Impact of Biological Therapies on the Immune Response after Pneumococcal Vaccination in Patients with Autoimmune Inflammatory Diseases
Abstract
1. Introduction
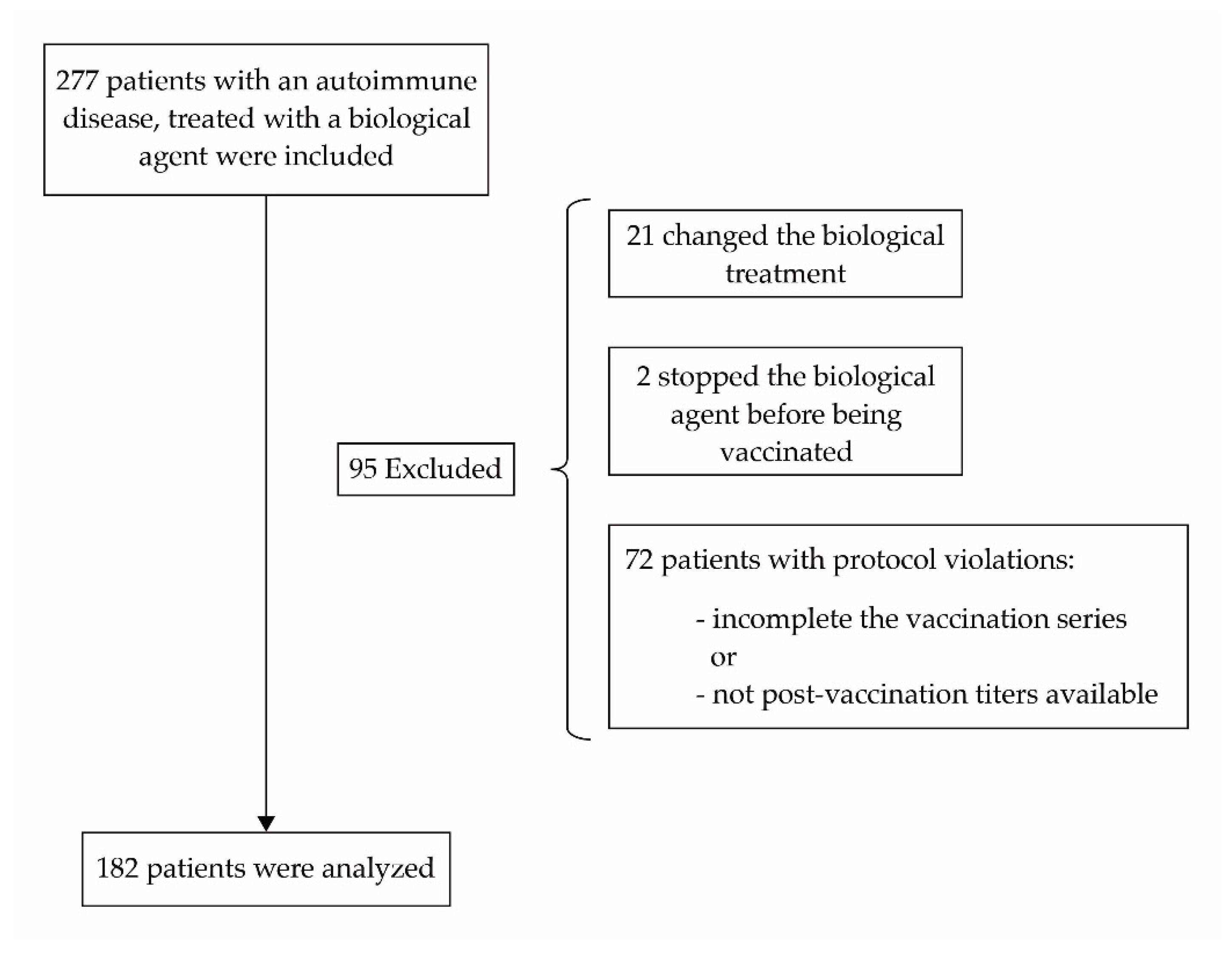
2. Materials and Methods
2.1. Participants
2.2. Ethical Approval Code
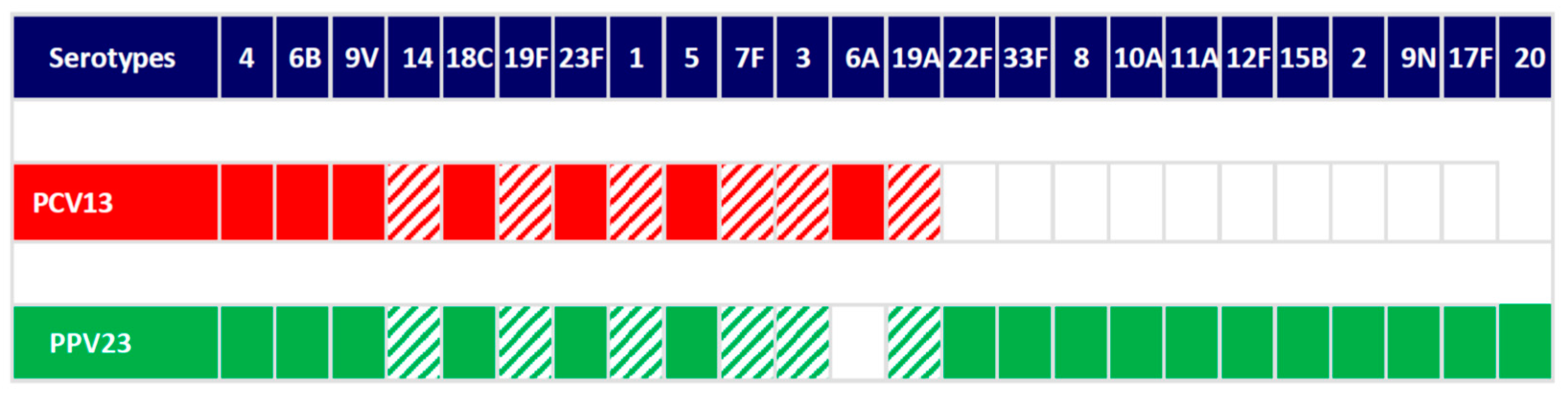
2.3. Vaccination, Immune Response and Microbiology Data
2.4. Statistics
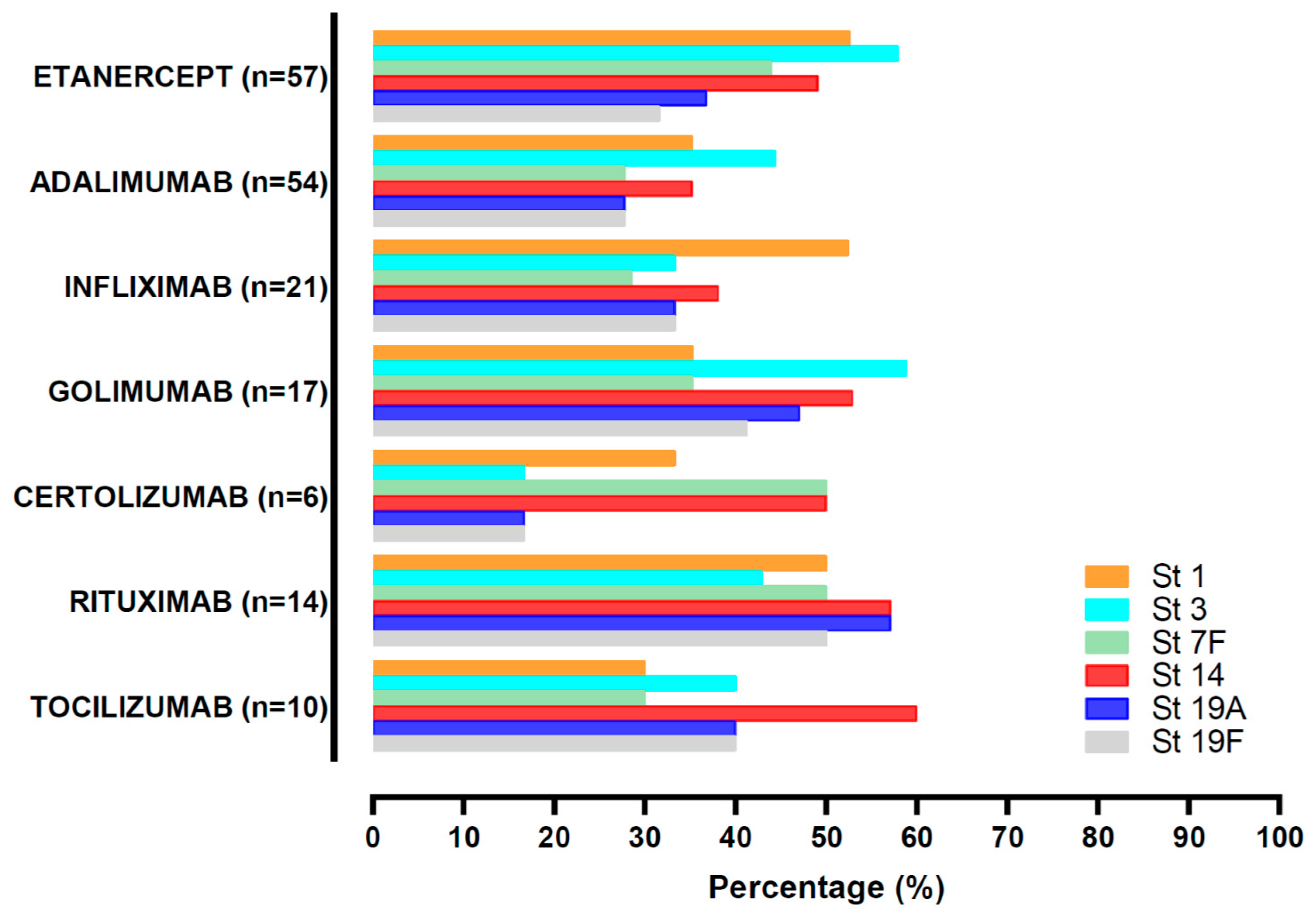
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wotton, C.J.; Goldacre, M.J. Risk of invasive pneumococcal disease in people admitted to hospital with selected immune-mediated diseases: Record linkage cohort analyses. J. Epidemiol. Community Health 2012, 66, 1177–1181. [Google Scholar] [CrossRef] [PubMed]
