Thymus Gland: A Double Edge Sword for Coronaviruses
Abstract
1. Introduction
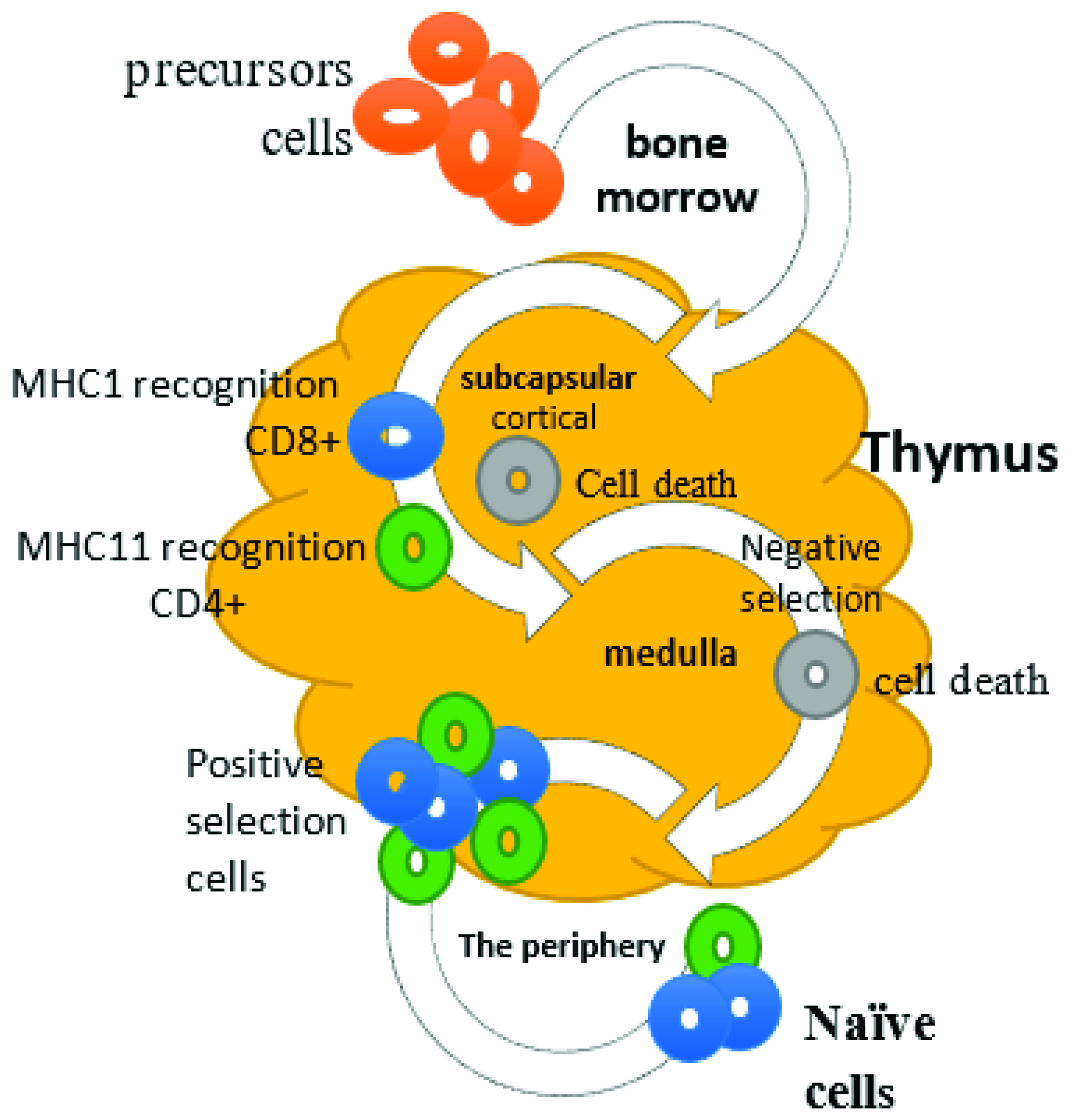
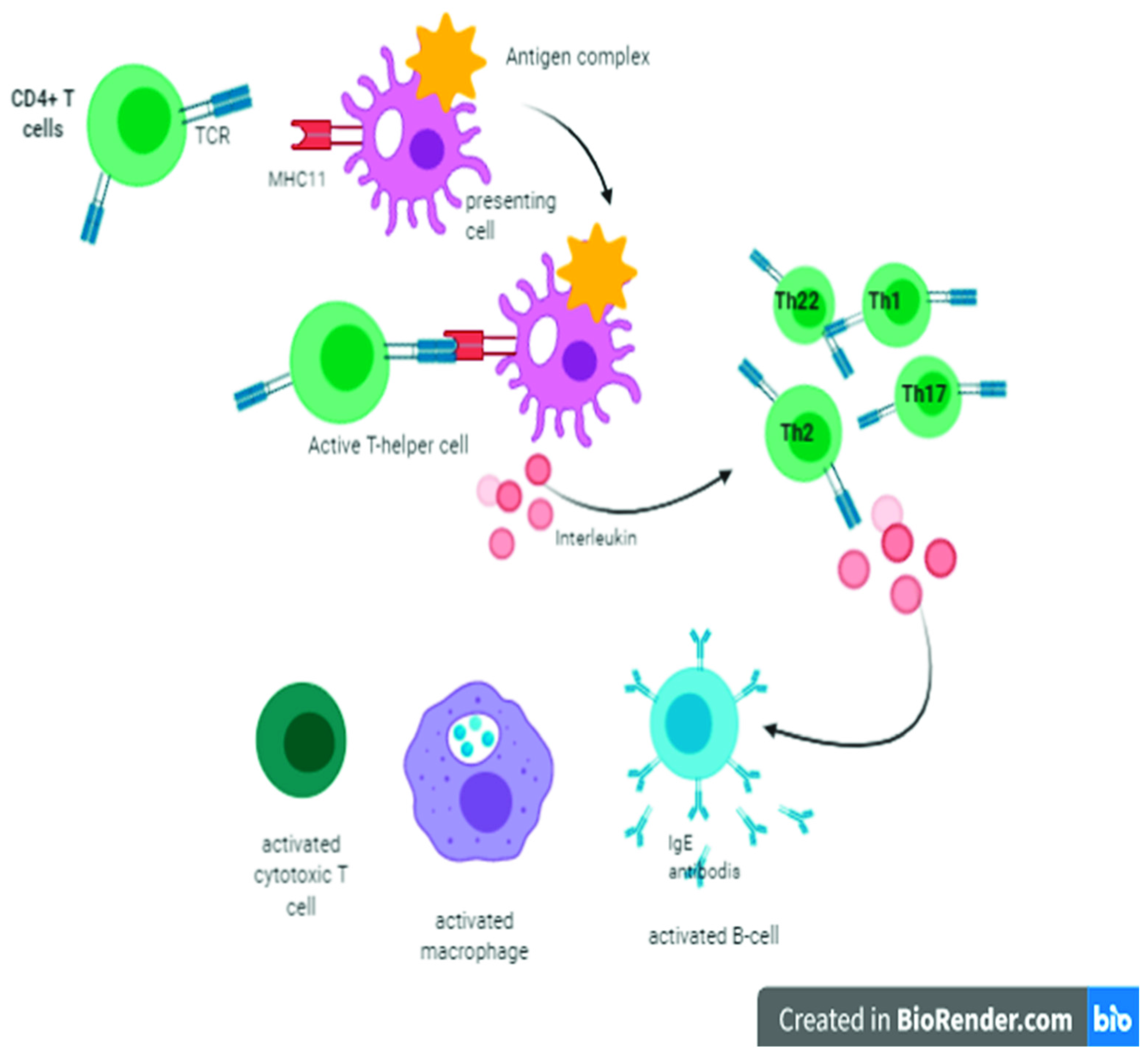
2. Cellular Immunity and Role of T Lymphocytes
3. Function of Hassall’s Corpuscles in Viral Infection
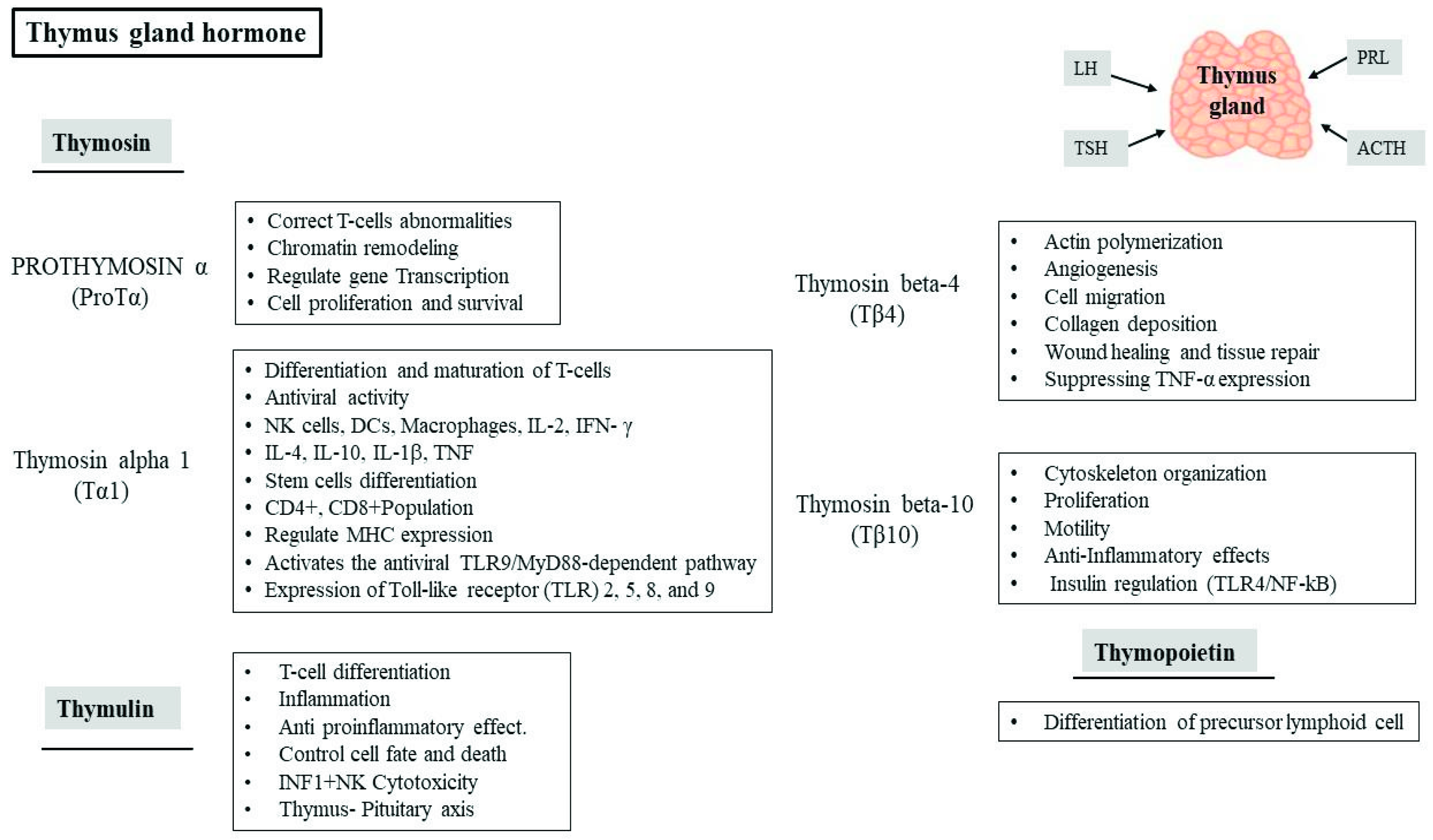
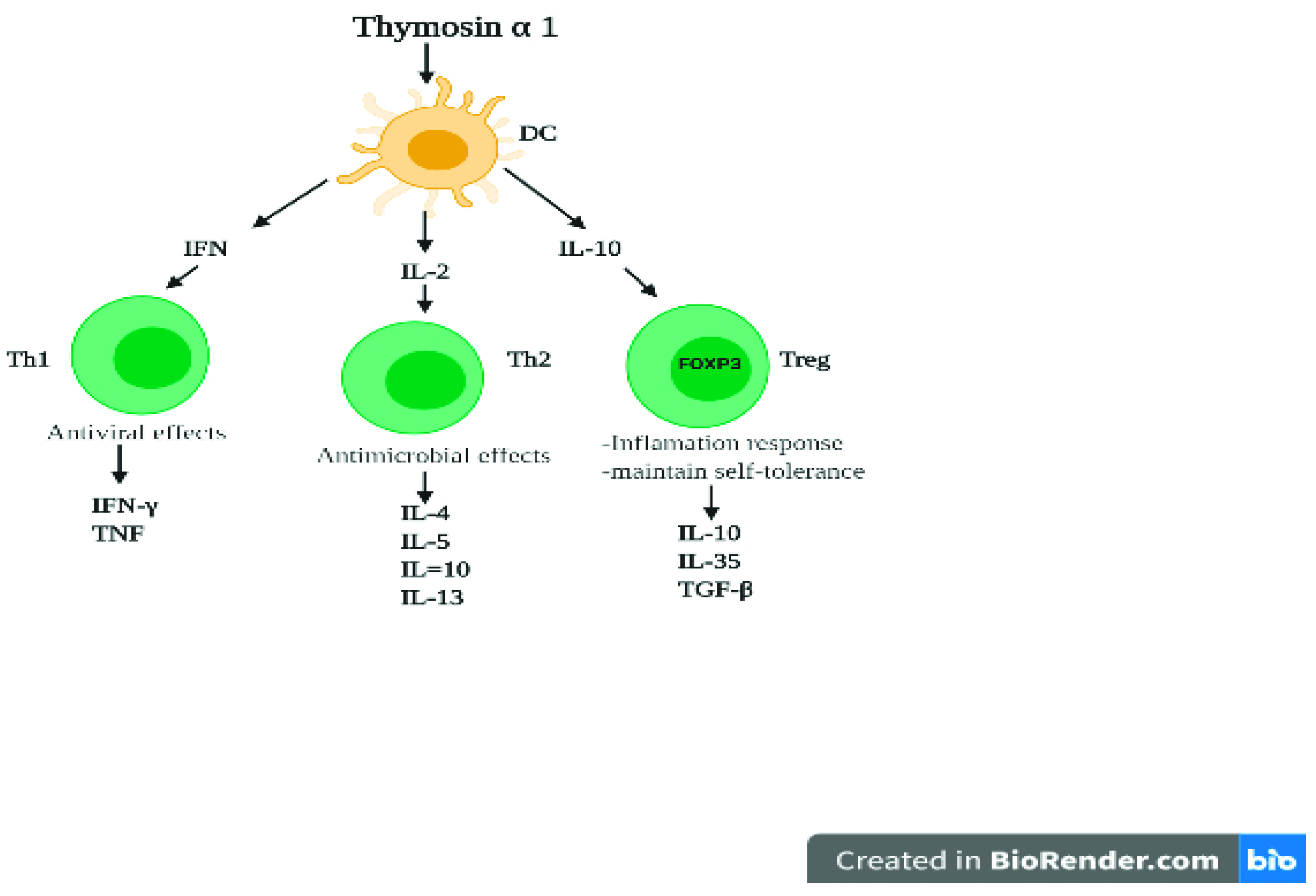
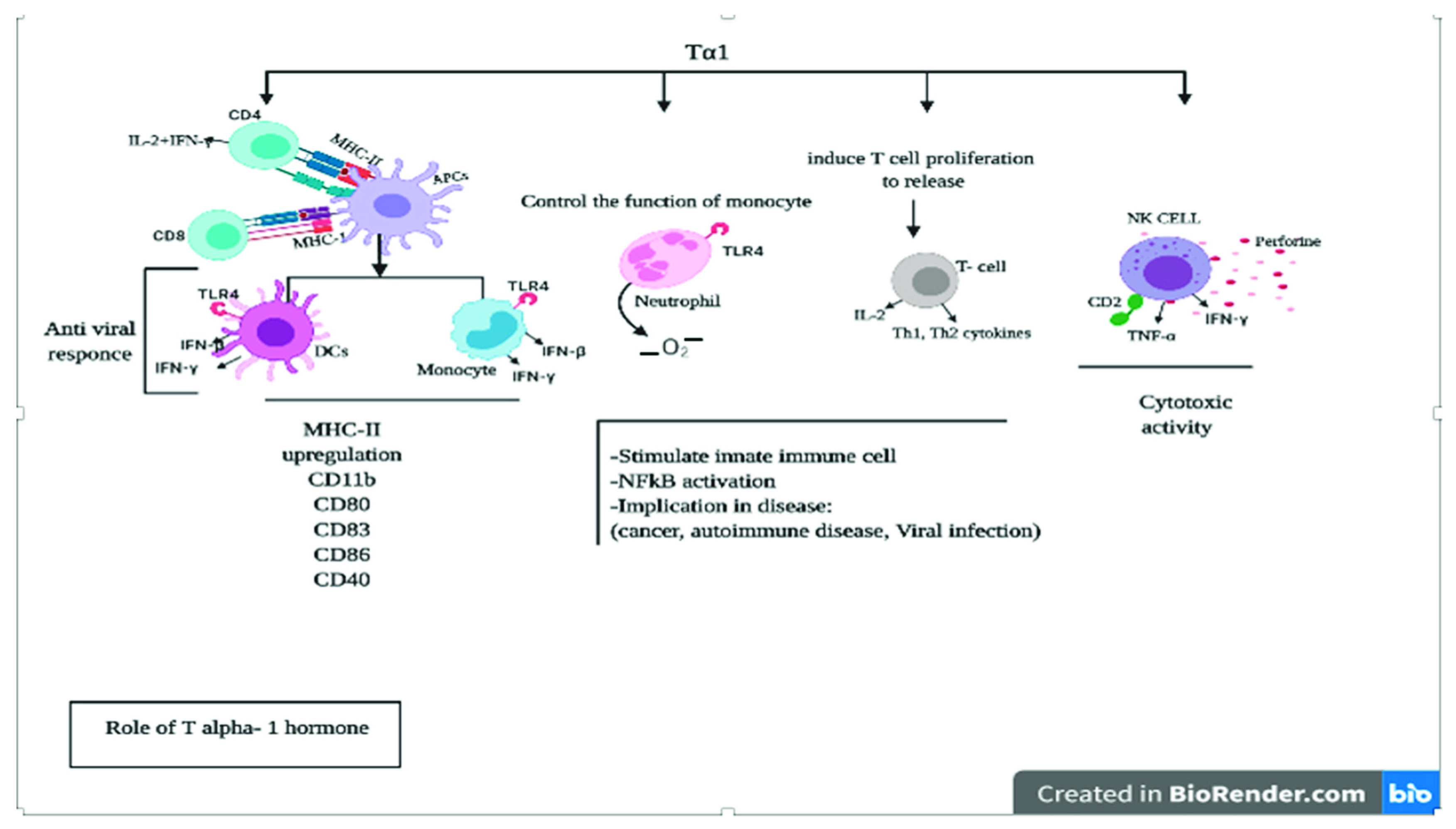
4. Role of Thymic Hormones in Viral Infection
5. Cytokines Activity against Coronaviruses
6. Role of the Thymus in Humoral Immunity against Coronavirus
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Principi, N.; Bosis, S.; Esposito, S. Effects of Coronavirus Infections in Children. Emerg. Infect. Dis. 2010, 16, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Abdul-Rasool, S.; Fielding, B.C. Understanding human coronavirus HCoV-NL63. Open Virol. J. 2010, 4, 76. [Google Scholar] [CrossRef] [PubMed]
- Meo, S.A.; Alhowikan, A.M.; Al-Khlaiwi, T.; Meo, I.M.; Halepoto, D.M.; Iqbal, M.; Usmani, A.M.; Hajjar, W.; Ahmed, N. Novel coronavirus 2019-nCoV: Prevalence, biological and clinical characteristics comparison with SARS-CoV and MERS-CoV. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 2012–2019. [Google Scholar] [PubMed]
- Prompetchara, E.; Ketloy, C.; Palaga, T. Immune responses in COVID-19 and potential vaccines: Lessons learned from SARS and MERS epidemic. Asian Pac. J. Allergy Immunol. 2020, 38, 1–9. [Google Scholar]
- Cao, M.; Zhang, D.; Wang, Y.; Lu, Y.; Zhu, X.; Li, Y.; Xue, H.; Lin, Y.; Zhang, M.; Sun, Y.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar]
- Drljača, J.N.; Vejnović, A.-M.T.; Miljković, D.M.; Popović, M.J.; Rakić, D.B.; Sekulić, S.R.; Čapo, I.Đ.; Petković, B.B. Changes in mouse thymus after exposure to tube-restraint stress. Arch. Biol. Sci. 2020, 72, 5–11. [Google Scholar] [CrossRef]
- Young, B.; O’Dowd, G.; Woodford, P. Wheater’s Functional Histology: A Text and Colour Atlas, 6th ed.; Elsevier: Philadelphia, PA, USA, 2013; pp. 204–206. ISBN 9780702047473. [Google Scholar]
- Miller, J. The early work on the discovery of the function of the thymus, an interview with Jacques Miller. Cell Death Differ. 2019, 27, 396–401. [Google Scholar] [CrossRef]
- Rousseau, M.A.; Chindelevitch, L.; An, G.; Hu, L.; Thareja, R.; Stephens, D.; Rish, i. Understanding Thethymus with Applications to Covid-19 Pathophysiology and Susceptibility with Potential Therapeutics. 2020. Available online: https://www.researchgate.net/publication/342882478_UNDERSTANDING_THE_THYMUS_WITH_APPLICATIONS_TO_SARS-COV-2_PATHOPHYSIOLOGY_AND_SUSCEPTIBILITY_WITH_POTENTIAL_THERAPEUTICS_A_PREPRINT#fullTextFileContent (accessed on 29 July 2020).
- Rehman, S.; Majeed, T.; Ansari, M.A.; Ali, U.; Sabit, H.; Al-Suhaimi, E.A. Current scenario of COVID-19 in pediatric age group and physiology of immune and thymus response. Saudi J. Biol. Sci. 2020, 27, 2567–2573. [Google Scholar] [CrossRef]
- Thapa, P.; Farber, D.L. The Role of the Thymus in the Immune Response. Thorac. Surg. Clin. 2019, 29, 123–131. [Google Scholar] [CrossRef]
- Naparstek, Y.; Holoshitz, J.; Eisenstein, S.; Reshef, T.; Rappaport, S.; Chemke, J.; Ben-Nun, A.; Cohen, I.R. Effector T lymphocyte line cells migrate to the thymus and persist there. Nature 1982, 300, 262–264. [Google Scholar] [CrossRef]
