Humoral Response to SARS-CoV-2 Vaccine of a Patient Receiving Methotrexate Treatment and Implications for the Need of Monitoring
Abstract
:1. Introduction
2. Case Report
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organization (WHO) COVID-19 Vaccines. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/covid-19-vaccines (accessed on 11 August 2021).
- Yousaf, A.; Gayam, S.; Feldman, S.; Zinn, Z.; Kolodney, M. Clinical outcomes of COVID-19 in patients taking tumor necrosis factor inhibitors or methotrexate: A multicenter research network study. J. Am. Acad. Dermatol. 2021, 84, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Winthrop, K.L.; E Brunton, A.; Beekmann, S.; Polgreen, P.; Baddley, J.; Saag, K.G.; Calabrese, C.; Calabrese, L.; Robinson, P.C.; Wallace, Z.S.; et al. SARS CoV-2 infection among patients using immunomodulatory therapies. Ann. Rheum. Dis. 2021, 80, 269–271. [Google Scholar] [CrossRef] [PubMed]
- Methotrexate and Glucocorticoids, but Not Anticytokine Therapy, Impair the Immunogenicity of A Single Dose of the BNT162b2 mRNA COVID-19 Vaccine in Patients with Chronic Inflammatory Arthritis. Available online: https://ard.bmj.com/content/annrheumdis/early/2021/06/25/annrheumdis-2021-220862.full.pdf (accessed on 11 September 2021).
- Furer, V.; Eviatar, T.; Zisman, D.; Peleg, H.; Paran, D.; Levartovsky, D.; Zisapel, M.; Elalouf, O.; Kaufman, I.; Meidan, R.; et al. Immunogenicity and safety of the BNT162b2 mRNA COVID-19 vaccine in adult patients with autoimmune inflammatory rheumatic diseases and in the general population: A multicentre study. Ann. Rheum. Dis. 2021, 80, 1330–1338. [Google Scholar] [CrossRef] [PubMed]
- Haberman, R.H.; Herati, R.; Simon, D.; Samanovic, M.; Blank, R.B.; Tuen, M.; Koralov, S.B.; Atreya, R.; Tascilar, K.; Allen, J.R.; et al. Methotrexate hampers immunogenicity to BNT162b2 mRNA COVID-19 vaccine in immune-mediated inflammatory disease. Ann. Rheum. Dis. 2021, 80, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Mahil, S.K.; Bechman, K.; Raharja, A.; Domingo-Vila, C.; Baudry, D.; A Brown, M.; Cope, A.P.; Dasandi, T.; Graham, C.; Lechmere, T.; et al. The effect of methotrexate and targeted immunosuppression on humoral and cellular immune responses to the COVID-19 vaccine BNT162b2: A cohort study. Lancet Rheumatol. 2021, 3, e627–e637. [Google Scholar] [CrossRef]
- Roche Diagnostic Elecsys Anti-SARS-CoV-2 S Assay Insert. Available online: https://www.fda.gov/media/144037/download (accessed on 11 August 2021).
- DiaSorin LIAISON® SARS-CoV-2 TrimericS IgG Assay Brochure. Available online: https://www.diasorin.com/sites/default/files/allegati_prodotti/liaisonr_sars-cov-2_trimerics_igg_assay_m0870004408_a_lr.pdf (accessed on 11 August 2021).
- Lukaszuk, K.; Kiewisz, J.; Rozanska, K.; Dabrowska, M.; Podolak, A.; Jakiel, G.; Woclawek-potocka, I.; Lukaszuk, A.; Rabalski, L. Usefulness of IVD kits for the assessment of SARS-CoV-2 antibodies to evaluate the humoral response to vaccination. Vaccines 2021, 2, 1–12. [Google Scholar]
- Subesinghe, S.; Bechman, K.; Rutherford, A.I.; Goldblatt, D.; Galloway, J.B. A systematic review and metaanalysis of antirheumatic drugs and vaccine immunogenicity in rheumatoid arthritis. J. Rheumatol. 2018, 45, 733–744. [Google Scholar] [CrossRef] [PubMed]
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Marc, G.P.; Moreira, E.D.; Zerbini, C.; et al. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef] [PubMed]
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Diemert, D.; Spector, S.A.; Rouphael, N.; Creech, C.B.; et al. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N. Engl. J. Med. 2021, 384, 403–416. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, A.; Bozzi, G.; Ungaro, R.; Villa, S.; Castelli, V.; Mangioni, D.; Muscatello, A.; Gori, A.; Bandera, A. Mini review immunological consequences of immunization with COVID-19 mRNA vaccines: Preliminary results. Front. Immunol. 2021, 12, 657711. [Google Scholar] [CrossRef] [PubMed]
- Turner, J.S.; O’Halloran, J.A.; Kalaidina, E.; Kim, W.; Schmitz, A.J.; Zhou, J.Q.; Lei, T.; Thapa, M.; Chen, R.E.; Case, J.B.; et al. SARS-CoV-2 mRNA vaccines induce persistent human germinal centre responses. Nature 2021, 596, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Favresse, J.; Gillot, C.; Di Chiaro, L.; Eucher, C.; Elsen, M.; Van Eeckhoudt, S.; David, C.; Morimont, L.; Dogné, J.-M.; Douxfils, J. Neutralizingantibodies in COVID-19 patients and vaccine recipients after two doses of BNT162b2. Viruses 2021, 13, 1364. [Google Scholar] [CrossRef] [PubMed]
- American College of Rheumatology ACR Announcements: Coronavirus Disease (COVID-19). Available online: https://www.rheumatology.org/Announcements (accessed on 12 August 2021).
