COVID-19 in Low- and Middle-Income Countries (LMICs): A Narrative Review from Prevention to Vaccination Strategy
Abstract
:1. Introduction
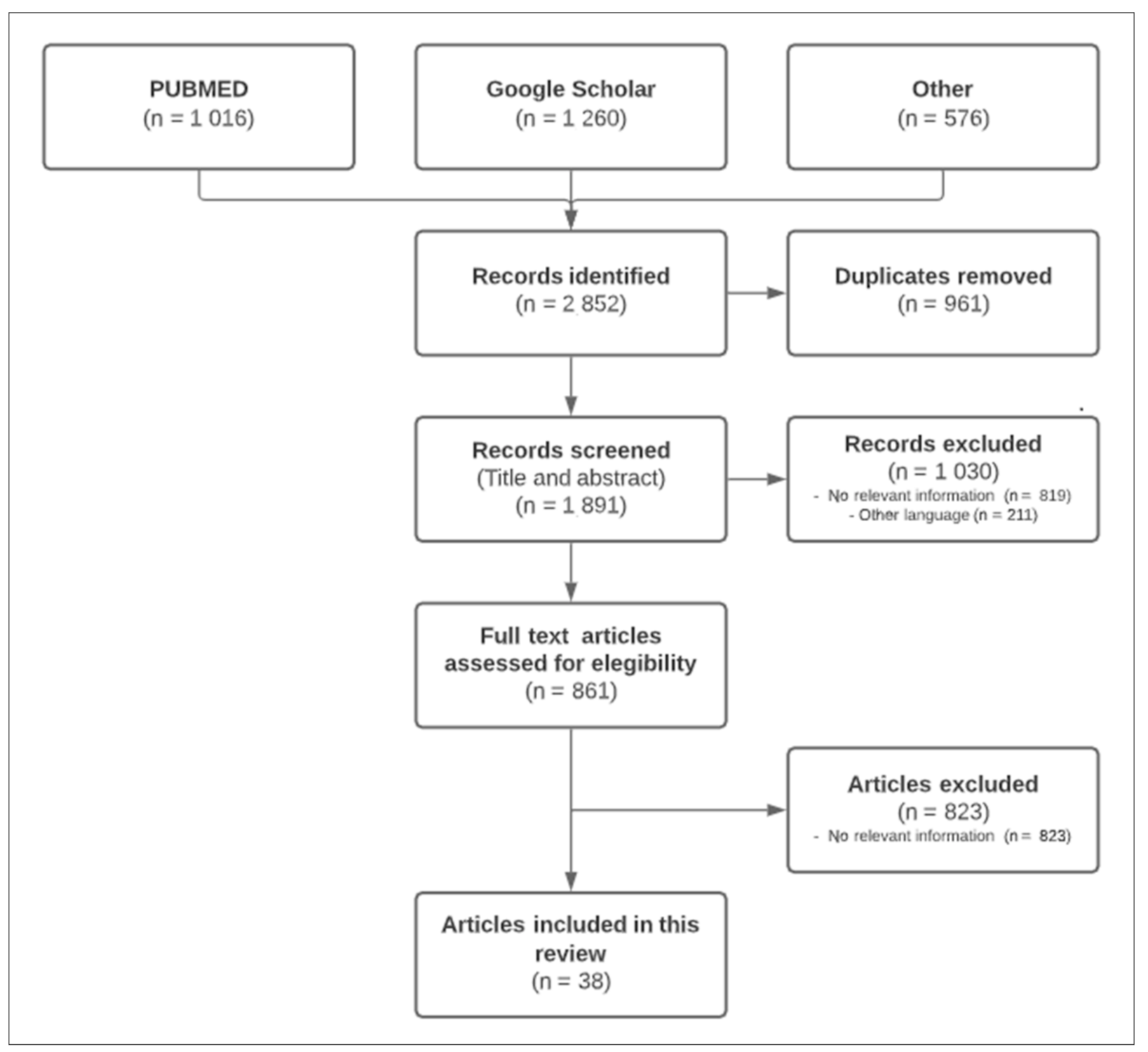
2. Materials and Methods
- publications reporting on COVID19 prevention and control, triage strategies, available infrastructure including Intensive Care Unit (ICU), treatment and vaccines;
- publications written in English.
