Impact of Obesity and Being Overweight on the Immunogenicity to Live Attenuated Hepatitis A Vaccine in Children and Young Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
Anthropometric Measurements
2.2. Study Vaccine
2.2.1. Assessment of Immunogenicity and Serological Assay
2.2.2. Assessment of Safety
2.3. Statistical Analysis
3. Results
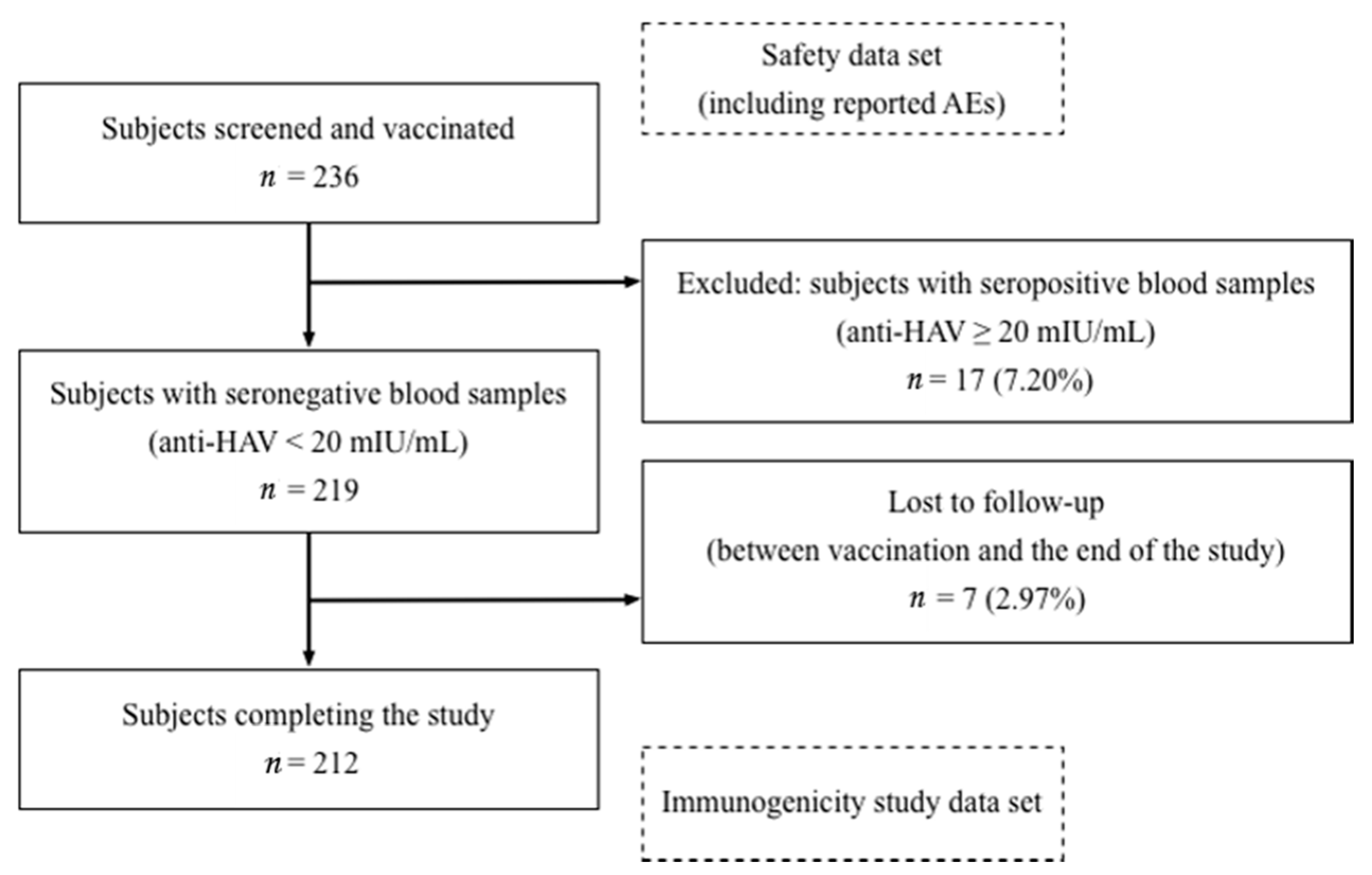
3.1. Participants
Baseline Demographic and Clinical Characteristics
3.2. Postvaccination Immunogenicity
3.3. Safety and Adverse Events
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Teerawattananon, Y.; Luz, A. Obesity in Thailand and Its Economic Cost Estimation; ADBI Working Paper 703; Asian Development Bank Institute: Tokyo, Japan, 2017; Available online: https://www.adb.org/sites/default/files/publication/236536/adbi-wp703.pdf (accessed on 1 January 2021).
