation study of an improved new real-time PCR assay with internal control for detection and laboratory diagnosis of African swine fever virus. J. Virol. Methods 2011, 178, 161–170. [Google Scholar] [CrossRef]
- Fernández-Pinero, J.; Gallardo, C.; Elizalde, M.; Robles, A.; Gómez, C.; Bishop, R.; Heath, L.E.; Couacy-Hymann, E.; Fasina, F.O.; Pelayo, V.; et al. Molecular Diagnosis of African Swine Fever by a New Real-Time PCR Using Universal Probe Library. Transbound. Emerg. Dis. 2013, 60, 48–58. [Google Scholar] [CrossRef]
- Wang, A.; Jia, R.; Liu, Y.; Zhou, J.; Qi, Y.; Chen, Y.; Liu, D.; Zhao, J.; Shi, H.; Zhang, J.; et al. Development of a novel quantitative real-time PCR assay with lyophilized powder reagent to detect African swine fever virus in blood samples of domestic pigs in China. Transbound. Emerg. Dis. 2019, 67, 284–297. [Google Scholar] [CrossRef]
- D’Agata, R.; Spoto, G. Artificial DNA and surface plasmon resonance. Artif. DNA PNA XNA 2012, 3, 45–52. [Google Scholar] [CrossRef]
- Biagetti, M.; Cuccioloni, M.; Bonfili, L.; Cecarini, V.; Sebastiani, C.; Curcio, L.; Giammarioli, M.; De Mia, G.M.; Eleuteri, A.M.; Angeletti, M. Chimeric DNA/LNA-based biosensor for the rapid detection of African swine fever virus. Talanta 2018, 184, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Luo, Y.; Accensi, F.; Ganges, L.; Rodríguez, F.; Shan, H.; Ståhl, K.; Qiu, H.-J.; Belák, S. Pre-Clinical Evaluation of a Real-Time PCR Assay on a Portable Instrument as a Possible Field Diagnostic Tool: Experiences from the Testing of Clinical Samples for African and Classical Swine Fever Viruses. Transbound. Emerg. Dis. 2017, 64, e31–e35. [Google Scholar] [CrossRef] [PubMed]
- Gill, P.; Ghaemi, A. Nucleic Acid Isothermal Amplification Technologies—A Review. Nucleosides Nucleotides Nucleic Acids 2008, 27, 224–243. [Google Scholar] [CrossRef]
- Piepenburg, O.; Williams, C.H.; Stemple, D.L.; Armes, N.A. DNA Detection Using Recombination Proteins. PLoS Biol. 2006, 4, e204. [Google Scholar] [CrossRef] [PubMed]
- Euler, M.; Wang, Y.; Nentwich, O.; Piepenburg, O.; Hufert, F.T.; Weidmann, M. Recombinase polymerase amplification assay for rapid detection of Rift Valley fever virus. J. Clin. Virol. 2012, 54, 308–312. [Google Scholar] [CrossRef]
- Wang, J.; Wang, J.; Geng, Y.; Yuan, W. A recombinase polymerase amplification-based assay for rapid detection of African swine fever virus. Can. J. Vet. Res. 2017, 81, 308–312. [Google Scholar]
- Fan, X.; Li, L.; Zhao, Y.; Liu, Y.; Liu, C.; Wang, Q.; Dong, Y.; Wang, S.; Chi, T.; Song, F.; et al. Clinical Validation of Two Recombinase-Based Isothermal Amplification Assays (RPA/RAA) for the Rapid Detection of African Swine Fever Virus. Front. Microbiol. 2020, 11, 1696. [Google Scholar] [CrossRef] [PubMed]
