Safety Profile of Recommended Vaccinations in Adolescents: Data from Surveillance of Adverse Events Following Immunization in Puglia (Italy), 2016–2020
Abstract
1. Introduction
- A booster dose of diphtheria–tetanus–acellular pertussis adsorbed-inactivated poliovirus vaccine (DTaP-IPV) at 11 to 12 years of age;
- A dose of quadrivalent meningococcal conjugated vaccine (MenACYW) at 12 to 18 years of age;
- Two or three doses of human papillomavirus (HPV) vaccine, according to the vaccine and to the patient’s age (two doses at 9 to 14 years of age with a 6- to 12-month interval or 3 doses for patients 15 to 26 years of age at 0, 1 to 2 and 6 months);
- One dose a year of influenza vaccine;
- Two doses of hepatitis A vaccine at 1 to 15 years of age, at 0 and 6 to 12 months in the case of subjects living in areas where hepatitis A virus (HAV) infection is endemic [6].
2. Materials and Methods
2.1. Data Sources
2.2. Population and Database
2.3. Data Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Principi, N.; Esposito, S. Adolescents and vaccines in the western world. Vaccine 2013, 31, 5366–5374. [Google Scholar] [CrossRef] [PubMed]
- Guarinoni, M.; Dignani, L. Effectiveness of the school nurse role in increasing the vaccination coverage rate: A narrative review. Ann. Ig 2021, 33, 55–66. [Google Scholar] [PubMed]
- Giambi, C.; D’Ancona, F.; Del Manso, M.; De Mei, B.; Giovannelli, I.; Cattaneo, C.; Possenti, V.; Declich, S.; Valore, L.R.F. Exploring reasons for non-vaccination against human papillomavirus in Italy. BMC Infect. Dis. 2014, 14, 545. [Google Scholar] [CrossRef] [PubMed]
- Simon, G.R.; Baker, C.; Barden, G.A., 3rd; Brown, O.W.; Hardin, A.; Lessin, H.R.; Committee on Practice and Ambulatory Medicine. 2014 recommendations for pediatric preventive health care. Pediatrics 2014, 133, 568–570. [Google Scholar]
- Broder, K.R.; Cohn, A.C.; Schwartz, B.; Klein, J.D.; Fisher, M.M.; Fishbein, D.B.; Mijalski, C.; Burstein, G.R.; Vernon-Smiley, M.E.; McCauley, M.M.; et al. Adolescent Immunizations and Other Clinical Preventive Services: A Needle and a Hook? Pediatrics 2008, 121, S25–S33. [Google Scholar] [CrossRef] [PubMed]
- Conferenza Permanente Per i Rapporti tra lo Stato le Regioni e le Province Autonome di Trento e Bolzano, Intesa, ai Sensi Dell’articolo 8, Comma 6, Della Legge 5 Giugno 2003, n. 131, tra il Governo, le Regioni e le Province Autonome di Trento e Bol-Zano sul Documento Recante “Piano Nazionale Prevenzione Vaccinale 2017–2019”. (Rep Atti n. 10/CSR) (17°01195). Available online: https://www.trovanorme.salute.gov.it/norme/dettaglioAtto?id=58185&completo=true (accessed on 11 October 2021).
- Tosti, M.E.; Spada, E.; Romanò, L.; Zanetti, A.; Mele, A.; SEIEVA Collaborating Group. Acute hepatitis A in Italy: Incidence, risk factors and preventive measures. J. Viral Hepat. 2008, 15, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Regione puglia. Calendario Vaccinale Per la Vita 2018–Regione Puglia. Available online: https://www.sanita.puglia.it/documents/20182/26673928/Calendario+vaccinale+per+la+vita+2018/f36429b9-5f76-44c2-b495-d5b9228959ef?version=1.0&t=1549553827908 (accessed on 11 October 2021).
- World Health Organization (WHO). Causality Assessment of an Adverse Event Following Immunization (AEFI): User Manual for the Revised WHO Classification. Available online: https://www.who.int/publications/i/item/causality-assessment-aefi-user-manual-2019 (accessed on 11 October 2021). 2019 Update.