- Jitnarin, N.; Kosulwat, V.; Rojroongwasinkul, N.; Boonpraderm, A.; Haddock, C.K.; Poston, W.S. Prevalence of overweight and obesity in Thai population: Results of the National Thai Food Consumption Survey. Eat. Weight Disord. 2011, 16, e242–e249. [Google Scholar] [CrossRef]
- Bandaru, P.; Rajkumar, H.; Nappanveettil, G. The impact of obesity on immune response to infection and vaccine: An insight into plausible mechanisms. Endocrinol. Metab. Synd. 2013, 2, 2. [Google Scholar] [CrossRef]
- Eliakim, A.; Schwindt, C.; Zaldivar, F.; Casali, P.; Cooper, D.M. Reduced tetanus antibody titers in overweight children. Autoimmunity 2006, 39, 137–141. [Google Scholar] [CrossRef]
- Andersen, C.J.; Murphy, K.E.; Fernandez, M.L. Impact of Obesity and Metabolic Syndrome on Immunity. Adv. Nutr. 2016, 7, 66–75. [Google Scholar] [CrossRef]
- Painter, S.D.; Ovsyannikova, I.G.; Poland, G.A. The weight of obesity on the human immune response to vaccination. Vaccine 2015, 33, 4422–4429. [Google Scholar] [CrossRef] [PubMed]
- Young, K.M.; Gray, C.M.; Bekker, L.G. Is obesity a risk factor for vaccine non-responsiveness? PLoS ONE 2013, 8, e82779. [Google Scholar] [CrossRef] [PubMed]
- Simó Miñana, J.; Gaztambide Ganuza, M.; Fernández Millán, P.; Peña Fernández, M. Hepatitis B vaccine immunoresponsiveness in adolescents: A revaccination proposal after primary vaccination. Vaccine 1996, 14, 103–106. [Google Scholar] [CrossRef]
- Averhoff, F.; Mahoney, F.; Coleman, P.; Schatz, G.; Hurwitz, E.; Margolis, H. Immunogenicity of Hepatitis B vaccines: Implications for persons at occupational risk of Hepatitis B virus infection. Am. J. Prev. Med. 1998, 15, 1–8. [Google Scholar] [CrossRef]
- Reuman, P.D.; Kubilis, P.; Hurni, W.; Brown, L.; Nalin, D. The effect of age and weight on the response to formalin inactivated, alum-adjuvanted hepatitis A vaccine in healthy adults. Vaccine 1997, 15, 1157–1161. [Google Scholar] [CrossRef]
- Van der Wielen, M.; Van Damme, P.; Chlibek, R.; Smetana, J.; von Sonnenburg, F. Hepatitis A/B vaccination of adults over 40 years old: Comparison of three vaccine regimens and effect of influencing factors. Vaccine 2006, 24, 5509–5515. [Google Scholar] [CrossRef]
- Lim, J.; Song, Y.J.; Park, W.S.; Sohn, H.; Lee, M.S.; Shin, D.H.; Kim, C.B.; Kim, H.; Oh, G.J.; Ki, M. The immunogenicity of a single dose of hepatitis A virus vaccines (Havrix(R) and Epaxal(R)) in Korean young adults. Yonsei Med. J. 2014, 55, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.; Mao, J.S.; Motlekar, S.; Fangcheng, Z.; Kadhe, G. A review of immunogenicity and tolerability of live attenuated Hepatitis A vaccine in children. Hum. Vaccin. Immunother. 2016, 12, 3160–3165. [Google Scholar] [CrossRef]
- WHO position paper on hepatitis A vaccines: June 2012-recommendations. Vaccine 2013, 31, 285–286. [CrossRef] [PubMed]
- Vashishtha, V.M.; Choudhury, P.; Kalra, A.; Bose, A.; Thacker, N.; Yewale, V.N.; Bansal, C.P.; Mehta, P.J. Indian Academy of Pediatrics (IAP) Recommended Immunization Schedule for Children Aged 0 through 18 years—India, 2014 and Updates on Immunization. Indian Pediatr. 2014, 51, 785–803. [Google Scholar] [CrossRef] [PubMed]
- The Asia-Pacific Perspective: Redefining Obesity and Its Treatment. Available online: https://apps.who.int/iris/handle/10665/206936 (accessed on 1 January 2021).