- Miao, F.; Zhang, J.; Li, N.; Chen, T.; Wang, L.; Zhang, F.; Mi, L.; Zhang, J.; Wang, S.; Wang, Y.; et al. Rapid and Sensitive Recombinase Polymerase Amplification Combined with Lateral Flow Strip for Detecting African Swine Fever Virus. Front. Microbiol. 2019, 10, 1004. [Google Scholar] [CrossRef] [PubMed]
- Bai, J.; Lin, H.; Li, H.; Zhou, Y.; Liu, J.; Zhong, G.; Wu, L.; Jiang, W.; Du, H.; Yang, J.; et al. Cas12a-Based On-Site and Rapid Nucleic Acid Detection of African Swine Fever. Front. Microbiol. 2019, 10, 2830. [Google Scholar] [CrossRef]
- Notomi, T.; Okayama, H.; Masubuchai, H.; Yonekawa, T.; Watanabe, K.; Amino, N.; Hase, T. Loop-mediated isothermal amplification of DNA. Nucleic Acids Res. 2000, 28, E63. [Google Scholar] [CrossRef] [PubMed]
- James, H.E.; Ebert, K.; Mcgonigle, R.; Reid, S.M.; Boonham, N.; Tomlinson, J.A.; Hutchings, G.H.; Denyer, M.; Oura, C.A.; Dukes, J.P.; et al. Detection of African swine fever virus by loop-mediated isothermal amplification. J. Virol. Methods 2010, 164, 68–74. [Google Scholar] [CrossRef]
- Tran, D.H.; Tran, H.T.; Le, U.P.; Vu, X.D.; Trinh, T.B.N.; Do, H.D.K.; Than, V.T.; Bui, L.M.; Van Vu, V.; Nguyen, T.L.; et al. Direct colorimetric LAMP assay for rapid detection of African swine fever virus: A validation study during an outbreak in Vietnam. Transbound. Emerg. Dis. 2020. [Google Scholar] [CrossRef]
- Yu, L.-S.; Chou, S.-Y.; Wu, H.-Y.; Chen, Y.-C.; Chen, Y.-H. Rapid and semi-quantitative colorimetric loop-mediated isothermal amplification detection of ASFV via HSV color model transformation. J. Microbiol. Immunol. Infect. 2020. [Google Scholar] [CrossRef]
- Zhu, Y.-S.; Shao, N.; Chen, J.-W.; Qi, W.-B.; Li, Y.; Liu, P.; Chen, Y.-J.; Bian, S.-Y.; Zhang, Y.; Tao, S.-C. Multiplex and visual detection of African Swine Fever Virus (ASFV) based on Hive-Chip and direct loop-mediated isothermal amplification. Anal. Chim. Acta 2020, 1140, 30–40. [Google Scholar] [CrossRef] [PubMed]
- Kuboki, N.; Inoue, N.; Sakurai, T.; Di Cello, F.; Grab, D.J.; Suzuki, H.; Sugimoto, C.; Igarashi, I. Loop-Mediated Isothermal Amplification for Detection of African Trypanosomes. J. Clin. Microbiol. 2003, 41, 5517–5524. [Google Scholar] [CrossRef]
- Xu, G.; Hu, L.; Zhong, H.; Wang, H.; Yusa, S.-I.; Weiss, T.C.; Romaniuk, P.J.; Pickerill, S.; You, Q. Cross Priming Amplification: Mechanism and Optimization for Isothermal DNA Amplification. Sci. Rep. 2012, 2, 246. [Google Scholar] [CrossRef]
- Feng, T.; Li, S.; Wang, S.; Pan, J. Cross priming amplification with nucleic acid test strip analysis of mutton in meat mixtures. Food Chem. 2018, 245, 641–645. [Google Scholar] [CrossRef] [PubMed]