- Stefanizzi, P.; De Nitto, S.; Spinelli, G.; Lattanzio, S.; Stella, P.; Ancona, D.; Dell’Aera, M.; Padovano, M.; Soldano, S.; Tafuri, S.; et al. Post-Marketing Active Surveillance of Adverse Reactions Following Influenza Cell-Based Quadrivalent Vaccine: An Italian Prospective Observational Study. Vaccines 2021, 9, 456. [Google Scholar] [CrossRef] [PubMed]
- Tafuri, S.; Fortunato, F.; Gallone, M.S.; Stefanizzi, P.; Calabrese, G.; Boccalini, S.; Martinelli, D.; Prato, R. Systematic causality assessment of adverse events following HPV vaccines: Analysis of current data from Apulia region (Italy). Vaccine 2018, 36, 1072–1077. [Google Scholar] [CrossRef] [PubMed]
- Tafuri, S.; Gallone, M.S.; Calabrese, G.; Germinario, C. Adverse events following immunization: Is this time for the use of WHO causality assessment? Expert Rev. Vaccines 2015, 14, 625–627. [Google Scholar] [CrossRef] [PubMed]
- AIFA–Gruppo di Lavoro Sull’analisi dei Segnali dei Vaccini. Guida Alla Valutazione Delle Reazioni Avverse Osservabili Dopo Vaccinazione. 2016. Available online: http://www.aifa.gov.it/sites/default/files/Guida_valutazione_reazioni_avverse_osservabili_dopo_vaccinazione_2.pdf (accessed on 25 September 2021).
- European Medical Agency. Important Medical Event Terms List Version (MedDRA)–Version 24.0. Available online: https://www.ema.europa.eu/en/human-regulatory/research-development/pharmacovigilance/eudravigilance/eudravigilance-system-overview (accessed on 25 September 2021).
- Stefanizzi, P.; Stella, P.; Ancona, D.; Malcangi, K.N.; Bianchi, F.P.; De Nitto, S.; Ferorelli, D.; Germinario, C.A.; Tafuri, S. Adverse Events Following Measles-Mumps-Rubella-Varicella Vaccination and the Case of Seizures: A Post Marketing Active Surveillance in Puglia Italian Region, 2017–2018. Vaccines 2019, 7, 140. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food and Drug Administration. Varivax. Package Insert. 2020. Available online: https://www.fda.gov/media/76000/download (accessed on 13 October 2021).
- U.S. Food and Drug Administration. Bexsero. Package Insert. 2020. Available online: https://www.fda.gov/media/90996/download (accessed on 13 October 2021).
- U.S. Food and Drug Administration. Cervarix. Package Insert. 2020. Available online: https://www.fda.gov/media/78013/download (accessed on 13 October 2021).
- U.S. Food and Drug Administration. Gardasil. Package Insert. 2020. Available online: https://www.fda.gov/media/74350/download (accessed on 13 October 2021).
- U.S. Food and Drug Administration. Gardasil 9. Package Insert. 2020. Available online: https://www.fda.gov/media/90064/download (accessed on 13 October 2021).
- U.S. Food and Drug Administration. Havrix. Package Insert. 2020. Available online: https://www.fda.gov/media/119388/download (accessed on 13 October 2021).
- U.S. Food and Drug Administration. Trumenba. Package Insert. 2020. Available online: https://www.fda.gov/media/89936/download (accessed on 13 October 2021).
- U.S. Food and Drug Administration. MMR-II. Package Insert. 2020. Available online: https://www.fda.gov/media/75191/download (accessed on 13 October 2021).
- AIFA. Polioboostrix. Summary of Product Characteristics. 2020. Available online: https://farmaci.agenziafarmaco.gov.it/aifa/servlet/PdfDownloadServlet?pdfFileName=footer_000200_036752_FI.pdf&sys=m0b1l3 (accessed on 13 October 2021).