- WHO Child Growth Standards based on length/height, weight and age. Acta. Paediatr. Suppl. 2006, 450, 76–85. [CrossRef]
- de Onis, M.; Onyango, A.W.; Borghi, E.; Garza, C.; Yang, H. Comparison of the World Health Organization (WHO) Child Growth Standards and the National Center for Health Statistics/WHO international growth reference: Implications for child health programmes. Public Health Nutr. 2006, 9, 942–947. [Google Scholar] [CrossRef]
- Ahmad, N.; Adam, S.I.; Nawi, A.M.; Hassan, M.R.; Ghazi, H.F. Abdominal Obesity Indicators: Waist Circumference or Waist-to-hip Ratio in Malaysian Adults Population. Int. J. Prev. Med. 2016, 7, 82. [Google Scholar] [CrossRef] [PubMed]
- Jolliffe, C.J.; Janssen, I. Development of age-specific adolescent metabolic syndrome criteria that are linked to the Adult Treatment Panel III and International Diabetes Federation criteria. J. Am. Coll. Cardiol. 2007, 49, 891–898. [Google Scholar] [CrossRef]
- Ma, G.S.; Ji, C.Y.; Ma, J.; Mi, J.; Yt Sung, R.; Xiong, F.; Yan, W.L.; Hu, X.Q.; Li, Y.P.; Du, S.M.; et al. Waist circumference reference values for screening cardiovascular risk factors in Chinese children and adolescents. Biomed. Environ. Sci. 2010, 23, 21–31. [Google Scholar] [CrossRef]
- Bhave, S.; Ghosh, A.; Sapru, A.; Mitra, M.; Chatterjee, S.; Bhattacharya, N.; Kadhe, G.; Mane, A.; Roy, S. Immunogenicity and safety of live attenuated hepatitis A vaccine (Biovac-A™) in healthy Indian children. Vaccine Dev. Ther. 2014, 4, 1–6. [Google Scholar] [CrossRef]
- Tabachnick, B.G.; Fidell, L.S. Multiple regression. In Using Multivariate Statistics; Tabachnick, B.G., Fidell, L.S., Eds.; Pearson Education: Boston, MA, USA, 2013; pp. 117–196. [Google Scholar]
- Siddiqui, K. Heuristics for sample size determination in multivariate statistical techniques. World Appl. Sci. J. 2013, 27, 285–287. [Google Scholar]
- Jacobsen, K.H.; Koopman, J.S. Declining hepatitis A seroprevalence: A global review and analysis. Epidemiol. Infect. 2004, 132, 1005–1022. [Google Scholar] [CrossRef] [PubMed]
- Sa-nguanmoo, P.; Posuwan, N.; Vichaiwattana, P.; Vuthitanachot, V.; Saelao, S.; Foonoi, M.; Fakthongyoo, A.; Makaroon, J.; Srisingh, K.; Asawarachun, D.; et al. Declining Trend of Hepatitis A Seroepidemiology in Association with Improved Public Health and Economic Status of Thailand. PLoS ONE 2016, 11, e0151304. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; An, J.; Tu, A.; Liang, X.; Cui, F.; Zheng, H.; Tang, Y.; Liu, J.; Wang, X.; Zhang, N.; et al. Comparison of immune persistence among inactivated and live attenuated hepatitis a vaccines 2 years after a single dose. Hum. Vaccin. Immunother. 2016, 12, 2322–2326. [Google Scholar] [CrossRef] [PubMed]
- Bhave, S.; Bavdekar, A.; Madan, Z.; Jha, R.; Bhure, S.; Chaudhari, J.; Pandit, A. Evaluation of immunogenicity and tolerability of a live attenuated hepatitis a vaccine in Indian children. Indian Pediatr. 2006, 43, 983–987. [Google Scholar]
- WHO Immunological Basis for Immunization Series: Module 18: Hepatitis A, Update 2019. Available online: https://apps.who.int/iris/handle/10665/326501 (accessed on 1 January 2021).