- Dembic, Z. The Cytokines of the Immune System: The Role of Cytokines in Disease Related to Immune Response; Academic Press: Cambridge, MA, USA, 2015. [Google Scholar]
- Mishalani, S.H.; Lones, M.A.; Said, J.W. Multilocular thymic cyst. A novel thymic lesion associated with human immunodeficiency virus infection. Arch. Pathol. Lab. Med. 1995, 119, 467. [Google Scholar]
- Inaba, H.; Rabah, R.; Meert, K.L.; Bhambhani, K. Herpes Simplex Virus Pneumonia in a Patient with Ependymoma. J. Pediatr. Hematol. 2004, 26, 108–111. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Zhao, C.; Dong, Q.; Zhuang, H.; Song, S.; Peng, G.; Dwyer, D.E. Effects of severe acute respiratory syndrome (SARS) coronavirus infection on peripheral blood lymphocytes and their subsets. Int. J. Infect. Dis. 2005, 9, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Da Guan, W.; Mok, C.K.P.; Chen, Z.L.; Feng, L.Q.; Li, Z.T.; Huang, J.C.; Ke, C.W.; Deng, X.; Ling, Y.; Wu, S.G.; et al. Characteristics of Traveler with Middle East Respiratory Syndrome, China, 2015. Emerg. Infect. Dis. 2015, 21, 2278–2280. [Google Scholar] [CrossRef] [PubMed]
- Alberola-Ila, J.; Hogquist, K.; Swan, K.A.; Bevan, M.J.; Perlmutter, R.M. Positive and negative selection invoke distinct signaling pathways. J. Exp. Med. 1996, 184, 9–18. [Google Scholar] [CrossRef]
- Abbas, A.K.; Lichtman, A.H.; Pillai, S. Cellular and Molecular Immunology; Elsevier Health Sciences: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Budd, R.C.; Fortner, K.A. T lymphocytes. In Kelley and Firestein’s Textbook of Rheumatology; Elsevier: Amsterdam, The Netherlands, 2017; pp. 189–206. [Google Scholar]
- Rich, R.R.; Fleisher, T.A.; Shearer, W.T.; Schroeder, H.W., Jr.; Frew, A.J.; Weyand, C.M. Clinical Immunology E-Book: Principles and Practice; Elsevier Health Sciences: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Maddur, M.S.; Bayry, J. B cells drive Th2 responses by instructing human dendritic cell maturation. OncoImmunology 2015, 4, e1005508. [Google Scholar] [CrossRef]
- Rathore, J.S.; Wang, Y. Protective role of Th17 cells in pulmonary infection. Vaccine 2016, 34, 1504–1514. [Google Scholar] [CrossRef]
- Le Rouzic, O.; Pichavant, M.; Frealle, E.; Guillon, A.; Si-Tahar, M.; Gosset, P. Th17 cytokines: Novel potential therapeutic targets for COPD pathogenesis and exacerbations. Eur. Respir. J. 2017, 50, 1602434. [Google Scholar] [CrossRef]
- Hotez, P.J.; Bottazzi, M.E.; Corry, D.B. The potential role of Th17 immune responses in coronavirus immunopathology and vaccine-induced immune enhancement. Microbes Infect. 2020, 22, 165–167. [Google Scholar] [CrossRef]
- Chen, X.; Du, Y.; Lin, X.; Qian, Y.; Zhou, T.; Huang, Z. CD4+ CD25+ regulatory T cells in tumor immunity. Int. Immunopharmacol. 2016, 34, 244–249. [Google Scholar] [CrossRef]
- Swerdlow, S.H.; Jaffe, E.S.; Brousset, P.; Chan, J.K.; de Leval, L.; Gaulard, P.; Harris, N.L.; Pileri, S.; Weiss, L.M. Cytotoxic T-cell and NK-cell Lymphomas. Am. J. Surg. Pathol. 2014, 38, e60–e71. [Google Scholar] [CrossRef]
- Liu, Q.; Sun, Z.; Chen, L. Memory T cells: Strategies for optimizing tumor immunotherapy. Protein Cell 2020, 11, 549–564. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Shi, X.; Chen, H.; Zhang, S.; Salker, M.S.; Mack, A.F.; Föller, M.; Mak, T.W.; Singh, Y.; Lang, F. DJ-1/Park7 Sensitive Na+/H+Exchanger 1 (NHE1) in CD4+T Cells. J. Cell. Physiol. 2016, 232, 3050–3059. [Google Scholar] [CrossRef]