- AIFA. Priorix Tetra. Summary of Product Characteristics. 2020. Available online: https://farmaci.agenziafarmaco.gov.it/aifa/servlet/PdfDownloadServlet?pdfFileName=footer_000200_038200_FI.pdf&retry=0&sys=m0b1l3 (accessed on 13 October 2021).
- AIFA. Rapporto vaccini 2019. Available online: https://www.aifa.gov.it/documents/20142/241052/Rapporto_Vaccini_2019.pdf (accessed on 2 November 2021).
- Vernon, N.; Jhaveri, P. Adverse Effects of Adolescent Immunizations. J. Osteopat. Med. 2014, 114, 13–17. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lee, S.Y.; Kwak, G.Y.; Nam, C.H.; Kim, J.H.; Hur, J.K.; Lee, K.Y.; Park, J.S.; Kim, H.M.; Kang, J.H. Immunogenicity and safety of diphtheria–tetanus vaccine in pre-adolescent and adolescent South Koreans. Vaccine 2009, 27, 3209–3212. [Google Scholar] [CrossRef] [PubMed]
- Stefanizzi, P.; Bianchi, F.P.; Spinelli, G.; Amoruso, F.; Ancona, D.; Stella, P.; Tafuri, S. Postmarketing surveillance of adverse events following meningococcal B vaccination: Data from Apulia Region, 2014–2019. Hum. Vaccines Immunother. 2021, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Phillips, A.; Hickie, M.; Totterdell, J.; Brotherton, J.; Dey, A.; Hill, R.; Snelling, T.; Macartney, K. Adverse events following HPV vaccination: 11 years of surveillance in Australia. Vaccine 2020, 38, 6038–6046. [Google Scholar] [CrossRef] [PubMed]