- Zheng, F.; Li, S.; Wang, S.; Feng, T.; Jiang, Z.; Pan, J. Cross-priming isothermal amplification combined with nucleic acid test strips for detection of meat species. Anal. Biochem. 2020, 597, 113672. [Google Scholar] [CrossRef]
- Frączyk, M.; Woźniakowski, G.; Kowalczyk, A.; Niemczuk, K.; Pejsak, Z. Development of cross-priming amplification for direct detection of the African Swine Fever Virus, in pig and wild boar blood and sera samples. Lett. Appl. Microbiol. 2016, 62, 386–391. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Meng, X.-Y.; Zhang, H.; Luo, Y.; Sun, Y.; Li, Y.; Abid, M.; Qiu, H.-J. Cross-priming amplification combined with immunochromatographic strip for rapid on-site detection of African swine fever virus. Sens. Actuators B Chem. 2018, 274, 304–309. [Google Scholar] [CrossRef]
- Oura, C.; Edwards, L.; Batten, C. Virological diagnosis of African swine fever—Comparative study of available tests. Virus Res. 2013, 173, 150–158. [Google Scholar] [CrossRef]
- Sanz, A.; García-Barreno, B.; Nogal, M.L.; Viñuela, E.; Enjuanes, L. Monoclonal antibodies specific for African swine fever virus proteins. J. Virol. 1985, 54, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Wardley, R.C.; Abu Elzein, E.M.E.; Crowther, J.R.; Wilkinson, P.J. A solid-phase enzyme linked immunosorbent assay for the detection of African swine fever virus antigen and antibody. J. Hyg. 1979, 83, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Vidal, M.I.; Stiene, M.; Henkel, J.; Bilitewski, U.; Costa, J.V.; Oliva, A.G. A solid-phase enzyme linked immunosorbent assay using monoclonal antibodies, for the detection of African swine fever virus antigens and antibodies. J. Virol. Methods 1997, 66, 211–218. [Google Scholar] [CrossRef]
- Hutchings, G.H.; Ferris, N.P. Indirect sandwich ELISA for antigen detection of African swine fever virus: Comparison of polyclonal and monoclonal antibodies. J. Virol. Methods 2006, 131, 213–217. [Google Scholar] [CrossRef] [PubMed]
- Sastre, P.; Gallardo, C.; Monedero, A.; Ruiz, T.; Arias, M.; Sanz, A.; Rueda, P. Development of a novel lateral flow assay for detection of African swine fever in blood. BMC Vet. Res. 2016, 12, 206. [Google Scholar] [CrossRef] [PubMed]
- Neilan, J.; Zsak, L.; Lu, Z.; Burrage, T.; Kutish, G.; Rock, D. Neutralizing antibodies to African swine fever virus proteins p30, p54, and p72 are not sufficient for antibody-mediated protection. Virology 2004, 319, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Pan, I.C.; De Boer, C.J.; Hess, W.R. African swine fever: Application of immunoelectroosmophoresis for the detection of antibody. Can. J. Comp. Med. Rev. Can. Med. Comp. 1972, 36, 309–316. [Google Scholar]
- Pastor, M.; Escribano, J. Evaluation of sensitivity of different antigen and DNA-hybridization methods in African swine fever virus detection. J. Virol. Methods 1990, 28, 67–77. [Google Scholar] [CrossRef]