3. Results
3.1. Infection Prevention and Control
3.2. Triage
3.3. Infrastructure and Intensive Care Unit
3.4. Treatment
3.5. Vaccine
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- World Health Organization. What Is a Pandemic? Available online: https://www.who.int/csr/disease/swineflu/frequently_asked_questions/pandemic/en/ (accessed on 5 January 2021).
- Centers for Disease Control and Prevention. Are We Prepared? Available online: https://www.cdc.gov/flu/pandemic-resources/1918-commemoration/are-we-prepared.htm (accessed on 25 April 2021).
- World Health Organization. WHO Coronavirus Disease (COVID-19) Dashboard. Available online: https://covid19.who.int/ (accessed on 31 January 2021).
- Kandel, N.; Chungong, S.; Omaar, A.; Xing, J. Health security capacities in the context of COVID-19 outbreak: An analysis of International Health Regulations annual report data from 182 countries. Lancet 2020, 395, 1047–1053. [Google Scholar] [CrossRef]
- The World Bank. The World by Income and Region. Available online: https://datatopics.worldbank.org/world-development-indicators/the-world-by-income-and-region.html (accessed on 5 January 2021).
- Anjum, F.R.; Anam, S.; Rahman, S.U. Novel Coronavirus disease 2019 (COVID-19): New challenges and new responsibilities in developing countries. Hum. Vaccines Immunother. 2020, 16, 2370–2372. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.W.; Schmitz, J.E.; Persing, D.H.; Stratton, C.W. Laboratory Diagnosis of COVID-19: Current Issues and Challenges. J. Clin. Microbiol. 2020, 58, e00512–e00520. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- The COVID Tracking Project. Totals for the US. Available online: https://covidtracking.com/data/national (accessed on 26 April 2021).
- Monjur, M.R.; Hassan, M.Z. Early Phases of COVID-19 management in a low-income country: Bangladesh. Infect. Control Hosp. Epidemiol. 2020, 41, 1116–1117. [Google Scholar] [CrossRef] [Green Version]
- Siow, W.T.; Liew, M.F.; Shrestha, B.R.; Muchtar, F.; See, K.C. Managing COVID-19 in resource-limited settings: Critical care considerations. Crit. Care 2020, 24, 167. [Google Scholar] [CrossRef] [Green Version]
- Rouzier, V.; Liautaud, B.; Deschamps, M.M. Facing the Monster in Haiti. N. Engl. J. Med. 2020, 383, e4. [Google Scholar] [CrossRef]
- World Health Organization. WHO Guide for the Stepwise Laboratory Improvement Process Towards Accreditation in the African Region (with Checklist). Available online: https://www.who.int/tb/laboratory/afro-slipta-checklist-guidance.pdf (accessed on 5 January 2021).
- Quaresima, V.; Naldini, M.M.; Cirillo, D.M. The prospects for the SARS-CoV-2 pandemic in Africa. EMBO Mol. Med. 2020, 12, e12488. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. A New Tool to Diagnose Tuberculosis: The Xpert MTB/RIF Assay. Available online: https://www.cdc.gov/tb/publications/factsheets/pdf/xpertmtb-rifassayfactsheet_final.pdf (accessed on 5 January 2021).