- Stefanizzi, P.; De Nitto, S.; Patano, F.; Bianchi, F.P.; Ferorelli, D.; Stella, P.; Ancona, D.; Bavaro, V.; Tafuri, S. Post-marketing surveillance of adverse events following measles, mumps, rubella and varicella (MMRV) vaccine: Retrospecive study in apulia region (ITALY), 2009-2017. Hum. Vaccines Immunother. 2020, 16, 1875–1883. [Google Scholar] [CrossRef]
- Tafuri, S.; Gallone, M.S.; Cappelli, M.G.; Martinelli, D.; Prato, R.; Germinario, C. Addressing the anti-vaccination movement and the role of HCWs. Vaccine 2014, 32, 4860–4865. [Google Scholar] [CrossRef]
- Faranda, D.; Alberti, T.; Arutkin, M.; Lembo, V.; Lucarini, V. Interrupting vaccination policies can greatly spread SARS-CoV-2 and enhance mortality from COVID-19 disease: The AstraZeneca case for France and Italy. Chaos: Interdiscip. J. Nonlinear Sci. 2021, 31, 041105. [Google Scholar] [CrossRef]
- Pomara, C.; Sessa, F.; Ciaccio, M.; Dieli, F.; Esposito, M.; Giammanco, G.; Garozzo, S.; Giarratano, A.; Prati, D.; Rappa, F.; et al. COVID-19 Vaccine and Death: Causality Algorithm According to the WHO Eligibility Diagnosis. Diagnostics 2021, 11, 955. [Google Scholar] [CrossRef]
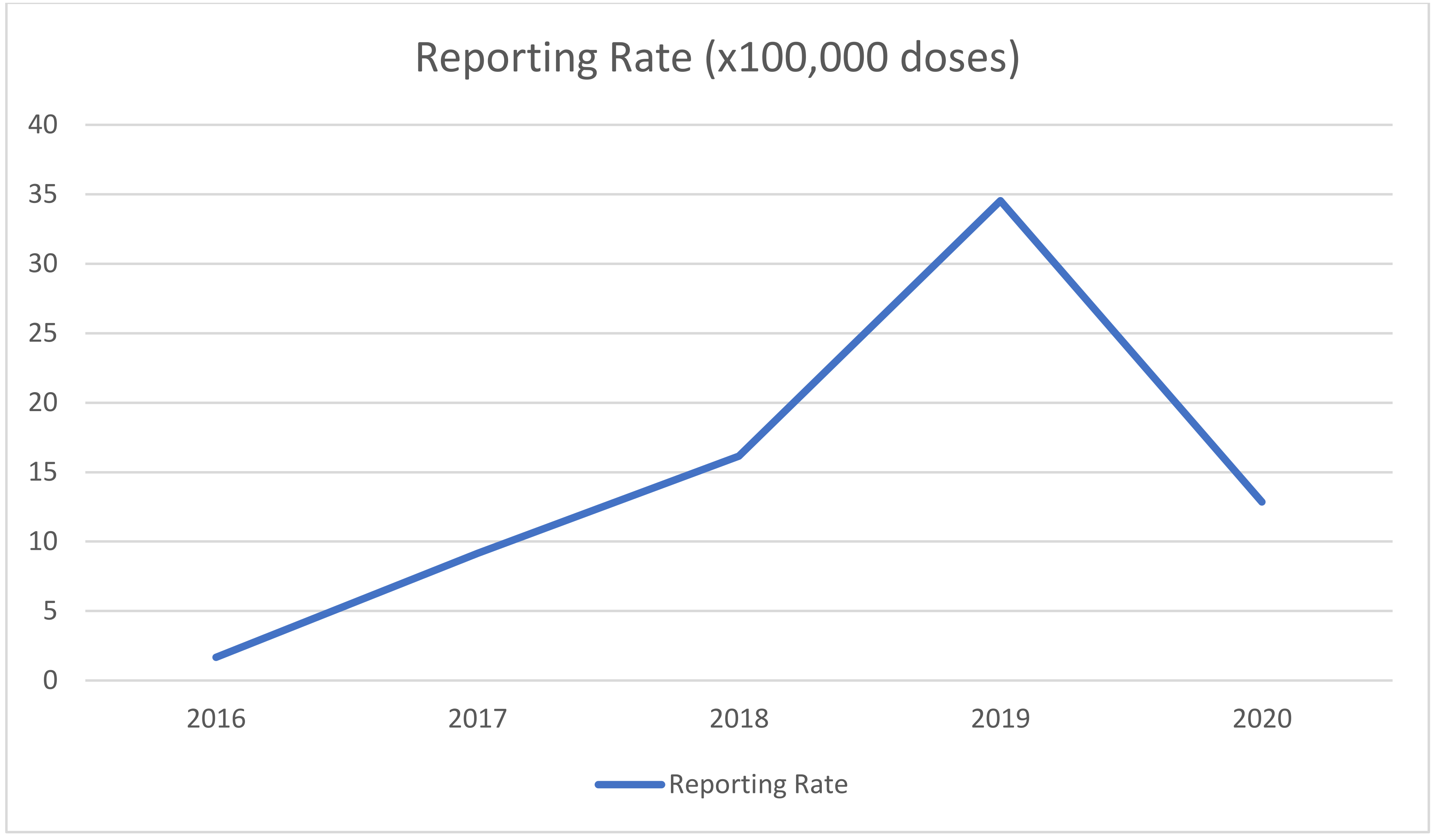
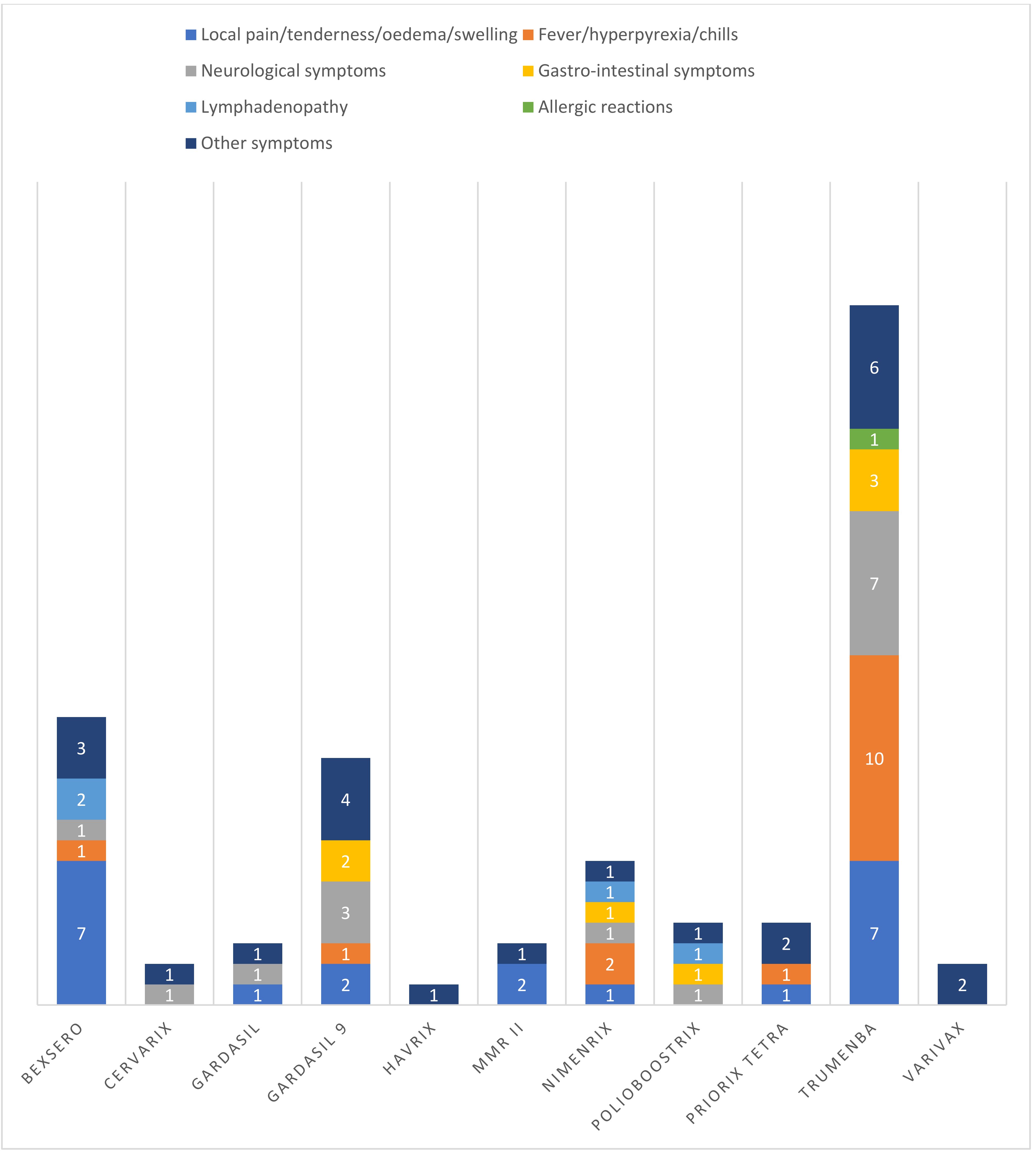
| Vaccine | 2016 | 2017 | 2018 | 2019 | 2020 | Total |
|---|---|---|---|---|---|---|
| DTaP (Boostrix) | 283 | 945 | 1962 | 65 | 5 | 3260 |
| DTaP-IPV (Polioboostrix) | 13,008 | 19,882 | 17,632 | 12,243 | 11,339 | 74,104 |
| HAV (Havrix) | 113 | 119 | 638 | 460 | 243 | 1573 |
| HPV2 (Cervarix) | 16,269 | 21,634 | 3140 | 245 | 18 | 41,306 |