- Fitzgerald, K.A.; O’Neill, L.A.; Gearing, A.J.; Callard, R.E. The Cytokine FactsBook and Webfacts, 2nd ed.; Academic Press: Cambridge, MA, USA, 2001. [Google Scholar] [CrossRef]
- Wu, H.-X.; Guo, P.-F.; Jin, L.-P.; Liang, S.-S.; Li, D.-J. Functional regulation of thymic stromal lymphopoietin on proliferation and invasion of trophoblasts in human first-trimester pregnancy. Hum. Reprod. 2010, 25, 1146–1152. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sakaguchi, S.; Miyara, M.; Costantino, C.; Hafler, D.A. FOXP3+ regulatory T cells in the human immune system. Nat. Rev. Immunol. 2010, 10, 490–500. [Google Scholar] [CrossRef] [PubMed]
- Le, P.T.; Lazorick, S.; Whichard, L.P.; Haynes, B.F.; Singer, K.H. Regulation of cytokine production in the human thymus: Epidermal growth factor and transforming growth factor alpha regulate mRNA levels of interleukin 1 alpha (IL-1 alpha), IL-1 beta, and IL-6 in human thymic epithelial cells at a post-transcriptional level. J. Exp. Med. 1991, 174, 1147–1157. [Google Scholar] [CrossRef] [PubMed]
- Odaka, C.; Hauri-Hohl, M.; Takizawa, K.; Nishikawa, Y.; Yano, M.; Matsumoto, M.; Boyd, R.; Holländer, G.A. TGF-β type II receptor expression in thymic epithelial cells inhibits the development of Hassall’s corpuscles in mice. Int. Immunol. 2013, 25, 633–642. [Google Scholar] [CrossRef]
- Wang, J.; Wang, Q.; Han, T.; Li, Y.-K.; Zhu, S.-L.; Ao, F.; Feng, J.; Jing, M.-Z.; Wang, L.; Ye, L.-B.; et al. Soluble interleukin-6 receptor is elevated during influenza A virus infection and mediates the IL-6 and IL-32 inflammatory cytokine burst. Cell. Mol. Immunol. 2014, 12, 633–644. [Google Scholar] [CrossRef] [PubMed]
- Henry, L.; Anderson, G. Immunoglobulins in Hassall’s corpuscles of the human thymus. J. Anat. 1990, 168, 185–197. [Google Scholar] [PubMed]
- Schmitz, N.; Kurrer, M.; Bachmann, M.F.; Kopf, M. Interleukin-1 Is Responsible for Acute Lung Immunopathology but Increases Survival of Respiratory Influenza Virus Infection. J. Virol. 2005, 79, 6441–6448. [Google Scholar] [CrossRef]
- Longhi, P.; Wright, K.; Lauder, S.N.; Nowell, M.; Jones, G.; Godkin, A.; Jones, S.A.; Gallimore, A.M. Interleukin-6 Is Crucial for Recall of Influenza-Specific Memory CD4+ T Cells. PLoS Pathog. 2008, 4, e1000006. [Google Scholar] [CrossRef] [PubMed]
- Paquette, S.G.; Banner, D.; Zhao, Z.; Fang, Y.; Huang, S.S.H.; Leόn, A.J.; Ng, D.C.K.; Almansa, R.; Martin-Loeches, I.; Ramírez, P.; et al. Interleukin-6 Is a Potential Biomarker for Severe Pandemic H1N1 Influenza A Infection. PLoS ONE 2012, 7, e38214. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Zhang, X.; Deng, W.; Liu, J.; Li, H.; Wen, M.; Bao, L.; Qu, J.; Liu, Y.; Li, F.; et al. Severe influenza A(H1N1)pdm09 infection induces thymic atrophy through activating innate CD8+CD44hi T cells by upregulating IFN-γ. Cell Death Dis. 2014, 5, e1440. [Google Scholar] [CrossRef]
- Velazquez-Salinas, L.; Verdugo-Rodriguez, A.; Rodriguez, L.L.; Borca, M.V. The Role of Interleukin 6 During Viral Infections. Front. Microbiol. 2019, 10, 1057. [Google Scholar] [CrossRef] [PubMed]