- British Society for Rheumatology COVID-19 Guidance. Available online: https://www.rheumatology.org.uk/practice-quality/covid-19-guidance (accessed on 12 August 2021).
- Canadian Rheumatology Association COVID-19 Resource Centre. Available online: https://rheum.ca/covid19/ (accessed on 12 August 2021).
- Available online: https://asm.org/Articles/2021/August/How-Effective-Are-COVID-19-Vaccines-in-Immunocompr (accessed on 1 October 2021).
- Haidar, G.; Agha, M.; Lukanski, A.; Linstrum, K.; Troyan, R.; Bilderback, A.; Rothenberger, S.; McMahon, D.K.; Crandall, M.; Enick, P.N. Immunogenicity of COVID-19 vaccination in immunocompromised patients: An observational, prospective cohort study interim analysis. medRxiv 2021. [Google Scholar] [CrossRef]
- Hall, V.G.; Ferreira, V.H.; Ku, T.; Ierullo, M.; Majchrzak-Kita, B.; Chaparro, C.; Selzner, N.; Schiff, J.; McDonald, M.; Tomlinson, G.; et al. Randomized Trial of a Third Dose of mRNA-1273 Vaccine in Transplant Recipients. N. Engl. J. Med. 2021, 385, 13. [Google Scholar] [CrossRef] [PubMed]
- Planas, D.; Bruel, T.; Grzelak, L.; Guivel-Benhassine, F.; Staropoli, I.; Porrot, F.; Planchais, C.; Buchrieser, J.; Rajah, M.M.; Bishop, E.; et al. Sensitivity of infectious SARS-CoV-2 B.1.1.7 and B.1.351 variants to neutralizing antibodies. Nat. Med. 2021, 27, 917–924. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Beltran, W.F.; Lam, E.C.; Denis, K.S.; Nitido, A.D.; Garcia, Z.H.; Hauser, B.M.; Feldman, J.; Pavlovic, M.N.; Gregory, D.J.; Poznansky, M.C.; et al. Multiple SARS-CoV-2 variants escape neutralization by vaccine-induced humoral immunity. Cell 2021, 184, 2372–2383.e9. [Google Scholar] [CrossRef] [PubMed]
- Mihai, C.; Dobrota, R.; Schröder, M.; Garaiman, A.; Jordan, S.; Becker, M.O.; Maurer, B.; Distler, O. COVID-19 in a patient with systemic sclerosis treated with tocilizumab for SSc-ILD. Ann. Rheum. Dis. 2020, 79, 668–669. [Google Scholar] [CrossRef] [PubMed]
- Connolly, C.M.; Paik, J.J. Impact of methotrexate on first-dose COVID-19 mRNA vaccination. Lancet Rheumatol. 2021, 3, e607–e609. [Google Scholar] [CrossRef]
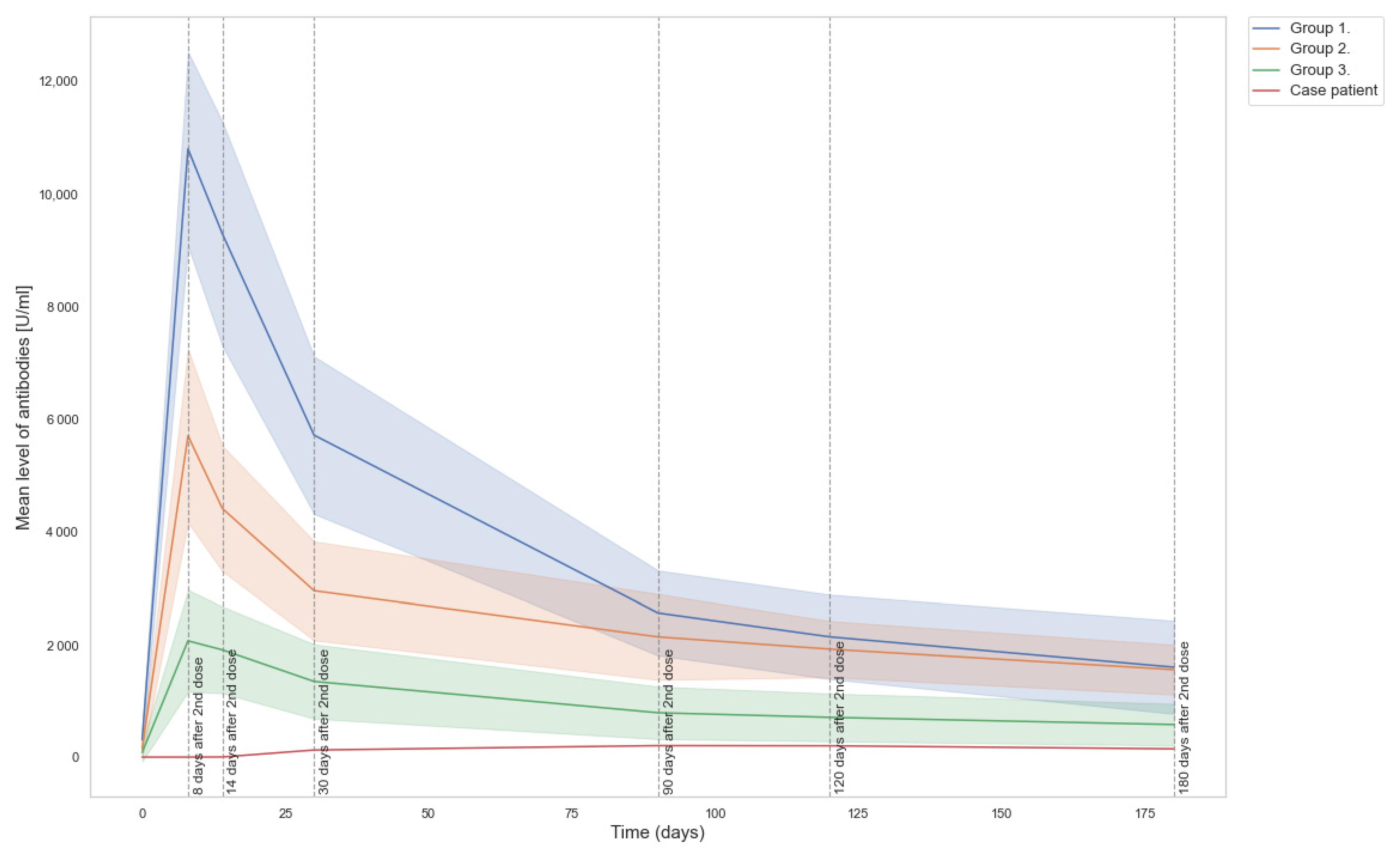
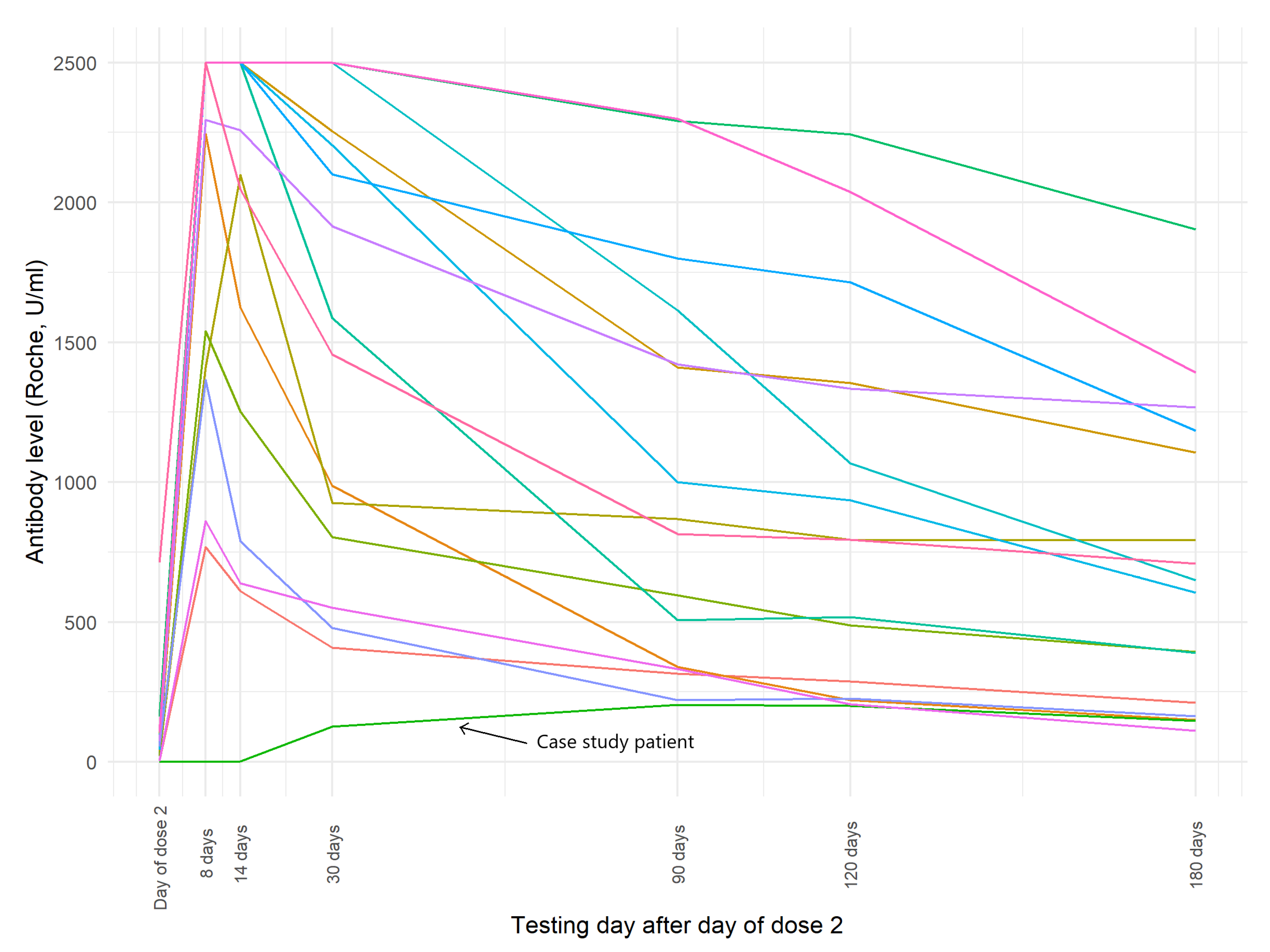
| Day | Roche U/mL | Roche BAU/mL | DiaSorin AU/mL | DiaSorin BAU/mL |
|---|---|---|---|---|
| 21 days after dose 1 | 0 | 0 | 5.71 | 14.846 |
| 8 days after dose 2 | 0.595 | 0.612 | 17.7 | 46.02 |
| 14 days after dose 2 | 2.87 | 2.953 | 34 | 88.4 |