| HPV4 (Gardasil) | 21,815 | 1950 | 511 | 95 | 8 | 24,379 |
| HPV9 (Gardasil 9) | 1 | 476 | 23,588 | 26,943 | 19,311 | 70,319 |
| Men B (Bexsero) | 1249 | 15,798 | 20,474 | 4351 | 836 | 42,708 |
| MenACYW (Nimenrix) | 1754 | 623 | 1277 | 8911 | 9566 | 22,131 |
| MenB (Trumenba) | 0 | 0 | 620 | 14,510 | 12,383 | 27,513 |
| MMR II (MMR II) | 5 | 15 | 9 | 3 | 2 | 34 |
| MMR (Priorix) | 466 | 56 | 14 | 9 | 3 | 548 |
| MMRV (Priorix Tetra) | 612 | 932 | 1070 | 45 | 0 | 2659 |
| Varicella (Varilrix) | 1715 | 39 | 9 | 656 | 691 | 3110 |
| Varicella (Varivax) | 2611 | 3074 | 3338 | 951 | 9 | 9983 |
| Total | 59,901 | 65,543 | 74,282 | 69,487 | 54,414 | 323,627 |
| Vaccine | Number of AEFIs | Reporting Rate (×100,000 Doses) |
|---|---|---|
| Bexsero | 9 | 21.1 |
| Cervarix | 1 | 2.42 |
| Gardasil | 2 | 8.20 |
| Gardasil 9 | 7 | 9.95 |
| Havrix | 1 | 63.6 |
| MMR II | 2 | 5882.35 |
| Nimenrix | 2 | 9.04 |
| Polioboostrix | 3 | 4.05 |
| Priorix Tetra | 2 | 75.2 |
| Trumenba | 19 | 69.1 |
| Varivax | 2 | 20.0 |
| Signs/Symptoms | N° | % (Out of 50 AEFIs) | Reporting Rate (×100,000 Doses) |
|---|---|---|---|
| Local pain/tenderness/oedema/swelling | 21 | 42.0 | 6.49 |
| Fever/hyperpyrexia/chills | 16 | 32.0 | 4.94 |
| Neurological symptoms | 15 | 30.0 | 4.63 |
| Gastro-intestinal symptoms | 7 | 14.0 | 2.16 |
| Lymphadenopathy | 4 | 8.00 | 1.24 |
| Allergic reactions | 1 | 2.0 | 0.309 |