- Backhaus, E.; Berg, S.; Andersson, R.; Ockborn, G.; Malmström, P.; Dahl, M.; Nasic, S.; Trollfors, B. Epidemiology of invasive pneumococcal infections: Manifestations, incidence and case fatality rate correlated to age, gender and risk factors. BMC Infect. Dis. 2016, 16, 367. [Google Scholar] [CrossRef] [PubMed]
- Shea, K.M.; Edelsberg, J.; Weycker, D.; Farkouh, R.A.; Strutton, D.R.; Pelton, S.I. Rates of pneumococcal disease in adults with chronic medical conditions. Open Forum. Infec. Dis. 2014, 1, ofu024. [Google Scholar] [CrossRef]
- Fernandez-Martinez, S.; Cortes, X.; Borrás-Blasco, J.; Gracia-Pérez, A.; Casterá, M.E. Effectiveness of a systematic vaccination program in patients with autoimmune inflammatory disease treated with anti-TNF alpha drugs. Expert. Opin. Biol. Ther. 2016, 16, 1317–1322. [Google Scholar] [CrossRef]
- van Assen, S.; Agmon-Levin, N.; Elkayam, O.; Cervera, R.; Doran, M.F.; Dougados, M.; Emery, P.; Geborek, P.; Ioannidis, J.P.A.; Jayne, D.R.W.; et al. EULAR recommendations for vaccination in adult patients with autoimmune inflammatory rheumatic diseases. Ann. Rheum. Dis. 2011, 70, 414–422. [Google Scholar] [CrossRef]
- Furer, V.; Rondaan, C.; Heijstek, M.W.; Agmon-Levin, N.; van Assen, S.; Bijl, M.; Breedveld, F.C.; D’Amelio, R.; Dougados, M.; Kapetanovic, M.C.; et al. 2019 update of EULAR recommendations for vaccination in adult patients with autoimmune inflammatory rheumatic diseases. Ann. Rheum. Dis. 2020, 79, 39–52. [Google Scholar] [CrossRef]
- Use of 13-Valent Pneumococcal Conjugate Vaccine and 23-Valent Pneumococcal Polysaccharide Vaccine for Adults with Immunocompromising Conditions: Recommendations of the Advisory Committee on Immunization Practices (ACIP). Available online: https://www.cdc.gov/mmwr/preview/mmwrhtml/mm6140a4.htm (accessed on 1 June 2020).