- Edston, E. Thymus Involution and Intravenous Drug Abuse. Am. J. Forensic Med. Pathol. 2020, 41, 32–34. [Google Scholar] [CrossRef]
- Lunin, S.M.; Novoselova, E.G. Thymus hormones as prospective anti-inflammatory agents. Expert Opin. Ther. Targets 2010, 14, 775–786. [Google Scholar] [CrossRef]
- Goldstein, A.L. History of the Discovery of the Thymosins. Ann. N. Y. Acad. Sci. 2007, 1112, 1–13. [Google Scholar] [CrossRef]
- Rafie, C.; Campa, A.; Smith, S.; Huffman, F.; Newman, F.; Baum, M.K. Cocaine Reduces Thymic Endocrine Function: Another Mechanism for Accelerated HIV Disease Progression. AIDS Res. Hum. Retrovir. 2011, 27, 815–822. [Google Scholar] [CrossRef]
- Severa, M.; Zhang, J.; Giacomini, E.; Rizzo, F.; Etna, M.P.; Cruciani, M.; Garaci, E.; Chopp, M.; Coccia, E.M. Thymosins in multiple sclerosis and its experimental models: Moving from basic to clinical application. Mult. Scler. Relat. Disord. 2018, 27, 52–60. [Google Scholar] [CrossRef]
- Romani, L.; Bistoni, F.; Gaziano, R.; Bozza, S.; Montagnoli, C.; Perruccio, K.; Pitzurra, L.; Bellocchio, S.; Velardi, A.; Rasi, G.; et al. Thymosin α 1 activates dendritic cells for antifungal Th1 resistance through Toll-like receptor signaling. Blood 2004, 103, 4232–4239. [Google Scholar] [CrossRef]
- Goldstein, A.L.; Badamchian, M. Thymosins: Chemistry and biological properties in health and disease. Expert Opin. Biol. Ther. 2004, 4, 559–573. [Google Scholar] [CrossRef]
- da Silva, A.L.; Martini, S.V.; Abreu, S.C.; Samary, C.d.S.; Diaz, B.L.; Fernezlian, S.; de Sá, V.K.; Capelozzi, V.L.; Boylan, N.J.; Goya, R.G.; et al. DNA nanoparticle-mediated thymulin gene therapy prevents airway remodeling in experimental allergic asthma. J. Control Release 2014, 180, 125–133. [Google Scholar] [CrossRef]
- Da Silva, A.L.; De Oliveira, G.P.; Kim, N.; Cruz, F.F.; Kitoko, J.Z.; Blanco, N.G.; Martini, S.V.; Hanes, J.; Rocco, P.R.M.; Suk, J.S.; et al. Nanoparticle-based thymulin gene therapy therapeutically reverses key pathology of experimental allergic asthma. Sci. Adv. 2020, 6, eaay7973. [Google Scholar] [CrossRef]
- Palaszynski, E.; Moody, T.; O’Donohue, T.; Goldstein, A. Thymosin α1-like peptides: Localization and biochemical characterization in the rat brain and pituitary gland. Peptides 1983, 4, 463–467. [Google Scholar] [CrossRef]
- Camerini, R.; Garaci, E. Historical review of thymosin α 1 in infectious diseases. Expert Opin. Biol. Ther. 2015, 15 (Suppl. 1), S117–S127. [Google Scholar] [CrossRef]
- Jia, R.; Liu, S.; Xu, J.; Liang, X. IL16 deficiency enhances Th1 and cytotoxic T lymphocyte response against influenza A virus infection. Biosci. Trends 2019, 13, 516–522. [Google Scholar] [CrossRef]
- Xie, M.; Chen, Q. Insight into 2019 novel coronavirus—An updated interim review and lessons from SARS-CoV and MERS-CoV. Int. J. Infect. Dis. 2020, 94, 119–124. [Google Scholar] [CrossRef] [PubMed]
- You, J.; Zhuang, L.; Cheng, H.-Y.; Yan, S.-M.; Yu, L.; Huang, J.-H.; Tang, B.-Z.; Huang, M.-L.; Ma, Y.-L.; Chongsuvivatwong, V.; et al. Efficacy of thymosin alpha-1 and interferon alpha in treatment of chronic viral hepatitis B: A randomized controlled study. World J. Gastroenterol. 2006, 12, 6715–6721. [Google Scholar] [CrossRef] [PubMed]