| 30 days after dose 2 | 125.8 | 129.424 | 98.3 | 255.58 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lukaszuk, K.; Woclawek-Potocka, I.; Jakiel, G.; Malinowska, P.; Wdowiak, A.; Rozanska, K.; Rabalski, L. Humoral Response to SARS-CoV-2 Vaccine of a Patient Receiving Methotrexate Treatment and Implications for the Need of Monitoring. Vaccines 2021, 9, 1151. https://doi.org/10.3390/vaccines9101151
Lukaszuk K, Woclawek-Potocka I, Jakiel G, Malinowska P, Wdowiak A, Rozanska K, Rabalski L. Humoral Response to SARS-CoV-2 Vaccine of a Patient Receiving Methotrexate Treatment and Implications for the Need of Monitoring. Vaccines. 2021; 9(10):1151. https://doi.org/10.3390/vaccines9101151
Chicago/Turabian StyleLukaszuk, Krzysztof, Izabela Woclawek-Potocka, Grzegorz Jakiel, Paulina Malinowska, Artur Wdowiak, Karolina Rozanska, and Lukasz Rabalski. 2021. "Humoral Response to SARS-CoV-2 Vaccine of a Patient Receiving Methotrexate Treatment and Implications for the Need of Monitoring" Vaccines 9, no. 10: 1151. https://doi.org/10.3390/vaccines9101151
APA StyleLukaszuk, K., Woclawek-Potocka, I., Jakiel, G., Malinowska, P., Wdowiak, A., Rozanska, K., & Rabalski, L. (2021). Humoral Response to SARS-CoV-2 Vaccine of a Patient Receiving Methotrexate Treatment and Implications for the Need of Monitoring. Vaccines, 9(10), 1151. https://doi.org/10.3390/vaccines9101151