- Pan, I.C.; Huang, T.S.; Hess, W.R. New method of antibody detection by indirect immunoperoxidase plaque staining for serodiagnosis of African swine fever. J. Clin. Microbiol. 1982, 16, 650–655. [Google Scholar] [CrossRef]
- Tabarés, E.; Fernández, M.; Salvador-Temprano, E.; Carnero, M.E.; Sanchez-Botija, C. A reliable enzyme linked immunosorbent assay for African swine fever using the major structural protein as antigenic reagent. Arch. Virol. 1981, 70, 297–300. [Google Scholar] [CrossRef]
- Pastor, M.J.; Arias, M.; Escribano, J.M. Comparison of two antigens for use in an enzyme-linked immunosorbent assay to detect African swine fever antibody. Am. J. Vet. Res. 1990, 51, 1540–1543. [Google Scholar] [PubMed]
- Kollnberger, S.D.; Gutierrez-Castañeda, B.; Foster-Cuevas, M.; Corteyn, A.; Parkhouse, R.M.E. Identification of the principal se-rological immunodeterminants of African swine fever virus by screening a virus cDNA library with antibody. J. Gen. Virol. 2002, 83 Pt 6, 1331. [Google Scholar] [CrossRef]
- Oviedo, J.; Rodriguez, F.; Gómez-Puertas, P.; Brun, A.; Gómez, N.; Alonso, C.; Escribano, J. High level expression of the major antigenic African swine fever virus proteins p54 and p30 in baculovirus and their potential use as diagnostic reagents. J. Virol. Methods 1997, 64, 27–35. [Google Scholar] [CrossRef]
- Perez-Filgueira, D.M.; Gonzalez-Camacho, F.; Gallardo, C.; Resino-Talavan, P.; Blanco, E.; Gomez-Casado, E.; Alonso, C.; Escribano, J.M. Optimization and Validation of Recombinant Serological Tests for African Swine Fever Diagnosis Based on Detection of the p30 Protein Produced in Trichoplusia ni Larvae. J. Clin. Microbiol. 2006, 44, 3114–3121. [Google Scholar] [CrossRef]
- Gallardo, C.; Blanco, E.; Rodriguez, J.M.; Carrascosa, A.L.; Sánchez-Vizcaíno, J.M. Antigenic Properties and Diagnostic Potential of African Swine Fever Virus Protein pp62 Expressed in Insect Cells. J. Clin. Microbiol. 2006, 44, 950–956. [Google Scholar] [CrossRef]
- Gallardo, C.; Reis, A.L.; Kalema-Zikusoka, G.; Malta, J.; Soler, A.; Blanco, E.; Parkhouse, R.M.E.; Leitão, A. Recombinant Antigen Targets for Serodiagnosis of African Swine Fever. Clin. Vaccine Immunol. 2009, 16, 1012–1020. [Google Scholar] [CrossRef]
- Gallardo, C.; Nieto, R.; Soler, A.; Pelayo, V.; Fernandezpinero, J.; Markowska-Daniel, I.; Pridotkas, G.; Nurmoja, I.; Granta, R.; Simon, A.O.; et al. Assessment of African Swine Fever Diagnostic Techniques as a Response to the Epidemic Outbreaks in Eastern European Union Countries: How To Improve Surveillance and Control Programs. J. Clin. Microbiol. 2015, 53, 2555–2565. [Google Scholar] [CrossRef]