- Rakotosamimanana, N.; Randrianirina, F.; Randremanana, R.; Raherison, M.S.; Rasolofo, V.; Solofomalala, G.D.; Spiegel, A.; Heraud, J.-M. GeneXpert for the diagnosis of COVID-19 in LMICs. Lancet Glob. Health 2020, 8, e1457–e1458. [Google Scholar] [CrossRef]
- Lai, C.C.; Shih, T.P.; Ko, W.C.; Tang, H.J.; Hsueh, P.R. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and coronavirus disease-2019 (COVID-19): The epidemic and the challenges. Int. J. Antimicrob. Agents 2020, 55, 105924. [Google Scholar] [CrossRef]
- Krishnakumar, B.; Rana, S. COVID 19 in INDIA: Strategies to combat from combination threat of life and livelihood. J. Microbiol. Immunol. Infect. 2020, 53, 389–391. [Google Scholar] [CrossRef]
- Wang, M.-W.; Zhou, M.-Y.; Ji, G.-H.; Ye, L.; Cheng, Y.-R.; Feng, Z.-H.; Chen, J. Mask crisis during the COVID-19 outbreak. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 3397–3399. [Google Scholar]
- Sudhir, A.; Mor, N. A primary care alternative to a hospital-based approach to COVID-19 in India. J. Glob. Health 2020, 10, 020346. [Google Scholar] [CrossRef]
- World Health Organization. Algorithm for COVID-19 Triage and Referral Patient Triage and Referral for Resource-Limited Settings during Community Transmission. Available online: https://apps.who.int/iris/bitstream/handle/10665/331915/COVID-19-algorithm-referral-triage-eng.pdf (accessed on 5 January 2021).
- Joebges, S.; Biller-Andorno, N. Ethics guidelines on COVID-19 triage—An emerging international consensus. Crit. Care 2020, 24, 201. [Google Scholar] [CrossRef]
- Barros, L.M.; Pigoga, J.L.; Chea, S.; Hansoti, B.; Hirner, S.; Papali, A.; Rudd, K.E.; Schultz, M.J.; Hynes, E.J.C. Pragmatic Recommendations for Identification and Triage of Patients with COVID-19 Disease in Low- and Middle-Income Countries. Am. J. Trop. Med. Hyg. 2021, 104, 3–11. [Google Scholar] [CrossRef]
- Ayebare, R.R.; Flick, R.; Okware, S.; Bodo, B.; Lamorde, M. Adoption of COVID-19 triage strategies for low-income settings. Lancet Respir. Med. 2020, 8, e22. [Google Scholar] [CrossRef]
- Nayan, N.; Kumar, M.K.; Nair, R.K.; Manral, I.; Ghosh, S.; Bhalla, S.; Singh, J.; Monga, A.; Afzal, M.; Kapoor, R. Clinical Triaging in Cough Clinic Alleviates COVID-19 Overload in Emergency Department in India. SN Compr. Clin. Med. 2021, 3, 22–27. [Google Scholar] [CrossRef]
- Atreya, A.; Nepal, B. Covid-19 pandemic and Nepal. Med. Leg. J. 2020, 88, 102–103. [Google Scholar] [CrossRef]
- Harris, C.; Carson, G.; Baillie, J.K.; Horby, P.; Nair, H. An evidence-based framework for priority clinical research questions for COVID-19. J. Glob. Health 2020, 10, 011001. [Google Scholar] [CrossRef]
- World Health Organization. World Health Statistics 2019: Monitoring Health for the SDGs, Sustainable Development Goals. Available online: https://apps.who.int/iris/handle/10665/324835 (accessed on 5 January 2021).
- Murthy, S.; Leligdowicz, A.; Adhikari, N.K. Intensive care unit capacity in low-income countries: A systematic review. PLoS ONE 2015, 10, e0116949. [Google Scholar] [CrossRef]
- Salluh, J.I.F.; Lisboa, T.; Bozza, F.A. Challenges for the care delivery for critically ill COVID-19 patients in developing countries: The Brazilian perspective. Crit. Care 2020, 24, 593. [Google Scholar] [CrossRef]
- Marson, F.A.L.; Ortega, M.M. COVID-19 in Brazil. Pulmonology 2020, 26, 241–244. [Google Scholar] [CrossRef]
- Federal Republic of Nigeria. National Strategy for the Scale-Up of Medical Oxygen in Health Facilities. Available online: https://www.health.gov.ng/doc/National%20Startegy%20for%20Scale-up%20of%20Medical%20Oxygen.pdf (accessed on 5 January 2021).
- Republic of Uganda. Ministry of Health Scale up of Medical Oxygen in Higher Level Facilities Implementation Plan. Available online: http://library.health.go.ug/sites/default/files/resources/MOH%20National%20Oxygen%20Scale%20up%20Plan%206%20April%202019.pdf (accessed on 5 January 2021).