- Beasley, L.E.; Koster, A.; Newman, A.B.; Javaid, M.K.; Ferrucci, L.; Kritchevsky, S.B.; Kuller, L.H.; Pahor, M.; Schaap, L.A.; Visser, M.; et al. Inflammation and race and gender differences in computerized tomography-measured adipose depots. Obes. (Silver Spring) 2009, 17, 1062–1069. [Google Scholar] [CrossRef]
- Park, H.S.; Park, J.Y.; Yu, R. Relationship of obesity and visceral adiposity with serum concentrations of CRP, TNF-alpha and IL-6. Diabetes Res. Clin. Pract. 2005, 69, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Ziccardi, P.; Nappo, F.; Giugliano, G.; Esposito, K.; Marfella, R.; Cioffi, M.; D’Andrea, F.; Molinari, A.M.; Giugliano, D. Reduction of inflammatory cytokine concentrations and improvement of endothelial functions in obese women after weight loss over one year. Circulation 2002, 105, 804–809. [Google Scholar] [CrossRef] [PubMed]
- Honce, R.; Schultz-Cherry, S. Impact of Obesity on Influenza A Virus Pathogenesis, Immune Response, and Evolution. Front. Immunol. 2019, 10, 1071. [Google Scholar] [CrossRef]
- Kulkarni, P.S.; Raut, S.K.; Patki, P.S.; Phadke, M.A.; Jadhav, S.S.; Kapre, S.V.; Dhorje, S.P.; Godse, S.R. Immunogenicity of a new, low-cost recombinant hepatitis B vaccine derived from Hansenula polymorpha in adults. Vaccine 2006, 24, 3457–3460. [Google Scholar] [CrossRef]
- Callahan, S.T.; Wolff, M.; Hill, H.R.; Edwards, K.M. Impact of body mass index on immunogenicity of pandemic H1N1 vaccine in children and adults. J. Infect. Dis. 2014, 210, 1270–1274. [Google Scholar] [CrossRef] [PubMed]
- Bollaerts, K.; Alexandridou, M.; Verstraeten, T. Risk factors for modified vaccine effectiveness of the live attenuated zoster vaccine among the elderly in England. Vaccine X 2019, 1, 100007. [Google Scholar] [CrossRef]
- Estrada, L.D.; Vazquez-Pagan, A.G.; Schultz-Cherry, S. Live attenuated influenza virus vaccine protects obese mice. J. Immunol. 2020, 204, 245.16. [Google Scholar]
- Sheridan, P.A.; Paich, H.A.; Handy, J.; Karlsson, E.A.; Hudgens, M.G.; Sammon, A.B.; Holland, L.A.; Weir, S.; Noah, T.L.; Beck, M.A. Obesity is associated with impaired immune response to influenza vaccination in humans. Int. J. Obes. (Lond.) 2012, 36, 1072–1077. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Cui, F.Q. The immunogenicity and impact factors of hepatitis A attenuated live vaccine and inactivated vaccine. Zhongguo Yi Miao He Mian Yi 2009, 15, 371–374. [Google Scholar]
- Wagner, G.; Lavanchy, D.; Darioli, R.; Pécoud, A.; Brulein, V.; Safary, A.; Frei, P.C. Simultaneous active and passive immunization against hepatitis A studied in a population of travellers. Vaccine 1993, 11, 1027–1032. [Google Scholar] [CrossRef]
- Chen, S.; Akbar, S.M.; Miyake, T.; Abe, M.; Al-Mahtab, M.; Furukawa, S.; Bunzo, M.; Hiasa, Y.; Onji, M. Diminished immune response to vaccinations in obesity: Role of myeloid-derived suppressor and other myeloid cells. Obes. Res. Clin. Pract. 2015, 9, 35–44. [Google Scholar] [CrossRef]
- Goubau, P.; Van Gerven, V.; Safary, A.; Delem, A.; Knops, J.; D’Hondt, E.; André, F.E.; Desmyter, J. Effect of virus strain and antigen dose on immunogenicity and reactogenicity of an inactivated hepatitis A vaccine. Vaccine 1992, 10 (Suppl. S1), S114–S118. [Google Scholar] [CrossRef]