- Cubillos, C.; Gómez-Sebastian, S.; Moreno, N.; Nuñez, M.C.; Mulumba-Mfumu, L.K.; Quembo, C.J.; Heath, L.; Etter, E.M.; Jori, F.; Escribano, J.M.; et al. African swine fever virus serodiagnosis: A general review with a focus on the analyses of African serum samples. Virus Res. 2013, 173, 159–167. [Google Scholar] [CrossRef]
- Pastor, M.J.; Laviada, M.D.; Sanchez-Vizcaino, J.M.; Escribano, J.M. Detection of African swine fever virus antibodies by im-munoblotting assay. Can. J. Vet. Res. 1989, 53, 105–107. [Google Scholar] [PubMed]
- Pastor, M.J.; Arias, M.; Alcaraz, C.; De Diego, M.; Escribano, J.M. A Sensitive Dot Immunobinding Assay for Serodiagnosis of African Swine Fever Virus with Application in Field Conditions. J. Vet. Diagn. Investig. 1992, 4, 254–257. [Google Scholar] [CrossRef] [PubMed]
- Gallardo, C.; Soler, A.; Nieto, R.; Sánchez, M.A.; Martins, C.; Pelayo, V.; Carrascosa, A.; Revilla, Y.; Simon, A.; Briones, V.; et al. Experimental Transmission of African Swine Fever (ASF) Low Virulent Isolate NH/P68 by Surviving Pigs. Transbound. Emerg. Dis. 2015, 62, 612–622. [Google Scholar] [CrossRef] [PubMed]
- Alcaraz, C.; Rodriguez, F.; Oviedo, J.; Eiras, A.; De Diego, M.; Alonso, C.; Escribano, J. Highly specific confirmatory Western blot test for African swine fever virus antibody detection using the recombinant virus protein p54. J. Virol. Methods 1995, 52, 111–119. [Google Scholar] [CrossRef]
- Kazakova, A.S.; Imatdinov, I.R.; Dubrovskaya, O.A.; Sidlik, M.V.; Balyshev, V.M.; Krasochko, P.A.; Sereda, A.D. Recombinant Protein p30 for Serological Diagnosis of African Swine Fever by Immunoblotting Assay. Transbound. Emerg. Dis. 2016, 64, 1479–1492. [Google Scholar] [CrossRef]
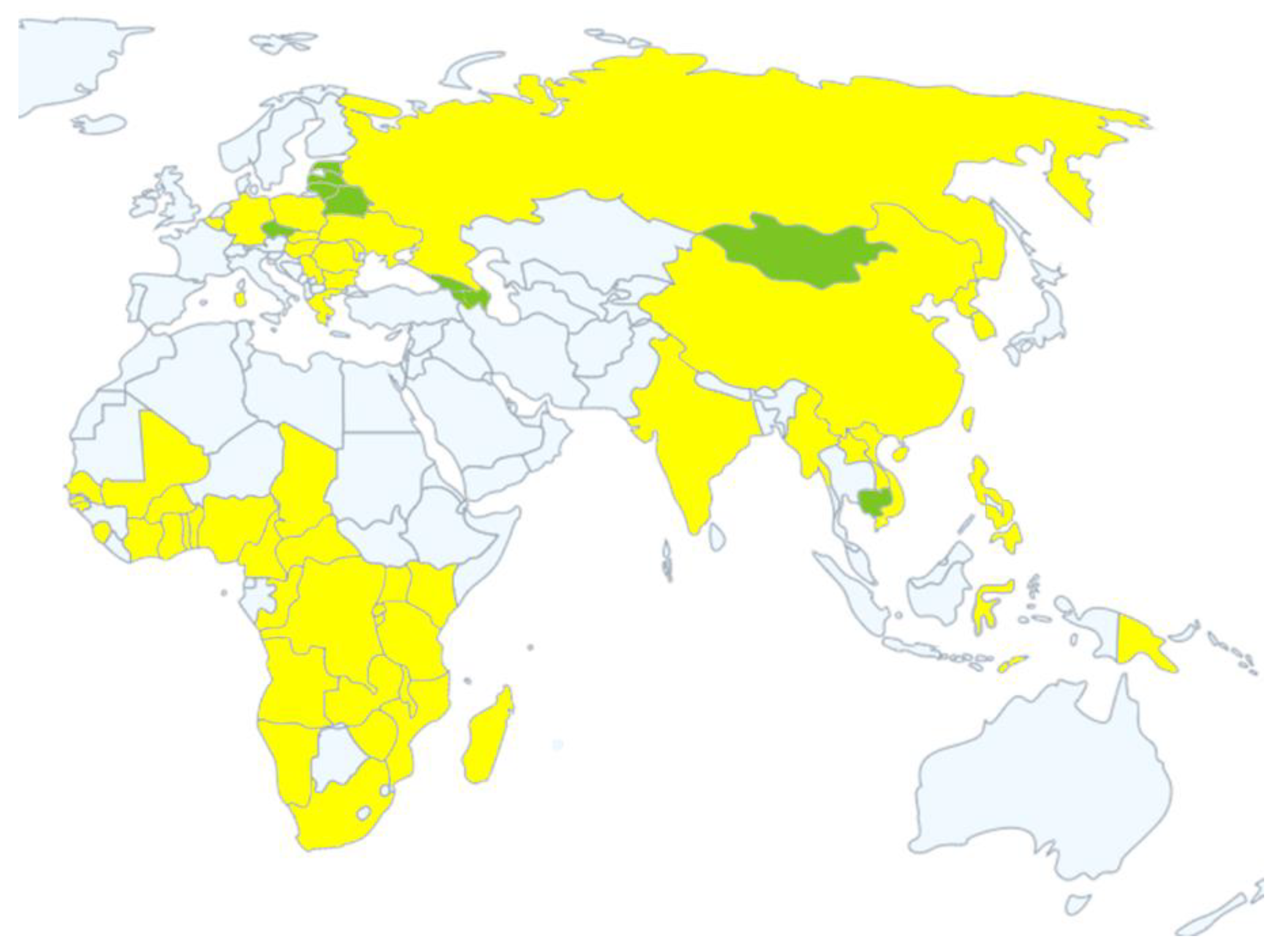
| Country | Year or Date Reported | Status |
|---|---|---|
| Georgia | 2007–2008 | Resolved |
| Armenia | 2007–2008, 2010–2011 | Resolved |
| Russian Federation | 2007 | Continuing |
| Azerbaijan | 2008 | Resolved |
| Latvia | 2014–2019 | Resolved |
| Estonia | 2014–2019 | Resolved |
| Ukraine | 2012, 2014 | Continuing |
| Belarus | 2013 | Resolved |
| Lithuania | 2014–2019 | Resolved |
| Poland | 2014 | Continuing |
| Moldova | 2016 | Continuing |
| Hungary | 2018 | Continuing |
| Czech Republic | 2017–2018 | Resolved |
| Romania | 2017 | Continuing |
| Belgium | 2018 | Continuing |
| Bulgaria | 2018 | Continuing |
| Slovakia | July, 2019 | Continuing |
| Serbia | 2019 | Continuing |
| Greece | February, 2020 | Continuing |
| Germany | September, 2020 | Continuing |
| Sardinia | 2007–2020 | Continuing |
| China | 3 August 2018 | Continuing |
| Democratic People’s Republic of Korea | 23 May 2019 | Continuing |
| Lao People’s Democratic Republic | 20 June 2019 | Continuing |
| Myanmar | 1 August 2019 | Continuing |
| The Philippines | 25 June 2019 | Continuing |