- Garaci, E.; Favalli, C.; Pica, F.; Vallebona, P.S.; Palamara, A.T.; Matteucci, C.; Pierimarchi, P.; Serafino, A.; Mastino, A.; Bistoni, F.; et al. Thymosin Alpha 1: From Bench to Bedside. Ann. N. Y. Acad. Sci. 2007, 1112, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Palamara, A.; Bue, M.; Savini, P. Thymosin alpha 1 inhibits Sendai virus replication: Involvement of intracellular redox state. In Proceedings of the 6th International Expert Forum of Immunotherapy and Gene Therapy, Florence, Italy, 6–8 May 1998; International Medical Press: London, UK, 1998. [Google Scholar]
- Skopeliti, M.; Iconomidou, V.A.; Derhovanessian, E.; Pawelec, G.; Voelter, W.; Kalbacher, H.; Tsitsilonis, O.E. Prothymosin α immunoactive carboxyl-terminal peptide TKKQKTDEDD stimulates lymphocyte reactions, induces dendritic cell maturation and adopts a β-sheet conformation in a sequence-specific manner. Mol. Immunol. 2009, 46, 784–792. [Google Scholar] [CrossRef]
- Bhat, M.A.; Rahman, S.; Rather, I.A.; Banday, I.; Syed, S.; Koser, H.; Kamal, M.A.; Minakshi, R.; Jan, A.T. Coronavirus Disease-2019 (COVID-19) in 2020: A Perspective Study of a Global Pandemic. Curr. Pharm. Des. 2020, 27. [Google Scholar] [CrossRef]
- Garaci, E.; Pica, F.; Serafino, A.; Balestrieri, E.; Matteucci, C.; Moroni, G.; Sorrentino, R.; Zonfrillo, M.; Pierimarchi, P.; Vallebona, P.S. Thymosin α1 and cancer: Action on immune effector and tumor target cells. Ann. N. Y. Acad. Sci. 2012, 1269, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Pica, F.; Chimenti, M.S.; Gaziano, R.; Buè, C.; Casalinuovo, I.A.; Triggianese, P.; Conigliaro, P.; Di Carlo, D.; Cordero, V.; Adorno, G.; et al. Serum thymosin α 1 levels in patients with chronic inflammatory autoimmune diseases. Clin. Exp. Immunol. 2016, 186, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Banchereau, J.; Steinman, R.M. Dendritic cells and the control of immunity. Nature 1998, 392, 245–252. [Google Scholar] [CrossRef]
- Sodhi, A.; Paul, S. Involvement of mitogen-activated protein kinases in the signal transduction pathway of bone marrow-derived macrophage activation in response to in vitro treatment with thymosin alpha 1. Int. Immunopharmacol. 2001, 2, 47–58. [Google Scholar] [CrossRef]
- Samara, P.; Ioannou, K.; Tsitsilonis, O.E. Prothymosin alpha and immune responses: Are we close to potential clinical applications? In Vitamins and Hormones; Academic Press: Cambridge, MA, USA, 2016; Volume 102, pp. 179–207. [Google Scholar]
- Yao, Q.; Doan, L.X.; Zhang, R.; Bharadwaj, U.; Li, M.; Chen, C. Thymosin-α1 modulates dendritic cell differentiation and functional maturation from human peripheral blood CD14+ monocytes. Immunol. Lett. 2007, 110, 110–120. [Google Scholar] [CrossRef]
- Zhang, P.; Chan, J.; Dragoi, A.-M.; Gong, X.; Ivanov, S.; Li, Z.-W.; Chuang, T.; Tuthill, C.; Wan, Y.; Karin, M.; et al. Activation of IKK by thymosin α1 requires the TRAF6 signalling pathway. EMBO Rep. 2005, 6, 531–537. [Google Scholar] [CrossRef]
- Tian, Y.; Grifoni, A.; Sette, A.; Weiskopf, D. Human T Cell Response to Dengue Virus Infection. Front. Immunol. 2019, 10, 2125. [Google Scholar] [CrossRef]
- Plüddemann, A.; Mukhopadhyay, S.; Gordon, S. Innate immunity to intracellular pathogens: Macrophage receptors and responses to microbial entry. Immunol. Rev. 2011, 240, 11–24. [Google Scholar] [CrossRef]