- Giefing-Kröll, C.; Berger, P.; Lepperdinger, G.; Grubeck-Loebenstein, B. How sex and age affect immune responses, susceptibility to infections, and response to vaccination. Aging Cell 2015, 14, 309–321. [Google Scholar] [CrossRef] [PubMed]
- Klein, S.L.; Jedlicka, A.; Pekosz, A. The Xs and Y of immune responses to viral vaccines. Lancet Infect. Dis. 2010, 10, 338–349. [Google Scholar] [CrossRef]
- Magnuson, A.; Fouts, J.; Booth, A.; Foster, M. Obesity-induced chronic low grade inflammation: Gastrointestinal and adipose tissue crosstalk. Integr. Obes. Diabetes 2015, 1, 103–108. [Google Scholar] [CrossRef]
- Batra, A.; Okur, B.; Glauben, R.; Erben, U.; Ihbe, J.; Stroh, T.; Fedke, I.; Chang, H.D.; Zeitz, M.; Siegmund, B. Leptin: A critical regulator of CD4+ T-cell polarization in vitro and in vivo. Endocrinology 2010, 151, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Batra, A.; Siegmund, B. The role of visceral fat. Dig. Dis. 2012, 30, 70–74. [Google Scholar] [CrossRef]
- Frühbeck, G. Overview of adipose tissue and its role in obesity and metabolic disorders. Methods Mol. Biol. 2008, 456, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Stoddart, M.L.; Blevins, K.S.; Lee, E.T.; Wang, W.; Blackett, P.R. Cherokee Diabetes Study. Association of acanthosis nigricans with hyperinsulinemia compared with other selected risk factors for type 2 diabetes in Cherokee Indians: The Cherokee Diabetes Study. Diabetes Care 2002, 25, 1009–1014. [Google Scholar] [CrossRef]
- Koh, Y.K.; Lee, J.H.; Kim, E.Y.; Moon, K.R. Acanthosis nigricans as a clinical predictor of insulin resistance in obese children. Pediatr. Gastroenterol. Hepatol. Nutr. 2016, 19, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Faridi, M.M.; Shah, N.; Ghosh, T.K.; Sankaranarayanan, V.S.; Arankalle, V.; Aggarwal, A.; Sathiyasekaran, M.; Bhattacharya, N.; Vasanthi, T.; Chatterjee, S.; et al. Immunogenicity and safety of live attenuated hepatitis A vaccine: A multicentric study. Indian Pediatr. 2009, 46, 29–34. [Google Scholar]
- Wang, X.Y.; Xu, Z.Y.; Ma, J.C.; von Seidlein, L.; Zhang, Y.; Hao, Z.Y.; Han, O.P.; Zhang, Y.L.; Tian, M.Y.; Ouyang, P.Y.; et al. Long-term immunogenicity after single and booster dose of a live attenuated hepatitis A vaccine: Results from 8-year follow-up. Vaccine 2007, 25, 446–449. [Google Scholar] [CrossRef]
| Total (n = 212) | Non-Obese Group (n = 117) | Obese Group (n = 95) | p | ||
|---|---|---|---|---|---|
| Gender, n (%) | Male | 81 (38.21) | 35 (29.91) | 46 (48.42) | 0.006 1,* |
| Female | 131 (61.79) | 82 (70.09) | 49 (51.58) | ||
| Age (years) [mean (SD)] | 13.95 (3.90) | 14.20 (3.47) | 13.63 (4.37) | 0.288 2 | |
| Age group 3, n (%) | Children | 172 (81.13) | 95 (81.20) | 77 (81.05) | 0.979 1 |
| Adults | 40 (18.87) | 22 (18.80) | 18 (18.95) | ||
| Truncal obesity, n (%) | No | 134 (63.21) | 113 (96.58) | 21 (22.11) | <0.001 2,* |
| Yes | 78 (36.79) | 4 (3.42) | 74 (77.89) | ||
| Anti-HAV titers, (mlU/mL) [GMT (95% CI)] | 2.60 (2.27, 2.97) | 2.63 (2.19, 3.15) | 2.56 (2.09, 3.13) | 0.851 2 |
| n | Postvaccination Anti-HAV Titers, GMT (95% CI) | Univariate p 1 | Multivariate p 2 | |
|---|---|---|---|---|
| Baseline anti-HAV titers | 212 | 2.60 (2.27, 2.97) | 0.416 | |
| Nutritional status 3 | ||||
| Non-obese group | 117 | 429.51 (401.97, 458.94) | Ref | |
| Obese group | 95 | 467.45 (424.47, 514.79) | 0.142 | |
| Age groups | ||||