- González-Romo, F.; Picazo, J.J.; García, A.; Labrador, M.; Barrios, V.; Magro, M.C.; Gil, P.; de la Cámara, G.; Rodríguez, A.; Barberán, J.; et al. Consenso sobre la vacunación anti-neumocócica en el adulto por riesgo de edad y patología de base. Actualización 2017. Rev. Esp. Quim. 2017, 30, 142–168. [Google Scholar]
- Nguyen, M.; Lindegaard, H.; Hendricks, O.; Friis-Møller, N. Factors associated with influenza and pneumococcal vaccine uptake among rheumatoid arthritis patients in Denmark invited to participate in a pneumococcal vaccine trial (Immunovax_RA). Scand. J. Rheumatol. 2017, 46, 446–453. [Google Scholar] [CrossRef]
- Brocq, O.; Acquacalda, E.; Berthier, F.; Albert, C.; Bolla, G.; Millasseau, E.; Destombe, C.; Azulay, J.; Asquier, C.; Florent, A.; et al. Influenza and pneumococcal vaccine coverage in 584 patients taking biological therapy for chronic inflammatory joint: A retrospective study. Jt. Bone Spine. 2016, 83, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Loubet, P.; Kernéis, S.; Groh, M.; Loulergue, P.; Blanche, P.; Verger, P.; Launay, O. Attitude, knowledge and factors associated with influenza and pneumococcal vaccine uptake in a large cohort of patients with secondary immune deficiency. Vaccine 2015, 33, 3703–3708. [Google Scholar] [CrossRef]
- Ramos-Sevillano, E.; Urzainqui, A.; de Andrés, B.; González-Tajuelo, R.; Domenech, M.; González-Camacho, F.; Sánchez-Madrid, F.; Brown, J.S.; García, E.; Yuste, J. PSGL-1 on Leukocytes is a Critical Component of the Host Immune Response against Invasive Pneumococcal Disease. PLoS Pathog. 2016, 12, e1005500. [Google Scholar] [CrossRef] [PubMed]
- Manoff, S.B.; Liss, C.; Caulfield, M.J.; Marchese, R.D.; Silber, J.; Boslego, J.; Romero-Steiner, S.; Rajam, G.; Glass, N.E.; Whitney, C.G.; et al. Revaccination with a 23-valent pneumococcal polysaccharide vaccine induces elevated and persistent functional antibody responses in adults aged 65 > or = years. J. Infect. Dis. 2010, 201, 525–533. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Nahm, M.H.; Kim, K.-H. The Effect of Age on the Response to the Pneumococcal Polysaccharide Vaccine. BMC Infect. Dis. 2010, 10, 60. Available online: https://pubmed.ncbi.nlm.nih.gov/20219110/ (accessed on 26 November 2020). [CrossRef] [PubMed]
- De Miguel, S.; Domenech, M.; González-Camacho, F.; Sempere, J.; Vicioso, D.; Sanz, J.C.; García Comas, L.; Ardanuy, C.; Fenoll, A.; Yuste, J. Nationwide trends of invasive pneumococcal disease in Spain (2009-2019) in children and adults during the pneumococcal conjugate vaccine era. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Van Aalst, M.; Langedijk, A.C.; Spijker, R.; de Bree, G.J.; Grobusch, M.P.; Goorhuis, A. The effect of immunosuppressive agents on immunogenicity of pneumococcal vaccination: A systematic review and meta-analysis. Vaccine 2018, 36, 5832–5845. [Google Scholar] [CrossRef]