| South Korea | 17 September 2019 | Continuing |
| Vietnam | 19 February 2019 | Continuing |
| Mongolia | 15 January 2019 | Resolved |
| Cambodia | 2 April 2019 | Resolved |
| Timor-Leste | 9 September 2019 | Continuing |
| Indonesia | September, 2019 | Continuing |
| Papua New Guinea | March, 2020 | Continuing |
| India | May, 2020 | Continuing |
| Detection Method | Target Gene | Internal Control | Display of Results | Sample Type | Limits of the Test | Validation | Reference | |
|---|---|---|---|---|---|---|---|---|
| No. of Diverse ASFV Isolates | No. of Clinical Samples | |||||||
| PCR | VP73 | None | Agarose gel electrophoresis | Tissue, blood, serum | 0.12 HADU50 | 22 | 70, including 18 serum, 4 blood | [88], OIE |
| PCR | VP72 | None | Agarose gel electrophoresis | Blood | 60 copes | 14 (4 genotypes) | 62 blood | [89] |
| Nested PCR | VP72 | Foreign DNA | Agarose gel electrophoresis | Ticks supernatant | 0.49 CPE50 | 20 (7 genotypes) | 60 ticks | [90] |
| Multiplex PCR | VP72 | None | Agarose gel electrophoresis | Tissue | 1.50 × 103 copes | Gene fragments | 10 pigs, 76 wild boars | [91] |
| Multiplex PCR | VP72 | None | Fluorescent intensity (FI) data | blood | 10 copes/μL | 4 (2 genotypes) | 24 | [92] |
| Bio-Plex multiplex PCR | VP72 | None | Bio-Plex | Tissue, blood | 103 copes | Gene fragments | 137 | [93] |
| Detection Method | Target Gene | Internal Control | Probes | Sample Type | Ct Value | Limits of the Test | Validation | Reference | |
|---|---|---|---|---|---|---|---|---|---|
| No. of Diverse ASFV Isolates | No. of Clinical Samples | ||||||||
| Real-time PCR | VP72 | Artificial template (mimic) | TaqMan | Tissue, ticks, cell lines | 21.79–30.59 | 10–100 | 25 (9 genotypes) | None | [95], OIE |
| Real-time PCR | VP72 | β-actin | TaqMan | Tissue, blood, serum, ticks, | 15–30 | 5.7–57 | 44 (7 genotypes) | 281 | [96] |
| Real-time PCR | VP72 | β-actin | UPL | Tissue, blood, serum, ticks, cell lines | 18.79–35.56 | below 18 | 46 (19 genotypes) | 260 | [97], OIE |
| Real-time PCR (LPR) | VP72 | None | TaqMan | Blood | <32 | 100 (plasmid) | Gene fragments | 218 (in China) | [98] |
| Real-time PCR (LNA) | VP72 | None | ssDNA/LNA | Tissue, blood | None | 178 | 14 (2 genotypes) | 20 | [100] |
| Real-time (T-COR4) | VP72 | None | None | Tissue, blood, serum | 34.3 | 10,000 | Gene fragments | 41 | [101] |
| Detection Method | Target Gene | Reaction Time | Reaction Temperature | Display of Results | Probe | Limits of the Test | Validation | Reference | |
|---|---|---|---|---|---|---|---|---|---|
| Compared withrPCR (OIE) | No. of Samples | ||||||||
| RPA | VP72 | 20 min | 39 °C | Genie III scanner device | Exo | 100 copes | Consistent | 40 | [105] |
| RPA | VP72 | 10 min | 38 °C | LFD | FITC | 150 copes | Consistent | 145 | [107] |