- Nowacki, T.M.; Kuerten, S.; Zhang, W.; Shive, C.L.; Kreher, C.R.; Boehm, B.O.; Lehmann, P.V.; Tary-Lehmann, M. Granzyme B production distinguishes recently activated CD8+ memory cells from resting memory cells. Cell. Immunol. 2007, 247, 36–48. [Google Scholar] [CrossRef]
- Shibabaw, T.; Molla, M.D.; Teferi, B.; Ayelign, B. Role of IFN and Complements System: Innate Immunity in SARS-CoV-2. J. Inflamm. Res. 2020, 13, 507–518. [Google Scholar] [CrossRef]
- Xia, H.; Shi, P.-Y. Antagonism of Type I Interferon by Severe Acute Respiratory Syndrome Coronavirus 2. J. Interf. Cytokine Res. 2020, 40, 543–548. [Google Scholar] [CrossRef]
- Nakhlband, A.; Fakhari, A.; Azizi, H. Interferon-beta offers promising avenues to COVID-19 treatment: A systematic review and meta-analysis of clinical trial studies. Naunyn-Schmiedeberg Arch. Pharmacol. 2021, 394, 829–838. [Google Scholar] [CrossRef]
- Aslam, M.; Zaidi, S.; Toor, R.; Gull, I.; Iqbal, M.; Abbas, Z.; Tipu, I.; Ahmed, A.; Athar, M.; Harito, C.; et al. Interferon α2–Thymosin α1 Fusion Protein (IFNα2–Tα1): A Genetically Engineered Fusion Protein with Enhanced Anticancer and Antiviral Effect. Materials 2021, 14, 3318. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Zhang, K.; Ye, Y.; Xing, J.; Wu, Y.; Ma, Y.; Li, Y. Li Effects of Castration on miRNA, lncRNA, and mRNA Profiles in Mice Thymus. Genes 2020, 11, 147. [Google Scholar] [CrossRef]
- Yan, F.; Mo, X.; Liu, J.; Ye, S.; Zeng, X.; Chen, D. Thymic function in the regulation of T cells, and molecular mechanisms underlying the modulation of cytokines and stress signaling. Mol. Med. Rep. 2017, 16, 7175–7184. [Google Scholar] [CrossRef]
- Ströher, U.; DiCaro, A.; Li, Y.; Strong, J.E.; Aoki, F.; Plummer, F.; Jones, S.M.; Feldmann, H. Severe Acute Respiratory Syndrome–Related Coronavirus Is Inhibited by Interferon-α. J. Infect. Dis. 2004, 189, 1164–1167. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.-C.; Chen, M.-Y.; Lee, W.-S.; Chang, Y.-L. Potential therapeutic agents against COVID-19: What we know so far. J. Chin. Med. Assoc. 2020, 83, 534–536. [Google Scholar] [CrossRef] [PubMed]
- Holdsworth, S.R.; Gan, P.Y. Cytokines: Names and numbers you should care about. Clin. J. Am. Soc. Nephrol. 2015, 10, 2243–2254. [Google Scholar] [CrossRef] [PubMed]
- Sainz, B., Jr.; Mossel, E.C.; Peters, C.J.; Garry, R.F. Interferon-beta and interferon-gamma synergistically inhibit the replication of severe acute respiratory syndrome-associated coronavirus (SARS-CoV). Virology 2004, 329, 11–17. [Google Scholar] [CrossRef] [PubMed]
- de Lang, A.; Osterhaus, A.; Haagmans, B.L. Interferon-γ and interleukin-4 downregulate expression of the SARS coronavirus receptor ACE2 in Vero E6 cells. Virology 2006, 353, 474–481. [Google Scholar] [CrossRef]
- Hofmann, H.; Pyrc, K.; van der Hoek, L.; Geier, M.; Berkhout, B.; Pohlmann, S. Human coronavirus NL63 employs the severe acute respiratory syndrome coronavirus receptor for cellular entry. Proc. Natl. Acad. Sci. USA 2005, 102, 7988–7993. [Google Scholar] [CrossRef] [PubMed]
- Sarzi-Puttini, P.; Giorgi, V.; Sirotti, S.; Marotto, D.; Ardizzone, S.; Rizzardini, G.; Antinori, S.; Galli, M. COVID-19, cytokines and immunosuppression: What can we learn from what can we learn from severe acute respiratory syndrome? Clin. Exp. Rheumatol. 2020, 38, 337–342. [Google Scholar] [PubMed]