- Visser, L.G. TNF-α Antagonists and Immunization. Curr. Infect. Dis. Rep. 2011, 13, 243–247. [Google Scholar] [CrossRef]
- Chiricozzi, A.; Gisondi, P.; Bellinato, F.; Girolomoni, G. Immune Response to Vaccination in Patients with Psoriasis Treated with Systemic Therapies. Vaccines 2020, 8, 769. [Google Scholar] [CrossRef]
- Mease, P.J.; Ritchlin, C.T.; Martin, R.W.; Gottlieb, A.B.; Baumgartner, S.W.; Burge, D.J.; Whitmore, J.B. Pneumococcal vaccine response in psoriatic arthritis patients during treatment with etanercept. J. Rheumatol. 2004, 31, 1356–1361. [Google Scholar]
- Rákóczi, É.; Perge, B.; Végh, E.; Csomor, P.; Pusztai, A.; Szamosi, S.; Bodnár, N.; Szántó, S.; Szücs, G.; Szekanecz, Z. Evaluation of the immunogenicity of the 13-valent conjugated pneumococcal vaccine in rheumatoid arthritis patients treated with etanercept. Jt. Bone Spine 2016, 83, 675–679. [Google Scholar] [CrossRef]
- Craft, J.E. Follicular helper T cells in immunity and systemic autoimmunity. Nat. Rev. Rheumatol. 2012, 8, 337–347. [Google Scholar] [CrossRef] [PubMed]
- Glaesener, S.; Quách, T.D.; Onken, N.; Weller-Heinemann, F.; Dressler, F.; Huppertz, H.-I.; Thon, A.; Meyer-Bahlburg, A. Distinct Effects of Methotrexate and Etanercept on the B Cell Compartment in Patients With Juvenile Idiopathic Arthritis. Arthritis. Rheumatol. Hoboken. Nj 2014, 66, 2590–2600. [Google Scholar] [CrossRef]
- Kaine, J.L.; Kivitz, A.J.; Birbara, C.; Luo, A.Y. Immune responses following administration of influenza and pneumococcal vaccines to patients with rheumatoid arthritis receiving adalimumab. J. Rheumatol. 2007, 34, 272–279. [Google Scholar]
- Bingham, C.O.; Looney, R.J.; Deodhar, A.; Halsey, N.; Greenwald, M.; Codding, C.; Trzaskoma, B.; Martin, F.; Agarwal, S.; Kelman, A. Immunization responses in rheumatoid arthritis patients treated with rituximab: Results from a controlled clinical trial. Arthritis Rheum. 2010, 62, 64–74. [Google Scholar] [CrossRef] [PubMed]
- Pham, T.; Fautrel, B.; Gottenberg, J.-E.; Goupille, P.; Hachulla, E.; Masson, C.; Morel, J.; Mouthon, L.; Saraux, A.; Schaeverbeke, T.; et al. Rituximab (MabThera) therapy and safety management. Clinical tool guide. Jt. Bone. Spine. Rev. Rhum. 2008, 75, S1–S99. [Google Scholar]
- Subesinghe, S.; Bechman, K.; Rutherford, A.I.; Goldblatt, D.; Galloway, J.B. A Systematic Review and Metaanalysis of Antirheumatic Drugs and Vaccine Immunogenicity in Rheumatoid Arthritis. J. Rheumatol. 2018, 45, 733–744. [Google Scholar] [CrossRef] [PubMed]
- Fischer, L.; Gerstel, P.F.; Poncet, A.; Siegrist, C.-A.; Laffitte, E.; Gabay, C.; Seebach, J.D.; Ribi, C. Pneumococcal polysaccharide vaccination in adults undergoing immunosuppressive treatment for inflammatory diseases—A longitudinal study. Arthritis Res. Ther. 2015, 17, 151. [Google Scholar] [CrossRef]