- World Health Organization. Severe Acute Respiratory Infections Treatment Center: Practical Manual to set up and Manage a SARI Treatment Center and SARI Screening Facility in Health Care Facilities. Available online: https://apps.who.int/iris/handle/10665/331603 (accessed on 5 January 2021).
- Sansone, P.; Giaccari, L.G.; Aurilio, C.; Coppolino, F.; Esposito, V.; Fiore, M.; Paladini, A.; Passavanti, M.B.; Pota, V.; Pace, M.C. Post-Infectious Guillain–Barré Syndrome Related to SARS-CoV-2 Infection: A Systematic Review. Life 2021, 11, 167. [Google Scholar] [CrossRef]
- Warren, T.K.; Jordan, R.; Lo, M.K.; Ray, A.S.; Mackman, R.L.; Soloveva, V.; Siegel, D.; Perron, M.; Bannister, R.; Hui, H.C.; et al. Therapeutic efficacy of the small molecule GS-5734 against Ebola virus in rhesus monkeys. Nature 2016, 531, 381–385. [Google Scholar] [CrossRef]
- Cvetkovic, R.S.; Goa, K.L. Lopinavir/ritonavir: A review of its use in the management of HIV infection. Drugs 2003, 63, 769–802. [Google Scholar] [CrossRef]
- Lei, Z.-N.; Wu, Z.-X.; Dong, S.; Yang, D.-H.; Zhang, L.; Ke, Z.; Zou, C.; Chen, Z.-S. Chloroquine and hydroxychloroquine in the treatment of malaria and repurposing in treating COVID-19. Pharmacol. Ther. 2020, 216, 107672. [Google Scholar] [CrossRef]
- National Institutes of Health. COVID-19 Treatment Guidelines Panel. Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. Available online: https://files.covid19treatmentguidelines.nih.gov/guidelines/covid19treatmentguidelines.pdf (accessed on 5 January 2021).
- FDA. Veklury (Remdesivir) EUA Letter of Approval. Available online: https://www.fda.gov/media/137564/download (accessed on 5 January 2021).
- FDA. Veklury (Remdesivir) EUA Letter of Authorization. Available online: https://www.fda.gov/media/143188/download (accessed on 5 January 2021).
- Dagens, A.; Sigfrid, L.; Cai, E.; Lipworth, S.; Cheung, V.; Harris, E.; Bannister, P.; Rigby, I.; Horby, P. Scope, quality, and inclusivity of clinical guidelines produced early in the COVID-19 pandemic: Rapid review. BMJ 2020, 369, m1936. [Google Scholar] [CrossRef]
- Liu, X.; Wang, X.J. Potential inhibitors against 2019-nCoV coronavirus M protease from clinically approved medicines. J. Genet. Genom. 2020, 47, 119–121. [Google Scholar] [CrossRef]
- Chen, F.; Chan, K.H.; Jiang, Y.; Kao, R.Y.T.; Lu, H.T.; Fan, K.W.; Cheng, V.C.C.; Tsui, W.H.W.; Hung, I.F.N.; Lee, T.S.W.; et al. In vitro susceptibility of 10 clinical isolates of SARS coronavirus to selected antiviral compounds. J. Clin. Virol. 2004, 31, 69–75. [Google Scholar] [CrossRef]
- Yao, X.; Ye, F.; Zhang, M.; Cui, C.; Huang, B.; Niu, P.; Liu, X.; Zhao, L.; Dong, E.; Song, C.; et al. In Vitro Antiviral Activity and Projection of Optimized Dosing Design of Hydroxychloroquine for the Treatment of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). Clin. Infect. Dis. 2020, 71, 732–739. [Google Scholar] [CrossRef] [Green Version]
- Saha, B.K.; Bonnier, A.; Chong, W. Antimalarials as Antivirals for COVID-19: Believe it or Not! Am. J. Med. Sci. 2020, 360, 618–630. [Google Scholar] [CrossRef]
- Rochwerg, B.; Siemieniuk, R.A.; Agoritsas, T.; Lamontagne, F.; Askie, L.; Lytvyn, L.; Agarwal, A.; Leo, Y.-S.; Macdonald, H.; Zeng, L.; et al. A living WHO guideline on drugs for COVID-19. BMJ 2020, 370, m3379, Erratum in 2020, 371, m4475. [Google Scholar]
- Hill, A.; Wang, J.; Levi, J.; Heath, K.; Fortunak, J. Minimum costs to manufacture new treatments for COVID-19. J. Virus. Erad. 2020, 6, 61–69. [Google Scholar] [CrossRef]
- The Global Fund. Available online: https://www.theglobalfund.org/en/ (accessed on 5 January 2021).