| RAA-CAS12a | VP72 | 1 h | 37 °C | LFD | CAS12a | 1 × 10−9 M | Decreased sensitivity | None | [108] |
| LAMP | Topoisomerase II gene, VP73 | 25 min | 64–66 °C | LFD | None | 330 copes | Consistent | 42 (from 7 pigs) | [110] |
| LAMP | Topoisomerase II gene | 30 min | 60 °C | Direct colorimetric | None | 10 HAD50/mL | Consistent | 97 (from Vietnam) | [111] |
| CPA | VP72 | 45 min | 56.2 °C | SYBR Green I | LightCycler 480 | 7.2 copes (plasmid) | Consistent | 10 pigs, 76 wild boars | [118] |
| CPA | VP72 | 60 min | 59 °C | LFD | Biotin | 200 copes | Consistent | 65 | [119] |
| Detection Method | Time | Sensitivity | Specificity | Sample Type | Cost | Comments |
|---|---|---|---|---|---|---|
| PCR | 5–6 h | XXX | XX | Tissues, blood, ticks or cell cultures | $$ | Most common method Susceptible to contamination Detects live or dead virus |
| HAD | 7–21 days | XX | XXX | Porcine macrophage cells | $$$$ | Gold standard, only used in a few reference laboratories |
| FAT | 75 min | XXX (for early detection) | XXX | Cryostat sections, impression smears, cultured cells | $$$ | Recommended when PCR is unavailable or lack of experience Needs a fluorescent microscope Lack of sensitivity after the first week post-infection |
| ELISA | 3 h | X (for early detection) | X | Serum, macerates | $ | Screening test In-house and commercial kits available |
| IBT | 3 h | X | X | Serum | $$$$ | Confirmatory test No commercial kits |
| IFA | 4 h | XXX | XX | Tissue exudates, serum or plasma | $$$ | Confirmatory test No commercially available reagents Needs a fluorescent microscope |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qiu, Z.; Li, Z.; Yan, Q.; Li, Y.; Xiong, W.; Wu, K.; Li, X.; Fan, S.; Zhao, M.; Ding, H.; et al. Development of Diagnostic Tests Provides Technical Support for the Control of African Swine Fever. Vaccines 2021, 9, 343. https://doi.org/10.3390/vaccines9040343
Qiu Z, Li Z, Yan Q, Li Y, Xiong W, Wu K, Li X, Fan S, Zhao M, Ding H, et al. Development of Diagnostic Tests Provides Technical Support for the Control of African Swine Fever. Vaccines. 2021; 9(4):343. https://doi.org/10.3390/vaccines9040343
Chicago/Turabian StyleQiu, Zilong, Zhaoyao Li, Quanhui Yan, Yuwan Li, Wenjie Xiong, Keke Wu, Xiaowen Li, Shuangqi Fan, Mingqiu Zhao, Hongxing Ding, and et al. 2021. "Development of Diagnostic Tests Provides Technical Support for the Control of African Swine Fever" Vaccines 9, no. 4: 343. https://doi.org/10.3390/vaccines9040343
APA StyleQiu, Z., Li, Z., Yan, Q., Li, Y., Xiong, W., Wu, K., Li, X., Fan, S., Zhao, M., Ding, H., & Chen, J. (2021). Development of Diagnostic Tests Provides Technical Support for the Control of African Swine Fever. Vaccines, 9(4), 343. https://doi.org/10.3390/vaccines9040343