- Ladhani, S.N.; Collins, S.; Djennad, A.; Sheppard, C.L.; Borrow, R.; Fry, N.K.; Andrews, N.J.; Miller, E.; Ramsay, M.E. Rapid increase in non-vaccine serotypes causing invasive pneumococcal disease in England and Wales, 2000–2017: A prospective national observational cohort study. Lancet. Infect. Dis. 2018, 18, 441–451. [Google Scholar] [CrossRef]
- Choi, E.H.; Zhang, F.; Lu, Y.-J.; Malley, R. Capsular Polysaccharide (CPS) Release by Serotype 3 Pneumococcal Strains Reduces the Protective Effect of Anti-Type 3 CPS Antibodies. Clin. Vaccin. Immunol. CVI 2016, 23, 162–167. [Google Scholar] [CrossRef]
- Domenech, M.; Sempere, J.; de Miguel, S.; Yuste, J. Combination of Antibodies and Antibiotics as a Promising Strategy Against Multidrug-Resistant Pathogens of the Respiratory Tract. Front. Immunol. 2018, 9, 2700. [Google Scholar] [CrossRef] [PubMed]
- Cafini, F.; Yuste, J.; Giménez, M.-J.; Sevillano, D.; Aguilar, L.; Alou, L.; Ramos-Sevillano, E.; Torrico, M.; González, N.; García, E.; et al. Enhanced in vivo activity of cefditoren in pre-immunized mice against penicillin-resistant S. pneumoniae (serotypes 6B, 19F and 23F) in a sepsis model. PLoS ONE 2010, 5, e12041. [Google Scholar] [CrossRef]
- Ramos-Sevillano, E.; Rodríguez-Sosa, C.; Díez-Martínez, R.; Giménez, M.-J.; Olmedillas, E.; García, P.; García, E.; Aguilar, L.; Yuste, J. Macrolides and β-lactam antibiotics enhance C3b deposition on the surface of multidrug-resistant Streptococcus pneumoniae strains by a LytA autolysin-dependent mechanism. Antimicrob. Agents. Chemother. 2012, 56, 5534–5540. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Sevillano, E.; Rodríguez-Sosa, C.; Cafini, F.; Giménez, M.-J.; Navarro, A.; Sevillano, D.; Alou, L.; García, E.; Aguilar, L.; Yuste, J. Cefditoren and ceftriaxone enhance complement-mediated immunity in the presence of specific antibodies against antibiotic-resistant pneumococcal strains. PLoS ONE 2012, 7, e44135. [Google Scholar] [CrossRef] [PubMed]

| Group | n (%) | Age (Mean ± SD) | Gender, n (%) Females | Disease Duration (Mean ± SD), in Years |
|---|---|---|---|---|
| Whole cohort | 182 (100) | 50 ± 13 | 109 (59.9) | 9.4 ± 8.8 |
| Diagnosis | ||||
| Spondyloarthritis | 65 (35.7) | 46 ± 11 | 28 (43.1) | 9.8 ± 10.0 |
| Rheumatoid arthritis | 63 (34.6) | 55 ± 12 | 50 (79.4) | 9.9 ± 7.9 |
| Psoriatic arthritis | 28 (15.3) | 50 ± 12 | 14 (50.0) | 7.6 ± 8.1 |
| Psoriasis | 8 (4.4) | 46 ± 10 | 5 (62.5) | 17.0 ± 10.5 |
| Inflammatory bowel disease | 7 (3.9) | 41 ± 11 | 3 (42.9) | 7.9 ± 6.6 |
| Connective tissue diseases * | 7 (3.9) | 54 ± 16 | 7 (100.0) | 2.4 ± 2.1 |
| Undifferentiated arthritis | 4 (2.2) | 33 ± 11 | 2 (50.0) | 4.7 ± 7.2 |
| Biological DMARDs | ||||
| Etanercept | 57 (31.3) | 49 ± 14 | 30 (52.6) | 6.3 ± 5.4 |
| Adalimumab | 54 (29.7) | 50 ± 10 | 29 (53.7) | 10.8 ± 9.5 |
| Infliximab | 21 (11.5) | 49 ± 13 | 13 (61.9) | 15.1 ± 12.7 |