- FDA. President’s Emergency Plan for AIDS Relief (PEPFAR). Available online: https://www.fda.gov/international-programs/presidents-emergency-plan-aids-relief-pepfar (accessed on 5 January 2021).
- Meara, J.G.; Leather, A.J.M.; Hagander, L.; Alkire, B.C.; Alonso, N.; Ameh, E.A.; Bickler, S.W.; Conteh, L.; Dare, A.J.; Davies, J.; et al. Global Surgery 2030: Evidence and solutions for achieving health, welfare, and economic development. Lancet 2015, 386, 569–624. [Google Scholar] [CrossRef]
- Bibiano-Guillen, C.; Arias-Arcos, B.; Collado-Escudero, C.; Mir-Montero, M.; Corella-Montoya, F.; Torres-Macho, J.; Buendía-Garcia, M.; Larrainzar-Garijo, R. Adapted Diving Mask (ADM) device as respiratory support with oxygen output during COVID-19 pandemic. Am. J. Emerg. Med. 2021, 39, 42–47. [Google Scholar] [CrossRef]
- Dondorp, A.M.; Hayat, M.; Aryal, D.; Beane, A.; Schultz, M.J. Respiratory Support in COVID-19 Patients, with a Focus on Resource-Limited Settings. Am. J. Trop. Med. Hyg. 2020, 102, 1191–1197. [Google Scholar] [CrossRef]
- World Health Organization. Oxygen Sources and Distribution for COVID-19 Treatment Centres. Available online: https://www.who.int/publications-detail/oxygensources-and-distribution-for-covid-19-treatment-centres (accessed on 5 January 2021).
- So, A.D.; Woo, J. Reserving coronavirus disease 2019 vaccines for global access: Cross sectional analysis. BMJ 2020, 371, m4750. [Google Scholar] [CrossRef]
- McAdams, D.; McDade, K.K.; Ogbuoji, O.; Johnson, M.; Dixit, S.; Yamey, G. Incentivising wealthy nations to participate in the COVID-19 Vaccine Global Access Facility (COVAX): A game theory perspective. BMJ Glob. Health 2020, 5, e003627. [Google Scholar] [CrossRef]
- Lie, R.K.; Miller, F.G. Allocating a COVID-19 Vaccine: Balancing National and International Responsibilities. Milbank Q. 2020, 99, 450–466. [Google Scholar] [CrossRef]

| Author | Country/Region | Topics |
|---|---|---|
| Anjum F.R. et al. [6] | – | Interventions to control COVID-19 infection. |
| Tang Y.W. et al. [7] | – | Laboratory diagnosis of COVID-19 infection. |
| Monjur M.R. et al. [9] | Bangladesh | Lack of a prevention policy in Bangladesh. |
| Siow W.T. et al. [10] | LMICs | COVID-19 infection management in resource-limited settings. |
| Rouzier V. et al. [11] | Haiti | Advice for LMICs on managing COVID-19 infection. |
| Quaresima V. et al. [13] | Sub-Saharan Africa | Africa Task Force for Novel Coronavirus (AFCOR) activities for COVID-19 infection management. |
| Rakotosamimanana N. et al. [15] | LMICs | GeneXpert for the diagnosis of COVID-19 infection in LMICs. |
| Lai C.C. et al. [16] | – | Interventions to control COVID-19 infection. |
| Krishnakumar B. et al. [17] | India | Interventions to control COVID-19 infection in India. |
| Wang M.W. et al. [18] | – | Lack of protective mask. |
| Sudhir A. et al. [19] | India | Interventions to control COVID-19 infection in India. |
| WHO [20] | LMICs | Algorithm for COVID-19 triage and referral in LMICs. |
| Joebges S. et al. [21] | – | Ethics guidelines on COVID-19 triage. |
| Barros L.M. et al. [22] | LMICs | Recommendations for identification and triage of patients with COVID-19 in LMICs. |