| Adults | 40 | 477.78 (411.03, 555.37) | Ref | |
| Children | 172 | 439.06 (413.22, 466.52) | 0.249 | |
| Truncal obesity | ||||
| No | 134 | 420.78 (395.28, 447.92) | Ref | Ref |
| Yes | 78 | 493.26 (442.86, 549.39) | 0.007 * | 0.002 * |
| Gender | ||||
| Male | 81 | 414.58 (372.97, 460.83) | Ref | Ref |
| Female | 131 | 466.81 (438.02, 497.49) | 0.044 * | 0.013 * |
| n | Postvaccination Anti-HAV Titers, GMT (95% CI) | Univariate p 1 | Multivariate p 2 | |
|---|---|---|---|---|
| Baseline anti-HAV titers | 95 | 2.56 (2.09, 3.13) | 0.050 | |
| Obesity levels | ||||
| Overweight | 34 | 436.69 (380.27, 501.45) | Ref | |
| Obesity | 31 | 474.37 (410.64, 547.99) | 0.485 | |
| Severe obesity | 30 | 497.38 (395.36, 625.72) | 0.277 | |
| Age groups | ||||
| Adults | 18 | 462.59 (355.63, 601.72) | Ref | |
| Children | 77 | 468.60 (421.82, 520.56) | 0.919 | |
| Truncal obesity | ||||
| No | 21 | 371.07 (314.18, 438.25) | Ref | Ref |
| Yes | 74 | 499.11 (446.37, 558.08) | 0.011 * | 0.011 * |
| Gender | ||||
| Male | 46 | 449.93 (381.09, 531.21) | Ref | |
| Female | 49 | 484.53 (434.63, 540.15) | 0.449 | |
| Acanthosis nigricans | ||||
| No | 62 | 442.11 (400.04, 488.61) | Ref | |
| Yes | 33 | 519.06 (421.40, 639.36) | 0.116 |
| Adverse Event | Number of Vaccinees | p | ||
|---|---|---|---|---|
| Total (n = 234) * | Non-Obese (n = 126) | Obese (n = 108) | ||
| No complication, n (%) | 165 (70.51) | 85 (67.46) | 80 (74.07) | 0.269 |
| Fever, n (%) | 26 (11.11) | 15 (11.90) | 11 (10.19) | 0.677 |
| Headache, n (%) | 30 (12.82) | 17 (13.49) | 13 (12.04) | 0.740 |
| Myalgia, n (%) | 36 (15.38) | 22 (17.46) | 14 (12.96) | 0.342 |
| Abdominal discomfort, n (%) | 4 (1.71) | 2 (1.59) | 2 (1.85) | 1.000 |
| Swelling and pain at the injection site, n (%) | 10 (4.27) | 5 (3.97) | 5 (4.63) | 1.000 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dumrisilp, T.; Wongpiyabovorn, J.; Buranapraditkun, S.; Tubjaroen, C.; Chaijitraruch, N.; Prachuapthunyachart, S.; Sintusek, P.; Chongsrisawat, V. Impact of Obesity and Being Overweight on the Immunogenicity to Live Attenuated Hepatitis A Vaccine in Children and Young Adults. Vaccines 2021, 9, 130. https://doi.org/10.3390/vaccines9020130
Dumrisilp T, Wongpiyabovorn J, Buranapraditkun S, Tubjaroen C, Chaijitraruch N, Prachuapthunyachart S, Sintusek P, Chongsrisawat V. Impact of Obesity and Being Overweight on the Immunogenicity to Live Attenuated Hepatitis A Vaccine in Children and Young Adults. Vaccines. 2021; 9(2):130. https://doi.org/10.3390/vaccines9020130
Chicago/Turabian StyleDumrisilp, Termpong, Jongkonnee Wongpiyabovorn, Supranee Buranapraditkun, Chomchanat Tubjaroen, Nataruks Chaijitraruch, Sittichoke Prachuapthunyachart, Palittiya Sintusek, and Voranush Chongsrisawat. 2021. "Impact of Obesity and Being Overweight on the Immunogenicity to Live Attenuated Hepatitis A Vaccine in Children and Young Adults" Vaccines 9, no. 2: 130. https://doi.org/10.3390/vaccines9020130
APA StyleDumrisilp, T., Wongpiyabovorn, J., Buranapraditkun, S., Tubjaroen, C., Chaijitraruch, N., Prachuapthunyachart, S., Sintusek, P., & Chongsrisawat, V. (2021). Impact of Obesity and Being Overweight on the Immunogenicity to Live Attenuated Hepatitis A Vaccine in Children and Young Adults. Vaccines, 9(2), 130. https://doi.org/10.3390/vaccines9020130