| Golimumab | 17 (9.3) | 49 ± 11 | 9 (52.9) | 8.6 ± 7.9 |
| Certolizumab | 6 (3.3) | 56 ± 10 | 3 (50.0) | 5.5 ± 3.7 |
| Rituximab | 14 (7.7) | 56 ± 11 | 12 (85.7) | 10.9 ± 9.6 |
| Tocilizumab | 10 (5.5) | 49 ± 17 | 10 (100.0) | 7.9 ± 7.3 |
| Abatacept | 2 (1.1) | 41 ± 16 | 2 (100.0) | 9.8 ± 5.9 |
| Anakinra | 1 (0.6) | 41 | 1 (100.0) | 5.5 |
| Synthetic DMARDs | ||||
| Methotrexate | 55 (30.2) | 51 ± 14 | 38 (69.1) | 9.3 ± 7.8 |
| Leflunomide | 15 (8.2) | 56 ± 14 | 10 (66.7) | 8.2 ± 6.8 |
| Sulfasalazine | 3 (1.6) | 50 ± 11 | 1 (33.3) | 7.5 ± 10.6 |
| Azathioprine | 2 (1.1) | 34 ± 15 | 2 (100.0) | 7.0 ± 7.0 |
| Hydroxychloroquine | 1 (0.6) | 50.8 | 1 (100.0) | 4.8 |
| Cyclosporine | 1 (0.6) | 51.8 | 1 (100.0) | 15.8 |
| Glucocorticoids | ||||
| Prednisone ≤7.5 mg/d | 23 (12.6) | 55 ± 16 | 18 (78.3) | 10.6 ± 9.0 |
| Prednisone >7.5 mg/d | 5 (2.7) | 49 ± 14 | 3 (60.0) | 3.0 ± 4.2 |
| Pneumococcal Serotypes | Patients with OT n (%) |
|---|---|
| 1 | 80 (44.0) |
| 3 | 86 (47.3) |
| 7F | 65 (35.7) |
| 14 | 81 (44.5) |
| 19A | 65 (35.7) |
| 19F | 60 (33.0) |
| Pneumococcal Serotypes | Patients with OT n (%) |
|---|---|
| 0 | 26 (14.3) |
| 1 | 40 (22.0) |
| 2 | 27 (14.8) |
| 3 | 39 (21.4) |
| 4 | 28 (15.4) |
| 5 | 20 (11.0) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Richi, P.; Yuste, J.; Navío, T.; González-Hombrado, L.; Salido, M.; Thuissard-Vasallo, I.; Jiménez-Díaz, A.; Llorente, J.; Cebrián, L.; Lojo, L.; et al. Impact of Biological Therapies on the Immune Response after Pneumococcal Vaccination in Patients with Autoimmune Inflammatory Diseases. Vaccines 2021, 9, 203. https://doi.org/10.3390/vaccines9030203
Richi P, Yuste J, Navío T, González-Hombrado L, Salido M, Thuissard-Vasallo I, Jiménez-Díaz A, Llorente J, Cebrián L, Lojo L, et al. Impact of Biological Therapies on the Immune Response after Pneumococcal Vaccination in Patients with Autoimmune Inflammatory Diseases. Vaccines. 2021; 9(3):203. https://doi.org/10.3390/vaccines9030203
Chicago/Turabian StyleRichi, Patricia, Jose Yuste, Teresa Navío, Laura González-Hombrado, Marina Salido, Israel Thuissard-Vasallo, Ana Jiménez-Díaz, Jesús Llorente, Laura Cebrián, Leticia Lojo, and et al. 2021. "Impact of Biological Therapies on the Immune Response after Pneumococcal Vaccination in Patients with Autoimmune Inflammatory Diseases" Vaccines 9, no. 3: 203. https://doi.org/10.3390/vaccines9030203
APA StyleRichi, P., Yuste, J., Navío, T., González-Hombrado, L., Salido, M., Thuissard-Vasallo, I., Jiménez-Díaz, A., Llorente, J., Cebrián, L., Lojo, L., Steiner, M., Cobo, T., Martín, M. D., García-Castro, M., Castro, P., & Muñoz-Fernández, S. (2021). Impact of Biological Therapies on the Immune Response after Pneumococcal Vaccination in Patients with Autoimmune Inflammatory Diseases. Vaccines, 9(3), 203. https://doi.org/10.3390/vaccines9030203