| Ayebare R.R. et al. [23] | Uganda | Algorithm for COVID-19 triage and referral in Uganda. |
| Nayan N. et al. [24] | India | Algorithm for COVID-19 triage and referral in India. |
| Atreya A. et al. [25] | Nepal | Health resources in Nepal during COVID-19. |
| Harris C. et al. [26] | – | State of clinical research for COVID-19 and recommendations for the implementation of standardised protocols. |
| Murthy S. et al. [28] | LMICs | Intensive Care Unit capacity in LMICs during COVID-19. |
| Salluh J.I.F. et al. [29] | Brazil | Intensive Care Unit capacity in Brazil during COVID-19. |
| Marson F.A.L. et al. [30] | Brazil | COVID-19 in Brazil. |
| Federal Republic of Nigeria [31] | Nigeria | Medical oxygen in health facilities in Nigeria. |
| Replabic of Uganda [32] | Uganda | Medical oxygen implementation plan in Uganda. |
| WHO [33] | LMICs | Severe acute respiratory infections (SARI) treatment centre. |
| Author | Topics |
|---|---|
| National Institutes of Health [38] | COVID-19 Treatment Guidelines. |
| Dagens A. et al. [41] | COVID-19 Treatment Guidelines. |
| Liu X. et al. [42] | Predicted commercial medicines as potential inhibitors against COVID-19. |
| Chen F. et al. [43] | Antiviral agents activity against COVID-19. |
| Yao X. et al. [44] | Hydroxychloroquine activity against COVID-19. |
| Saha Bket al. [45] | Antimalarials activity against COVID-19. |
| Siemieniuk R. et al. [46] | COVID-19 Treatment Guidelines. |
| Hill A. et al. [47] | Minimum costs to manufacture new treatments for COVID-19. |
| Bibiano-Guillen C. et al. [51] | Adapted Diving Mask (ADM) device as respiratory support with oxygen output during COVID-19 |
| Dondorp A.M. et al. [52] | Respiratory support in LMICs during COVID-19. |
| WHO [53] | Oxygen sources and distribution for COVID-19 in LMICs. |
| So A.D. et al. [54] | COVID-19 vaccines for global access. |
| McAdams D. et al. [55] | COVID-19 vaccine Global Access Facility (COVAX). |
| Lie R.K. et al. [56] | COVID-19 vaccine allocation. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pasquale, S.; Gregorio, G.L.; Caterina, A.; Francesco, C.; Beatrice, P.M.; Vincenzo, P.; Caterina, P.M. COVID-19 in Low- and Middle-Income Countries (LMICs): A Narrative Review from Prevention to Vaccination Strategy. Vaccines 2021, 9, 1477. https://doi.org/10.3390/vaccines9121477
Pasquale S, Gregorio GL, Caterina A, Francesco C, Beatrice PM, Vincenzo P, Caterina PM. COVID-19 in Low- and Middle-Income Countries (LMICs): A Narrative Review from Prevention to Vaccination Strategy. Vaccines. 2021; 9(12):1477. https://doi.org/10.3390/vaccines9121477
Chicago/Turabian StylePasquale, Sansone, Giaccari Luca Gregorio, Aurilio Caterina, Coppolino Francesco, Passavanti Maria Beatrice, Pota Vincenzo, and Pace Maria Caterina. 2021. "COVID-19 in Low- and Middle-Income Countries (LMICs): A Narrative Review from Prevention to Vaccination Strategy" Vaccines 9, no. 12: 1477. https://doi.org/10.3390/vaccines9121477
APA StylePasquale, S., Gregorio, G. L., Caterina, A., Francesco, C., Beatrice, P. M., Vincenzo, P., & Caterina, P. M. (2021). COVID-19 in Low- and Middle-Income Countries (LMICs): A Narrative Review from Prevention to Vaccination Strategy. Vaccines, 9(12), 1477. https://doi.org/10.3390/vaccines9121477