| Other symptoms | 21 | 42.0 | 6.49 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Lorenzo, A.; Bianchi, F.P.; Martinelli, A.; Lattanzio, S.; Carbonara, A.; Diella, G.; Tafuri, S.; Stefanizzi, P. Safety Profile of Recommended Vaccinations in Adolescents: Data from Surveillance of Adverse Events Following Immunization in Puglia (Italy), 2016–2020. Vaccines 2021, 9, 1302. https://doi.org/10.3390/vaccines9111302
Di Lorenzo A, Bianchi FP, Martinelli A, Lattanzio S, Carbonara A, Diella G, Tafuri S, Stefanizzi P. Safety Profile of Recommended Vaccinations in Adolescents: Data from Surveillance of Adverse Events Following Immunization in Puglia (Italy), 2016–2020. Vaccines. 2021; 9(11):1302. https://doi.org/10.3390/vaccines9111302
Chicago/Turabian StyleDi Lorenzo, Antonio, Francesco Paolo Bianchi, Andrea Martinelli, Sabrina Lattanzio, Antonella Carbonara, Giusy Diella, Silvio Tafuri, and Pasquale Stefanizzi. 2021. "Safety Profile of Recommended Vaccinations in Adolescents: Data from Surveillance of Adverse Events Following Immunization in Puglia (Italy), 2016–2020" Vaccines 9, no. 11: 1302. https://doi.org/10.3390/vaccines9111302
APA StyleDi Lorenzo, A., Bianchi, F. P., Martinelli, A., Lattanzio, S., Carbonara, A., Diella, G., Tafuri, S., & Stefanizzi, P. (2021). Safety Profile of Recommended Vaccinations in Adolescents: Data from Surveillance of Adverse Events Following Immunization in Puglia (Italy), 2016–2020. Vaccines, 9(11), 1302. https://doi.org/10.3390/vaccines9111302