- Yoshikawa, T.; Hill, T.; Li, K.; Peters, C.J.; Tseng, C.-T.K. Severe Acute Respiratory Syndrome (SARS) Coronavirus-Induced Lung Epithelial Cytokines Exacerbate SARS Pathogenesis by Modulating Intrinsic Functions of Monocyte-Derived Macrophages and Dendritic Cells. J. Virol. 2009, 83, 3039–3048. [Google Scholar] [CrossRef]
- Lo, A.W.; Tang, N.L.; To, K.-F. How the SARS coronavirus causes disease: Host or organism? J. Pathol. 2005, 208, 142–151. [Google Scholar] [CrossRef]
- Okabayashi, T.; Kariwa, H.; Yokota, S.-I.; Iki, S.; Indoh, T.; Yokosawa, N.; Takashima, I.; Tsutsumi, H.; Fujii, N. Cytokine regulation in SARS coronavirus infection compared to other respiratory virus infections. J. Med. Virol. 2006, 78, 417–424. [Google Scholar] [CrossRef]
- Law, H.K.-W.; Cheung, C.Y.; Ng, H.Y.; Sia, S.F.; Chan, Y.O.; Luk, W.; Nicholls, J.M.; Peiris, J.S.M.; Lau, Y.L. Chemokine up-regulation in SARS-coronavirus–infected, monocyte-derived human dendritic cells. Blood 2005, 106, 2366–2374. [Google Scholar] [CrossRef]
- Spencer, J.; Choy, M.; Hussell, T.; Papadaki, L.; Kington, J.P.; Isaacson, P.G. Properties of human thymic B cells. Immunology 1992, 75, 596–600. [Google Scholar]
- Isaacson, P.G.; Norton, A.J.; Addis, B.J. The human thymus contains a novel population of B lymphocytes. Lancet 1987, 2, 1488–1491. [Google Scholar] [CrossRef]
- Perera, J.; Meng, L.; Meng, F.; Huang, H. Autoreactive thymic B cells are efficient antigen-presenting cells of cognate self-antigens for T cell negative selection. Proc. Natl. Acad. Sci. USA 2013, 110, 17011–17016. [Google Scholar] [CrossRef] [PubMed]
- Yamano, T.; Nedjic, J.; Hinterberger, M.; Steinert, M.; Koser, S.; Pinto, S.; Gerdes, N.; Lutgens, E.; Ishimaru, N.; Busslinger, M.; et al. Thymic B Cells Are Licensed to Present Self Antigens for Central T Cell Tolerance Induction. Immunity 2015, 42, 1048–1061. [Google Scholar] [CrossRef]
- Lalle, M.; Minellli, M.; Tarantini, P.; Marino, M.; Cerasoli, V. Cellular and humoral immune alterations in thymectomized patients for thymoma. Ann. Hematol. Springer Verl. 2009, 88, 847–853. [Google Scholar] [CrossRef]
- Vidarsson, G.; Dekkers, G.; Rispens, T. IgG subclasses and allotypes: From structure to effector functions. Front. Immunol. 2014, 5, 520. [Google Scholar] [CrossRef]
- Frasca, D.; Diaz, A.; Romero, M.; Mendez, N.V.; Landin, A.M.; Blomberg, B.B. Effects of age on H1N1-specific serum IgG1 and IgG3 levels evaluated during the 2011–2012 influenza vaccine season. Immun. Ageing 2013, 10, 14. [Google Scholar] [CrossRef]
- Nutt, S.L.; Taubenheim, N.; Hasbold, J.; Corcoran, L.M.; Hodgkin, P.D. The genetic network controlling plasma cell differentiation. Semin. Immunol. 2011, 23, 341–349. [Google Scholar] [CrossRef]
- Reinholdt, J.; Husby, S. IgA and Mucosal Homeostasis. In Madame Curie BioScience Database; Landes Bioscience: Austin, TX, USA, 2013. [Google Scholar]
- Klimpel, G.R. Immune defenses. In Medical Microbiology, 4th ed.; University of Texas Medical Branch at Galveston: Galveston, TX, USA, 1996. [Google Scholar]
- Schroeder, H.W., Jr.; Cavacini, L. Structure and function of immunoglobulins. J. Allergy Clin. Immunol. 2010, 125, S41–S52. [Google Scholar] [CrossRef]
- Janeway, C.A.T.; Walport, M.; Shlomchik, M. Immunobiology, Part III, The Development of Mature Lymphocyte Receptor Repertoires; Garland Science: New York, NY, USA, 2001. [Google Scholar]
- Perera, J.; Huang, H. The development and function of thymic B cells. Experientia 2015, 72, 2657–2663. [Google Scholar] [CrossRef] [PubMed]
- Nuñez, S.; Moore, C.; Gao, B.; Rogers, K.; Hidalgo, Y.; del Nido, P.J.; Restaino, S.; Naka, Y.; Bhagat, G.; Madsen, J.C.; et al. The human thymus perivascular space is a functional niche for viral-specific plasma cells. Sci. Immunol. 2016, 1, eaah4447. [Google Scholar] [CrossRef] [PubMed]
- Frieman, M.; Heise, M.; Baric, R. SARS coronavirus and innate immunity. Virus Res. 2008, 133, 101–112. [Google Scholar] [CrossRef] [PubMed]
- Iwasaki, A.; Medzhitov, R. Control of adaptive immunity by the innate immune system. Nat. Immunol. 2015, 16, 343–353. [Google Scholar] [CrossRef]
- Qin, C.; Zhou, L.; Hu, Z.; Zhang, S.; Yang, S.; Tao, Y.; Xie, C.; Ma, K.; Shang, K.; Wang, W.; et al. Dysregulation of immune response in patients with COVID-19 in Wuhan, China. Clin. Infect. Dis. 2020, 71, 762–768. [Google Scholar] [CrossRef]
- Cuvelier, P.; Roux, H.; Couëdel-Courteille, A.; Dutrieux, J.; Naudin, C.; de Muylder, B.C.; Cheynier, R.; Squara, P.; Marullo, S. Protective reactive thymus hyperplasia in COVID-19 acute respiratory distress syndrome. Crit. Care 2021, 25, 4. [Google Scholar] [CrossRef]
- Minakshi, R.; Jan, A.T.; Rahman, S.; Kim, J. A Testimony of the Surgent SARS-CoV-2 in the Immunological Panorama of the Human Host. Front. Cell. Infect. Microbiol. 2020, 10, 575404. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.; Li, D.; Xue, H.; Zhang, L.; Liu, Z.; Zhang, B.; Zhang, L.; Yang, W.; Xie, B.; Duan, X.; et al. Progress and prospect on imaging diagnosis of COVID-19. Chin. J. Acad. Radiol. 2020, 3, 4–13. [Google Scholar] [CrossRef]
- Fisher, D.; Heymann, D. Q&A: The novel coronavirus outbreak causing COVID-19. BMC Med. 2020, 18, 57. [Google Scholar]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Suhaimi, E.A.; Aljafary, M.A.; Alkhulaifi, F.M.; Aldossary, H.A.; Alshammari, T.; AL-Qaaneh, A.; Aldahhan, R.; Alkhalifah, Z.; Gaymalov, Z.Z.; Shehzad, A.; et al. Thymus Gland: A Double Edge Sword for Coronaviruses. Vaccines 2021, 9, 1119. https://doi.org/10.3390/vaccines9101119
Al-Suhaimi EA, Aljafary MA, Alkhulaifi FM, Aldossary HA, Alshammari T, AL-Qaaneh A, Aldahhan R, Alkhalifah Z, Gaymalov ZZ, Shehzad A, et al. Thymus Gland: A Double Edge Sword for Coronaviruses. Vaccines. 2021; 9(10):1119. https://doi.org/10.3390/vaccines9101119
Chicago/Turabian StyleAl-Suhaimi, Ebtesam A., Meneerah A. Aljafary, Fadwa M. Alkhulaifi, Hanan A. Aldossary, Thamer Alshammari, Ayman AL-Qaaneh, Razan Aldahhan, Zahra Alkhalifah, Zagit Z. Gaymalov, Adeeb Shehzad, and et al. 2021. "Thymus Gland: A Double Edge Sword for Coronaviruses" Vaccines 9, no. 10: 1119. https://doi.org/10.3390/vaccines9101119
APA StyleAl-Suhaimi, E. A., Aljafary, M. A., Alkhulaifi, F. M., Aldossary, H. A., Alshammari, T., AL-Qaaneh, A., Aldahhan, R., Alkhalifah, Z., Gaymalov, Z. Z., Shehzad, A., & Homeida, A. M. (2021). Thymus Gland: A Double Edge Sword for Coronaviruses. Vaccines, 9(10), 1119. https://doi.org/10.3390/vaccines9101119






